Molecular Markers Improve Abiotic Stress Tolerance in Crops: A Review
Abstract
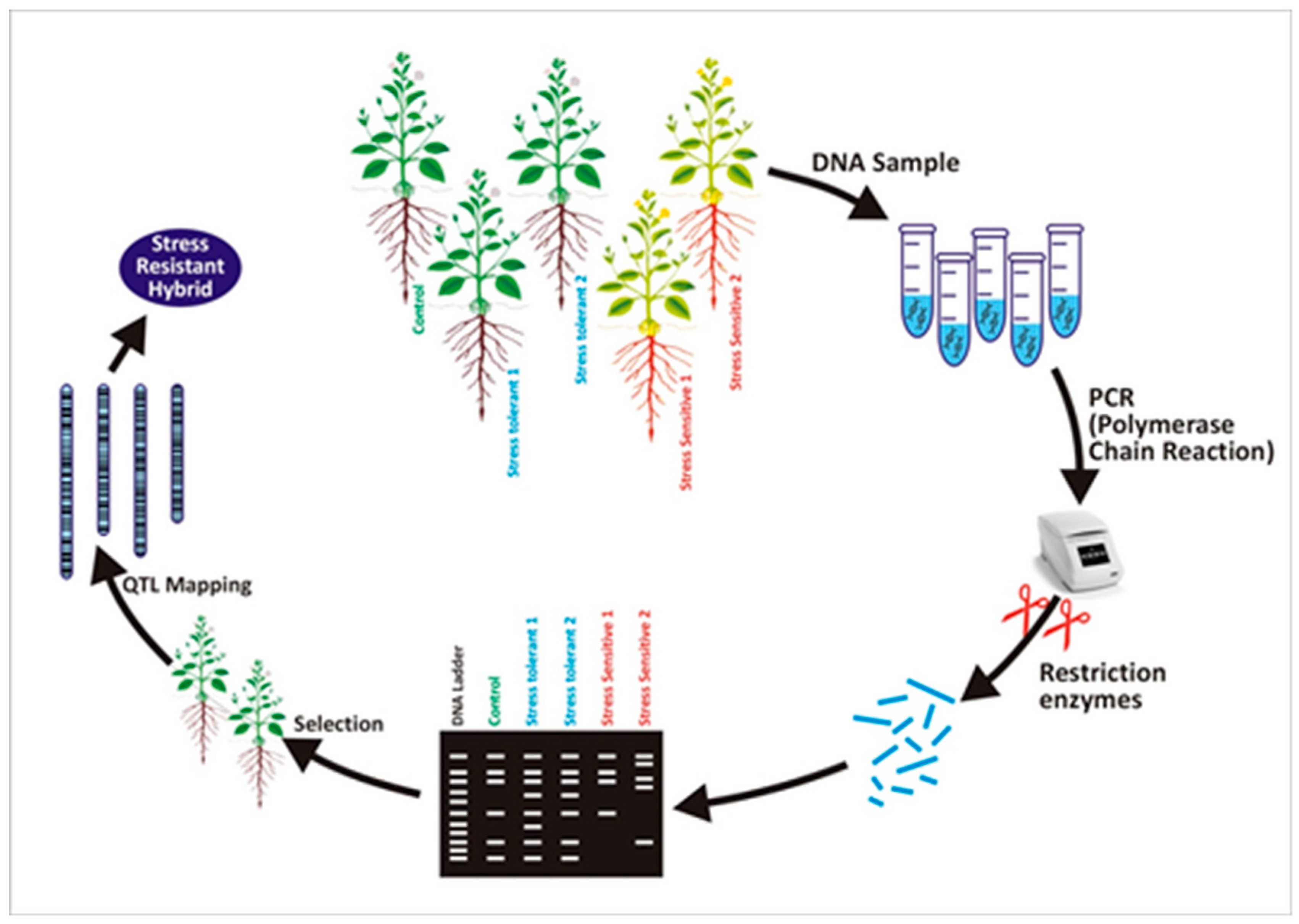
:1. Introduction
2. Abiotic Stress Impact on Agricultural Crops
3. DNA Marker Applications for Abiotic Stress Tolerance
3.1. RAPD Marker Analysis for Salinity and Drought Stresses
3.2. Stress Tolerance in Hybrids
3.3. Genetic Diversity Identification under Heat and Frost Stress
4. Mapping of QTL Genes Related to Abiotic Stresses by DNA Markers
4.1. Saltol
4.2. Dehydrin
5. QTL Mapping by DNA Markers
6. Marker-Assisted Selection by SNP Marker
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fedoroff, N.V.; Battisti, D.S.; Beachy, R.N.; Cooper, P.J.; Fischhoff, D.A.; Hodges, C.N.; Knauf, V.C.; Lobell, D.; Mazur, B.J.; Molden, D.; et al. Radically rethinking agriculture for the 21st century. Science 2010, 327, 833–834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Younis, A.; Siddique, M.I.; Kim, C.K.; Lim, K.B. RNA Interference (RNAi) induced gene silencing: A promising approach of hi-tech plant breeding. Int. J. Biol. Sci. 2014, 10, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Rejeb, I.; Pastor, V.; Mauch-Mani, B. Plant responses to simultaneous biotic and abiotic stress: Molecular mechanisms. Plants 2014, 3, 458–475. [Google Scholar] [CrossRef] [PubMed]
- Boyer, J.S. Plant productivity and environment. Science 1982, 218, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Taiz, L.; Zeiger, E. Plant Physiology; The Benjamin Cummings Publishing Co., Inc.: Redwood City, CA, USA, 1991; pp. 100–119. [Google Scholar]
- Larcher, W. Physiological Plant Ecology: Ecophysiology and Stress Physiology of Functional Groups; Springer: Berlin/Heidelberg, Germany, 2003; pp. 345–437. [Google Scholar]
- Farooq, A.; Nadeem, M.; Abbas, G.; Shabbir, A.; Khalid, M.S.; Javeed, H.M.R.; Saeed, M.F.; Akram, A.; Younis, A.; Akhtar, G. Cadmium partitioning, physiological and oxidative stress responses in Marigold (Calendula calypso) grown on contaminated soil: Implications for phytoremediation. Bull. Environ. Contam. Toxicol. 2020, 105, 270–276. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Younis, A.; Riaz, A.; Mansoor, F.; Hameed, M.; Akram, N.A.; Abideen, Z. Morpho-anatomical adaptations of two Tagetes erecta L. cultivars with contrasting response to drought stress. Pak. J. Bot. 2020, 52, 801–810. [Google Scholar] [CrossRef]
- Chen, D.; Shao, Q.; Yin, L.; Younis, A.; Zheng, B. Polyamine function in plants: Metabolism, regulation on development, and roles in abiotic stress responses. Front. Plant Sci. 2019, 9, 1945. [Google Scholar] [CrossRef] [PubMed]
- Younis, A.; Riaz, A.; Tariq, U.; Nadeem, M.; Khan, N.A.; Ahsan, M.; Adil, W.; Naseem, M.K. Drought tolerance of Leucophyllum frutescens: Physiological and morphological studies reveal the potential xerophyte. Acta Sci. Pol. Hortorum Cultus 2017, 16, 85–94. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxic. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Al-Juburi, H.J.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Lipiec, J.; Doussan, C.; Nosalewicz, A.; Kondracka, K. Effect of drought and heat stresses on plant growth and yield: A review. Int. Agrophys. 2013, 27, 463–477. [Google Scholar] [CrossRef]
