Morpho-Anatomical and Physiological Responses Can Predict the Ideal Period for the Transplantation of Hydroponic Seedlings of Hymenaea courbaril, a Neotropical Fruit Tree
Abstract
:1. Introduction
2. Material and Methods
2.1. Acquisition of the Seedlings and Cultivation Conditions
2.2. Morpho-Anatomical Evaluations
2.3. Physiological Evaluation
2.4. Growth
2.5. Statistical Analyses
3. Results
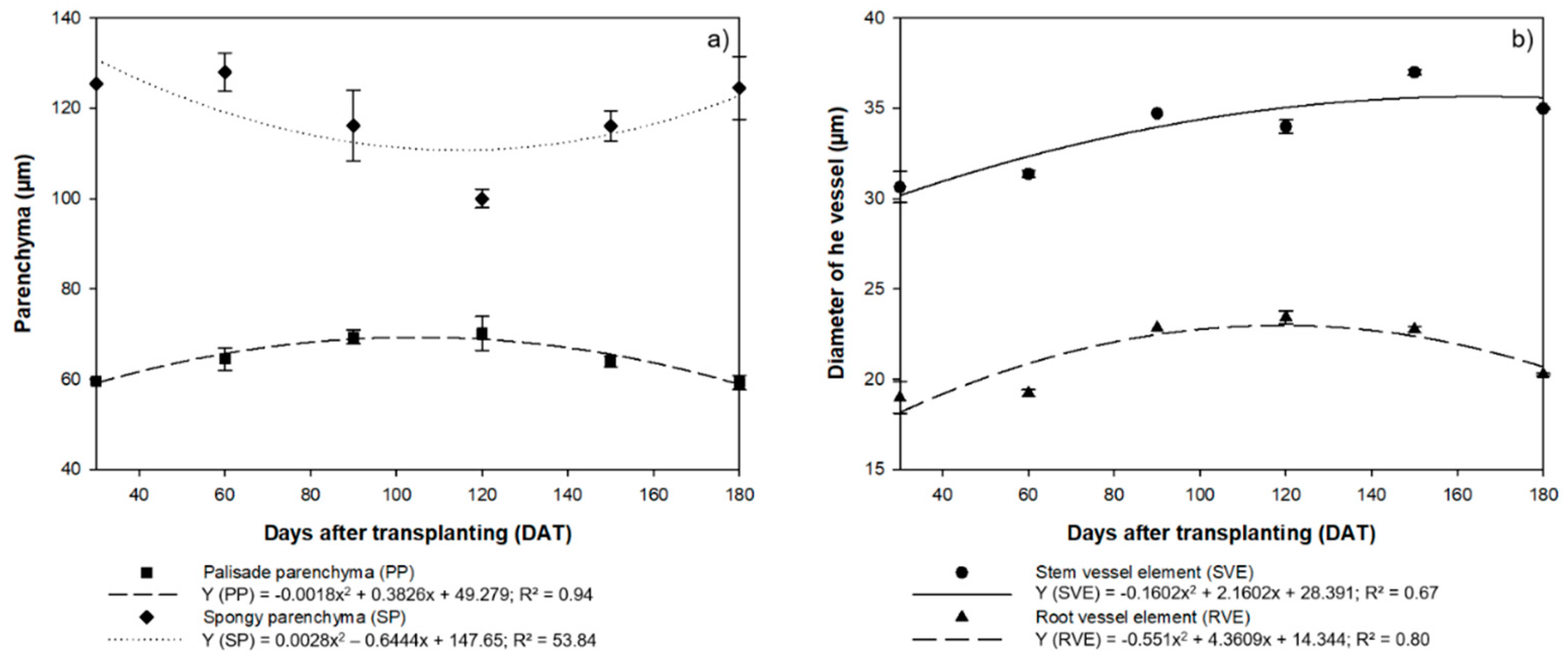
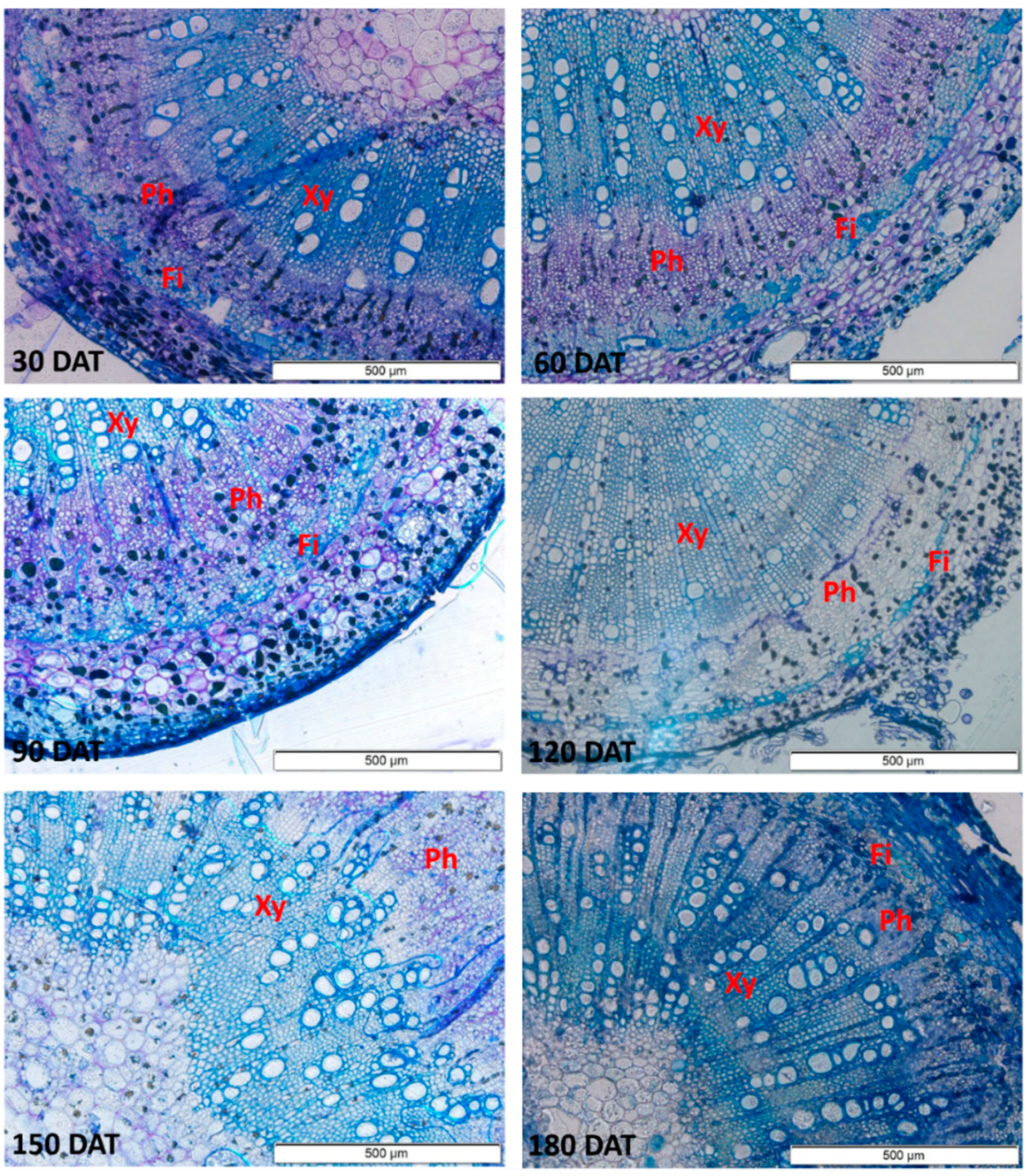
3.1. Morpho-Anatomical Assessment
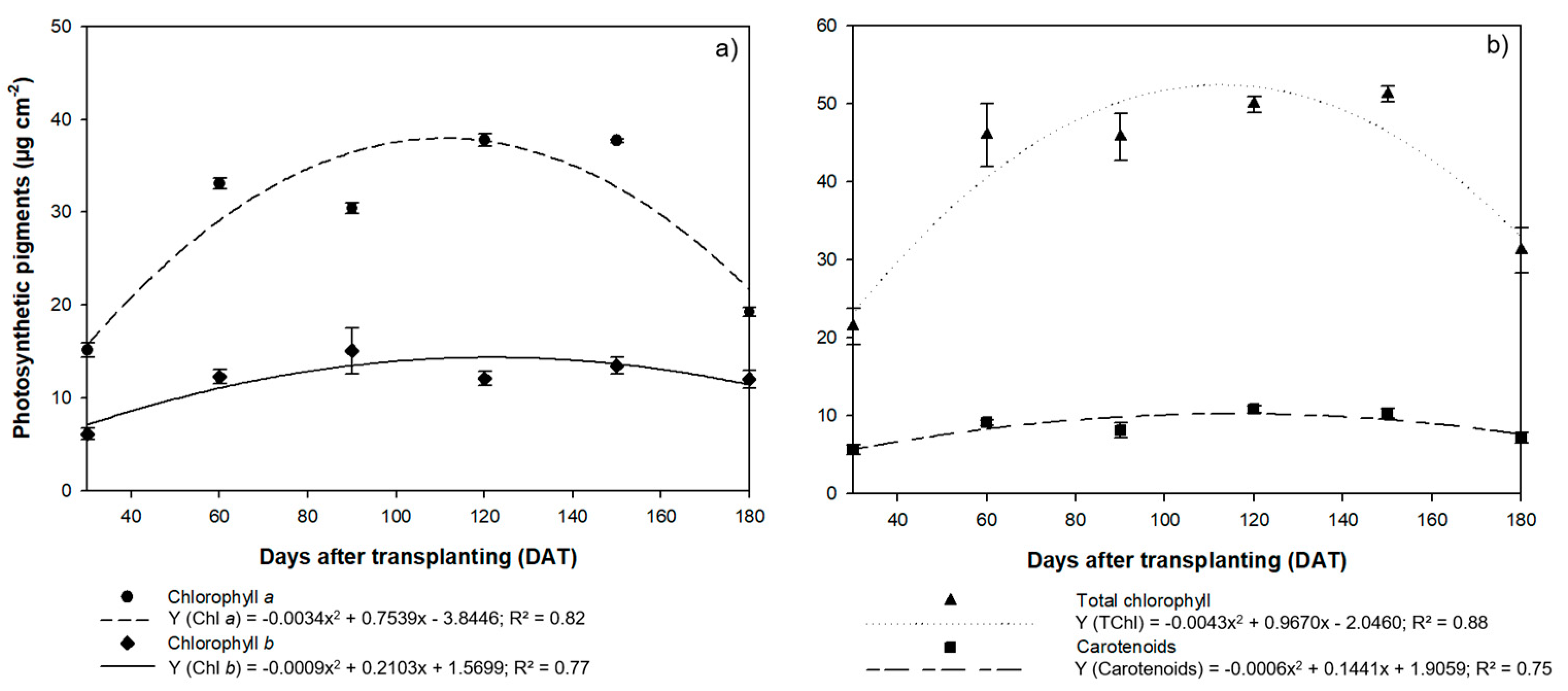
3.2. Physiological Assessment
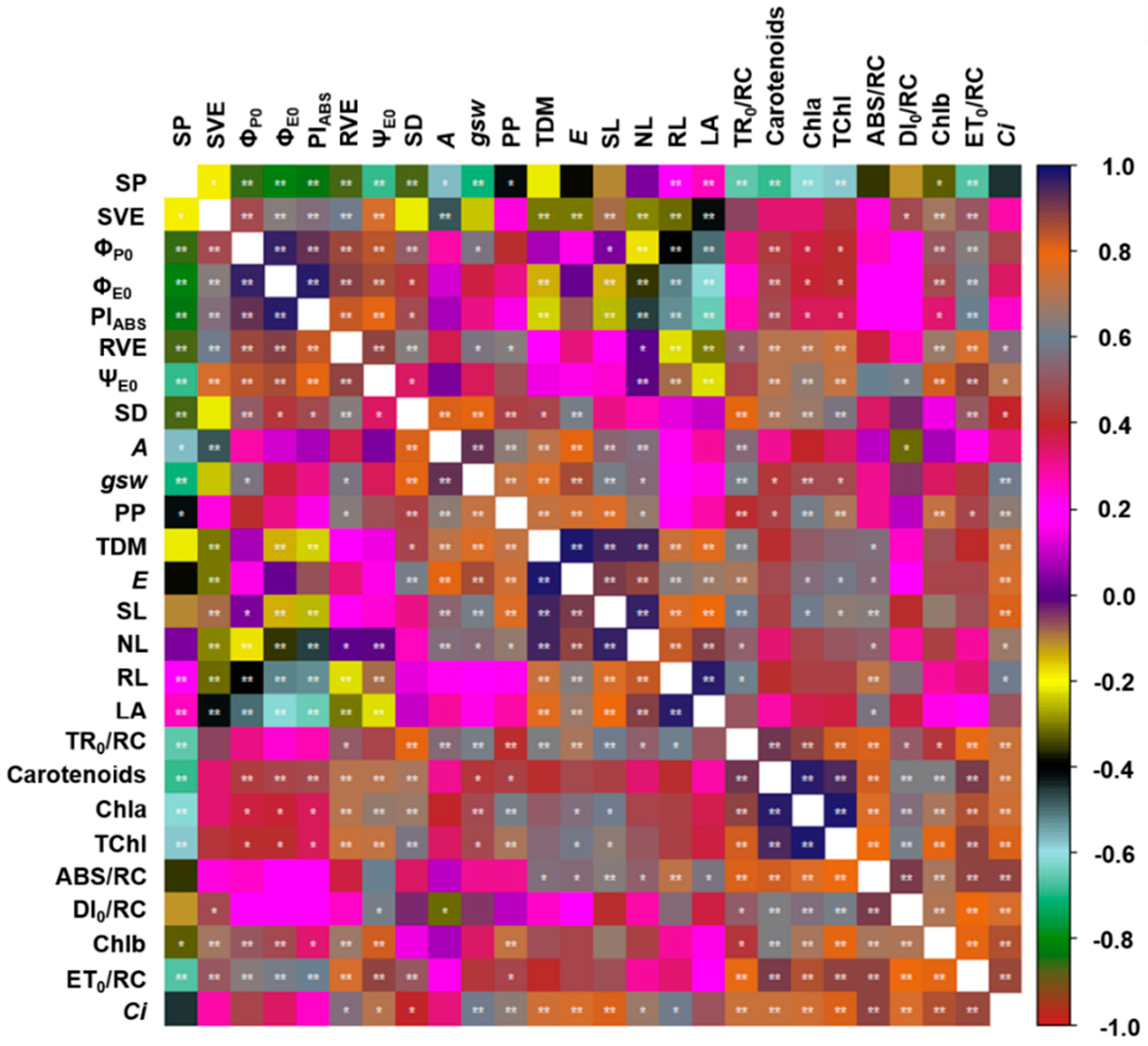
3.3. Assessment of Growth and the Interactions among the Variables
4. Discussion
4.1. Morpho-Anatomical Patterns
4.2. Physiological Patterns
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Maucieri, C.; Nicoletto, C.; Os, E.V.; Anseeeuw, D.; Havermaet, R.V.; Jungle, R. Aquaponics Food Production Systems. Combined Aquaculture and Hydroponic Production Technologies for the Future; Goddek, S., Joyce, A., Kotzen, B., Burnell, G., Eds.; Springer: Cham, Switzerland, 2019; pp. 97–110. [Google Scholar]
- Wootton-Beard, P. Growing without Soil—An Overview of Hydroponics; Aberystwyth University: Aberystwyth, UK, 2019; pp. 1–10. [Google Scholar]
- Venter, G. Different Types of Hydroponic Systems: Farming for Tomorrow. The Origins of Hydroponic Farming. Farmer’s Wkly. 2017, 2017, 26–27. [Google Scholar]
- Jia, X.; Wang, H.; Svetla, S.; Zhu, Y.F.; Hu, Y.; Cheng, L.; Zhao, T.; Wang, Y.X. Comparative physiological responses and adaptive strategies of apple Malus halliana to salt, alkali and saline-alkali stress. Sci. Hortic. 2019, 254, 154–162. [Google Scholar] [CrossRef]
