Ascorbic Acid and Ozone: Novel Perspectives to Explain an Elusive Relationship
Abstract
:1. Introduction
2. Is AA Content Always Related to Ozone Tolerance?
- Exogenous AA induces protection against ozone injury.
- Light pre-treatment increases AA content and improves ozone tolerance.
- Young tissues generally have more AA and are less affected by ozone.
3. What Do We Talk About, When We Talk About “Total Ascorbic Acid”?
4. AA and ROS Scavenging in Relation to Ozone Tolerance/Sensitivity
5. The Role of Ascorbate in the Signal Transduction Pathway
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heggestad, H.E.; Middleton, J.T. Ozone in high concentrations as cause of tobacco leaf injury. Science 1959, 129, 208–210. [Google Scholar] [CrossRef] [PubMed]
- Freebairn, H.T. The Prevention of air pollution damage to plants by the use of vitamin C sprays. J. Air Pollut. Control Assoc. 1960, 10, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Mächler, F.; Wasescha, M.R.; Krieg, F.; Oertli, J.J. Damage by ozone and protection by ascorbic acid in barley leaves. J. Plant Physiol. 1995, 147, 469–473. [Google Scholar] [CrossRef]
- Maddison, J.; Lyons, T.; Plöchl, M.; Barnes, J. Hydroponically cultivated radish fed L-galactono-1,4-lactone exhibit increased tolerance to ozone. Planta 2002, 214, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Feng, Z.; Dai, L.; Shang, B.; Paoletti, E. Large variability in ambient ozone sensitivity across 19 ethylenediurea-treated Chinese cultivars of soybean is driven by total ascorbate. J. Environ. Sci. 2018, 64, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Rakwal, R.; Agrawal, S.B.; Shibato, J.; Ogawa, Y.; Yoshida, Y.; Agrawal, G.K.; Agrawal, M. Investigating the impact of elevated levels of ozone on tropical wheat using integrated phenotypical, physiological, biochemical, and proteomics approaches. J. Proteome Res. 2010, 9, 4565–4584. [Google Scholar] [CrossRef]
- Cho, K.; Tiwari, S.; Agrawal, S.B.; Torres, N.L.; Agrawal, M.; Sarkar, A.; Shibato, J.; Agrawal, G.K.; Kubo, A.; Rakwal, R. Tropospheric ozone and plants: Absorption, responses and consequences. In Reviews of Environmental Contamination and Toxicology; Whitacre, D., Ed.; Springer: New York, NY, USA, 2011; Volume 212, pp. 61–111. [Google Scholar]
- Rai, R.; Agrawal, M. Impact of tropospheric ozone on crop plants. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2012, 82, 241–257. [Google Scholar] [CrossRef]
- Vainonen, J.P.; Kangasjärvi, J. Plant signalling in acute ozone exposure. Plant Cell Environ. 2015, 38, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Hayes, F.; Jones, M.L.M.; Mills, G.; Ashmore, M. Meta-analysis of the relative sensitivity of semi-natural vegetation species to ozone. Environ. Pollut. 2007, 146, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Agathokleous, E.; Saitanis, C.J.; Koike, T. Tropospheric O3, the nightmare of wild plants: A review study. J. Agric. Meteorol. 2015, 71, 142–152. [Google Scholar] [CrossRef]
- Frei, M. Breeding of ozone resistant rice: Relevance, approaches and challenges. Environ. Pollut. 2015, 197, 144–155. [Google Scholar] [CrossRef]
