Gas and Liquid Chromatography Mass Spectrometry as a Tool for Elucidating Volatile Organic Compounds (VOCs) and Metabolites in Maternal Milk: A Perspective on Infants’ Health Risk Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Sample Collection
2.3. Sample Preparation
2.3.1. GC-MS/MS Targeted Analysis for VOCs
2.3.2. LC-ESI-MS/MS Targeted Analysis of Metabolites
2.3.3. GC-MS/MS Untargeted Analysis and SPME Selection
2.4. Instrumental Analysis
2.4.1. GC-MS/MS Targeted Analysis
2.4.2. LC-ESI-MS/MS Targeted Analysis
2.4.3. GC-MS Untargeted Analysis
2.4.4. MS-DIAL Untargeted Workflow
2.5. Validation Study
2.6. Infants’ Health Risk Assessment
3. Results
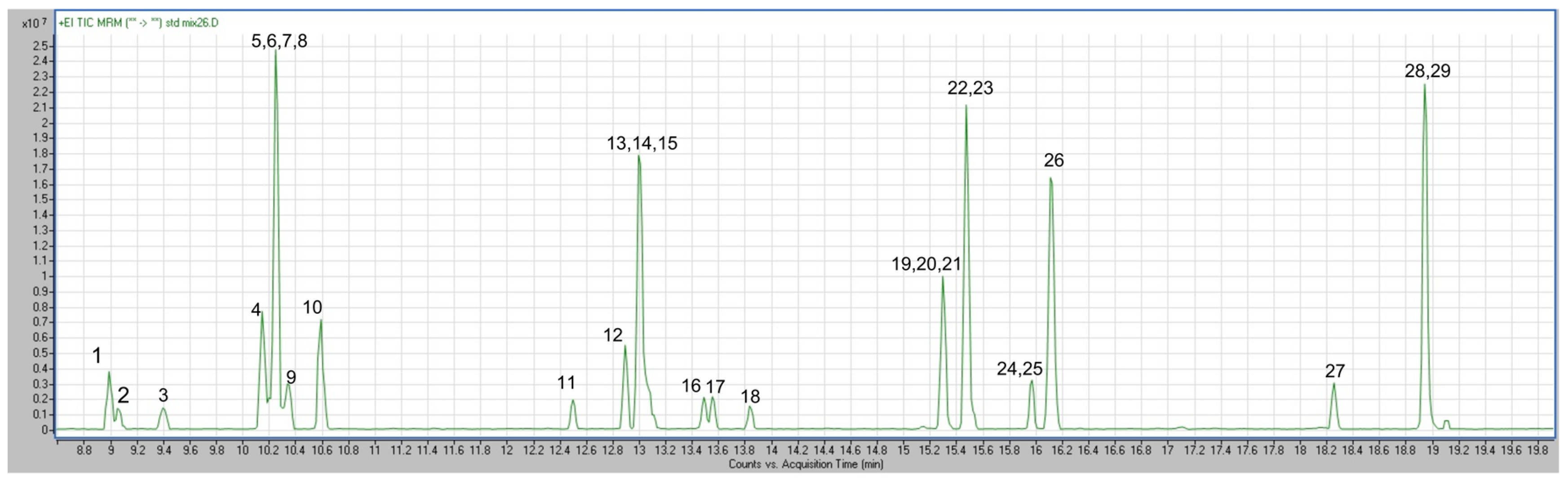
3.1. Optimization of Targeted and Untargeted Analytical Methods
3.2. Compound Identification via Targeted and Untargeted Analysis to Support Characterization of VOCs in Human Milk
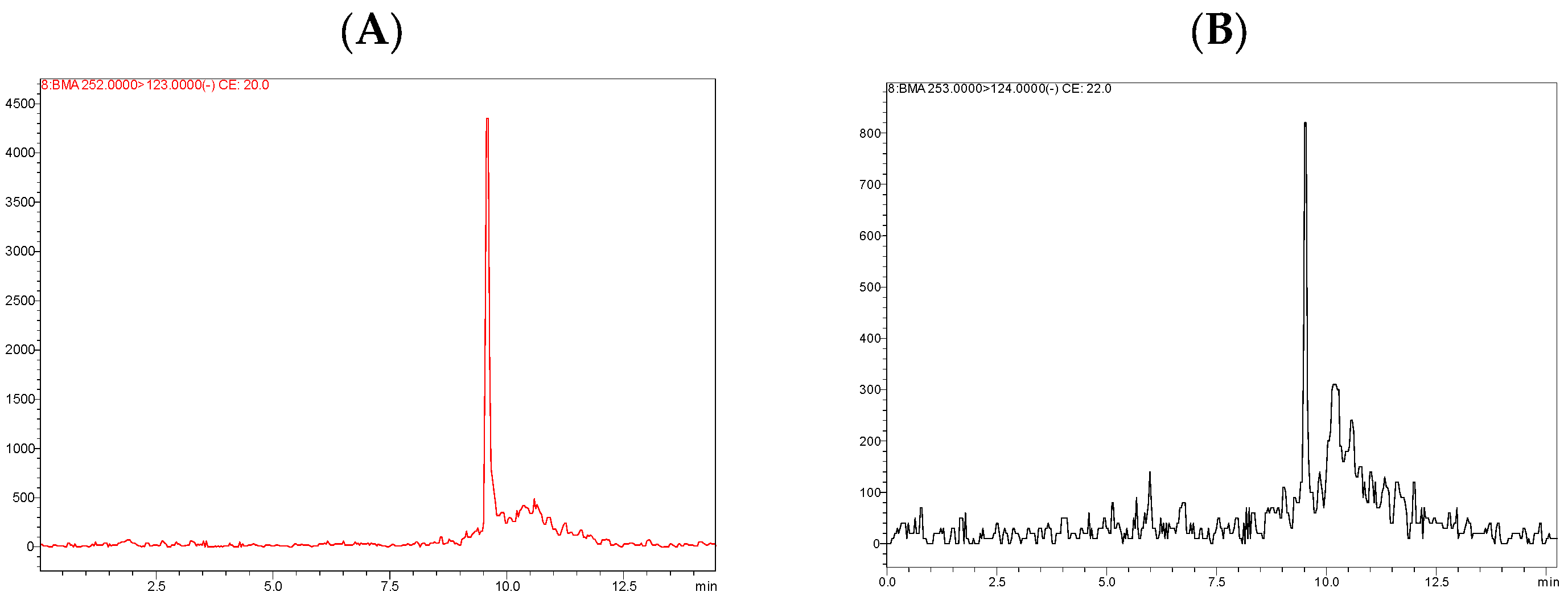
3.2.1. Targeted Chemical Analysis
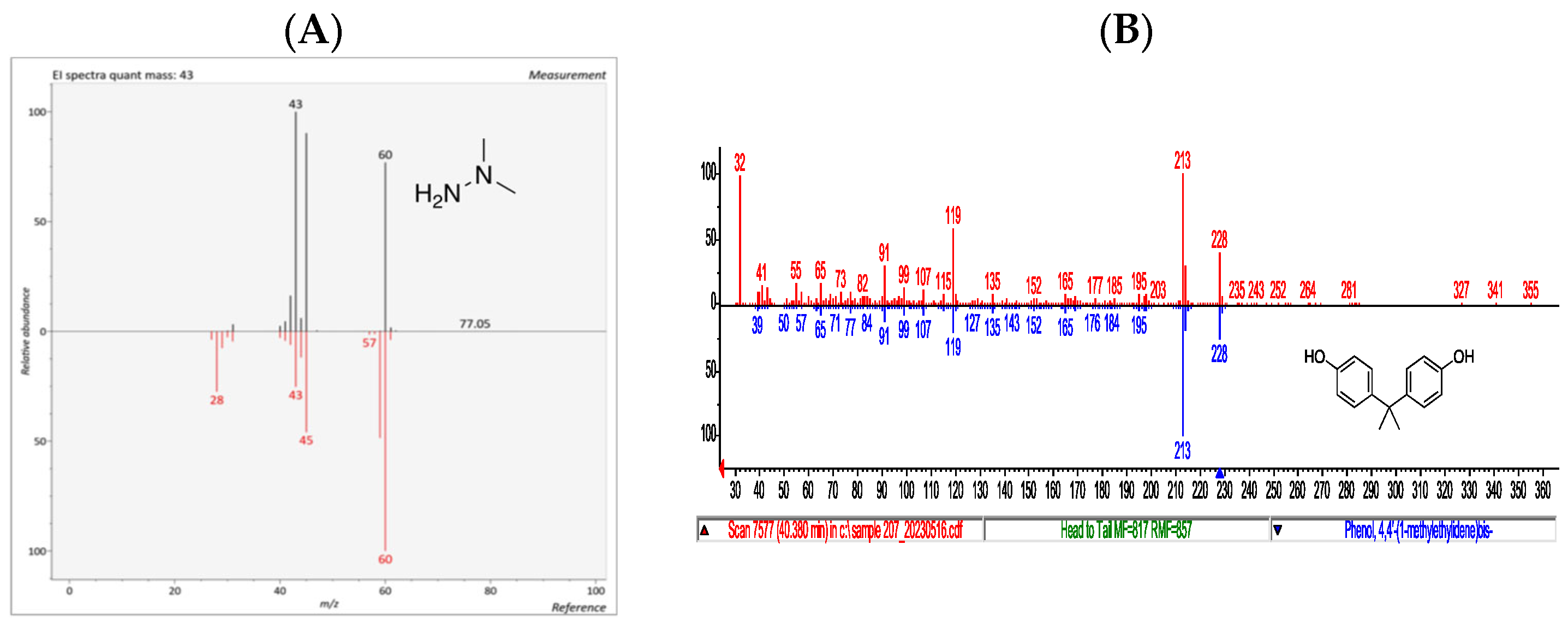
3.2.2. Untargeted Chemical Analysis
3.3. Infants’ Health Risk Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO-Breastfeeding. Available online: https://www.who.int/tools/elena/interventions/exclusive-breastfeeding (accessed on 2 September 2023).
- Theurich, M.A.; Davanzo, R.; Busck-Rasmussen, M.; Diaz-Gomez, N.M.; Brennan, C.; Kylberg, E.; Baerug, A.; McHugh, L.; Weikert, C.; Abraham, K.; et al. Breastfeeding Rates and Programs in Europe: A Survey of 11 National Breastfeeding Committees and Representatives. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Tigka, M.; Metallinou, D.; Nanou, C.; Iliodromiti, Z.; Lykeridou, K. Frequency and Determinants of Breastfeeding in Greece: A Prospective Cohort Study during the COVID-19 Pandemic. Children 2022, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- Li, A.J.; Pal, V.K.; Kannan, K. A review of environmental occurrence, toxicity, biotransformation and biomonitoring of volatile organic compounds. Environ. Chem. Ecotoxicol. 2021, 3, 91–116. [Google Scholar] [CrossRef]
- Ratel, J.; Engel, E. Determination of benzenic and halogenated volatile organic compounds in animal-derived food products by one-dimensional and comprehensive two-dimensional gas chromatography-mass spectrometry. J. Chromatogr. A 2009, 1216, 7889–7898. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhou, X.; Wang, C.; Zhou, H. Environmental and human health impacts of volatile organic compounds: A perspective review. Chemosphere 2023, 313, 137489. [Google Scholar] [CrossRef]
- Soni, V.; Singh, P.; Goel, V. Effects of VOCs on Human Health. In Air Pollution and Control; Energy, Environment, and Sustainability Book Series (ENENSU); Springer: Singapore, 2017; pp. 119–142. [Google Scholar]
- Blount, B.C.; McElprang, D.O.; Chambers, D.M.; Waterhouse, M.G.; Squibb, K.S.; LaKind, J.S. Methodology for collecting, storing, and analyzing human milk for volatile organic compounds. J. Environ. Monit. 2010, 12, 1265–1273. [Google Scholar] [CrossRef]
- DIRECTIVE-2008/50/EC. Directive 2008/50/EC of the European Parliament and of the Council of 21 May 2008 on Ambient Air Quality and Cleaner Air for Europe. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX%3A32008L0050, (accessed on 5 June 2023).
- Limit—Value-Benzene. Available online: https://limitvalue.ifa.dguv.de (accessed on 5 June 2023).