- Kang, J.S.; Singh, H.; Singh, G.; Kang, H.; Kalra, V.P.; Kaur, J. Abiotic Stress and Its Amelioration in Cereals and Pulses: A Review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1019–1045. [Google Scholar]
- Mullis, K.B.; Faloona, F.A. Specific synthesis of DNA in vitro via a polymerase-catalyzed chain reaction. Methods Enzymol. 1987, 155, 335–350. [Google Scholar] [PubMed]
- Ullah, I. Molecular Genetic Studies for Drought Tolerance in Cotton. Ph.D. Thesis, Quaid-i-Azam University, Islamabad, Pakistan, 2009. [Google Scholar]
- Ramzan, F.; Kim, H.T.; Shim, K.K.; Choi, Y.H.; Younis, A.; Lim, K.B. Genetic diversity and relationship assessment of Lilium lancifolium × Asiatic hybrid ‘Chianti’ progeny by ISSR markers. Eur. J. Hortic. Sci. 2018, 83, 142–150. [Google Scholar] [CrossRef]
- Daryanto, S.; Wang, L.; Jacinthe, P.A. Global synthesis of drought effects on maize and wheat production. PLoS ONE 2016, 11, e0156362. [Google Scholar] [CrossRef]
- Farooq, M.; Gogoi, N.; Barthakur, S.; Baroowa, B.; Bharadwaj, N.; Alghamdi, S.S.; Siddique, K.H.M. Drought stress in grain legumes during reproduction and grain filling. J. Agron. Crop Sci. 2017, 203, 81–102. [Google Scholar] [CrossRef]
- Zlatev, Z.; Lidon, F.C. An overview on drought induced changes in plant growth, water relations and photosynthesis. Emir. J. Food Agric. 2012, 24, 57–72. [Google Scholar]
- Król, A. The Growth and Water Uptake by Yellow Seed and Black Seed Rape Depending on the State of Soil Compaction. Ph.D. Thesis, Bohdan Dobrzañski, Institute of Agrophysics PAS, Lublin, Poland, 2013. [Google Scholar]
- Minhas, P.S.; Rane, J.; Pasala, R.K. Abiotic stresses in agriculture: An overview. In Abiotic Stress Management for Resilient Agriculture; Springer: New York, NY, USA, 2017. [Google Scholar]
- Francini, A.; Sebastiani, L. Abiotic Stress Effects on Performance of Horticultural Crops. Horticulturae 2019, 5, 67. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Liu, B.; Piao, S.; Wang, X.; Lobell, D.B.; Huang, Y.; Huang, M.; Yao, Y.; Bassu, S.; Ciais, P.; et al. Temperature increase reduces global yields of major crops in four independent estimates. Proc. Natl. Acad. Sci. USA 2017, 114, 9326–9331. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M. Crop Plants and Abiotic Stresses. J. Biomol. Res. Ther. 2013, 3, e125. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; Burucs, Z.; Tucher, S.V.; Schmidhalter, U. Short term effects of drought and salinity on mineral nutrient distribution along growing leaves of maize seedlings. Environ. Exp. Bot. 2007, 60, 268–275. [Google Scholar] [CrossRef]
- Younis, A.; Riaz, A.; Qasim, M.; Mansoor, F.; Zulfiqar, F.; Tariq, U.; Bhatti, Z.M. Screening of marigold (Tagetes erecta L.) cultivars for drought stress based on vegetative and physiological characteristics. Int. J. Food Allied Sci. 2017, 3, 56–63. [Google Scholar] [CrossRef]
- Bechtold, U.; Field, B. Molecular mechanisms controlling plant growth during abiotic stress. J. Exp. Bot. 2018, 69, 2753–2758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rupnarayan, S. Tolerance of plants in response to abiotic stress factors. Recent Adv. Petrochem. Sci. 2017, 1, 555–573. [Google Scholar]
