Root Foraging Capacity in Bambara Groundnut (Vigna Subterranea (L.) Verdc.) Core Parental Lines Depends on the Root System Architecture during the Pre-Flowering Stage
Abstract
:1. Introduction
2. Results
2.1. Plant Emergence, Size, Biomass Production, and Root/Shoot Ratio
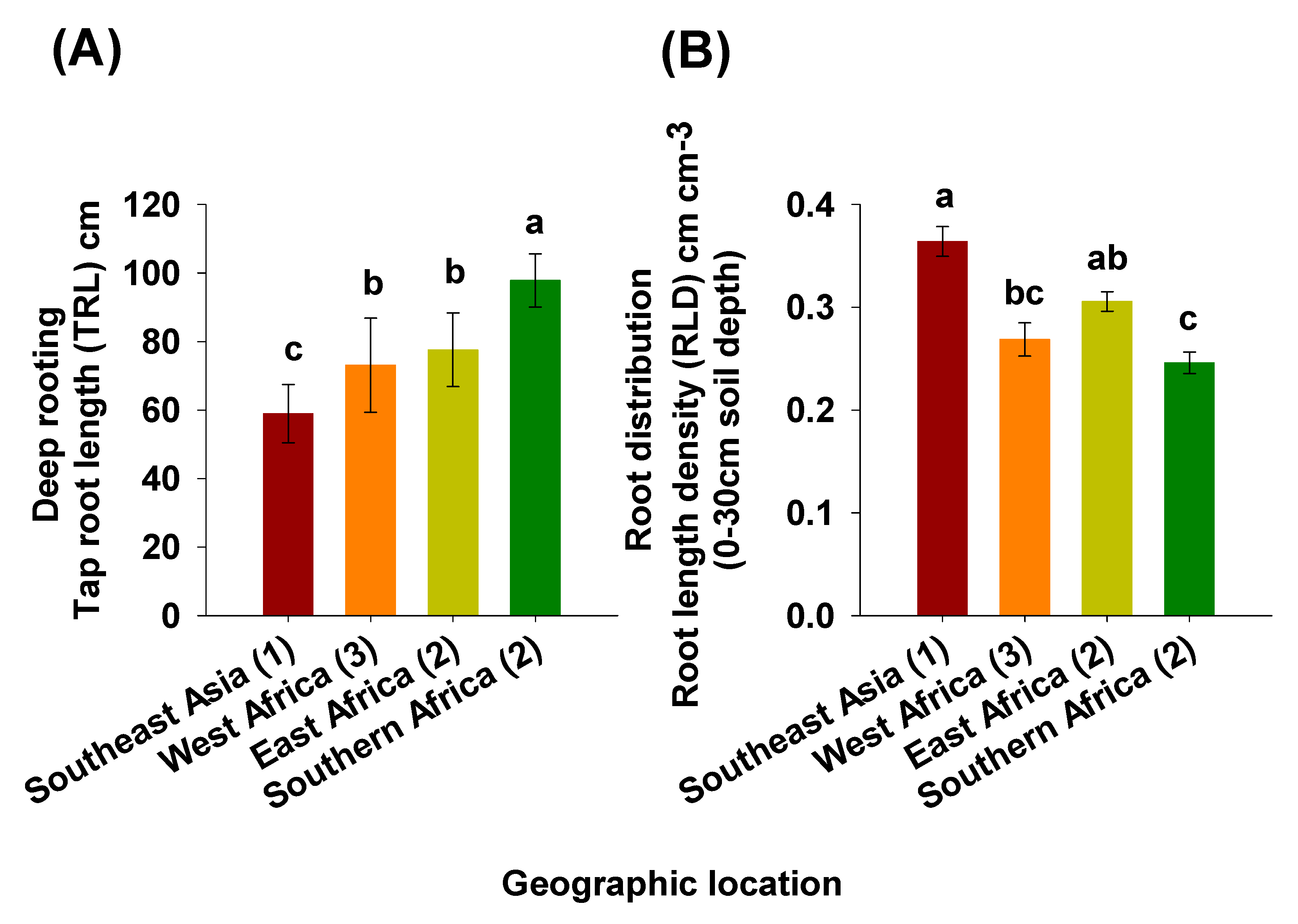
2.2. Deep Rooting Profile
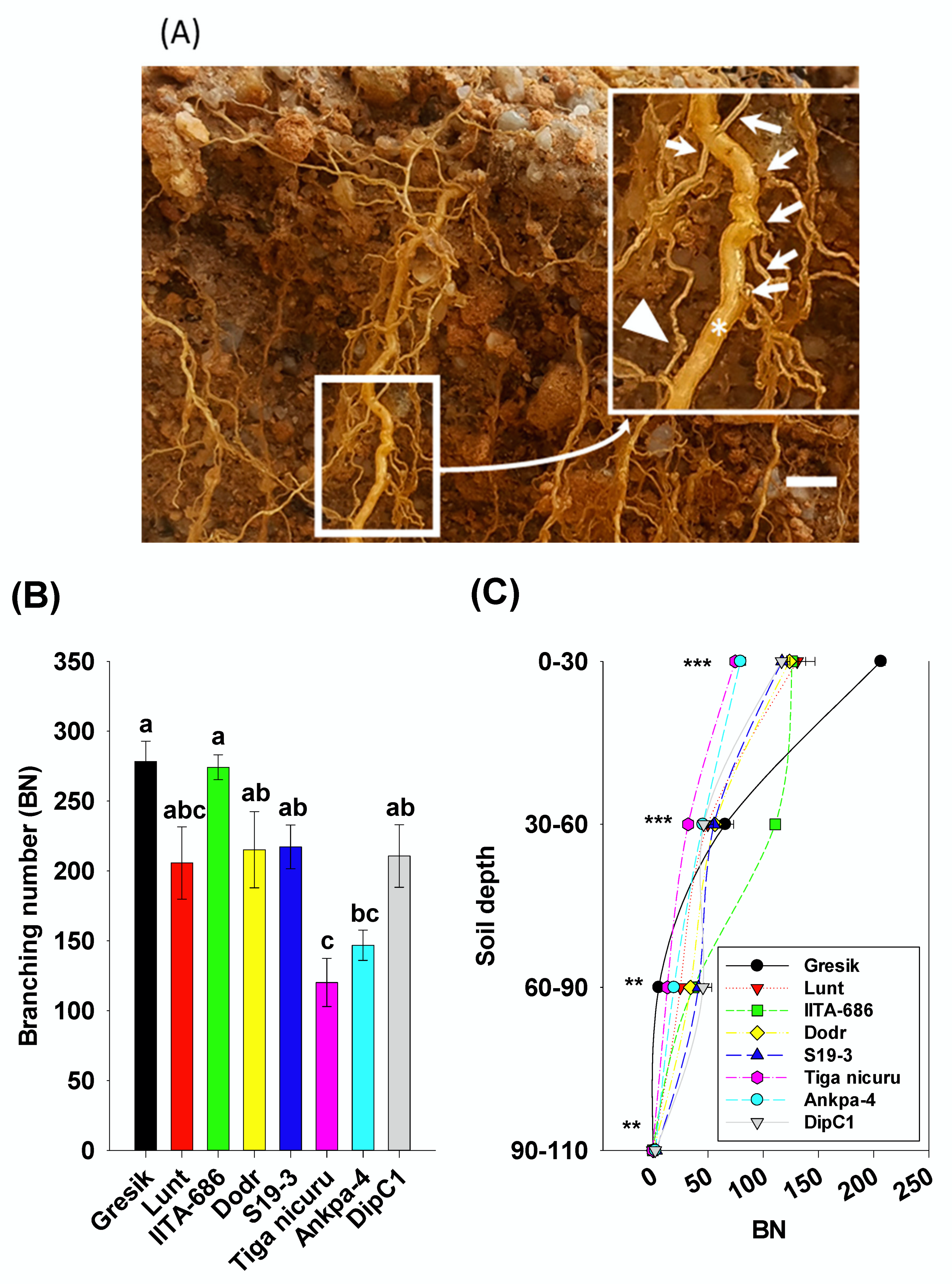
2.3. Root System Branching and Density Dynamics
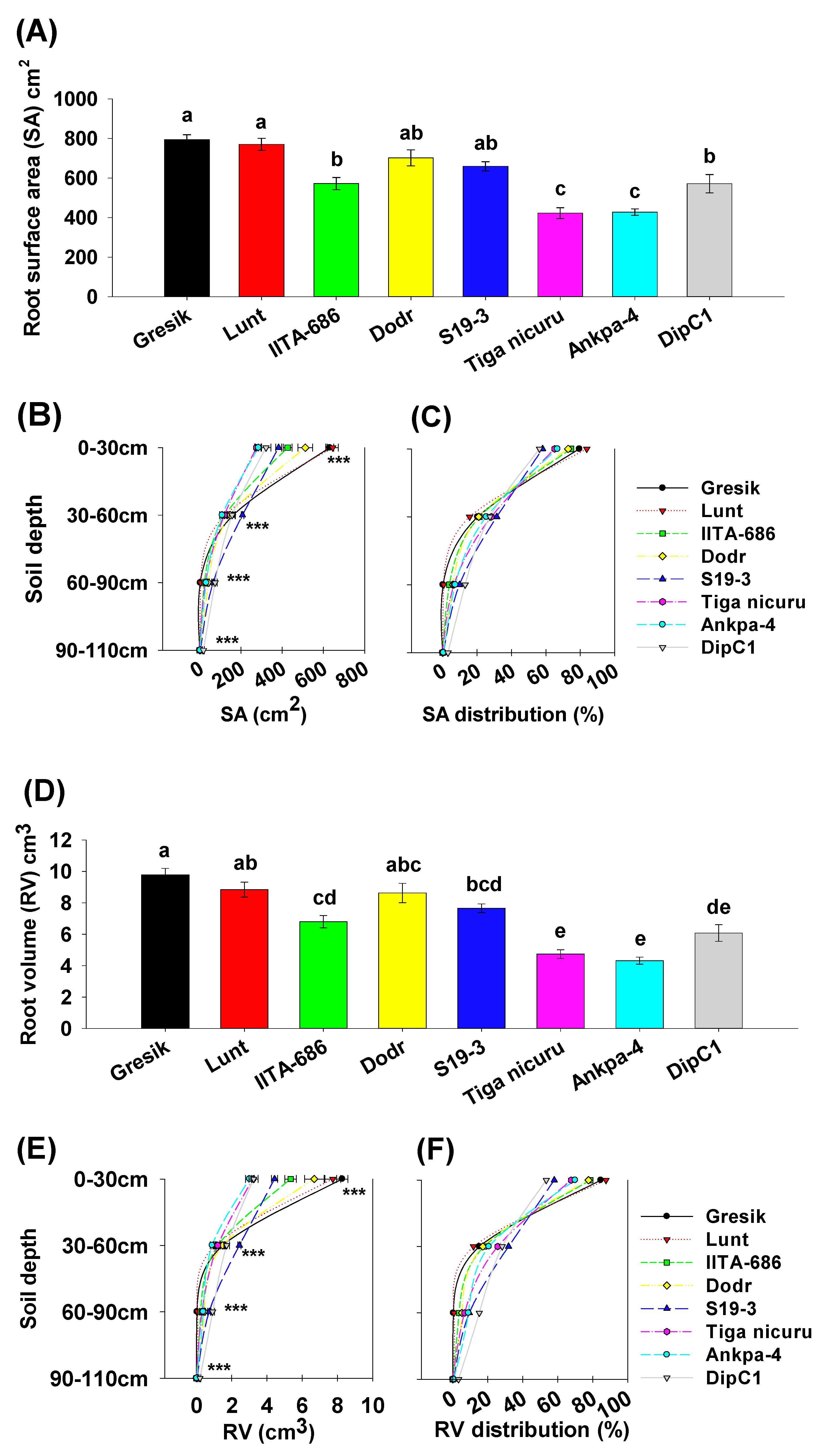
2.4. Root Surface Area, Volume, and Diameter
2.5. Identification of Grouping of Genotypes with Relatively Homogeneous Root Traits
3. Discussion
4. Materials and Methods
4.1. Plant Material
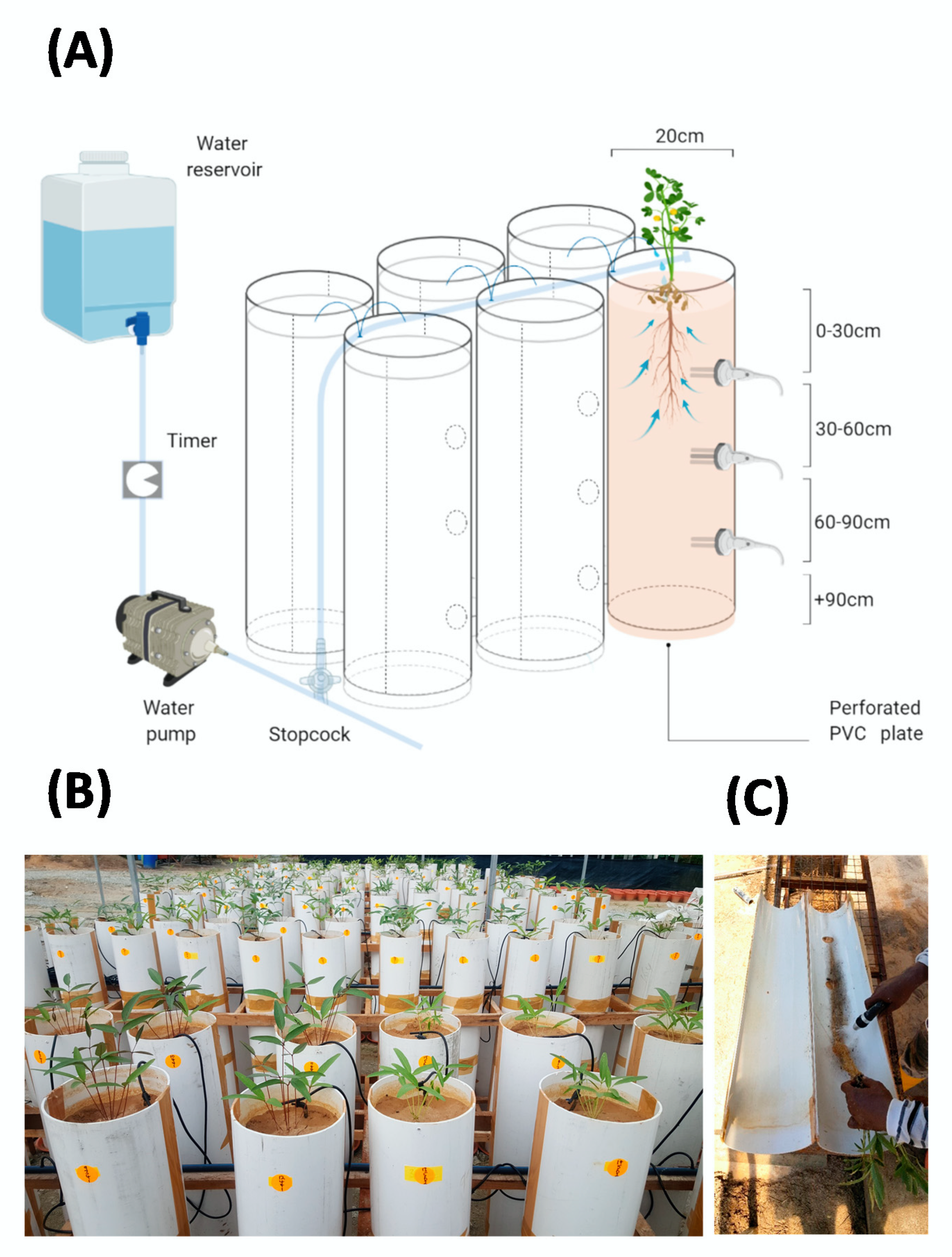
4.2. Study Site and PVC Column Screening System
4.3. Plant Sampling and Measurements
4.4. Developmental Traits
4.5. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rogers, E.D.; Benfey, P.N. Regulation of plant root system architecture: Implications for crop advancement. Curr. Opin. Biotechnol. 2015, 32, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Ho, M.D. Rhizoeconomics: Carbon costs of phosphorus acquisition. Plant Soil 2005, 269, 45–56. [Google Scholar] [CrossRef]
- Paula, S.; Pausas, J.G. Root traits explain different foraging strategies between resprouting life histories. Oecologia 2011, 165, 321–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feldman, A.; Ho, W.K.; Massawe, F.; Mayes, S. Bambara groundnut is a climate-resilient crop: How could a drought-tolerant and nutritious legume improve community resilience in the face of climate change? In BT—Sustainable Solutions for Food Security: Combating Climate Change by Adaptation; Sarkar, A., Sensarma, S.R., van Loon, G.W., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 151–167. ISBN 978-3-319-77878-5. [Google Scholar]
- Doku, E.V.; Karikari, S.K. Bambara groundnut. Econ. Bot 1971, 25, 225–252. [Google Scholar] [CrossRef]
- Taub, D.R.; Goldberg, D. Root System Topology of Plants from Habitats Differing in Soil Resource Availability. Funct. Ecol. 1996, 10, 258–264. [Google Scholar] [CrossRef]
- Chen, Y.L.; Dunbabin, V.M.; Diggle, A.J.; Siddique, K.H.M.; Rengel, Z. Development of a novel semi-hydroponic phenotyping system for studying root architecture. Funct. Plant Biol. 2011, 38, 355–363. [Google Scholar] [CrossRef] [Green Version]
