Changes in the Phenolic Compounds of Hop (Humulus lupulus L.) Induced by Infection with Verticillium nonalfalfae, the Causal Agent of Hop Verticillium Wilt
Abstract
:1. Introduction
2. Results
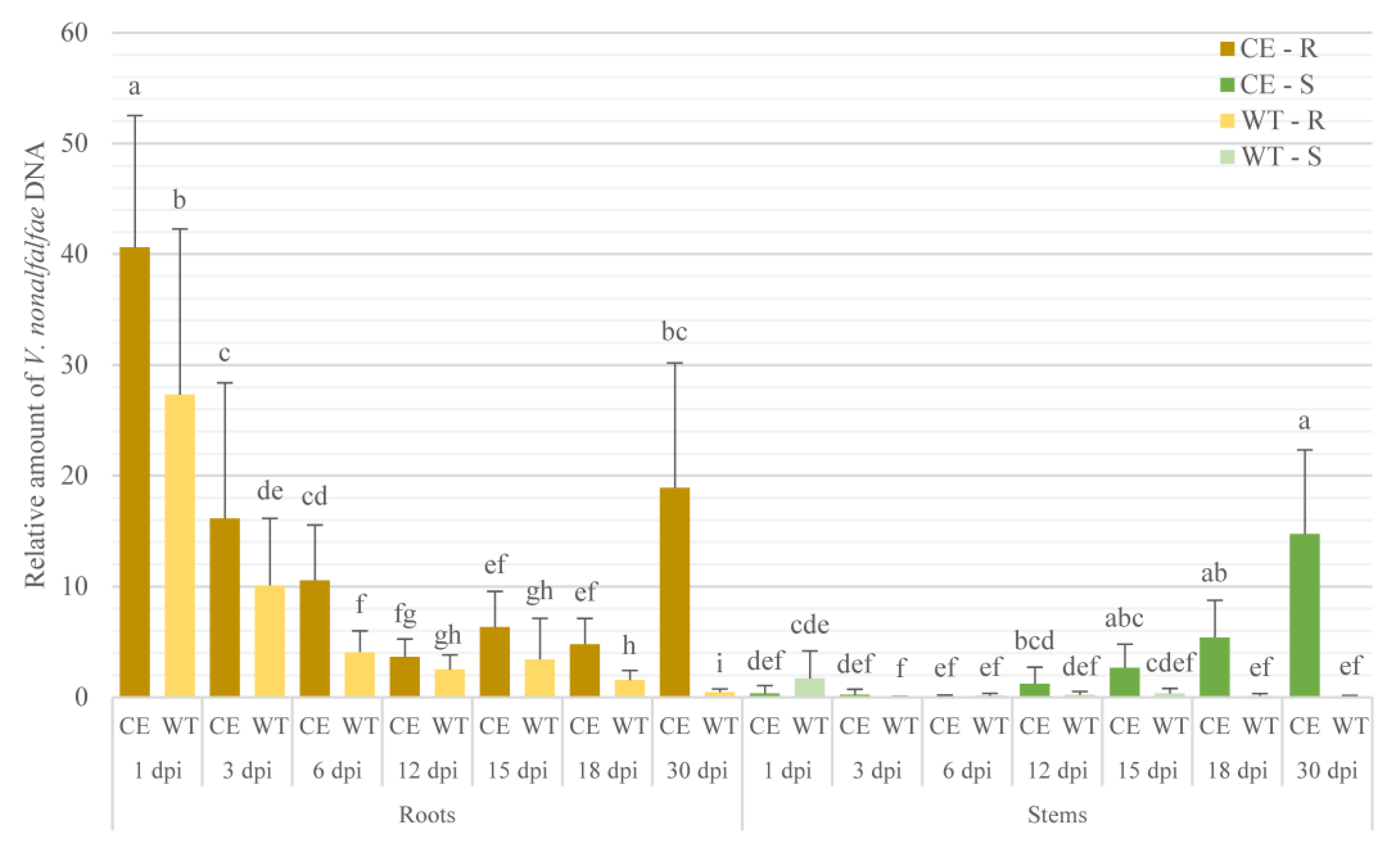
2.1. V. nonalfalfae Colonization Profile Analysis in Susceptible and Resistant Hop Cultivars
