Abstract
This study presents a comprehensive inventory of bryophytes in Serra dos Órgãos National Park (PARNASO), aiming to evaluate species richness, floristic composition and threatened taxa. Despite the state of Rio de Janeiro being one of the most extensively sampled regions for bryophytes in Brazil, detailed surveys of its conservation units remain scarce. Data were obtained through bibliographic review, herbarium specimen analysis, and new field collections. A total of 504 species were recorded, belonging to 202 genera and 76 families. The park harbors three locally endemic species, eight endemic to Rio de Janeiro, and sixty-nine species endemic to Brazil. Additionally, eleven species were identified as threatened, comprising seven Endangered (EN), two Critically Endangered (CR), and two Vulnerable (VU) according to the IUCN guidelines. PARNASO includes four distinct ecosystems along an altitudinal gradient: sub-montane forest (up to 500 m), montane forest (500–1500 m), upper-montane forest (1500–2000 m), and high-altitude fields (above 2000 m). Montane Forest showed the highest species richness, followed by high-altitude fields, upper-montane forest, and sub-montane forest. The findings highlight PARNASO’s importance in preserving bryophyte diversity in a highly diverse yet understudied region. This work contributes valuable baseline data to support conservation strategies and future ecological studies in Atlantic Forest remnants.
1. Introduction
Serra dos Órgãos National Park (PARNASO), established on November 30th, 1939, holds the distinction of being the third national park created in Brazil. This protected area is situated within the Atlantic Forest domain, specifically in the Serra do Mar massifs and called “Órgãos” in Portuguese because its peaks resemble and are named as “pipe organs”. The Serra do Mar is an extensive mountain range approximately 1500 km along the Brazilian coast, from the northern Santa Catarina State to Rio de Janeiro State [1,2].
PARNASO currently represents one of the most important remaining biodiversity hotspots in Rio de Janeiro State, since its ecosystems are one of the largest centers of flower plant endemism in the Atlantic Forest [3,4]. It also plays a vital role in preserving essential water sources that feed two main watersheds—Paraíba do Sul and several streams of the Baía de Guanabara [5,6,7,8].
The Atlantic Forest domain is one of the most species-rich forests in the tropics [4,9,10]. It is also recognized for having high levels of biodiversity and endemism, expressed via its complex structural environments [11,12]. Tropical forests harbor the highest diversity of bryophyte species in the world, not only due to their structural complexity and microhabitat heterogeneity, but also because of favorable climatic conditions such as high humidity, frequent precipitation, and relatively stable temperatures [13,14]. Some authors have reported that this diversity may vary along the altitudinal range, especially concerning structural and floristic composition [15,16,17,18].
The Atlantic Forest within PARNASO comprises well-preserved forests characterized by four distinct vegetation formations based on altitude, climate, and soil conditions, among other factors [6,19,20,21,22]. In the lower parts around Sede Guapimirim, sub-montane forest prevails, while near Sede at Teresópolis, the forest transitions from montane forest to upper-montane forest, culminating above 2000 m.a.s.l. and high-altitude fields along the trail from Teresópolis to Petrópolis [19].
The species list of the vascular plants in Flora Organensis [19] is strongly based on the work of 18th-century botanists and on collections made especially by A.C. Brade (1881–1971) and C.T. Rizzini (1921–1992) between the 1930s and 1950s of the 20th century. These collectors also recorded bryophytes, and the specimens were studied by Bartram (1954) [19], who identified 146 species. Most bryophyte specimens from Serra dos Órgãos are deposited in the Herbarium of Jardim Botânico do Rio de Janeiro (RB).
Bryophytes from PARNASO were not sufficiently explored, and even considering that, though several researchers collected and published new taxa, the study was dispersed as examined material in taxonomical revisionary work. For example, Faria et al. (2021) [23] attempted to re-collect species founded by Glaziou in Serra dos Órgaos and the taxonomic reclassification of an Atlantic Forest endemic, and Peralta et al. (2020) [24] reported species of the Andreaceae family from the region.
There is no other information on bryophytes in PARNASO; thus, there is no detailed data on the floristic structure and composition of bryophytes. A comprehensive species list will contribute to filling a significant knowledge gap for the state of Rio de Janeiro and support the assessment of threatened bryophyte species. In addition, the revision of herbarium specimens and the incorporation of new collections will help stimulate further research on the ecology and biogeography of bryophytes within this important national park. This work aims to provide an inventory of the bryophyte species in PARNASO and discuss the following topics: (i) species richness and floristic composition; (ii) substrate preferences; and (iii) endemism and conservation status.
2. Results
2.1. Species Richness and Floristic Composition
The present study documented 504 species in 202 genera and 76 families (Table 1). The Serra dos Órgaos National Park preserves 57% of the known bryophyte flora of Rio de Janeiro State and 31% of Brazil. A total of 3000 specimens were analyzed, resulting in the identification of 428 species and an additional 79 species (indicated by * in Table 1) that remain known only from the literature.

Table 1.
Richness and diversity of bryophyte species. Symbols on the species voucher: species only known from the literature (*).
Three species are locally endemic, being known only from the type specimens collected over 100 years ago: Southbya organensis (Lützelburg 6003 p.p. [holotype JE]), Plagiochila pseudoradicans (Lützelburg 6029b [holotype JE]) and Dicranodontium pulchroalare ssp. brasiliense (Lützelburg 1519 [holotype JE]). Additional field research is urgently needed in order to obtain new records and data of these three endemic species.
The most species-rich families of liverworts are Lejeuneaceae (65 spp.) and Lepidoziaceae (16 spp.), while for moss they are Leucobryaceae (30 spp.) and Sphagnaceae (25 spp.). A total of 8 species of hornworts were found, distributed among the families Dendrocerotaceae (4 spp.), Anthocerotaceae (2 spp.) and Nothotyladaceae (2 spp.). The most species-rich genera were Campylopus (Leucobryaceae, 23 spp.); Plagiochila (Plagiochilaceae, 21 spp.); Fissidens (Fissidentaceae, 20 spp.); Lejeunea (Lejeuneaceae, 16 spp.); and Metzgeria (Metzgeriaceae, 14 spp.). Considering the samples analyzed, the most common species with the largest number of records were Campylopus arctocarpus (7 specimens); Lejeunea acanthogona (7); Lejeunea flava (7); Radula nudicaulis (7); Bryum capillare (6); Fissidens wallisii (6); Lejeunea serpillifolioides (6); Rigodium toxarion (6); Trichocolea brevifissa (6); and Zelometeorium ambiguum (6).