- Collinson, S.T.; Clawson, E.J.; Azam-Ali, S.N.; Black, C.R. Effects of soil moisture deficits on the water relations of bambara groundnut (Vigna subterranea L. Verdc.). J. Exp. Bot. 1997, 48, 877–884. [Google Scholar] [CrossRef] [Green Version]
- Collinson, S.T.; Berchie, J.; Azam-Ali, S.N. The effect of soil moisture on light interception and the conversion coefficient for three landraces of bambara groundnut (Vigna subterranea). J. Agric. Sci. 1999, 133, 151–157. [Google Scholar] [CrossRef] [Green Version]
- Sesay, A.; Mpuisang, T.; Morake, T.S.; Al-Shareef, I.; Chepete, H.J.; Moseki, B. Preliminary assessment of bambara groundnut (Vigna subterranea L.) landraces for temperature and water stress tolerance under field conditions in botswana. S. Afr. J. Plant Soil 2010, 27, 312–321. [Google Scholar] [CrossRef]
- Jørgensen, S.T.; Liu, F.; Ouédraogo, M.; Ntundu, W.H.; Sarrazin, J.; Christiansen, J.L. Drought Responses of Two Bambara Groundnut (Vigna subterranea L. Verdc.) Landraces Collected from a Dry and a Humid Area of Africa. J. Agron. Crop Sci. 2010, 196, 412–422. [Google Scholar] [CrossRef]
- Vurayai, R.; Emongor, V.; Moseki, B. Effect of water stress imposed at different growth and development stages on morphological traits and yield of bambara groundnuts (Vigna subterranea L. Verdc). Am. J. Plant Physiol. 2011, 6, 17–27. [Google Scholar] [CrossRef] [Green Version]
- Mabhaudhi, T.; Modi, A.T. Growth, phenological and yield responses of a bambara groundnut (Vigna subterranea (L.) Verdc.) landrace to imposed water stress under field conditions. S. Afr. J. Plant Soil 2013, 30, 69–79. [Google Scholar] [CrossRef]
- Mabhaudhi, T.; Modi, A.T.; Beletse, Y.G. Growth, phenological and yield responses of a bambara groundnut (Vigna subterranea L. Verdc) landrace to imposed water stress: IL. Rain shelter conditions. Water SA 2013, 39, 191–198. [Google Scholar] [CrossRef] [Green Version]
- Chibarabada, T.P.; Modi, A.T.; Mabhaudhi, T. Bambara groundnut (Vigna subterranea) seed quality in response to water stress on maternal plants. Acta Agric. Scand. Sect. B Soil Plant Sci. 2015, 65, 364–373. [Google Scholar] [CrossRef]
- Chibarabada, T.P.; Modi, A.T.; Mabhaudhi, T. Water use characteristics of a bambara groundnut (Vigna subterranea L. Verdc) landrace during seedling establishment. Water SA 2015, 41, 472–482. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, Y.Y.; Mayes, S.; Massawe, F. Effects of short-Term water deficit stress on physiological characteristics of Bambara groundnut (Vigna subterranea (L.) Verdc.). S. Afr. J. Plant Soil 2016, 33, 51–58. [Google Scholar] [CrossRef]
