Unravelling Chemical Composition of Agave Spines: News from Agave fourcroydes Lem.
Abstract
1. Introduction
2. Results and Discussion
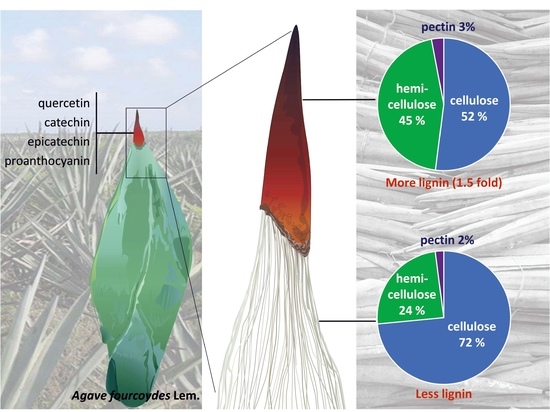
2.1. Lignocellulosic Composition of Fiber and Spines
2.2. Metabolomic Profiling and In Situ Secondary Polyphenols Detection of Spine Tissues
3. Materials and Methods
3.1. Leaves and Spines Sampling and Preparation
3.2. Monosaccharide Composition Analysis
3.3. Lignin Composition
3.4. Extraction and Metabolic Profiling of Spines
3.5. Cellulose and Lignin Visualization in Spines
3.6. Histological Staining for PAs and Condensed Tannins
3.7. Statistical Analyses and Images Processing
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Burns, K.C. Are there general patterns in plant defense against megaherbivores? Biol. J. Linn. Soc. 2014, 111, 38–48. [Google Scholar] [CrossRef]
- Zhang, F.; Pascale, R.; Huang, T.; Wang, Y.; May, A.; Dupont, C.; Orbovic, V.; Irish, V.F. Reprogramming of stem cell activity to convert thorns into branches. Curr. Biol. 2020, 30, 2951–2961. [Google Scholar] [CrossRef] [PubMed]
- Mauseth, J.D. Structure–function relationships in highly modified shoots of Cactaceae. Ann. Bot. 2006, 98, 901–926. [Google Scholar] [CrossRef] [PubMed]
- Lev-Yadun, S. Aposematic (warning) coloration associated with thorns in higher plants. J. Theor. Biol. 2001, 210, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, J.H.C.; Lavorel, S.; Garnier, E.; Díaz, S.; Buchmann, N.; Gurvich, D.E.; Reich, P.B.; ter Steege, H.; Morgan, H.D.; van der Heijden, M.G.A.; et al. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- García-Mendoza, A.J. Revisión taxonómica del complejo Agave potatorum Zucc. (Agavaceae): Nuevos taxa y neotipificación. Acta Bot. Mex. 2010, 91, 71–93. [Google Scholar] [CrossRef]
- Davis, S.C.; Simpson, J.; Gil-Vega, K.C.; Niechayev, N.A.; Tongerlo, E.; Hurtado Castano, N.; Dever, L.V.; Búrquez, A. Undervalued potential of crassulacean acid metabolism for current and future agricultural production. J. Exp. Bot. 2019, 70, 6521–6537. [Google Scholar] [CrossRef]
- Gentry, H.S. Agaves of Continental North America; University of Arizona Press: Tucson, AZ, USA, 1982. [Google Scholar]
- Mora-López, J.L.; Reyes-Aguero, J.A.; Flores-Flores, J.L.; Peña-Valdivia, C.B.; Aguirre-Rivera, J.R. Morphological variation and humanization of Agave genus, salmianae section. Agrociencia 2011, 45, 465–477. [Google Scholar]
- Thiede, J. Agave Agavaceae. In Illustrated Handbook of Succulent Plants: Monocotyledons, 2nd ed.; Eggli, U., Nyffeler, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Malainine, M.E.; Dufresne, A.; Dupeyre, D.; Mahrouz, M.; Vuong, R.; Vignon, M.R. Structure and morphology of cladodes and spines of Opuntia ficus-indica. Cellulose extraction and characterization. Carbohydr. Polym. 2003, 51, 77–83. [Google Scholar] [CrossRef]
- Vignon, M.R.; Heux, L.; Malainine, M.-E.; Mahrouz, M. Arabinan-cellulose composite in Opuntia ficus-indica prickly pear spines. Carbohydr. Res. 2004, 339, 123–131. [Google Scholar] [CrossRef]