- Chi, Z.H.; Goodyer, C.G.; Hales, B.F.; Bayen, S. Characterization of different contaminants and current knowledge for defining chemical mixtures in human milk: A review. Environ. Int. 2023, 171, 107717. [Google Scholar] [CrossRef]
- Barr, D.B.; Wang, R.Y.; Needham, L.L. Biologic monitoring of exposure to environmental chemicals throughout the life stages: Requirements and issues for consideration for the National Children’s Study. Environ. Health Perspect. 2005, 113, 1083–1091. [Google Scholar] [CrossRef]
- Drabinska, N.; Flynn, C.; Ratcliffe, N.; Belluomo, I.; Myridakis, A.; Gould, O.; Fois, M.; Smart, A.; Devine, T.; Costello, B.L. A literature survey of all volatiles from healthy human breath and bodily fluids: The human volatilome. J. Breath Res. 2021, 15, 034001. [Google Scholar] [CrossRef]
- Lin, Y.J.; Feng, C.; Le, S.Y.; Qiu, X.L.; Xu, Q.; Jin, S.P.; Fang, Y.M.; Jin, Y.E.; Wen, Y.M.; Wang, G.Q.; et al. Infant Exposure to PCBs and PBDEs Revealed by Hair and Human Milk Analysis: Evaluation of Hair as an Alternative Biomatrix. Environ. Sci. Technol. 2022, 56, 15912–15919. [Google Scholar] [CrossRef]
- Kim, S.R.; Halden, R.U.; Buckley, T.J. Volatile organic compounds in human milk: Methods and measurements. Environ. Sci. Technol. 2007, 41, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, M.; Ewers, U.; Wittsiepe, J.; Furst, P.; Holzer, J.; Eberwein, G.; Angerer, J.; Marczynski, B.; Ranft, U. Human biomonitoring studies in North Rhine-Westphalia, Germany. Int. J. Hyg. Environ. Health 2007, 210, 307–318. [Google Scholar] [CrossRef]
- Rondanelli, M.; Perdoni, F.; Infantino, V.; Faliva, M.A.; Peroni, G.; Iannello, G.; Nichetti, M.; Alalwan, T.A.; Perna, S.; Cocuzza, C. Volatile Organic Compounds as Biomarkers of Gastrointestinal Diseases and Nutritional Status. J. Anal. Methods Chem. 2019, 2019, 7247802. [Google Scholar] [CrossRef]
- Costello, B.D.; Amann, A.; Al-Kateb, H.; Flynn, C.; Filipiak, W.; Khalid, T.; Osborne, D.; Ratcliffe, N.M. A review of the volatiles from the healthy human body. J. Breath Res. 2014, 8, 014001. [Google Scholar] [CrossRef]
- Smith, D. Worldwide trends in DDT levels in human breast milk. Int. J. Epidemiol. 1999, 28, 179–188. [Google Scholar] [CrossRef]
- Oliveira, M.; Duarte, S.; Delerue-Matos, C.; Pena, A.; Morais, S. Exposure of nursing mothers to polycyclic aromatic hydrocarbons: Levels of un-metabolized and metabolized compounds in breast milk, major sources of exposure and infants’ health risks. Environ. Pollut. 2020, 266, 115243. [Google Scholar] [CrossRef] [PubMed]
- Cok, I.; Mazmanci, B.; Mazmanci, M.A.; Turgut, C.; Henkelmann, B.; Schramm, K.W. Analysis of human milk to assess exposure to PAHs, PCBs and organochlorine pesticides in the vicinity Mediterranean city Mersin, Turkey. Environ. Int. 2012, 40, 63–69. [Google Scholar] [CrossRef]
- Lehmann, G.M.; LaKind, J.S.; Davis, M.H.; Hines, E.P.; Marchitti, S.A.; Alcala, C.; Lorber, M. Environmental Chemicals in Breast Milk and Formula: Exposure and Risk Assessment Implications. Environ. Health Perspect. 2018, 126, 96001. [Google Scholar] [CrossRef]
- Louis, L.M.; Kavi, L.K.; Boyle, M.; Pool, W.; Bhandari, D.; De Jesús, V.R.; Thomas, S.; Pollack, A.Z.; Sun, A.; McLean, S.; et al. Biomonitoring of volatile organic compounds (VOCs) among hairdressers in salons primarily serving women of color: A pilot study. Environ. Int. 2021, 154, 106655. [Google Scholar] [CrossRef] [PubMed]
- Bolden, A.L.; Kwiatkowski, C.F.; Colborn, T. New Look at BTEX: Are Ambient Levels a Problem? Environ. Sci. Technol. 2015, 49, 5261–5276. [Google Scholar] [CrossRef] [PubMed]
- Malik, P.; Hashim, T.; Varma, S.; Diaz, L.; Chowdhary, A.; Bapat, P.; Alkhatib, L.; Centeno, L.; Poursina, O.; Pan, H.; et al. BTEX (benzene, toluene, ethylbenzene, and xylene) and risk of cancer—A study from Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey. Am. J. Clin. Pathol. 2022, 158, S102–S103. [Google Scholar] [CrossRef]
- Gould, O.; Nguyen, N.; Honeychurch, K.C. New Applications of Gas Chromatography and Gas Chromatography-Mass Spectrometry for Novel Sample Matrices in the Forensic Sciences: A Literature Review. Chemosensors 2023, 11, 527. [Google Scholar] [CrossRef]
- Atsdr-Styrene. In Atsdr Toxicological Profile for Styrene; Agency for Toxic Substances and Disease Registry, U.S. Department of Human and Health Services: Atlanta, GA, USA, 2010. Available online: https://www.atsdr.cdc.gov/toxprofiles/tp53.pdf (accessed on 25 June 2023).
- Branch, V.; Pirkle, J.L. Laboratory Procedure Manual Analyte: Volatile Organic Compounds (VOCs) Metabolites Matrix: Urine Method: Ultra Performance Liquid Chromatography with Electro Spray Tandem Mass Spectrometry [UPLC ESI/MSMS]. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2015-2016/labmethods/UVOC_UVOCS_I_MET.pdf (accessed on 5 June 2023).
- INCHEM. Available online: https://inchem.org (accessed on 5 August 2023).
- Atsdr-Ethylbenzene. In Atsdr Toxicological Profile for Ethylbenzene; Agency for Toxic Substances and Disease Registry, U.S. Department of Health and Human Services: Atlanta, GA, USA, 2010. Available online: https://www.atsdr.cdc.gov/ToxProfiles/tp110.pdf (accessed on 25 June 2023).
- Atsdr-Xylene. In Atsdr Toxicological Profile for Xylene; Agency for Toxic Substances and Disease Registry, U.S. Department of Health and Human Services: Atlanta GA, USA, 2007. Available online: https://www.atsdr.cdc.gov/ToxProfiles/tp71.pdf (accessed on 25 June 2023).
- ICH-M10-Guideline-2022. ICH Harmonised Guideline: Bioanalytical Method Validation and Study Sample Analysis-M10 ICH Consensus Guideline. Available online: https://database.ich.org/sites/default/files/M10_Guideline_Step4_2022_0524.pdf (accessed on 3 April 2023).
- Bioanalytical Method Validation: Guidance for Industry. Available online: https://www.fda.gov/media/70858/download (accessed on 3 April 2023).
- SWGTOX. Scientific Working Group for Forensic Toxicology: Standard Practices for Method Validation in Forensic Toxicology. J. Anal. Toxicol. 2013, 37, 452–474. [Google Scholar] [CrossRef] [PubMed]
- 2002/657/EC. Commission Decision of 12 August 2002 Implementing Council Directive 96/23/EC Concerning the Performance of Analytical Methods and the Interpretation of Results. Available online: https://eur-lex.europa.eu/eli/dec/2002/657/oj, (accessed on 3 April 2023).
- US-EPA. Risk Based Concentration Table; United States Environmental Protection Agency: Philadelphia, PA, USA; Washington, DC, USA, 2000.
- Milk-Consumption. Available online: https://www2.hse.ie/babies-children/breastfeeding/expressing-pumping/how-much-breast-milk-express/ (accessed on 3 March 2023).
- Weight-WHO. Available online: https://www.who.int/tools/child-growth-standards/standards/weight-velocity (accessed on 6 April 2023).
- Rogan, W.J.; Ragan, N.B. Chemical contaminants, pharmacokinetics, and the lactating mother. Environ. Health Perspect. 1994, 102 (Suppl. S11), 89–95. [Google Scholar] [CrossRef]
- US-EPA. 2020. Available online: https://iris.epa.gov/AtoZ/?list_type=alpha (accessed on 21 May 2021).