- Ainsworth, E.A. Understanding and improving global crop response to ozone pollution. Plant J. 2017, 90, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Dumont, J.; Keski-Saari, S.; Keinänen, M.; Cohen, D.; Ningre, N.; Kontunen-Soppela, S.; Baldet, P.; Gibon, Y.; Dizengremel, P.; Vaultier, M.N.; et al. Ozone affects ascorbate and glutathione biosynthesis as well as amino acid contents in three Euramerican poplar genotypes. Tree Physiol. 2014, 34, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Höller, S.; Ueda, Y.; Wu, L.; Wang, Y.; Hajirezaei, M.-R.; Ghaffari, M.-R.; von Wirén, N.; Frei, M. Ascorbate biosynthesis and its involvement in stress tolerance and plant development in rice (Oryza sativa L.). Plant Mol. Biol. 2015, 88, 545–560. [Google Scholar] [CrossRef]
- Barnes, J.; Zheng, Y.; Lyons, T. Plant tolerance to ozone: The role of ascorbate. In Air Pollution and Plant Biotechnology; Omasa, K., Saji, H., Youssefian, S., Kondo, N., Eds.; Springer: Tokyo, Japan, 2002; pp. 235–252. ISBN 978-4-431-68390-2. [Google Scholar]
- Conklin, P.L.; Pallanca, J.E.; Last, R.L.; Smirnoff, N. L-Ascorbic acid metabolism in the ascorbate-deficient Arabidopsis mutant vtc1. Plant Physiol. 1997, 115, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Conklin, P.L.; Williams, E.H.; Last, R.L. Environmental stress sensitivity of an ascorbic acid-deficient Arabidopsis mutant. Proc. Natl. Acad. Sci. USA 1996, 93, 9970–9974. [Google Scholar] [CrossRef] [PubMed]
- Conklin, P.L.; Norris, S.R.; Wheeler, G.L.; Williams, E.H.; Smirnoff, N.; Last, R.L. Genetic evidence for the role of GDP-mannose in plant ascorbic acid (vitamin C) biosynthesis. Proc. Natl. Acad. Sci. USA 1999, 96, 4198–4203. [Google Scholar] [CrossRef] [PubMed]
- Menser, H.A. Response of Plants to Air Pollutants. III. A relation between ascorbic acid levels and ozone susceptibility of light-preconditioned tobacco plants. Plant Physiol. 1964, 39, 564–567. [Google Scholar] [CrossRef]
- Heggestad, H.E. Origin of Bel-W3, Bel-C and Bel-B tobacco varieties and their use as indicators of ozone. Environ. Pollut. 1991, 74, 264–291. [Google Scholar] [CrossRef]
- Robinson, J.M.; Britz, S.J. Tolerance of a field grown soybean cultivar to elevated ozone level is concurrent with higher leaflet ascorbic acid level, higher ascorbate-dehydroascorbate redox status, and long term photosynthetic productivity. Photosynth. Res. 2000, 64, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Gallie, D.R. Increasing tolerance to ozone by elevating foliar ascorbic acid confers greater protection against ozone than increasing avoidance. Plant Physiol. 2005, 138, 1673–1689. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Lyons, T.; Ollerenshaw, J.H.; Barnes, J.D. Ascorbate in the leaf apoplast is a factor mediating ozone tolerance in Plantago major. Plant Physiol. Biochem. 2000, 38, 403–411. [Google Scholar] [CrossRef]
- Burkey, K.O. Effects of ozone on apoplast/cytoplasm partitioning of ascorbic acid in snap bean. Physiol. Plant. 1999, 107, 188–193. [Google Scholar] [CrossRef]