- Molero, G.; Tcherkez, G.; Roca, R.; Mauve, C.; Cabrera-Bosquet, L.; Araus, J.L.; Nogués, S.; Aranjuelo, I. Do metabolic changes underpin physiological responses to water limitation in alfalfa (Medicago sativa) plants during a regrowth period? Agric. Water Manag. 2019, 212, 1–11. [Google Scholar] [CrossRef]
- Carillo, P.; Cirillo, C.; Micco, V.; Arena, C.; Pascale, S.; Rouphael, Y. Morpho-anatomical, physiological and biochemical adaptive responses to saline water of Bougainvillea spectabilis Willd. trained to different canopy shapes. Agric. Water Manag. 2019, 212, 12–22. [Google Scholar] [CrossRef]
- Kaur, A.; Thind, S.K. Phisiological adjustments of wheat to drought stress the influenced by osmoprotectants and cytokinins. Agric. Res. J. 2018, 55, 86–91. [Google Scholar] [CrossRef]
- Caine, R.S.; Yin, X.; Sloan, J.; Harrison, E.L.; Mohammed, U.; Fulton, T.; Biswal, A.K.; Dionora, J.; Chater, C.C.; Coe, R.A.; et al. Rice with reduced stomatal density conserves water and has improved drought tolerance under future climate conditions. New Phyt. 2019, 221, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Mäkelä, P.S.A.; Jokinen, K.; Himanen, K. Roles of endogenous glycinebetaine in plant abiotic stress responses. In Osmoprotectant-Mediated Abiotic Stress Tolerance in Plants; Hossain, M.A., Kumar, V., Burritt, D.J., Eds.; Springer: Cham, Switzerland, 2019; pp. 153–173. [Google Scholar] [CrossRef]
- Lee, Y.T.; Langenheim, J. Systematics of the Genus Hymenaea L. (Leguminosae, Caesalpinioideae, Detarieae); University of California Press: Berkley, CA, USA, 1975; pp. 1–190. [Google Scholar]