2.2. Substrate Preferences
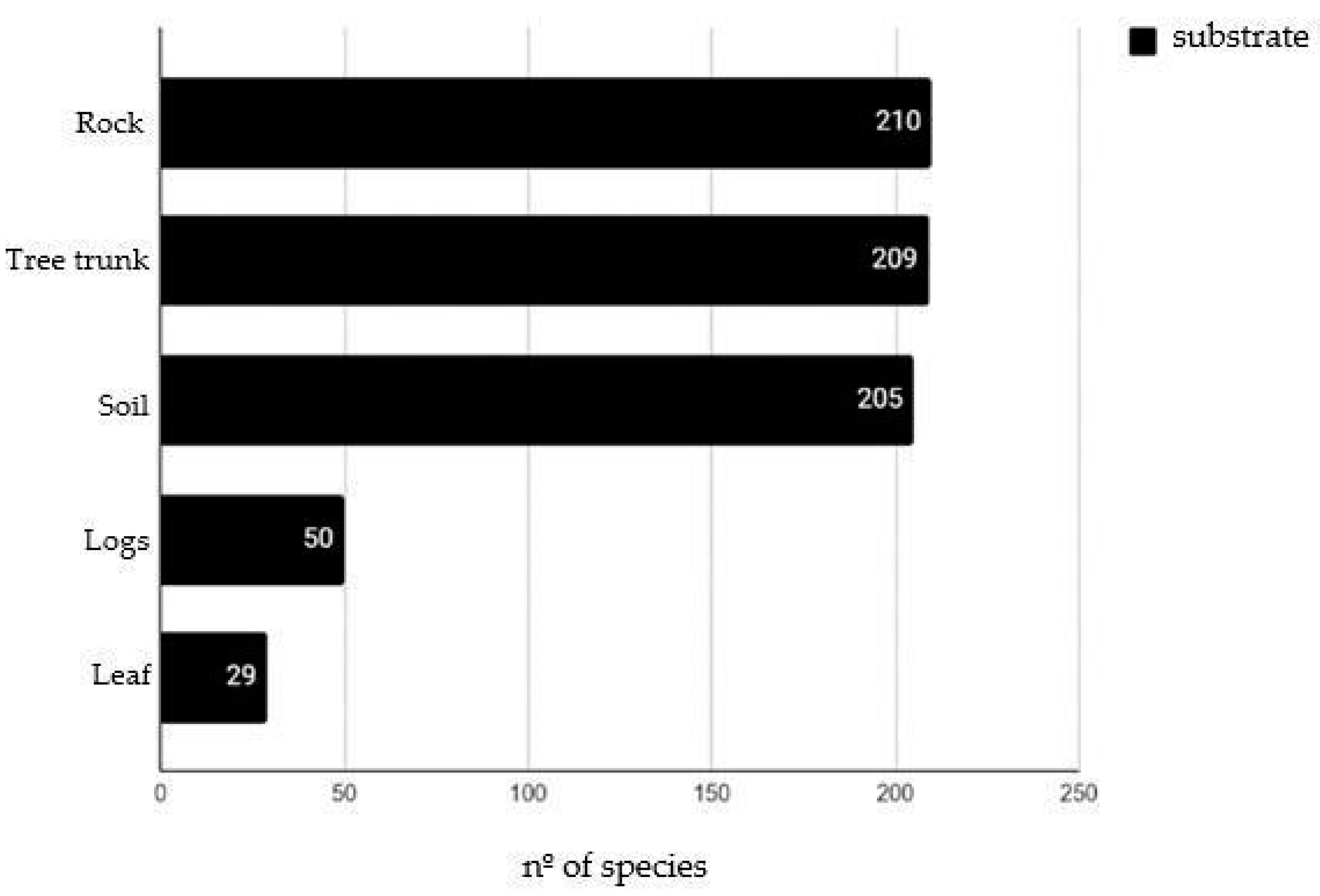
The bryophyte species predominantly colonized rocks (210 spp.), followed by tree trunks (209 spp.), soil (205 spp.), logs (50 spp.), and leaves (29 spp.) (Figure 1).
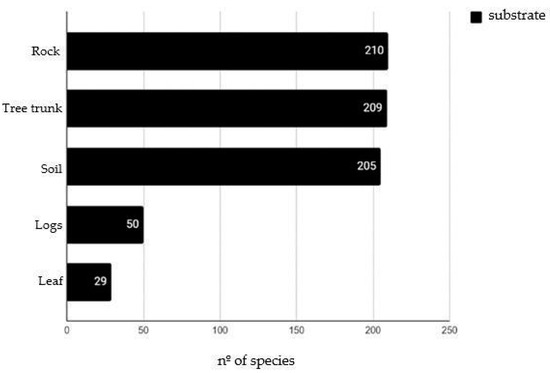
Figure 1.
Substrates colonized by bryophytes in Serra dos Órgãos National Park.
The availability of trees within the forests of PARNASO clearly influences the bryophyte species occurrence. Mosses predominantly colonize tree trunks, with only four species being found on leaves: Paraleucobryum longifolium (Dicranaceae); Philophyllum tenuifolium (Hookeriaceae); Toloxis imponderosa (Meteoriaceae); and Schlotheimia tecta (Orthotrichaceae).
The lowest bryophyte colonization was for leaves (epiphyllous). The leaf-colonizing species most frequent were the leafy liverworts of the family Lejeuneaceae (21 spp.), followed by Radulaceae (2 spp.) and the thallose liverwort family Metzgeriaceae (2 spp.). According to Gradstein et al. (2001) [17] and Ilkiu-Borges & Lisboa (2004) [57], species of Lejeuneaceae can colonize different types of substrates, but their occurrence on leaves is indicative of preserved native areas.
