Fungal Hyphae on the Assimilation Branches Are Beneficial for Haloxylon ammodendron to Absorb Atmospheric Water Vapor: Adapting to an Extreme Drought Environment
Abstract
:1. Introduction
2. Results
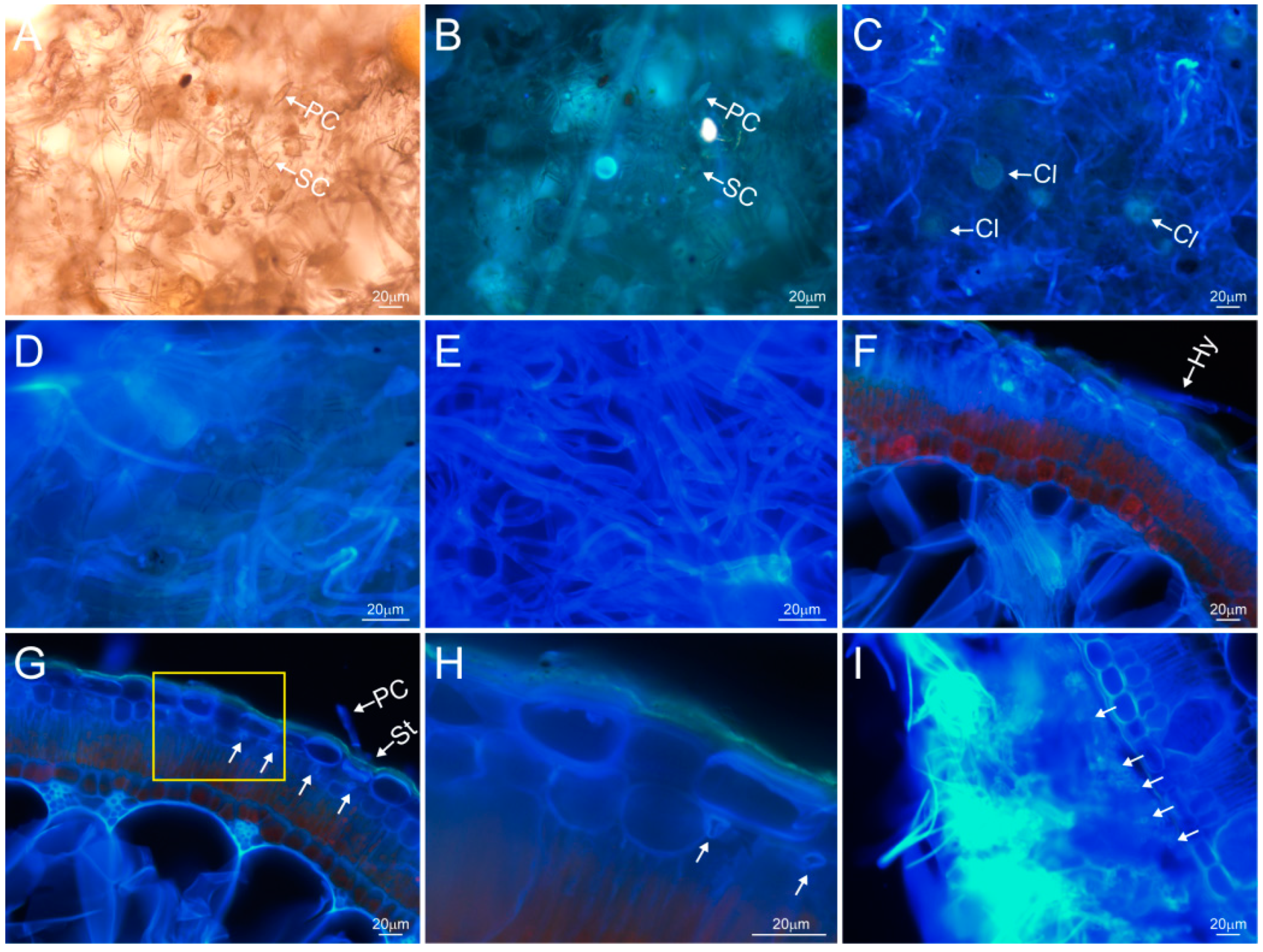
2.1. Fungal Infiltration Law, Morphology, and Mycelium Structure Characteristics
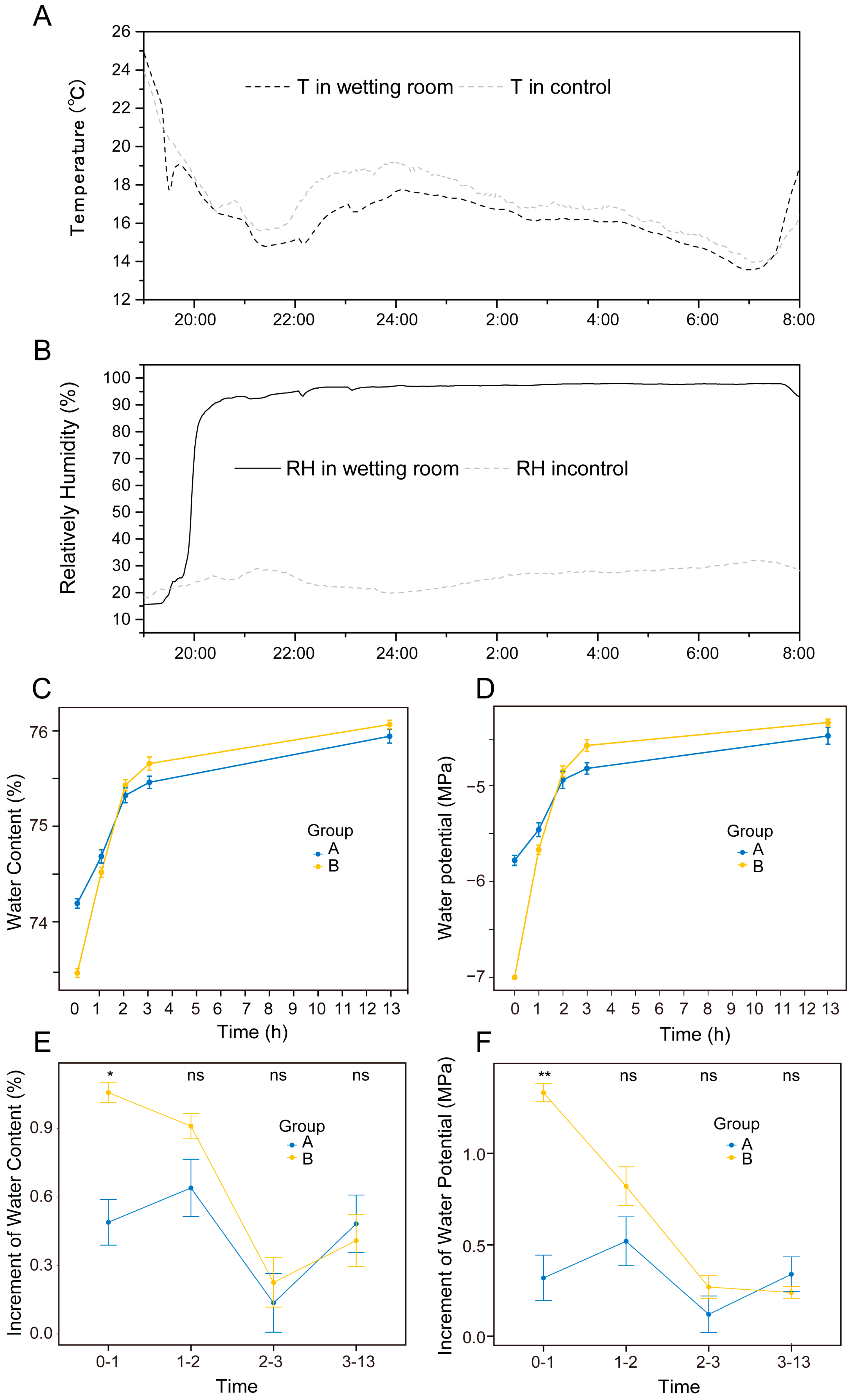
2.2. Comparison of Water Content and Water Potential of Assimilation Branches, with Hyphae and without Hyphae, under Ultrapure Water Humidification
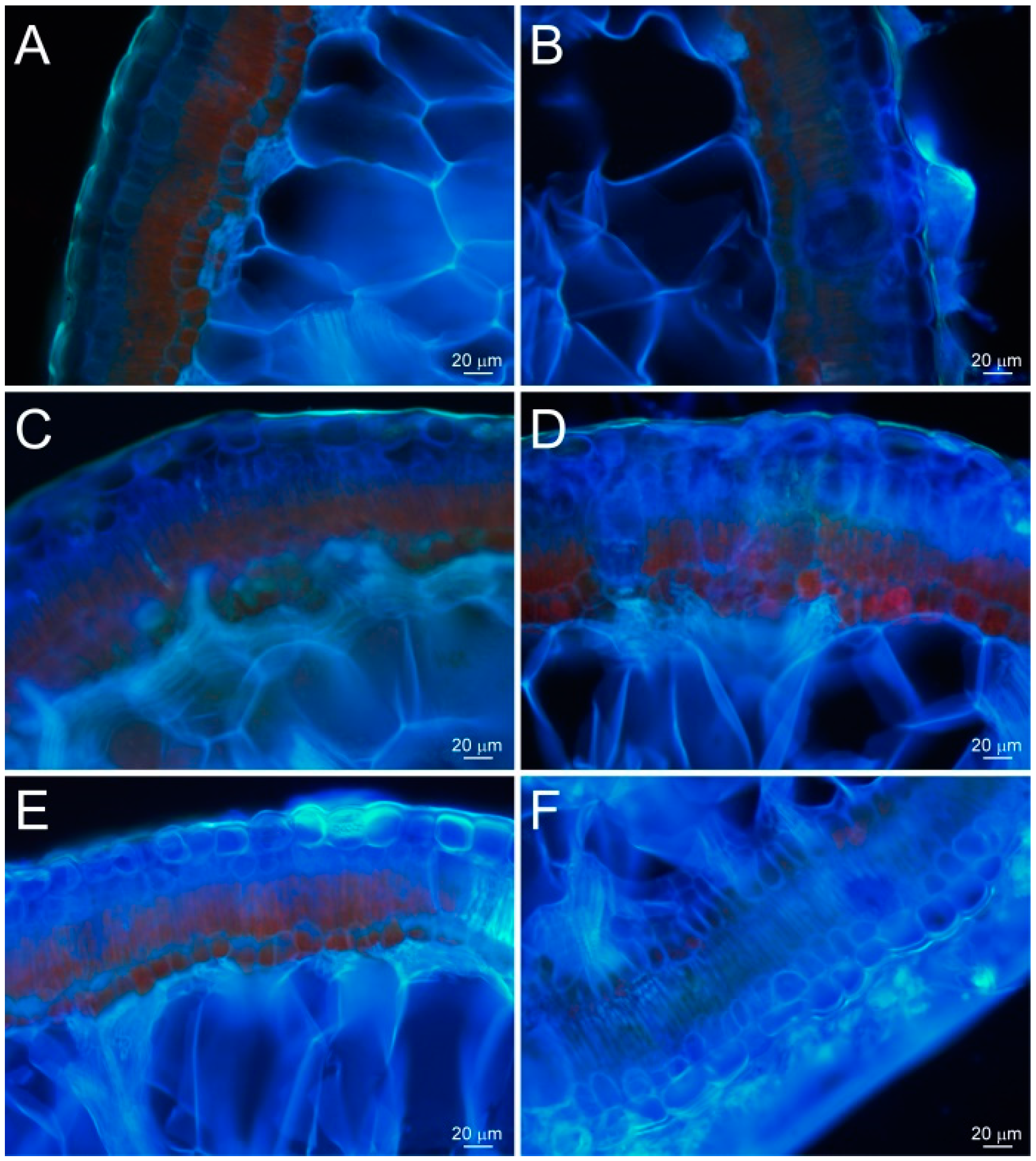
2.3. Water Absorption Sites and Characteristics of Assimilating Branches with Hyphae
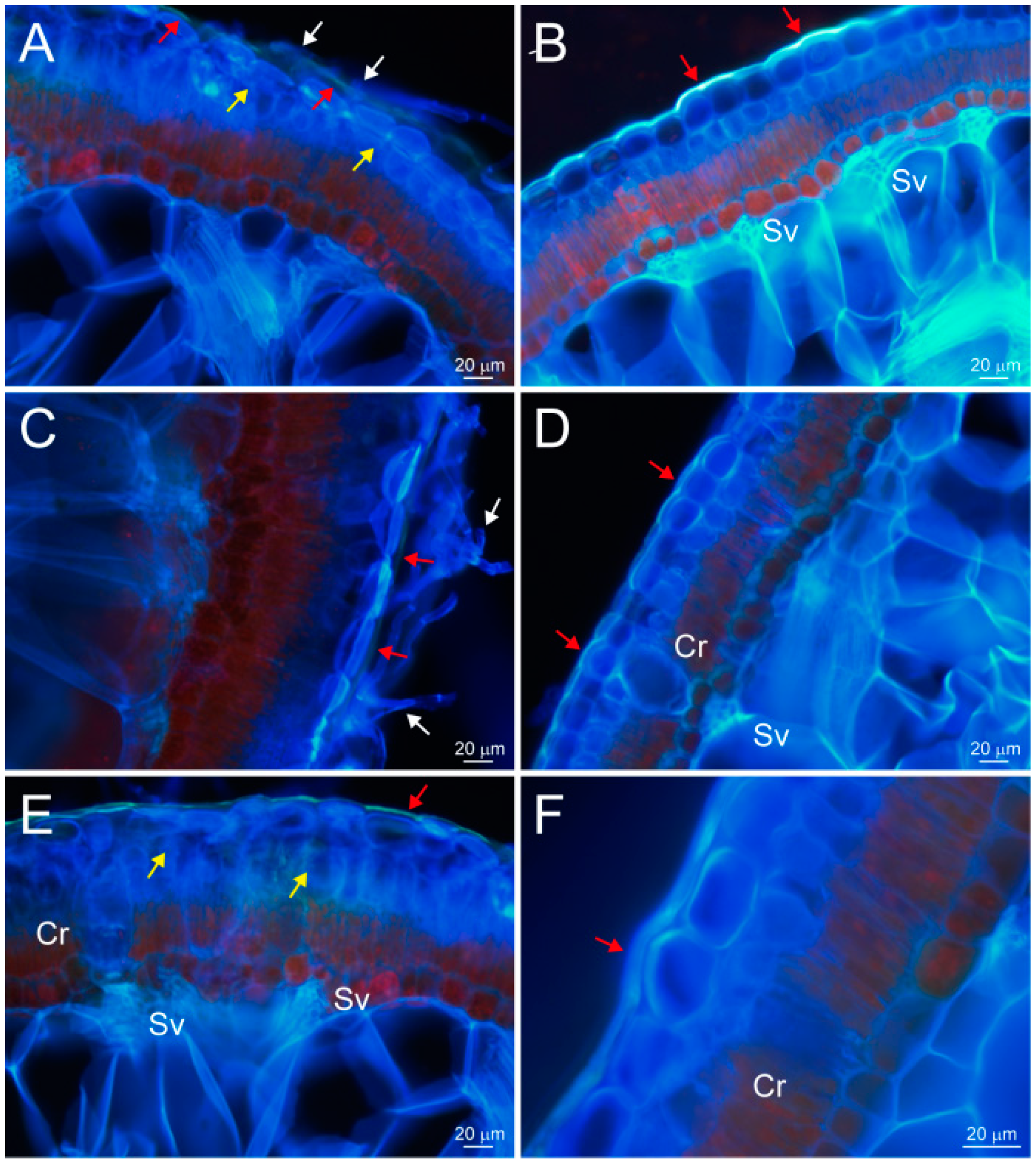
2.4. Transcuticular Transport of Hyphae Promotes Water Absorption through the Assimilating Branches of Haloxylon ammodendron
3. Discussion
3.1. Analysis of the Mechanism of Hyphae Promoting Water Absorption
3.2. The Growth of Hyphae on the Assimilation Branches of Haloxylon ammodendron Is an Inevitable Ecological Process of Arid Ecological Adaptation
3.3. New Insights and Significance between White Powder Fungi and Haloxylon ammodendron
4. Experimental Site, Materials, and Methods
4.1. Experimental Site and Materials
4.2. Experimental Methods
4.2.1. Humidification Experiment
4.2.2. Plant Water Potential
4.2.3. Moisture Content Determination
4.2.4. Fluorescence Tracer Detection
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, S.H.; Xu, X.F.; Sun, Y.M.; Jun, L.; Li, C.Z. Influence of drought hardening on the resistance physiology of potato seedlings under drought stress. J. Integr. Agric. 2018, 17, 336–347. [Google Scholar] [CrossRef]