- Burkey, K.O.; Eason, G. Ozone tolerance in snap bean is associated with elevated ascorbic acid in the leaf apoplast. Physiol. Plant. 2002, 114, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Moldau, H.; Bichele, I.; Hüve, K. Dark-induced ascorbate deficiency in leaf cell walls increases plasmalemma injury under ozone. Planta 1998, 207, 60–66. [Google Scholar] [CrossRef]
- Robinson, J.M.; Britz, S.J. Ascorbate-dehydroascorbate level and redox status in leaflets of field-grown soybeans exposed to elevated ozone. Int. J. Plant Sci. 2001, 162, 119–125. [Google Scholar] [CrossRef]
- Turcsányi, E.; Lyons, T.; Plöchl, M.; Barnes, J. Does ascorbate in the mesophyll cell walls form the first line of defence against ozone? Testing the concept using broad bean (Vicia faba L.). J. Exp. Bot. 2000, 51, 901–910. [Google Scholar] [CrossRef]
- Castillo, F.J.; Greppin, H. Extracellular ascorbic acid and enzyme activities related to ascorbic acid metabolism in Sedum album L. leaves after ozone exposure. Environ. Exp. Bot. 1988, 28, 231–238. [Google Scholar] [CrossRef]
- Sanmartin, M.; Drogoudi, P.D.; Lyons, T.; Pateraki, I.; Barnes, J.; Kanellis, A.K. Over-expression of ascorbate oxidase in the apoplast of transgenic tobacco results in altered ascorbate and glutathione redox states and increased sensitivity to ozone. Planta 2003, 216, 918–928. [Google Scholar] [PubMed]
- Conklin, P.L.; Saracco, S.A.; Norris, S.R.; Last, R.L. Identification of ascorbic acid-deficient Arabidopsis thaliana mutants. Genetics 2000, 154, 847–858. [Google Scholar]
- Buettner, G.R.; Schafer, F.Q. Ascorbate as an antioxidant. In Vitamin C—Functions and Biochemistry in Animals and Plants; Asard, H., May, J.M., Smirnoff, N., Eds.; Bios Scientific Publishers: London, UK; New York, NY, USA, 2004; pp. 173–188. ISBN 1-85996-293-9. [Google Scholar]
- Polle, A. Dissecting the superoxide dismutase-ascorbate-glutathione-pathway in chloroplasts by metabolic modeling. Computer simulations as a step towards flux analysis. Plant Physiol. 2001, 126, 445–462. [Google Scholar] [CrossRef] [PubMed]
- De Tullio, M.C.; De Gara, L.; Paciolla, C.; Arrigoni, O. Dehydroascorbate-reducing proteins in maize are induced by the ascorbate biosynthesis inhibitor lycorine. Plant Physiol. Biochem. 1998, 36, 433–440. [Google Scholar] [CrossRef]
- Kollist, H.; Moldau, H.; Mortensen, L.; Rasmussen, S.K.; Jørgensen, L.B. Ozone flux to plasmalemma in barley and wheat is controlled by stomata rather than by direct reaction of ozone with cell wall ascorbate. J. Plant Physiol. 2000, 156, 645–651. [Google Scholar] [CrossRef]
- Kangasjӟrvi, J.; Jaspers, P.; Kollist, H. Signalling and cell death in ozone-exposed plants. Plant Cell Environ. 2005, 28, 1021–1036. [Google Scholar]
- Laisk, A.; Kull, O.; Moldau, H. Ozone concentration in leaf intercellular air spaces is close to zero. Plant Physiol. 1989, 90, 1163–1167. [Google Scholar] [CrossRef]
- Kangasjärvi, J.; Talvinen, J.; Utriainen, M.; Karjalainen, R. Plant defence systems induced by ozone. Plant Cell Environ. 1994, 17, 783–794. [Google Scholar]