- Younis, A.; Riaz, A.; Ikram, S.; Nawaz, T.; Hameed, M.; Fatima, S.; Batool, R.; Ahmad, F. Salinity-induced structural and functional changes in three cultivars of Alternanthra bettzikiana (Regel) G. Nicholson. Turk. J. Agric. For. 2013, 37, 674–687. [Google Scholar] [CrossRef]
- Shasany, A.K.; Darokar, M.P.; Dhawan, S.; Gupta, A.K.; Gupta, S.; Shukla, A.K.; Patra, N.K.; Khanuja, S.P. Use of RAPD and AFLP markers to identify interand intraspecific hybrids of Mentha. J. Hered. 2005, 96, 542–549. [Google Scholar] [CrossRef] [Green Version]
- Bhutta, W.M.; Amjad, M. Molecular characterization of salinity tolerance in wheat (Triticum aestivum L.). Arch. Agron. Soil Sci. 2015, 61, 1641–1648. [Google Scholar]
- Dogan, I.; Kekec, G.; Ozyigit, I.I.; Sakcali, M.S. Salinity induced changes in cotton (Gossypium hirsutum L.). Pak. J. Bot. 2012, 44, 21–25. [Google Scholar]
- Jain, S.M. Tissue culture-derived variation in crop improvement. Euphytica 2001, 118, 153–166. [Google Scholar] [CrossRef]
- Balkrishna, R.A.; Shankarrao, S.S. In vitro screening and molecular genetic markers associated with salt tolerance in maize. Afr. J. Biotechnol. 2013, 12, 4251–4255. [Google Scholar]
- Karp, A.; Kresovich, S.; Bhat, K.V.; Ayad, W.G.; Hodgkin, T. Molecular Tools in Plant Genetic Resources Conservation: A Guide to the Technologies; IPGRI: Rome, Italy, 1993; pp. 278–286. [Google Scholar]
- Saleh, B. DNA changes in cotton (Gossypium hirsutum L.) under salt stress as revealed by RAPD marker. Adv. Hortic. Sci. 2016, 60, 13–21. [Google Scholar]
- Shao, H.B.; Liang, Z.S.; Shao, M.A.; Sun, Q. Dynamic changes of anti-oxidative enzymes of 10 wheat genotypes at soil water deficits. Coll. Surf. B Biointerfaces 2005, 42, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Kirigwi, F.M.; Van Ginkel, M.; Brown-Guedira, G.; Gill, B.S.; Paulsen, G.M.; Fritz, A.K. Markers associated with a QTL for grain yield in wheat under drought. Mol. Breed. 2007, 20, 401–413. [Google Scholar] [CrossRef]
- Zhou, G.A.; Chang, R.Z.; Qiu, L.J. Overexpression of soybean ubiquitin-conjugating enzyme gene GmUBC2 confers enhanced drought and salt tolerance through modulating abiotic stress-responsive gene expression in Arabidopsis. Plant Mol. Biol. 2010, 72, 357–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huseynova, I.M.; Rustamova, S.M. Screening for drought stress tolerance in wheat genotypes using molecular markers. Proc. ANAS (Biol. Sci.) 2010, 65, 132–139. [Google Scholar]
- Rashed, M.A.; Sabry, S.B.S.; Atta, A.H.; Mostafa, A.M. Development of RAPD markers associated drought tolerance in bread wheat (Triticum aestivum L.). Egypt. J. Genet. Cytol. 2010, 39, 131–142. [Google Scholar]
- Lin, K.H.; Lo, H.F.; Lee, S.P.; Kuo, C.G.; Chen, J.T.; Yeh, W.L. RAPD markers for the identification of yield traits in tomatoes under heat stress via bulked segregant analysis. Hereditas 2006, 143, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Krupa-Małkiewicz, M.; Bienias, A. BSA and molecular markers screening for salt stress tolerant mutant of Petunia obtained in in vitro culture. Ciência Rural St. Maria 2018, 48, e20170042. [Google Scholar] [CrossRef]
- Yuan, X.; Bao, Z.; He, Y.; Chen, Q.; Wang, G. Development of SCAR marker related to summer stress tolerance in tall fescue (Festuca arundinacea). Not. Bot. Horti Agrobot. 2014, 42, 81–87. [Google Scholar] [CrossRef] [Green Version]
