Evaluation of Anticancer Activity of Satureja montana Supercritical and Spray-Dried Extracts on Ehrlich’s Ascites Carcinoma Bearing Mice
Abstract
:1. Introduction
2. Results
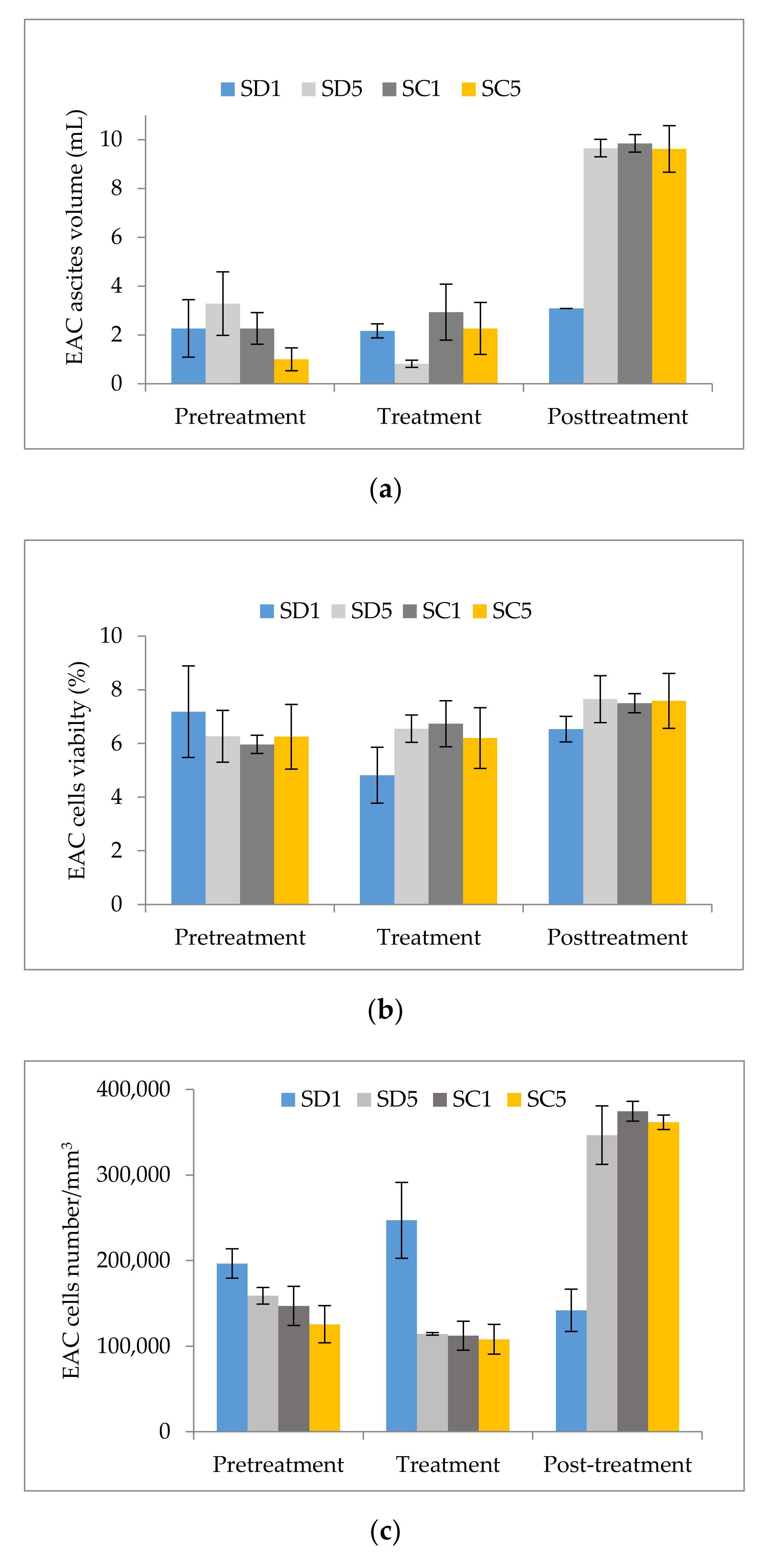
2.1. Impact of S. montana Extracts on the Volume of Ascites, Viability, and Number of Cells
2.2. Impact of S. montana Extracts on the Antioxidant Status Malign Cells of EAC
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Preparation of Extracts
4.3. Chemical Analysis
4.4. Antioxidant Activity
4.5. Animals and Treatments
- Group EAC—animals with implanted Ehrlich ascites carcinoma (EAC) cells treated with 2 mL/kg of saline, i.p. (n = 6).