- OEHHA. Office of Environmental Health Hazards Assessment. 2020. Available online: https://oehha.ca.gov (accessed on 21 May 2021).
- RAIS. The Risk Assessment Information System. 2020. Available online: https://rais.ornl.gov (accessed on 2 June 2021).
- Cailleux, A.; Cogny, M.; Allain, P. Blood isoprene concentrations in humans and in some animal species. Biochem. Med. Metab. Biol. 1992, 47, 157–160. [Google Scholar] [CrossRef]
- Biren, C.; Zhang, L.Y.; Bhandari, D.; Blount, B.C.; De Jesús, V.R. Isoprene Exposure in the United States Based on Urinary IPM3: NHANES 2015–2016. Environ. Sci. Technol. 2020, 54, 2370–2378. [Google Scholar] [CrossRef]
- Sukul, P.; Richter, A.; Junghanss, C.; Schubert, J.K.; Miekisch, W. Origin of breath isoprene in humans is revealed via multi-omic investigations. Commun. Biol. 2023, 6, 999. [Google Scholar] [CrossRef]
- Nolan, L.S.; Lewis, A.N.; Gong, Q.; Sollome, J.J.; DeWitt, O.N.; Williams, R.D.; Good, M. Untargeted Metabolomic Analysis of Human Milk from Mothers of Preterm Infants. Nutrients 2021, 13, 3604. [Google Scholar] [CrossRef]
- Marincola, F.C.; Noto, A.; Caboni, P.; Reali, A.; Barberini, L.; Lussu, M.; Murgia, F.; Santoru, M.L.; Atzori, L.; Fanos, V. A metabolomic study of preterm human and formula milk by high resolution NMR and GC/MS analysis: Preliminary results. J. Matern. Fetal Neonatal Med. 2012, 25, 62–67. [Google Scholar] [CrossRef]
- Kus-Yamashita, M.M.M.; Cano, C.B.; Monteiro, V.C.B.; Catarino, R.M. Human Milk: Fast Determination of Docosahexaenoic Acid (DHA). Analytica 2023, 4, 54–65. [Google Scholar] [CrossRef]
- EFSA-Citronellol. Safety and efficacy of non-conjugated and accumulated unsaturated straight-chain and branched-chain, aliphatic primary alcohols, aldehydes, acids, acetals and esters belonging to chemical group 4 when used as flavourings for all animal species EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). EFSA J. 2016, 14, 4559. [Google Scholar]
- Kumar, A.; Mishra, A.K. Biological Importance of Phenol Derivatives as Potent Bioactive Compound: A Review. Lett. Org. Chem. 2018, 15, 251–264. [Google Scholar] [CrossRef]
- Maela, M.P.; van der Walt, H.; Serepa-Dlamini, M.H. The Antibacterial, Antitumor Activities, and Bioactive Constituents’ Identification of Alectra sessiliflora Bacterial Endophytes. Front. Microbiol. 2022, 13, 870821. [Google Scholar] [CrossRef] [PubMed]
- EC-SCCS-Opinion-1, 4-THB. European Commission: Scientific Committee on Consumer Safety (SCCS), Opinion on Hair Dye 1,2,4-trihydroxybenzene (1,2,4-THB): COLIPA n° A33. Available online: https://health.ec.europa.eu/system/files/2021-08/sccs_o_222_0.pdf (accessed on 25 January 2024).
- Niu, Y.M.; Wang, B.; Zhao, Y.F.; Zhang, J.; Shao, B. Highly Sensitive and High-Throughput Method for the Analysis of Bisphenol Analogues and Their Halogenated Derivatives in Breast Milk. J. Agric. Food Chem. 2017, 65, 10452–10463. [Google Scholar] [CrossRef] [PubMed]
- Rovira, J.; Martinez, M.A.; Mari, M.; Cunha, S.C.; Fernandes, J.O.; Marmelo, I.; Marques, A.; Haug, L.S.; Thomsen, C.; Nadal, M.; et al. Mixture of environmental pollutants in breast milk from a Spanish cohort of nursing mothers. Environ. Int. 2022, 166, 107375. [Google Scholar] [CrossRef] [PubMed]
- TDI-Toluene-Xylenes. 2023. Available online: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-toluene-ethylbenzene-xylenes/guidelines-canadian-drinking-water-quality-guideline-technical-document-toluene-ethylbenzene-xylenes-page-11.html (accessed on 22 August 2023).
- TDI-Styrene. Available online: https://inchem.org/documents/jecfa/jecmono/v19je15.htm (accessed on 25 June 2023).
- MADL-Benzene. Available online: https://oehha.ca.gov/media/downloads/crnr/benzene20madlfinal.pdf (accessed on 22 August 2023).
- LOAEL-Isoprene. Summary of Initial Risk Assessment Report: Isoprene-PRTR No of Japan: 28. Available online: https://www.nite.go.jp/en/chem/chrip/chrip_search/dt/pdf/CI_02_001/risk/pdf_gaiyou_en/028sum.pdf (accessed on 5 September 2023).
- ECHA-LOEC/2. Available online: https://echa.europa.eu/documents/10162/13632/information_requirements_r10_en.pdf/bb902be7-a503-4ab7-9036-d866b8ddce69 (accessed on 5 August 2023).
- ECHA-Isoprene. Available online: https://echa.europa.eu/documents/10162/431f6bc0-a4b3-a1a1-4e8f-6c8eb48ab64a (accessed on 5 September 2023).
- EFSA-LOQ/2. Use of cut-off values on the limits of quantification reported in datasets used to estimate dietary exposure to chemical contaminants. EFSA J. 2018, 15, 1452E. [Google Scholar]
- Seeber, A.; Demes, P.; Kiesswetter, E.; Schäper, M.; van Thriel, C.; Zupanic, M. Changes of neurobehavioral and sensory functions due to toluene exposure below 50 ppm? Environ. Toxicol. Pharmacol. 2005, 19, 635–643. [Google Scholar] [CrossRef]
- Seeber, A.; Schäper, M.; Zupanic, M.; Blaszkewicz, M.; Demes, P.; Kiesswetter, E.; van Thriel, C. Toluene exposure below 50 ppm and cognitive function: A follow-up study with four repeated measurements in rotogravure printing plants. Int. Arch. Occup. Environ. Health 2004, 77, 1–9. [Google Scholar] [CrossRef]
- Korsak, Z.; Wisniewska-Knypl, J.; Swiercz, R. Toxic effects of subchronic combined exposure to n-butyl alcohol and m-xylene in rats. Int. J. Occup. Med. Environ. Health 1994, 7, 155–166. [Google Scholar]
- EFSA-BMD-EFSA-Scientific-Committee. Guidance on the use of the benchmark dose approach in risk assessment. EFSA J. 2022, 20, e07584. [Google Scholar]
- US-EPA-Benzene. Available online: https://iris.epa.gov/ChemicalLanding/&substance_nmbr=276 (accessed on 25 January 2024).