- Nie, G.; Tang, L.; Zhang, Y.; Huang, L.; Ma, X.; Cao, X.; Pan, L.; Zhang, X.; Zhang, X. Development of SSR markers based on transcriptome sequencing and association analysis with drought tolerance in Perennial grass miscanthus from China. Front. Plant Sci. 2017, 8, 801. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Guo, L.; Shu, Z.; Sun, Y.; Chen, Y.; Liang, Z.; Guo, H. Identification of amplified fragment Length Polymorphism (AFLP) markers tightly associated with drought stress gene in male sterile and fertile salvia miltiorrhiza bunge. Int. J. Mol. Sci. 2013, 14, 6518–6528. [Google Scholar] [CrossRef] [PubMed]
- Razavi, F.; Keyser, E.D.; Riek, J.D.; Labeke, M.C.V. A method for testing drought tolerance in Fragaria based on fast screening for water deficit response and use of associated AFLP and EST candidate gene markers. Euphytica 2011, 180, 385–409. [Google Scholar] [CrossRef]
- Raga, V.; Intrigliolo, D.S.; Bernet, G.P.; Carbonell, E.A.; Asins, M.J. Genetic analysis of salt tolerance in a progeny derived from the citrus rootstocks Cleopatra mandarin and trifoliate orange. Tree Genet. Genomes 2016, 12, 34. [Google Scholar] [CrossRef]
- Gharsallah, C.; Abdelkrim, A.B.; Fakhfakh, H.; Salhi-Hannachi, A.; Gorsane, F. SSR marker-assisted screening of commercial tomato genotypes under salt stress. Breed. Sci. 2016, 66, 823–830. [Google Scholar] [CrossRef] [Green Version]
- Damra, E.M.; Kasrawi, M.; Akash, M.W. Development of scar marker linked to heat stress tolerance in tomato. In Proceedings of the 65th ISERD International Conference, Mecca, Saudi Arabia, 23–24 January 2017; pp. 122–130. [Google Scholar]
- Comlekcioglu, N.; Simsek, O.; Boncuk, M.; Aka-Kacar, Y. Genetic characterization of heat tolerant tomato (Solanum lycopersicon) genotypes by SRAP and RAPD markers. Genet. Mol. Res. 2010, 9, 2263–2274. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, F.; Weng, Y.; Sun, M.; Shi, X.; Zhou, Y.; Yu, L.; Wu, Z. Identification of heat-tolerance QTLs and high-temperature stress-responsive genes through conventional QTL mapping, QTLseq and RNA-seq in tomato. BMC Plant Biol. 2019, 19, 398. [Google Scholar] [CrossRef]
- Wang, B.; Guo, X.; Zhao, P.; Ruan, M.; Yu, X.; Zou, L.; Yang, Y.; Li, X.; Deng, D.; Xiao, J.; et al. Molecular diversity analysis, drought related marker-traits association mapping and discovery of excellent alleles for 100-day old plants by EST-SSRs in cassava germplasms (Manihot esculenta Cranz). PLoS ONE 2017, 12, e0177456. [Google Scholar] [CrossRef]
- Foolad, M.R. Breeding for abiotic stress tolerances in tomato. In Abiotic Stresses: Plant Resistance Through Breeding and Molecular Approaches; Ashraf, M., Harris, P.J.C., Eds.; The Haworth Press Inc.: New York, NY, USA, 2005; pp. 613–684. [Google Scholar]
- Zhang, G.L.; Chen, L.Y.; Xiao, G.Y.; Xiao, Y.H.; Chen, X.B.; Zhang, S.T. Bulked segregant analysis to detect QTL related to heat tolerance in rice (Oryza sativa L.) using SSR markers. Agric. Sci. China 2009, 8, 482–487. [Google Scholar] [CrossRef]
- Barakat, M.N.; Al-Doss, A.A.; Elshafei, A.A.; Moustafa, K.A. Identification of new microsatellite marker linked to the grainfilling rate as indicator for heat tolerance genes in F2 wheat population. Aust. J. Crop Sci. 2011, 5, 104–110. [Google Scholar]