- Nascimento, H.H.C.; Nogueira, R.J.M.C.; Silva, E.C.; Silva, M.A. Analysis do growth de seedlings of jatobá (Hymenaea courbaril L.) em different níveis de water no soil. Rev. Árvore 2011, 35, 617–626. [Google Scholar] [CrossRef] [Green Version]
- Nascimento, H.H.C.; Santos, C.A.; Freire, C.S.; Silva, M.A.; Nogueira, R.J.M.C. Ajustamento osmótico em seedlings of jatobá submetidas à salinidade em meio hidropônico. Rev. Árvore 2015, 39, 641–653. [Google Scholar] [CrossRef] [Green Version]
- Silva-Júnior, M.C.; Felfili, J.M.; Walter, B.M.T.; Nogueira, P.E.; Rezende, A.V.; Morais, R.O.; Nóbrega, M.G.G. Analysis da flora arbórea de Matas de Galeria no Distrito Federal: 21 levantamentos. In Cerrado: Caracterização and Recuperação de Matas de Galeria; Ribeiro, J.F., Fonseca, C.E.L., Sousa-Silva, J.C., Eds.; Embrapa Cerrados: Planaltina, Brazil, 2001; pp. 143–191. [Google Scholar]
- Oliveira, W.L.; Medeiros, M.B.; Moser, P.; Pinheiro, R.; Olsen, L.B. Regeneração and estrutura populacional de jatobá-da-mata (Hymenaea courbaril L.), em dois fragmentos com different graus de perturbação antrópica. Act. Bot. Bras. 2011, 25, 876–884. [Google Scholar] [CrossRef]
- Bessa, L.A.; Moreira, M.A.; Silva, F.G.; Vitorino, L.C.; Megguer, C.A. The influence of nitrogen in nutrient solution on growth, nutrient uptake and enzymatic activity of Anacardium othonianum Rizz. Curr. Sci. 2019, 117, 12–25. [Google Scholar] [CrossRef]
- Bessa, L.A.; Vitorino, L.C.; Silva, F.G. Macronutrient Omission Affects the Seedling Performance of Eugenia dysenterica DC., an Important Fruiting Species of the Cerrado Biome. J. Agric. Sci. 2019, 11. [Google Scholar] [CrossRef]