- Mosco, A. Micro-morphology and anatomy of Turbinicarpus (Cactaceae) spines. Rev. Mex. Biodivers. 2009, 80, 119–128. [Google Scholar]
- Reyes-Rivera, J.; Solano, E.; Terrazas, T.; Soto-Hernández, M.; Arias, S.; Almanza-Arjona, Y.C.; Polindara-García, L.A. Classification of lignocellulosic matrix of spines in Cactaceae by Py-GC/MS combined with omic tools and multivariate analysis: A chemotaxonomic approach. J. Anal. Appl. Pyrol. 2020, 148, 104796. [Google Scholar] [CrossRef]
- Colunga-GarcíaMarín, P.; May-Pat, F. Morphological variation of henequen (Agave fourcroydes, Agavaceae) germoplasm and its wild ancestor (A. angustifolia) under uniform growth conditions: Diversity and domestication. Am. J. Bot. 1997, 11, 1449–1465. [Google Scholar] [CrossRef]
- Vargas-Ponce, O.; Zizumbo-Villareal, D.; Marin, P.C.G. In situ diversity and maintenance of traditional Agave landraces used in spirits production in West-Central Mexico. Econ. Bot. 2007, 61, 362–375. [Google Scholar] [CrossRef]
- García-Mendoza, A. Los agaves de México. Ciencias. 2007, 87, 14–23. [Google Scholar]
- Porras-Ramírez, E.S.; Sosa-Marcos, Y.; Palma-Cruz, F.; Cisneros, A. Phenotyping genetic diversity of wild Agave species that coexist in the same spatial region. Notulae Botanicae Horti Agrobotanici 2016, 44, 640–648. [Google Scholar] [CrossRef]
- Trejo-Torres, J.; Gann, G.D.; Christenhusz, M.J.M. The Yucatan Peninsula is the place of origin of sisal (Agave sisalana, Asparagaceae): Historical accounts, phytogeography and current populations. Bot. Sci. 2018, 96, 366–379. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar Naik, P.; Patlan, S.; Chhokar, V. Assessment of genetic variation among Asparagus racemosus genotypes using molecular and biochemical markers. J. Med. Plants Stud. 2016, 6, 117–123. [Google Scholar]
- Pasha, S.; Khanam, S.; Afsar, Z. Isolation and characterization of chemical constituents of Asparagus racemosus as markers. Int. J. Res. Dev. Pharm Life Sci. 2016, 5, 2255–2263. [Google Scholar]
- Onlom, C.; Nuengchamnong, N.; Phrompittayarat, W.; Putalun, W.; Waranuch, N.; Ingkaninan, K. Quantification of saponins in Asparagus racemosus by HPLC-Q-TOF-MS/MS. Nat. Prod. Commun. 2017, 12, 7–10. [Google Scholar] [CrossRef]
- Lyczakowski, J.J.; Wicher, K.B.; Terrett, O.M.; Faria‑Blanc, N.; Yu, X.; Brown, D.; Krogh, K.B.R.M.; Dupree, P.; Busse‑Wicher, M. Removal of glucuronic acid from xylan is a strategy to improve the conversion of plant biomass to sugars for bioenergy. Biotechnol. Biofuels 2017, 10, 224. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Abad, A.; Berglund, J.; Toriz, G.; Gatenholm, P.; Henriksson, G.; Lindstrom, M.; Wohlert, J.; Vilaplana, F. Regular motifs in xylan modulate molecular flexibility and interactions with cellulose surfaces. Plant Physiol. 2017, 175, 1579–1592. [Google Scholar] [CrossRef]