2.3. Endemism and Conservation Status
The Serra dos Órgaos National Park protects three species locally endemic, eight from Rio de Janeiro state, and 69 Brazilian (Table 2). Endemism and species richness are high with an increase in altitude, as was also found in Itatiaia National Park, located in Serra da Mantiqueira [18].

Table 2.
List of endemic bryophyte species found in PARNASO. (EB = endemic to Brazil; ERJ = endemic to Rio de Janeiro State; and CS = conservation status category.
Of the 28 species of Brazilian endemic bryophytes exclusive to high-altitude fields, 12 belong to the family Sphagnaceae (42%). The species Sphagnum luetzelburgii and S. sehnemii are endemic to Brazil, occurring in the states of Paraná, Santa Catarina, Rio Grande do Sul, and Rio de Janeiro. However, in Rio de Janeiro State, these species are restricted to mountains of the Atlantic Forest and have only been reported for PARNASO by Costa (2021) [58] and not by the present work.
The lower number of endemic species recorded in sub-montane forest probably reflects the effects of ecological and biogeographic processes, as well as human interference [18]. Among the 19 endemic species exclusively found in montane forest, 13 are liverworts and 6 are mosses. The predominant liverwort family is Lejeuneaceae, with nine species, followed by Metzgeriaceae with three and Jamesoniellaceae with two.
We have re-collected some species considered rare, including Bryum limbatum. Ochi (1980) [59] cites a collection from Metzgeria furcata, collected by Bartram (1954) [19]; Sphagnum cuspidatum, collected by Luetzelburg in 1923; and Andreaea acutifolia, collected by Mueller in 1845. Andreaea acutifolia is always mistakenly identified as A. rupestris, the two differing mainly by perichaetial leaves [24]. The species S. cuspidatum is widespread in Brazil [60], but has been collected only once in PARNASO.
The Serra dos Orgaos Park also protects eleven of the species indicated as threatened by CNCFlora and Flora e Funga do Brasil (Figure 2 and Table 2) in the Rio de Janeiro state: seven as Endangered (EN), two as Critically Endangered (CR) and two as Vulnerable (VU). Among these threatened species, three are known only from the type material, and were not found in the field trips. Southbya organensis was described by Herzog (1949) [61] and is classified as Critically Endangered (CR) by CNCFlora (2012) [58] due to its distribution being restricted to Serra dos Órgãos, specifically Morro do Açu peak, which has excessive tourism and recurrent arson instead. Plagiochila pseudoradicans and Dicranodontium pulchroalare ssp. brasiliense were described by Herzog (1949) [61], and neither was assessed by CNCFlora (2012) [62] because they were considered as Data Deficient (DD) at that time.

Figure 2.
Pictures of some species indicated as threatened in Serra dos Orgaos National Park. (A) Gongylanthus liebmannianus (Lindenb. & Gottsche) Steph., (B) Metzgeria hegewaldii Kuwah., (C) Plagiochila boryana Gottsche ex Steph., (D) Atractylocarpus longisetus (Hook.) E.B.Bartram, (E) Leptodontium wallisii (Müll.Hal.) Kindb., (F) Sphagnum perforatum Warnst.
3. Discussion
The Serra dos Órgãos National Park (PARNASO) exhibits remarkable bryophyte diversity. This richness highlights the park as a key conservation area for bryophytes in the Atlantic Forest. These findings suggest that even in areas already considered well-studied, significant knowledge gaps remain, highlighting the dynamic nature of bryophyte diversity and distribution.
One important factor contributing to this diversity is the presence of forested areas with abundant tree trunks, which serve as critical substrates for epiphytic bryophytes. The architectural, morphological, phenological, and chemical traits of different tree species create varied microhabitats that support a wide range of bryophyte taxa [57,63]. Shade-loving epiphytes, in particular, are commonly found in these forest environments but are highly sensitive to environmental disturbances. As such, they are often among the first species to disappear following canopy opening or habitat degradation [64], making them reliable indicators of forest health.
In contrast, mosses exhibit a broader ecological amplitude due to their desiccation tolerance and structurally complex adaptations, such as costa development, hyaline leaf apices, papillae, and specialized conducting tissues like hydroids and leptoids [29,65,66]. These features enable many mosses to colonize exposed and drier substrates, including rocky outcrops. Similar patterns of moss dominance on rock surfaces have been observed in other highland regions of Brazil, such as Chapada Diamantina [67,68], Serra do Caraça [69], and Serra da Canastra [70].
The high-altitude ecosystems of PARNASO further enhance bryophyte diversity by creating isolated and unique habitats. Mountain summits, often compared to ecological islands due to their environmental isolation, support specialized flora adapted to harsh conditions such as strong winds, low temperatures, and nutrient-poor soils [71,72,73]. These rupestrian fields form natural refugia, acting as barriers to typical forest species and favoring the emergence of narrow endemics and rare taxa [74,75,76].
Overall, the findings of this study underscore the ecological importance and conservation value of Serra dos Órgãos National Park. Alongside Itatiaia National Park, PARNASO remains one of the most critical remnants of Atlantic Forest in Rio de Janeiro and Brazil, particularly in terms of bryophyte conservation. The park’s diverse topography, wide range of microhabitats, and climatic heterogeneity—marked by high altitudes, low temperatures, and high light availability—create optimal conditions for bryophyte diversity. Furthermore, the presence of endemic and threatened species reinforces the park’s role as a conservation priority. Such studies are valuable as they reveal the significance of endemism and species richness, crucial factors in determining conservation priorities [77], and emphasize the importance of preserving Serra dos Órgãos National Park.