- Souza, A.G.; Chalfun, N.N.J.; Faquin, V.; Souza, A.A. Production of peach grafts under hydroponic conditions. Ciênc. Agrotec. 2011, 35, 322–326. [Google Scholar] [CrossRef]
- Corrêa, R.M.; Pinto, S.I.C.; Reis, É.S.; Carvalho, V.A.M. Hydroponic Prodzuction of Fruit Tree Seedlings in Brazil; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef] [Green Version]
- Mendoza Castilho, V.M.; Pineda, J.P.; Canales, J.M.V.; Arguello, E.H. Nutrition of fig (Ficus carica L.) under hydroponics and greenhouse conditions. J. Plant Nutr. 2019, 42, 1350–1365. [Google Scholar] [CrossRef]
- Goulas, Y.; Cerovic, Z.G.; Cartelat, A.; Moya, I. Dualex: A new instrument for field measurements of epidermal ultraviolet absorbance by chlorophyll fluorescence. Appl. Opt. 2004, 43, 4488–4496. [Google Scholar] [CrossRef]
- Dong, T.; Shang, J.; Chen, J.M.; Liu, J.; Qian, B.; Ma, B.; Morrison, M.J.; Zhang, C.; Liu, Y.; Shi, Y.; et al. The sessment of portable chlorophyll meters for measuring crop leaf chlorophyll concentration. Remote Sens. 2019, 11, 2706. [Google Scholar] [CrossRef] [Green Version]
- Majeed, A.; Muhammad, Z. Salinity: A Major agricultural problem—Causes, impacts on crop productivity and management strategies. Plant Abiotic Stress Toler. 2019, 83–99. [Google Scholar] [CrossRef]
- Anjum, S.A.; The hraf, U.; Zohaib, A.; Tanveer, M.; Naeem, M.; Ali, I.; Tabassum, T.; Nazir, U. Growth and developmental responses of crop plants under drought stress: A review. Zemdirbyste 2017, 104, 267–276. [Google Scholar] [CrossRef]