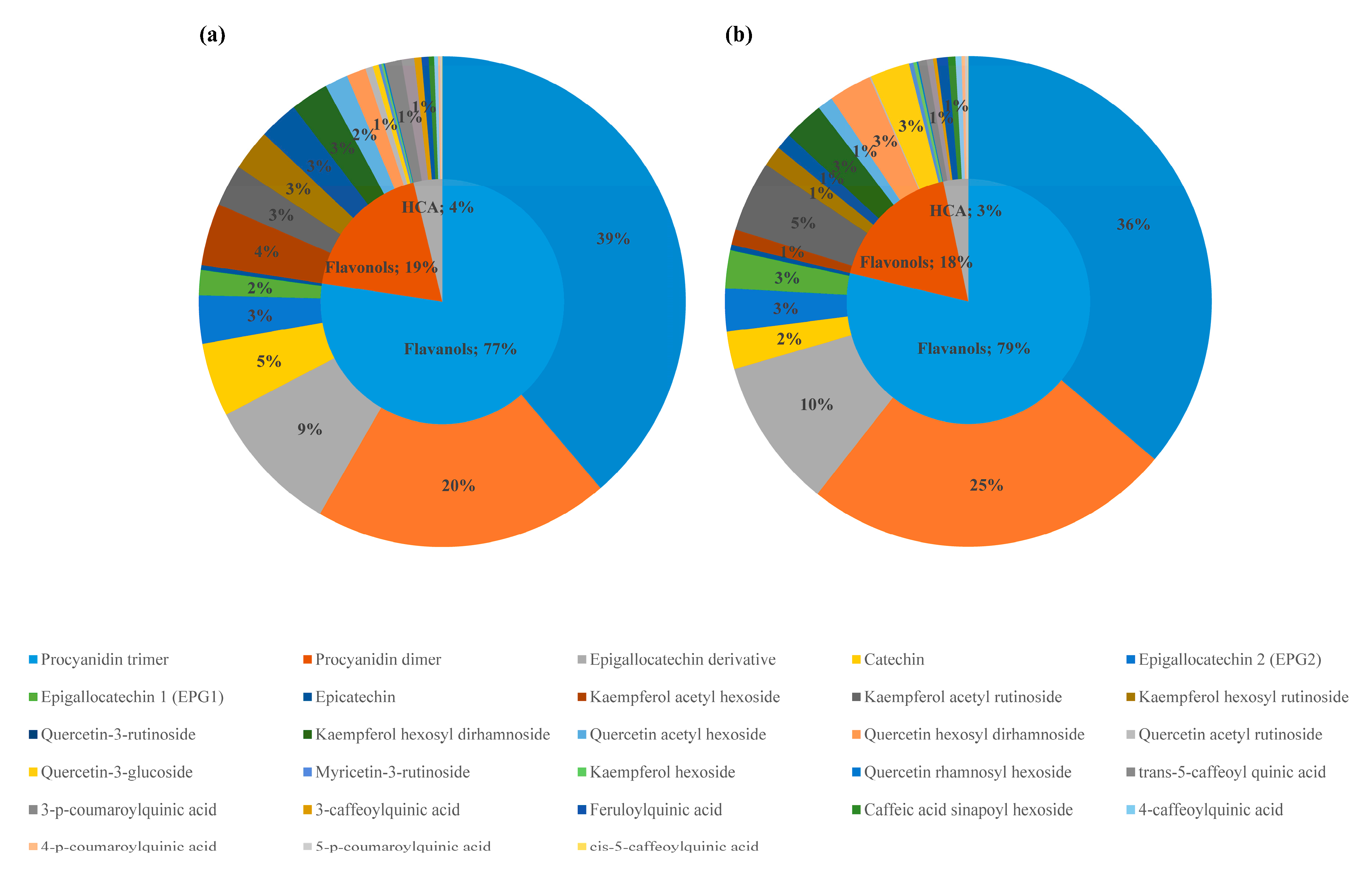
2.2. Phenolic Compounds
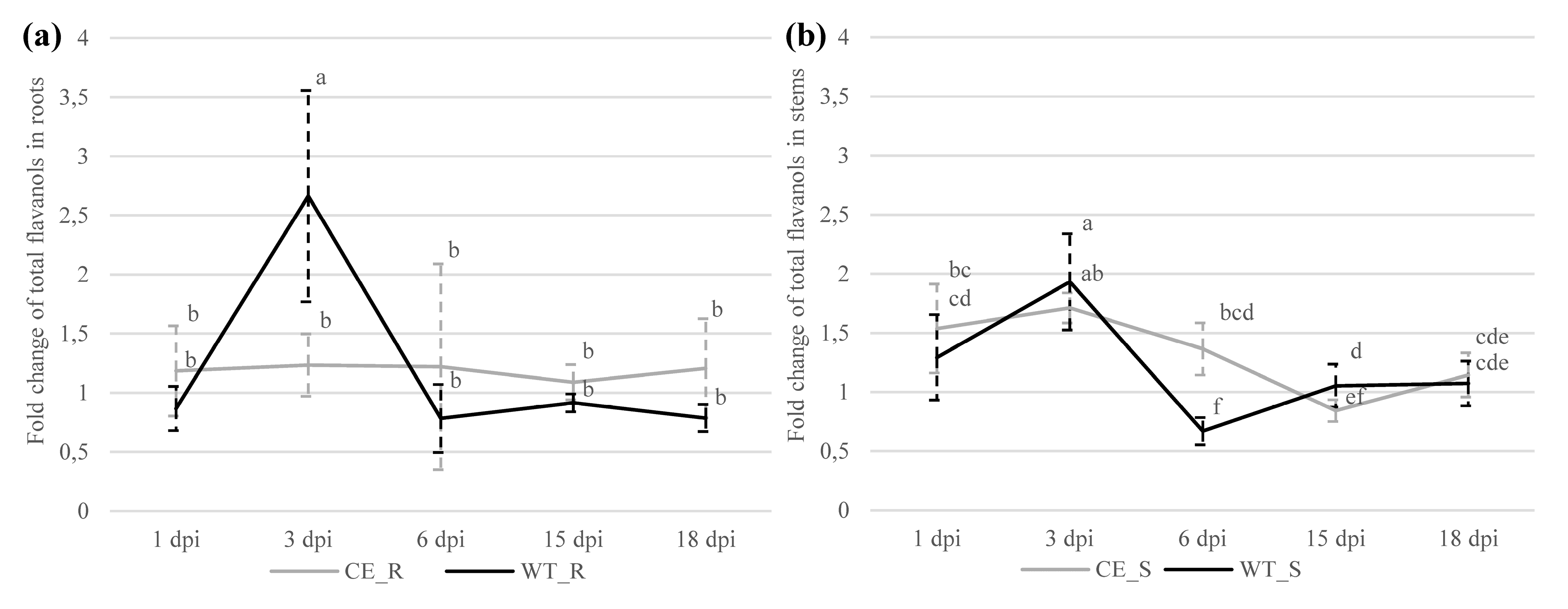
2.2.1. Flavanols
2.2.2. Flavonol Derivatives
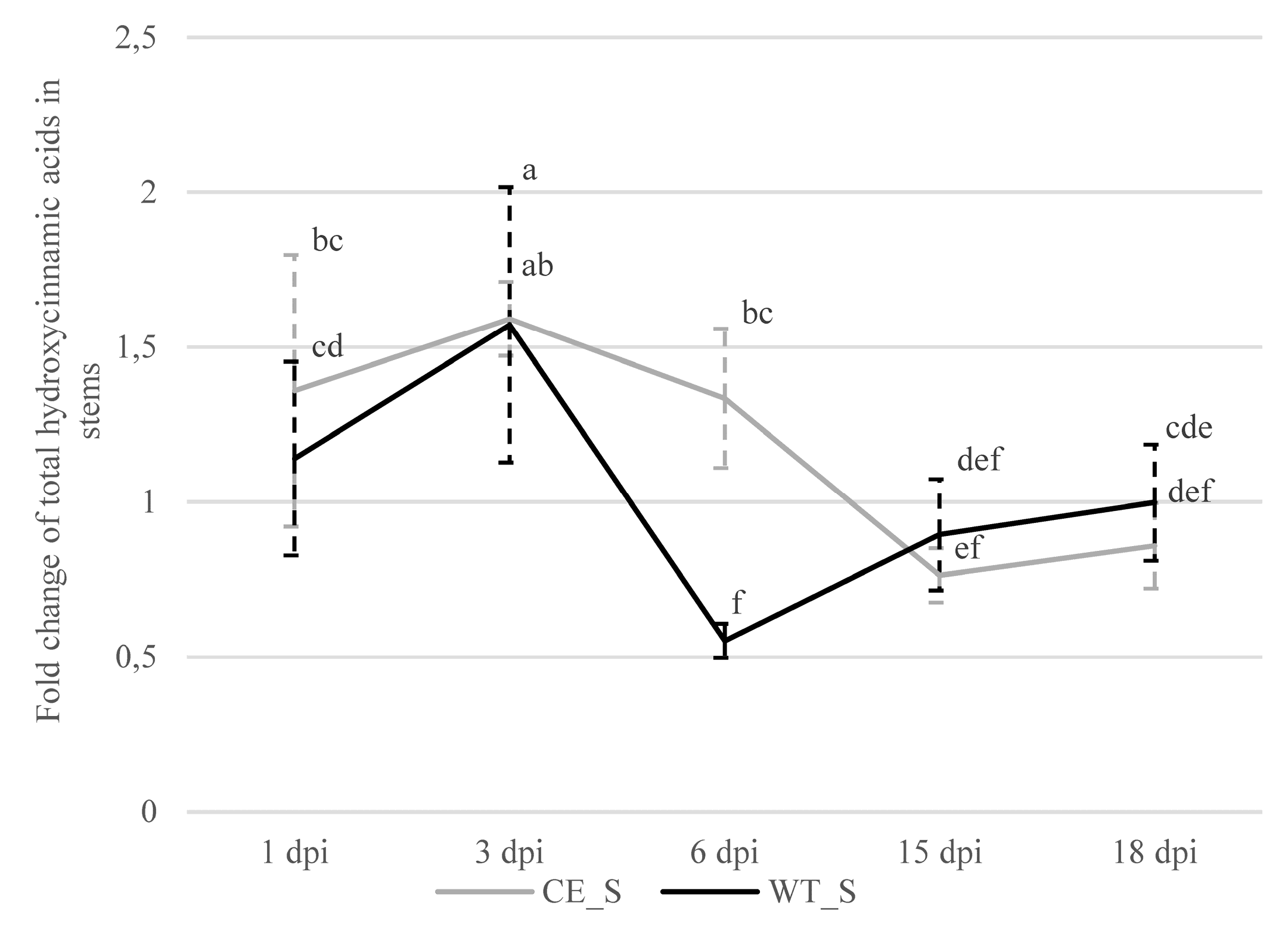
2.2.3. Hydroxycinnamic Acids
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. V. nonalfalfae qPCR Quantification
4.3. Extraction and Determination of Phenolic Compounds Using HPLC–Diode-Array Detector (DAD)–MSn Analysis
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Steenackers, B.; De Cooman, L.; De Vos, D. Chemical Transformations of Characteristic Hop Secondary Metabolites in Relation to Beer Properties and the Brewing Process: A Review. Food Chem. 2015, 172, 742–756. [Google Scholar] [CrossRef] [PubMed]
- Neve, R.A. Hops; Springer: Dordrecht, The Netherlands, 1991. [Google Scholar] [CrossRef]
- IHGC. “International Hop Growers’ Convention: Economic Commission—Summary Reports.” International Hop Growers’ Convention. Available online: http://www.hmelj-giz.si/ihgc/doc/2019%20APR%20IHGC%20EC%20Report_final.pdf (accessed on 22 November 2019).