- Chai, H.H.; Massawe, F.; Mayes, S. Assessment of a segregating population for the improvement of drought tolerance in Bambara groundnut. Acta Hortic. 2016, 1127, 339–345. [Google Scholar] [CrossRef]
- Chai, H.H.; Massawe, F.; Mayes, S. Effects of mild drought stress on the morpho-physiological characteristics of a bambara groundnut segregating population. Euphytica 2016, 208, 225–236. [Google Scholar] [CrossRef]
- De Dorlodot, S.; Forster, B.; Pagès, L.; Price, A.; Tuberosa, R.; Draye, X. Root system architecture: Opportunities and constraints for genetic improvement of crops. Trends Plant Sci. 2007, 12, 474–481. [Google Scholar] [CrossRef]
- Lynch, J.P.; Brown, K.M. New roots for agriculture: Exploiting the root phenome. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 1598–1604. [Google Scholar] [CrossRef]
- Lynch, J.P.; Chimungu, J.G.; Brown, K.M. Root anatomical phenes associated with water acquisition from drying soil: Targets for crop improvement. J. Exp. Bot. 2014, 65, 6155–6166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kundy, A.C. Soil Water Deficit Stress on Bambara Groundnut (Vigna subterranea [L.] Verdc) and Groundnut (Arachis hypogaea [L.]). Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2019. [Google Scholar]
- Abu, H.B.; Buah, S.S.J. Characterization of Bambara Groundnut Landraces and Their Evaluation by Farmers in the Upper West Region of Ghana. J. Dev. Sustain. Agric. 2011, 6, 64–74. [Google Scholar]
- Hammer, G.L.; Dong, Z.S.; McLean, G.; Doherty, A.; Messina, C.; Schussler, J.; Zinselmeier, C.; Paszkiewicz, S.; Cooper, M. Can changes in canopy and/or root system architecture explain historical maize yield trends in the U.S. Corn Belt? Crop Sci. 2009, 49, 299–312. [Google Scholar] [CrossRef]
- Palta, J.A.; Fillery, I.R. Nitrogen accumulation and remobilisation in wheat of sup 15N-urea applied to a duplex soil at seeding. Aust. J. Exp. Agric. 1993, 33, 233–238. [Google Scholar] [CrossRef]
- York, L.M.; Galindo-Castañeda, T.; Schussler, J.R.; Lynch, J.P. Evolution of US maize (Zea mays L.) root architectural and anatomical phenes over the past 100 years corresponds to increased tolerance of nitrogen stress. J. Exp. Bot. 2015, 66, 2347–2358. [Google Scholar] [CrossRef] [Green Version]
- Wasson, A.P.; Richards, R.A.; Chatrath, R.; Misra, S.C.; Prasad, S.V.S.; Rebetzke, G.J.; Kirkegaard, J.A.; Christopher, J.; Watt, M. Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J. Exp. Bot. 2012, 63, 3485–3498. [Google Scholar] [CrossRef] [Green Version]