- Carstensen, A.; Szameitat, A.E.; Frydenvang, J.; Husted, S. Chlorophyll a fluorescence analysis can detect phosphorus deficiency under field conditions and is an effective tool to prevent grain yield reductions in spring barley (Hordeum vulgare L.). Plant Soil 2019, 434, 79–91. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence the a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef] [Green Version]
- Farias, D.B.S.; Silva, P.S.O.; Lucas, A.A.T.; Freitas, M.I.; Santos, T.J.; Fontes, P.T.N.; Júnior, L.F.G.O. Physiological and productive parameters of the okra under irrigation levels. Sci. Hortic. 2019, 252, 1–6. [Google Scholar] [CrossRef]
- Silva, E.M.; Magalhães, P.C.; Castro, E.M.; Ávila, R.G.; Almeida, L.G. Modifications in the root system, gas exchanges and productivity of sorghum hybrids cultivated under water déficit. Rev. Bras. Milho Sorgo. 2018, 17, 380–389. [Google Scholar] [CrossRef]
- Embiale, A.; Hussein, M.; Husen, A.; Sahile, S.; Mohammed, K. differential sensitivity of Pisum sativum L. Cultivars to water-deficit stress: Changes in growth, water status, chlorophyll fluorescence and gas exchange attributes. J. Agron. 2016, 15, 45–57. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [Green Version]
- Posudin, Y.I.; Godlevska, O.O.; Zaloilo, I.A.; Kozhem’yako, Y.V. Application of portable fluorometer for estimation of plant tolerance to abiotic factors. Int. Agrophys. 2010, 24, 363–368. [Google Scholar]
- Douthe, C.; Gago, J.; Ribas-Carbó, M.; Núñez, R.; Pedro, N.; Flexas, J. Measuring photosynthesis and respiration with infrared gas analysers. Adv. Plant Ecophysiol. Tech. 2018, 51–75. [Google Scholar] [CrossRef]