- Poortavakoli, S.; Sheidai, M.; Alishah, O.; Noormohammadi, Z. Genetic diversity analysis in drought stress tolerant cottons. Nucleus 2017, 60, 57–62. [Google Scholar] [CrossRef]
- Collins, N.C.; Tardieu, F.; Tuberosa, R. Quantitative trait loci and crop performance under abiotic stress: Where do we stand? Plant Phys. 2008, 147, 469–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.Y.; Quiros, C.F. Sequence-Related Amplified Polymorphism (SRAP) a new marker system based on a simple PCR reaction: Its application to mapping and gene tagging in Brassica. Theor. Appl. Genet. 2001, 103, 455–461. [Google Scholar] [CrossRef]
- Howarth, C.J. Genetic improvements of tolerance to high temperature. In Abiotic Stresses: Plant Resistance through Breeding and Molecular Approaches; Ashraf, M., Harris, P.J.C., Eds.; Howarth Press Inc.: New York, NY, USA, 2005. [Google Scholar]
- Bohnert, H.J.; Gong, Q.; Li, P.; Ma, S. Unraveling abiotic stress tolerance mechanisms-getting genomics going. Curr. Opin. Plant Biol. 2006, 9, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Fang, L.; Yang, T.; Zhang, X.; Hu, J.; Zhang, H.; Han, W.; Hua, Z.; Hao, J.; Zong, X. Marker-trait association analysis of frost tolerance of 672 worldwide pea (Pisum sativum L.) collections. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- De Leon, T.B.; Linscombe, S.; Gregorio, G.; Subudhi, P.K. Genetic variation in Southern USA rice genotypes for seedling salinity tolerance. Front. Plant Sci. 2015, 6, 374. [Google Scholar] [CrossRef] [Green Version]
- Mohammadi-Nejad, G.; Arzani, A.; Rezai, A.M.; Singh, R.K.; Gregorio, G.B. Assessment of rice genotypes for salt tolerance using microsatellite markers associated with the saltol QTL. Afr. J. Biotechnol. 2008, 7, 730–736. [Google Scholar]
- Linh, L.H.; Linh, T.H.; Xuan, T.D.; Ham, L.H.; Ismail, A.M.; Khanh, T.D. Molecular breeding to improve salt tolerance of rice (Oryza sativa L.) in the Red River Delta of Vietnam. Int. J. Plant Genom. 2012, 2012. [Google Scholar] [CrossRef] [Green Version]
- Kosava, K.; Vitamvas, P.; Prasil, I.T. Wheat and barley dehydrins under cold, drought, and salinity-what can LEA-II proteins tell us about plant stress response? Front. Plant Sci. 2014, 5, 343. [Google Scholar]
- Du, J.B.; Yuan, S.; Chen, Y.E.; Sun, X.; Zhang, Z.W.; Xu, F.; Yuan, M.; Shang, J.; Lin, H.H. Comparative expression analysis of dehydrins between two barley varieties, wild barley and Tibetan hulless barley associated with different stress resistance. Acta Physiol. Plant. 2011, 33, 567–574. [Google Scholar] [CrossRef]
- Mare, C.; Mazzucotelli, E.; Crosatti, C.; Francia, E.; Stanca, A.M.; Cattivelli, L. Hv-WRKY38: A new transcription factor involved in cold- and drought-response in barley. Plant Mol. Biol. 2004, 55, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.W.; Close, T.J. A newly identified barley gene, Dhn12 encoding a YSK2 DHN, is located on chromosome 6H and has embryo-specific expression. Theo. Appl. Genet. 2000, 100, 1274–1278. [Google Scholar] [CrossRef]
- Walia, H.; Wilson, C.; Wahid, A.; Condamine, P.; Cui, X.; Close, T.J. Expression analysis of barley (Hordeum vulgare L.) during salinity stress. Funct. Integr. Genom. 2006, 6, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.; Younis, R.; Rashed, M.; Eldomyati, F.; Rashed, N. Molecular markers associated with drought tolerance in Citrullus colocynthis. Egypt. J. Genet. Cytol. 2012, 41, 311–325. [Google Scholar] [CrossRef]
