Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane
Abstract
1. Introduction
2. Results
2.1. Combined Variance Analysis for Sugarcane Photosynthetic Characteristics
2.2. Principal Component Analysis for Sugarcane Photosynthetic Efficiency
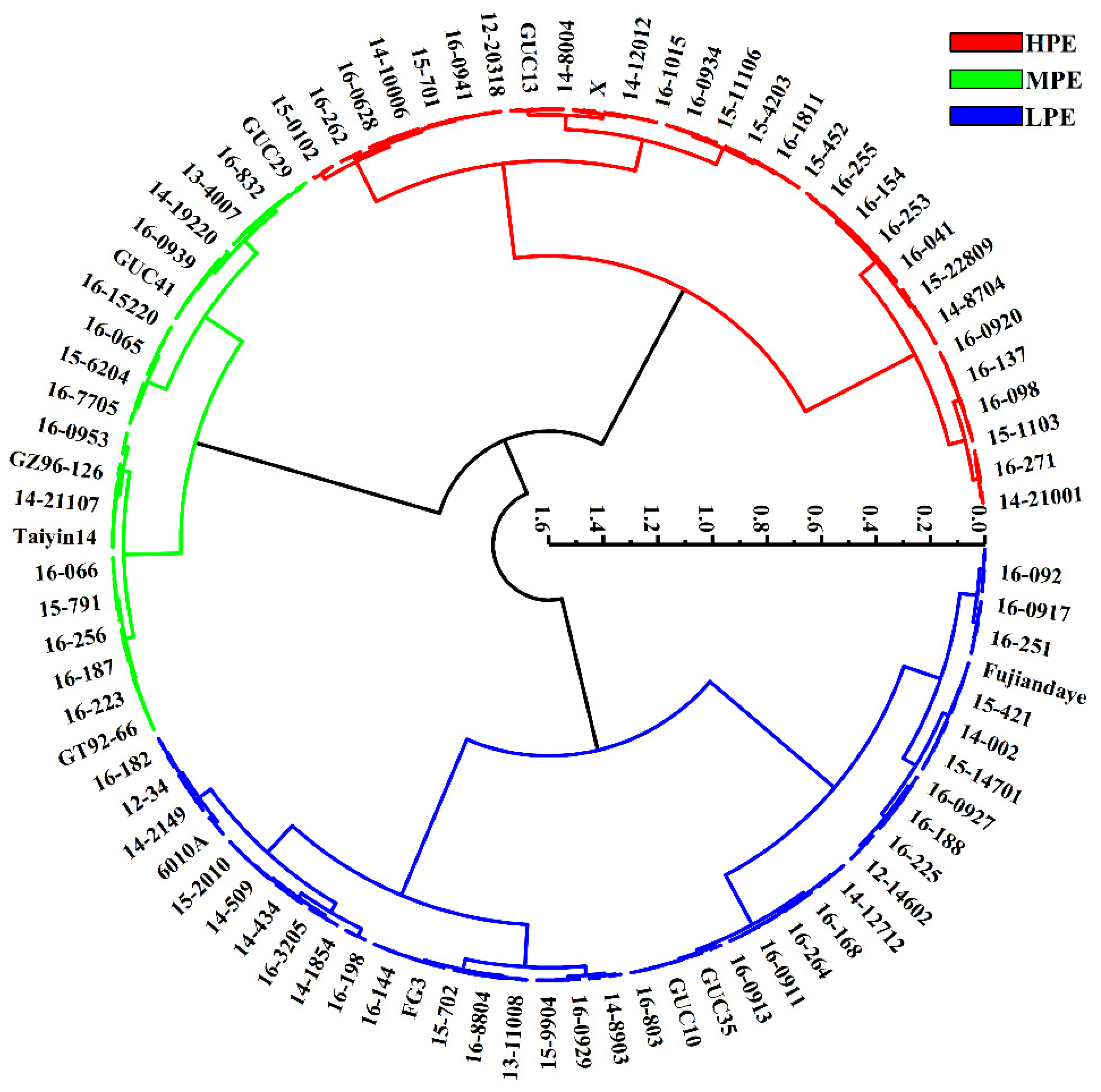
2.3. Cluster Analysis and Discriminant Analysis for Sugarcane Photosynthetic Efficiency
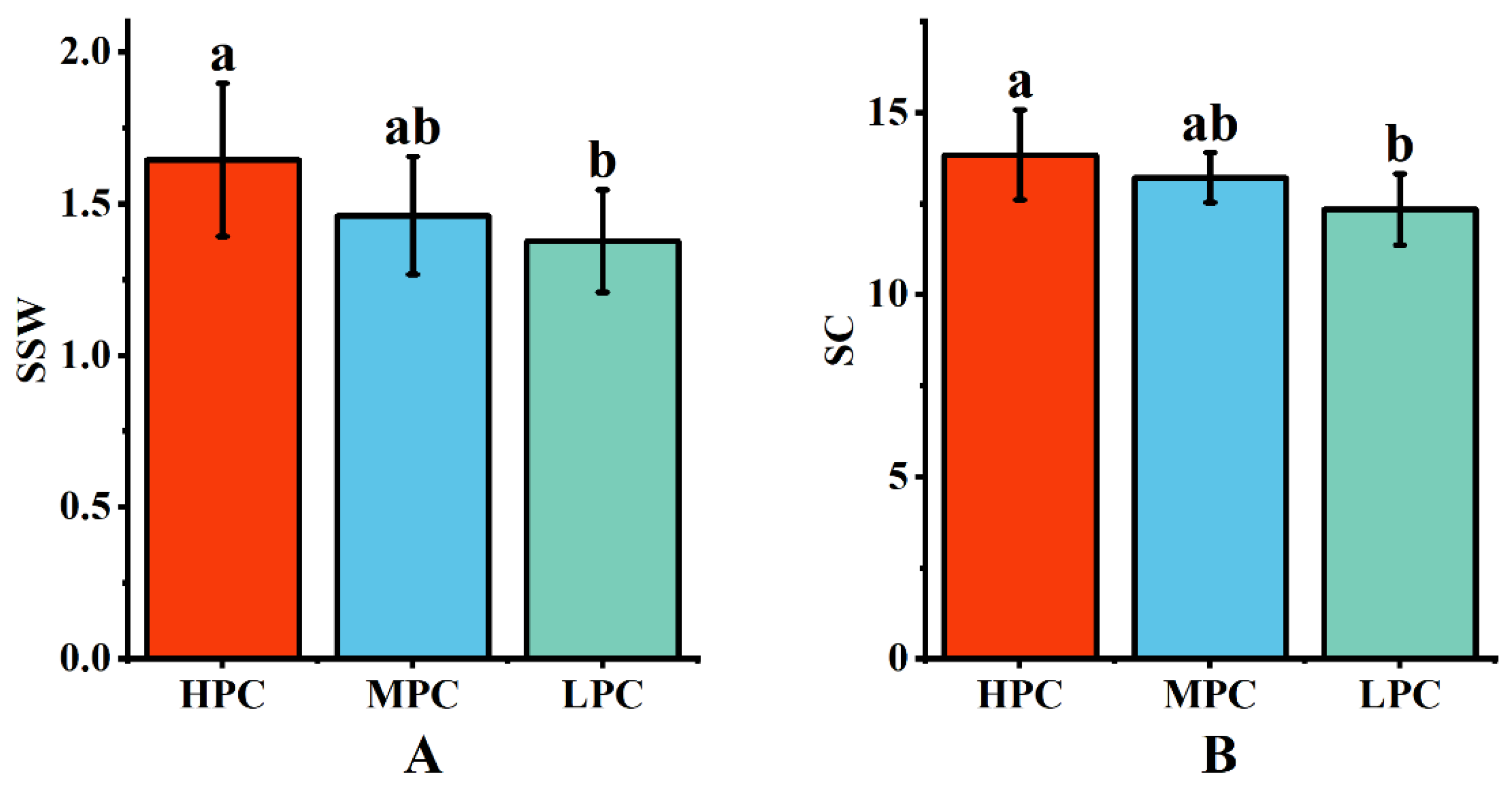
2.4. Quality Efficiency of Sugarcane with Different Photosynthetic Traits
3. Discussion
4. Materials and Methods
4.1. Experimental Location and Design
4.2. Field Data Collection
4.3. Sugarcane Single-Stem Weight and Sugar Content Measurement
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boaretto, L.F.; Labate, M.T.V.; Franceschini, L.M.; Cataldi, T.R.; Budzinski, I.G.F.; de Moraes, F.E.; Labate, C.A. Proteomics Reveals an Increase in the Abundance of Glycolytic and Ethanolic Fermentation Enzymes in Developing Sugarcane Culms During Sucrose Accumulation. Front. Plant Sci. 2021, 12, 716964. [Google Scholar] [CrossRef] [PubMed]
- Mattiello, L.; Riaño-Pachón, D.M.; Martins, M.C.; da Cruz, L.P.; Bassi, D.; Marchiori, P.E.; Ribeiro, R.V.; Labate, M.T.; Labate, C.A.; Menossi, M. Physiological and transcriptional analyses of developmental stages along sugarcane leaf. BMC Plant Biol. 2015, 15, 300. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Singh, S.N.; Tiwari, A.K.; Pathak, S.K.; Singh, A.K.; Srivastava, S.; Mohan, N. Integration of sugarcane production technologies for enhanced cane and sugar productivity targeting to increase farmers’ income: Strategies and prospects. 3 Biotech 2019, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Passioura, J.B.; Angus, J.F. Improving productivity of crops in water-limited environments. Adv. Agron. 2010, 106, 37–75. [Google Scholar]
- Li, C.; Jackson, P.; Lu, X.; Xu, C.; Cai, Q.; Basnayake, J.; Lakshmanan, P.; Ghannoum, O.; Fan, Y. Genotypic variation in transpiration efficiency due to differences in photosynthetic capacity among sugarcane-related clones. J. Exp. Bot. 2017, 68, 2377–2385. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.; Basnayake, J.; Lakshmanan, P.; Fukai, S. Genotypic variation in intrinsic transpiration efficiency correlates with sugarcane yield under rainfed and irrigated field conditions. Physiol. Plant. 2020, 172, 976–989. [Google Scholar] [CrossRef]
- Pretty, J. Agricultural sustainability: Concepts, principles and evidence. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 447–465. [Google Scholar] [CrossRef]
- Robinson, N.; Brackin, R.; Vinall, K.; Soper, F.; Holst, J.; Gamage, H.; Paungfoo-Lonhienne, C.; Rennenberg, H.; Lakshmanan, P.; Schmidt, S. Nitrate paradigm does not hold up for sugarcane. PLoS ONE 2011, 6, e19045. [Google Scholar] [CrossRef]
- Schmidt Rivera, X.C.; Bacenetti, J.; Fusi, A.; Niero, M. The influence of fertiliser and pesticide emissions model on life cycle assessment of agricultural products: The case of Danish and Italian barley. Sci. Total Environ. 2017, 592, 745–757. [Google Scholar] [CrossRef]