- Poljakoff-Mayber, A. Morphological and anatomical changes in plants the a response to salinity stress. Plants Saline Environ. 1975, 97–117. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The Water Culture Method for Growing Plants without Soils; California Agricultural Experimental Station: Berkeley, CA, USA, 1950; pp. 1–347. [Google Scholar]
- Karnovsky, M.J. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol. 1965, 27, 137–138. [Google Scholar]
- O’brien, T.P.; Mccully, T.P.M.E. The Study of Plant Structure: Principles and Selected Methods; Termarcarphy Pty.: Melbourne, Australia, 1981. [Google Scholar]
- Ferreira, T.; Rasband, W. The ImageJ Guia do Usuário-Version. 2010, 1.43. Available online: http://rsbweb.nih.gov/ij/docs/user-guide.pdf (accessed on 5 March 2020).
- Jorge, L.I.F.; Mello, A.R.P.; Reis, C.M.P.V.; Ferro, V.O. Exame microscÛpico de Ficus enormis (Mart. ex Miq.) Miq. (gameleira) and de Hymenaea courbaril L. var. stilbocarpa (Hayne) Lee & Langenheim (jatobá). Rev. Bras. Farm. 2006, 87, 85–88. [Google Scholar]
- Barnes, J.D.; Balaguer, L.; Manrique, E.; Elvira, S.; Davison, A.W. A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environ. Exp. Bot. 1992, 32, 85–100. [Google Scholar] [CrossRef]
- Wellburn, A.R. The spectral determination of chlorophylls a and b, the well the total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1997, 144, 307–313. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The fluorescence transient the a tool to characterize and screen photosynthetic samples. In Probing Photosynthesis: Mechanism, Regulation and Adaptation; Yunus, M., Pathre, U., Mohanty, P., Eds.; Taylor and Francis: New York, NY, USA, 2000; pp. 445–483. [Google Scholar]
- Core Team, R. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 30 January 2020).