- Group PRETREATMENT—animals pretreated with 2 mL/kg of the investigated extract, i.p. during seven days (n = 6).
- Group TREATMENT—animals were treated with 2 mL/kg of the investigated extract, i.p. seven days after the implantation of EAC (n = 6).
- Group POSTTREATMENT—animals post-treated with 2 mL/kg of the investigated extract, i.p. during seven days, seven days after implantation (n = 6).
- After 14 days from the day of implementation of EAC, all animals were sacrificed and the ascites was collected for further biochemical analyses.
4.6. Determination of Ascites Volume, Tumor Cell Number, and Cell Viability
4.7. Biochemical Assays
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Group | EAC Ascites Volume (mL) | |||
|---|---|---|---|---|
| EAC Control | 7.117 ± 0.458 | |||
| Extract | SD1 | SD5 | SC1 | SC5 |
| Pretreatment | 2.267 ± 1.178 a,* | 3.283 ± 1.301 a,* | 2.267 ± 0.674 a,* | 1.000 ± 0.469 a,* |
| Treatment | 2.167 ± 0.288 ab,* | 0.817 ± 0.147 b,* | 2.933 ± 1.147 a,* | 2.267 ± 1.065 ab,* |
| Post-treatment | 3.083 ± 0.382 b,* | 9.657 ± 0.359 a,* | 9.851 ± 0.932 a,* | 9.622 ± 0.954 a,* |
| Group | EAC Cells Viability (%) | |||
|---|---|---|---|---|
| EAC Control | 7.243 ± 1.425 | |||
| Extract | SD1 | SD5 | SC1 | SC5 |
| Pretreatment | 7.183 ± 1.705 a | 6.267 ± 0.965 a | 5.967 ± 0.339 a | 6.250 ± 1.205 a |
| Treatment | 4.817 ± 1.042 a,* | 6.550 ± 0.509 a | 6.733 ± 0.857 a | 6.200 ± 1.131 a |
| Post-treatment | 6.533 ± 0.476 a | 7.650 ± 0.874 a | 7.499 ± 0.356 a | 7.586 ± 1.023 a |
| Group | EAC Cells Number/mm3 | |||
|---|---|---|---|---|
| EAC Control | 119 583.333 ± 6 755.862 | |||
| Extract | SD1 | SD5 | SC1 | SC5 |
| Pretreatment | 196,583.333 ± 17,133.058 a,* | 158,833.333 ± 9739.952 ab,* | 147,000.000 ± 22,858.259 b | 125,583.333 ± 21,731.122 b |
| Treatment | 247,000.000 ± 44,340.730 a,* | 114,333.300 ± 1505.545 b | 112,166.700 ± 16,898.720 b | 108,000.000 ± 17,378.150 b |
| Post-treatment | 141,833.333 ± 24,717.740 b,* | 346,569.58 ± 34,156.81 a,* | 374,511.22 ± 11,568.13 a,* | 361,581.47 ± 8455.36 a,* |
References
- Dunkić, V.; Kremer, D.; Dragojević Müller, I.; Stabentheiner, E.; Kuzmić, S.; Jurišić Grubešić, R.; Vujić, L.; Kosalec, I.; Randić, M.; Srečec, S.; et al. Chemotaxonomic and micromorphological traits of Satureja montana L. and S. subspicata Vis. (Lamiaceae). Chem. Biodivers. 2012, 9, 2825–2842. [Google Scholar]
- Ćetković, G.S.; Mandić, A.I.; Čanadanović-Brunet, J.M.; Djilas, S.M.; Tumbas, V.T. HPLC screening of phenolic compounds in winter savory (Satureja montana L.) extracts. J. Liq. Chromatogr. Relat. Technol. 2007, 30, 293–306. [Google Scholar]
- Parsaei, P.; Bahmani, M.; Naghdi, N.; Asadi-Samani, M.; Rafieian-Kopaei, M. A review of therapeutic and pharmacological effects of thymol. Der Pharm. Lett. 2016, 8, 150–154. [Google Scholar]
- Sharifi-Rad, M.; Varoni, E.M.; Iriti, M.; Martorell, M.; Setzer, W.N.; del Mar Contreras, M.; Salehi, B.; Soltani-Nejad, A.; Rajabi, S.; Tajbahshs, M.; et al. Carvacrol and human health: A comprehensive review. Phytother. Res. 2018, 32, 1675–1687. [Google Scholar] [CrossRef] [PubMed]
