Osteosarcoma MRI Image-Assisted Segmentation System Base on Guided Aggregated Bilateral Network
Abstract
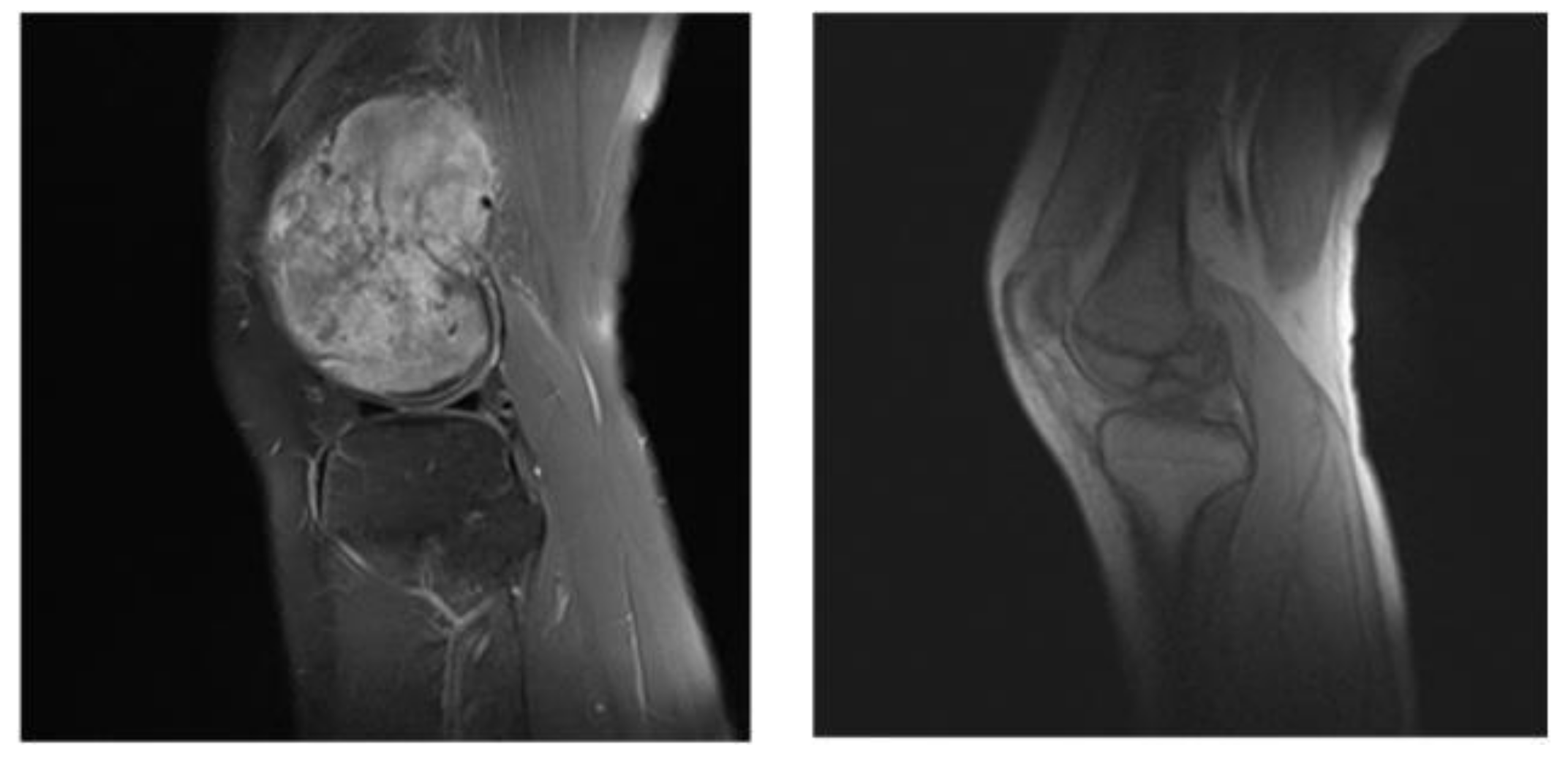
:1. Introduction
- (1)
- This paper uses the mean teacher model to optimize the classification of the data set, divide the data set into US and NS, and input them into the network in different orders for training, which further improves the training efficiency.
- (2)
- We use the fusion noise reduction method to preprocess the image, which improves the accuracy of model training and improves the robustness of the model.