- Lamb, C.; Dixon, R.A. The oxidative burst in plant disease tolerance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 251–275. [Google Scholar] [CrossRef]
- Mahalingam, R.; Fedoroff, N. Stress response, cell death and signalling: The many faces of reactive oxygen species. Physiol. Plant. 2003, 119, 56–68. [Google Scholar] [CrossRef]
- Gadjev, I.; Stone, J.M.; Gechev, T.S. Programmed cell death in plants: New insights into redox regulation and the role of hydrogen peroxide. In International Review of Cell and Molecular Biology; Academic Press: Waltham, MA, USA, 2008; Volume 270, pp. 87–144. [Google Scholar]
- Wang, P.; Song, C.-P. Guard-cell signalling for hydrogen peroxide and abscisic acid. New Phytol. 2008, 178, 703–718. [Google Scholar] [CrossRef]
- Chang, C.; Damiani, I.; Puppo, A.; Frendo, P. Redox changes during the legume–Rhizobium symbiosis. Mol. Plant 2009, 2, 370–377. [Google Scholar] [CrossRef]
- Miller, G.; Schlauch, K.; Tam, R.; Cortes, D.; Torres, M.A.; Shulaev, V.; Dangl, J.L.; Mittler, R. The plant NADPH oxidase RBOHD mediates rapid systemic signaling in response to diverse stimuli. Sci. Signal 2009, 2, ra45. [Google Scholar] [CrossRef]
- Jaspers, P.; Kangasjärvi, J. Reactive oxygen species in abiotic stress signaling. Physiol. Plant. 2010, 138, 405–413. [Google Scholar] [CrossRef]
- Swanson, S.; Gilroy, S. ROS in plant development. Physiol. Plant. 2010, 138, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Noctor, G.; Foyer, C.H. ASCORBATE AND GLUTATHIONE: Keeping active oxygen under control. Annu. Rev. Plant Phys. Plant Mol. Biol. 1998, 49, 249–279. [Google Scholar] [CrossRef]
- Shigeoka, S.; Ishikawa, T.; Tamoi, M.; Miyagawa, Y.; Takeda, T.; Yabuta, Y.; Yoshimura, K. Regulation and function of ascorbate peroxidase isoenzymes. J. Exp. Bot. 2002, 53, 1305–1319. [Google Scholar] [CrossRef]
- Willekens, H.; Camp, W.V.; Montagu, M.V.; Inze, D.; Langebartels, C.; Sandermann, H., Jr. Ozone, sulfur dioxide, and ultraviolet B have similar effects on mRNA accumulation of antioxidant genes in Nicotiana plumbaginifolia L. Plant Physiol. 1994, 106, 1007–1014. [Google Scholar] [CrossRef]
- Conklin, P.L.; Last, R.L. Differential accumulation of antioxidant mRNAs in Arabidopsis thaliana exposed to ozone. Plant Physiol. 1995, 109, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Kubo, A.; Saji, H.; Tanaka, K.; Kondo, N. Expression of Arabidopsis cytosolic ascorbate peroxidase gene in response to ozone or sulfur dioxide. Plant Mol. Biol. 1995, 29, 479–489. [Google Scholar] [CrossRef]
- Örvar, B.L.; McPherson, J.; Ellis, B.E. Pre-activating wounding response in tobacco prior to high-level ozone exposure prevents necrotic injury. Plant J. 1997, 11, 203–212. [Google Scholar] [CrossRef]
- Örvar, B.L.; Ellis, B.E. Transgenic tobacco plants expressing antisense RNA for cytosolic ascorbate peroxidase show increased susceptibility to ozone injury. Plant J. 1997, 11, 1297–1305. [Google Scholar] [CrossRef]
