Effect of Light Intensity and Quality on Growth Rate and Composition of Chlorella vulgaris
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microalga Cultivation
2.1.1. Cultivation of C. vulgaris in the Greenhouse Environment
2.1.2. Cultivation of C. vulgaris in a Closed Laboratory Bioreactor
2.1.3. Culture Medium
2.2. Measurements
2.3. Nutrient Composition Analyses
2.4. Algal Growth Kinetic Model as a Function of Light Intensity
2.5. Statistical Analysis
3. Results
3.1. Growth Rate of C. vulgaris in the Greenhouse
3.2. Composition of C. vulgaris in the Greenhouse
3.3. Growth rate of C. vulgaris in the Closed Bioreactor
3.4. Composition of C. vulgaris in the Closed Bioreactor
3.5. Algal Growth Kinetic Model as a Function of Light Intensity
4. Discussion
4.1. Effect of Light Intensity on the Growth Rate of C. vulgaris
4.2. Effect of Light Intensity on Composition of C. vulgaris
4.2.1. Experiments in the Greenhouse
4.2.2. Experiments in the Closed Bioreactor—Effect of Wavelength and Artificial Irradiation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mata, T.M.; Martins, M.M.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Chojnacka, K.; Marquez-Rocha, F.-J. Kinetic and stoichiometric relationships of the energy and carbon metabolism in the culture of microalgae. Biotechnology 2004, 3, 21–34. [Google Scholar]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borowitzka, M.A. High-value products from microalgae—Their development and commercialisation. J. Appl. Phycol. 2013, 25, 743–756. [Google Scholar] [CrossRef]
- Yeo, U.; Lee, I.; Seo, I.; Kim, R. Identification of the key structural parameters for the design of a large-scale PBR. Biosyst. Eng. 2018, 171, 165–178. [Google Scholar] [CrossRef]
- Adarme-Vega, T.C.; Lim, D.K.Y.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Factories 2012, 11, 96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aramrueang, N.; Rapport, J.; Zhang, R. Effects of hydraulic retention time and organic loading rate on performance and stability of anaerobic digestion of Spirulina platensis. Biosyst. Eng. 2016, 147, 174–182. [Google Scholar] [CrossRef]
- Chen, J.; Dong, W.; Zhang, X.; Tyagi, R.D.; Drogui, P.; Surampalli, R.D. The potential of microalgae in biodiesel production. Renew. Sustain. Energy Rev. 2018, 90, 336–346. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. Algal culturing. In Algae: Anatomy, Biochemistry, and Biotechnology Taylor and Francis, 1st ed.; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Pulz, O. Photobioreactors: Production systems for phototrophic microorganisms. Appl. Microbiol. Biotechnol. 2001, 57, 287–293. [Google Scholar] [CrossRef]
- Seo, I.; Lee, I.; Hwang, H.; Hong, S.; Bitog, J.P.; Kwon, K.; Lee, C.; Kim, Z.; Cuello, J. Numerical investigation of a bubble-column photo-bioreactor design for microalgae cultivation. Biosyst. Eng. 2012, 113, 229–241. [Google Scholar] [CrossRef]
- Rösch, C.; Posten, C. Challenges and Perspectives of Microalgae Production; Karlsruhe Institute of Technology (KIT): Karlsruhe, Germany, 2012; p. 5. [Google Scholar]
- Metsoviti, M.N.; Papapolymerou, G.; Karapanagiotidis, I.T.; Katsoulas, N. Comparison of growth rate and nutrient content of five microalgae species cultivated in greenhouses. Plants 2019, 8, 279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Zhou, W.; Hu, B.; Min, M.; Chen, P.; Ruan, R.R. Effect of light intensity on algal biomass accumulation and biodiesel production for mixotrophic strains Chlorella kessleri and Chlorella protothecoide cultivated in highly concentrated municipal wastewater. Biotechnol. Bioeng. 2012, 109, 2222–2229. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Ibrahim, I.M.; Harvey, P.J. The influence of photoperiod and light intensity on the growth and photosynthesis of Dunaliella salina (chlorophyta) CCAP 19/30. Plant Physiol. Biochem. 2016, 106, 305–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, S.P.; Singh, P. Effect of temperature and light on the growth of algae species: A review. Renew. Sustain. Energy Rev. 2015, 50, 431–444. [Google Scholar] [CrossRef]