- (3)
- This paper adopts the image segmentation model based on the fast bilateral segmentation network. While achieving high-precision segmentation, it uses an extremely lightweight model which greatly improves the speed of segmentation, so it has great significance in practical applications.
- (4)
- We used more than 80,000 samples collected by the Second Xiangya Hospital of Central South University for experimental analysis. The structure shows that our osteosarcoma segmentation method has a better segmentation effect than other methods, and the model is highly lightweight, which is convenient for training and application. This method is of great significance to the diagnosis, treatment and prognosis of osteosarcoma. Doctors can use the diagnosis results as an auxiliary basis for diagnosis and treatment, and greatly reduce the pressure of doctors and the time of diagnosis without affecting the accuracy.
2. Related Works
3. Methods
- (1)
- Data set optimization. We use the mean-teacher model to first divide the dataset into useful-slices (US) and Normal-Slices (NS), and then input them to the next step of processing.
- (2)
- Preprocessing. In the denoising process, we improve the median filter by adding more features, which results in a simple and effective denoising method for osteosarcoma MRI images and significantly reduces various noise interference. At the same time, we use curvelet transform for image enhancement to locally enhance the tumor region to further reduce unwanted noise in the inputted image.
- (3)
- Guided aggregation bilateral network segmentation algorithm. In this algorithm, we deal with the low-level details and high-level semantics separately to achieve high-precision and high-efficiency real-time semantic segmentation. In addition, we design a guided aggregation layer to enhance the interconnection and fusion of two types of feature representations, with good segmentation performance.
3.1. Dataset Optimization
- (1)
- Input and into the student model, and the output is the predicted probability ; input into the teacher model, and output the predicted probability ;
- (2)
- Calculate the loss value according to and , and the calculation equation of is:
- (3)
- Calculate the loss value and the loss equation according to and ;
- (4)
- The loss value of the student model is , the update parameter using this gradient descent is , and the teacher network updates the parameter by moving average to , and the update process is expressed by the Equation (2).
3.2. Preprocessing
3.2.1. Noise Reduction Process
3.2.2. Data Augmentation Process
3.3. Detailed Design of Bilateral Networks
- (1)
- The channel capacity of the detail branch used to process the underlying details of the osteosarcoma image is high and shallow, and the receptive field for spatial details is relatively small. The feature representation of this branch has a large spatial size and a wide channel, so it is best not to use residual connections, otherwise, it may increase the cost and reduce the speed.
- (2)
- The semantic branch is used to capture the high-level semantics in osteosarcoma MRI images, and its channel capacity is small, the level is relatively deep, and the receptive field for categorical semantics is relatively large. This branch has a lower channel capacity than the detail branch because spatial details can be provided by the detail branch. Compared to the channel with ω (ω < 1) detail branches in our experiments, such branches are lightweight.
- (3)
- The features of the above two branches are complementary, and one of them does not know the information of the other, so we design an aggregation layer to combine these two types of feature representations. Because of the fast downsampling strategy, the spatial dimension of the output of the semantic branch is smaller than that of the detail branch. Therefore, we need to upsample the output feature map of the semantic branch to match the output of the detail branch.
3.3.1. Detailed Design of Detail Branches
3.3.2. Detailed Design of Semantic Branches
- (1)
- 3 × 3 convolution. Feature responses can be efficiently aggregated and scaled to higher dimensional spaces.
- (2)
- A 3 × 3 depthwise convolution can be performed independently on each output channel of the expansion layer.
- (3)
- The 1 × 1 network can be used as a projection layer to project the output of the depth-wise convolution to the space with low channel capacity. When stride = 2, we will use two 3 × 3 depth convolutions to further expand the receptive field, and a 3 × 3 separable convolution is used as a shortcut. Many people use a 5 × 5 separable convolution to expand the receptive field. Under certain conditions, its FLOPS will be better than two. There are fewer 3 × 3 separable convolutions. But in this layer, we use two 3 × 3 depthwise convolutions instead of 5 × 5 depthwise convolutions. Because this structure has fewer FLOPS and the same receptive field in our model, the model becomes more lightweight.
3.3.3. Bilateral Guided Aggregation
3.3.4. Booster Training Strategy
4. Data Collection, Analysis and Discussion
4.1. Data Collection
4.2. Evaluation Indexes
4.3. Contrast Algorithm
- (1)
- The core module of the pyramid scene parsing network (PSPNet) [38] is the pyramid pooling module, which is used to aggregate the context information of different regions in order to obtain global information. The size of the pooling kernel at the pyramid level can be set.