4. Materials and Methods
4.1. Study Area
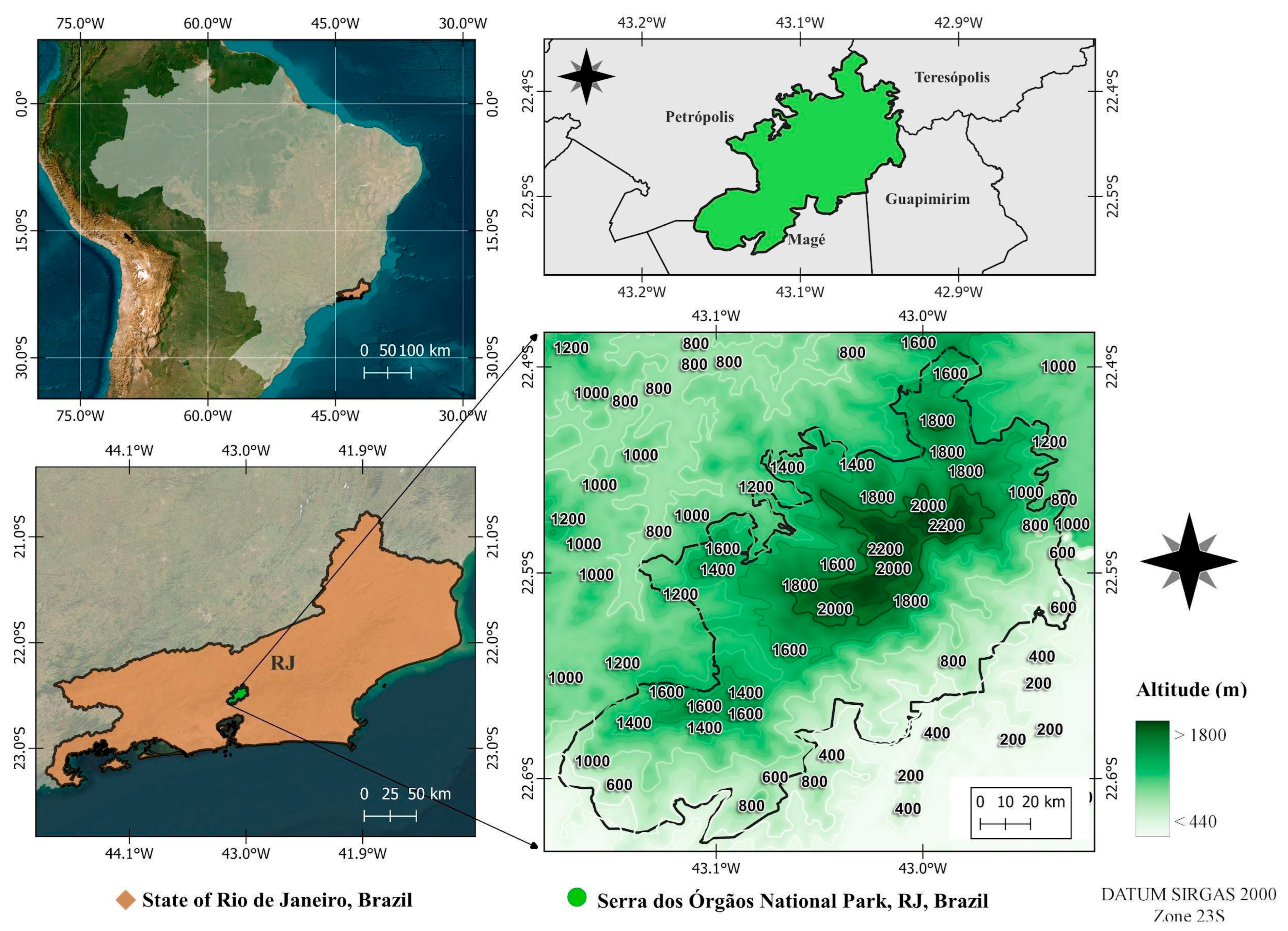
Serra dos Órgãos National Park (PARNASO) is located in the highlands region of Rio de Janeiro State, Brazil, ranging the municipalities of Teresópolis, Petrópolis, Guapimirim and Magé (22°23′36.96″–22°34′57.72″ S and 43°10′57.72″–42°58′43.68″ W). It is a protected area with 20,024 hectares of Atlantic Forest in a central position in the Serra do Mar Ecological Corridor. Ranging in altitude from 80 to 2275 m.a.s.l. (Figure 3), it is the highest portion of the entire Serra do Mar [2]. The climate is tropical and super humid (80–90% relative humidity), with the annual average temperature ranging from 13 to 23 °C, although minimums can reach −5 °C during winter above 800 m.a.s.l. [6,8,74]. The average annual rainfall ranges from 1500 mm to 3000 mm, with higher concentrations of rain during summer (December to March) and a dry season during winter (June to August), while heat is well distributed throughout the year [78,79].
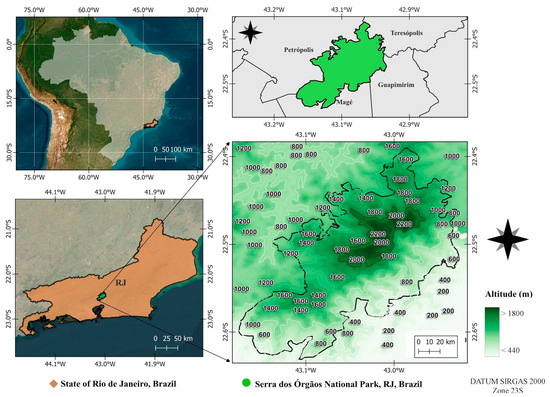
Figure 3.
Location of the Serra dos Órgãos National Park, Rio de Janeiro State, Brazil.
The vegetation classification adopted follows Veloso et al. (1991) [20] and Safford (1999) [80]: Sub-montane Forest (below 600 m.a.s.l., typically evergreen, with a high canopy and rich floristic diversity; dominant tree species include Protium spp., Virola spp., and members of the Myrtaceae and Fabaceae families); montane forest (600–1500, characterized by shorter trees and more frequent presence of cloud cover; common tree species include Clethra spp. and various species of Melastomataceae); upper-montane forest (above 1500, vegetation here is more stunted, often with gnarled trees adapted to frequent mist and lower temperatures). Dominant elements include Weinmannia paulliniifoli and Drimys brasiliensis, and high-altitude fields (above 1800–2000) are open grassland formations interspersed with shrubs, herbs, and occasional low woody elements. Common species include Vellozia spp., Paepalanthus spp., and Eryngium spp. (Figure 4).