- Farooq, M.; Gogoi, N.; Barthakur, S.; Baroowa, B.; Bharadwaj, N.; Alghamdi, S.S.; Siddique, K.H.M. Drought Stress in Grain Legumes during Reproduction and Grain Filling. J. Agron. Crop Sci. 2017, 203, 81–102. [Google Scholar] [CrossRef]
- Gregory, P.J.; McGowan, M.; Biscoe, P.V.; Hunter, B. Water relations of winter wheat: 1. Growth of the root system. J. Agric. Sci. 1978, 91, 91–102. [Google Scholar] [CrossRef]
- Krishnamurthy, L.; Ito, O.; Johansen, C. Genotypic differences in root growth dynamics and its implications for drought resistance chickpea. Dvnamics of Roots and Nitrogen in Cropping Systems of the Semi-arid Tropics. JIRCAS J. 1996, 3, 33–48. [Google Scholar]
- Kashiwagi, J.; Krishnamurthy, L.; Upadhyaya, H.D.; Krishna, H.; Chandra, S.; Vadez, V.; Serraj, R. Genetic variability of drought-avoidance root traits in the mini-core germplasm collection of chickpea (Cicer arietinum L.). Euphytica 2005, 146, 213–222. [Google Scholar] [CrossRef] [Green Version]
- Polanía, J.A.; Rao, I.M.; Beebe, S.; García, R. Desarrollo y distribución de raíces bajo estrés por sequía en fríjol común (Phaseolus vulgaris L.) en un sistema de tubos con suelo. Agron. Colomb. 2009, 27, 25–32. [Google Scholar]
- Chen, Y.L.; Dunbabin, V.M.; Postma, J.A.; Diggle, A.J.; Palta, J.A.; Lynch, J.P.; Siddique, K.H.M.; Rengel, Z. Phenotypic variability and modelling of root structure of wild Lupinus angustifolius genotypes. Plant Soil 2011, 348, 345–364. [Google Scholar] [CrossRef]
- León, M.F.; Squeo, F.A.; Gutiérrez, J.R.; Holmgren, M. Rapid root extension during water pulses enhances establishment of shrub seedlings in the Atacama Desert. J. Veg. Sci. 2011, 22, 120–129. [Google Scholar] [CrossRef]
- Mwale, S.S.; Azam-Ali, S.N.; Massawe, F.J. Growth and development of bambara groundnut (Vigna subterranea) in response to soil moisture. 1. Dry matter and yield. Eur. J. Agron. 2007, 26, 345–353. [Google Scholar] [CrossRef]
- Jørgensen, S.T.; Ntundu, W.H.; Ouédraogo, M.; Christiansen, J.L.; Liu, F. Effect of a short and severe intermittent drought on transpiration, seed yield, yield components, and harvest index in four landraces of bambara groundnut. Int. J. Plant Prod. 2011, 5, 25–36. [Google Scholar]
