Effect of Long-Term of He-Ne Laser Light Irradiation on Selected Physiological Processes of Triticale
Abstract
:1. Introduction
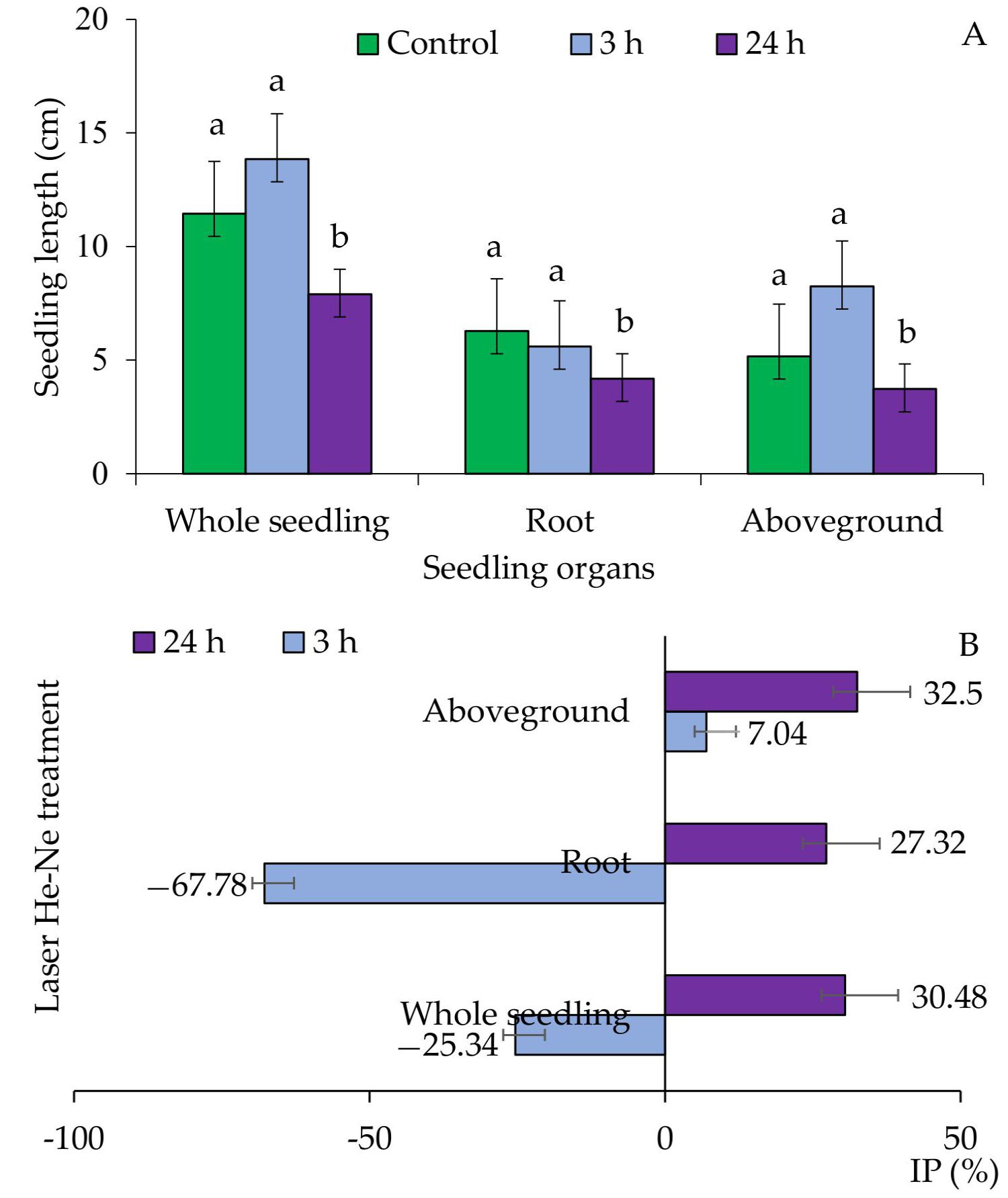
2. Results
3. Discussion
4. Materials and Methods
4.1. Grains Irradiation
4.2. Germination Condition
4.3. Germination Parameters
4.4. Plant Growth Conditions
4.5. Biometric Analysis
4.6. Fresh and Dry Mass and Tissue Water Content
4.7. Chlorophyll a Fluorescence
4.8. Chlorophyll Content
4.9. Electrolyte Leakage
4.10. Emission Fluorescence
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brugnoli, E.; Björkman, O. Chloroplast movements in leaves—influence on chlorophyll fluorescence and measurements of light-induced absorbency changes related to delta-ph and zeaxanthin formation. Photosynth. Res. 1992, 32, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Carvalhoab, S.D.; Foltaa, K.M. Environmentally modified organisms—expanding genetic potential with light. Crit. Rev. Plant Sci. 2014, 33, 486–508. [Google Scholar] [CrossRef]
- Young, J.E. Effects of spectral composition of light on the growth of a higher plant. In Light as an Ecological Factor; Evans, G.C., Bainbridge, R., Rackham, O., Eds.; Blackwell: Oxford, UK, 1975; pp. 135–159. [Google Scholar]
- Théry, M. Forest light and its influence on habitat selection. Plant Ecol. 2001, 153, 251–261. [Google Scholar] [CrossRef]
- Elemans, M. Light, nutrients and the growth of herbaceous forest species. Acta Oecolo. 2004, 26, 197–202. [Google Scholar] [CrossRef]
- Ellenberg, H.; Weber, H.; Dull, R.; Wirth, V.; Werner, W.; Paulissen, D. Zegerverte von Pflanzen in Mitteleuropa. Scr. Geobot. 1992, 18, 1–258. [Google Scholar]
- Mathiassen, S.K.; Bak, T.; Christensen, S.; Kudsk, P. The effect of laser treatment as a weed control method. Biosyst. Eng. 2006, 94, 497–505. [Google Scholar] [CrossRef] [Green Version]
- Muszyński, S.; Gładyszewska, B. Representation of He-Ne laser irradiation effect on radish seeds with selected germination indices. Int. Agrophys. 2008, 22, 151–157. [Google Scholar]
- Klimont, K. The influence of the laser light on yield and seed quality of amaranth (Amaranthus cruentus L.). Biul. IHAR 2002, 223, 249–255. [Google Scholar]
- Dziwulska, A.; Koper, R. Effect of pre-sowing laser biostimulation on germination of lucerne seeds. Acta Agrophys. 2003, 82, 33–39. [Google Scholar]
- Dziwulska, A.; Koper, R.; Wilczek, M. Estimation of effect of He-Ne laser beam on germination capacity of white clover variety of Anda cultivar. Acta Agrophys. 2004, 3, 435–441. [Google Scholar]
- Chen, Y.-P.; Yue, M.; Wang, X.-L. Influence of He–Ne laser irradiation on seeds thermodynamic parameters and seedlings growth of Isatis indogotica. Plant Sci. 2005, 168, 601–606. [Google Scholar] [CrossRef]