Figure 4.
Collection sites in Serra dos Órgãos National Park. (A) Trail of the “Mirante”, (B) aspect of the montane forest with streams, (C) “Véu da Noiva” waterfall, (D) aspect of the upper-montane forest, (E) aspect of high-altitude fields, with Sphagnum among Chusquea sp.
4.2. Studied Taxa
The species list developed here is based on (i) a bibliographic survey conducted by consulting published volumes of major Brazilian botanical journals, as well as international bibliographic databases (Web of Science, SciELO, and Google Scholar), in search of peer-reviewed articles; the search focused on article titles containing combinations of the keywords “Serra dos Órgãos,” “bryophytes,” “mosses,” and “liverworts,” followed by a careful and detailed reading of each selected publication; (ii) herbarium specimens collected in the study area; and (iii) new collections (Figure 4).
Herbarium specimens were obtained from online sources like (GBIF [81], jabot, JStorTypes and SpeciesLink accessed several times). The specimens are requested on loan and re-determined and carefully examined for small species associations, with one of each species cited as voucher in the Table 1. The authors conducted four field excursions to the PARNASO, each lasting one week, two during the rainy season and two during the dry season, with the aim of locating species not previously recorded in the herbarium. The collection, preservation and herborization methodologies follow Frahm (2003) and employ free walks [82] for covering all available substrates like tree trunks, logs, soil, rocks and leaves.
Field trips in the Park intended to check if species collected in the past are yet present, to collect new specimens and to record new taxa. Collected samples were deposited in the herbarium of the University of Brasília (UB), with duplicates sent to the herbarium of Instituto de Pesquisas Ambientais (SP). The classification system used here follows Crandall-Stotler et al. [83] for Marchantiophyta, Goffinet et al. [65] for Bryophyta and Renzaglia et al. [84] for Anthocerotophyta. Data referring to endemic species of Brazil and of Rio de Janeiro State came from Flora e Funga do Brasil (floradobrasil.jbrj.gov.br), while conservation status followed CNCFlora (cncflora.jbrj.gov.br).
5. Conclusions
The Serra dos Órgãos National Park stands out as a key refuge for bryophyte diversity in the Atlantic Forest, hosting numerous endemic and threatened species. The park’s altitudinal variation, diverse microhabitats, and unique environmental conditions favor high species richness and endemism. These findings reinforce the ecological value of PARNASO and underscore the urgency of conserving this unique mountainous ecosystem.
Author Contributions
Methodology, M.T.A.G. and D.F.P.; Validation, D.F.P.; Investigation, J.S.d.L., A.L.A.F., M.T.A.G. and D.F.P.; Resources, D.F.P.; Data curation, J.S.d.L., A.L.A.F. and D.F.P.; Writing—original draft, A.L.A.F.; Writing—review & editing, J.S.d.L., M.T.A.G. and D.F.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Fundação de Amparo à Pesquisa do Distrito Federal (FAP-DF).
Data Availability Statement
The original contributions presented in this study are included in the article; further inquiries can be directed to the corresponding author.
Acknowledgments
We thank the Fundação de Amparo à Pesquisa do Distrito Federal (FAP-DF) for financial support to this project and the park coordinator PARNASO (Jorge Nascimento). We fully thank the support in field collections of the colleagues Amanda Leal, Anderson Eustáquio, Clapton Moura, Cosme Ponte and Flávio Rick.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviation
| PARNASO | Serra dos Órgãos National Park |
References
- Mello, D.; Mello, G.; Mallet-Rodrigues, F. Aves da Serra dos Órgãos e Adjacências: Guia de Campo (Birds of the Serra dos Órgãos and Surrounding Areas: Field Guide). Neotrop. Bird. 2015, 19, 87. [Google Scholar]
- Cronemberger, C.; Delciellos, A.C.; de Barros, C.S.; Gentile, R.; Weksler, M.; Braz, A.G.; Teixeira, B.R.; Loretto, D.; Vilar, E.M.; Pereira, F.A.; et al. Mamíferos do Parque Nacional da Serra dos Órgãos: Atualização da lista de espécies e implicações para a conservação. Oecologia Aust. 2019, 23, 191–214. [Google Scholar] [CrossRef]
- Mori, S.A.; Boom, B.M.; Prance, G.T. Distribution patterns and conservation of Eastern Brazilian coastal forest tree species. Brittonia 1981, 33, 233–245. [Google Scholar] [CrossRef]
- Lima, H.C.; Guedes-Bruni, R.R. Plantas arbóreas da Reserva Ecológica de Macaé de Cima. In Serra de Macaé de Cima: Diversidade Florística e Conservação em Mata Atlântica; Lima, H.C., Guedes-Bruni, R.R., Eds.; Jardim Botânico do Rio de Janeiro: Rio de Janeiro, Brazil, 1997; pp. 53–63. [Google Scholar]