- Tanveer, M.; Shahzad, B.; Sharma, A.; Khan, E.A. 24-Epibrassinolide application in plants: An implication for improving drought stress tolerance in plants. Plant Physiol. Biochem. 2019, 135, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xin, K.; Liu, H.; Cheng, J.; Shen, X.; Wang, Y.; Zhang, L. Pantoea alhagi, a novel endophytic bacterium with ability to improve growth and drought tolerance in wheat. Sci. Rep. 2017, 7, 41564. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; He, X.; Hou, L.; Ren, Y.; Wang, S.; Su, F. Dark septate endophytes isolated from a xerophyte plant promote the growth of Ammopiptanthus mongolicus under drought condition. Sci. Rep. 2018, 8, 7896. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.E.D. Plant growth-promoting activities for bacterial and fungal endophytes isolated from medicinal plant of Teucrium polium L. J. Adv. Res. 2017, 8, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Arnold, A.E.; Mejía, L.C.; Kyllo, D.; Rojas, E.I.; Maynard, Z.; Robbins, N.; Herre, E.A. Fungal endophytes limit pathogen damage in a tropical tree. Proc. Natl. Acad. Sci. USA 2003, 100, 15649–15654. [Google Scholar] [CrossRef] [PubMed]
- Arnold, A.E.; Henk, D.A.; Eells, R.L.; Lutzoni, F.; Vilgalys, R. Diversity and phylogenetic affinities of foliar fungal endophytes in loblolly pine inferred by culturing and environmental PCR. Mycologia 2007, 99, 185–206. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, M.A.; Brachmann, A.; Begerow, D.; Peršoh, D. Transient leaf endophytes are the most active fungi in 1-year-old beech leaf litter. Fungal Divers. 2018, 89, 237–251. [Google Scholar] [CrossRef]