- OEHHA-Ethylbenzene. Available online: https://oehha.ca.gov/chemicals/ethylbenzene (accessed on 26 January 2024).
- CDC-Environmental-Health. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2013-2014/labmethods/UVOC_H_MET.pdf (accessed on 25 January 2024).
- Thomas, S.N.; French, D.; Jannetto, P.J.; Rappold, B.A.; Clarke, W.A. Liquid chromatography-tandem mass spectrometry for clinical diagnostics. Nat. Rev. Methods Primers 2022, 2, 96. [Google Scholar] [CrossRef]
- Fabietti, F.; Ambruzzi, A.; Delise, M.; Sprechini, M.R. Monitoring of the benzene and toluene contents in human milk. Environ. Int. 2004, 30, 397–401. [Google Scholar] [CrossRef]
- Muelbert, M.; Galante, L.; Alexander, T.; Harding, J.E.; Pook, C.; Bloomfield, F.H. Odor-active volatile compounds in preterm breastmilk. Pediatr. Res. 2022, 91, 1493–1504. [Google Scholar] [CrossRef]
- Contador, R.; Delgado, F.J.; García-Parra, J.; Garrido, M.; Ramírez, R. Volatile profile of breast milk subjected to high-pressure processing or thermal treatment. Food Chem. 2015, 180, 17–24. [Google Scholar] [CrossRef]
- Pazo, D.Y.; Moliere, F.; Sampson, M.M.; Reese, C.M.; Agnew-Heard, K.A.; Walters, M.J.; Holman, M.R.; Blount, B.C.; Watson, C.H.; Chambers, D.M. Mainstream Smoke Levels of Volatile Organic Compounds in 50 US Domestic Cigarette Brands Smoked with the ISO and Canadian Intense Protocols. Nicotine Tob. Res. 2016, 18, 1886–1894. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.J.; Gong, S.W.; He, Y.; Liu, D. Smoking is associated with elevated blood level of volatile organic compounds: A population-based analysis of NHANES 2017-2018. Arch. Public Health 2023, 81, 55. [Google Scholar] [CrossRef]
- Yaqub, G.; Hamid, A.; Khan, N.; Ishfaq, S.; Banzir, A.; Javed, T. Biomonitoring of Workers Exposed to Volatile Organic Compounds Associated with Different Occupations by Headspace GC-FID. J. Chem. 2020, 2020, 6956402. [Google Scholar] [CrossRef]
- Longo, V.; Forleo, A.; Giampetruzzi, L.; Siciliano, P.; Capone, S. Human Biomonitoring of Environmental and Occupational Exposures by GC-MS and Gas Sensor Systems: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 10236. [Google Scholar] [CrossRef]
- Wolff, M.S. Occupationally Derived Chemicals in Breast-Milk. Am. J. Ind. Med. 1983, 4, 259–281. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.; Mahle, D.; Bankston, L.; Greene, R.; Gearhart, J. Lactational transfer of volatile chemicals in breast milk. Am. Ind. Hyg. Assoc. J. 1997, 58, 425–431. [Google Scholar] [CrossRef]
- US-EPA-Water-HA. Available online: https://www.epa.gov/system/files/documents/2022-01/dwtable2018.pdf (accessed on 25 January 2024).
- Fleming-Jones, M.E.; Smith, R.E. Volatile organic compounds in foods: A five year study. J. Agric. Food Chem. 2003, 51, 8120–8127. [Google Scholar] [CrossRef] [PubMed]

| Number | RT (min) | Compound | Molecular Formula | CAS No. | Octanol/Water Partition Coefficient (Log Pow)/Solubility in Water [29] | Molecular Mass (Da) | Boiling Point (°C) | Quantifier | Collision Energy CE (V) | Qualifier 1 Qualifier 2 | CE, V | SRM Ratio % |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 9.03 | 1,2-Dichloroethene | C2H2Cl2 | 540-59-0 | 2.0/poor solubility in water | 96.95 | 55 | 96 > 96 | 1 | 61, 98 * | na ** | na ** |
| 2 | 9.12 | Methyl tert-butyl-ether | C5H12O | 1634-04-4 | 1.06/4.24 g/100 mL at 20 °C | 88.2 | 55 | 73 > 73 | 0 | 73 > 43 | 5 | 16 |
| 3 | 9.51 | Tetrahydrofuran | C₄H₈O | 109-99-9 | 0.46 (estimated)/freely soluble | 72.1 | 66 | 71 > 71 | 1 | 72 > 42 72 > 72 | 10 1 | 8 |
| 4 | 10.11 | Dichloromethane | CH2Cl2 | 75-09-2 | 1.25/1.3 g/100 mL at 20 °C: (moderate) | 84.9 | 40 | 75 > 75 | 1 | 86, 84.9, 84, 75 * | na ** | na ** |
| 5 | 10.21 | Isoprene | C5H8 | 78-79-5 | 2.3/642 mg/L at 25 °C: (very poor), insoluble in water | 68.1 | 34 | 67 > 67 | 1 | 67 > 41 | 17 | 20 |
| 6 | 10.30 | 1,2-Dichloroethane | C2H4Cl2 | 203-458-1 | 1.48/0.87 g/100 mL | 98.96 | 83.5 | 98 > 62 | 1 | 97 > 92 | 10 | 12 |
| 7 | 10.30 | 1,2-Dichloroethane-D4 | C2H3Cl2 | 107-06-2 | 102.98 | 83 | 66 > 65 | 1 | 102 > 65 | 17 | 40 | |
| 8 | 10.31 | Benzene | C6H6 | 71-43-2 | 2.13/0.18 g/100 mL at 25 °C | 78.1 | 80 | 78 > 78 | 1 | 52 > 52 78 > 52 | 18 1 | 20 |
| 9 | 10.33 | Hexane | C6H14 | 110-54-3 | 3.9/0.0013 g/100 mL at 20 °C: | 86.2 | 68 | 57 > 57 | 0 | 57 > 41 | 5 | 45 |