- Teixeira, G.C.M.; de Prado, R.M.; Rocha, A.M.S.; de Oliveira Filho, A.S.B.; da Sousa Junior, G.S.; Gratão, P.L. Action of silicon on the activity of antioxidant enzymes and on physiological mechanisms mitigates water deficit in sugarcane and energy cane plants. Sci. Rep. 2022, 12, 17487. [Google Scholar] [CrossRef]
- Raines, C.A. Increasing photosynthetic carbon assimilation in C3 plants to improve crop yield: Current and future strategies. Plant Physiol. 2011, 155, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Mo, F.; Yan, M.; Zhang, X.; Zhang, B.; Huang, X.; Huang, D.; Pan, Y.; Verma, K.K.; Li, Y.-R. Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane. Life 2022, 12, 1201. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Yao, W.; Yu, K.; Qin, L.; Ruan, M.; Powell, C.A.; Chen, B.; Zhang, M. Photosynthetic characterization and expression profiles of sugarcane infected by Sugarcane mosaic virus (SCMV). Photosynth. Res. 2020, 150, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Sales, C.R.G.; Wang, Y.; Evers, J.B.; Kromdijk, J. Improving C4 photosynthesis to increase productivity under optimal and suboptimal conditions. J. Exp. Bot. 2021, 72, 5942–5960. [Google Scholar] [CrossRef]
- He, Y.; Matthews, M.L. Seasonal climate conditions impact the effectiveness of improving photosynthesis to increase soybean yield. Field Crops Res. 2023, 296, 108907. [Google Scholar] [CrossRef]
- Kumar, A.; Pandey, S.S.; Kumar, D.; Tripathi, B.N. Genetic manipulation of photosynthesis to enhance crop productivity under changing environmental conditions. Photosynth. Res. 2022, 155, 1–21. [Google Scholar] [CrossRef]
- Makino, A. Photosynthesis, Grain Yield, and Nitrogen Utilization in Rice and Wheat. Plant Physiol. 2011, 155, 125–129. [Google Scholar] [CrossRef]
- McCormick, A.J.; Cramer, M.D.; Watt, D.A. Sink strength regulates photosynthesis in sugarcane. New Phytol. 2006, 171, 759–770. [Google Scholar] [CrossRef]
- De Souza, A.P.; Gaspar, M.; Da Silva, E.A.; Ulian, E.C.; Waclawovsky, A.J.; Nishiyama, M.Y., Jr.; Dos Santos, R.V.; Teixeira, M.M.; Souza, G.M.; Buckeridge, M.S. Elevated CO2 increases photosynthesis, biomass and productivity, and modifies gene expression in sugarcane. Plant Cell Environ. 2008, 31, 1116–1127. [Google Scholar] [CrossRef]
- Monteith, J.L. Climate and the Efficiency of Crop Production in Britain [and Discussion]. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1977, 281, 277–294. [Google Scholar]
- Pfündel, E.E. Simultaneously measuring pulse-amplitude-modulated (PAM) chlorophyll fluorescence of leaves at wavelengths shorter and longer than 700 nm. Photosynth. Res. 2021, 147, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Shapiguzov, A.; Kangasjärvi, J. Studying Plant Stress Reactions In Vivo by PAM Chlorophyll Fluorescence Imaging. In Reactive Oxygen Species in Plants; Humana: New York, NY, USA, 2022; pp. 43–61. [Google Scholar] [CrossRef]
- Oliver, T.; Kim, T.D.; Trinugroho, J.P.; Cordón-Preciado, V.; Wijayatilake, N.; Bhatia, A.; Rutherford, A.W.; Cardona, T. The Evolution and Evolvability of Photosystem II. Annu. Rev. Plant Biol. 2023, 74, 225–257. [Google Scholar] [CrossRef] [PubMed]