- Gładyszewska, B.; Koper, R.; Kornaczyński, K. Technology and effects of pre-sowing laser biostimulation of cucumber seeds. Zesz. Probl. Post. Nauk Rol. 1998, 454, 213–219. [Google Scholar]
- Hernández, A.C.; Carballo, C.A.; Artola, A.; Michtchenko, A. Laser irradiation effects on maize seed field performance. Seed Sci. Technol. 2006, 4, 193–197. [Google Scholar] [CrossRef]
- Michalik, B. Plant Breeding; Państwowe Wydawnictwo Rolnicze i Leśne: Poznań, Poland, 2009; pp. 264–266. ISBN 978-83-09-01056-2. [Google Scholar]
- Qiu, Z.B.; Li, J.T.; Yue, M. The damage repair role of He-Ne laser on wheat exposed to osmotic stress. Can. J. Plant Sci. 2010, 90, 691–698. [Google Scholar] [CrossRef]
- Dziwulska, A.; Wilczek, M.; Wintal, M. Effect of laser stimulation on crop yield of alfalfa and hybrid alfalfa studied in years of full land use. Acta Agrophys. 2006, 7, 327–336. [Google Scholar]
- Khalifa, N.S.; Ghandoor, H.E. Short term effect of he-ne laser on Jatropha curcas leaf development and amino acid biosynthesis: More insights in developmental genetics of oil producing plants. In Proceedings of the 7th International Conference “Plant & Microbial Biotechnology & Their Role in the Development of the Society”, Tanta, Egypt, 19–20 April 2017; pp. 119–132. [Google Scholar]
- Smith, H. Light quality, photoreception, and plant strategy. Annu. Rev. Plant Physiol. 1982, 33, 481–518. [Google Scholar] [CrossRef]
- Steyn, W.J.; Wand, S.J.E.; Holcroft, D.M.; Jacobs, G. Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef]
- Albert, N.W.; Lewis, D.H.; Zhang, H.; Irving, L.J.; Jameson, P.E.; Davies, K.M. Light-induced vegetative anthocyanin pigmentation in Petunia. J. Exp. Bot. 2009, 60, 2191–2202. [Google Scholar] [CrossRef] [Green Version]
- Zivack, M.; Brestic, M.; Kalaji, H.M. Govindjee Photosynthetic responses of sun- and shade-grown barley leaves to high light: Is the lower PSII connectivity in shade leaves associated with protection against excess of light? Photosynth. Res. 2014, 119, 339–354. [Google Scholar] [CrossRef] [Green Version]
- Frank, J.; Sterck, F.J.; Duursma, R.W.; Valladares, F.; Cieslak, M.; Weemstra, M. Plasticity influencing the light compensation point offsets the sspecialization for light niches across shrub species in a tropical forest understorey. J. Ecol. 2013, 101, 971–980. [Google Scholar] [CrossRef] [Green Version]
- Klimont, K. Wpływ światła lasera na plon roślin i wartość siewną fasoli (Phaseolus vulgaris L.) i grochu siewnego (Pisum sativum L.). Biul. IHAR 2001, 217, 263–277. [Google Scholar]
