1. Introduction
Plant-parasitic nematodes (PPNs) are pervasive pests of a multitude of economically important crop plants, with an impact resulting in economic damage of over
$100 billion a year due to yield losses. Of these, root-knot nematodes (RKNs;
Meloidogyne spp.) are the most damaging PPNs as they can infest a broad range of horticultural and agricultural crop plants [
1,
2].
Meloidogyne spp. (Nematoda: Tylenchida: Meloidogynidae) are soil-borne pathogens infesting roots and causing deformations—the characteristic root galls, limiting the uptake of water and nutrients by the plant and thus causing plant stress [
3]. Furthermore, some RKN species, such as
Meloidogyne luci, have an unresolved taxonomic status, which can hinder official regulatory action against this pest [
4,
5]. Chemical nematicides are usually the principal control option against PPNs. Historically, fumigant, carbamate or organophosphate nematicides have been most widely used. However, they often pose unacceptable environmental and human health concerns [
6]. Many older nematicide types are being replaced by a newer generation of chemicals like fluopyram, secondary metabolites from bacteria, such as avermectins, and various biological agents. Microbiological bionematicides include various fungi (
Pochonia chlamydosporium,
Myrothecium verrucaria,
Purpureocillium lilacinus,
Trichoderma spp. and
Metarhizium spp.) and bacteria (
Pasteuria spp. and
Bacillus spp.). Such bionematicides also represent an important facet of environmentally conscious strategies for pest management. Of these, rhizobacteria
Bacillus firmus (Bacillales: Bacillaceae) are one of the most widely used control agents in bionematicide preparations [
7,
8].
Apart from being utilised in the agricultural sector for crop protection,
B. firmus is gaining importance for biotechnological applications, such as the production of bioactive compounds, food and feed additives, and expression of industrial enzymes [
9]. It has been suggested that
B. firmus’ nematicidal properties are due to various secondary metabolites, enzymes and toxins causing nematode mortality or suppressing nematode reproduction, egg-hatching and juvenile survival [
7,
10,
11,
12].
Bacillus proteinases, however, are the only nematode-virulence factor to have been confirmed experimentally [
10,
11]. Isolated from cultivated soil in Israel, isolate
B. firmus I-1582 has been used commercially as a bionematicide since the early 2000s. Strain I-1582 significantly reduced infestation of different RKN species on tomatoes (
Solanum lycopersicum L.) and cucumbers (
Cucumis sativus L.) in pot, greenhouse and field experiments [
13,
14,
15,
16]; it induced paralysis and mortality of the RKN
M. incognita, as well as the burrowing nematode
Radopholus similis and stem nematode
Ditylenchus dipsaci [
17], and reduced sting nematode
Belonolaimus longicaudatus infestation on bermudagrass (
Cynodon spp.) [
18], while at the same time increasing plant height and yield [
19], and also showing the ability of I-1582 to promote plant growth. Many
Bacillus spp. have previously been observed to promote plant growth [
20].
B. firmus bacteria were shown to induce biochemical changes in nematode-parasitised eggplant (
Solanum melongena L.), aiding stress alleviation [
21], while
B. firmus SW5 was shown to alleviate salt-induced stress in soybean (
Glycine max L.) in various ways, including the promotion of biomass yield, chlorophyll synthesis and nutrient uptake, as well as influencing the antioxidant defence systems and stress-responsive genes [
22].
