Potential Response Patterns of Endogenous Hormones in Cliff Species Opisthopappus taihangensis and Opisthopappus longilobus under Salt Stress
Abstract
:1. Introduction
2. Results
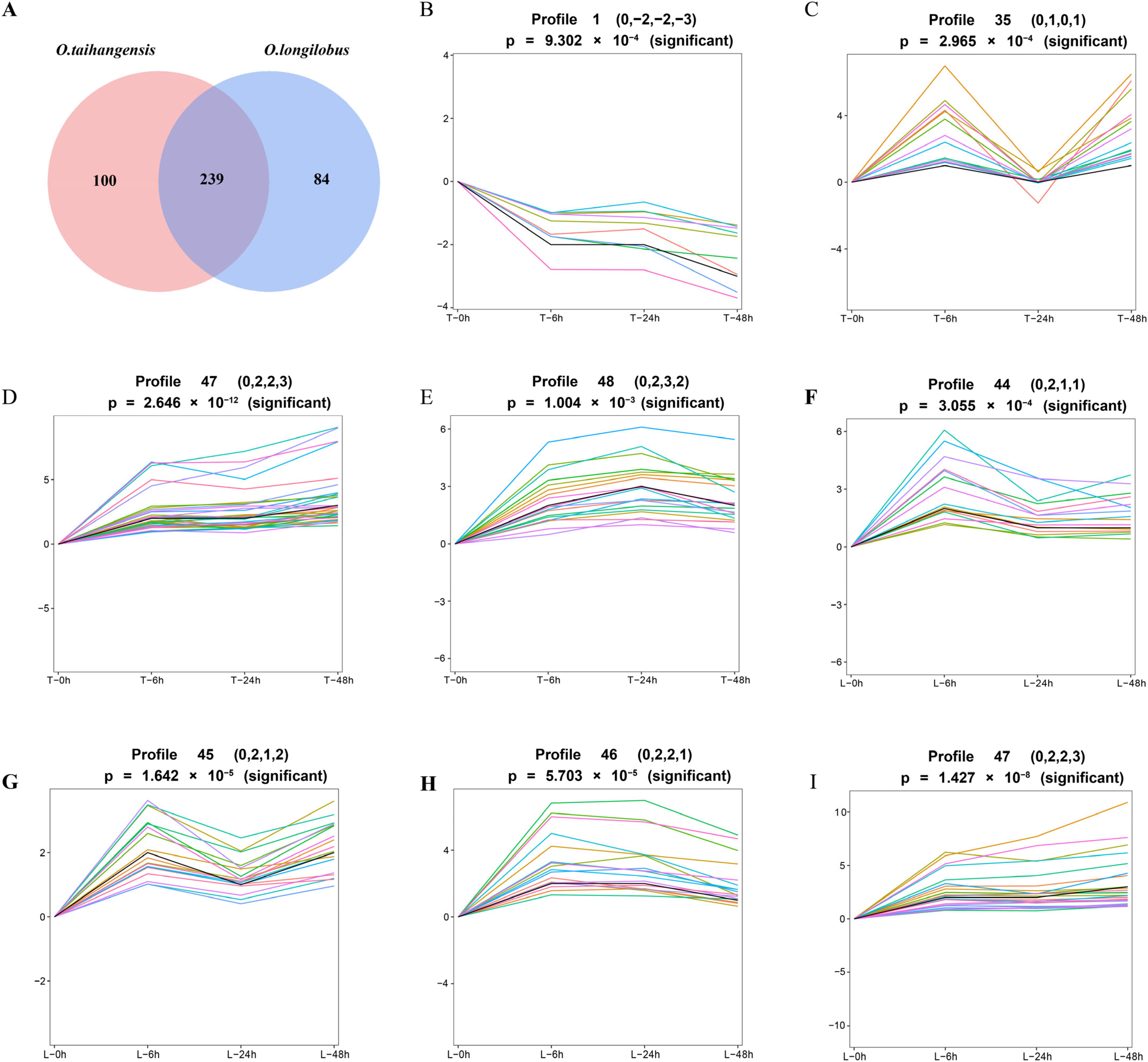
2.1. DEGs Related to Endogenous Opisthopappus Hormones
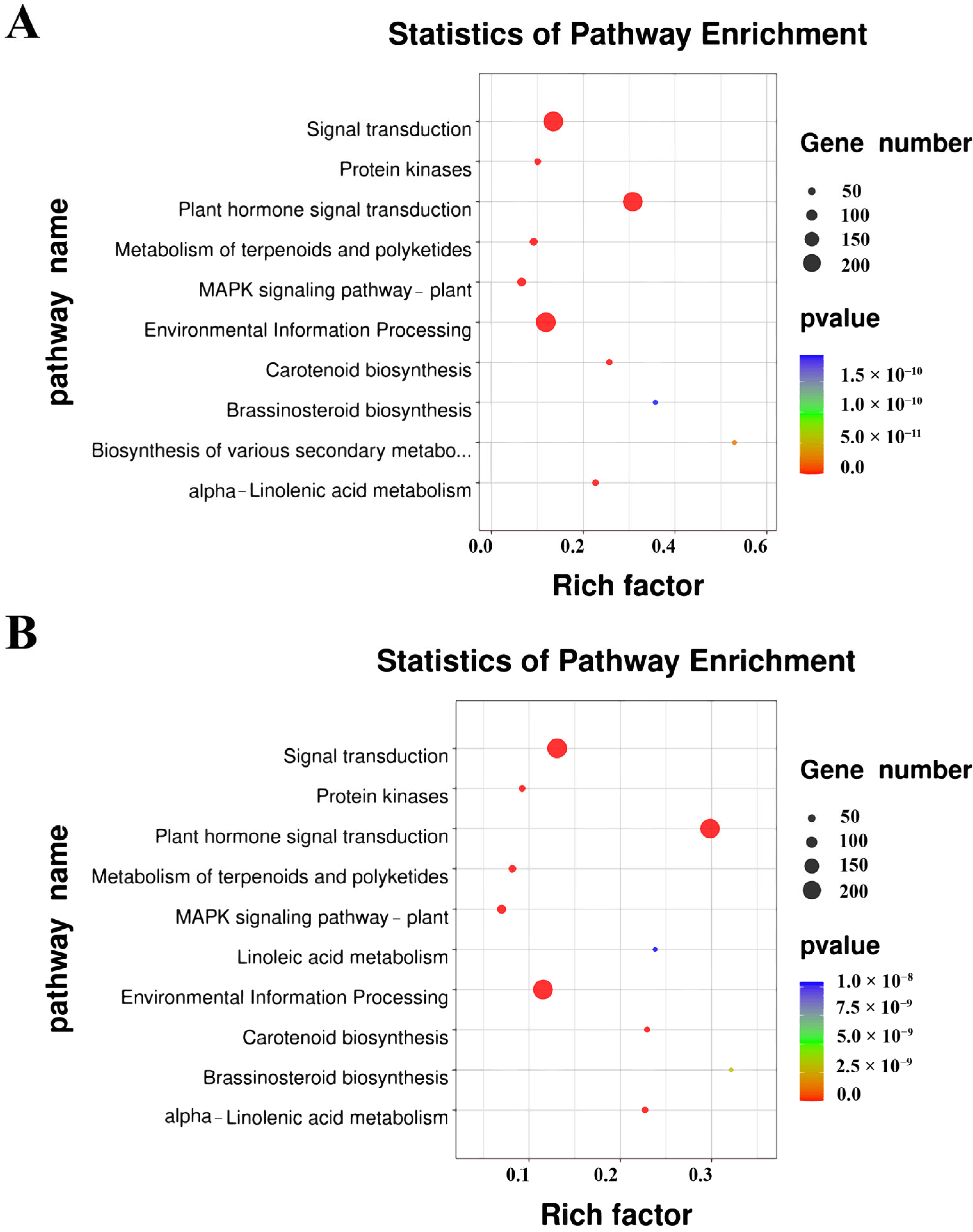
2.2. KEGG of DEGs
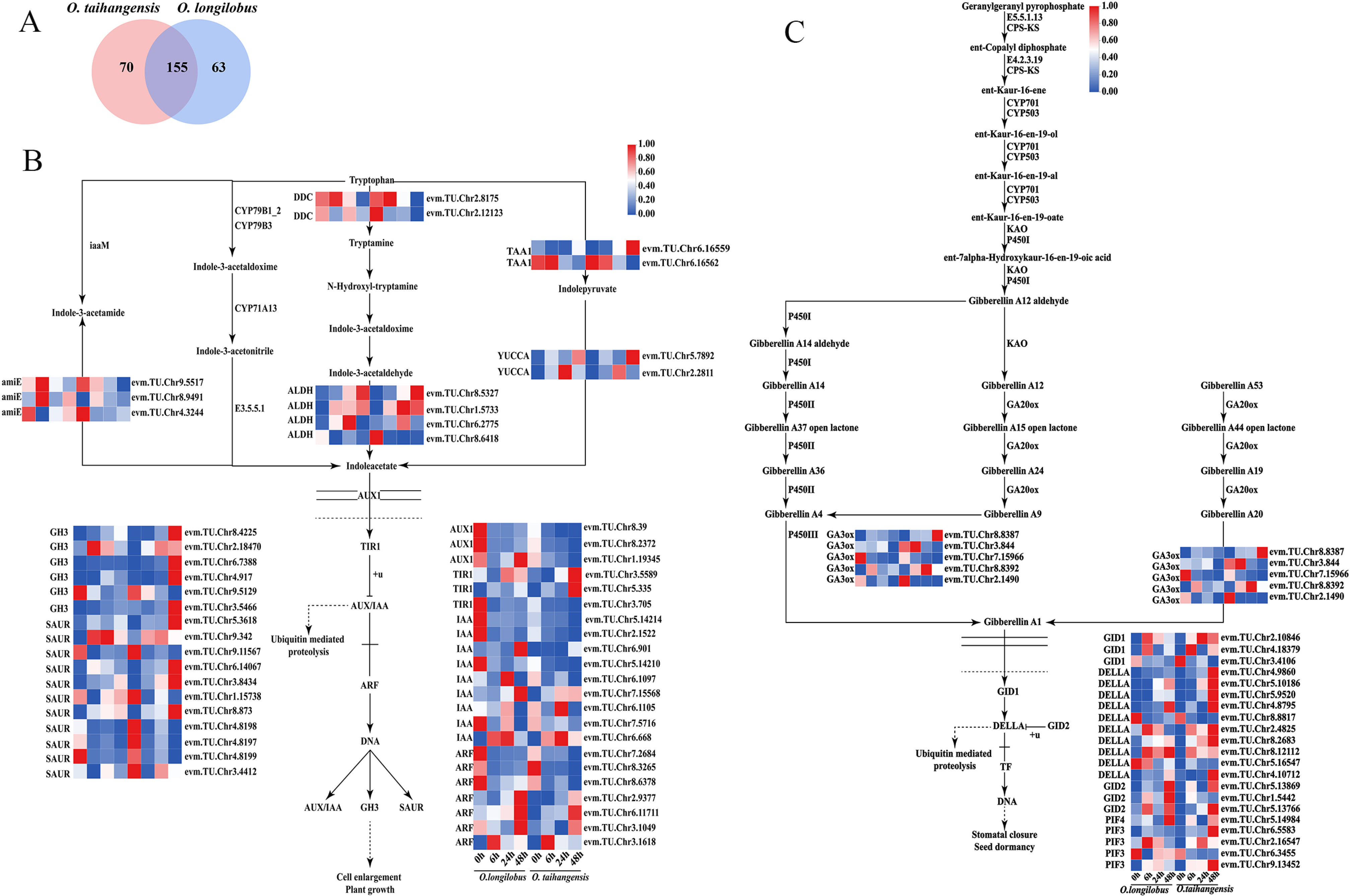
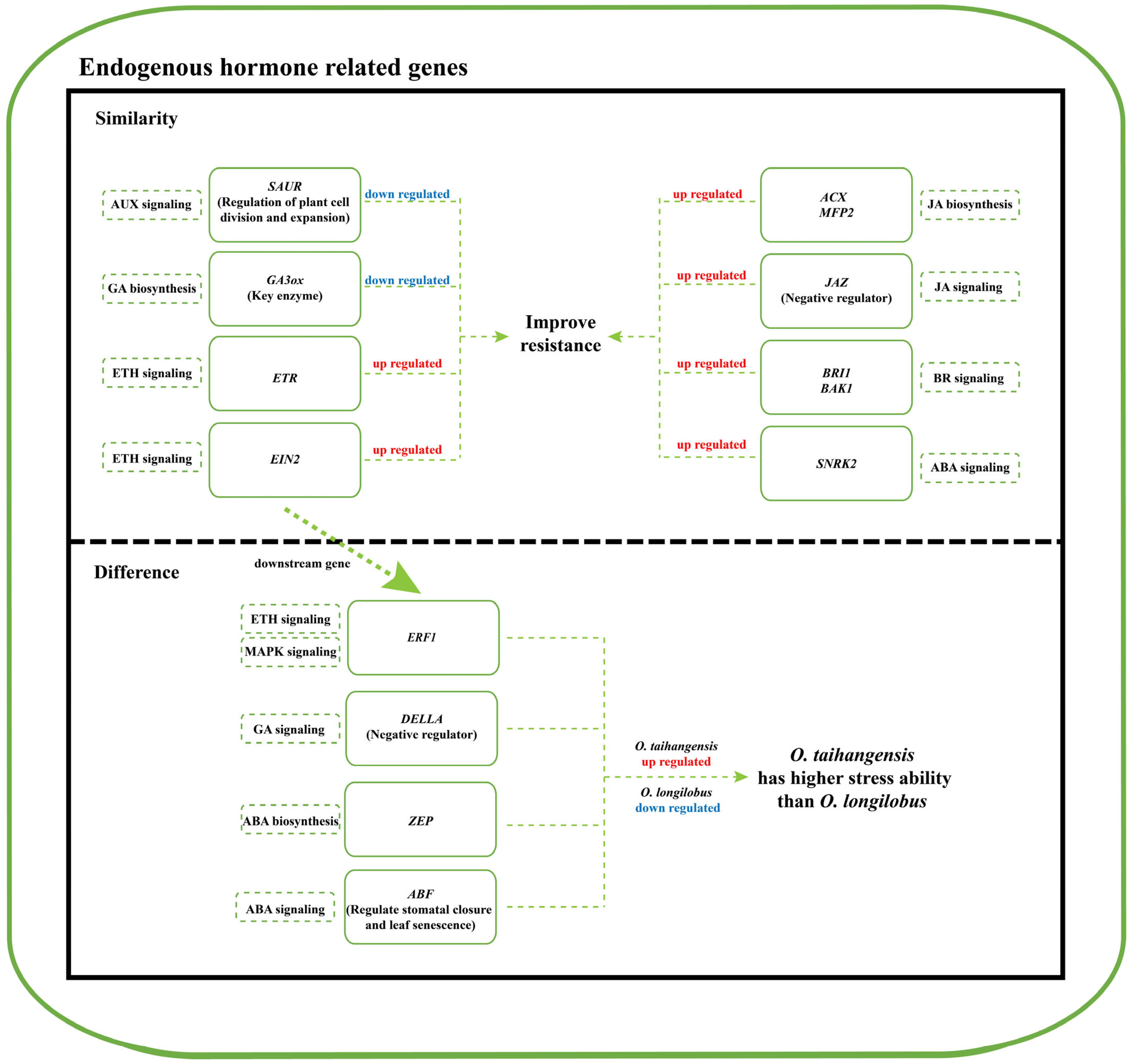
2.3. Biosynthesis and Signal Transduction Pathways of Endogenous Hormones
2.3.1. AUX and GA Biosynthesis and Signal Transduction
2.3.2. JA and BR Biosynthesis and Signal Transduction
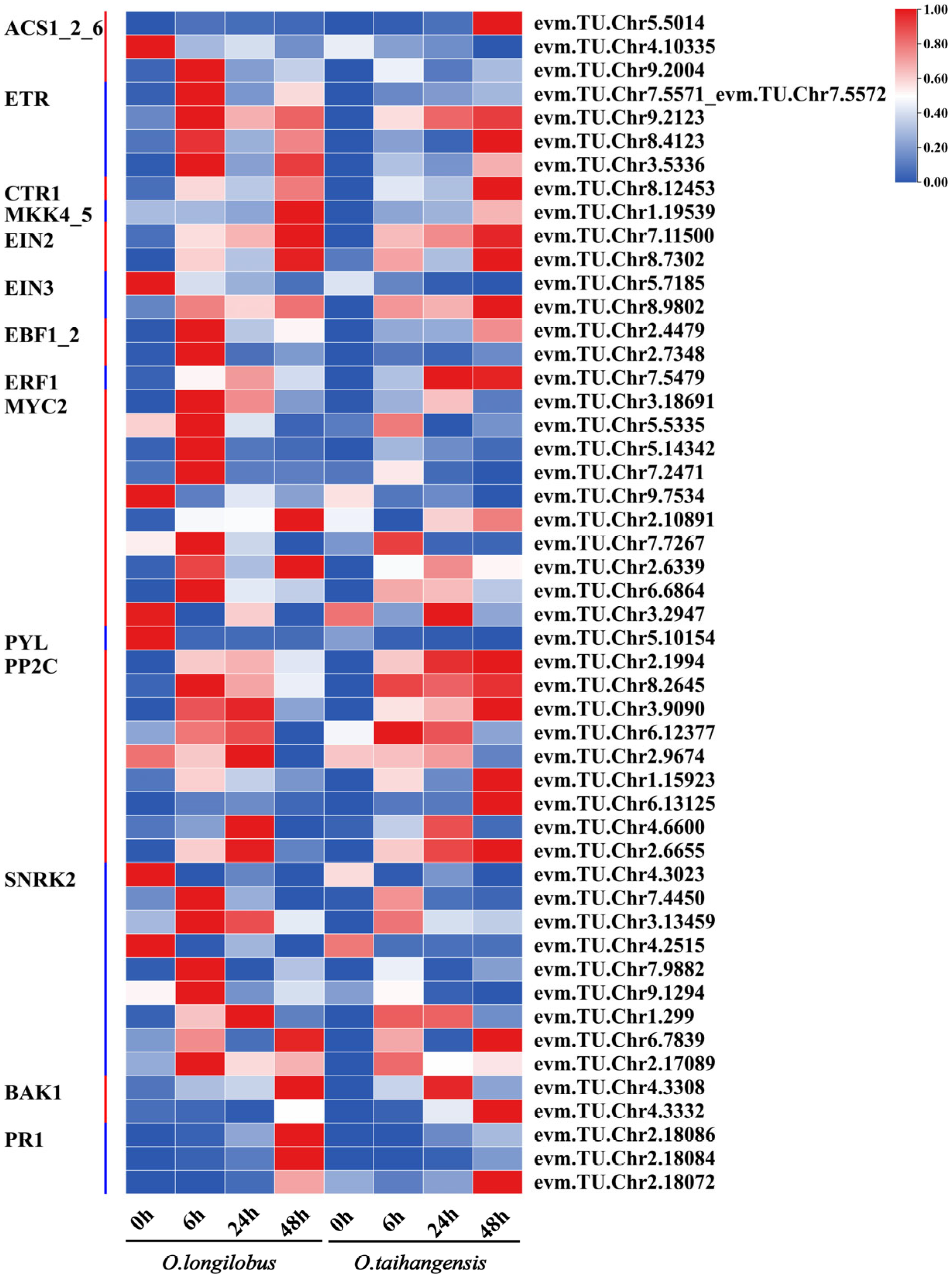
2.3.3. ABA and ETH Biosynthesis and Signal Transduction
2.3.4. CK and SA Biosynthesis and Signal Transduction
2.4. MAPK Signaling Pathway
2.5. Interactive Network Analysis
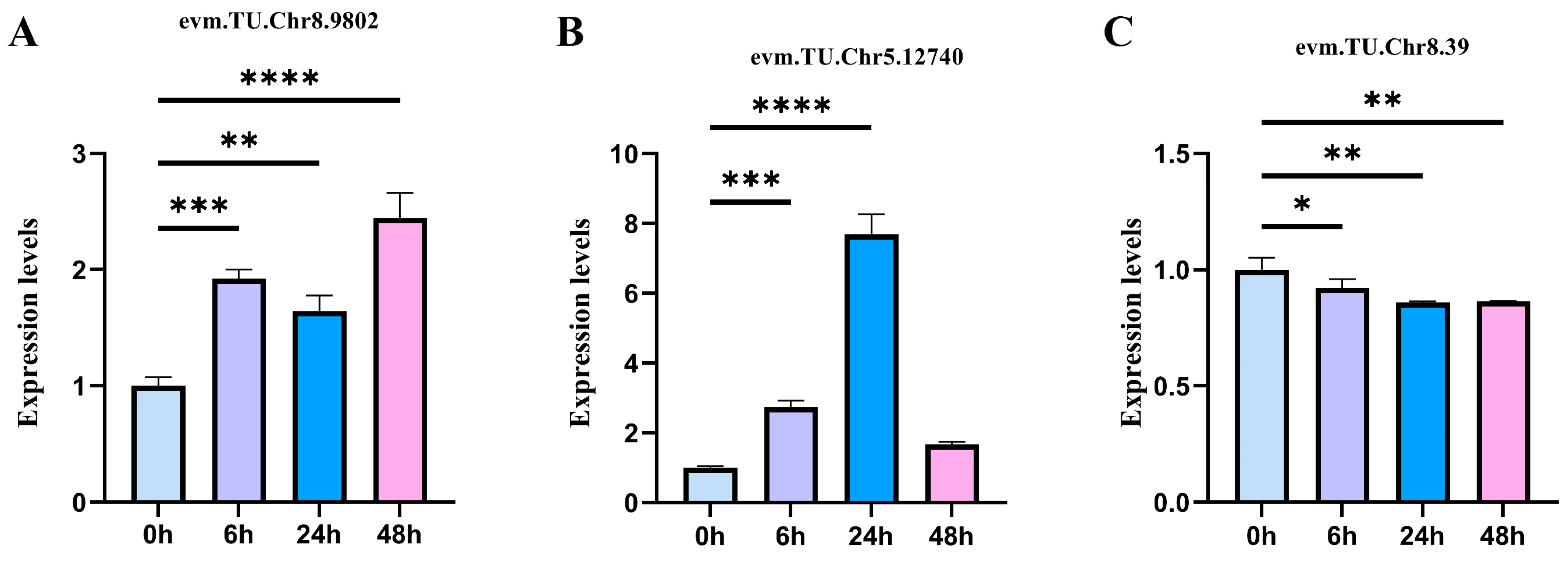
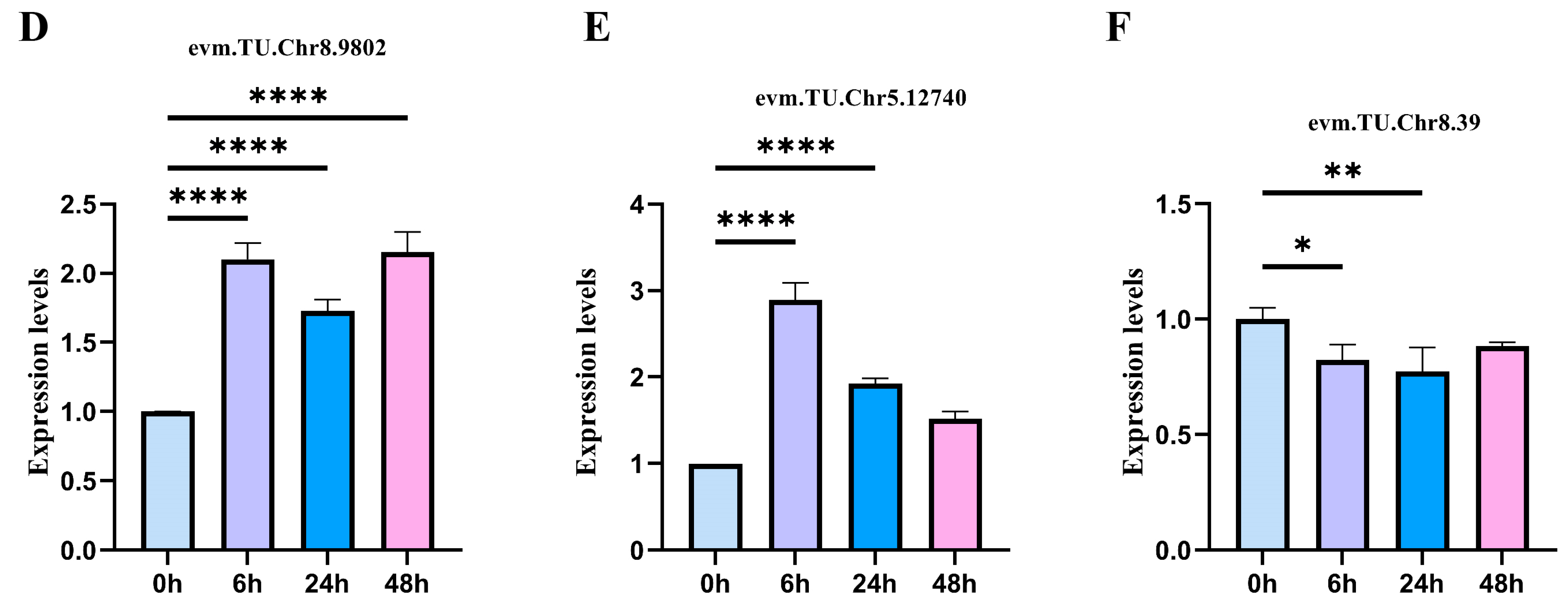
2.6. qRT-PCR Quantitative Verification
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Salt Treatments
4.2. DEGs Related to Endogenous Hormones
4.3. Enrichment Analysis
4.4. C-DEGs Analysis
4.5. PPI Network of DEGs
4.6. Quantitative Real-Time PCR (qRT-PCR)
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Sample | Raw Data | Valid Data | Valid Ratio (Reads) | Q20% | Q30% | GC Content% | ||
|---|---|---|---|---|---|---|---|---|
| Read | Base | Read | Base | |||||
| L_0h_1 | 37182186 | 5.58G | 36394348 | 5.46G | 97.88 | 99.98 | 97.52 | 43 |
| L_0h_2 | 44414876 | 6.66G | 43497750 | 6.52G | 97.94 | 99.98 | 97.45 | 43 |
| L_0h_3 | 44227778 | 6.63G | 43322712 | 6.50G | 97.95 | 99.98 | 97.29 | 43 |
| L_6h_1 | 42627342 | 6.39G | 41662700 | 6.25G | 97.74 | 99.97 | 97.17 | 42 |
| L_6h_2 | 46401950 | 6.96G | 45473926 | 6.82G | 98 | 99.97 | 97.07 | 42 |
| L_6h_3 | 45438882 | 6.82G | 44639616 | 6.70G | 98.24 | 99.98 | 97.21 | 42.5 |
| L_24h_1 | 43147800 | 6.47G | 42196388 | 6.33G | 97.79 | 99.97 | 97.1 | 42.5 |
| L_24h_2 | 44085774 | 6.61G | 43199864 | 6.48G | 97.99 | 99.97 | 97.23 | 43 |