- Torsethaugen, G.; Pitcher, L.H.; Zilinskas, B.A.; Pell, E.J. Overproduction of ascorbate peroxidase in the tobacco chloroplast does not provide protection against ozone. Plant Physiol. 1997, 114, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Luwe, M.; Takahama, U.; Heber, U. Role of ascorbate in detoxifying ozone in the apoplast of spinach (Spinacia oleracea L.) leaves. Plant Physiol. 1993, 101, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Polle, A.; Wieser, G.; Havranek, W.M. Quantification of ozone influx and apoplastic ascorbate content in needles of Norway spruce trees (Picea abies L., Karst) at high altitude. Plant Cell Environ. 1995, 18, 681–688. [Google Scholar] [CrossRef]
- Ranieri, A.; D’Urso, G.; Nali, C.; Lorenzini, G.; Soldatini, G.F. Ozone stimulates apoplastic antioxidant systems in pumpkin leaves. Physiol. Plant. 1996, 97, 381–387. [Google Scholar] [CrossRef]
- Plöchl, M.; Lyons, T.; Ollerenshaw, J.; Barnes, J. Simulating ozone detoxification in the leaf apoplast through the direct reaction with ascorbate. Planta 2000, 210, 454–467. [Google Scholar] [CrossRef] [PubMed]
- Booker, F.L.; Burkey, K.O.; Jones, A.M. Re-evaluating the role of ascorbic acid and phenolic glycosides in ozone scavenging in the leaf apoplast of Arabidopsis thaliana L.: Ozone effects on leaf apoplast chemistry. Plant Cell Environ. 2012, 35, 1456–1466. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.-Y.; Burkey, K.O.; Robinson, J.M.; Booker, F.L. Leaf extracellular ascorbate in relation to O3 tolerance of two soybean cultivars. Environ. Pollut. 2007, 150, 355–362. [Google Scholar] [CrossRef] [PubMed]
- D’Haese, D.; Vandermeiren, K.; Asard, H.; Horemans, N. Other factors than apoplastic ascorbate contribute to the differential ozone tolerance of two clones of Trifolium repens L. Plant Cell Environ. 2005, 28, 623–632. [Google Scholar]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- De Tullio, M.C. Beyond the antioxidant: The double life of Vitamin C. Subcell. Biochem. 2012, 56, 49–65. [Google Scholar] [CrossRef]
- Smirnoff, N. Ascorbic acid metabolism and functions: A comparison of plants and mammals. Free Radic. Biol. Med. 2018, 122, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Saga, G.; Giorgetti, A.; Fufezan, C.; Giacometti, G.M.; Bassi, R.; Morosinotto, T. Mutation analysis of violaxanthin de-epoxidase identifies substrate-binding sites and residues involved in catalysis. J. Biol. Chem. 2010, 285, 23763–23770. [Google Scholar] [CrossRef] [PubMed]
- Shikita, M.; Fahey, J.W.; Golden, T.R.; Holtzclaw, W.D.; Talalay, P. An unusual case of ‘uncompetitive activation’ by ascorbic acid: Purification and kinetic properties of a myrosinase from Raphanus sativus seedlings. Biochem. J. 1999, 341, 725–732. [Google Scholar] [CrossRef]
- Dilley, D.R.; Kuai, J.; Poneleit, L.; Zhu, Y.; Pekker, Y.; Wilson, I.D.; Burmeister, D.M.; Gran, C.; Bowers, A. Purification and characterization of ACC oxidase and Its expression during ripening in apple fruit. In Cellular and Molecular Aspects of the Plant Hormone Ethylene; Current Plant Science and Biotechnology in Agriculture; Pech, J.C., Latché, A., Balagué, C., Eds.; Springer: Dordrecht, The Netherlands, 1993; Volume 16. [Google Scholar] [CrossRef]
- Hedden, P. The oxidases of gibberellin biosynthesis: Their function and metabolism. Physiol. Plant. 1997, 101, 709–719. [Google Scholar] [CrossRef]