- Skočibušić, M.; Bezić, N. Chemical composition and antidiarrhoeal activities of winter savory (Satureja montana L.) essential oil. Pharm. Biol. 2003, 41, 622–626. [Google Scholar] [CrossRef]
- Stanic, G.; Samaržija, I. Diuretic activity of Satureja montana subsp. montana extracts and oil in rats. Phytother. Res. 1993, 7, 363–366. [Google Scholar] [CrossRef]
- Yamasaki, K.; Nakano, M.; Kawahata, T.; Mori, H.; Otake, T.; Ueda, N.; Oishi, I.; Inami, R.; Yamane, M.; Nakamura, M.; et al. Anti-HIV-1 activity of herbs in Labiatae. Biol. Pharm. Bull. 1998, 21, 829–833. [Google Scholar] [CrossRef] [Green Version]
- Oussalah, M.; Caillet, S.; Saucier, L.; Lacroix, M. Inhibitory effects of selected plant essential oils on the growth of four pathogenic bacteria: E. coli O157: H7, Salmonella typhimurium, Staphylococcus aureus and Listeria monocytogenes. Food Control. 2007, 18, 414–420. [Google Scholar] [CrossRef]
- Malongane, F.; McGaw, L.J.; Mudau, F.N. The synergistic potential of various teas, herbs and therapeutic drugs in health improvement: A review. J. Sci. Food Agric. 2017, 97, 4679–4689. [Google Scholar] [CrossRef]
- Surh, Y.J. Molecular mechanisms of chemopreventive effects of selected dietary and medicinal phenolic substances. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 1999, 428, 305–327. [Google Scholar] [CrossRef]
- Fitsiou, E.; Anestopoulos, I.; Chlichlia, K.; Galanis, A.; Kourkoutas, I.; Panayiotidis, M.I.; Pappa, A. Antioxidant and antiproliferative properties of the essential oils of Satureja thymbra and Satureja parnassica and their major constituents. Anticancer Res. 2016, 36, 5757–5763. [Google Scholar] [CrossRef] [Green Version]
- Tahmasbpour, E.; Mohammadpour, G.; Tahmasbpour, R.; Noureini, S.; Bagherpour, G. In vitro Antimicrobial and cytotoxicity assays of Satureja bakhtiarica and Zataria multiflora essential oils. AJPCT 2015, 3, 502–511. [Google Scholar]
- Yousefzadi, M.; Riahi-Madvar, A.; Hadian, J.; Rezaee, F.; Rafiee, R.; Biniaz, M. Toxicity of essential oil of Satureja khuzistanica: In vitro cytotoxicity and anti-microbial activity. J. Immunotoxicol. 2014, 11, 50–55. [Google Scholar] [CrossRef] [Green Version]
- Stammati, A.; Bonsi, P.; Zucco, F.; Moezelaar, R.; Alakomi, H.L.; Von Wright, A. Toxicity of selected plant volatiles in microbial and mammalian short-term assays. Food Chem. Toxicol. 1999, 37, 813–823. [Google Scholar] [CrossRef]
- Zeytinoglu, M.; Aydin, S.; Oztürk, Y.; Baser, K. Inhibitory effects of carvacrol on DMBA induced pulmonary tumorigenesis in rats. Acta Pharm. Turc. 1998, 2, 93–98. [Google Scholar]
- Horvathova, E.; Turcaniova, V.; Slamenova, D. Comparative study of DNA-damaging and DNA-protective effects of selected components of essential plant oils in human leukemic cells K562. Neoplasma 2006, 54, 478–483. [Google Scholar]
- Zeytinoglu, H.; Incesu, Z.; Baser, K.H.C. Inhibition of DNA synthesis by carvacrol in mouse myoblast cells bearing a human N-RAS oncogene. Phytomedicine 2003, 10, 292–299. [Google Scholar] [CrossRef]