- Radisek, S.; Jakse, J.; Javornik, B. Genetic Variability and Virulence among Verticillium albo-atrum Isolates from Hop. Eur. J. Plant Pathol. 2006, 116, 301–314. [Google Scholar] [CrossRef]
- Yadeta, K.; Thomma, B.P.H.J. The Xylem as Battleground for Plant Hosts and Vascular Wilt Pathogens. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.Y.; Xiao, H.L.; Gui, Y.J.; Zhang, D.D.; Li, L.; Bao, Y.M.; Dai, X.F. Characterization of the Verticillium Dahliae Exoproteome Involves in Pathogenicity from Cotton-Containing Medium. Front. Microbiol. 2016, 7, 1709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fradin, E.F.; Thomma, B.P.H.J. Physiology and Molecular Aspects of Verticillium Wilt Diseases Caused by V. dahliae and V. albo-atrum. Mol. Plant Pathol. 2006, 7, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Carroll, C.L.; Carter, C.A.; Goodhue, R.E.; Lawell, C.Y.C.L.; Subbarao, K.V. A Review of Control Options and Externalities for Verticillium Wilts. Phytopathology 2018, 108, 160–171. [Google Scholar] [CrossRef] [Green Version]
- Vermerris, W.; Nicholson, R. (Eds.) The Role of Phenols in Plant Defense. In Phenolic Compound Biochemistry; Springer: Dordrecht, The Netherlands, 2008; pp. 211–234. [Google Scholar] [CrossRef]
- Osbourn, A.E. Preformed Antimicrobial Compounds and Plant Defense against Fungal Attack. Plant Cell 1996, 8, 1821–1831. [Google Scholar] [CrossRef]
- VanEtten, H.D.; Mansfield, J.W.; Bailey, J.A.; Farmer, E.E. 2 Classes of Plant Antibiotics—Phytoalexins Versus Phytoanticipins. Plant Cell 1994, 6, 1191–1192. [Google Scholar] [CrossRef]
- Jones, J.D.; Dangl, J.L. The Plant Immune System. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Mikulic-Petkovsek, M.; Ivancic, A.; Todorovic, B.; Veberic, R.; Stampar, F. Fruit Phenolic Composition of Different Elderberry Species and Hybrids. J. Food Sci. 2015, 80, C2180–C2190. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Schmitzer, V.; Slatnar, A.; Weber, N.; Veberic, R.; Stampar, F.; Munda, A.; Koron, D. Alteration of the Content of Primary and Secondary Metabolites in Strawberry Fruit by Colletotrichum Nymphaeae Infection. J. Agric. Food Chem. 2013, 61, 5987–5995. [Google Scholar] [CrossRef] [PubMed]
- Obermeier, C.; Hossain, M.A.; Snowdon, R.; Knufer, J.; von Tiedemann, A.; Friedt, W. Genetic Analysis of Phenylpropanoid Metabolites Associated with Resistance against Verticillium longisporum in Brassica napus. Mol. Breed. 2013, 31, 347–361. [Google Scholar] [CrossRef]
- Eynck, C.; Koopmann, B.; Karlovsky, P.; von Tiedemann, A. Internal Resistance in Winter Oilseed Rape Inhibits Systemic Spread of the Vascular Pathogen Verticillium longisporum. Phytopathology 2009, 99, 802–811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mikulic-Petkovsek, M.; Stampar, F.; Veberic, R. Accumulation of Phenolic Compounds in Apple in Response to Infection by the Scab Pathogen, Venturia inaequalis. Physiol. Mol. Plant Pathol. 2009, 74, 60–67. [Google Scholar] [CrossRef]
- El-Bebany, A.F.; Adam, L.R.; Daayf, F. Differential Accumulation of Phenolic Compounds in Potato in Response to Weakly and Highly Aggressive Isolates of Verticillium dahliae. Can. J. Plant Pathol. 2013, 35, 232–240. [Google Scholar] [CrossRef]
- Bily, A.C.; Reid, L.M.; Taylor, J.H.; Johnston, D.; Malouin, C.; Burt, A.J.; Bakan, B.; Regnault-Roger, C.; Pauls, K.P.; Arnason, J.T.; et al. Dehydrodimers of Ferulic Acid in Maize Grain Pericarp and Aleurone: Resistance Factors to Fusarium graminearum. Phytopathology 2003, 93, 712–719. [Google Scholar] [CrossRef] [Green Version]