Plant growth-promoting rhizobacteria (PGPR), such as various bacilli, are rhizosphere-inhabiting bacteria associated with plant roots. PGPR promote plant growth through direct and/or indirect mechanisms. Direct mechanisms include the production and/or metabolism of plant growth regulators (plant hormones) by PGPR and facilitating the uptake of nutrients to the plant (nitrogen fixation and phosphate solubilisation). Indirect mechanisms include the production of siderophores, pathogen-inhibiting factors (cell-wall degrading enzymes and antibiotics) and induced systemic resistance (ISR), eliciting plant host defences by PGPR [
20]. The multimodal action of some PGPR—pest control and the promotion of plant growth—can be harnessed in the context of sustainable, environmentally friendly agricultural practices. Apart from a decreased need for chemical pesticides, PGPR can promote more efficient and improved nutrient uptake to crop plants, thus enabling enhanced fertiliser use in agricultural production systems. The long-term overuse of chemical fertilisers to improve productivity often leads to wider negative environmental impact due to nitrate leaching into ground water, phosphorus run-off and subsequent eutrophication of aquatic ecosystems, as well as the release of greenhouse gasses, ozone layer degradation and acid rain, among other things [
23]. PGPR can therefore be used for precision agriculture practices, which seek to optimise the usage of seeds, fertiliser and pesticides, while at the same time lowering costs and maximising yield quality and the economic return of a given agricultural production system [
24,
25]. Precision agriculture relies heavily on remote sensing applications to enable site-specific crop management. Such approaches are enabled by hyperspectral imaging, which can record images containing spectral data for a range of wavelengths in the light spectrum, typically in the visible and near-infrared (VNIR: 400–1000 nm) and shortwave infrared (SWIR: 1000–2500 nm) spectral regions [
26]. Spectral information is spatially resolved and extended to parts of the light spectrum invisible to the human eye [
27]. Hyperspectral imaging can be used to resolve different plant stresses that change the biochemical composition of leaf tissue, enabling non-destructive plant phenotyping [
28]. Internal leaf structure and pigment composition vary depending on the stressor and can result in different spectral signatures, depending on biotic or abiotic stress [
29], while chlorophyll and carotenoid pigment content and spatial distribution can be used to determine the presence of leaf diseases [
30]. Previous research has also shown that hyperspectral imaging can be used to distinguish winter wheat (
Triticum aestivum L.) growing in microbiologically richer soils [
31] and to detect the beneficial effects of some PGPR, since PGPR-mediated higher uptake of nutrients to plants leads to improved leaf biochemistry [
32].
The efficacy of
B. firmus I-1582 as a bionematicide has been documented, and possible nematode virulence factors that could explain the nematicidal properties of this strain have been described within the assembly of the
B. firmus I-1582 genome [
12]. However, to the best of our knowledge, there are no studies examining the genetic capability of
B. firmus I-1582 to promote plant-growth and concurrent nematicidal and plant-growth promoting effects on nematode-stressed tomato plants using hyperspectral imaging. The aims of this study were therefore (1) to investigate the nematicidal performance of
B. firmus I-1582 against
M. luci on tomatoes in a controlled environment in a glasshouse, as well as in a microplot experimental site serving as a field-analogue; (2) to evaluate the genetic capability of
B. firmus I-1582 as a PGPR through bioinformatics analysis and compare it with the measured elemental composition of plant tissue; (3) to measure the effects of
B. firmus I-1582 inoculation on plant physiology and substrate/soil biological activity; and (4) to test the ability of hyperspectral imaging to differentiate and characterise plants with different treatments. Our hypotheses were in line with the aims stated above: (1)
B. firmus I-1582 has a marked nematicidal influence on
M. luci on tomatoes; (2) as a PGPR,
B. firmus I-1582 has the genetic capability to facilitate nutrient uptake in tomato plants; (3)
B. firmus I-1582 has a positive effect on plant physiology and soil biological activity; and (4) hyperspectral imaging, combined with machine learning methods, can successfully distinguish between different treatments and identify relevant spectral regions for these differences.
3. Discussion
Inoculation of the tomato rhizosphere with
B. firmus I-1582 had a dual effect on the plants.
B. firmus reduced the severity of
M. luci infestation in both pot and microplot experiments by approximately 50%, while at the same time having beneficial effects on plant morphology, physiology and nutrient uptake.