- Jayakumar, S.; Madankumar, A.; Asokkumar, S.; Raghunandhakumar, S.; Kamaraj, S.; Divya, M.G.J.; Devaki, T. Potential preventive effect of carvacrol against diethylnitrosamine-induced hepatocellular carcinoma in rats. Mol. Cell. Biochem. 2012, 360, 51–60. [Google Scholar] [CrossRef]
- Elgndi, M.A.; Filip, S.; Pavlić, B.; Vladić, J.; Stanojković, T.; Žižak, Ž.; Zeković, Z. Antioxidative and cytotoxic activity of essential oils and extracts of Satureja montana L., Coriandrum sativum L. and Ocimum basilicum L. obtained by supercritical fluid extraction. J. Supercrit. Fluids 2017, 128, 128–137. [Google Scholar] [CrossRef]
- Četojević-Simin, D.D.; Čanadanvić-Brunet, J.M.; Bogdanović, G.N.; Ćetković, G.S.; Tumbas, V.T.; Djilas, S.M. Antioxidative and antiproliferative effects of Satureja montana L. extracts. J. BUON 2004, 9, 443–449. [Google Scholar]
- Grosso, C.; Figueiredo, A.C.; Burillo, J.; Mainar, A.M.; Urieta, J.S.; Barroso, J.G.; Coelho, A.; Palavra, A.M. Enrichment of the thymoquinone content in volatile oil from Satureja montana using supercritical fluid extraction. J. Sep. Sci. 2009, 32, 328–334. [Google Scholar] [CrossRef]
- Grosso, C.; Oliveira, A.C.; Mainar, A.M.; Urieta, J.S.; Barroso, J.G.; Palavra, A.M.F. Antioxidant activities of the supercritical and conventional Satureja montana extracts. J. Food Sci. 2009, 74, C713–C717. [Google Scholar] [CrossRef] [PubMed]
- Damjanović-Vratnica, B.; Perović, S.; Lu, T.; Santos, R. Effect of matrix pretreatment on the supercritical CO2 extraction of Satureja montana essential oil. Chem. Ind. Chem. Eng. Q. 2016, 22, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Vidović, S.; Zeković, Z.; Marošanović, B.; Todorović, M.P.; Vladić, J. Influence of pre-treatments on yield, chemical composition and antioxidant activity of Satureja montana extracts obtained by supercritical carbon dioxide. J. Supercrit. Fluids 2014, 95, 468–473. [Google Scholar] [CrossRef]
- Vladić, J.; Canli, O.; Pavlić, B.; Zeković, Z.; Vidović, S.; Kaplan, M. Optimization of Satureja montana subcritical water extraction process and chemical characterization of volatile fraction of extracts. J. Supercrit. Fluids 2017, 120, 86–94. [Google Scholar] [CrossRef]
- Jakovljević, M.; Vladić, J.; Vidović, S.; Pastor, K.; Jokić, S.; Molnar, M.; Jerković, I. Application of deep eutectic solvents for the extraction of rutin and rosmarinic acid from Satureja montana L. and evaluation of the extracts antiradical activity. Plants 2020, 9, 153. [Google Scholar] [CrossRef] [Green Version]
- Rezvanpanah, S.; Rezaei, K.; Razavi, S.H.; Moini, S. Use of microwave-assisted hydrodistillation to extract the essential oils from Satureja hortensis and Satureja montana. Food Sci. Technol. Res. 2008, 14, 311–314. [Google Scholar] [CrossRef] [Green Version]
- Zekovic, Z.; Gavaric, A.; Pavlic, B.; Vidovic, S.; Vladic, J. Optimization: Microwave irradiation effect on polyphenolic compounds extraction from winter savory (Satureja montana L.). Sep. Sci. Technol. 2017, 52, 1377–1386. [Google Scholar] [CrossRef]