| L_24h_3 | 44295014 | 6.64G | 43489158 | 6.52G | 98.18 | 99.97 | 97.17 | 43 |
| L_48h_1 | 34481428 | 5.17G | 33942430 | 5.09G | 98.44 | 99.97 | 96.88 | 42 |
| L_48h_2 | 38597360 | 5.79G | 37896306 | 5.68G | 98.18 | 99.97 | 96.65 | 42 |
| L_48h_3 | 47607662 | 7.14G | 46791990 | 7.02G | 98.29 | 99.97 | 96.86 | 42 |
| T_0h_1 | 43742304 | 6.56G | 42479114 | 6.37G | 97.11 | 99.97 | 97.18 | 43 |
| T_0h_2 | 37577538 | 5.64G | 36945886 | 5.54G | 98.32 | 99.98 | 97.52 | 44 |
| T_0h_3 | 43842334 | 6.58G | 43069372 | 6.46G | 98.24 | 99.98 | 97.37 | 44 |
| T_6h_1 | 44450660 | 6.67G | 43272680 | 6.49G | 97.35 | 99.97 | 97.07 | 43 |
| T_6h_2 | 44165698 | 6.62G | 43040866 | 6.46G | 97.45 | 99.98 | 97.34 | 43 |
| T_6h_3 | 43733578 | 6.56G | 42559804 | 6.38G | 97.32 | 99.97 | 97.22 | 43 |
| T_24h_1 | 44239528 | 6.64G | 43304948 | 6.50G | 97.89 | 99.98 | 97.19 | 45 |
| T_24h_2 | 47837072 | 7.18G | 46370698 | 6.96G | 96.93 | 99.97 | 97.19 | 42 |
| T_24h_3 | 43705198 | 6.56G | 42782336 | 6.42G | 97.89 | 99.98 | 97.14 | 42 |
| T_48h_1 | 44105106 | 6.62G | 43564398 | 6.53G | 98.77 | 99.97 | 96.57 | 42 |
| T_48h_2 | 44198704 | 6.63G | 43665264 | 6.55G | 98.79 | 99.97 | 96.71 | 42 |
| T_48h_3 | 43200034 | 6.48G | 42677700 | 6.40G | 98.79 | 99.97 | 96.63 | 42 |
| Gene ID | Primers (5′ to 3′) |
|---|---|
| evm. TU. Chr8.9802 | F: GATGACCAGCAGCCACCAA |
| R: TGTTCCGCCACGATTGACTT | |
| evm. TU. Chr5.12740 | F: GACGCTCGGACCACAAGATAT |
| R: GAGGACAGTATAGCCACCATCA | |
| evm. TU. Chr8.39 | F: AGGTTGCCAGTTGTGATTCCA |
| R: GGCTTCTCCGCAGCATTCT | |
| evm. TU. Chr8.13443 (Actin) | F: CCTACAACGCCACACTCTCA |
| R: ACAGCAAGTTACACCACTCATG |
References
- Yang, Y.; Guo, Y. Unraveling salt stress signaling in plants. J. Integr. Plant Biol. 2018, 60, 796–804. [Google Scholar] [CrossRef]
- Shahid, S.A.; Zaman, M.; Heng, L. Soil Salinity: Historical Perspectives and a World Overview of the Problem. In Guideline for Salinity Assessment, Mitigation and Adaptation Using Nuclear and Related Techniques; Springer: Cham, Switzerland, 2018; pp. 43–53. [Google Scholar]