B. firmus-treated plants exhibited PGP effects compared to the control plants (PC), where nematode infestation was completely eliminated (pot experiment) or inhibited to the same level (microplot experiment). The results indicated PGP effects of
B. firmus were not associated solely with lower nematode counts, although the specific
B. firmus PGP effects indicated by the results were not controlled using a separate treatment of
B. firmus-treated plants not inoculated with nematodes. In the pot experiment, the plant physiology measurements only detected differences between treated (PC, BfL and BfH) and untreated plants (NC). Photosynthesis rate measurements did not show statistically significant differences between treatments despite seemingly clearly separated means. This was due to the high variance of the data in the negative control (NC) and a single aberrant measurement in the BfL treatment. Removal of this outlier resulted in statistically significant differences in photosynthesis rates for the treated (PC, BfL and BfH) and untreated (NC) plants (F
3, 11 = 7.05,
p = 0.0065). In the microplot experiment, a significant difference between treatments was only observed in the effective quantum yield of the PSII and Fv/Fm values, while the other parameters were not significantly different. In general, the highest rates of photosynthetic parameters were recorded for the BfH treatment. The small data sample, together with the high variability of the measured parameters recorded for the PC treatment, could explain the lack of significance for this treatment. Some morphological and biochemical parameters indicated plant growth promotion (PGP) of
B. firmus, examples being leaf area, number of flowers, plant dry weight and fresh root weight in the pot experiment, as well as relative leaf chlorophyll content and the content of some elements in the microplot experiment. These PGP effects were evident even when compared to the positive control (PC), where the chemical nematicide was used to either completely eliminate the nematode infestation in the pot experiment (
Table 1), or suppress it to the same level as in
B. firmus-treated plants in the microplot experiment (
Table 2), thus eliminating or levelling the effects of RKN-induced biological stress on tomatoes in the PC treatment compared to BfH. The efficacy of the chemical RKN control treatment (PC; active ingredient fluopyram) was inconsistent in the pot and microplot experiments. Lower nematode suppression in the PC in the microplot experiment could be attributed to experimental design and environmental factors. To approach field-like RKN infestation conditions in the microplot experiment, highly infested tomato roots were incorporated into the microplot soil.
M. luci eggs were contained within the gelatinous matrix of the egg-masses on the root surface or even inside the roots, and were therefore shielded from the effects of chemical treatment to a greater extent than the eggs in suspension used in the pot experiment. A greater day/night temperature difference in the microplot experiment could also have resulted in more gradual J2 larvae hatching than in the controlled glasshouse environment. Thus, in the microplot experiment, a higher number of J2 larvae were invading plant roots between nematicide applications, when fluopyram was less efficient [
33]. Additionally, the microplot soil was much more heterogeneous in terms of particle size and composition than the defined substrate used in the pot experiment, leading to possible areas of sub-optimal nematicide concentrations. Fluopyram has previously been shown to have a limited effect on
Heterodera glycines inhibition beyond the applied area of the soil [
34]. Although
B. firmus consistently reduced
M. luci infestation levels in the pot and microplot experiments (
Table 1 and
Table 2), previous studies have highlighted more variable results [
35] or inconsistent success in controlling plant-parasitic nematode populations and crop yield [
36,
37]. However, the inconsistencies observed in these studies may be the result of variable environmental conditions, unsuccessful rhizosphere colonisation and microbial competition. Beeman and Tylka [
37] further pointed out the need to evaluate the nematicidal action of
B. firmus over longer experiments, which may also be important when characterising its PGP effects.
Inoculation with
B. firmus I-1582 significantly increased the total microbial activity of the substrate or soil in both the pot and microplot experiments (
Figure 2). Activity at the end of the experiment did not depend on the size of the bacterial inoculum, although the robustness of this observation was not tested in the microplots. Total microbial activity was lower in the pot experiment than in microplots, probably due to a higher sand content in the substrate used in the pot experiment. A high sand content can correlate with low fluorescein release, measured using the fluorescein diacetate (FDA) hydrolysis method [
38]. The level of microbial activity in the BfH microplot treatment was similar to the results obtained by Giannakou et al. [
14] when testing the same bacterial strain. Additionally, spatial analysis of FDA hydrolysis data in our experiment indicated the predominantly rhizosphere activity of
B. firmus I-1582, since in the BfH microplot the highest fluorescence was measured in the areas closest to the plants, while the PC and NC microplots showed a more random distribution (
Figure 2c).