- Saeed, M.; Guo, W.; Zhang, T. Association mapping for salinity tolerance in cotton (Gossypium hirsutum L) germplasm from US and diverse regions of China. Aust. J. Crop Sci. 2014, 8, 338–346. [Google Scholar]
- Abdelraheem, A.; Mahdy, Z.; Zhang, J.F. The first linkage map for a recombinant inbred line population in cotton (Gossypium barbadense) and its use in studies of PEG-induced dehydration tolerance. Euphytica 2015, 205, 941–958. [Google Scholar] [CrossRef]
- Cui, D.Z.; Wu, D.D.; Somarathna, Y.; Xu, C.Y.; Li, S.; Li, P.; Zhang, H.; Chen, H.; Li, Z. QTL mapping for salt tolerance based on snp markers at the seedling stage in maize (Zea mays L.). Euphytica 2015, 203, 273–283. [Google Scholar] [CrossRef]
- Kebede, H.; Subudhi, P.K.; Rosenow, D.T.; Nguyen, H.T. Quantitative trait loci influencing drought tolerance in grain sorghum (Sorghum bicolor L. Moench). Theor. Appl. Genet. 2001, 103, 266–276. [Google Scholar] [CrossRef]
- Sbei, H.; Sato, K.; Shehzad, T.; Harrabi, M.; Okuno, K. Detection of QTLs for salt tolerance in Asian barley (Hordeum vulgare L.). Breed. Sci. 2014, 64, 378–388. [Google Scholar] [CrossRef] [Green Version]
- Xue, D.W.; Huang, Y.Z.; Zhang, X.Q.; Wei, K.; Westcott, S.; Li, C.; Chen, M.; Zhang, G.; Lance, R. Identification of QTLs associated with salinity tolerance at late growth stage in barley. Euphytica 2009, 169, 187–196. [Google Scholar] [CrossRef]
- Mohammadi, V.; Zali, A.A.; Bihamta, M.R. Mapping QTLs for heat tolerance in wheat. J. Agric. Sci. Technol. 2008, 10, 261–267. [Google Scholar]
- Dolferus, R.; Thavamanikumar, S.; Sangma, H.; Kleven, S.; Wallace, X.; Forrest, K.; Rebetzke, G.; Hayden, M.; Borg, L.; Smith, A.; et al. Determining the genetic architecture of reproductive stage drought tolerance in wheat using a correlated trait and correlated marker effect model. Genes Genomes Genet. 2019, 9, 473–489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, M.A.; Khan, S.H.; Khan, A.S.; Kazi, A.M.; Basra, S.M.A. Identification of QTLs for drought tolerance traits on wheat chromosome 2A using association mapping. Int. J. Agric. Biol. 2014, 16, 862–870. [Google Scholar]
- Hussain, B.; Lucas, S.J.; Ozturk, L.; Budak, H. Mapping QTLs conferring salt tolerance and micronutrient concentrations at seedling stage in wheat. Sci. Rep. 2017, 7, 15662. [Google Scholar] [CrossRef] [PubMed]
- Tadesse, W.; Suleiman, S.; Tahir, I.; Sanchez-Garcia, M.; Jighly, A.; Hagras, A.; Thabet, S.; Baum, M. Heat tolerant QTL associated with grain yield and its components in spring bread wheat under heat stressed environments of Sudan and Egypt. Crop Sci. 2018, 59, 199–211. [Google Scholar] [CrossRef] [Green Version]
- Wainaina, C.M.; Makihara, D.; Nakamura, M.; Ikeda, A.; Suzuki, T.; Mizukami, Y.; Nonoyama, T.; Doi, K.; Kikuta, M.; Samejima, H.; et al. Identification and validation of QTLs for cold tolerance at the booting stage and other agronomic traits in a rice cross of a Japanese tolerant variety, Hananomai, and a NERICA parent, WAB56-104. Plant Prod. Sci. 2018, 21, 132–143. [Google Scholar] [CrossRef] [Green Version]
- Munns, R.; Tester, M. Mechanisms of Salinity Tolerance. Ann. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [Green Version]