- Berchie, J.N.; Opoku, M.; Adu-Dapaah, H.; Agyemang, A.; Sarkodie-Addo, J.; Asare, E.; Addo, J.; Akuffo, H. Evaluation of five bambara groundnut (Vigna subterranea (L.) Verdc.) landraces to heat and drought stress at Tono-Navrongo, Upper East Region of Ghana. Afr. J. Agric. Res. 2012, 7, 250–256. [Google Scholar] [CrossRef]
- Al-Shareef, I.; Sparkes, D.; Karunaratne, A.S.; Azam-Ali, S. The effect of temperature and drought stress on radiation use efficiency of Bambara groundnut (Vigna subterranea (L.) Verde.) landraces. Acta Hortic. 2013, 979, 675–682. [Google Scholar] [CrossRef]
- Al Shareef, I.; Sparkes, D.; Azam-Ali, S. Temperature and drought stress effects on growth and development of bambara groundnut (Vigna Subterranea L.). Exp. Agric. 2014, 50, 72–89. [Google Scholar] [CrossRef]
- Nautiyal, P.C.; Kulkarni, G.; Singh, A.L.; Basu, M.S. Evaluation of water-deficit stress tolerance in Bambara groundnut landraces for cultivation in sub-tropical environments in India. Indian J. Plant Physiol. 2017, 22, 190–196. [Google Scholar] [CrossRef]
- Mayes, S.; Basu, S.M.; Molosiwa, O.; Redjeki, E.S.; Ahmad, N.S.; Khan, F.; Zehra, S.; Noah, S.; Mayes, K.; Roberts, J.; et al. Molecular analysis of Bambara groundnut, an underutilised African legume crop as part of the BAMLINK project—What lessons can we learn? Acta Hortic. 2013, 979, 451–458. [Google Scholar] [CrossRef] [Green Version]
- Mayes, S.; Ho, W.K.; Chai, H.H.; Gao, X.; Kundy, A.C.; Mateva, K.I.; Zahrulakmal, M.; Hahiree, M.K.I.M.; Kendabie, P.; Licea, L.C.S.; et al. Bambara groundnut: An exemplar underutilised legume for resilience under climate change. Planta 2019, 250, 803–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuijken, R.C.P.; Van Eeuwijk, F.A.; Marcelis, L.F.M.; Bouwmeester, H.J. Root phenotyping: From component trait in the lab to breeding. J. Exp. Bot. 2015, 66, 5389–5401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lalitha, N.; Upadhyaya, H.D.; Krishnamurthy, L.; Kashiwagi, J.; Kavikishor, P.B.; Singh, S. Assessing genetic variability for root traits and identification of trait-specific germplasm in chickpea reference set. Crop Sci. 2015, 55, 2034–2045. [Google Scholar] [CrossRef]
- Serraj, R.; Krishnamurthy, L.; Kashiwagi, J.; Kumar, J.; Chandra, S.; Crouch, J.H. Variation in root traits of chickpea (Cicer arietinum L.) grown under terminal drought. Field Crop. Res. 2004, 88, 115–127. [Google Scholar] [CrossRef] [Green Version]