- M’hamdi, O.; Égei, M.; Pék, Z.; Ilahy, R.; Nemeskéri, E.; Helyes, L.; Takács, S. Root Development Monitoring under Different Water Supply Levels in Processing Tomato Plants. Plants 2023, 12, 3517. [Google Scholar] [CrossRef] [PubMed]
- Rossato, J.A.S., Jr.; Madaleno, L.L.; Mutton, M.J.R.; Higley, L.G.; Fernandes, O.A. Photosynthesis, yield and raw material quality of sugarcane injured by multiple pests. PeerJ 2019, 7, e6166. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Yang, H.; Wu, Y.; Gong, W.; Gul, H.; Yan, Y.; Yang, W. Photosynthetic Acclimation of Shade-Grown Soybean Seedlings to a High-Light Environment. Plants 2023, 12, 2324. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Sonoike, K. Screening of mutants using chlorophyll fluorescence. J. Plant Res. 2021, 134, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Scholes, J.D.; Rolfe, S.A. Chlorophyll fluorescence imaging as tool for understanding the impact of fungal diseases on plant performance: A phenomics perspective. Funct. Plant Biol. 2009, 36, 880–892. [Google Scholar] [CrossRef]
- Pérez-Bueno, M.L.; Pineda, M.; Barón, M. Phenotyping Plant Responses to Biotic Stress by Chlorophyll Fluorescence Imaging. Front. Plant Sci. 2019, 10, 1135. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef]
- Chaerle, L.; Leinonen, I.; Jones, H.G.; Van Der Straeten, D. Monitoring and screening plant populations with combined thermal and chlorophyll fluorescence imaging. J. Exp. Bot. 2007, 58, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef] [PubMed]
- Chaerle, L.; Van Der Straeten, D. Seeing is believing: Imaging techniques to monitor plant health. Biochim. Biophys. Acta 2001, 1519, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Sytar, O.; Brestic, M.; Zivcak, M.; Olsovska, K.; Kovar, M.; Shao, H.; He, X. Applying hyperspectral imaging to explore natural plant diversity towards improving salt stress tolerance. Sci. Total Environ. 2017, 578, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, C.; Belin, E.; Bove, E.; Rousseau, D.; Fabre, F.; Berruyer, R.; Guillaumès, J.; Manceau, C.; Jacques, M.A.; Boureau, T. High throughput quantitative phenotyping of plant resistance using chlorophyll fluorescence image analysis. Plant Methods 2013, 9, 17. [Google Scholar] [CrossRef]
- Robson, J.K.; Ferguson, J.N.; McAusland, L.; Atkinson, J.A.; Tranchant-Dubreuil, C.; Cubry, P.; Sabot, F.; Wells, D.M.; Price, A.H.; Wilson, Z.A.; et al. Chlorophyll fluorescence-based high-throughput phenotyping facilitates the genetic dissection of photosynthetic heat tolerance in African (Oryza glaberrima) and Asian (Oryza sativa) rice. J. Exp. Bot. 2023, 74, 5181–5197. [Google Scholar] [CrossRef]
- Sibret, T.; Verbruggen, W.; Peaucelle, M.; Verryckt, L.T.; Bauters, M.; Combe, M.; Boeckx, P.; Verbeeck, H. High photosynthetic capacity of Sahelian C(3) and C(4) plants. Photosynth. Res. 2021, 147, 161–175. [Google Scholar] [CrossRef]