- Kaiser, C.S.; Römpp, H.; Schmidt, P.C. Pharmaceutical applications of supercritical carbon dioxide. Die Pharm. 2001, 56, 907. [Google Scholar]
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of spray-drying in microencapsulation of food ingredients: An overview. Food Res. Int. 2007, 40, 1107–1121. [Google Scholar] [CrossRef]
- Vladić, J.; Ambrus, R.; Szabó-Révész, P.; Vasić, A.; Cvejin, A.; Pavlić, B.; Vidović, S. Recycling of filter tea industry by-products: Production of A. millefolium powder using spray drying technique. Ind. Crop. Prod. 2016, 80, 197–206. [Google Scholar] [CrossRef]
- Vladić, J.; Zeković, Z.; Jokić, S.; Svilović, S.; Kovačević, S.; Vidović, S. Winter savory: Supercritical carbon dioxide extraction and mathematical modeling of extraction process. J. Supercrit. Fluids 2016, 117, 89–97. [Google Scholar] [CrossRef]
- Vidović, S.S.; Vladić, J.Z.; Vaštag, Ž.G.; Zeković, Z.P.; Popović, L.M. Maltodextrin as a carrier of health benefit compounds in Satureja montana dry powder extract obtained by spray drying technique. Powder Technol. 2014, 258, 209–215. [Google Scholar] [CrossRef]
- El Tawab, A.M.A.; Shahin, N.N.; Abdel Mohsen, M.M. Protective effect of Satureja montana extract on cyclophosphamide-induced testicular injury in rats. Chem. Biol. Interact. 2014, 224, 196–205. [Google Scholar] [CrossRef]
- Silva, F.V.; Martins, A.; Salta, J.; Neng, N.R.; Nogueira, J.M.; Mira, D.; Gaspar, N.; Justino, J.; Grosso, C.; Urieta, J.S.; et al. Phytochemical profile and anticholinesterase and antimicrobial activities of supercritical versus conventional extracts of Satureja montana. J. Agric. Food Chem. 2009, 57, 11557–11563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ćebovic, T.; Vladić, J.; Gavarić, A.; Zeković, Z.; Vidović, S. Assessment of antioxidant and hepatoprotective potential of Satureja montana extracts against CCl4 induced liver damage. Nat. Med. Mater. 2019, 39, 5–10. [Google Scholar] [CrossRef] [Green Version]
- Serrano, C.; Matos, O.; Teixeira, B.; Ramos, C.; Neng, N.; Nogueira, J.; Nunes, M.L.; Marques, A. Antioxidant and antimicrobial activity of Satureja montana L. extracts. J. Sci. Food Agric. 2011, 91, 1554–1560. [Google Scholar] [CrossRef]
- Ipek, E.; Tüylü, B.A.; Zeytinoglu, H. Effects of carvacrol on sister chromatid exchanges in human lymphocyte cultures. In Animal Cell Technology: Basic & Applied Aspects; Springer: Dordrecht, The Netherlands, 2003; pp. 471–474. [Google Scholar]
- Arunasree, K.M. Anti-proliferative effects of carvacrol on a human metastatic breast cancer cell line, MDA-MB 231. Phytomedicine 2010, 17, 581–588. [Google Scholar] [CrossRef]
- Yin, Q.H.; Yan, F.X.; Zu, X.Y.; Wu, Y.H.; Wu, X.P.; Liao, M.C.; Deng, S.W.; Yin, L.L.; Zhuang, Y.Z. Anti-proliferative and pro-apoptotic effect of carvacrol on human hepatocellular carcinoma cell line HepG-2. Cytotechnology 2012, 64, 43–51. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, S.S.; Saraswati, S.; Mathur, R.; Pandey, M. Antitumor properties of Boswellic acid against Ehrlich ascites cells bearing mouse. Food Chem. Toxicol. 2011, 49, 1924–1934. [Google Scholar] [CrossRef]