- (2)
- The multi-scale fully convolutional network (MSFCN) [39] is a fully convolutional network based on multi-supervised output layers which can be used for the automatic segmentation of tumors. It uses multiple feature channels in the up-sampling part.
- (3)
- U-net [40] is a segmentation network that adopts a u-shaped structure. It uses convolution to first encode and then decode. It includes two parts: feature extraction and upper sampling. This model is relatively simple, but the effect is better.
- (4)
- Feature pyramid networks (FPN) [41] utilize high-resolution images of low-level features and semantic information of high-level features to fuse features of different layers to achieve prediction.
- (5)
- The multi-scale residual network (MSRN) [20] is based on residual blocks, and introduces convolution kernels of different sizes, so that the features of images of different scales can be adaptively detected, and the most effective image information can be obtained at the same time. This method is a state-of-the-art osteosarcoma MRI image segmentation model which can effectively utilize the characteristics of low-resolution images.
- (6)
- A fully convolutional network (FCN) [37] is a tumor detection method that uses a skip structure to achieve fine segmentation to perform the pixel-level classification of images. We select FCN-8s and FCN-16s, two networks with 8 and 16 upsampling, respectively, for comparative experiments.
4.4. Training Strategy
4.5. Segmentation Effect Evaluation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, G.; Wu, J. Efficacy prediction based on attribute and multi-source data collaborative for auxiliary medical system in developing countries. Neural Comput. Appl. 2022, 34, 5497–5512. [Google Scholar] [CrossRef]
- Li, X.; Qi, H.; Wu, J. Efficient path-sense transmission based on IoT system in opportunistic social networks. Peer-to-Peer Netw. Appl. 2022, 15, 811–826. [Google Scholar] [CrossRef]
- Wu, J.; Yang, S.; Gou, F.; Zhou, Z.; Xie, P.; Xu, N.; Dai, Z. Intelligent Segmentation Medical Assistance System for MRI Images of Osteosarcoma in Developing Countries. Comput. Math. Methods Med. 2022, 2022, 7703583. [Google Scholar] [CrossRef]
- Wu, J.; Gou, F.; Tian, X. Disease Control and Prevention in Rare Plants Based on the Dominant Population Selection Method in Opportunistic Social Networks. Comput. Intell. Neurosci. 2022, 2022, 1489988. [Google Scholar] [CrossRef] [PubMed]
- Gou, F.; Wu, J. Message Transmission Strategy Based on Recurrent Neural Network and Attention Mechanism in Iot System. J. Circuits, Syst. Comput. 2022, 31, 2250126. [Google Scholar] [CrossRef]
- Wu, J.; Gou, F.; Xiong, W.; Zhou, X. A Reputation Value-Based Task-Sharing Strategy in Opportunistic Complex Social Networks. Complexity 2021, 2021, 8554351. [Google Scholar] [CrossRef]
- Wu, J.; Qu, J.; Yu, G. Behavior prediction based on interest characteristic and user communication in opportunistic social networks. Peer-to-Peer Netw. Appl. 2021, 14, 1006–1018. [Google Scholar] [CrossRef]
- Ouyang, W.; Chen, Z.; Wu, J.; Yu, G.; Zhang, H. Dynamic Task Migration Combining Energy Efficiency and Load Balancing Optimization in Three-Tier UAV-Enabled Mobile Edge Computing System. Electronics 2021, 10, 190. [Google Scholar] [CrossRef]
- Wu, J.; Gou, F.; Tan, Y. A staging auxiliary diagnosis model for non-small cell lung cancer based the on intelligent medical system. Comput. Math. Methods Med. 2021, 2021, 6654946. [Google Scholar] [CrossRef]
- Dong, Y.; Chang, L.; Luo, J.; Wu, J. A Routing Query Algorithm Based on Time-Varying Relationship Group in Opportunistic Social Networks. Electronics 2021, 10, 1595. [Google Scholar] [CrossRef]