Multiple genes associated with various PGP traits were found in the
B. firmus I-1582 genome. Genes associated with phytohormone IAA production (
ipdC,
aat and
iaaH) found in the I-1582 assembly did not show any similarity to query sequences that would be considered to correspond to function (
Figure 3). Bacterially produced IAA can have PGP as well as deleterious effects on plant growth. IAA is associated with stimulating effects on root system architecture, increasing root hair formation and the number and length of the roots [
39]. We did not observe significant differences in root system architecture in
B. firmus-inoculated and non-inoculated plants in our experiments (results not shown). Fresh root weight was significantly higher in BfH plants in the pot experiment, but not in the BfH treatment in the microplot experiment (
Table 2 and
Table 3). The higher root weight in the BfH-treated plants in the pot experiment could be attributed to the larger plant canopy of these plants and not to the bacterial IAA-related effects on the root system.
With regard to nutrient uptake mechanisms, I-1582 was found to contain various gene homologues or metabolic pathways that could contribute to PGP (
Figure 3). Nutrient measurements for leaves at the end of the microplot experiment were inconclusive in relation to the extent of actual PGP by I-1582 (
Table 5). I-1582 did not appear to have the genetic capability for atmospheric nitrogen fixation, since the majority of genes from the
nif cluster (
nifBHDKYENXhesAnifV) did not have any matches in I-1582—especially the
nifH gene, encoding the reaction-catalysing reductase subunit of nitrogenase [
40]. Other nitrogen cycle-related genes without matches in I-1582 were
amoA and
amoB genes encoding the α- and β-subunits of the ammonia monooxygenase enzyme from
Nitrosomonas europaea active in ammonia oxidation (NH
3 to NO
2−) as part of the nitrification process [
41], and
norB encoding the nitric oxide reductase enzyme involved in denitrification [
42]. Homologues to the
nirK and
nosZ (weak identity) genes associated with denitrification were found in the I-1582 assembly. I-1582 also appeared to have the complete bacterial
nas gene cluster (
nasABCDEF) used for nitrate assimilation from dissolved and particulate organic nitrogen [
43], but nitrate assimilated inside bacterial cells in such a manner would not be available to plants [
41]. Sequences with weak identity to
napA and
narG involved in dissimilatory nitrate reduction to ammonium (DNRA) were found in I-1582. However, the gene
nrfA, encoding the key nitrite reductase [
44], was not found, indicating that I-1582 did not have the genetic capability for DNRA-associated PGP. Based on the bioinformatics analysis, this strain did not have the genetic capability for nitrogen fixation or DNRA, but rather used assimilated N in its own metabolism or facilitated its escape into the atmosphere through denitrification. On the other hand, various PGPR, such as
Bacillus spp., have been demonstrated to enhance plant growth by influencing the expression of the nitrate (NO
3−) and ammonium (NH
4+) uptake genes of plants [
45], which could potentially explain the higher average N content in BfH plants in the microplot experiment (
Table 5). I-1582 was found to contain many gene homologues associated with mineral phosphate solubilisation through gluconic acid production (
gdh and
gad), phosphonate solubilisation (phosphonate gene cluster
phn) and phosphate transport (
pst operon:
pstSCAB and
phoP-
phoR system). The phosphonate
phn gene cluster enables the solubilisation of organophosphorus phosphonate compounds that are not otherwise bioavailable to plants. I-1582 did not contain all the genes from the clusters (
Figure 3), similar to various
B. cereus strains [
46].