- Jumpponen, A.; Jones, K.L. Massively parallel 454 sequencing indicates hyperdiverse fungal communities in temperate Quercus macrocarpa phyllosphere. New Phytol. 2009, 184, 438–448. [Google Scholar] [CrossRef]
- Kannadan, S.; Rudgers, J.A. Endophyte symbiosis benefits a rare grass under low water availability. Funct. Ecol. 2008, 22, 706–713. [Google Scholar] [CrossRef]
- Waller, F.; Achatz, B.; Baltruschat, H.; Fodor, J.; Becker, K.; Fischer, M.; Heier, T.; Hückelhoven, R.; Neumann, C.; Wettstein, D.V.; et al. The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 2005, 102, 13386–13391. [Google Scholar] [CrossRef]
- Xu, G.Q.; Mcdowell, N.G.; Li, Y. A possible link between life and death of a xeric tree in desert. J. Plant Physiol. 2016, 194, 35–44. [Google Scholar] [CrossRef]
- Xu, H.; Li, Y.; Xu, G.Q.; Zou, T. Ecophysiological response and morphological adjustment of two Central Asian desert shrubs towards variation in summer precipitation. Plant Cell Environ. 2007, 30, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Tashpolat, T.; Li, Y.; Tang, L.S.; Fan, L.L. The survival and above/below ground growth of H. ammodendron ammodendron seedling. Acta Ecol. Sin. 2014, 34, 2012–2019. [Google Scholar]
- Chen, Y.C.; Li, M.; Wu, M.Z.; Song, Y.X. Structure and composition of roots in two species of H. ammodendron Bunge. Plant Physiol. J. 2013, 49, 1273–1276. [Google Scholar]