- Martínez-Abad, A.; Jiménez-Quero, A.; Wohlert, J.; Vilaplana, F. Influence of the molecular motifs of mannan and xylan populations on their recalcitrance and organization in spruce softwoods. Green Chem. 2020, 22, 3956–3970. [Google Scholar] [CrossRef]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant. Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Voragen, A.G.J.; Coenen, G.-J.; Verhoef, R.P.; Schols, H.A. Pectin, a versatile polysaccharide present in plan cell walls. Struct. Chem. 2009, 20, 263–275. [Google Scholar] [CrossRef]
- Mitchell, V.D.; Taylor, C.M.; Bauer, S. Comprehensive analysis of monomeric phenolics in dilute acid plant hydrolysates. BioEnergy Res. 2014, 7, 654–669. [Google Scholar] [CrossRef]
- Pérez-Pimienta, J.A.; López-Ortega, M.G.; Sanchez, A. Recent developments in Agave performance as a drought-tolerant biofuel feedstock: Agronomics, characterization, and biorefining. Biofuels Bioprod. Biorefining 2017, 11, 732–748. [Google Scholar] [CrossRef]
- Delfín, R.M.E.; Calderón, S.M.; Ragazzo, S.J.A.; Gómez, R.J.; López, Z.L.; Aguilar, U.M.G. Acid pretreatment optimization for xylose production from Agave tequilana Weber var. azul, Agave americana var. oaxacensis, Agave karwinskii, and Agave potatorum bagasses using a Box-Behnken design. Biomass Conv. Biorefin. 2019, 1–10. [Google Scholar] [CrossRef]
- Del Rio, J.C.; Gutierrez, A.; Martınez, A.T. Identifying acetylated lignin units in non-wood fibers using pyrolysis-gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 1181–1185. [Google Scholar] [CrossRef]
- Martínez, Á.T.; Rencoret, J.; Marques, G.; Gutiérrez, A.; Ibarra, D.; Jiménez, B.J.; del Río, J.C. Monolignol acylation and lignin structure in some nonwoody plants: A 2D NMR study. Phytochemistry 2008, 69, 2831–2843. [Google Scholar] [CrossRef]
- José, C.; Prinsen, P.; Cadena, E.M.; Martínez, Á.T.; Gutiérrez, A.; Rencoret, J. Lignin–carbohydrate complexes from sisal (Agave sisalana) and abaca (Musa textilis): Chemical composition and structural modifications during the isolation process. Planta 2016, 243, 1143–1158. [Google Scholar]
- Zhou, L.; Li, D.; Wang, J.; Liu, Y.; Wu, J. Antibacterial phenolic compounds from the spines of Gleditsia sinensis Lam. Nat. Prod. Res. 2007, 21, 283–291. [Google Scholar] [CrossRef] [PubMed]
- López-Palacios, C.; Peña-Valdivia, C.B. Screening of secondary metabolites in cladodes to further decode the domestication process in the genus Opuntia (Cactaceae). Planta 2020, 251, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-H.; Hu, Y.-L.; Liu, T.-X. Plant distribution and pharmacological activity of flavonoids. Tradit. Med. Res. 2019, 4, 269–287. [Google Scholar]
- Ekalu, A.; Habila, J.D. Flavonoids: Isolation, characterization, and health benefits. Beni Suef Univ. J. Basic Appl. Sci. 2020, 45, 1–14. [Google Scholar] [CrossRef]
- Almaraz-Abarca, N.; Delgado-Alvardo, E.A.; Ávila-Reyes, J.A.; Uribe-Soto, J.N.; González-Valdez, L.S. The phenols of the genus Agave (Agavaceae). J. Biomater. Nanobiotechnol. 2013, 4, 9–16. [Google Scholar] [CrossRef]