- Kreslavski, V.D.; Shirshikova, G.N.; Lyubimov, V.Y.; Shmarev, A.N.; Boutanaev, A.M.; Kosobryukhov, A.A.; Schmitt, F.J.; Friedrich, T.; Allakhverdiev, S.I. Effect of preillumination with red light on photosynthetic parameters and oxidant-/antioxidant balance in Arabidopsis thaliana in response to UV-A. J. Photochem. Photobiol. B Biol. 2013, 127, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Dudareva, L.V.; Lankevich, S.V.; Sumtsova, V.M.; Rudikovcka, E.G.; Salyaev, R.K. Possible paths of the impact oflow-intensity laser radiation on membrane structures in plant cells. Annu. Wheat Newsl. 2007, 53, 70–72. [Google Scholar]
- Dinoev, S.; Antonov, M.; Stoyanov, T.; Georgieva, C. Spectral impact of low-power laser radiation on wheat and maize parameters. Bulg. Acad. Sci. Probl. Eng. Cybern. Robot. Sofia 2004, 54, 74–85. [Google Scholar]
- Ciupak, A.; Szczurowska, I.; Gadyszewska, B.; Pietruszewski, S. Impact of laser light and magnetic field stimulation on the process of buckwheat seed germination. Tech. Sci. 2007, 10, 1–10. [Google Scholar] [CrossRef]
- Flores, M.; Carbonell, M.V.; Martinez, E. Exposure of maize seeds to stationary magnetic fields: Effects of germination and early growth. Environ. Exp. Bot. 2007, 59, 68–75. [Google Scholar] [CrossRef]
- Qiu, Z.B.; Li, J.T.; Zhang, M.M.; Bi, Z.Z.; Li, Z.L. He-Ne laser pretreatment protects wheat seedlings against cadmium-induced oxidative stress. Ecotoxicol. Environ. Saf. 2013, 88, 135–141. [Google Scholar] [CrossRef]
- Govil, S.R.; Agrawal, D.C.; Rai, K.P.; Thakur, S.N. Growth responses of Vigna radiata seeds to laser irradiation in the UV-A region. Physiol. Plant. 2006, 63, 133–134. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, J.Z.; Hang, T.; Li, P.P. Photosynthetic characteristics and growth performance of lettuce (Lactuca sativa L.) under different light/dark cycles in mini plant factories. Photosynthetica 2020, 58, 740–747. [Google Scholar] [CrossRef] [Green Version]
- Vasilevski, G. Perspectives of the application of biophysical methods in sustainable agriculture. Bulg. J. Plant Physiol. 2003, 29, 179–186. [Google Scholar]
- Danie, R.M. The upper limits of enzyme thermal stability. Enzyme Microb. Technol. 1996, 19, 74–79. [Google Scholar] [CrossRef]
- Wilczek, M.; Ćwintal, M.; Kornas-Czuwar, B.; Koper, R. Influence of pre-sowing laser stimulation and microelement nutrition (B, Mo) on yielding of seed of tetraploid red clover during four-year performance. Acta Agrophys. 2006, 8, 527–536. [Google Scholar]
- Qi, Z.; Yue, M.; Wang, X.L. Laser pretreatment protects cells of broad bean from UV-B radiation damage. J. Photochem. Photobiol. B Biol. 2002, 59, 33–37. [Google Scholar] [CrossRef]
- Podleśny, J.; Podleśna, A. Morphological changes and yield of selected species of leguminous plants under the influence of seed treatment with laser light. Int. Agrophys. 2004, 18, 253–260. [Google Scholar]