- Jongrungklang, N.; Toomsan, B.; Vorasoot, N.; Jogloy, S.; Boote, K.J.; Hoogenboom, G.; Patanothai, A. Drought tolerance mechanisms for yield responses to pre-flowering drought stress of peanut genotypes with different drought tolerant levels. Field Crop. Res. 2013, 144, 34–42. [Google Scholar] [CrossRef]
- Figueroa-Bustos, V.; Palta, A.J.; Chen, Y.; Siddique, H.M.K. Early Season Drought Largely Reduces Grain Yield in Wheat Cultivars with Smaller Root Systems. Plants 2019, 8, 305. [Google Scholar] [CrossRef] [Green Version]
- Nicotra, A.; Babicka, N.; Westoby, M. Seedling root anatomy and morphology: An examination of ecological differentiation with rainfall using phylogenetically independent contrasts. Oecologia 2002, 130, 136–145. [Google Scholar] [CrossRef]
- Lynch, J.P. Steep, cheap and deep: An ideotype to optimize water and N acquisition by maize root systems. Ann. Bot. 2013, 112, 347–357. [Google Scholar] [CrossRef] [Green Version]
- Graham, P.H.; Vance, C.P. Legumes: Importance and Constraints to Greater Use. Plant Physiol. 2003, 131, 872–877. [Google Scholar] [CrossRef] [Green Version]
- Comas, L.H.; Becker, S.R.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 442. [Google Scholar] [CrossRef] [Green Version]
- Reynolds, J.F.; Kemp, P.R.; Ogle, K.; Fernández, R.J. Modifying the ‘pulse–reserve’ paradigm for deserts of North America: Precipitation pulses, soil water, and plant responses. Oecologia 2004, 141, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Flores, R.; Winkel, T.; Nguyen-Thi-Truc, A.; Joffre, R. Root foraging capacity depends on root system architecture and ontogeny in seedlings of three Andean Chenopodium species. Plant Soil 2014, 380, 415–428. [Google Scholar] [CrossRef]
- Redjeki, E.S.; Ho, W.K.; Shah, N.; Molosiwa, O.O.; Ardiarini, N.R.; Kuswanto, K.; Mayes, S. Understanding the genetic relationships of Indonesian bambara groundnut cultivars and investigating their origins. Genome 2020. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P. Roots of the Second Green Revolution. Aust. J. Bot. 2007, 55, 493–512. [Google Scholar] [CrossRef]
- Blum, A. Plant Breeding for Water-Limited Environments; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7490-7. [Google Scholar]
- Sponchiado, B.N.; White, J.W.; Castillo, J.A.; Jones, P.G. Root growth of four common bean cultivars in relation to drought tolerance in environments with contrasting soil types. Exp. Agric. 1989, 25, 249–257. [Google Scholar] [CrossRef]