- Difusa, A.; Talukdar, J.; Kalita, M.C.; Mohanty, K.; Goud, V.V. Effect of light intensity and pH condition on the growth, biomass and lipid content of microalgae Scenedesmus species. Biofuels 2015, 6, 37–44. [Google Scholar] [CrossRef]
- Lee, E.; Jalalizadeh, M.; Zhang, Q. Growth kinetic models for microalgae cultivation: A review. Algal Res. 2015, 12, 497–512. [Google Scholar] [CrossRef]
- Minhas, A.K.; Hodgson, P.; Barrow, S.J.; Adholeya, A. A review on the assessment of stress conditions for simultaneous production of microalgal lipids and carotenoids. Front. Microbiol. 2016, 7, 546. [Google Scholar] [CrossRef] [Green Version]
- Wijffels, R.H.; Barbosa, M.J.; Eppink, M.H.M. Microalgae for the production of bulk chemicals and biofuels. Biofuels Bioprod. Bioref. 2010, 4, 287–295. [Google Scholar] [CrossRef] [Green Version]
- Chávez-Fuentes, P.; Ruiz-Marin, A.; Canedo-López, Y. Biodiesel synthesis from Chlorella vulgaris under effect of nitrogen limitation, intensity and quality light: Estimation on the based fatty acids profiles. Mol. Biol. Rep. 2018, 45, 1145–1154. [Google Scholar] [CrossRef]
- Khalili, A.; Najafpour, G.D.; Amini, G.; Samkhaniyani, F. Influence of nutrients and LED light intensities on biomass production of microalgae Chlorella vulgaris. Biotechnol. Bioprocess Eng. 2015, 20, 284–290. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Wang, G.C.; Zhou, B.C. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour. Technol. 2008, 99, 4717–4722. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, L.; Oliveira, A.C. Microalgae as a raw material for biofuels production. J. Ind. Microbiol. Biotechnol. 2009, 36, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Metsoviti, M.N.; Katsoulas, N.; Karapanagiotidis, I.T.; Papapolymerou, G. Effect of nitrogen concentration, two-stage and prolonged cultivation on growth rate, lipid and protein content of Chlorella vulgaris. J. Chem. Technol. Biotechnol. 2019, 94, 1466–1473. [Google Scholar] [CrossRef]
- George, B.; Pancha, I.; Dessai, C.; Chokshi, K.; Paliwal, C.; Ghosh, T.; Mishra, S. Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus—A potential strain for bio-fuel production. Bioresour. Technol. 2014, 171, 367–374. [Google Scholar] [CrossRef]
- He, Q.; Yang, H.; Wu, L.; Hu, C. Effect of light intensity on physiological changes, carbon allocation and neutral lipid accumulation in oleaginous microalgae. Bioresour. Technol. 2015, 191, 219–228. [Google Scholar] [CrossRef]
- Ruangsomboon, S. Effect of light, nutrient, cultivation time and salinity on lipid production of newly isolated strain of the green microalga, Botryococcus braunii KMITL 2. Bioresour. Technol. 2012, 109, 261–265. [Google Scholar] [CrossRef]
- Baiee, M.A.; Salman, J.M. Effect of phosphorus concentration and light intensity on protein content of microalga Chlorella vulgaris. Mesop. Environ. J. 2016, 2, 75–86. [Google Scholar]
- SAG. Sammlung von Algenkulturen der Universität Göttingen. Culture Collection of Algae, Abteilung Experimentelle Phykologie und Sammlung von Algenkulturen (EPSAG), Universität Göttingen, Deutschland. 2007. Available online: http://epsag.uni-goettingen.de (accessed on 4 November 2019).