- Deng, Y.; Gou, F.; Wu, J. Hybrid data transmission scheme based on source node centrality and community reconstruction in opportunistic social networks. Peer-to-Peer Netw. Appl. 2021, 14, 3460–3472. [Google Scholar] [CrossRef]
- Wu, J.; Zou, W.; Long, H. Effective path prediction and data transmission in opportunistic social networks. IET Commun. 2021, 15, 2202–2211. [Google Scholar] [CrossRef]
- Gou, F.; Wu, J. Triad link prediction method based on the evolutionary analysis with IoT in opportunistic social networks. Comput. Commun. 2021, 181, 143–155. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, Z.G.; Wu, J.; Yu, G. MNSRQ: Mobile node social relationship quantification algorithm for data transmission in Internet of things. IET Commun. 2021, 15, 748–761. [Google Scholar] [CrossRef]
- Lu, Y.; Chang, L.; Luo, J.; Wu, J. Routing Algorithm Based on User Adaptive Data Transmission Scheme in Opportunistic Social Networks. Electronics 2021, 10, 1138. [Google Scholar] [CrossRef]
- Cui, R.; Chen, Z.; Wu, J.; Tan, Y.; Yu, G. A Multiprocessing Scheme for PET Image Pre-Screening, Noise Reduction, Segmentation and Lesion Partitioning. IEEE J. Biomed. Heal. Inform. 2021, 25, 1699–1711. [Google Scholar] [CrossRef]
- Zhang, X.; Chang, L.; Luo, J.; Wu, J. Effective communication data transmission based on community clustering in opportunistic social networks in IoT system. J. Intell. Fuzzy Syst. 2021, 41, 2129–2144. [Google Scholar] [CrossRef]
- Nasor, M.; Obaid, W. Segmentation of osteosarcoma in MRI images by K-means clustering, Chan-Vese segmentation, and iterative Gaussian filtering. IET Image Process. 2020, 15, 1310–1318. [Google Scholar] [CrossRef]
- Shuai, L.; Gao, X.; Wang, J. Wnet ++: A Nested W-shaped Network with Multiscale Input and Adaptive Deep Supervision for Osteosarcoma Segmentation. In Proceedings of the 2021 IEEE 4th International Conference on Electronic Information and Communication Technology (ICEICT), Xi’an, China, 18–20 August 2021. [Google Scholar] [CrossRef]
- Zhang, R.; Huang, L.; Xia, W.; Zhang, B.; Qiu, B.; Gao, X. Multiple supervised residual network for osteosarcoma segmentation in CT images. Comput. Med. Imaging Graph. 2018, 63, 1–8. [Google Scholar] [CrossRef]
- Arunachalam, H.B.; Mishra, R.; Daescu, O.; Cederberg, K.; Rakheja, D.; Sengupta, A.; Leonard, D.; Hallac, R.; Leavey, P. Viable and necrotic tumor assessment from whole slide images of osteosarcoma using ma-chine-learning and deep-learning models. PLoS ONE 2019, 14, e0210706. [Google Scholar] [CrossRef]
- Nabid, R.A.; Rahman, M.L.; Hossain, M.F. Classification of Osteosarcoma Tumor from Histological Image Using Sequential RCNN. In Proceedings of the 2020 11th International Conference on Electrical and Computer Engineering (ICECE), Dhaka, Bangladesh, 17–19 December 2020. [Google Scholar]
- Fu, Y. Deep Model with Siamese Network for Viability and Necrosis Tumor Assessment in Osteosarcoma. arXiv 2019, arXiv:1910.12513. [Google Scholar]
- Qiao, Y.-J.; Gao, B.-L.; Shi, R.-X.; Liu, X.; Wang, Z.-H. Improved FCM Brain MRI Image Segmentation Algorithm Based on Tamura Texture Feature. Ji Suan Ji Ke Xue 2021, 48, 111–117. [Google Scholar]
- Hua, L.; Gu, Y.; Gu, X.; Xue, J.; Ni, T. A Novel Brain MRI Image Segmentation Method Using an Improved Multi-View Fuzzy c-Means Clustering Algorithm. Front. Neurosci. 2021, 15, 662674. [Google Scholar] [CrossRef] [PubMed]
- Tarkhaneh, O.; Shen, H. An adaptive differential evolution algorithm to optimal multi-level thresholding for MRI brain image segmentation. Expert Syst. Appl. 2019, 138, 112820. [Google Scholar] [CrossRef]