- Mullineaux, P.; Exposito-Rodriguez, M.; Laissue, P.P.; Smirnoff, N. ROS-dependent signalling pathways in in plants and algae exposed to high light: Comparison with other eukaryotes. Free Radic. Biol. Med. 2018, 122, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Mattson, M.P. How does hormesis impact biology, toxicology, and medicine? NPJ Aging Mech. Disease 2017, 3, 13. [Google Scholar] [CrossRef]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. Hormesis, a compelling platform for sophisticated plant science. Trends Plant Sci. 2019, 24, 318–327. [Google Scholar] [CrossRef]
- Calabrese, E.J. Pre-conditioning is hormesis I: Documentation, dose-response features and mechanistic foundations. Pharmacol. Res. 2016, 110, 242–264. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.F.; Geihs, M.A.; França, T.F.A.; Moreira, D.C.; Hermes-Lima, M. Is “preparation for Oxidative Stress” a case of physiological conditioning hormesis? Front. Physiol. 2018, 9, 945. [Google Scholar] [CrossRef]
- Vaiserman, A.M. Hormesis and epigenetics: Is there a link? Ageing Res. Rev. 2011, 10, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, S.; Uno, M.; Okabe, E.; Nono, M.; Nishida, E. Environmental stresses induce transgenerationally inheritable survival advantages via germline-to-soma communication in Caenorhabditis elegans. Nat. Commun. 2017, 8, 14031. [Google Scholar] [CrossRef]
- Costantini, D. Hormesis promotes evolutionary change. Dose Response 2019, 17, 1–4. [Google Scholar] [CrossRef]
- Trewavas, A. The foundations o plant intelligence. Interface Focus 2017, 7, 20160098. [Google Scholar] [CrossRef]
- Agathokleous, E.; Belz, R.G.; Calatayud, V.; De Marco, A.; Hoshika, Y.; Kitao, M.; Saitanis, C.J.; Sicard, P.; Paoletti, E.; Calabrese, E.J. Predicting the effect of ozone on vegetation via linear non-threshold (LNT), threshold and hormetic dose-response models. Sci. Total Environ. 2019, 649, 61–74. [Google Scholar] [CrossRef]
- Sasek, T.W.; Richardson, C.J.; Fendick, E.A.; Bevington, S.R.; Kress, L.W. Carryover effects of acid rain and ozone on the physiology of multiple flushes of loblolly pine seedlings. Forest Sci. 1991, 37, 1078–1098. [Google Scholar]
- Langebartels, C.; Heller, W.; Führer, G.; Lippert, M.; Simons, S.; Sandermann, H. Memory effects in the action of ozone on conifers. Ecotoxicol. Environ. Saf. 1998, 41, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Thellier, M.; Lüttge, U. Plant memory: A tentative model. Plant Biol. 2013, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Issakidis-Bourguet, E.; Zhou, D.-X. Perspectives on the interaction between metabolism, redox, and epigenetics in plants. J. Exp. Bot. 2016, 67, 5291–5300. [Google Scholar] [CrossRef]
- Yamamuro, C.; Zhu, J.-K.; Yang, Z. Epigenetic modifications and plant hormone action. Mol. Plant 2016, 9, 57–70. [Google Scholar] [CrossRef] [PubMed]
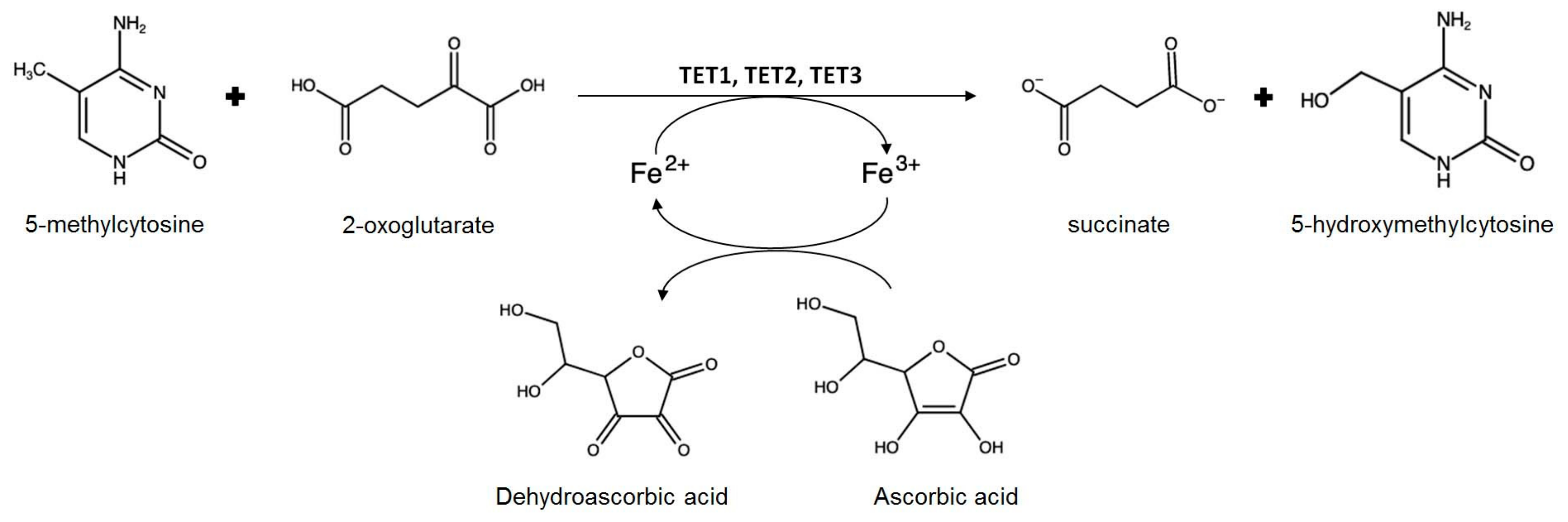
- Young, J.I.; Zuchner, S.; Wang, G. Regulation of the epigenome by Vitamin C. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef]