- Marcotrigiano, M. A role for leaf epidermis in the control of leaf size and the rate and extent of mesophyll cell division. Am. J. Bot. 2010, 97, 224–233. [Google Scholar] [CrossRef]
- Liu, R.; Meng, Q.; Zheng, F.; Kong, L.; Yuan, J.; Lübberstedt, T. Genetic mapping of QTL for maize leaf width combining RIL and IF2 populations. PLoS ONE 2017, 12, e0189441. [Google Scholar] [CrossRef]
- Han, L.; Jiang, C.; Zhang, W.; Wang, H.; Li, K.; Liu, X.; Liu, Z.; Wu, Y.; Huang, C.; Hu, X. Morphological Characterization and Transcriptome Analysis of New Dwarf and Narrow-Leaf (dnl2) Mutant in Maize. Int. J. Mol. Sci. 2022, 23, 795. [Google Scholar] [CrossRef]
- Mathan, J.; Bhattacharya, J.; Ranjan, A. Enhancing crop yield by optimizing plant developmental features. Development 2016, 143, 3283–3294. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Solanki, M.K.; Yu, Z.-X.; Yang, L.-T.; An, Q.-L.; Dong, D.-F.; Li, Y.-R. Draft Genome Analysis Offers Insights Into the Mechanism by Which Streptomyces chartreusis WZS021 Increases Drought Tolerance in Sugarcane. Front. Microbiol. 2019, 9, 3262. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D.H.; Kumar, R.; Headland, L.R.; Ranjan, A.; Covington, M.F.; Ichihashi, Y.; Fulop, D.; Jiménez-Gómez, J.M.; Peng, J.; Maloof, J.N.; et al. A quantitative genetic basis for leaf morphology in a set of precisely defined tomato introgression lines. Plant Cell 2013, 25, 2465–2481. [Google Scholar] [CrossRef] [PubMed]
- Recchia, I.; Sparla, F.; Pupillo, P. Photosynthetic properties of spring geophytes assessed by chlorophyll fluorescence analysis. Plant Physiol. Biochem. 2017, 118, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Motos, J.R.; Noguera-Vera, L.; Barba-Espín, G.; Piqueras, A.; Hernández, J.A. Antioxidant Metabolism and Chlorophyll Fluorescence during the Acclimatisation to Ex Vitro Conditions of Micropropagated Stevia rebaudiana Bertoni Plants. Antioxidants 2019, 8, 615. [Google Scholar] [CrossRef] [PubMed]
- Peršić, V.; Ament, A.; Antunović Dunić, J.; Drezner, G.; Cesar, V. PEG-induced physiological drought for screening winter wheat genotypes sensitivity—Integrated biochemical and chlorophyll a fluorescence analysis. Front. Plant Sci. 2022, 13, 987702. [Google Scholar] [CrossRef]
- Falcioni, R.; Antunes, W.C.; Demattê, J.A.M.; Nanni, M.R. Biophysical, Biochemical, and Photochemical Analyses Using Reflectance Hyperspectroscopy and Chlorophyll a Fluorescence Kinetics in Variegated Leaves. Biology 2023, 12, 704. [Google Scholar] [CrossRef]
- Yano, K.; Morinaka, Y.; Wang, F.; Huang, P.; Takehara, S.; Hirai, T.; Ito, A.; Koketsu, E.; Kawamura, M.; Kotake, K.; et al. GWAS with principal component analysis identifies a gene comprehensively controlling rice architecture. Proc. Natl. Acad. Sci. USA 2019, 116, 21262–21267. [Google Scholar] [CrossRef]
- Cowling, S.B.; Treeintong, P.; Ferguson, J.; Soltani, H.; Swarup, R.; Mayes, S.; Murchie, E.H.; Lawson, T. Out of Africa: Characterizing the natural variation in dynamic photosynthetic traits in a diverse population of African rice (Oryza glaberrima). J. Exp. Bot. 2022, 73, 3283–3298. [Google Scholar] [CrossRef]