| 10 | 10.61 | 2-Methyl-pentane | C6H14 | 107-83-5 | 3.2 (estimated)/no solubility in water | 86.2 | 60 | 71 > 71 | 0 | 71 > 43 | 12 | 70 |
| 11 | 12.50 | Heptane | C7H16 | 142-82-5 | 4.66/2.2 mg/L at 25 °C: (very poor) | 100.2 | 98.4 | 43 > 43 | 1 | 71 > 43 71 > 71 | 4 1 | 42 |
| 12 | 12.91 | 2,2,4-Trimethyl-pentane | C8H18 | 540-84-1 | No water solubility | 114.3 | 99 | 57 > 57 | 1 | 85 > 43 43 > 43 | 24 1 | 15 |
| 13 | 12.92 | Toluene-D8 | C7D8 | 2037-26-5 | 0.5 g/L in water at 20 °C | 100.19 | 111 | 100 > 100 | 1 | 100 > 98 98 > 98 | 5 1 | 35 |
| 14 | 13.01 | Toluene | C7H8 | 108-88-3 | 2.69/no water solubility | 92.1 | 111 | 91 > 91 | 1 | 92 > 65 91 > 65 91 > 39 | 5 15 24 | 45 |
| 15 | 13.10 | Octane | C8H18 | 111-65-9 | 4.00/5.18/no water solubility | 114.22 | 126 | 43 > 43 | 1 | 85 > 43 | 11 | 15 |
| 16 | 13.41 | 1,3-Dichloropropene | C3H4Cl2 | 542-75-6 | 1.82/0.2 g/100 mL at 20 °C | 111 | 108 | 75 > 75 | 1 | 75 > 49 110 > 75 | 10 13 | 5 |
| 17 | 13.50 | 1,1,2-Trichloroethane | C2H3Cl3 | 79-00-5 | 2.35/0.45 g/100 mL at 20 °C: (very poor) | 133.4 | 114 | 96 > 96 | 1 | 132 > 131 133 > 83 | 5 17 | 10 |
| 18 | 13.81 | Tetrachloroethylene | C2Cl4 | 127-18-4 | 3.4/0.015 g/100 mL at 20 °C | 165.8 | 121 | 96 > 96 | 1 | 165 > 94 165 > 131 129 > 94 | 27 18 15 | 35 |
| 19 | 15.22 | Chlorobenzene-D5 | C6D5Cl | 3114-55-4 | 0.49 g/L in water at 20 °C | 117.59 | 132 | 117 > 117 | 1 | 117 > 85 | 15 | 45 |
| 20 | 15.23 | Chlorobenzene | C6H5Cl | 108-90-7 | 2.18/2.84/g/100 mL at 20 °C: 0.05 | 112.6 | 132 | 112 > 77 | 13 | 112 > 112 | 1 | 35 |
| 21 | 15.25 | Ethyl benzene | C8H10 | 100-41-4 | 3.1/g/100 mL at 20 °C: 0.015 | 106.2 | 136 | 106 > 91 | 14 | 106 > 106 91 > 65 91 > 91 | 1 20 1 | 80 |
| 22 | 15.48 | p-Xylene | C6H4(CH3)2 | 106-42-3 | 3.15/no solubility in water | 106.2 | 138 | 106 > 91 | 14 | 106 > 106 91 > 65 91 > 91 | 1 20 1 | 70 |
| 23 | 15.48 | m-Xylene | C6H4(CH3)2 | 108-38-3 | 3.20/no solubility in water | 106.17 | 138 | 106 > 91 | 14 | 106 > 106 91 > 65 91 > 91 | 1 20 1 | 70 |
| 25 | 16.01 | o-Xylene | C6H4(CH3)2 | 95-47-6 | 3.12/no solubility in water | 106.2 | 144 | 106 > 91 | 14 | 106 > 106 91 > 65 91 > 91 | 1 20 1 | 100 |
| 24 | 16.11 | o-Xylene-D10 | C6D4(CD3)2 | 56004-61-6 | 146 mg/L in water at 25 °C | 116.23 | 142 | 116 > 116 | 1 | 116 > 98 | 17 | 16 |
| 26 | 16.14 | Styrene | C8H8 | 100-42-5 | 3.0/g/100 mL at 20 °C: 0.03 | 104.2 | 145 | 104 > 78 | 20 | 103 > 77 104 > 104 | 18 1 | 80 |
| 27 | 18.27 | 1,2,4 Trimethyl benzene | C9H12 | 95-63-6 | 3.8/very poor solubility in water | 120.2 | 169 | 105 > 105 | 1 | 120 > 120 120 > 105 | 1 10 | 32 |
| 28 | 18.97 | 1,2,3 Trimethyl benzene | C9H12 | 526-73-8 | 3.7/g/100 mL: 0.005 | 120.2 | 176 | 105 > 105 | 1 | 120 > 120 120 > 105 | 1 10 | 70 |
| 29 | 19.01 | p-Dichlorobenzene | C6H4Cl2 | 106-46-7 | 3.37/mg/L at 20 °C: 49 (practically insoluble) | 147.0 | 174 | 146 > 75 | 27 | 146 > 111 111 > 111 111 > 75 | 10 1 7 | 65 |
| tR (min) | Compound | Parent Compound | Biological Half-Life | Molecular Formula | CAS | Molecular Mass (Da) | Quantifier/ Qualifier | CE, V | Dwell Time | Q1 Pre Bias | Q3 Pre Bias | SRM Ratio % |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2.3 | DL-MA | Styrene | several hours in the blood and about 2–4 days in subcutaneous adipose tissue [27] | C₈H₈O₃ | 90-64-2 | 152.15 | 151 > 107 | 11 | 100 | 19 | 16 | 10 |
| 152 > 108 | 18 | 100 | 25 | 21 | ||||||||
| 2.3 | R-MA | Ethyl Benzene | 27.5 h [30] | C₈H₈O₃ | 611-71-2 | 152.15 | 151 > 107 | 13 | 100 | 19 | 26 | 10 |
| 152 > 108 | 13 | 100 | 26 | 10 | ||||||||
| 3.3 | PGA | Ethyl Benzene Styrene | 27.5 h [30] | C₈H6O₃ | 611-73-4 | 150.13 | 149 > 77 | 11 | 100 | 11 | 18 | 15 |
| 148.6 > 105.1 | 18 | 100 | 11 | 21 | ||||||||
| 3.7 | HA | Toluene | No ref | C9H9NO3 | 495-69-2 | 179.17 | 177.9 > 133.8 | 14 | 100 | 13 | 23 | 90 |
| 178 > 76.9 | 22 | 100 | 15 | 15 | ||||||||
| 4.6 | 2-MHA | Xylene | 1 h [31] | C10H11NO3 | 42013-20-7 | 193.20 | 192.2 > 148.1 | 11 | 100 | 14 | 17 | 25 |
| 191.9 > 91.2 | 18 | 100 | 16 | 18 | ||||||||
| 8.2 | 3-MHA | Xylene | 1 h [31] | C10H11NO3 | 27115-49-7 | 193.20 | 192.2 > 148.2 | 16 | 100 | 15 | 17 | 20 |
| 192.2 > 91.2 | 10 | 100 | 20 | 21 | ||||||||
| 8.2 | 4-MHA | Xylene | 1 h | C10H11NO3 | 27115-50-0 | 193.20 | 192.2 > 148.3 | 10 | 100 | 20 | 21 | 20 |
| 192.2 > 91 | 16 | 100 | 23 | 19 | ||||||||
| 9.2 | PMA | Benzene | No ref | C11H13NO3S | 4775-80-8 | 239.30 | 237.8 > 109 | 100 | 12 | 21 | 22 | 32 |