- Chen, C.Y.; Yeh, K.L.; Aisyah, R.; Lee, D.J.; Chang, J.S. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: A critical review. Bioresour. Technol. 2011, 102, 71–81. [Google Scholar] [CrossRef]
- Perez, L.; Salgueiro, J.L.; Maceiras, R.; Cancela, A.; Sanchez, A. An effective method for harvesting of marine microalgae: pH induced flocculation. Bioresour. Technol. 2017, 97, 20–26. [Google Scholar] [CrossRef]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.Y.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef] [Green Version]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of the Association of Official Analytical Chemists International, 16th ed.; AOAC: Arlington, VA, USA, 1995. [Google Scholar]
- Biancarosa, I.; Espe, M.; Bruckner, C.G.; Heesch, S.; Liland, N.; Waagbø, R.; Torstensen, B.; Lock, E.J. Amino acid composition, protein content, and nitrogen-to-protein conversion factors of 21 seaweed species from Norwegian waters. J. Appl. Phycol. 2017, 29, 1001–1009. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Ryckebosch, E.; Muylaert, K.; Foubert, I. Optimization of an analytical procedure for extraction of lipids from microalgae. J. Am. Oil Chem. Soc. 2012, 89, 189–198. [Google Scholar] [CrossRef]
- Dean, A.; Sigee, D.; Estrada, B.; Pittman, J. Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour. Technol. 2010, 101, 4499–4507. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Singh, G.P.; Sharma, V.K. Effects of culture conditions on growth and biochemical profile of Chlorella vulgaris. J. Plant Pathol. Microb. 2012, 3, 5. [Google Scholar] [CrossRef] [Green Version]
- Daliry, S.; Hallajisani, A.; Mohammadi, R.J.; Nouri, H.; Golzary, A. Investigation of optimal condition for Chlorella vulgaris microalgae growth. Glob. J. Environ. Sci. Manag. 2017, 3, 217–230. [Google Scholar] [CrossRef]
- Khoeyi, Z.A.; Seyfabadi, J.; Ramezanpour, Z. Effect of light intensity and photoperiod on biomass and fatty acid composition of the microalgae, Chlorella vulgaris. Aquac. Int. 2010, 20, 41–49. [Google Scholar] [CrossRef]
- Seyfabadi, J.; Ramezanpour, Z.; Khoeyi, Z.A. Protein, fatty acid, and pigment content of Chlorella vulgaris under different light regimes. J. Appl. Phycol. 2011, 23, 721–726. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–383. [Google Scholar] [CrossRef]
- Das, P.; Lei, W.; Aziz, S.S.; Obbar, S. Enhanced algae growth in both phototrophic and mixotrophic culture under blue light. Bioresour. Technol. 2011, 102, 3883–3887. [Google Scholar] [CrossRef]