- Puente-Garza, C.A.; García-Lara, S.; Gutiérrez-Uribe, J.A. Enchanced of saponins and flavonols by micropropagation of Agave salmiana. Ind. Crops Prod. 2017, 105, 225–230. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; El-Kammar, H.A.; Farag, M.A.; Saleh, D.O.; El Dine, R.S. Metabolomic profiling of five Agave leaf taxa via UHPLC/PDA/ESI-MS inrelation to their anti-inflammatory, immunomodulatory and ulceroprotective activities. Steroids 2020, 160, 108648. [Google Scholar] [CrossRef]
- Retamales, H.A.; Scharaschkin, T. A staining protocol for identifying secondary compounds in Myrtaceae. Appl. Plant. Sci. 2014, 10, 1400063. [Google Scholar] [CrossRef]
- Carvalho Ribeiro, V.; Espolador Leitao, C.A. Utilisation of Toluidine blue O pH 4.0 and histochemical inferences in plant sections obtained by free-hand. Protoplasma 2020, 272, 993–1008. [Google Scholar] [CrossRef]
- Pourcel, L.; Routaboul, J.M.; Kerhoas, L.; Caboche, M.; Lepiniec, L.; Debeaujon, I. TRANSPARENT TESTA10 encodes a laccase-like enzyme involved in oxidative polymerization of flavonoids in Arabidopsis seed coat. Plant Cell 2005, 17, 2966–2980. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.M.; Wong, K.K.Y.; Fernyhough, A. The laccase/ABTS system oxidizes (+)-catechin to oligomeric products. Enzyme Microb. Technol. 2007, 40, 1272–1279. [Google Scholar] [CrossRef]
- Hu, Q.; Luo, C.; Zhang, Q.; Luo, Z. Isolation and characterization of a Laccase gene potentially involved in proanthocyanidin polymerization in oriental persimmon (Diospyros kaki Thunb.) fruit. Mol. Biol. Rep. 2013, 40, 2809–2820. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.; Hu, K.; Tian, T.; Li, X.; Chen, L.; Zhang, Y.; Yi, B.; Wen, J.; Ma, C.; Shen, J.; et al. Transcriptomic analysis of seed coats in yellow-seeded Brassica napus reveals novel genes that influence proanthocyanidin biosynthesis. Front. Plant. Sci. 2017, 8, 1674. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.K.; and Kuhad, R.C. Laccase: Enzyme revisited and function redefined. Indian J. Microbiol. 2008, 48, 309–316. [Google Scholar] [CrossRef]
- Janusz, G.; Pawlik, A.; Świderska-Burek, U.; Polak, J.; Sulej, J.; Jarosz-Wilkołazka, A.; Paszczyński, A. Laccase Properties, Physiological Functions, and Evolution. Int. J. Mol. Sci. 2020, 21, 966. [Google Scholar] [CrossRef]
- García-Castillo, M.J.; Hernández-Zepeda, C.; Tamayo-Ordoñez, M.C.; Canche-Escamilla, G.; Duarte-Aranda, S.; Sánchez-Teyer, L.F. Expression analysis of CesA gene related to fiber production in Agave fourcroydes Lem. In Sustainable and Integrated Use of Agave; Gutiérrez-Mora, A., Ed.; Conacyt-Ciatej-Agared: Jalisco, Mexico, 2016; pp. 27–32. [Google Scholar]
- Morais de Carvalho, D.; Martínez-Abad, A.; Evtuguin, D.V.; Colodette, J.L.; Lindström, M.E.; Vilaplana, F.; Sevastyanova, O. Isolation and characterization of acetylated glucuronoxylan from sugarcane bagasse and straw. Carbohydr. Polym. 2017, 156, 223–234. [Google Scholar] [CrossRef]