- Travassos, K.D.; Soares, F.A.L.; Gheyi, H.R.; Silva, D.R.S.; Nascimento, A.K.S.; Dias, N.S. Produção de aquênio do girassol irrigado com water salobra. Rev. Bras. Eng. Agríc. 2011, 15, 371–376. [Google Scholar] [CrossRef]
- Orsini, F.; Alnayef, M.; Bona, S.; Maggio, A.; Gianquinto, G. Low stomatal density and reduced transpiration facilitate strawberry adaptation to salinity. Environ. Exp. Bot. 2012, 81, 1–10. [Google Scholar] [CrossRef]
- Hughes, J.; Hepworth, C.; Dutton, C.; Dunn, J.A.; Hunt, L.; Stephens, J.; Waugh, R.; Cameron, D.D.; Gray, J.E. Reducing stomatal density in barley improves drought tolerance without impacting on yield. Plant Physiol. 2017, 174, 776–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cruz, Y.C.; Scarpa, A.L.M.; Pereira, M.P.; Castro, E.M.; Pereira, F.J. Growth of Typha domingensis the related to leaf physiological and anatomical modifications under drought conditions. Acta Physiol. Plant. 2019, 64, 41–64. [Google Scholar] [CrossRef]
- Silva, J.S.; Paz, V.P.S.; Soares, T.M.; Almeida, W.F.; Fernandes, J.P. Production of lettuce with brackish water in NFT hydroponic system. Semina Ciênc. Agrár. 2018, 39, 947–962. [Google Scholar] [CrossRef]
- Chadirin, Y.; Matsuoka, T.; Suhardiyanto, H.; Susila, A.D. Application of deep sea water (DSW) for nutrient supplement in hydroponics cultivation of tomato: Effect of supplemented DSW at different EC levels on fruit properties. Bul. Agron. 2007, 35, 118–126. [Google Scholar] [CrossRef]
- Sakamoto, K.; Kogi, M.; Yanagisawa, T. Effects of salinity and nutrients in seawater on hydroponic culture of red leaf lettuce. Environ. Control Biol. 2014, 52, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Soares, H.R.; Santos Júnior, J.A.; França e Silva, Ê.F.; Rolim, M.M.; Silva, G.F. Water and physiological relationships of lettuce cultivated in hydroponics with brackish Waters. Rev. Ciênc. Agron. 2019, 50, 216–222. [Google Scholar] [CrossRef]
- Martins, J.B.; Santos Júnior, J.A.; Silva Júnior, F.J.D.; Silva, G.F.D.; Medeiros, S.D.S. Production of parsley in hydroponic conditions under isosmotic brackish nutrient solutions. Ciênc. Agrotec. 2019, 43. [Google Scholar] [CrossRef] [Green Version]
- Pompelli, M.F.; Mendes, K.R.; Ramos, M.V.; Santos, J.N.B.; Youssef, D.T.A.; Pereira, J.D.; Endres, L.; Jarma-Orozco, A.; Solano-Gomes, R.; Jarma-Arroyo, B.; et al. Mesophyll thickness and sclerophylly among Calotropis procera morphotypes reveal water-saved adaptation to environments. J. Arid Land 2019, 11, 795–810. [Google Scholar] [CrossRef] [Green Version]
- Thangthong, N.; Jogloy, S.; Punjansing, T.; Kvien, C.K.; Kesmala, T.; Vorasoot, N. Changes in root anatomy of peanut (Arachis hypogaea L.) under different durations of early season drought. Agronomy 2019, 9, 215. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Guo, P.; Peng, H. An ignored anatomical variable: Pore shape shows a nonrandom variation pattern in xylem cross sections. Nord. J. Bot. 2019, 37, 5. [Google Scholar] [CrossRef]
- Dai, Y.; Yuan, L.; Zhang, S.; Wang, J.; Xie, S.; Zhao, M.; Chen, G.; Sun, R.; Wang, C. Comprehensive evaluation for cold tolerance in wucai (Brassica campestris L.) by the performance index on an absorption basis (PIabs). Agronomy 2019, 9, 61. [Google Scholar] [CrossRef] [Green Version]
- Galić, V.; Mazur, M.; Šimić, D.; Zdunić, Z.; Franic, M. Plant biomass in salt-stressed young maize plants can be modelled with photosynthetic performance. Photosynthetica 2019, 57, 9–19. [Google Scholar] [CrossRef] [Green Version]