B. firmus is known to produce 2-ketogluconic acid and this organic acid secretion is considered the primary mechanism for phosphate, along with K and Zn solubilisation [
20]. Various secondary mechanisms include the production of chelating substances and inorganic acids. Soil bacteria usually uptake solubilised phosphate and K through various types of transporters, such as Trk, Kdp and Ktr [
47]. Homologues to genes from
ktrABCD, but not to the
kdpFABC operon, were found in the I-1582 genome. Nutrient measurements showed that although the BfH-treated plants averaged the highest K content in leaves, there were no significant differences between treatments, and all plants had borderline-deficient K concentrations [
48]. No significant differences in P content were observed between treatments either, but the BfH-treated plants had a significantly higher Zn content (
Table 5). Additionally, genes homologous to the siderophore petrobactin (anthrachelin) biosynthetic cluster (
asbABCDEF), but not the bacillibactin (
dhbABCEF) biosynthetic cluster, were found in the I-1582 genome. Siderophores are low molecular weight Fe
3+ chelators expressed into an extracellular medium, and petrobactin and bacillibactin are known to be produced by various
Bacillus spp. under Fe-limited conditions [
49,
50]. Siderophores are considered PGP factors since solubilisation of Fe
3+ increases Fe bioavailability to plants as well as microorganisms [
20]. The presence of homologues to petrobactin biosynthesis and transport genes within the genome (
Figure 3) indicated the genetic capability of I-1582 for some kind of siderophore production and possible PGP through enhancement of Fe uptake, but no significant differences were observed between treatments. In the microplot experiment, the BfH-treated plants had a significantly lower Ca content and higher (not statistically significant) average content of Na compared to the NC and PC. In low K
+ conditions plants can uptake more Na
+, which negatively affects the assimilation of Ca
2+ [
51], so the observed results were probably due to the plant’s physiological response and were not influenced by
B. firmus, since PGPR are more likely to increase ion homeostasis, such as the K
+/Na
+ and Ca
2+/Na
+ ratios [
52].
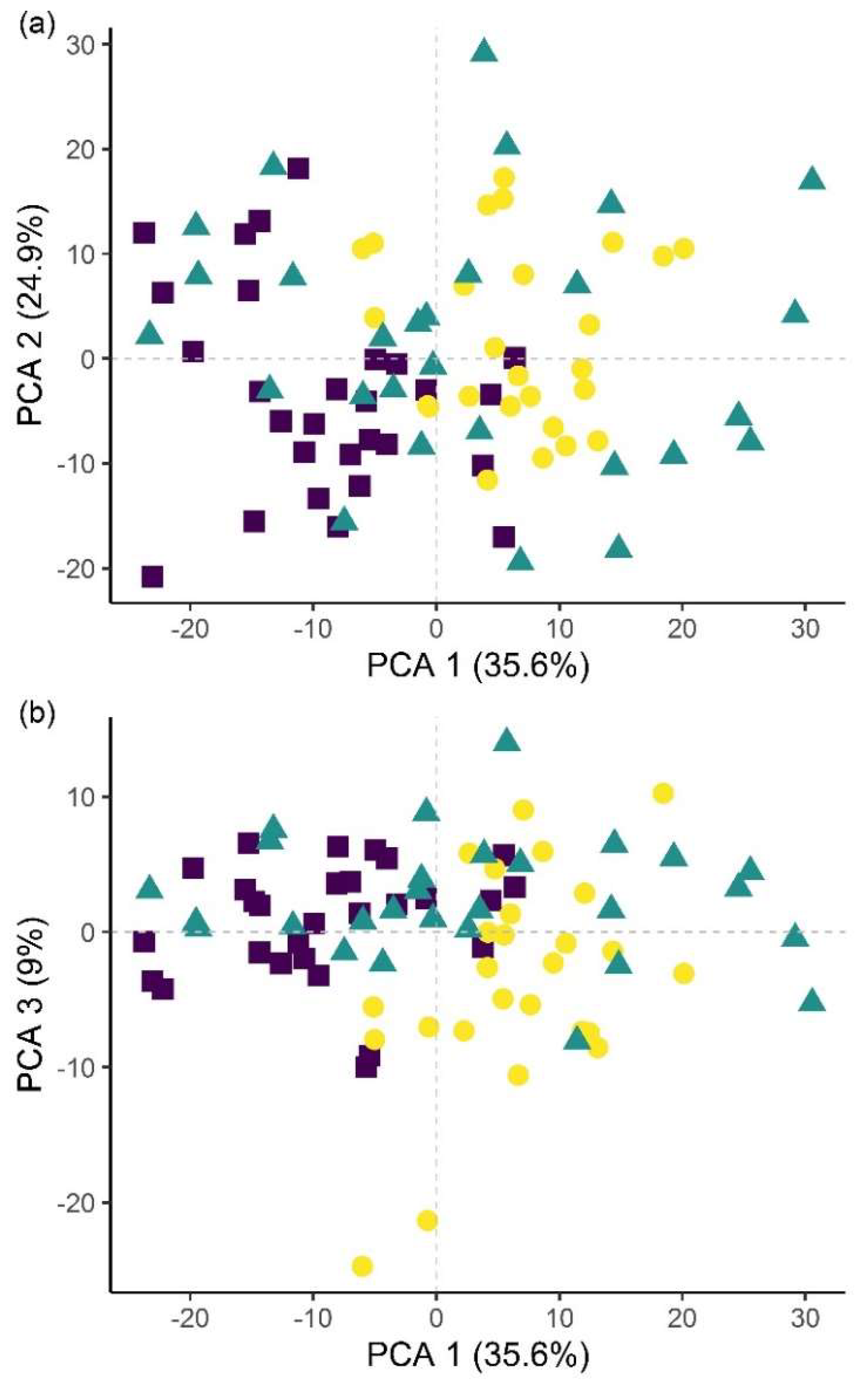
Plants in the pot experiment showed a typical spectral curve for green plants, exhibiting a well-defined reflectance peak around 550 nm (green band) and absorbance at 650–700 nm (red) and 400–500 nm (blue) by chlorophyll (
Figure 7). The red-edge spectral region (690–750 nm) was characterised by a sudden increase in reflectance, indicating normally functioning plants. Reflectance curves in the NIR (approximately 750–1300 nm) and SWIR (1300–2500 nm) regions were also typical, with weaker water absorption bands at 980 and 1150 nm and well-defined water absorption features at 1450 and 1950 nm [