- Babu, R.C.; Nguyen, B.D.; Chamarerk, V.P.; Shanmugasundaram, P.; Chezhian, P.; Jeyaprakash, S.K.; Ganesh, S.K.; Palchamy, A.; Sadasivam, S.; Sarkarung, S.; et al. Genetic analysis of drought resistance in rice by molecular markers, Association between Secondary Traits and Field Performance. Crop Sci. 2003, 43, 1457–1469. [Google Scholar] [CrossRef]
- Xiao, Y.; Yi, P.; Luo, L.; Zhang, G.; Deng, H.; Dai, L.; Liu, X.; Tang, W.; Chen, L.; Wang, G.L. Quantitative trait loci associated with seed set under high-temperature stress at the flowering stage in rice (Oryza sativa L.). Euphytica 2011, 178, 331–338. [Google Scholar] [CrossRef]
- Buu, B.C.; Ha, P.T.T.; Tam, B.P.; Nhien, T.T.T.; Hieu, N.V.; Phuoc, N.T. Quantitative trait loci associated with heat tolerancein rice (Oryza sativa L.). Plant Breed. Biotechnol. 2014, 2, 14–24. [Google Scholar] [CrossRef]
- Sabouri, H.; Sabouri, A. New evidence of QTLs attributed to salinity tolerance in rice. Afr. J. Biotechnol. 2009, 7, 4376–4383. [Google Scholar]
- Bizimana, J.B.; Luzi Kihupi, A.; Murori, R.W.; Singh, R.K. Identification of quantitative trait loci for salinity tolerance in rice (Oryza sativa L.) using IR29/Hasawi mapping population. J. Genet. 2017, 96, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-García, R.; Prats, E.; Fondevilla, S.; Satovic, Z.; Rubiales, D. Quantitative trait loci asso-ciated to drought adaptation in pea (Pisum sativum L.). Plant Mol. Biol. Rep. 2015, 33, 1768. [Google Scholar] [CrossRef] [Green Version]
- Klein, A.; Houtin, H.; Rond, C.; Marget, P.; Jacquin, F.; Boucherot, K.; Huart, M.; Rivière, N.; Boutet, G.; Lejeune-Hénaut, I.; et al. QTL analysis of frost damage in pea suggests different mechanisms involved in frost tolerance. Theor. Appl. Genet. 2014, 127, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Gore, M.; Buckler, E.; Yu, J. Status and prospects of association mapping in plants. Plant Genome 2008, 1, 5–20. [Google Scholar] [CrossRef]
- Yan, J.B.; Warburton, M.; Crouch, J. Association mapping for enhancing maize (Zea mays L.) Genetic improvement. Crop Sci. 2011, 51, 433–449. [Google Scholar] [CrossRef]
- Yan, J.B.; Kandianis, C.B.; Harjes, C.E.; Bai, L.; Kim, E.H.; Yang, X.H.; Skinner, D.J.; Fu, Z.; Mitchell, S.; Li, Q.; et al. Rare genetic variation at Zea mays crtRB1 increases b-carotene in maize grain. Nat. Genet. 2010, 42, 322–327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hao, Z.; Li, X.; Xie, C.; Weng, J.; Li, M.; Zhang, D.; Liang, X.; Liu, L.; Liu, S.; Zhang, S. Identification of functional genetic variations underlying drought tolerance in maize using SNP markers. J. Integr. Plant Biol. 2011, 53, 641–652. [Google Scholar] [CrossRef]
| Crop | DNA Marker | Abiotic Stress | Objective | Reference |
|---|---|---|---|---|
| Petunia × atkinsiana | RAPD | Salinity |
| [44] |
| D. Don cv. Prism Red. | ISSR | |||
| Tall fescue (Festuca arundinacea) | Sequence characterized amplified region (SCAR) | Summer stress | Development of sequence-related markers to screen summer stress-resistant plants | [45] |
| Perennial grass (Miscanthus sinensis) | SSR | Drought | To formulate SSR markers linked to drought resistance by utilizing transcriptome sequencing | [46] |