- Song, Y.Y.; Li, R.; Luo, H.W.; Zhang, W.H. Reproductive allocation characteristics of H. ammodendron ammodendron populations in three habitats of Gurbantunggut desert. Chin. J. Ecol. 2012, 31, 837–843. [Google Scholar]
- Huang, Z.; Zhang, X.; Zheng, G.; Gutterman, Y. Influence of light, temperature, salinity and storage on seed germination of H. ammodendron. J. Arid Environ. 2003, 55, 453–464. [Google Scholar] [CrossRef]
- Dai, Y.; Zheng, X.J.; Tang, L.S.; Li, Y. Stable oxygen isotopes reveal distinct water use patterns of two H. ammodendron species in the Gurbantonggut Desert. Plant Soil 2015, 389, 73–87. [Google Scholar] [CrossRef]
- Yang, Q.Y.; Zhao, W.Z.; Liu, B.; Liu, H. Physiological responses of H. ammodendron to rainfall pulses in temperate desert regions, Northwestern China. Trees 2014, 28, 709–722. [Google Scholar]
- Yan, Q.D.; Su, P.X.; Chen, H.B.; Zhang, L.M. Comparative studies on crystal idioblasts of five desert C4 plants. J. Plant Ecol. (Chin. Version) 2008, 32, 873–882. [Google Scholar]
- Zhao, Z.Y. Xinjiang Powdery Mildew Flora; Xinjiang People’s Publishing House: Urumqi, Xinjiang, 1979. [Google Scholar]
- Kong, T.T.; Liu, A.H.; Zhang, J.W. The occurrence and prevention of major diseases of H. ammodendron in Xinjiang. Prot. For. Sci. Technol. 2019, 9, 91–93. [Google Scholar]
- Tursun, T. Integrated control technology of H. ammodendron mildew. Mod. Hortic. 2015, 2, 56. [Google Scholar]