53]. The differences in spectral signatures for the treatments were subtle and most apparent in BfH plants. In contrast to the observed morphology (
Table 3,
Figure 1), the reflectance spectra of the BfH pot experiment plants indicated stress responses. The BfH treatment resulted in higher reflectance in the green band, indicating lower chlorophyll content, while reflectance levelled with the BfL plants in the red-edge and NIR up until about 980 nm, where it again deviated from other treatments in the SWIR region. Higher average reflectance at 550–680 nm has previously been observed with various forms of stress, such as heavy-metal toxicity [
54] or bacterial infestation of leaf tissue, leading to a lower chlorophyll content [
30]. Interestingly, this effect was only observed in BfH-treated plants, but not in plants treated with low
B. firmus-inoculum (BfL). As there were no significant differences in nematicidal effects between the BfL and BfH treatments (
Table 1), the observed responses of the BfL plants could be attributed to induced systemic stress tolerance elicited by low inoculum of
B. firmus, while high bacterial loads in the rhizosphere are known to occasionally have phytotoxic effects [
16]. Furthermore, PGPR can induce a jasmonic acid (JA)-dependent signalling pathway, as seen with
Serattia marcescens and
Bacillus pumilus, leading to systemic stress resistance in
Arabidopsis thaliana against viruses [
55]. JA was shown to augment resistance to a broad range of herbivores, including RKN [
56], and also increase the pigment content in leaves of
Nicotiana tabacum [
57]. On the other hand, the apparently lower chlorophyll content in BfH, as indicated by reflectance, could simply occur due to structural changes in leaf mesophyll, as shown by the spectral signatures in NIR (700–1300 nm). Gitelson et al. [
58] observed that an increase in leaf thickness and/or density led to an increase in reflectance in NIR and to a per volume decrease in chlorophyll concentration (thus causing an increase in reflectance in the green band). Relative chlorophyll content measurements, however, do not appear to be affected by leaf thickness in this way [
59]. The BfH treatment also led to higher reflectance in parts of the NIR and SWIR spectrum, which has previously been observed in plants subject to salt [
60] or water stress [
29,
61,
62]. This observation again contradicted the morphological data, since the BfH plants had larger canopies and a greater plant dry weight and fresh root weight—not indicative of persistent water stress. The spectral data could be explained by brief periodic water-limiting conditions due to high plant growth and limiting substrate volume towards the end of the pot experiment, especially since the discrepancies described above were not observed in the BfH-treated plants in the microplot experiment. Apart from foliar water content, however, the significant spectral ranges identified for discrimination of the treatment groups in both the pot and microplot experiments underlined the importance of dry matter-associated spectral regions (1.7, 2.1 and 2.3 μm) in the SWIR [