| Salvia (Salvia miltiorrhiza) | AFLP | Drought |
| [47] |
| ||||
| Strawberry Fragaria ananassa Duch.) | Expressed sequence tag (EST) | Drought |
| [48] |
| Amplified fragment length polymorphism (AFLP) |
| |||
| Citrus | Quantitative trait loci (QTL) | Salinity | Genetic evaluation of salt resistance concerning physiological and vegetative characteristics | [49] |
| Citrus reshni | ||||
| Poncirus trifoliata | ||||
| Tomato (Solanum lycopersicum L.) | SSR | Salinity |
| [50] |
| ||||
| Tomato (Solanum lycopersicum L.) | RAPD | Heat |
| [51] |
| Sequence characterized amplified region (SCAR) |
| |||
| Tomato (Solanum lycopersicum L.) | Sequence-related amplified polymorphism (SRAP) | Heat |
| [52] |
| Randomly amplified polymorphic DNA (RAPD) |
| |||
| Tomato (Solanum lycopersicum L.) | Quantitative trait loci (QTL) | Heat |
| [53] |
| ||||
| Cassava (Manihot esculenta Cranz) | Expressed sequence tags–simple sequence repeat (EST–SSR) markers | Drought |
| [54] |
|
| Crop | Botanical Name | DNA Marker | No. of QTLs | No. of Chromosomes with QTLs Loci | Objective | Reference |
|---|---|---|---|---|---|---|
| Cotton | Gossypium hirsutum | Simple sequence repeats (SSR); |
| [73] | ||
| Single strand conformation polymorphic (SSCP) | 14 | 11 |
| [74] | ||
| Maize | Zea mays | Single nucleotide polymorphism (SNP) | 29 | 1, 3, and 5 |
| [75] |
| Sorghum | Sorghum bicolor | Restriction fragment length polymorphism (RFLP) | 7 | 1 and 2 |
| [76] |
| Barley | Hordeum vulgare | Single nucleotide polymorphism (SNP) | 2 (salt tolerance indices) | 1 and 2 |
| [77] |
| Barley | Simple sequence repeats (SSR) | 13 | 4 |
| [78] |
| DNA Markers | No. of QTLs Related to Tolerance | Chromosome No. with QTL Loci | Research Objectives | References |
|---|---|---|---|---|
| Simple sequence repeats (SSR) and amplified fragment length polymorphism (AFLP) | 3 | 1, 5, 7 |
| [79] |
| Simple sequence repeats (SSR), diversity array technology (DarT), gene-based marker for Vrn-A1 | 1 | 5A |
| [80] |
| Simple sequence repeats (SSR) | 8 | 2A |
| [81] |
| Single nucleotide polymorphism (SNP) | 6 | 7A |
| [82] |
| Single nucleotide polymorphism (SNP) | 3 | 3B, 4A, and 5A |
| [83] |
| Simple sequence repeats (SSR) | 2 | 8 and 10 |
| [84] |
| DNA Markers | No. of QTLs Related to Tolerance | No. of Chromosomes with QTLs Loci | Research Objectives | References |
|---|---|---|---|---|
| Simple sequence repeats (SSR) | 47 | 1–4 and 6–12 |
| [86] |
| Single feature polymorphism, simple sequence repeats (SSR) | 2 | 4 and 10 |
| [87] |
| Simple sequence repeats (SSR) | 1 | 3 |
| [88] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Younis, A.; Ramzan, F.; Ramzan, Y.; Zulfiqar, F.; Ahsan, M.; Lim, K.B. Molecular Markers Improve Abiotic Stress Tolerance in Crops: A Review. Plants 2020, 9, 1374. https://doi.org/10.3390/plants9101374
Younis A, Ramzan F, Ramzan Y, Zulfiqar F, Ahsan M, Lim KB. Molecular Markers Improve Abiotic Stress Tolerance in Crops: A Review. Plants. 2020; 9(10):1374. https://doi.org/10.3390/plants9101374
Chicago/Turabian StyleYounis, Adnan, Fahad Ramzan, Yasir Ramzan, Faisal Zulfiqar, Muhammad Ahsan, and Ki Byung Lim. 2020. "Molecular Markers Improve Abiotic Stress Tolerance in Crops: A Review" Plants 9, no. 10: 1374. https://doi.org/10.3390/plants9101374