- Guedes, M.L.; Batista, M.A.; Ramalho, M.; Freitas, H.M.B.; Silva, E.M. Breve incursão sobre a biodiversidade da Mata Atlântica. In Mata Atlântica e Biodiversidade; Franke, C.R., Rocha, P.L.B., Klein, W., Gomes, S.L., Eds.; Edufba: Salvador, Brazil, 2005; pp. 39–92. [Google Scholar]
- Faria, C.C.; de Castro, E.B.V. Envolvendo a Comunidade Científica na Gestão do Parque Nacional da Serra dos Órgãos. Biodiversidade Bras. 2015, 5, 4–20. [Google Scholar]
- Coelho, M.A.N.; Baumgratz, J.F.A.; Lobão, A.Q.; Sylvestre, L.S.; Trovó, M.; Silva, L.A.E. Flora of Rio de Janeiro state: An overview of Atlantic Forest diversity. Rodriguésia 2017, 68, 1–11. [Google Scholar] [CrossRef]
- de Carvalho-E-Silva, S.P.; de Carvalho-E-Silva, A.M.P.T.; Folly, M.; de Luna-Dias, C.; Bezerra, A.d.M.; Gomes, M.d.R.; Caram, J.; Peixoto, O.L.; Izecksohn, E. Parque Nacional da Serra dos Órgãos: The highest Amphibian diversity within an Atlantic Forest protected area. Biota Neotrop. 2020, 20, e20201033. [Google Scholar] [CrossRef]
- Primack, R.B. Essentials of Conservation Biology; Sinauer: Sunderland, UK, 1993; 608p. [Google Scholar]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Leigh, E.G.; Davidar, P.; Dick, C.W.; Terborgh, J.; Puyravaud, J.P.; ter Steege, H.; Wright, S.J. Why do some tropical forests have so many species of trees? Biotropica 2004, 36, 447–473. [Google Scholar]
- Corlett, R.T.; Primack, R.B. Tropical Rain Forests: An Ecological and Biogeographical Comparison; Wiley Blackwell: Oxford, UK, 2011. [Google Scholar]
- Pócs, T. Tropical Forest Bryophytes. In Bryophyte Ecology; Smith, A.J.E., Ed.; Chapman and Hall: New York, NY, USA, 1982; pp. 59–103. [Google Scholar]
- Gradstein, S.R. Bryophyte diversity of the tropical rainforest. Arch. Des Sci. 1995, 48, 91–96. [Google Scholar]
- Frahm, J.P.; Gradstein, S.R. An altitudinal zonation of tropical rain forests using bryophytes. J. Biogeogr. 1991, 18, 669–678. [Google Scholar] [CrossRef]
- Pócs, T. The altitudinal distribution of Kilimanjaro bryophytes. In Proceedings of the XIIIth Plenary Meeting AETFAT, Zomba, Malawi, 2 April 1994; Seyani, J.H., Chikuni, A.C., Eds.; pp. 797–812. [Google Scholar]
- Gradstein, S.R.; Churchill, S.P.; Salazar, A.N. Guide to the Bryophytes of Tropical America; Memoirs of The New York Botanical Garden: New York, NY, USA, 2001; Volume 86, pp. 1–577. [Google Scholar]
- Costa, D.P.; Santos, N.D.; Rezende, M.A.; Buck, W.R.; Schäfer-Verwimp, A. Bryoflora of the Itatiaia National Park along an elevation gradient: Diversity and conservation. Biodivers. Conserv. 2015, 24, 2199–2212. [Google Scholar] [CrossRef]
- Bartram, E.B. Musci. In Flora Organensis. Lista Preliminar dos Cormophyta da Serra dos Órgãos; Rizzini, C.T., Ed.; Archivos do Jardim Botânico do Rio de Janeiro: Rio de Janeiro, Brazil, 1954; Volume 13, pp. 116–243. [Google Scholar]
- Veloso, H.P.; Rangel-Filho, A.L.R.; Lima, J.C.A. Classificação da Vegetação Brasileira, Adaptada a um Sistema Universal; IBGE: Rio de Janeiro, Brazil, 1991; 124p.
- Amador, E.S. Baía de Guanabara e Ecossistemas Periféricos: Homem e Natureza; Scientific Research: Rio de Janeiro, Brazil, 1997; 539p. [Google Scholar]
- Aguiar, A.P.; Chiarello, A.G.; Mendes, S.L.; Matos, E.N. Os corredores Central e da Serra do Mar na Mata Atlântica Brasileira. In Mata Atlântica: Biodiversidade, Ameaças e Perspectivas; Galindo-Leal, C., Câmara, I.G., Eds.; S.O.S. Mata Atlântica e Conservação Internacional e Centro de Ciências Aplicadas à Biodiversidade: Belo Horizonte, Brazil, 2005; pp. 119–132. [Google Scholar]
- Faria, A.L.A.; Peralta, D.F.; Amorim, E.T.; Carvalho-Silva, M.; Câmara, P.E.A.S. Bryophytes from Glaziou’s historical collections: Revisiting the flora of Rio de Janeiro and implications for conservation. Cryptogam. Bryol. 2021, 42, 197–204. [Google Scholar]
- Peralta, D.F. The genus Andreaea Hedw. (Andreaceae, Bryophyta) in Brazil. Phytotaxa 2020, 451, 169–174. [Google Scholar] [CrossRef]
- Winter, G.; Schäfer-Verwimp, A. Re-evaluation of the taxonomic status of Frullania caulisequa and Frullania obcordata (Frullaniaceae, Marchantiophyta). Frahmia 2020, 19, 1–21. [Google Scholar]
- Gottsche, C.M.; Lindenberg, J.B.W.; Nees von Esenbeck, C.G.D. Synopsis Hepaticarum; Meissner: Hamburg, Germany, 1844; 834p. [Google Scholar]
- Fulford, M.H. Manual of the leafy Hepaticae of Latin America. Part I. In Memoirs of The New York Botanical Garden; The New York Botanical Garden: New York, NY, USA, 1963; Volume 11, pp. 1–172. [Google Scholar]
- Grolle, R. Miscellanea hepaticologicae (11–20). J. Jpn. Bot. 1964, 39, 173–178. [Google Scholar]
- Stotler, R.E.; Crandall-Stotler, B.J. A Monograph of the Genus Bryopteris (Swartz) Nees von Esenbeck; Bryophytorum Bibliotheca: Berlin, Germany, 1974; Volume 3, 159p. [Google Scholar]