- Sykes, R.; Yung, M.; Novaes, E.; Kirst, M.; Peter, G.; Davis, M. High-Throughput screening of plant cell-wall composition using pyrolysis molecular beam mass spectroscopy. In Biofuels: Methods and Protocols; Mielenz, J.R., Ed.; Humana Press: Totowa, NJ, USA, 2009. [Google Scholar]
- Infante-Rodríguez, D.A.; Monribot-Villanueva, J.L.; Mehltreter, K.; Carrión, G.L.; Lachaud, J.P.; Velázquez-Narváez, A.C.; Vásquez-Reyes, V.M.; Valenzuela-González, J.E.; Guerrero-Analco, J.A. Phytochemical characteristics of leaves determine foraging rate of the leaf-cutting ant Atta mexicana (Smith)(Hymenoptera: Formicidae). Chemoecology 2020, 30, 147–159. [Google Scholar] [CrossRef]
- Landa-Cansigno, C.; Hernández-Domínguez, E.E.; Monribot-Villanueva, J.L.; Licea-Navarro, A.F.; Mateo-Cid, L.E.; Segura-Cabrera, A.; Guerrero-Analco, J.A. Screening of Mexican tropical seaweeds as sources of α-amylase and α-glucosidase inhibitors. Algal Res. 2020, 49, 101954. [Google Scholar] [CrossRef]
- Smith, C.A.; O’Maille, G.; Want, E.J.; Qin, C.; Trauger, S.A.; Brandon, T.R.; Custodio, D.E.; Abagyan, R.; Siuzdak, G. METLIN: A metabolite mass spectral database. Ther. Drug Monit. 2005, 27, 747–751. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Andres-Lacueva, C.; Arita, M.; Kroon, P.; Manach, C.; Urpi-Sarda, M.; Wishart, D. Databases on food phytochemicals and their health-promoting effects. J. Agric. Food Chem. 2011, 2011 59, 4331–4348. [Google Scholar] [CrossRef]
- Horai, H.; Arita, M.; Kanaya, S.; Nihei, Y.; Ikeda, T.; Suwa, K.; Ojima, Y.; Tanaka, K.; Tanaka, S.; Aoshima, K.; et al. MassBank: A public repository for sharing mass spectral data for life sciences. J. Mass Spectrom. 2010, 45, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, H.; Alvarado, C.; Bouchet, B.; Kalthoum-Chérif, J.; Trabelsi-Ayadi, M.; Guyot, S. Tissue and cellular localization of tannins in tunisian dates (Phoenix dactylifera L.) by light and transmission electron microscopy. J. Agric. Food Chem. 2014, 62, 6650–6654. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Hu, X.; Li, C. Overexpression of the LoMYB29 gene of Larix olgensis contributes to the regulation of proanthocyanidin biosynthesis in Arabidopsis thaliana. J. For. Res. 2019, 30, 1793–1804. [Google Scholar] [CrossRef]






| Number | RT (min) | m/z | Formula | Ion/Adduct | Error (ppm) | Fragments | Candidate | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Positive mode | ||||||||||
| 1 | 2.37 | 291.0871 | C15H15O6+ | [M + H]+ | 2 | 165.055 | 147.0441 | 139.0391 | 123.0439 | (+)-Catechin |
| 2 | 3.64 | 633.1428 | C27H30O16Na+ | [M + Na]+ | 2 | 303.0502 | 257.0445 | 153.0185 | 137.0231 | Rutin |
| 3 | 4.13 | 577.1341 | C30H25O12+ | [M + H − H2O]+ | 2 | 425.0869 | 317.0659 | Epicatechin-gallocatechin | ||
| 4 | 4.41 | 559.1236 | C30H23O11+ | [M + H − H2O]+ | 0 | Proanthocyanidin | ||||
| 5 | 4.65 | 559.1235 | C30H23O11+ | [M + H − H2O]+ | 0 | Proanthocyanidin | ||||