- Perveen, R.; Jamil, Y.; Ashraf, M.; Ali, Q.; Iqbal, M.; Ahmad, M.R. He-Ne laser-induced improvement in biochemical, physiological, growth and yield characteristics in sunflower (Helianthus annuus L.). Photochem. Photobiol. 2011, 87, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Wilczek, M.; Fordoński, G. Influence of pre-sowing laser stimulation of seeds on photosynthesis and transpiration intensity and on yielding of red clover. Acta Agrophys. 2007, 9, 517–524. [Google Scholar]
- Kalaji, M.H.; Carpentier, R.; Allakhverdiev, S.I.; Bosa, K. Fluorescence parameters as early indicators of light stress in barley. J. Photochem. Photobiol. B Biol. 2012, 112, 1–6. [Google Scholar] [CrossRef]
- Kalaji, M.H.; Goltsev, V.; Bosa, K.; Allakhverdiev, S.I.; Strasser, R.J. Govindjee Experimental in vivo measurements of light emission in plants: A perspective dedicated to David Walker. Photosynth. Res. 2012, 114, 69–96. [Google Scholar] [CrossRef]
- Chen, H.; Han, R. He-Ne laser treatment improves the photosynthetic efficiency of wheat exposed to enhanced UV-B radiation. Laser Phys. 2014, 24, 105602. [Google Scholar] [CrossRef] [Green Version]
- Cholakov, D.; Petkova, V. Morphological and physiological characteristics of seedlings caused by laser and gamma irradiation of cucumber seeds. Acta Hortic. 2002, 579, 285–287. [Google Scholar] [CrossRef]
- Ruban, A.X. Plants in light. Commun. Integr. Biol. 2009, 2, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Enriquez, S.; Sand-Jensen, K. Variation in light absorption properties of Mentha aquatica L. as a function of leaf form: Implications for plant growth. Int. J. Plant Sci. 2003, 164, 125–136. [Google Scholar] [CrossRef]
- Rybiński, W.; Garczyński, S. Influence of laser light on leaf area and parameters of photosynthetic activity in DH lines of spring barley (Hordeum vulgare L.). Int. Agrophys. 2004, 18, 261–267. [Google Scholar]
- Kalaji, M.H.; Pietkiewicz, S. Some physiological indices to be exploited as a crucial tool in plant breeding. Plant Breed. Seed Sci. 2004, 49, 19–39. [Google Scholar]
- Zhu, X.G.; Govindjee; Baker, N.; deSturler, E.; Ort, D.; Long, S. Chlorophyll a fluorescence induction kinetics in leaves predicted from a model describing each discrete step of excitation energy and electron transfer associated with photosystem II. Planta 2005, 223, 114–133. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Schansker, G.; Ladle, R.J.; Goltsev, V.; Bosa, K.; Allakhverdiev, S.I.; Brestic, M.; Bussotti, F.; Calatayud, A.; Dąbrowski, P.; et al. Frequently asked questions about in vivo chlorophyll fluorescence: Practical issues. Photosynth. Res. 2014, 122, 121–158. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Zhang, T.-J.; Chow, W.S.; Xie, X.; Chen, Y.J.; Peng, C.L. Photosynthetic characteristics and light energy conversions under different light environments in five tree species occupying dominant status at different stages of subtropical forest succession. Funct. Plant Biol. 2015, 42, 609–619. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The fluorescence transient as a tool to characterise and screen photosynthetic samples. In Probing Photosynthesis: Mechanisms, Regulation and Adaptation; Yunus, M., Pathre, U., Mohanty, P., Eds.; Taylor and Francis: London, UK, 2000; pp. 445–483. [Google Scholar]