- Mandal, S.; Mitra, A. Accumulation of Cell Wall-Bound Phenolic Metabolites and Their Upliftment in Hairy Root Cultures of Tomato (Lycopersicon esculentum Mill). Biotechnol. Lett. 2008, 30, 1253–1258. [Google Scholar] [CrossRef]
- Talboys, P.W. Some Mechanisms Contributing to Verticillium-Resistance in the Hop Root. Trans. Br. Mycol. Soc. 1958, 41, 227–241. [Google Scholar] [CrossRef]
- Talboys, P.W. Degradation of Cellulose by Verticillium albo-atrum. Trans. Br. Mycol. Soc. 1958, 41, 242–248. [Google Scholar] [CrossRef]
- Talboys, P.W. Association of Tylosis and Hyperplasia of the Xylem with Vascular Invasion of the Hop by Verticillium albo-atrum. Trans. Br. Mycol. Soc. 1958, 41, 249–260. [Google Scholar] [CrossRef]
- Trevisan, M.T.S.; Valdivia, A.C.R.; Scheffer, J.J.C.; Verpoorte, R. Enzyme Activities in Cell Suspension Cultures of Two Hop Cultivars after Elicitation by a Fungal Culture Filtrate. Biotechnol. Lett. 1997, 19, 207–211. [Google Scholar] [CrossRef]
- Lawton, M.A.; Lamb, C.J. Transcriptional Activation of Plant Defense Genes by Fungal Elicitor, Wounding, and Infection. Mol. Cell Biol. 1987, 7, 335–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landa, B.B.; Perez, A.G.; Luaces, P.; Montes-Borrego, M.; Navas-Cortes, J.A.; Sanz, C. Insights into the Effect of Verticillium dahliae Defoliating-Pathotype Infection on the Content of Phenolic and Volatile Compounds Related to the Sensory Properties of Virgin Olive Oil. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markakis, E.A.; Tjamos, S.E.; Antoniou, P.P.; Roussos, P.A.; Paplomatas, E.J.; Tjamos, E.C. Phenolic Responses of Resistant and Susceptible Olive Cultivars Induced by Defoliating and Nondefoliating Verticillium dahliae Pathotypes. Plant Dis. 2010, 94, 1156–1162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talboys, P.W. Verticillium Wilt in English Hops—Retrospect and Prospect. Can. J. Plant Pathol. 1987, 9, 68–77. [Google Scholar] [CrossRef]
- Toueni, M.; Ben, C.; Le Ru, A.; Gentzbittel, L.; Rickauer, M. Quantitative Resistance to Verticillium Wilt in Medicago truncatula Involves Eradication of the Fungus from Roots and Is Associated with Transcriptional Responses Related to Innate Immunity. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cregeen, S.; Radisek, S.; Mandelc, S.; Turk, B.; Stajner, N.; Jakse, J.; Javornik, B. Different Gene Expressions of Resistant and Susceptible Hop Cultivars in Response to Infection with a Highly Aggressive Strain of Verticillium albo-atrum. Plant Mol. Biol. Rep. 2015, 33, 689–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jakse, J.; Cerenak, A.; Radisek, S.; Satovic, Z.; Luthar, Z.; Javornik, B. Identification of Quantitative Trait Loci for Resistance to Verticillium Wilt and Yield Parameters in Hop (Humulus lupulus L.). Theor. Appl. Genet. 2013, 126, 1431–1443. [Google Scholar] [CrossRef]
- Sewell, G.W.F.; Wilson, J.F. The Influence of Normal Tillage and of Non—Cultivation on Verticillium Wilt of the Hop. Ann. Appl. Biol. 1974, 76, 37–47. [Google Scholar] [CrossRef]
- Sewell, G.W.F.; Wilson, J.F. Verticillium Wilt of the Hop: Field Studies on Wilt in a Resistant Cultivar in Relation to Nitrogen Fertilizer Applications. Ann. Appl. Biol. 1966, 59, 265–273. [Google Scholar] [CrossRef]
- Zhang, W.W.; Jiang, T.F.; Cui, X.; Qi, F.J.; Jian, G.L. Colonization in Cotton Plants by a Green Fluorescent Protein Labelled Strain of Verticillium dahliae. Eur. J. Plant Pathol. 2013, 135, 867–876. [Google Scholar] [CrossRef]
- Eynck, C.; Koopmann, B.; Grunewaldt-Stoecker, G.; Karlovsky, P.; von Tiedemann, A. Differential Interactions of Verticillium longisporum and V-dahliae with Brassica napus Detected with Molecular and Histological Techniques. Eur. J. Plant Pathol. 2007, 118, 259–274. [Google Scholar] [CrossRef]