- Shabala, S.; Bose, J.; Hedrich, R. Salt bladders: Do they matter? Trends Plant Sci. 2014, 19, 687–691. [Google Scholar] [CrossRef]
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Khan, A.; AL-Harrasi, A. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32. [Google Scholar] [CrossRef]
- Yu, Z.; Duan, X.; Luo, L.; Dai, S.; Ding, Z.; Xia, G. How Plant Hormones Mediate Salt Stress Responses. Trends Plant Sci. 2020, 25, 1117–1130. [Google Scholar] [CrossRef]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef] [PubMed]
- Ribba, T.; Garrido-Vargas, F.; O’Brien, J.A. Auxin-mediated responses under salt stress: From developmental regulation to biotechnological applications. J. Exp. Bot. 2020, 71, 3843–3853. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, M.J.; Terrile, M.C.; Bartoli, C.G.; D’Ippólito, S.; Casalongué, C.A. Auxin signaling participates in the adaptative response against oxidative stress and salinity by interacting with redox metabolism in Arabidopsis. Plant Mol. Biol. 2010, 74, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, M.J.; Terrile, M.C.; Windels, D.; Lombardo, M.C.; Bartoli, C.G.; Vazquez, F.; Estelle, M.; Casalongué, C.A. MiR393 regulation of auxin signaling and redox-related components during acclimation to salinity in Arabidopsis. PLoS ONE 2014, 9, e107678. [Google Scholar] [CrossRef] [PubMed]
- Colebrook, E.H.; Thomas, S.G.; Phillips, A.L.; Hedden, P. The role of gibberellin signalling in plant responses to abiotic stress. J. Exp. Biol. 2014, 217, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Davière, J.M.; Achard, P. Gibberellin signaling in plants. Development 2013, 140, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Rashotte, A.M.; Wysocka-Diller, J.W. ABA and GA signaling pathways interact and regulate seed germination and seedling development under salt stress. Acta Physiol. Plant. 2011, 33, 261–271. [Google Scholar] [CrossRef]
- Bao, S.; Hua, C.; Shen, L.; Yu, H. New insights into gibberellin signaling in regulating flowering in Arabidopsis. J. Integr. Plant Biol. 2020, 62, 118–131. [Google Scholar] [CrossRef]