- Chen, Y.-P. Isatis indigotica seedlings derived from laser stimulated seeds showed improved resistance to elevated UV-B. Plant Growth Regul. 2008, 55, 73–79. [Google Scholar] [CrossRef]
- Holt, N.E.; Zigmantas, D.; Valkunas, L.; Li, X.P.; Niyogi, K.K.; Fleming, G.R. Carotenoid cation formation and the regulation of photosynthetic light harvesting. Science 2005, 307, 433–436. [Google Scholar] [CrossRef] [Green Version]
- Campos, M.D.; Nogales, A.; Cardoso, H.G.; Campos, C.; Grzebelus, D.; Velada, I.; Arnholdt-Schmitt, B. Carrot plastid terminal oxidase gene (DcPTOX) responds early to chilling and harbors intronic pre-mirnas related to plant disease defense. Plant Gene 2016, 7, 21–25. [Google Scholar] [CrossRef]
- Ramel, F.; Birtic, S.; Cuiné, S.; Triantaphylidès, C.; Ravant, J.-L.; Havaux, M. Chemical quenching of singlet oxygen by carotenoids in plants. Plant Physiol. 2012, 158, 1267–1278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santabarbara, S.; Casazza, A.P.; Ali, K.; Economou, C.K.; Wannathong, T.; Zito, F.; Redding, K.E.; Rappaport, F.; Purton, S. The requirement for carotenoids in the assembly and function of the photosynthetic complexes in Chlamydomonas reinhardtii. Plant Physiol. 2013, 161, 535–546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demmig-Adams, B.; Gilmore, A.M.; Adams, W.W., III. In vivo functions of carotenoids in higher plants. FASEB J. 1996, 10, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Janků, M.; Luhová, L.; Petřivalský, M. On the origin and fate of reactive oxygen species in plant cell compartments. Antioxidants 2019, 8, 105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duderova, L.; Tarasenko, V.; Rudikovskaya, E. Involvement of photoprotective compounds of a phenolic nature in the response of Arabidopsis thaliana leaf tissues to low-intensity laser radiation. J. Photochem. Photobiol. 2020, 96, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Król, A.; Amarowicz, R.; Weidner, S. Changes in the composition of phenolic compounds and antioxidant properties of grapevine roots and leaves (Vitis vinifera L.) under continuous of long-term drought stress. Acta Physiol. Plant. 2014, 36, 1491–1499. [Google Scholar] [CrossRef] [Green Version]
- Mazur, A. The role of seed coat in the germination and early stages of growth of bean (Phaseolus vulgaris L.) in the presence of chickweed (Stellaria media (L.) Vill.). AUPC Stud. Nat. 2019, 4, 103–118. [Google Scholar] [CrossRef]