- Ćebović, T.; Spasić, S.; Popović, M. Cytotoxic effects of the Viscum album L. extract on Ehrlich tumour cells in vivo. Phytother. Res. 2008, 22, 1097–1103. [Google Scholar] [CrossRef]
- Vulić, J.; Ćebović, T.; Čanadanović, V.; Ćetković, G.; Djilas, S.; Čanadanović-Brunet, J.; Velicanski, A.; Cvetkovic, D.; Tumbas, V. Antiradical, antimicrobial and cytotoxic activities of commercial beetroot pomace. Food Funct. 2013, 4, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Espín, J.C.; Soler-Rivas, C.; Wichers, H.J. Characterization of the total free radical scavenger capacity of vegetable oils and oil fractions using 2,2-diphenyl-1-picrylhydrazyl radical. J. Agric. Food Chem. 2000, 48, 648–656. [Google Scholar] [CrossRef]
- Bergmayer, U.H. Methoden der Enzymatischen Analyse; Chemie: Weinheim, Germany, 1970; pp. 483–484. [Google Scholar]
- Beers, R.F.J.; Sizer, J.W. Spectrophotometric method for measuring of breakdown of hydrogen peroxide by catalase. J. Biol. Chem. 1950, 195, 133–140. [Google Scholar]
- Simon, L.M.; Fatrai, Z.; Jonas, D.J.; Matkovics, B. Study of metabolism enzymes during the development of Phaseolus vulgaris. Biochem. Physiol. Pflanz. 1974, 166, 389–393. [Google Scholar] [CrossRef]
- Beuthler, E. Glutathione reductase, glutathione peroxidase, catalase, glutathione. In Red Cell Metabolism: A Manual of Biochemical Methods; Beutler, E., Ed.; Grune and Stratton: New York, NY, USA, 1984. [Google Scholar]
- Goldberg, D.M.; Spooner, R.J. Glutathione reductase. In Methods of Enzymatic Analysis; Bergmayer, H.U., Ed.; Chemie: Weinheim, Germany, 1983; Volume 3. [Google Scholar]
- Beuthler, E.; Duron, O.; Kelly, B. Improved methods for the determination of blood glutathione. J. Lab. Clin. Med. 1983, 61, 882–889. [Google Scholar]
- Buege, A.L.; Aust, D.S. Microsomal lipid peroxidation. In Methods in Enzymology; Fleisher, S., Parker, L., Eds.; Academic Press: New York, NY, USA, 1978. [Google Scholar]
| Parameter | Group | EAC | SD1 | SD5 | SC1 | SC5 |
|---|---|---|---|---|---|---|
| XOD | Pretreatment | 0.156 ± 0.009 | 0.142 ± 0.004 b | 0.151 ± 0.005 b | 0.807 ± 0.138 a,* | 0.822 ± 0.057 *,a |
| Treatment | 0.104 ± 0.011 c,* | 0.836 ± 0.069 b,* | 1.035 ± 0.064 a,* | 1.012 ± 0.014 a,* | ||
| Posttreatment | 0.220 ± 0.014 a,* | 0.102 ± 0.005 b,* | 0.097 ± 0.012 b,* | 0.101 ± 0.004 b,* | ||
| CAT | Pretreatment | 0.503 ± 0.013 | 0.585 ± 0.019 a,* | 0.429 ± 0.002 b,* | 0.233 ± 0.016 c,* | 0.136 ± 0.002 d* |
| Treatment | 0.636 ± 0.027 a,* | 0.138 ± 0.002 c,* | 0.194 ± 0.006 b,* | 0.191 ± 0.003 b,* | ||
| Post-treatment | 0.316 ± 0.008 b,* | 0.698 ± 0.052 a,* | 0.713 ± 0.002 a,* | 0.742 ± 0.041 a,* | ||
| Px | Pretreatment | 0.325 ± 0.023 | 0.302 ± 0.016 c | 0.432 ± 0.010 c,* | 0.618 ± 0.003 b,* | 0.881 ± 0.019 a,* |
| Treatment | 0.120 ± 0.006 c,* | 0.875 ± 0.035 a,* | 0.715 ± 0.004 b,* | 0.710 ± 0.002 b,* | ||
| Post-treatment | 0.474 ± 0.035 a,* | 0.100 ± 0.008 b,* | 0.114 ± 0.023 b,* | 0.118 ± 0.007 b,* | ||
| GR | Pretreatment | 2.187 ± 0.107 | 2.115 ± 0.165 b | 3.012 ± 0.105 b,* | 4.957 ± 0.135 a,* | 5.707 ± 0.667 a,* |