- Kashiwagi, J.; Krishnamurthy, L.; Crouch, J.H.; Serraj, R. Variability of root length density and its contributions to seed yield in chickpea (Cicer arietinum L.) under terminal drought stress. Field Crop. Res. 2006, 95, 171–181. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.K.; Gaur, P.M.; Chamarthi, S.K.; Krishnamurthy, L.; Tripathi, S.; Kashiwagi, J.; Samineni, S.; Singh, V.K.; Thudi, M.; Jaganathan, D. Fast-track introgression of “QTL-hotspot” for root traits and other drought tolerance traits in JG 11, an elite and leading variety of chickpea. Plant Genome 2013, 6, 14. [Google Scholar] [CrossRef] [Green Version]
- Kirkegaard, J.A.; Lilley, J.M.; Howe, G.N.; Graham, J.M. Impact of subsoil water use on wheat yield. Aust. J. Agric. Res. 2007, 58, 303–315. [Google Scholar] [CrossRef]
- Zhan, A.; Lynch, J.P. Reduced frequency of lateral root branching improves N capture from low-N soils in maize. J. Exp. Bot. 2015, 66, 2055–2065. [Google Scholar] [CrossRef] [Green Version]
- Massawe, F.; Nwale, S.; Azam-Ali, S.; Roberts, J. Breeding in bambara groundnut (Vigna subterranea (L.) Verdc): Strategic considerations. Afr. J. Biotechnol. 2005, 4, 463–471. [Google Scholar]
- Gregory, P.J.; Mayes, S.; Hui, C.H.; Jahanshiri, E.; Julkifle, A.; Kuppusamy, G.; Kuan, H.W.; Lin, T.X.; Massawe, F.; Suhairi, T.A.S.T.M.; et al. Crops For the Future (CFF): An overview of research efforts in the adoption of underutilised species. Planta 2019, 250, 979–988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, J.; Sjöstrom, J. Optimizing the experimental design of soil columns in saturated and unsaturated transport experiments. J. Contam. Hydrol. 2010, 115, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hardie, M.; Lisson, S.; Doyle, R.; Cotching, W. Determining the frequency, depth and velocity of preferential flow by high frequency soil moisture monitoring. J. Contam. Hydrol. 2013, 144, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Wald, A. Tests of Statistical Hypotheses Concerning Several Parameters When the Number of Observations is Large. Trans. Am. Math. Soc. 1943, 54, 426–482. [Google Scholar] [CrossRef]
- Manschadi, A.M.; Hammer, G.L.; Christopher, J.T.; DeVoil, P. Genotypic variation in seedling root architectural traits and implications for drought adaptation in wheat (Triticum aestivum L.). Plant Soil 2008, 303, 115–129. [Google Scholar] [CrossRef]
- Singh, V.; van Oosterom, E.J.; Jordan, D.R.; Messina, C.D.; Cooper, M.; Hammer, G.L. Morphological and architectural development of root systems in sorghum and maize. Plant Soil 2010, 333, 287–299. [Google Scholar] [CrossRef]