- Wang, J.; Qin, H.; Zhou, S.; Wei, P.; Zhang, H.; Zhou, Y.; Miao, Y.; Huang, R. The Ubiquitin-Binding Protein OsDSK2a Mediates Seedling Growth and Salt Responses by Regulating Gibberellin Metabolism in Rice. Plant Cell 2020, 32, 414–428. [Google Scholar] [CrossRef]
- Chen, K.; Li, G.J.; Bressan, R.A.; Song, C.P.; Zhu, J.K.; Zhao, Y. Abscisic acid dynamics, signaling, and functions in plants. J. Integr. Plant Biol. 2020, 62, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Edel, K.H.; Kudla, J. Integration of calcium and ABA signaling. Curr. Opin. Plant Biol. 2016, 33, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chan, Z.; Gao, J.; Xing, L.; Cao, M.; Yu, C.; Hu, Y.; You, J.; Shi, H.; Zhu, Y. ABA receptor PYL9 promotes drought resistance and leaf senescence. Proc. Natl. Acad. Sci. USA 2016, 113, 1949–1954. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Chen, G.; Wang, Y.; Huang, Y.; Marchant, D.B.; Wang, Y.; Yang, Q.; Dai, F.; Hills, A.; Blatt, M.R. Evolutionary Conservation of ABA Signaling for Stomatal Closure. Plant Physiol. 2017, 174, 732–747. [Google Scholar] [CrossRef]
- Chen, H.; Bullock, D.A., Jr.; Alonso, J.M.; Stepanova, A.N. To Fight or to Grow: The Balancing Role of Ethylene in Plant Abiotic Stress Responses. Plants 2022, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wang, Q.; Gao, Z.; Wang, Y.; Liu, Y.; Ma, Z.; Chen, Y.; Zhang, Y.; Yan, F.; Li, J. Analysis of Phytohormone Signal Transduction in Sophora alopecuroides under Salt Stress. Int. J. Mol. Sci. 2021, 22, 7313. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Niu, M.; Gao, T.; Shen, Y.; Zhou, X.; Zhang, Y.; Liu, L.; Chai, M.; Sun, G.; Wang, Y. Responsive Alternative Splicing Events of Opisthopappus Species against Salt Stress. Int. J. Mol. Sci. 2024, 25, 1227. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, H.; Zang, E.; Liu, Z.; Lan, Y.; Hao, W.; He, S.; Fan, X.; Sun, G.; Wang, Y. Adaptation insights from comparative transcriptome analysis of two Opisthopappus species in the Taihang mountains. BMC Genom. 