- Sommer, S.G.; Han, E.; Li, X.; Rosenqvist, E.; Liu, F. The Chlorophyll Fluorescence Parameter Fv/Fm Correlates with Loss of Grain Yield after Severe Drought in Three Wheat Genotypes Grown at Two CO2 Concentrations. Plants 2023, 12, 436. [Google Scholar] [CrossRef]
- Quagliata, G.; Abdirad, S.; Celletti, S.; Sestili, F.; Astolfi, S. Screening of Triticum turgidum genotypes for tolerance to drought stress. Plant Physiol. Biochem. PPB 2023, 194, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Yang, J.; Huang, Y.; Liu, J.; Ma, X.; Zhao, L.; Ma, C.; Tu, J.; Shen, J.; Fu, T.; et al. Bulk segregant analysis-sequencing and RNA-Seq analyses reveal candidate genes associated with albino phenotype in Brassica napus. Front. Plant Sci. 2022, 13, 994616. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Zhang, J.; Li, D.; Yan, L.; You, M.; Zhang, J.; Lei, X.; Chang, D.; Ji, X.; et al. Physiological and Proteomic Responses of Contrasting Alfalfa (Medicago sativa L.) Varieties to High Temperature Stress. Front. Plant Sci. 2021, 12, 753011. [Google Scholar] [CrossRef] [PubMed]
- Basu, P.S.; Pratap, A.; Gupta, S.; Sharma, K.; Tomar, R.; Singh, N.P. Physiological Traits for Shortening Crop Duration and Improving Productivity of Greengram (Vigna radiata L. Wilczek) Under High Temperature. Front. Plant Sci. 2019, 10, 1508. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Chen, K.C.; Cheng, T.S.; Lee, C.; Lin, S.H.; Tung, C.W. Chlorophyll fluorescence analysis in diverse rice varieties reveals the positive correlation between the seedlings salt tolerance and photosynthetic efficiency. BMC Plant Biol. 2019, 19, 403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Sun, D.; Cheng, K.W.; Chen, F. Investigation of carbon and energy metabolic mechanism of mixotrophy in Chromochloris zofingiensis. Biotechnol. Biofuels 2021, 14, 36. [Google Scholar] [CrossRef] [PubMed]
- Kolar, C.; Adám, E.; Schäfer, E.; Nagy, F. Expression of tobacco genes for light-harvesting chlorophyll a/b binding proteins of photosystem II is controlled by two circadian oscillators in a developmentally regulated fashion. Proc. Natl. Acad. Sci. USA 1995, 92, 2174–2178. [Google Scholar] [CrossRef]
- Lu, G.; Pan, Y.-B.; Wang, Z.; Xu, F.; Cheng, W.; Huang, X.; Ren, H.; Pang, C.; Que, Y.; Xu, L. Utilization of a Sugarcane100K Single Nucleotide Polymorphisms Microarray-Derived High-Density Genetic Map in Quantitative Trait Loci Mapping and Function Role Prediction of Genes Related to Chlorophyll Content in Sugarcane. Front. Plant Sci. 2021, 12, 817875. [Google Scholar] [CrossRef]
- Basnayake, J.; Jackson, P.A.; Inman-Bamber, N.G.; Lakshmanan, P. Sugarcane for water-limited environments. Variation in stomatal conductance and its genetic correlation with crop productivity. J. Exp. Bot. 2015, 66, 3945–3958. [Google Scholar] [CrossRef]
- Nasyrov, Y.S. Genetic Control of Photosynthesis and Improving of Crop Productivity. Annu. Rev. Plant Physiol. 1978, 29, 215–237. [Google Scholar] [CrossRef]