- AOSA (Association of Official Seed Analysis). Seed vigour testing. In Handbook on Seed Testing; Contribution No. 32; Association of Official Seed Analysis: Ithaca, NY, USA, 1983. [Google Scholar]
- Islam, A.K.M.A.; Anuar, N.; Yaakob, Z. Effect of genotypes and pre-sowing treatments on seed germination behavior of Jatropha. Asian J. Plant Sci. 2009, 8, 433–439. [Google Scholar] [CrossRef]
- Chiapusio, G.; Sanchez, A.M.; Reigosa, M.J.; Gonzalez, L.; Pellissier, F. Do germination indices adequately reflect allelochemical effects on the germination process? J. Chem. Ecol. 1997, 23, 2445–2453. [Google Scholar] [CrossRef]
- Farooq, M.; Basra, S.M.A.; Afzal, I.; Khaliq, A. Optimization of hydropriming techniques for rice seed invigoration. Seed Sci. Technol. 2006, 34, 507–512. [Google Scholar] [CrossRef]
- Steiner, A.A. A universal method for preparing nutrient solutions of a certain desired composition. Plant Soil 1961, 15, 134–154. [Google Scholar] [CrossRef] [Green Version]
- Mominul Islam, A.K.M.; Kato-Noguchi, H. Allelopathic potentiality of medicinal plant Leucas aspera. Int. J. Sustain. Agric. 2012, 4, 1–7. [Google Scholar]
- Lichtenthaler, H.K.; Buschmann, C.; Knapp, M. Measurement of chlorophyll fluorescence kinetics (Kautsky effect) and the chlorophyll fluorescence decrease ratio (RFd–values) with the PAM–Fluorometer. In Analytical Methods in Plant Stress Biology; Filek, N., Biesaga-Kościelniak, J., Marcińska, I., Eds.; The Franciszek Gorski Institute of Plant Physiology of the Polish Academy of Sciences: Kraków, Poland, 2004; pp. 93–111. [Google Scholar]
- Barnes, J.D.; Balaguer, L.; Manrique, E.; Elvira, S.; Davison, A.W. A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environ. Exp. Bot. 1992, 32, 83–100. [Google Scholar] [CrossRef]
- Możdżeń, K.; Barabasz-Krasny, B.; Zandi, P.; Turisová, I. Influence of allelopathic activity of Galinsoga parviflora Cav. and Oxalis fontana Bunge on the early growth stages of cultivars Raphanus sativus L. var. radicula Pers. Biologia 2018, 73, 1187–1195. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Knapp, M.; Buschmann, C. Recording chlorophyll fluorescence emission spectra with the Perkin Elmer fluorescence spectrometer LS 50. In Analytical Methods in Plant Stress Biology; Filek, N., Biesaga-Kościelniak, J., Marcińska, I., Eds.; The Franciszek Gorski Institute of Plant Physiology of the Polish Academy of Sciences: Kraków, Poland, 2004; pp. 112–124. [Google Scholar]
- Jena, S.; Acharya, S.; Mohapatra, P.K. Variation in effects of four OP insecticides on photosynthetic pigment fluorescence of Chlorella vulgaris Beij. Ecotoxicol. Environ. Saf. 2012, 80, 111–117. [Google Scholar] [CrossRef] [PubMed]

| Treatment (h) | Day (24 h) | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | |
| Control | 12.0 b ±1.03 | 36.0 ab ±1.19 | 56.8 ab ±0.89 | 80.8 a ±1.66 | 89.6 a ±1.18 | 100.0 a ±0.76 | 100.0 a ±1.35 |
| 3 | 20.8 a ±0.84 | 50.4 a ±0.79 | 70.4 a ±1.14 | 88.0 a ±1.22 | 91.2 a ±1.10 | 94.4 a ±0.55 | 97.6 a ±0.55 |
| 24 | 7.2 b ±0.84 | 16.8 b ±0.84 | 36.8 b ±0.84 | 57.6 b ±1.14 | 65.6 ab ±1.14 | 69.6 b ±0.84 | 72.8 ab ±1.00 |
| Treatment (h) | SE | GI | CRG | SVI | T50 |
|---|---|---|---|---|---|
| Control | 12.00 b ±2.83 | 23.63 b ±1.54 | 18.40 b ±0.24 | 11.44 b ±2.06 | 0.46 a ±0.001 |
| 3 | 21.17 a ±3.57 | 37.92 a ±1.25 | 19.24 a ±0.25 | 13.60 a ±1.96 | 0.46 a ±0.001 |
| 24 | 9.50 b ±4.62 | 21.72 c ±1.57 | 17.93 c ±0.23 | 6.04 c ±1.47 | 0.44 b ±0.003 |
| Treatment (h) | FM | DM | TWC |
|---|---|---|---|
| 0 | 214 a ±0.04 | 33 b ± 0.003 | 84.39 a ±2.02 |
| 3 | 235 a ±0.06 | 45 a ±0.003 | 80.87 ab ±3.55 |
| 24 | 192 b ±0.05 | 44 a ±0.005 | 76.41 b ±4.94 |
| F | 1.209 | 4.699 | 5.836 |
| * p | 0.332 | 0.031 * | 0.017 * |
| Treatment (h) | F450/F535 | F450/F685 | F450/F735 | F685/F735 | PSIIA/C | PSIA/C | PSI/PSII |
|---|---|---|---|---|---|---|---|
| 0 | 1.12 a ±0.01 | 1.44 a ±0.21 | 3.18 a ±0.34 | 2.23 a ±0.20 | 1.00 a ±0.07 | 1.45 a ±0.01 | 0.69 a ±0.05 |
| 3 | 1.08 a ±0.04 | 1.33 a ±0.28 | 2.34 ab ±0.35 | 1.78 b ±0.17 | 0.95 a ±0.05 | 1.53 a ±0.04 | 0.62 a ±0.03 |
| 24 | 1.03 a ±0.06 | 1.75 a ±0.55 | 3.37 a ±1.08 | 1.93 a ±0.11 | 0.96 a ±0.03 | 1.48 a ±0.08 | 0.65 a ±0.03 |
| F | 3.598 | 0.992 | 1.906 | 5.650 | 0.757 | 1.670 | 2.345 |
| * p | 0.094 | 0.424 | 0.229 | 0.042 * | 0.509 | 0.265 | 0.177 |
| Treatment (h) | F0 | Fm | Fv | Fv/Fm | Fv/F0 | ChlSPAD Total | Chl a | Chl b |
|---|---|---|---|---|---|---|---|---|
| (mg × g–1) | ||||||||
| 0 | 175.2 a ±34.33 | 879.4 a ±12.86 | 704.2 a ±9.61 | 0.802 a ±0.013 | 4.06 a ±0.33 | 15.44 b ±6.83 | 1.20 a ±0.31 | 0.50 a ±0.10 |
| 3 | 165.0 ab ±17.65 | 804.2 a ±17.27 | 639.2 a ±5.57 | 0.795 a ±0.006 | 3.88 a ±0.13 | 21.20 a ±8.07 | 1.40 a ±0.48 | 0.58 a ±0.15 |
| 24 | 189.0 a ±33.43 | 937.4 a ±15.28 | 748.4 a ±11.99 | 0.799 a ±0.006 | 3.97 a ±0.15 | 15.96 b ±5.01 | 1.03 ab ±0.58 | 0.40 ab ±0.25 |
| F | 0.834 | 1.481 | 1.692 | 0.679 | 0.802 | 2.989 | 0.925 | 1.249 |
| * p | 0.458 | 0.266 | 0.225 | 0.525 | 0.471 | 0.046 * | 0.423 | 0.322 |
| Treatment (h) | Plant Part | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Root | Stem | Leaves | Remaining Part of the Shoot | |||||||||||
| I | II | III | IV | |||||||||||
| (cm) | IP(%) | (cm) | IP(%) | (cm) | IP(%) | (cm) | IP(%) | (cm) | IP(%) | (cm) | IP(%) | (cm) | IP(%) | |
| 0 | 27.78 ab ±6.81 | 5.56 a ±1.03 | 12.14 a ±1.82 | 24.68 a ±2.13 | 26.66 a ±2.53 | 16.04 a ±5.53 | 10.78 a ±6.78 | |||||||
| 3 | 29.22 ab ±9.83 | −13.10 | 5.50 a ±1.15 | −1.74 | 10.26 ab ±3.40 | 11.21 | 20.32 b ±2.61 | 17.70 | 26.84 a ±2.64 | −1.08 | 17.96 a ±3.38 | −26.95 | 9.88 a ±1.72 | −39.75 |