| Treatment | 1.202 ± 0.095 b,* | 5.627 ± 0.363 a,* | 5.172 ± 0.111 a,* | 5.195 ± 0.024 a,* | ||
| Post-treatment | 3.930 ± 0.217 a,* | 1.195 ± 0.002 b,* | 1.254 ± 0.028 b,* | 1.284 ± 0.185 b,* | ||
| GSHPx | Pretreatment | 0.779 ± 0.048 | 0.796 ± 0.024 d | 0.933 ± 0.022 c,* | 1.302 ± 0.049 b,* | 1.697 ± 0.038 a,* |
| Treatment | 0.474 ± 0.034 b,* | 1.91 ± 0.345 a,* | 2.043 ± 0.020 a,* | 2.007 ± 0.031 a,* | ||
| Post-treatment | 1.131 ± 0.151 a,* | 0.395 ± 0.065 b,* | 0.401 ± 0.068 b,* | 0.421 ± 0.005 b,* | ||
| GSH | Pretreatment | 1.603 ± 0.110 | 1.417 ± 0.056 a,* | 1.358 ± 0.029 a,* | 0.871 ± 0.033 b,* | 0.884 ± 0.043 b,* |
| Treatment | 1.513 ± 0.058 a,* | 0.926 ± 0.069 b,* | 0.621 ± 0.011 c,* | 0.635 ± 0.005 c,* | ||
| Post-treatment | 1.105 ± 0.063 b,* | 1.801 ± 0.045 a,* | 1.799 ± 0.029 a,* | 1.842 ± 0.003 a,* | ||
| LPx | Pretreatment | 0.032 ± 0.008 | 0.030 ± 0.009 c | 0.041 ± 0.003 c | 0.100 ± 0.007 a,* | 0.080 ± 0.005 b,* |
| Treatment | 0.022 ± 0.005 c | 0.042 ± 0.521 b | 0.077 ± 0.005 a | 0.083 ± 0.005 a | ||
| Post-treatment | 0.056 ± 0.018 a,* | 0.011 ± 0.006 b,* | 0.019 ± 0.006 b,* | 0.014 ± 0.001 b,* |
| Compound | SC | SD |
|---|---|---|
| α-Terpinene | 0.15 | n.i. |
| p-Cymene | 3.96 | 0.36 |
| γ-Terpinene | 0.80 | n.i. |
| α-Terpineol | n.i. | 0.21 |
| Eucalyptol | 0.42 | n.i. |
| Trans-sabinene hydrate | 0.33 | n.i. |
| Cis-sabinene hydrate | 0.15 | n.i. |
| Linalool | 0.29 | 0.18 |
| Borneol | 1.56 | n.i. |
| Terpinen 4-ol | 0.84 | 0.42 |
| Carvacrol | 78.61 | 71.82 |
| Trans-caryophyllene | 2.40 | 0.24 |
| Caryophyllene oxide | 1.26 | 1.31 |
| α-Amorphen | 0.46 | n.i. |
| β-Bisabolene | 0.74 | n.i. |
| γ-Cadinene | 0.53 | n.i. |
| δ-Cadinene | 0.78 | n.i. |
| β-Cadinene | n.i. | 0.24 |
| Spatulenol | n.i. | 0.21 |
| Heptakosane | 0.17 | n.i. |
| Nonakosane | 0.19 | n.i. |
| Group | XOD | CAT | Px | GR | GSH-Px | GSH | LPx |
|---|---|---|---|---|---|---|---|
| Control | 1.93 ± 0.02 a | 9.56 ± 0.37 b | 10.68 ± 1.38 b | 6.13 ± 0.07 a | 8.70 ± 0.38 a | 4.98 ± 0.16 a | 3.04 ± 0.07 a |
| SC | 1.87 ± 0.03 ab | 13.4 ± 0.59 a | 14.88 ± 0.38 a | 7.03 ± 0.46 a | 7.54 ± 2.23 a | 4.99 ± 0.17 a | 2.62 ± 0.54 a |
| SD | 1.86 ± 0.03 b | 12.37 ± 0.18 a | 14.57 ± 0.80 a | 6.53 ± 1.61 a | 6.67 ± 1.06 a | 4.92 ± 0.39 a | 2.21 ± 0.24 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vladić, J.; Ćebović, T.; Vidović, S.; Jokić, S. Evaluation of Anticancer Activity of Satureja montana Supercritical and Spray-Dried Extracts on Ehrlich’s Ascites Carcinoma Bearing Mice. Plants 2020, 9, 1532. https://doi.org/10.3390/plants9111532
Vladić J, Ćebović T, Vidović S, Jokić S. Evaluation of Anticancer Activity of Satureja montana Supercritical and Spray-Dried Extracts on Ehrlich’s Ascites Carcinoma Bearing Mice. Plants. 2020; 9(11):1532. https://doi.org/10.3390/plants9111532
Chicago/Turabian StyleVladić, Jelena, Tatjana Ćebović, Senka Vidović, and Stela Jokić. 2020. "Evaluation of Anticancer Activity of Satureja montana Supercritical and Spray-Dried Extracts on Ehrlich’s Ascites Carcinoma Bearing Mice" Plants 9, no. 11: 1532. https://doi.org/10.3390/plants9111532