- Sasikala, E.; Kanmani, P.; Gopalakrishnan, R.; Radha, R. Identification of lesion using an efficient hybrid algorithm for MRI brain image segmentation. J. Ambient Intell. Humaniz. Comput. 2021. [Google Scholar] [CrossRef]
- Anisuzzaman, D.; Barzekar, H.; Tong, L.; Luo, J.; Yu, Z. A deep learning study on osteosarcoma detection from histological images. Biomed. Signal Process. Control 2021, 69, 102931. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, X.; Wang, X.; Quan, X.; Deng, Y.; Lu, M.; Wei, Q.; Ye, Q.; Zhou, Q.; Xiang, Z.; et al. MRI-based radiomics signature for pretreatment prediction of pathological response to neoadjuvant chemo-therapy in osteosarcoma: A multicenter study. Eur. Radiol. 2021, 31, 7913–7924. [Google Scholar] [CrossRef]
- Dionísio, F.; Oliveira, L.; Hernandes, M.; Engel, E.; Rangayyan, R.; Azevedo-Marques, P.; Nogueira-Barbosa, M. Manual and semiautomatic segmentation of bone sarcomas on MRI have high similarity. Braz. J. Med Biol. Res. 2020, 53, e8962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baidya Kayal, E.; Kandasamy, D.; Sharma, R.; Sharma, M.C.; Bakhshi, S.; Mehndiratta, A. SLIC-supervoxels-based response evaluation of osteosarcoma treated with neoadjuvant chemother-apy using multi-parametric MR imaging. Eur. Radiol. 2020, 30, 3125–3136. [Google Scholar] [CrossRef]
- Dai, Y.; Yin, P.; Mao, N.; Sun, C.; Wu, J.; Cheng, G.; Hong, N. Differentiation of Pelvic Osteosarcoma and Ewing Sarcoma Using Radiomic Analysis Based on T2-Weighted Images and Contrast-Enhanced T1-Weighted Images. BioMed Res. Int. 2020, 2020, 9078603. [Google Scholar] [CrossRef]
- Parlak, Ş.; Ergen, F.B.; Yüksel, G.Y.; Karakaya, J.; Aydın, G.B.; Kösemehmetoğlu, K.; Aydıngöz, Ü. Diffusion-weighted imaging for the differentiation of Ewing sarcoma from osteosarcoma. Skelet. Radiol. 2021, 50, 2023–2030. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.R.; Gikas, P.; Gerrand, C.; Briggs, T.W.; Saifuddin, A. The sensitivity, specificity, and diagnostic accuracy of whole-bone MRI for identifying skip metastases in appendicular osteosarcoma and Ewing sarcoma. Skelet. Radiol. 2020, 49, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Guerra, R.B.; Tostes, M.D.; Miranda LD, C.; Camargo OP, D.; Baptista, A.M.; Caiero, M.T.; dos Santos Machado, T.M.; Abadi, M.D.; de Oliveira, C.R.G.C.M.M.; Filippi, R.Z. Comparative analysis between osteosarcoma and Ewing’s sarcoma: Evaluation of the time from onset of signs and symptoms until diagnosis. Clinics 2006, 61, 99–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhan, X.; Long, H.; Gou, F.; Duan, X.; Kong, G.; Wu, J. A Convolutional Neural Network-Based Intelligent Medical System with Sensors for Assistive Diagnosis and Decision-Making in Non-Small Cell Lung Cancer. Sensors 2021, 21, 7996. [Google Scholar] [CrossRef] [PubMed]
- Long, J.; Shelhamer, E.; Darrell, T. Fully convolutional networks for semantic segmentation. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Boston, MA, USA, 7–12 June 2015; pp. 3431–3440. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Shi, J.; Qi, X.; Wang, X.; Jia, J. Pyramid scene parsing network. In Proceedings of the 2017 IEEE Conference on Computer Vision and Pattern Recognition, Honolulu, HI, USA, 21–26 July 2017; pp. 2881–2890. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Xia, W.; Zhang, B.; Qiu, B.; Gao, X. MSFCN-multiple supervised fully convolutional networks for the osteosarcoma segmentation of CT images. Comput. Methods Programs Biomed. 2017, 143, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional networks for biomedical image segmentation. In Medical Image Computing and Computer-Assisted Intervention 2015; Navab, N., Hornegger, J., Wells, W.M., Frangi, A.F., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 234–241. [Google Scholar] [CrossRef] [Green Version]