- Koussevitzky, S.; Nott, A.; Mockler, T.C.; Hong, F.; Sachetto-Martins, G.; Surpin, M.; Lim, J.; Mittler, R.; Chory, J. Signals from chloroplasts converge to regulate nuclear gene expression. Science 2007, 316, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Calleja, M.; Monteagudo, A.; Casas, A.M.; Boutin, C.; Pin, P.A.; Morales, F.; Igartua, E. Rapid On-Site Phenotyping via Field Fluorimeter Detects Differences in Photosynthetic Performance in a Hybrid—Parent Barley Germplasm Set. Sensors 2020, 20, 1486. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Greaves, I.K.; Groszmann, M.; Wu, L.M.; Dennis, E.S.; Peacock, W.J. Hybrid mimics and hybrid vigor in Arabidopsis. Proc. Natl. Acad. Sci. USA 2015, 112, E4959–E4967. [Google Scholar] [CrossRef] [PubMed]
- Banco, A.; Trentacoste, E.; Monasterio, R.P. Characterization of virgin olive oils from Spanish olive varieties introduced in Mendoza, Argentina, and their comparison with the autochthonous variety. J. Sci. Food Agric. 2021, 101, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Mahenderakar, M.D.; Jugran, A.K.; Singh, R.K.; Srivastava, R.K. Assessing genetic diversity and population structure of sugarcane cultivars, progenitor species and genera using microsatellite (SSR) markers. Gene 2020, 753, 144800. [Google Scholar] [CrossRef]
- Lauterberg, M.; Saranga, Y.; Deblieck, M.; Klukas, C.; Krugman, T.; Perovic, D.; Ordon, F.; Graner, A.; Neumann, K. Precision phenotyping across the life cycle to validate and decipher drought-adaptive QTLs of wild emmer wheat (Triticum turgidum ssp. dicoccoides) introduced into elite wheat varieties. Front. Plant Sci. 2022, 13, 965287. [Google Scholar] [CrossRef]
- Li, X.; Ran, R.; Chen, G.; Zhao, P. Genomic Variation Underlying the Breeding Selection of Quinoa Varieties Longli-4 and CA3-1 in China. Int. J. Mol. Sci. 2022, 23, 4030. [Google Scholar] [CrossRef]
- Rajan, N.; Debnath, S.; Perveen, K.; Khan, F.; Pandey, B.; Srivastava, A.; Khanam, M.N.; Subramaniyan, V.; Kumarasamy, V.; Paul, P.J.; et al. Optimizing hybrid vigor: A comprehensive analysis of genetic distance and heterosis in eggplant landraces. Front. Plant Sci. 2023, 14, 1238870. [Google Scholar] [CrossRef]
- Lu, S.; Zhao, X.; Hu, Y.; Liu, S.; Nan, H.; Li, X.; Fang, C.; Cao, D.; Shi, X.; Kong, L.; et al. Natural variation at the soybean J locus improves adaptation to the tropics and enhances yield. Nat. Genet. 2017, 49, 773–779. [Google Scholar] [CrossRef]
- Kokilananthan, S.; Bulugahapitiya, V.P.; Manawadu, H.; Gangabadage, C.S. Sesquiterpenes and monoterpenes from different varieties of guava leaf essential oils and their antioxidant potential. Heliyon 2022, 8, e12104. [Google Scholar] [CrossRef]
- Nagano, Y.; Tashiro, H.; Nishi, S.; Hiehata, N.; Nagano, A.J.; Fukuda, S. Genetic diversity of loquat (Eriobotrya japonica) revealed using RAD-Seq SNP markers. Sci. Rep. 2022, 12, 10200. [Google Scholar] [CrossRef] [PubMed]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 201–247. [Google Scholar]
- Wang, M.; Li, X.; Shen, Y.; Adnan, M.; Mao, L.; Lu, P.; Hu, Q.; Jiang, F.; Khan, M.T.; Deng, Z.; et al. A systematic high-throughput phenotyping assay for sugarcane stalk quality characterization by near-infrared spectroscopy. Plant Methods 2021, 17, 76. [Google Scholar] [CrossRef] [PubMed]