- Bruijnzeel, L.A.; Waterloo, M.J.; Proctor, J.; Kuiters, A.T.; Kotterink, B. Hydrological observations in montane rain forests on Gunung Silam, Sabah, Malaysia, with special reference to the Massenerhebung effect. J. Ecol. 1993, 81, 145–167. [Google Scholar] [CrossRef]
- Eller, C.B.; Lima, A.L.; Oliveira, R.S. Foliar uptake of fog water and transport belowground alleviates drought effects in the cloud forest tree species, Drimys brasiliensis (Winteraceae). New Phytol. 2013, 199, 151–162. [Google Scholar] [CrossRef]
- Oliveira, R.S.; Dawson, T.E.; Burgess, S.S.O. Evidence for direct water absorption by the shoot of the desiccation–tolerant plant Vellozia flavicans in the savannas of central Brazil. J. Trop. Ecol. 2005, 21, 585–588. [Google Scholar] [CrossRef]
- Hao, X.M.; Li, C.; Guo, B.; Ma, J.X.; Ayup, M.; Chen, Z.S. Dew formation and its long–term in a desert riparian forest ecosystem on the eastern edge of the Taklimakan Desert in China. J. Hydrol. 2012, 472, 90–98. [Google Scholar] [CrossRef]
- Zhang, Y.; Hao, X.M.; Sun, H.T.; Hua, D.; Qin, J.X. How Populus euphratica utilizes dew in an extremely arid region. Plant Soil 2019, 443, 493–508. [Google Scholar] [CrossRef]
- Li, Z.K.; Gong, X.W.; Wang, J.L.; Chen, Y.D.; Liu, F.Y.; Li, H.P.; Lv, G.H. Foliar water uptake improves branch water potential and photosynthetic capacity in Calligonum mongolicum. Ecol. Indic. 2023, 146, 109825. [Google Scholar] [CrossRef]
- Stone, E.C.; Went, F.W.; Young, C.L. Water absorption from the atmosphere by plants growing in dry soil. Science 1950, 111, 546–548. [Google Scholar] [CrossRef]