2022, 23, 466. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, Y.; Zhong, J.; Zhang, T.; Li, D.; Ba, T.; Xu, T.; Chang, L.; Zhang, Q.; Sun, M. Root Physiological Traits and Transcriptome Analyses Reveal that Root Zone Water Retention Confers Drought Tolerance to Opisthopappus taihangensis. Sci. Rep. 2020, 10, 2627. [Google Scholar] [CrossRef]
- Swarup, R.; Bhosale, R. Developmental Roles of AUX1/LAX Auxin Influx Carriers in Plants. Front. Plant Sci. 2019, 10, 1306. [Google Scholar] [CrossRef] [PubMed]
- Strader, L.C.; Zhao, Y. Auxin perception and downstream events. Curr. Opin. Plant Biol. 2016, 33, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Gray, W.M. SAUR proteins as effectors of hormonal and environmental signals in plant growth. Mol. Plant 2015, 8, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Stortenbeker, N.; Bemer, M. The SAUR gene family: The plant’s toolbox for adaptation of growth and development. J. Exp. Bot. 2018, 70, 17–27. [Google Scholar] [CrossRef]
- Luo, C.; Yan, J.; He, C.; Liu, W.; Xie, D.; Jiang, B. Genome-Wide Identification of the SAUR Gene Family in Wax Gourd (Benincasa hispida) and Functional Characterization of BhSAUR60 during Fruit Development. Int. J. Mol. Sci. 2022, 23, 14021. [Google Scholar] [CrossRef] [PubMed]
- Murase, K.; Hirano, Y.; Sun, T.P.; Hakoshima, T. Gibberellin-induced DELLA recognition by the gibberellin receptor GID1. Nature 2008, 456, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Li, Z.; Xiao, G.; Zhai, M.; Pan, X.; Huang, R.; Zhang, H. CYP71D8L is a key regulator involved in growth and stress responses by mediating gibberellin homeostasis in rice. J. Exp. Bot. 2020, 71, 1160–1170. [Google Scholar] [PubMed]
- Hernández-García, J.; Sun, R.; Serrano-Mislata, A.; Inoue, K.; Vargas-Chávez, C.; Esteve-Bruna, D.; Arbona, V.; Yamaoka, S.; Nishihama, R.; Kohchi, T.; et al. Coordination between growth and stress responses by DELLA in the liverwort Marchantia polymorpha. Curr. Biol. 2021, 31, 3678–3686.e11. [Google Scholar] [CrossRef]