| 239 > 110 | 100 | 12 | 19 | 24 | ||||||||
| 9.4 | BMA | Toluene | 3–738 min (depending on the tissue) | C12H15NO3S | 19542-77-9 | 253.32 | 252 > 123 | 20 | 100 | 17 | 13 | 25 |
| 253 > 124 | 22 | 100 | 17 | 15 |
| Compound | ILIS | Calibration Range (ng/mL) | Regression Equation | R2 | LOD (ng/mL) | LLOQ (ng/mL) | Mean Recovery (LLOQ) ± RSD % | Precision, %CV |
|---|---|---|---|---|---|---|---|---|
| Isoprene | 1,2-Dichloroethane-D4 | 0.1–100 | y = 7.2441x − 8.0326 | 0.9975 | 0.04 | 0.1 | 81 ± 12 | 8 |
| 2-Methyl pentane | Toluene-D8 | 1–100 | y = 0.017x − 0.0327 | 0.9962 | 0.40 | 1 | 85 ± 9 | 10 |
| Methyl tert-butyl-ether | 1,2-Dichloroethane-D4 | 1–100 | y = 0.0134x − 0.0193 | 0.9948 | 0.40 | 1 | 80 ± 10 | 13 |
| Hexane | Toluene-D8 | 0.5–100 | y = 0.1083x − 0.2439 | 0.9950 | 0.15 | 0.5 | 90 ± 13 | 18 |
| Dichloromethane | 1,2-Dichloroethane-D4 | 0.2–100 | y = 0.0894x − 0.2337 | 0.9961 | 0.06 | 0.2 | 92 ± 7 | 15 |
| Tetrahydrofuran | 1,2-Dichloroethane-D4 | 1–100 | y = 0.0042x − 0.0014 | 0.9985 | 0.40 | 1 | 100 ± 13 | 8 |
| Benzene | Toluene-D8 | 0.5–100 | y = 0.1113x − 0.2993 | 0.9981 | 0.06 | 0.2 | 83 ± 6 | 7 |
| Heptane | Toluene-D8 | 0.5–100 | y = 0.7784x − 2.2274 | 0.9982 | 0.20 | 0.5 | 80 ± 9 | 14 |
| Octane | Toluene-D8 | 0.2–100 | y = 0.8472x − 1.3177 | 0.9987 | 0.08 | 0.2 | 87 ± 11 | 15 |
| 2,2,4-Trimethylpentane | Toluene-D8 | 1–100 | y = 0.0095x − 0.024 | 0.9963 | 0.40 | 1 | 86 ± 9 | 11 |
| Toluene | Toluene-D8 | 0.2–100 | y = 0.8548x − 1.8007 | 0.9982 | 0.08 | 0.2 | 88 ± 14 | 12 |
| 1,3-Dichloropropene | Chlorobenzene-D5 | 1–100 | y = 0.0014x − 0.0031 | 0.9973 | 0.40 | 1 | 82 ± 10 | 18 |
| 1,1,2-Trichloroethane | Chlorobenzene-D5 | 1–100 | y = 0.0019x − 0.0017 | 0.9988 | 0.40 | 1 | 80 ± 9 | 17 |
| Tetrachloroethylene | Chlorobenzene-D5 | 0.5–100 | y = 0.003x − 0.0042 | 0.9955 | 0.20 | 0.5 | 84 ± 6 | 20 |
| Chlorobenzene | Chlorobenzene-D5 | 0.5–100 | y = 0.003x − 0.0046 | 0.9958 | 0.20 | 0.5 | 91 ± 7 | 7 |
| Ethyl benzene | Chlorobenzene-D5 | 0.5–100 | y = 0.0052x + 0.0003 | 0.9984 | 0.20 | 0.5 | 92 ± 15 | 6 |
| p-Xylene | o-Xylene-D10 | 0.2–100 | y = 0.0031x + 0.0021 | 0.9984 | 0.08 | 0.2 | 87 ± 12 | 5 |
| m-Xylene | o-Xylene-D10 | 0.2–100 | y = 0.0308x + 0.0194 | 0.998 | 0.08 | 0.2 | 89 ± 14 | 8 |
| o-Xylene | o-Xylene-D10 | 0.5–100 | y = 0.0307x + 0.0243 | 0.9988 | 0.20 | 0.5 | 89 ± 8 | 9 |
| Styrene | o-Xylene-D10 | 0.2–100 | y = 1.0526x + 2.0068 | 0.9983 | 0.08 | 0.2 | 83 ± 5 | 13 |
| 1,2,4-Trimethylbenzene | Chlorobenzene-D5 | 0.5–100 | y = 0.0094x − 0.0063 | 0.996 | 0.20 | 0.5 | 90 ± 10 | 6 |
| 1,2,3-Trimethylbenzene | Chlorobenzene-D5 | 0.5–100 | y = 0.0017x + 0.0026 | 0.9972 | 0.20 | 0.5 | 89 ± 14 | 7 |
| p-Dichlorobenzene | Chlorobenzene-D5 | 0.2–100 | y = 0.0024x + 0.0022 | 0.9954 | 0.08 | 0.2 | 83 ± 8 | 16 |
| 1,2-Dichloroethene | 1,2-Dichloroethane-D4 | 1–100 | y = 0.0013x − 0.0022 | 0.9908 | 0.40 | 1 | 78 ± 6 | 15 |
| 1,2-Dichloroethane | 1,2-Dichloroethane-D4 | 1–100 | y = 0.0021x + 0.9932 | 0.9932 | 0.40 | 1 | 81 ± 8 | 12 |
| Metabolite | Calibration Range (ng mL−1) | LOD (ng/mL) | LLOQ (ng/mL) | Mean Recovery (LLOQ) ± RSD % | Precision, %CV | |||
| DL-MA | 0.5–100 | y = 110,009x − 8866 | 0.20 | 0.5 | 91 ± 15 | 15 | ||
| R-MA | 0.5–100 | y = 811,288x – 13,330 | 0.20 | 0.5 | 89 ± 12 | 11 | ||
| PGA | 0.4–100 | y = 9,803,147x + 208,237 | 0.15 | 0.4 | 81 ± 11 | 9 | ||
| 2-MHA | 0.4–100 | y = 2,553,189x + 90,707 | 0.15 | 0.4 | 84 ± 8 | 18 | ||
| 3-MHA | 0.4–100 | y = 3,723,117x + 58,001 | 0.15 | 0.4 | 86 ± 9 | 15 | ||
| 4-MHA | 0.4–100 | y = 2,540,019x + 70,005 | 0.15 | 0.4 | 81 ± 12 | 14 | ||
| BMA | 0.2–100 | y = 12,223,177x + 855,458 | 0.08 | 0.2 | 89 ± 7 | 9 | ||
| PMA | 0.2–100 | y = 6,690,883x – 411,307 | 0.08 | 0.2 | 92 ± 15 | 10 | ||
| HA | 0.4–100 | y = 4,412,407x + 1,077,341 | 0.15 | 0.4 | 104 ± 16 | 12 | ||
| Substance Concentration (ng mL−1) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample Code | Isoprene | Benzene | Toluene | p,m-Xylene | Styrene | BMA | MA (DL and R) | 3-MHA |
| Sample 2 | n.d. a | n.d. | n.d. | 0.39 ± 0.14 | n.d. | n.d. | 0.79 ± 0.21 | <LLOQ |
| Sample 3 | n.d. | n.d. | n.d. | n.d. | n.d. | <LLOQ | n.d. | n.d. |
| Sample 4 | n.d. | 0.58 ± 0.15 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Sample 5 | <LLOQ | n.d. | n.d. | n.d. | n.d. | n.d. | <LLOQ | n.d. |