- Sampaio, O.M.; Silva, M.F.G.F. Evaluation de furanocumarinas como inibidores da fotossíntese através de ensaios de fluorescência da chlorophyll a. Química Nova 2012, 35, 2115–2118. [Google Scholar] [CrossRef] [Green Version]
- Estaji, A.; Kalaji, H.M.; Karimi, H.R.; Roosta, H.R.; Moosavi-Nezhad, S.M. How glycine betaine induces tolerance of cucumber plants to salinity stress? Photosynthetica 2019, 57, 3. [Google Scholar] [CrossRef] [Green Version]
- Khatri, K.; Rathore, M.S. Photosystem photochemistry, prompt and delayed fluorescence, photosynthetic responses and electron flow in tobacco under drought and salt stress. Photosynthetica 2019, 57, 61–74. [Google Scholar] [CrossRef]
- Xia, J.; Tian, Q. Early stage toxicity of excess copper to photosystem II of Chlorella pyrenoidosa–OJIP chlorophyll a fluorescence analysis. Int. J. Environ. Sci. 2009, 21, 1569–1574. [Google Scholar] [CrossRef]
- Umar, M.; Uddin, Z.; Siddiqui, Z.S. Responses of photosynthetic apparatus in sunflower cultivars to combined drought and salt stress. Photosynthetica 2019, 57, 627–639. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.Y.; Nie, H.S.; Yu, H.Y.; Kong, D.S.; Wu, J.Y. Effect of salt stress on the growth and photosystem II photochemical characteristics of Lycium ruthenicum Murr. Seedlings. Photosynthetica 2019, 57, 564–571. [Google Scholar] [CrossRef] [Green Version]
- Bertolino, L.T.; Caine, R.S.; Gray, J.E. Impact of stomatal density and morphology on water-use efficiency in a changing world. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunn, J.; Hunt, L.; Afsharinafar, M.; Meselmani, M.A.; Mitchell, A.; Howells, R.; Wallington, E.; Fleming, A.J.; Gray, J. Reduced stomatal density in bread wheat leads to increased water-use efficiency. J. Exp. Bot. 2019, 4737–4748. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, H.; Uragami, C.; Cogdell, R.J. Carotenoids and photosynthesis. Carotenoids Nat. 2016, 111–139. [Google Scholar] [CrossRef]
- Dautermann, O.; Lohr, M. A functional zeaxanthin epoxidase from red algae shedding light on the evolution of light-harvesting carotenoids and the xanthophyll cycle in photosynthetic eukaryotes. Plant J. 2017, 92, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Gago, J.; Daloso, D.M.; Figueroa, C.M.; Flexas, J.; Fernie, A.R.; Nikoloski, Z. Relationships of leaf net photosynthesis, stomatal conductance, and mesophyll conductance to primary metabolism: A multispecies meta-analysis approach. Plant Physiol. 2016, 171, 265–279. [Google Scholar] [CrossRef]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, D.d.C.V.d.; Bessa, L.A.; Silva, F.G.; Rosa, M.; Vasconcelos Filho, S.d.C.; Vitorino, L.C. Morpho-Anatomical and Physiological Responses Can Predict the Ideal Period for the Transplantation of Hydroponic Seedlings of Hymenaea courbaril, a Neotropical Fruit Tree. Plants 2020, 9, 721. https://doi.org/10.3390/plants9060721
Sousa DdCVd, Bessa LA, Silva FG, Rosa M, Vasconcelos Filho SdC, Vitorino LC. Morpho-Anatomical and Physiological Responses Can Predict the Ideal Period for the Transplantation of Hydroponic Seedlings of Hymenaea courbaril, a Neotropical Fruit Tree. Plants. 2020; 9(6):721. https://doi.org/10.3390/plants9060721
Chicago/Turabian StyleSousa, Daniele de Cássia Vieira de, Layara Alexandre Bessa, Fabiano Guimarães Silva, Márcio Rosa, Sebastião de Carvalho Vasconcelos Filho, and Luciana Cristina Vitorino. 2020. "Morpho-Anatomical and Physiological Responses Can Predict the Ideal Period for the Transplantation of Hydroponic Seedlings of Hymenaea courbaril, a Neotropical Fruit Tree" Plants 9, no. 6: 721. https://doi.org/10.3390/plants9060721