- Jovet-Ast, S. Le genre Colura. Hépatiques, Lejeuneaceae, Diplasieae. Rev. Bryol. Lichénologique 1953, 22, 206–312. [Google Scholar]
- Gradstein, S.R.; Costa, D.P. The Hepaticae and Anthocerotae of Brazil; Memoirs of the New York Botanical Garden: New York, NY, USA, 2003; Volume 87, pp. 1–336. [Google Scholar]
- Bischler, H. Le genre Drepanolejeunea Steph. en Amérique Centrale et Méridionale. Rev. Bryol. Lichénologique 1964, 33, 15–179. [Google Scholar]
- Schäfer-Verwimp, A.; VitaL, D.M. New or interesting records of Brazilian bryophytes. J. Hattori Bot. Lab. 1989, 66, 255–261. [Google Scholar]
- Nees von Esenbeck, C.G.D. Beitrage zur Naturgeschichte der deutschen Lebermoose (Schluss). Flora 1833, 16, 401–412. [Google Scholar]
- Raddi, G. Crittogamie Brasiliene; Dalla Società Tipografica: Modena, Italy, 1823; 33p. [Google Scholar]
- Costa, D.P. Metzgeriaceae (Hepaticae). Flora Neotrop. Monogr. 2008, 102, 1–169. [Google Scholar]
- Costa, D.P.; Imbassahy, C.A.D.; Silva, V.P.A.V. Checklist and distribution of mosses, liverworts and hornworts of Rio de Janeiro state, Brazil. J. Hattori Bot. Lab. 2005, 98, 259–298. [Google Scholar]
- Heinrichs, J. New synonyms in Plagiochila (Hepaticae) II. Cryptogam. Bryol. 2002, 23, 351–353. [Google Scholar]
- Heinrichs, J.; Gradstein, S.R. A revision of Plagiochila sect. Crispatae and sect. Hypnoides (Hepaticae) in the Neotropics. I. Plagiochila disticha, P. montagnei and P. raddiana. Nova Hedwig. 2000, 70, 161–184. [Google Scholar] [CrossRef]
- Brotherus, V.F. Musci. In Die natürlichen Pflanzenfamilien; Engler, A., Prantl, K., Eds.; W. Engelmann: Leipzig, Germany, 1924; Volume 2, p. 11. [Google Scholar]
- Allen, B.H.; Crosby, M.R. Revision of the genus Squamidium (Musci: Meteoriaceae). J. Hattori Bot. Lab. 1986, 61, 423–476. [Google Scholar]
- Manuel, M.G. The genus Meteoridium (C. Müll.) Manuel, stat. Nov. (Bryopsida: Meteoriaceae). Lindbergia 1977, 4, 45–55. [Google Scholar]
- Brotherus, V.F. Musci (Laubmoose). In Ergebnisse der Botanischen Expedition der Kaiserlichen Akademie der Wissenschaften Nach Südbrasilien 1901, 1924b, Band II (Thallophyta und Bryophyta); Springer: Wien, Austria, 1924; Volume 4, pp. 45–55. [Google Scholar]
- Brotherus, V.F. Musci Schenckiani. Ein Beitrag zur Kenntniss der Moosflora Brasiliens. Hedwigia 1894, 33, 127–136. [Google Scholar]
- Hampe, E. Enumeratio muscorum frondosorum Brasiliae centralis, praecipue provinciarum Rio de Janeiro et S. Paulo, adhuc cognitorum. Vidensk. Meddel. Naturhist. Foren. Kjøbenhavn. Ser. 1879, 1, 73–164. [Google Scholar]
- Bartram, E.B. New mosses from southern Brazil. J. Wash. Acad. Sci. 1952, 42, 178–182. [Google Scholar]
- Luetzelburg, P. Estudo Botanico do Nordéste; Inspectoria Federal de Obras Contra as Seccas: Rio de Janeiro, Brazil, 1923; Volume 1, p. 126.
- Vattimo-Gil, I.; Vattimo, I. Bryophyta (Musci) do herbário do Jardim Botânico do Rio de Janeiro—II. Rodriguésia 1980, 32, 257–267. [Google Scholar] [CrossRef]
- Müller, C. Nachtragliche Bemerkungen über die von Gardner in Brasilien gesammelten Laubmoose. Bot. Ztg. 1845, 3, 89–94. [Google Scholar]
- Frahm, J.P. Taxonomische Notizen zur Gattung Campylopus. Rev. Bryol. Lichénologie 1975, 41, 321–332. [Google Scholar]
- Herzog, T.H. Geographie der Moose; Gustav Fischer: Jena, Germany, 1926; 439p. [Google Scholar]
- Li, D.D.; Guo, S.L.; Fang, Y.M. Taxonomic notes on Macromitrium catharinense (Bryopsida, Orthotrichaceae) with its new synonyms. Nord. J. Bot. 2019, 37, e02320. [Google Scholar] [CrossRef]
- Bednarek-Ochyra, H.; Ochyra, R. Bucklandiella allanfifei (Grimmiaceae), a new moss species from New Zealand, with a note on South American B. striatipila. J. Bryol. 2010, 32, 245–255. [Google Scholar] [CrossRef]
- Buck, W.R. New combinations and new synonymy in Brazilian Sematophyllaceae. Nova Hedwig. 1998, 66, 241–246. [Google Scholar] [CrossRef]
- Andrews, A.L. Notes on the Warnstorf Sphagnum herbarium III. The subgenus Inophloea in South America. Bryologist 1941, 44, 155–160. [Google Scholar] [CrossRef]
- Hooker, W.J.; Wilson, W. Enumeration of the mosses and Hepaticae, collected in Brazil by George Gardner, Esq. Lond. J. Bot. 1844, 3, 149–167. [Google Scholar]
- Ilkiu-Borges, A.L.; Lisboa, R.C.L. Cololejeuneae (Lejeuneaceae, Hepaticae) na Estação Científica Ferreira Penna, Melgaço, PA, Brasil. Acta Bot. Bras. 2004, 18, 887–902. [Google Scholar] [CrossRef]
- de Costa, D.P. A synopsis of the family Sphagnaceae in Brazil. Syst. Bot. Monogr. 2021, 111, 1–142. [Google Scholar]
- Ochi, H. A revision of the neotropical Bryoideae, Musci (First part). J. Fac. Educ. Tottori Univ. Nat. Sci. 1980, 29, 49–154. [Google Scholar]
- Flora e Funga do Brasil. Jardim Botânico do Rio de Janeiro. Available online: https://floradobrasil.jbrj.gov.br/ (accessed on 30 July 2023).