- Vallad, G.E.; Subbarao, K.V. Colonization of Resistant and Susceptible Lettuce Cultivars by a Green Fluorescent Protein-Tagged Isolate of Verticillium dahliae. Phytopathology 2008, 98, 871–885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ullah, C.; Unsicker, S.B.; Fellenberg, C.; Constabel, C.P.; Schmidt, A.; Gershenzon, J.; Hammerbacher, A. Flavan-3-Ols Are an Effective Chemical Defense against Rust Infection. Plant Physiol. 2017, 175, 1560–1578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammerbacher, A.; Paetz, C.; Wright, L.P.; Fischer, T.C.; Bohlmann, J.; Davis, A.J.; Fenning, T.M.; Gershenzon, J.; Schmidt, A. Flavan-3-Ols in Norway Spruce: Biosynthesis, Accumulation, and Function in Response to Attack by the Bark Beetle-Associated Fungus Ceratocystis polonica. Plant Physiol. 2014, 164, 2107–2122. [Google Scholar] [CrossRef] [Green Version]
- Mace, M.E.; Howell, C.R. Histochemistry and Identification of Condensed Tannin Precursors in Roots of Cotton Seedlings. Can. J. Bot. 1974, 52, 2423–2426. [Google Scholar] [CrossRef]
- Daayf, F.; Nicole, M.; Boher, B.; Pando, A.; Geiger, J.P. Early Vascular Defense Reactions of Cotton Roots Infected with a Defoliating Mutant Strain of Verticillium dahliae. Eur. J. Plant Pathol. 1997, 103, 125–136. [Google Scholar] [CrossRef]
- El Modafar, C.; Clerivet, A.; Macheix, J.J. Flavan Accumulation in Stems of Platanus X Acerifolia Seedlings Inoculated with Ceratocystis fimbriata f sp. Platani, the Canker Stain Disease Agent. Can. J. Bot. 1996, 74, 1982–1987. [Google Scholar] [CrossRef]
- Madadkhah, E.; Lotfi, M.; Nabipour, A.; Rahmanpour, S.; Banihashemi, Z.; Shoorooei, M. Enzymatic Activities in Roots of Melon Genotypes Infected with Fusarium oxysporum f. sp. Melonis Race 1. Sci. Hortic. 2012, 135, 171–176. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Schmitzer, V.; Stampar, F.; Veberic, R.; Koron, D. Changes in Phenolic Content Induced by Infection with Didymella applanata and Leptosphaeria coniothyrium, the Causal Agents of Raspberry Spur and Cane Blight. Plant Pathol. 2014, 63, 185–192. [Google Scholar] [CrossRef]
- Baidez, A.G.; Gomez, P.; Del Rio, J.A.; Ortuno, A. Dysfunctionality of the Xylem in Olea europaea L. Plants Associated with the Infection Process by Verticillium dahliae Kleb. Role of Phenolic Compounds in Plant Defense Mechanism. J. Agric. Food Chem. 2007, 55, 3373–3377. [Google Scholar] [CrossRef] [PubMed]
- De Farias Viegas Aquije, G.M.; Zorzal, P.B.; Buss, D.S.; Ventura, J.A.; Fernandes, P.M.; Fernandes, A.A.R. Cell Wall Alterations in the Leaves of Fusariosis-Resistant and Susceptible Pineapple Cultivars. Plant Cell Rep. 2010, 29, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- El Modafar, C.; Tantaoui, A.; El Boustani, E. Changes in Cell Wall-Bound Phenolic Compounds and Lignin in Roots of Date Palm Cultivars Differing in Susceptibility to Fusarium oxysporum f. sp. Albedinis. J. Phytopathol. 2000, 148, 405–411. [Google Scholar] [CrossRef]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin Biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef]
- Kang, Z.; Buchenauer, H. Ultrastructural and Immunocytochemical Investigation of Pathogen Development and Host Responses in Resistant and Susceptible Wheat Spikes Infected by Fusarium culmorum. Physiol. Mol. Plant Pathol. 2000, 57, 255–268. [Google Scholar] [CrossRef]
- Parker, D.; Beckmann, M.; Zubair, H.; Enot, D.P.; Caracuel-Rios, Z.; Overy, D.P.; Snowdon, S.; Talbot, N.J.; Draper, J. Metabolomic Analysis Reveals a Common Pattern of Metabolic Re-Programming During Invasion of Three Host Plant Species by Magnaporthe grisea. Plant J. 2009, 59, 723–737. [Google Scholar] [CrossRef]