| Genotypes | D50% | SH | NoL | R:S Ratio |
|---|---|---|---|---|
| number of days | cm plant −1 | number plant −1 | RDW/SDW | |
| Gresik | 4.67 ± 1.03c | 24.65 ± 1.1a | 17.33 ± 1.91 | 0.35 ± 0.03a |
| Lunt | 5.67 ± 1.03bc | 23.02 ± 0.91ab | 14.67 ± 0.95 | 0.19 ± 0.02bc |
| IITA-686 | 6.5 ± 1.05abc | 24.1 ± 1.04ab | 13.33 ± 0.8 | 0.21 ± 0.02bc |
| Dodr | 5.33 ± 1.03bc | 23.76 ± 0.96ab | 13.83 ± 0.48 | 0.23 ± 0.02bc |
| S19-3 | 6.17 ± 1.47abc | 22.7 ± 0.66ab | 13.83 ± 0.54 | 0.28 ± 0.02ab |
| Tiga nicuru | 5.5 ± 1.05bc | 19.23 ± 2.15b | 14.25 ± 1.09 | 0.23 ± 0.04bc |
| Ankpa-4 | 7.83 ± 1.47a | 19.19 ± 1.52b | 14.33 ± 0.71 | 0.16 ± 0.02c |
| DipC1 | 7.33 ± 1.03ab | 23.08 ± 0.88ab | 15.25 ± 1.42 | 0.21 ± 0.02bc |
| Mean | 6.13 | 22.47 | 14.60 | 0.23 |
| F probability | ||||
| Genotype | 0.000 *** | 0.009 ** | 0.27 ns | 0.000 *** |
| Geographical Region | Genotypes 1 | Seed Color | Country Collected | Climate | Rainfall Mean (mm year−1) |
|---|---|---|---|---|---|
| Southeast Asia | Gresik | Dark | Indonesia | Tropical wet | ˃2000 |
| West Africa | Lunt | Cream | Sierra Leone | Tropical wet | ˃2000 |
| Ankpa-4 | Brown | Nigeria | Tropical dry | ˃2000 | |
| Tiga nicuru | Cream | Mali | Subtropical | 450 | |
| East Africa | IITA-686 | Dark | Tanzania | Tropical dry | ˃750 |
| Dodr | Red | Tanzania | Tropical dry | ˃570 | |
| Southern Africa | S19-3 | Dark | Namibia | Subtropical desert | 350 |
| DipC1 | Cream with black eye | Botswana | Semi-arid | 500 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mateva, K.I.; Chai, H.H.; Mayes, S.; Massawe, F. Root Foraging Capacity in Bambara Groundnut (Vigna Subterranea (L.) Verdc.) Core Parental Lines Depends on the Root System Architecture during the Pre-Flowering Stage. Plants 2020, 9, 645. https://doi.org/10.3390/plants9050645
Mateva KI, Chai HH, Mayes S, Massawe F. Root Foraging Capacity in Bambara Groundnut (Vigna Subterranea (L.) Verdc.) Core Parental Lines Depends on the Root System Architecture during the Pre-Flowering Stage. Plants. 2020; 9(5):645. https://doi.org/10.3390/plants9050645
Chicago/Turabian StyleMateva, Kumbirai Ivyne, Hui Hui Chai, Sean Mayes, and Festo Massawe. 2020. "Root Foraging Capacity in Bambara Groundnut (Vigna Subterranea (L.) Verdc.) Core Parental Lines Depends on the Root System Architecture during the Pre-Flowering Stage" Plants 9, no. 5: 645. https://doi.org/10.3390/plants9050645