- Ra, C.H.; Kang, C.H.; Jung, J.H.; Jeong, G.T.; Kim, S.K. Effects of light-emitting diodes (LEDs) on the accumulation of lipid content using a two-phase culture process with three microalgae. Bioresour. Technol. 2016, 212, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Blair, M.F.; Kokabian, B.; Gude, V.G. Light and growth medium effect on Chlorella vulgaris biomass production. J. Environ. Chem. Eng. 2014, 2, 665–674. [Google Scholar] [CrossRef]
- Wong, Y.K.; Ho, Y.H.; Ho, K.C.; Leung, H.M.; Chow, K.P.; Yung, K.K.L. Effect of different light sources on algal biomass and lipid production in internal Leds-illuminated photobioreactor. J. Mar. Biol. Aquac. 2016, 2, 1–8. [Google Scholar] [CrossRef]
- Atta, M.; Idris, A.; Bukhari, A.; Wahidin, S. Intensity of blue LED light: A potential stimulus for biomass and lipid content in fresh water microalgae Chlorella vulgaris. Bioresour. Technol. 2013, 148, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Rubio, F.C.; Camacho, F.G.; Sevilla, J.M.F.; Chisti, Y.; Grima, E.M. A mechanistic model of photosynthesis in microalgae. Bioresour. Technol. 2002, 81, 459–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanken, W.; Postma, P.R.; Winter, L.; Wijffels, R.H.; Janssen, M. Predicting microalgae growth. Algal Res. 2016, 14, 28–38. [Google Scholar] [CrossRef] [Green Version]
- Takache, H.; Pruvost, J.; Cornet, J.F. Kinetic modeling of the photosynthetic growth of Chlamydomonas reinhardtii in a photobioreactor. Biotechnol. Prog. 2012, 28, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Sasi, D.; Mitra, P.; Vigueras, A.; Hill, G.A. Growth kinetics and lipid production using Chlorella vulgaris in a circulating loop photobioreactor. J. Chem. Technol. Biotechnol. 2011, 86, 875–880. [Google Scholar] [CrossRef]
- Solovchenko, A.E.; Khozin-Goldberg, I.; Didi-Cohen, S.; Cohen, Z.; Merzlyak, M.N. Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. J. Phycol. 2008, 20, 245–251. [Google Scholar] [CrossRef]
- Liu, J.; Yuan, C.; Hu, G.; Li, F. Effects of light intensity on the growth and lipid accumulation of microalga Scenedesmus sp. 11-1 under nitrogen limitation. Appl. Biochem. Biotechnol. 2012, 166, 2127–2137. [Google Scholar] [CrossRef]
- Xia, S.; Wan, L.; Li, A.; Sang, M.; Zhang, C. Effects of nutrients and light intensity on the growth and biochemical composition of a marine microalga Odontella aurita. Chin. J. Oceanol. Limnol. 2013, 3, 1–6. [Google Scholar] [CrossRef]
- Pruvost, J.; Vooren, G.V.; Gouic, B.L.; Couzinet-Mossion, A.; Legrand, J. Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour. Technol. 2011, 102, 150–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Dong, F.; Jin, P. Effects of Light-emitting Diodes (LEDs) on the Accumulation of Lipid Content in Microalgae. In Proceedings of the 2nd International Conference on Sustainable Energy and Environment Protection (ICSEEP 2017), Changsha, China, 23–25 June 2017. [Google Scholar] [CrossRef] [Green Version]