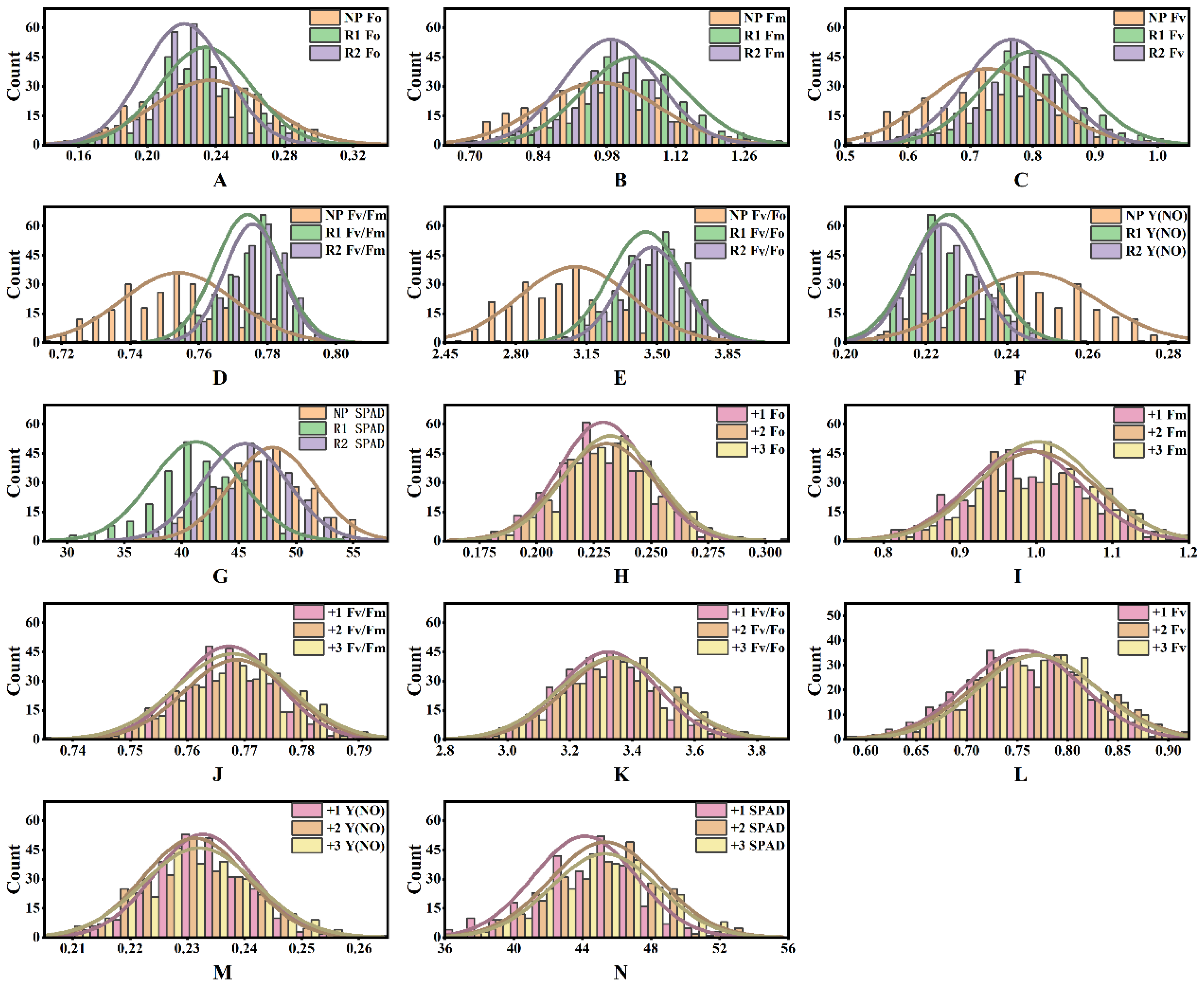
| Source of Variation | df | Fm | Fo | Fv | Fv/Fm | Fv/Fo | Y(NO) | SPAD | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Square | SS (%) | Mean Square | SS (%) | Mean Square | SS (%) | Mean Square | SS (%) | Mean Square | SS (%) | Mean Square | SS (%) | Mean Square | SS (%) | ||
| Genotype (G) | 257 | 0.1271 *** | 20.1 | 0.00905 *** | 23.0 | 0.076 *** | 19.0 | 0.0019 *** | 18.6 | 0.62 *** | 19.5 | 0.0019 *** | 18.6 | 236 *** | 28.5 |
| Year (Y) | 2 | 3.0716 *** | 3.8 | 0.15122 *** | 3.0 | 3.215 *** | 6.3 | 0.3431 *** | 26.2 | 102.73 *** | 25.2 | 0.3431 *** | 26.2 | 26540 *** | 25.0 |
| Leaf Position (L) | 2 | 0.1803 *** | 0.2 | 0.00569 *** | 0.1 | 0.124 *** | 0.23 | 0.0011 *** | 0.1 | 0.44 *** | 0.1 | 0.0011 *** | 0.1 | 1066 *** | 1.1 |
| Rep (R) | 2 | 0.0695 * | 0.1 | 0.00553 * | 0.1 | 0.036 * | 0.1 | 0.0001 | 0.0 | 0.07 | 0.0 | 0.0001 | 0.0 | 11 | 0.0 |
| G × Y | 514 | 0.0974 *** | 30.8 | 0.0064 *** | 32.5 | 0.058 *** | 29.0 | 0.001 *** | 19.6 | 0.31 *** | 19.3 | 0.001 *** | 19.6 | 77 *** | 18.5 |
| G × L | 514 | 0.0163 *** | 5.6 | 0.00095 *** | 4.9 | 0.01 *** | 5.1 | 0.0002 *** | 4.0 | 0.06 *** | 4.1 | 0.0002 *** | 4.0 | 12 *** | 2.9 |
| Y × L | 4 | 0.3466 *** | 0.9 | 0.00957 *** | 0.4 | 0.241 *** | 0.9 | 0.0016 *** | 0.2 | 0.53 *** | 0.3 | 0.0016 *** | 0.2 | 1187 *** | 2.2 |
| G × Y × L | 1028 | 0.014 *** | 8.9 | 0.00076 *** | 7.7 | 0.009 *** | 9.0 | 0.0002 *** | 6.6 | 0.05 ** | 6.4 | 0.0002 *** | 6.6 | 12 *** | 5.9 |
| Residuals | 4642 | 0.0106 | 30.2 | 0.00062 | 28.3 | 0.007 | 30.3 | 0.0001 | 24.7 | 0.04 | 25.0 | 0.0001 | 24.7 | 7 | 16.0 |
| h2 (%) | 76.13 | 77.97 | 76.06 | 81.69 | 82.63 | 81.69 | 88.3 | ||||||||
| Traits | PC1 | PC2 | PC3 |
|---|---|---|---|
| Fm | 0.507 | 0.856 | −0.102 |
| Fo | 0.838 | 0.537 | −0.074 |
| Fv | 0.366 | 0.924 | −0.107 |
| Fv/Fm | −0.880 | 0.473 | −0.024 |
| Fv/Fo | −0.878 | 0.475 | −0.013 |
| Y(NO) | 0.880 | −0.473 | 0.024 |
| SPAD | 0.103 | 0.266 | 0.958 |
| Eigenvalue | 3.42 | 2.62 | 0.95 |
| Proportion of Variance | 48.92 | 37.40 | 13.53 |
| Cumulative Proportion | 48.92 | 86.31 | 99.85 |
| SS (%) | 48.99 | 37.46 | 13.55 |
| Grade of Photosynthetic | High Photosynthetic Efficiency (HPE) | Moderate Photosynthetic Efficiency (MPE) | Low Photosynthetic Efficiency (LPE) |
|---|---|---|---|
| Tested genotype | 6105, 24201, 09-175, 11-11319, 12-20318, 12-6403, 14-10006, 14-12012, 14-12506, 14-14707, 14-15239, 14-15418, 14-18504, 14-21001, 14-2244, 14-2802, 14-3508, 14-8004, 14-8704, 15-0102, 15-1103, 15-11106, 15-16850, 15-18106, 15-22809, 15-23304, 15-3303, 15-42, 15-4203, 15-451, 15-452, 15-4818, 15-701, 15-793, 15-W3, 16-041, 16-0628, 16-063, 16-0812, 16-084, 16-0916, 16-0920, 16-0924, 16-0930, 16-0934, 16-0941, 16-098, 16-1015, 16-104, 16-106, 16-11708, 16-12026, 16-12509, 16-1322, 16-1342, 16-137, 16-151, 16-154, 16-1811, 16-184, 16-192, 16-195, 16-2026, 16-226, 16-231, 16-253, 16-255, 16-262, 16-271, 16-401, 16-7010, 16-7506, 