- Erdmann, R.M.; Souza, A.L.; Clish, C.B.; Gehring, M. 5-hydroxymethylcytosine is not present in appreciable quantities in Arabidopsis DNA. G3 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Song, S.; Wu, Y.; Li, Y.; Chen, T.; Huang, Z.; Liu, S.; Dunwell, T.L.; Pfeifer, G.P.; Dunwell, J.M.; et al. Genome-wide mapping of 5-hydroxymethylcytosine in three rice cultivars reveals its preferential localization in transcriptionally silent transposable element genes. J. Exp. Bot. 2015, 66, 6651–6663. [Google Scholar] [CrossRef] [PubMed]
- Tsukada, Y.; Fang, J.; Erdjument-Bromage, H.; Warren, M.E.; Borchers, C.H.; Tempst, P.; Zhang, Y. Histone demethylation by a family of JmjC domain-containing proteins. Nature 2006, 439, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Li, G.; Cui, X.; Liu, C.; Wang, X.-J.; Cao, X. Comparative analysis of JmjC domain-containing proteins reveals the potential histone demethylases in Arabidopsis and rice. J. Integr. Plant Biol. 2008, 50, 886–896. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Zhang, Q.; Wang, H.; Han, J.; Xu, Z.; Yan, S.; Zhu, Z. OsJMJ703, a rice histone demethylase gene, plays key roles in plant development and responds to drought stress. Plant Physiol. Biochem. 2018, 132, 183–188. [Google Scholar] [CrossRef]
- Chowrasia, S.; Panda, A.K.; Rawal, H.C.; Kaur, H.; Mondal, T.K. Identification of JumonjiC domain containing gene family among the Oryza species and their expression analysis in FL478, a salt tolerant rice genotype. Plant Physiol. Biochem. 2018, 130, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Kacienè, G.; Miškelytè, D.; AbdElgawad, H.; Beemster, G.; Asard, H.; Dikšaitytè, A.; Žaltauskaitè, J.; Sujetovienè, G.; Januškaitienè, I.; Juknys, R. O3 pollution in a future climate increases the competition between summer rape and wild mustard. Plant Physiol. Biochem. 2019, 135, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Bulley, S.; Laing, W. The regulation of ascorbate biosynthesis. Curr. Opin. Plant Biol. 2016, 33, 15–22. [Google Scholar] [CrossRef]
- Frei, M.; Wissuwa, M.; Pariasca-Tanaka, J.; Chen, C.P.; Südekum, K.-H.; Kohno, Y. Leaf ascorbic acid level—Is it really important for ozone tolerance in rice? Plant Physiol. Biochem. 2012, 59, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Lojacono, F.V.; De Tullio, M.C. Why we should stop inferring simple correlations between antioxidants and plant stress tolerance: Towards the antioxidomic era. OMICS 2012, 16, 160–167. [Google Scholar] [CrossRef] [PubMed]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellini, E.; De Tullio, M.C. Ascorbic Acid and Ozone: Novel Perspectives to Explain an Elusive Relationship. Plants 2019, 8, 122. https://doi.org/10.3390/plants8050122
Bellini E, De Tullio MC. Ascorbic Acid and Ozone: Novel Perspectives to Explain an Elusive Relationship. Plants. 2019; 8(5):122. https://doi.org/10.3390/plants8050122
Chicago/Turabian StyleBellini, Erika, and Mario C. De Tullio. 2019. "Ascorbic Acid and Ozone: Novel Perspectives to Explain an Elusive Relationship" Plants 8, no. 5: 122. https://doi.org/10.3390/plants8050122