- Zhao, Y.; Dong, W.; Zhang, N.; Ai, X.; Wang, M.; Huang, Z.; Xiao, L.; Xia, G. A wheat allene oxide cyclase gene enhances salinity tolerance via jasmonate signaling. Plant Physiol. 2014, 164, 1068–1076. [Google Scholar] [CrossRef]
- Garcia-Abellan, J.O.; Fernandez-Garcia, N.; Lopez-Berenguer, C.; Egea, I.; Flores, F.B.; Angosto, T.; Capel, J.; Lozano, R.; Pineda, B.; Moreno, V.; et al. The tomato res mutant which accumulates JA in roots in non-stressed conditions restores cell structure alterations under salinity. Physiol. Plant. 2015, 155, 296–314. [Google Scholar] [CrossRef]
- Wasternack, C.; Song, S. Jasmonates: Biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J. Exp. Bot. 2017, 68, 1303–1321. [Google Scholar] [CrossRef]
- Valenzuela, C.E.; Acevedo-Acevedo, O.; Miranda, G.S.; Vergara-Barros, P.; Holuigue, L.; Figueroa, C.R.; Figueroa, P.M. Salt stress response triggers activation of the jasmonate signaling pathway leading to inhibition of cell elongation in Arabidopsis primary root. J. Exp. Bot. 2016, 67, 4209–4220. [Google Scholar] [CrossRef]
- Yao, D.; Zhang, X.; Zhao, X.; Liu, C.; Wang, C.; Zhang, Z.; Zhang, C.; Wei, Q.; Wang, Q.; Yan, H. Transcriptome analysis reveals salt-stress-regulated biological processes and key pathways in roots of cotton (Gossypium hirsutum L.). Genomics 2011, 98, 47–55. [Google Scholar] [CrossRef]
- Chini, A.; Ben-Romdhane, W.; Hassairi, A.; Aboul-Soud, M.A. Identification of TIFY/JAZ family genes in Solanum lycopersicum and their regulation in response to abiotic stresses. PLoS ONE 2017, 12, e0177381. [Google Scholar] [CrossRef]
- Ebel, C.; BenFeki, A.; Hanin, M.; Solano, R.; Chini, A. Characterization of wheat (Triticum aestivum) TIFY family and role of Triticum Durum Td TIFY11a in salt stress tolerance. PLoS ONE 2018, 13, e0200566. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Li, J. Regulation of Three Key Kinases of Brassinosteroid Signaling Pathway. Int. J. Mol. Sci. 2020, 21, 4340. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.H.; Li, J. BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 2002, 110, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Zhu, J.K. Regulation of abscisic acid biosynthesis. Plant Physiol. 2003, 133, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Seok, H.Y.; Park, B.K.; Kim, S.H.; Goh, C.H.; Lee, B.H.; Lee, C.H.; Moon, Y.H. Overexpression of Arabidopsis ZEP enhances tolerance to osmotic stress. Biophy. Res. 2008, 375, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.I.; Hong, J.H.; Ha, J.O.; Kang, J.Y.; Kim, S.Y. ABFs, a family of ABA-responsive element binding factors. J. Biol. Chem. 2000, 275, 1723–1730. [Google Scholar] [CrossRef]
- Fujita, Y.; Fujita, M.; Satoh, R.; Maruyama, K.; Parvez, M.M.; Seki, M.; Hiratsu, K.; Ohme-Takagi, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. AREB1 is a transcription activator of novel ABRE-dependent ABA signaling that enhances drought stress tolerance in Arabidopsis. Plant Cell 2005, 17, 3470–3488. [Google Scholar] [CrossRef] [PubMed]
- Abdeen, A.; Schnell, J.; Miki, B. Transcriptome analysis reveals absence of unintended effects in drought-tolerant transgenic plants overexpressing the transcription factor ABF3. BMC Genom. 2010, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.D.; Singh, N.; Ahuja, P.S.; Reddy, T.V. Abscisic acid response element binding factor 1 is required for establishment of Arabidopsis seedlings during winter. Mol. Biol. Rep. 2011, 38, 5147–5159. [Google Scholar] [CrossRef]
- Song, X.; Yu, X.; Hori, C.; Demura, T.; Ohtani, M.; Zhuge, Q. Heterologous Overexpression of Poplar SnRK2 Genes Enhanced Salt Stress Tolerance in Arabidopsis thaliana. Front. Plant Sci. 2016, 7, 612. [Google Scholar] [CrossRef]
- Cebrián, G.; Iglesias-Moya, J.; García, A.; Martínez, J.; Romero, J.; Regalado, J.J.; Martínez, C.; Valenzuela, J.L.; Jamilena, M. Involvement of ethylene receptors in the salt tolerance response of Cucurbita pepo. Hortic. Res. 2021, 8, 73. [Google Scholar] [CrossRef]