63]. Wavelengths in the ranges 740–759 nm, linked to biomass reflectance, 1437–1448 nm, linked to water absorption [
64], and 1145 nm and 2054–2065 nm, linked to the N–H stretch in proteins [
65], were important in differentiating between the treated and untreated plants in the pot experiment; in turn, discrimination between
B. firmus-inoculated and non-inoculated plants was determined by differences in the ranges 2005–2033 nm, linked to foliar water, lignin or cellulose, 2157 nm, linked to the C–N and N–H bonds in proteins, and 2325–2417 nm, linked to cellulose, starch, amylose and proteins. Apart from spectral ranges linked to biomass (759–770 nm) and water absorption (2005–2016 nm), the C–N and N–H stretches (2152–2157 nm) were also significant for separation of the two
B. firmus inoculum sizes (
Figure 9a). Nitrogen, probably contained in the molecular structures of the proteins, energy storage metabolites and structural molecules, were thus implicated as significant factors in discrimination between
B. firmus-inoculated and non-inoculated plants. Significant spectral ranges in the BfH-treated plants in the microplot experiment were especially linked to chlorophyll (
Figure 9c) and pigment content (482–773 nm), which was supported by relative chlorophyll measurements, as well as to proteins (2162–2173 nm). It has been reported that the relationship between relative chlorophyll content and leaf N content per leaf area is affected by environmental factors and the leaf features of crop species [
66]. The spectral ranges linked to the C–N and N–H stretches of the proteins were equally important in the NC plants (1085–1090, 1177, 2152 nm;
Figure 9b), which corresponded with the nutrient measurement data, since the BfH and NC plants in the microplot did not significantly differ in total leaf N content (
Table 5,
Figure S3). Differences between the treatments described above made it possible to differentiate between
B. firmus-treated and untreated plants using hyperspectral remote sensing, with a reliability of 96.3% (microplot experiment) and 97.4% (pot experiment). This analysis showcased the possibility of using hyperspectral remote sensing and image analysis to determine successful application of nematicidal and plant-growth-promoting bacterial agents in the rhizosphere. Successful application of a biological control agent means successful growth of added bacteria in the rhizosphere, through which they produce metabolites having nematicidal and PGP effects. Detailed plant-by-plant determination of a successful biological control agent application could be determined using hyperspectral remote sensing and image analysis, and a secondary application of bacteria could be prescribed for specific plants not yet protected by the first application.