- Lin, T.Y.; Dollár, P.; Girshick, R.; He, K.; Hariharan, B.; Belongie, S. Feature pyramid networks for object detection. In Proceedings of the 2017 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Honolulu, HI, USA, 21–26 July 2017; pp. 936–944. [Google Scholar] [CrossRef] [Green Version]













| Symbol | Represented Meaning |
|---|---|
| Parameter group for the student model in formula | |
| Parameter group for the teacher model in the above formula | |
| Randomly assign dataset 1 | |
| Randomly assign dataset 2 | |
| Labels for dataset | |
| Represent student model output probability in formula | |
| Represent teacher model output probability in the above formula | |
| one of the loss values of the student function | |
| one of the loss values of the teacher function | |
| , | Updated student and teacher model parameter Set |
| Loss value of the student model | |
| Represent subimage window in formula | |
| each pixel of the subimage window in the above formula | |
| detected target | |
| Multistage filter | |
| Channel size |
| Characteristics | Total | Training Set | Test Set | |
|---|---|---|---|---|
| Age | <15 | 48 (23.5%) | 38 (23.2%) | 10 (25%) |
| 15~25 | 131 (64.2%) | 107 (65.2%) | 24 (60.0%) | |
| >25 | 25 (12.3%) | 19 (11.6%) | 6 (15.0%) | |
| Sex | Female | 92 (45.1%) | 69 (42.1%) | 23 (57.5%) |
| Male | 112 (54.9%) | 95 (57.9%) | 17 (42.5%) | |
| Marital status | Married | 32 (15.7%) | 19 (11.6%) | 13 (32.5%) |
| Unmarried | 172 (84.3%) | 145 (88.4%) | 27 (67.5%) | |
| Surgery | Yes | 181 (88.8%) | 146 (89.0%) | 35 (87.5%) |
| No | 23 (11.2%) | 18 (11.0%) | 5 (12.5%) | |
| SES | Low SES | 78 (38.2%) | 66 (40.2%) | 12 (30.0%) |
| High SES | 126 (61.8%) | 98 (59.8%) | 28 (70.0%) | |
| Grade | Low grade | 41 (20.1%) | 15 (9.1%) | 26 (65%) |
| High grade | 163 (79.9%) | 149 (90.9%) | 14 (35%) | |
| Location | Axial | 29 (14.2%) | 21 (12.8%) | 8 (20%) |
| Extremity | 138 (67.7%) | 109 (66.5%) | 29 (72.5%) | |
| Other | 37 (18.1%) | 34 (20.7%) | 3 (7.5%) | |
| Model | Pr | Re | F1 | IoU | DSC | Params |
|---|---|---|---|---|---|---|
| FCN-16s | 0.922 | 0.882 | 0.900 | 0.824 | 0.859 | 134.3 M |
| FCN-8s | 0.941 | 0.873 | 0.901 | 0.830 | 0.876 | 134.3 M |
| PSPNet | 0.856 | 0.888 | 0.872 | 0.772 | 0.870 | 46.70 M |
| MSFCN | 0.881 | 0.936 | 0.906 | 0.841 | 0.874 | 23.8 M |
| MSRN | 0.893 | 0.945 | 0.918 | 0.853 | 0.887 | 14.27 M |
| FPN | 0.914 | 0.924 | 0.919 | 0.852 | 0.888 | 48.20 M |
| UNet | 0.922 | 0.924 | 0.923 | 0.867 | 0.892 | 17.26 M |
| Ours | 0.915 | 0.923 | 0.919 | 0.853 | 0.915 | 2.33 M |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.; Gou, F.; Dai, Z. Osteosarcoma MRI Image-Assisted Segmentation System Base on Guided Aggregated Bilateral Network. Mathematics 2022, 10, 1090. https://doi.org/10.3390/math10071090
Shen Y, Gou F, Dai Z. Osteosarcoma MRI Image-Assisted Segmentation System Base on Guided Aggregated Bilateral Network. Mathematics. 2022; 10(7):1090. https://doi.org/10.3390/math10071090
Chicago/Turabian StyleShen, Yedong, Fangfang Gou, and Zhehao Dai. 2022. "Osteosarcoma MRI Image-Assisted Segmentation System Base on Guided Aggregated Bilateral Network" Mathematics 10, no. 7: 1090. https://doi.org/10.3390/math10071090
APA StyleShen, Y., Gou, F., & Dai, Z. (2022). Osteosarcoma MRI Image-Assisted Segmentation System Base on Guided Aggregated Bilateral Network. Mathematics, 10(7), 1090. https://doi.org/10.3390/math10071090