- Gaytan-Luna, D.E.; Ochoa-Alfaro, A.E.; Rocha-Uribe, A. Effect of green and red light in lipid accumulation and transcriptional profile of genes implicated in lipid biosynthesis in Chlamydomonas reinhardtii. Biotechnol. Prog. 2016, 32, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Kendirlioglu, G.; Cetin, A.K. Effect of different wavelengths of light on growth, pigment content and protein amount of Chlorella vulgaris. Fresenius Environ. Bull. 2017, 26, 7974–7980. [Google Scholar]
- Asuthkar, M.; Gunti, Y.; Ramgopal, R.S.; Rao, C.S.; Yadavalli, R. Effect of different wavelengths of light on the growth of Chlorella pyrenoidosa. Int. J. Pharm. Sci. Res. 2016, 7, 847–851. [Google Scholar] [CrossRef]
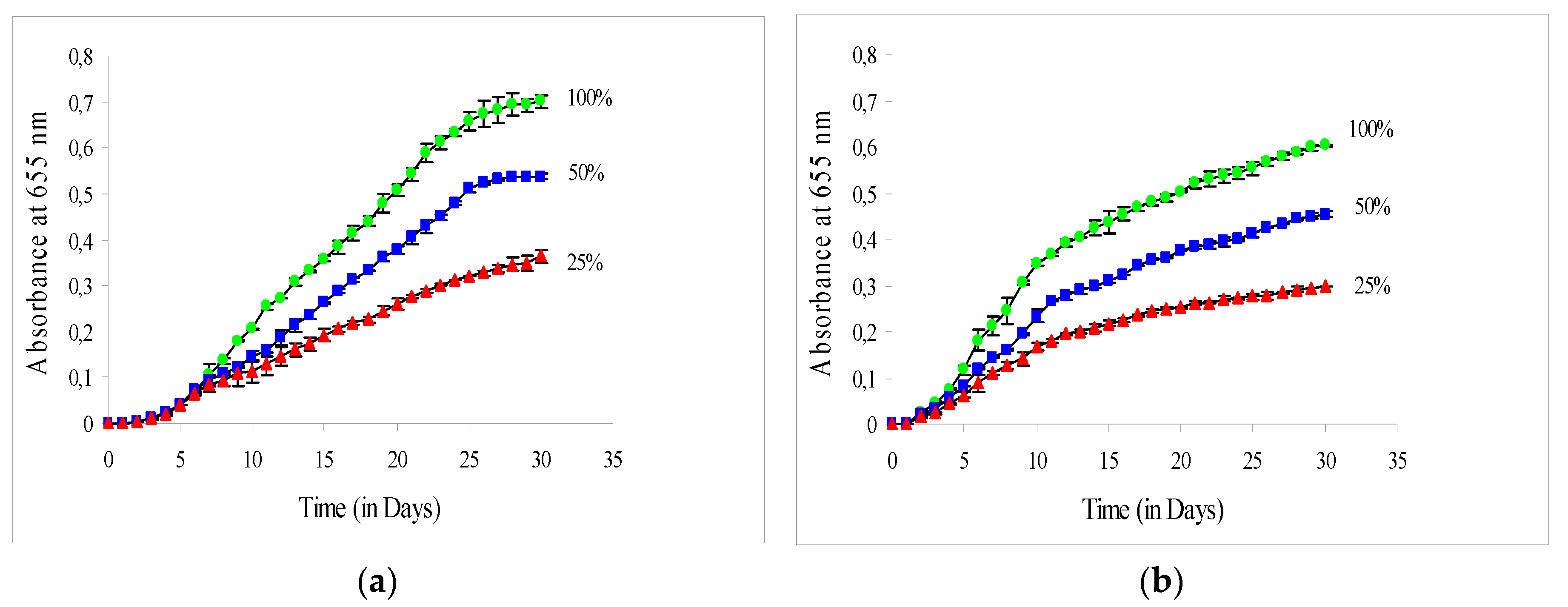

| Growth Rate (in d−1) and Productivity (in g L−1 d−1) | Solar Irradiance | ||
|---|---|---|---|
| 100% | 50% | 25% | |
| June | |||
| μexp | 0.329 ± 0.030 a | 0.252 ± 0.022 b | 0.181 ± 0.020 c |
| Pb | 0.022 ± 0.001 a | 0.015 ± 0.002 b | 0.011 ± 0.000 c |
| Pl | 0.006 ± 0.000 a | 0.003 ± 0.000 b | 0.002 ± 0.000 c |
| Pp | 0.005 ± 0.000 a | 0.004 ± 0.000 b | 0.003 ± 0.000 c |
| September | |||
| μexp | 0.293 ± 0.033 a | 0.221 ± 0.022 b | 0.169 ± 0.020 c |
| Pb | 0.018 ± 0.001 a | 0.014 ± 0.001 b | 0.010 ± 0.001 c |
| Pl | 0.004 ± 0.000 a | 0.003 ± 0.000 b | 0.002 ± 0.000 c |
| Pp | 0.005 ± 0.000 a | 0.004 ± 0.000 b | 0.003 ± 0.000 c |
| Solar Irradiance | Moisture | Lipids | Proteins | Ash | Fiber | NFE |
|---|---|---|---|---|---|---|
| June | ||||||
| 100% | 7.925 ± 0.263 a | 25.567 ± 0.301 a | 23.735 ± 0.371 a | 12.275 ± 0.411 a | 9.425 ± 0.275 a | 21.073 ± 0.483 c |
| 50% | 7.950 ± 0.311 a | 22.279 ± 0.157 b | 23.606 ± 0.817 a | 12.375 ± 0.299 a | 9.325 ± 0.189 a | 24.466 ± 0.990 b |
| 25% | 8.050 ± 0.191 a | 19.456 ± 0.342 c | 23.801 ± 0.651 a | 12.275 ± 0.359 a | 9.225 ± 0.320 a | 27.193 ± 0.730 a |
| September | ||||||
| 100% | 7.725 ± 0.171 a | 24.716 ± 0.200 a | 27.634 ± 0.520 a | 12.125 ± 0.330 a | 9.250 ± 0.208 a | 18.550 ± 0.488 c |
| 50% | 7.975 ± 0.171 a | 21.300 ± 0.269 b | 27.563 ± 0.878 a | 12.338 ± 0.229 a | 9.300 ± 0.365 a | 21.524 ± 1.200 b |
| 25% | 8.000 ± 0.082 a | 17.292 ± 0.267 c | 27.121 ± 0.794 a | 12.275 ± 0.386 a | 9.475 ± 0.206 a | 25.837 ± 0.681 a |
| Growth Rate (in d−1) and Productivity (in g L−1 d−1) | I420–520/I580–680 | |||
|---|---|---|---|---|