- Maurya, S.; Singh, R.; Singh, D.P.; Singh, H.B.; Srivastava, J.S.; Singh, U.P. Phenolic Compounds of Sorghum vulgare in Response to Sclerotium rolfsii Infection. J. Plant Interact. 2007, 2, 25–29. [Google Scholar] [CrossRef]
- Njoroge, S.M.C.; Vallad, G.E.; Park, S.Y.; Kang, S.; Koike, S.T.; Bolda, M.; Burman, R.; Polonik, W.; Subbarao, K.V. Phenological and Phytochemical Changes Correlate with Differential Interactions of Verticillium dahliae with Broccoli and Cauliflower. Phytopathology 2011, 101, 523–534. [Google Scholar] [CrossRef] [Green Version]
- Osbourn, A.E.; Clarke, B.R.; Lunness, P.; Scott, P.R.; Daniels, M.J. An Oat Species Lacking Avenacin Is Susceptible to Infection by Gaeumannomyces-Graminis Var Tritici. Physiol. Mol. Plant Pathol. 1994, 45, 457–467. [Google Scholar] [CrossRef]
- Lo, S.C.C.; De Verdier, K.; Nicholson, R.L. Accumulation of 3-Deoxyanthocyanidin Phytoalexins and Resistance to Colletotrichum Sublineolum in Sorghum. Physiol. Mol. Plant Pathol. 1999, 55, 263–273. [Google Scholar] [CrossRef]
- Tweedy, B.G. Inorganic Sulfur as a Fungicide. Residue Rev. 1981, 78, 43–68. [Google Scholar]
- Cooper, R.M.; Williams, J.S. Elemental Sulphur as an Induced Antifungal Substance in Plant Defence. J. Exp. Bot. 2004, 55, 1947–1953. [Google Scholar] [CrossRef] [PubMed]
- Bollig, K.; Specht, A.; Myint, S.S.; Zahn, M.; Horst, W.J. Sulphur Supply Impairs Spread of Verticillium Dahliae in Tomato. Eur. J. Plant Pathol. 2013, 135, 81–96. [Google Scholar] [CrossRef]
- Flajsman, M.; Radisek, S.; Javornik, B. Pathogenicity Assay of Verticillium nonalfalfae on Hop Plants. Bio-Protocol 2017, 7. [Google Scholar] [CrossRef]
- Kump, B.; Javornik, B. Evaluation of Genetic Variability among Common Buckwheat (Fagopyrum esculentum Moench) Populations by Rapd Markers. Plant Sci. 1996, 114, 149–158. [Google Scholar] [CrossRef]
- Walker, N.J. A Technique Whose Time Has Come. Science 2002, 296, 557–559. [Google Scholar] [CrossRef]
- Stajner, N.; Cregeen, S.; Javornik, B. Evaluation of Reference Genes for Rt-Qpcr Expression Studies in Hop (Humulus lupulus L.) During Infection with Vascular Pathogen Verticillium albo-atrum. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative Pcr and the 2−Δδct Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Koron, D.; Zorenc, Z.; Veberic, R. Do Optimally Ripe Blackberries Contain the Highest Levels of Metabolites? Food Chem. 2017, 215, 41–49. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 22 November 2019).
- de Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.2.8. 2017. Available online: https://CRAN.R-project.org/package=agricolae (accessed on 22 November 2019).

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunej, U.; Mikulič-Petkovšek, M.; Radišek, S.; Štajner, N. Changes in the Phenolic Compounds of Hop (Humulus lupulus L.) Induced by Infection with Verticillium nonalfalfae, the Causal Agent of Hop Verticillium Wilt. Plants 2020, 9, 841. https://doi.org/10.3390/plants9070841
Kunej U, Mikulič-Petkovšek M, Radišek S, Štajner N. Changes in the Phenolic Compounds of Hop (Humulus lupulus L.) Induced by Infection with Verticillium nonalfalfae, the Causal Agent of Hop Verticillium Wilt. Plants. 2020; 9(7):841. https://doi.org/10.3390/plants9070841
Chicago/Turabian StyleKunej, Urban, Maja Mikulič-Petkovšek, Sebastjan Radišek, and Nataša Štajner. 2020. "Changes in the Phenolic Compounds of Hop (Humulus lupulus L.) Induced by Infection with Verticillium nonalfalfae, the Causal Agent of Hop Verticillium Wilt" Plants 9, no. 7: 841. https://doi.org/10.3390/plants9070841