- Limm, E.B.; Simonin, K.A.; Bothman, A.G.; Dawson, T.E. Foliar water uptake: A common water acquisition strategy for plants of the redwood forest. Oecologia 2009, 161, 449–459. [Google Scholar] [CrossRef]
- Ebner, M.; Miranda, T.; Nebelsick, A.R. Efficient fog harvesting by Stipagrostis sabulicola (Namib dune bushman grass). J. Arid Environ. 2011, 75, 524–531. [Google Scholar] [CrossRef]
- Limm, E.B.; Dawson, T.E. Polystichum munitum (Dryopteridaceae) varies geographically in its capacity to absorb fog water by foliar uptake within the redwood forest ecosystem. Am. J. Bot. 2010, 97, 1121–1128. [Google Scholar] [CrossRef]
- Li, Z.K.; Chen, Y.D.; Wang, H.M.; Zhang, X.N. Foliar Water Uptake and Its Relationship with Photosynthetic Capacity and Anatomical Structure between Female and Male Populus euphratica at Different Growth Stages. Forests 2023, 14, 1444. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Barthlott, W. Multifunctional surface structures of plants: An inspiration for biomimetics. Prog. Mater. Sci. 2009, 54, 137–178. [Google Scholar] [CrossRef]
- Burkhardt, J. Hygroscopic particles on leaves: Nutrients or desiccants? Ecol. Monogr. 2010, 80, 369–399. [Google Scholar] [CrossRef]
- Fernandez, V.; Gil-Pelegrin, E.; Eichert, T. Foliar water and solute absorption: An update. Plant J. 2021, 105, 870–883. [Google Scholar] [CrossRef]
- Rundel, P.W. Encyclopedia of Plant Physiology Plant Ecology II Water Relations and Carbon Assimilation; Springer: Berlin/Heidelberg, Germany, 1982. [Google Scholar]
- Schreel, J.D.; Steppe, K. Foliar water uptake in trees: Negligible or necessary? Trends Plant Sci. 2020, 25, 590–603. [Google Scholar] [CrossRef]