| 6 | 4.86 | 559.1237 | C30H23O11+ | [M + H − H2O]+ | 0 | Proanthocyanidin | ||||
| 7 | 5.29 | 303.0504 | C15H11O7+ | [M + H]+ | 1 | 257.0444 | 229.0497 | 153.018 | 137.0233 | Quercetin |
| 8 | 6.089 | 287.0555 | C15H11O6+ | [M + H]+ | 1 | 241.0499 | 165.0181 | 153.0181 | 121.0282 | Kaempferol |
| Negative mode | ||||||||||
| 9 | 1.17 | 345.0829 | C14H17O10− | [M − H]− | 0 | Methyl 6-O-galloyl-beta-D-glucopyranoside | ||||
| 1 | 2.37 | 289.0717 | C15H13O6− | [M − H]− | 0 | 245.0814 | 203.0707 | 137.0237 | (+)-Catechin | |
| 10 | 2.54 | 771.2003 | C33H39O21− | [M − H]− | 2 | 609.1455 | 463.087 | 301.0339 | 151.0027 | Quercetin 3-rutinoside-7-galactoside |
| 11 | 2.85 | 289.0718 | C15H13O6− | [M − H]− | 0 | 245.0813 | 203.0705 | 137.0234 | (−)-Epicatechin | |
| 2 | 3.64 | 609.1458 | C27H29O16− | [M − H]− | 0 | 547.1238 | 331.0454 | 300.0263 | 151.0028 | Rutin |
| 12 | 3.95 | 243.0659 | C14H11O4− | [M − H]− | 1 | 225.0555 | 201.0552 | 183.0295 | 159.0447 | Suberenone |
| 4 | 4.41 | 557.1097 | C30H21O11− | [M − H − H2O]− | 2 | Proanthocyanidin | ||||
| 5 | 4.66 | 557.1092 | C30H21O11− | [M – H − H2O]− | 1 | Proanthocyanidin | ||||
| 6 | 4.87 | 557.1084 | C30H21O11− | [M − H − H2O]− | 0 | Proanthocyanidin | ||||
| 7 | 5.29 | 301.0351 | C15H9O7− | [M − H]− | 0 | 271.0244 | 227.0342 | 151.0031 | 121.0288 | Quercetin |
| 8 | 6.09 | 285.0406 | C15H9O6− | [M − H]− | 0 | 255.0298 | 229.0498 | 151.0031 | 93.0339 | Kaempferol |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morán-Velázquez, D.C.; Monribot-Villanueva, J.L.; Bourdon, M.; Tang, J.Z.; López-Rosas, I.; Maceda-López, L.F.; Villalpando-Aguilar, J.L.; Rodríguez-López, L.; Gauthier, A.; Trejo, L.; et al. Unravelling Chemical Composition of Agave Spines: News from Agave fourcroydes Lem. Plants 2020, 9, 1642. https://doi.org/10.3390/plants9121642
Morán-Velázquez DC, Monribot-Villanueva JL, Bourdon M, Tang JZ, López-Rosas I, Maceda-López LF, Villalpando-Aguilar JL, Rodríguez-López L, Gauthier A, Trejo L, et al. Unravelling Chemical Composition of Agave Spines: News from Agave fourcroydes Lem. Plants. 2020; 9(12):1642. https://doi.org/10.3390/plants9121642
Chicago/Turabian StyleMorán-Velázquez, Dalia C., Juan L. Monribot-Villanueva, Matthieu Bourdon, John Z. Tang, Itzel López-Rosas, Luis F. Maceda-López, José L. Villalpando-Aguilar, Lorena Rodríguez-López, Adrien Gauthier, Laura Trejo, and et al. 2020. "Unravelling Chemical Composition of Agave Spines: News from Agave fourcroydes Lem." Plants 9, no. 12: 1642. https://doi.org/10.3390/plants9121642
APA StyleMorán-Velázquez, D. C., Monribot-Villanueva, J. L., Bourdon, M., Tang, J. Z., López-Rosas, I., Maceda-López, L. F., Villalpando-Aguilar, J. L., Rodríguez-López, L., Gauthier, A., Trejo, L., Azadi, P., Vilaplana, F., Guerrero-Analco, J. A., & Alatorre-Cobos, F. (2020). Unravelling Chemical Composition of Agave Spines: News from Agave fourcroydes Lem. Plants, 9(12), 1642. https://doi.org/10.3390/plants9121642