| Sample 12 | 0.11 ± 0.06 | n.d. | 0.63 ± 0.18 | n.d. | 0.40 ± 0.12 | 0.31 ± 0.11 | n.d. | n.d. |
| Sample 18 | n.d. | 0.34 ± 0.14 | n.d. | n.d. | n.d | n.d. | n.d | n.d. |
| Substance | Concentration (ng mL−1) | EDI (mg/kg bw per day) | NOAEL (mg/kg bw per day) b | Toxicological Endpoint | TDI (mg/kg bw per day) | TDI/10 (mg/kg bw per day) *** | HQ |
|---|---|---|---|---|---|---|---|
| Toluene | 0.94 a | 9.40 × 10−5 | 9.7 × 10−2 [55] | Neurological (cognitive function) [62,63] | 9.7 × 10−3 | 9.7 × 10−4 | 9.69 × 10−2 |
| Benzene | 0.58 | 5.80 × 10−5 | 8.4 × 10−1 [57] | Reproductive toxicity [57] | 2.4 × 10−2 * | 2.4 × 10−3 | 2.42 × 10−2 |
| Styrene | 0.79 c | 7.90 × 10−5 | 20 ppm [27] | Depression of the central nervous system (CNS) [56] | 4.0 × 10−2 | 4.0 × 10−3 | 1.97 × 10−2 |
| Xylene | 0.59 d | 5.90 × 10−5 | 1.00 [55] | Neurobehavioral alterations (motor coordination disturbances) [64] | 1.3 × 10−2 | 1.3 × 10−3 | 4.54 × 10−2 |
| Ethyl benzene | 0.79 e | 7.90 × 10−5 | 10.17 [55,65] b | Carcinogenicity (oxidative stress, lung tumors) [55] | 4.1 × 10−1 | 4.1 × 10−2 | 1.93 × 10−3 |
| Isoprene | 0.11 | 1.10 × 10−5 | 29.5 [58] | Repeated dose toxicity (spinal cord degeneration) [58] | 2.95 ** [58] | 2.95 × 10−1 | 3.70 × 10−5 |
| HQs | |||||||
| Sample | Isoprene | Benzene | Toluene | p,m-Xylene | Styrene | Ethyl benzene | HI |
| 2 (A) | na (C) | na | na | 4.54 × 10−2 | 1.97 × 10−2 | na | 6.51 × 10−2 |
| 2 (B) | na | na | na | 4.54 × 10−2 | na | 1.93 × 10−3 | 4.73 × 10−2 |
| 3 | na | na | 1.00 × 10−2 | na | na | na | 1.00 × 10−2 |
| 4 | na | 2.42 × 10−2 | na | na | na | na | 2.42 × 10−2 |
| 5 (A) | 1.60 × 10−5 | na | na | na | 6.20 × 10−3 | na | 6.22 × 10−3 |
| 5 (B) | 1.60 × 10−5 | na | na | na | na | 6.00 × 10−4 | 6.16 × 10−4 |
| 12 | 3.70 × 10−5 | na | 9.69 × 10−2 | na | 1.00 × 10−2 | na | 1.07 × 10−1 |
| 18 | na | 1.41 × 10−2 | na | na | na | na | 1.41 × 10−2 |
| VOC | CPFo (mg/kg per Day) | CEDI (mg/kg bw per Day) | CR |
|---|---|---|---|
| Benzene | 1.5 × 10−2 [66] | 5.8 × 10−5 | 8.7 × 10−7 |
| Ethylbenzene | 1.1 × 10−2 [67] | 7.9 × 10−5 | 8.7 × 10−7 |
| Substances’ Concentrations and Ranges (ng mL−1) | |||||
|---|---|---|---|---|---|
| Toluene | Benzene | Styrene | p,m-Xylene | BMA | Reference |
| 0.63 | 0.58 | 0.40 | 0.39 | 0.31 | This work |
| 0.46 | 0.08 | 0.13 | 0.54 | na | [8] |
| 0.46–0.56 | 0.06–0.12 | na 1 | na | na | [15] |
| 0.04–2.54 a | 0.01–0.18 a | na | na | na | [70] |
| VOC | Concentration (mg L−1) | HA One-Day (mg L−1) | HA Ten-Day (mg L−1) |
|---|---|---|---|
| Benzene | 0.00058 | 0.2 | 0.2 |
| Ethylbenzene | 0.00079 | 30 | 3 |
| Styrene | 0.00040 | 20 | 2 |
| Toluene | 0.00094 | 20 | 2 |
| Χylenes | 0.00059 | 40 | 40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tzanetou, E.N.; Manea-Karga, E.; Baira, E.; Boutsikou, T.; Iliodromiti, Z.; Iacovidou, N.; Machera, K.; Kasiotis, K.M. Gas and Liquid Chromatography Mass Spectrometry as a Tool for Elucidating Volatile Organic Compounds (VOCs) and Metabolites in Maternal Milk: A Perspective on Infants’ Health Risk Assessment. Chemosensors 2024, 12, 30. https://doi.org/10.3390/chemosensors12030030
Tzanetou EN, Manea-Karga E, Baira E, Boutsikou T, Iliodromiti Z, Iacovidou N, Machera K, Kasiotis KM. Gas and Liquid Chromatography Mass Spectrometry as a Tool for Elucidating Volatile Organic Compounds (VOCs) and Metabolites in Maternal Milk: A Perspective on Infants’ Health Risk Assessment. Chemosensors. 2024; 12(3):30. https://doi.org/10.3390/chemosensors12030030
Chicago/Turabian StyleTzanetou, Evangelia N., Electra Manea-Karga, Eirini Baira, Theodora Boutsikou, Zoi Iliodromiti, Nicoletta Iacovidou, Kyriaki Machera, and Konstantinos M. Kasiotis. 2024. "Gas and Liquid Chromatography Mass Spectrometry as a Tool for Elucidating Volatile Organic Compounds (VOCs) and Metabolites in Maternal Milk: A Perspective on Infants’ Health Risk Assessment" Chemosensors 12, no. 3: 30. https://doi.org/10.3390/chemosensors12030030
APA StyleTzanetou, E. N., Manea-Karga, E., Baira, E., Boutsikou, T., Iliodromiti, Z., Iacovidou, N., Machera, K., & Kasiotis, K. M. (2024). Gas and Liquid Chromatography Mass Spectrometry as a Tool for Elucidating Volatile Organic Compounds (VOCs) and Metabolites in Maternal Milk: A Perspective on Infants’ Health Risk Assessment. Chemosensors, 12(3), 30. https://doi.org/10.3390/chemosensors12030030