| 24 | 38.66 a ±14.61 | −52.02 | 5.30 a ±0.68 | 0.91 | 11.58 ab ±2.64 | 1.77 | 25.60 a ±1.32 | −4.08 | 27.62 a ±3.44 | −3.79 | 19.12 a ±5.46 | −32.85 | 10.80 a ±4.98 | −58.9 |
| F | 1.469 | 0.098 | 0.641 | 9.086 | 0.155 | 0.505 | 0.056 | |||||||
| * p | 0.269 | 0.908 | 0.544 | 0.004 * | 0.858 | 0.616 | 0.946 | |||||||
| Treatment (h) | Plant Part | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Root | Stem | Leaves | Remaining Part of the Shoot | |||||||||||
| I | II | III | IV | |||||||||||
| A | B | A | B | A | B | A | B | A | B | A | B | A | B | |
| 0 | 428 ab ±0.163 | 49 b ±0.006 | 87 ab ±0.015 | 15 a ±0.002 | 53 a ±0.008 | 5 a ±0.001 | 81 a ±0.019 | 9 ab ±0.002 | 85 ab ±0.012 | 12 b ±0.002 | 50 abc ±0.019 | 8 ab ±0.003 | 91 a ±0.047 | 14 ab ±0.007 |
| 3 | 358 ab ±0.073 | 44 b ±0.012 | 94 a ±0.030 | 15 a ±0.004 | 36 ab ±0.020 | 5 a ±0.003 | 75 ab ±0.016 | 7 b ±0.004 | 92 ab ±0.017 | 13 ab ±0.002 | 61 ab ±0.019 | 11 a ±0.003 | 78 ab ±0.017 | 18 a ±0.013 |
| 24 | 514 a ±0.185 | 76 a ±0.027 | 109 a ±0.015 | 17 a ±0.003 | 45 a ±0.020 | 6 a ±0.001 | 98 a ±0.010 | 13 a ±0.001 | 113 a ±0.022 | 16 a ±0.002 | 84 a ±0.037 | 14 a ±0.005 | 108 a ±0.044 | 17 a ±0.007 |
| F | 1.382 | 4.634 | 1.422 | 0.648 | 1.164 | 0.037 | 2.844 | 5.763 | 3.524 | 6.184 | 2.111 | 3.503 | 0.736 | 0.324 |
| * p | 0.288 | 0.032 * | 0.279 | 0.541 | 0.345 | 0.964 | 0.097 | 0.018 * | 0.063 | 0.014 * | 0.164 | 0.063 | 0.499 | 0.729 |
| Treatment (h) | Plant Part | ||||||
|---|---|---|---|---|---|---|---|
| Root | Stem | Leaves | Remainder Part of the Shoot | ||||
| I | II | III | IV | ||||
| 0 | 87.48 a ±3.42 | 83.24 a ±0.90 | 89.76 a ±0.40 | 89.28 a ±1.12 | 85.88 a ±5.12 | 83.41 a ±1.71 | 85.32 a ±1.09 |
| 3 | 87.68 a ±1.70 | 83.50 a ±1.74 | 81.79 a ±1.28 | 91.09 a ±4.08 | 86.29 a ±2.23 | 81.27 a ±1.85 | 76.90 ab ±1.41 |
| 24 | 84.97 a ±4.04 | 84.77 a ±0.70 | 83.07 a ±1.29 | 86.67 ab ±1.29 | 85.35 a ±0.92 | 82.34 a ±1.68 | 84.41 a ±0.89 |
| F | 1.105 | 2.323 | 0.810 | 3.782 | 0.103 | 1.860 | 1.428 |
| p | 0.363 | 0.140 | 0.468 | 0.053 | 0.903 | 0.198 | 0.278 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Możdżeń, K.; Barabasz-Krasny, B.; Zandi, P. Effect of Long-Term of He-Ne Laser Light Irradiation on Selected Physiological Processes of Triticale. Plants 2020, 9, 1703. https://doi.org/10.3390/plants9121703
Możdżeń K, Barabasz-Krasny B, Zandi P. Effect of Long-Term of He-Ne Laser Light Irradiation on Selected Physiological Processes of Triticale. Plants. 2020; 9(12):1703. https://doi.org/10.3390/plants9121703
Chicago/Turabian StyleMożdżeń, Katarzyna, Beata Barabasz-Krasny, and Peiman Zandi. 2020. "Effect of Long-Term of He-Ne Laser Light Irradiation on Selected Physiological Processes of Triticale" Plants 9, no. 12: 1703. https://doi.org/10.3390/plants9121703