16-8716, 19-607, FG2, FN0335, FN10-0574, GT03-351, GT42, GT05-378, GUC13, GUC23, GZ74-141, ROC22, X, YG39 | 3203, 12-1801, 13-1105, 13-4007, 14-14325, 14-18509, 14-19220, 14-20701, 14-21107, 14-2720, 14-4315, 15-1005, 15-1106, 15-2007, 15-4513, 15-5404, 15-6204, 15-791, 15-794, 16-064, 16-065, 16-066, 16-0914, 16-0928, 16-0936, 16-0939, 16-0953, 16-0954, 16-12506, 16-1330, 16-136, 16-15220, 16-1612, 16-167, 16-186, 16-187, 16-222, 16-223, 16-224, 16-22402, 16-256, 16-453, 16-7019, 16-7705, 16-7722, 16-832, 16-8801, GT92-66, GT94-119, GUC17, GUC25, GUC29, GUC41, GUC8, GZ96-126, ROC27, Taiyin14, TB3, Xi096, ZZ9 | 3717, 6101, 8914, 35365, 40375, 11-2819, 14-15220, 06-0918, 10-228, 11-20318, 11-601, 12-106, 12-12803, 12-14602, 12-17204, 12-34, 13-11008, 13-11919, 13-14812, 13-18402, 13-21501, 14-002, 14-12712, 14-1854, 14-2149, 14-3902, 14-434, 14-509, 14-5603, 14-8009, 14-8705, 14-8903, 15-14701, 15-1743, 15-2010, 15-421, 15-453, 15-5306, 15-6008, 15-6201, 15-6402, 15-702, 15-9904, 16-043, 16-087, 16-088, 16-091, 16-0911, 16-0913, 16-0917, 16-092, 16-0926, 16-0927, 16-0929, 16-093, 16-0931, 16-0942, 16-096, 16-10002, 16-11203, 16-11905, 16-12512, 16-1329, 16-1331, 16-1335, 16-142, 16-144, 16-163, 16-168, 16-1715, 16-182, 16-188, 16-198, 16-22419, 16-225, 16-232, 16-251, 16-264, 16-3205, 16-3417, 16-5402, 16-615, 16-7719, 16-803, 16-831, 16-8701, 16-8804, 20-718, 6010A, CP01-1372, FG3, FN04-3504, Fujiandaye, Ganjiang18, GT02-390, GUC10, GUC16, GUC2, GUC21, GUC3, GUC31, GUC35, GUC7, LC05-129, ROC16, Shuidian25, TB1, TB11, YC64-389, YG24, YR03-425, YR99-596 |
| Fm | 1.001 ± 0.062 B | 1.042 ± 0.052 A | 0.963 ± 0.065 C |
| Fo | 0.225 ± 0.016 B | 0.242 ± 0.016 A | 0.229 ± 0.019 B |
| Fv/Fm | 0.776 ± 0.005 A | 0.768 ± 0.005 B | 0.762 ± 0.006 C |
| Fv/Fo | 3.484 ± 0.103 A | 3.333 ± 0.097 B | 3.228 ± 0.106 C |
| Fv | 0.78 ± 0.047 B | 0.800 ± 0.038 A | 0.734 ± 0.047 C |
| Y(NO) | 0.224 ± 0.005 C | 0.232 ± 0.005 B | 0.238 ± 0.006 A |
| SPAD | 46.3 ± 2.359 A | 42.6 ± 2.3 C | 45.1 ± 2.9 B |
| NO. | 86 | 60 | 112 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, Y.; Xu, Y.; Khan, A.; Jiang, C.; Li, H.; Wu, Y.; Zhang, C.; Wang, M.; Chen, J.; Zeng, L.; et al. Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane. Plants 2024, 13, 587. https://doi.org/10.3390/plants13050587
Wei Y, Xu Y, Khan A, Jiang C, Li H, Wu Y, Zhang C, Wang M, Chen J, Zeng L, et al. Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane. Plants. 2024; 13(5):587. https://doi.org/10.3390/plants13050587
Chicago/Turabian StyleWei, Yibin, Yuzhi Xu, Abdullah Khan, Chunxiu Jiang, Huojian Li, Yuling Wu, Chi Zhang, Maoyao Wang, Jun Chen, Lifang Zeng, and et al. 2024. "Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane" Plants 13, no. 5: 587. https://doi.org/10.3390/plants13050587
APA StyleWei, Y., Xu, Y., Khan, A., Jiang, C., Li, H., Wu, Y., Zhang, C., Wang, M., Chen, J., Zeng, L., & Zhang, M. (2024). Analysis of Photosynthetic Characteristics and Screening High Light-Efficiency Germplasm in Sugarcane. Plants, 13(5), 587. https://doi.org/10.3390/plants13050587