- Herzog, T.H. Miscellanea Bryologica. I. Neotropica. Memo. Soc. Pro Fauna Et Flora Fenn. 1949, 25, 53–54. [Google Scholar]
- CNCFlora. Southbya organensis. In Lista Vermelha da Flora Brasileira Versão 2012; Centro Nacional de Conservação da Flora: Washington, DC, USA, 2012. Available online: https://cncflora.jbrj.gov.br/ficha/97186 (accessed on 27 July 2023).
- Zotz, G.; Vollrath, B. The epiphyte vegetation of the palm Socratea exorrhiza-correlations with tree size, tree age and bryophyte cover. J. Trop. Ecol. 2003, 19, 81–90. [Google Scholar] [CrossRef]
- Gradstein, S.R. The taxonomic diversity of epiphyllous bryophytes. Abstr. Bot. 1997, 21, 15–19. [Google Scholar]
- Goffinet, B.; Buck, W.R.; Shaw, A.J. Morphology, anatomy and classification of the Bryophyta. In Bryophyte Biology, 2nd ed.; Goffinet, B., Shaw, A.J., Eds.; Cambridge University Press: New York, NY, USA, 2009; pp. 56–138. [Google Scholar]
- Gao, B.; Li, X.; Zhang, D.; Liang, Y.; Yang, H.; Chen, M.; Zhang, Y.; Zhang, J.; Wood, A.J. Desiccation tolerance in bryophytes: The dehydration and rehydration transcriptomes in the desiccation-tolerant bryophyte Bryum argenteum. Sci. Rep. 2017, 7, 7571. [Google Scholar] [CrossRef] [PubMed]
- Bastos, C.J.P.; Yano, O.; Vilas Bôas-Bastos, S.B. Briófitas de campos rupestres da Chapada Diamantina, estado da Bahia, Brasil. Rev. Bras. Botânica 2000, 23, 357–368. [Google Scholar] [CrossRef]
- Valente, E.B.; Pôrto, P.K.C.; Bastos, C.J.P. Habitat heterogeneity and diversity of bryophytes in campos rupestres. Acta Bot. Bras. 2017, 31, 241–249. [Google Scholar] [CrossRef]
- Carmo, D.M.; Lima, J.S.; Silva, M.I.; Amélio, L.A.; Peralta, D.F. Briófitas da Reserva Particular do Patrimônio Natural da Serra do Caraça, Estado de Minas Gerais, Brasil. Hoehnea 2018, 45, 484–508. [Google Scholar] [CrossRef]
- Carmo, D.M.; Peralta, D.F. Survey of bryophytes in Serra da Canastra National Park, Minas Gerais, Brazil. Acta Bot. Bras. 2016, 30, 254–265. [Google Scholar] [CrossRef]
- Schuster, R.M. Phytogeography of bryophytes. In New Manual of Bryophytes; Schuster, R.M., Ed.; Hattori Botanical Laboratory: Nichinan, Japan, 1983; pp. 463–626. [Google Scholar]
- Gotelli, N.J. Island Biogeography. In A Primer of Ecology, 4th ed.; Gotelli, N.J., Ed.; Sinauer Associates: Boston, MA, USA, 2009. [Google Scholar]
- Lomolino, M.V.; Riddle, B.R.; Whittaker, R.J. Biogeography: Biological Diversity Across Space and Time, 5th ed.; Sinauer Associates: Oxford, UK, 2016. [Google Scholar]
- Ribeiro, K.T.; Medina, B.M.O.; Scarano, F.R. Species Composition and Biogeographic Relations of the Rock Outcrop Flora on the High Plateau of Itatiaia, SE-Brazil. Braz. J. Bot. 2007, 30, 623–639. [Google Scholar] [CrossRef]
- Costa, D.P.; Couto, G.P.; Siqueira, M.F.; Churchill, S.P. Bryofloritic affinities between Itatiaia National Park and tropical Andean countries. Phytotaxa 2018, 346, 203–220. [Google Scholar] [CrossRef]
- Santos, N.D.; Oliveira, J.R.P.M.; Silva, L.T.P.; Gonçalves, M.T.A.; Peralta, D.F. Campos de altitude: Ecossistemas prioritários para a conservação de briófitas. In Botânica: Para Que e Para Quem? Desafios, Avanços e Perspectivas na Sociedade Contemporânea; Moura, C.W.N., Shimizu, G.H., Eds.; Sociedade Botânica do Brasil: Brasília-DF, Brazil, 2022; pp. 92–99. [Google Scholar]
- Kier, G.; Kreft, H.; Lee, T.M.; Jetz, W.; Ibisch, P.L.; Nowicki, C.; Mutke, J.; Barthlott, W. A global assessment of endemism and species richness across island and mainland regions. Proc. Natl. Acad. Sci. USA 2009, 106, 9322–9327. [Google Scholar] [CrossRef]
- Fiderj. Indicadores Climatológicos do Estado do Rio de Janeiro; Fundação de Desenvolvimento Econômico e Social do Rio de Janeiro: Rio de Janeiro, Brazil, 1978; 156p. [Google Scholar]
- Davis, E.G.; Naghettini, M.C. Estudos de Chuvas Intensas no Estado do Rio de Janeiro, 2nd ed.; CPRM: Brasília, Brazil, 2000; 142p. [Google Scholar]
- Safford, H.D. Brazilian páramos I. An introduction to the physical environment and vegetation of the campos de altitude. J. Biogeogr. 1999, 26, 693–712. [Google Scholar] [CrossRef]
- GBIF.org. GBIF Occurrence. Available online: https://doi.org/10.15468/dl.4ysrw3 (accessed on 11 June 2025).
- Frahm, J.P. Manual of tropical bryology. Trop. Bryol. 2003, 23, 1–195. [Google Scholar]
- Crandall-Stotler, B.J.; Stotler, R.E.; Long, D.G. Phylogeny and classification of the Marchantiophyta. Edinb. J. Bot. 2009, 66, 155–198. [Google Scholar] [CrossRef]
- Renzaglia, K.S.; Villarreal, J.C.; Duff, R.J. New insights into morphology, anatomy, and systematics of hornworts. In Bryophyte Biology, 2nd ed.; Goffinet, B., Shaw, A.J., Eds.; Cambridge University Press: Cambridge, UK, 2009; 565p. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).