| 1.30 | 0.90 | 0.60 | 0.30 | |
| μexp | 0.394 ± 0.021 a | 0.346 ± 0.022 b | 0.266 ± 0.031 c | 0.183 ± 0.023 d |
| Pb | 0.080 ± 0.004 a | 0.060 ± 0.001 b | 0.041 ± 0.002 c | 0.030 ± 0.002 d |
| Pl | 0.010 ± 0.000 a | 0.009 ± 0.000 a | 0.008 ± 0.000 b | 0.006 ± 0.000 c |
| Pp | 0.021 ± 0.000 a | 0.015 ± 0.000 b | 0.010 ± 0.000 c | 0.008 ± 0.000 d |
| Light Intensity (μmol Photons m−2 s−1) | ||||
| 520 | 390 | 260 | 130 | |
| μexp | 0.363 ± 0.023 a | 0.318 ± 0.020 b | 0.276 ± 0.011 c | 0.166 ± 0.020 d |
| Pb | 0.085 ± 0.001 a | 0.054 ± 0.001 b | 0.041 ± 0.001 c | 0.032 ± 0.003 d |
| Pl | 0.019 ± 0.000 a | 0.008 ± 0.000 b | 0.004 ± 0.000 c | 0.003 ± 0.000 d |
| Pp | 0.022 ± 0.000 a | 0.014 ± 0.000 b | 0.010 ± 0.000 c | 0.008 ± 0.000 d |
| I420–520/I580–680 | Moisture | Lipids | Proteins | Ash | Fiber | NFE |
|---|---|---|---|---|---|---|
| 1.30 | 8.067 ± 0.252 a | 12.680 ± 0.191 d | 26.710 ± 0.274 a | 12.367 ± 0.451 a | 9.267 ± 0.252 a | 30.910 ± 0.476 a |
| 0.90 | 7.800 ± 0.100 a | 16.219 ± 0.655 c | 25.509 ± 0.141 b | 12.267 ± 0.252 a | 9.467 ± 0.250 a | 28.738 ± 0.725 b |
| 0.60 | 7.867 ± 0.351 a | 18.447 ± 0.436 b | 25.249 ± 0.304 b | 12.333 ± 0.473 a | 9.333 ± 0.255 a | 26.771 ± 0.643 b |
| 0.30 | 8.000 ± 0.100 a | 19.984 ± 0.439 a | 25.819 ± 0.126 b | 12.217 ± 0.225 a | 9.433 ± 0.252 a | 24.547 ± 0.839 c |
| Light Intensity (in μmol Photons m−2 s−1) | ||||||
| 520 | 8.000 ± 0.265 a | 22.240 ± 0.305 a | 25.741 ± 0.122 a | 12.233 ± 0.306 a | 9.533 ± 0.208 a | 22.252 ± 0.469 d |
| 390 | 7.700 ± 0.200 a | 14.696 ± 0.307 b | 25.565 ± 0.415 a | 12.250 ± 0.391 a | 9.333 ± 0.153 a | 30.456 ± 0.643 c |
| 260 | 7.900 ± 0.100 a | 10.948 ± 0.306 c | 25.545 ± 0.322 a | 12.400 ± 0.400 a | 9.300 ± 0.265 a | 33.907 ± 0.221 b |
| 130 | 8.100 ± 0.100 a | 7.879 ± 0.288 d | 25.100 ± 0.629 a | 12.200 ± 0.300 a | 9.233 ± 0.252 a | 37.487 ± 0.840 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Metsoviti, M.N.; Papapolymerou, G.; Karapanagiotidis, I.T.; Katsoulas, N. Effect of Light Intensity and Quality on Growth Rate and Composition of Chlorella vulgaris. Plants 2020, 9, 31. https://doi.org/10.3390/plants9010031
Metsoviti MN, Papapolymerou G, Karapanagiotidis IT, Katsoulas N. Effect of Light Intensity and Quality on Growth Rate and Composition of Chlorella vulgaris. Plants. 2020; 9(1):31. https://doi.org/10.3390/plants9010031
Chicago/Turabian StyleMetsoviti, Maria N., George Papapolymerou, Ioannis T. Karapanagiotidis, and Nikolaos Katsoulas. 2020. "Effect of Light Intensity and Quality on Growth Rate and Composition of Chlorella vulgaris" Plants 9, no. 1: 31. https://doi.org/10.3390/plants9010031