- Feng, G.; Liu, G.; Xiao, J. The Arabidopsis EIN2 restricts organ growth by retarding cell expansion. Plant Signal. Behav. 2015, 10, 5. [Google Scholar] [CrossRef]
- Xie, Z.; Nolan, T.M.; Jiang, H.; Yin, Y. AP2/ERF Transcription Factor Regulatory Networks in Hormone and Abiotic Stress Responses in Arabidopsis. Front. Plant Sci. 2019, 10, 228. [Google Scholar] [CrossRef]
- Gehart, H.; Kumpf, S.; Ittner, A.; Ricci, R. MAPK signalling in cellular metabolism: Stress or wellness. EMBO Rep. 2010, 11, 834–840. [Google Scholar] [CrossRef]
- Coulthard, L.R.; White, D.E.; Jones, D.L.; McDermott, M.F.; Burchill, S.A. p38MAPK: Stress responses from molecular mechanisms to therapeutics. Trends Mol. Med. 2009, 15, 369–379. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Shen, Y.; Han, M.; Su, Y.; Feng, X.; Gao, T.; Zhou, X.; Wu, Q.; Sun, G.; Wang, Y. Potential Response Patterns of Endogenous Hormones in Cliff Species Opisthopappus taihangensis and Opisthopappus longilobus under Salt Stress. Plants 2024, 13, 557. https://doi.org/10.3390/plants13040557
Zhang Y, Shen Y, Han M, Su Y, Feng X, Gao T, Zhou X, Wu Q, Sun G, Wang Y. Potential Response Patterns of Endogenous Hormones in Cliff Species Opisthopappus taihangensis and Opisthopappus longilobus under Salt Stress. Plants. 2024; 13(4):557. https://doi.org/10.3390/plants13040557
Chicago/Turabian StyleZhang, Yimeng, Yuexin Shen, Mian Han, Yu Su, Xiaolong Feng, Ting Gao, Xiaojuan Zhou, Qi Wu, Genlou Sun, and Yiling Wang. 2024. "Potential Response Patterns of Endogenous Hormones in Cliff Species Opisthopappus taihangensis and Opisthopappus longilobus under Salt Stress" Plants 13, no. 4: 557. https://doi.org/10.3390/plants13040557
APA StyleZhang, Y., Shen, Y., Han, M., Su, Y., Feng, X., Gao, T., Zhou, X., Wu, Q., Sun, G., & Wang, Y. (2024). Potential Response Patterns of Endogenous Hormones in Cliff Species Opisthopappus taihangensis and Opisthopappus longilobus under Salt Stress. Plants, 13(4), 557. https://doi.org/10.3390/plants13040557