- Wenzel, R.N. Resistance of solid surface to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Wang, H.X.; Xiao, H.L.; Ren, J.; Chen, Y.B.; Yang, Q. An ultrasonic humidification fluorescent tracing method for detecting unsaturated atmospheric water absorption by the aerial parts of desert plants. J. Arid Land 2016, 8, 272–283. [Google Scholar] [CrossRef]
- Fahn, A. Structural and functional properties of trichomes of xeromorphic leaves. Ann. Bot. 1986, 57, 631–637. [Google Scholar] [CrossRef]
- O’Brien, T.P.; McCully, M.E. The study of plant structure. In Principles and Selected Methods; Temacarphi: Melbourne, Australia, 1981. [Google Scholar]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Xiao, H.; Pang, L.; Wang, F. Fungal Hyphae on the Assimilation Branches Are Beneficial for Haloxylon ammodendron to Absorb Atmospheric Water Vapor: Adapting to an Extreme Drought Environment. Plants 2024, 13, 1233. https://doi.org/10.3390/plants13091233
Wang X, Xiao H, Pang L, Wang F. Fungal Hyphae on the Assimilation Branches Are Beneficial for Haloxylon ammodendron to Absorb Atmospheric Water Vapor: Adapting to an Extreme Drought Environment. Plants. 2024; 13(9):1233. https://doi.org/10.3390/plants13091233
Chicago/Turabian StyleWang, Xiaohua, Honglang Xiao, Lei Pang, and Fang Wang. 2024. "Fungal Hyphae on the Assimilation Branches Are Beneficial for Haloxylon ammodendron to Absorb Atmospheric Water Vapor: Adapting to an Extreme Drought Environment" Plants 13, no. 9: 1233. https://doi.org/10.3390/plants13091233




