Abstract
Background: Thymic epithelial tumors (TET) are rare neoplasms of the anterior mediastinum. Surgery is the mainstay treatment for resectable TET, whereas systemic treatments are reserved for unresectable and metastatic tumors. The development of new treatments, such as immune checkpoint inhibitors (ICI) and targeted therapies, with promising results in other types of solid tumors, has led to the investigation of their potential efficacy in TET. The study of tumor microenvironments (TME) is another field of investigation that has gained the interest of researchers. Taking into account the complex structure of the thymus and its function in the development of immunity, researchers have focused on TME elements that could predict ICI efficacy. Materials and Methods: The primary objective of this systematic review was to investigate the efficacy of ICI in TET. Secondary objectives included the toxicity of ICI, the efficacy of targeted therapies in TET, and the evaluation of the elements of TME that may be predictive factors of ICI efficacy. A literature search was conducted in February 2023 using the Ovid Medline and SciVerse Scopus databases. Results: 2944 abstracts were retrieved, of which 31 were retained for the systematic review. Five phase II and one retrospective study assessed ICI efficacy. The overall response rate (ORR) varied from 0% to 34%. Median progression-free survival (PFS) ranged from 3.8 to 8.6 months, being lower in thymic carcinoma (TC) (3.8–4.2 months). Median overall survival (OS) ranged from 14.1 to 35.4 months. Treatment-related adverse events occurred in 6.6% to 27.3% of patients. Sixteen studies assessed targeted therapies. The most active molecule was lenvatinib, with 38% ORR in patients with TC while no activity was detected for imatinib, erlotinib plus bevacizumab, and saracatinib. Ten studies assessed TME elements that could predict ICI efficacy. Four studies focused on the tumor-infiltrating immune cells suggesting improved outcomes in patients with TC and high tumor-infiltrating lymphocyte densities. Another study showed that CD8+, CD20+, and CD204+ tumor-infiltrating immune cells in cancer stroma might be prognostic biomarkers in TC. Another study identified the immune-related long non-coding RNAs as a predictor of response to ICI. Tumor mutational burden was identified as a predictive factor of ICI efficacy in one study. Conclusions: Despite study heterogeneity, this review shows that ICI could be a therapeutic option for selected patients with TET that are not amenable to curative radical treatment after first-line chemotherapy.
1. Introduction
Thymic epithelial tumors (TET), which account for 15% of all anterior mediastinal tumors, are uncommon neoplasms of the prevascular mediastinum [1]. They are derived from the epithelial cells of the thymus and are categorized in relation to the fraction of the non-tumoral lymphocytic part, and to their resemblance to normal thymic architecture. This heterogeneous group of neoplastic lesions includes thymomas and thymic carcinomas (TC). The 2015 revised World Health Organization (WHO) classification system classified TET as A, AB, B1, B2, and B3 thymoma, and TC [2]. Thymomas may present an indolent course and, for that reason, were formerly considered benign neoplasms. However, they are nowadays classified as malignant lesions. The overall prognosis is good for thymomas that are amenable to complete surgical resection [3]. On the contrary, TC are characterized by a more clinically aggressive behavior [3,4]. The majority of patients are eligible for surgical treatment, which may be combined with adjuvant radiation. Chemotherapy is prescribed to patients with unresectable TET, at advanced stages (stages III–IV according to either the Masaoka–Koga or the ITMIG classification), or for recurring diseases [4,5]. Platinum-based chemotherapy is the most often used regimen but there is no formal consensus on the best regimen due to the absence of randomized trials in these rare entities [3,4,5].
Recently, the introduction of new treatments, such as immune checkpoint inhibitors (ICI), with promising results in other types of solid tumors, has led researchers to investigate their potential efficacy in TET [6,7,8,9,10,11,12,13]. However, there is limited evidence about their clinical advantage.
Similarly, there is an increased interest in the detection of molecular alterations that could be modified by targeted therapies [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29]. New drugs are selectively targeting the pathways that play an important role in oncogenesis, tumor growth, and proliferation. The rarity and the histological heterogeneity of TET constitute a major obstacle in the conduct of large-scale trials and, until now, the majority of evidence has derived from case reports and small case series.
Another research field that has gained recent interest is that regarding the tumor microenvironment (TME) [30]. The TME consists mainly of endothelial cells of the vascular epithelium, cells of innate and adaptive immunity, fibroblasts, pericytes, signal-carrying molecules, and the extracellular matrix. A close dynamic relationship exists between the tumor and its microenvironment [31,32,33]. This complex interaction is a key element in oncogenesis, growth, and tumor spread. Taking into account the complex structure of the thymus and its function in the development of immunity, researchers are focusing on the TME elements that could predict ICI efficacy [32,33]. The expression of PD-L1 on tumor cells contributes to the prediction of clinical efficacy of ICI in some tumor types, such as non-small cell lung cancer, which take advantage of the crucial roles played by the PD-L1/PD-1 and CTLA4/CD80/CD86 axes in the evasion of immune surveillance. As a result, the distinct composition of TME within various TET histological categories, along with the variations seen among them in the PD-L1 expression patterns, constitutes an essential component of their biological background and directly regulate both the response to checkpoint-inhibitory receptor blockade and the predisposition to autoimmune disease [31,32,33].
The objective of this systematic review was to assess the most recent data on the effectiveness of immunotherapy treatments and the advantages of targeted therapies against potentially treatable mutations in TET. Additionally, components of the TME that might serve as indicators of ICI effectiveness were evaluated.
2. Material and Methods
The literature search, which was designed by a scientific librarian (VD) with competence in medical literature research, was carried out in February 2023 utilizing the Ovid Medline and SciVerse Scopus databases. The search parameters were converted into MeSH terms and free-text keywords, which were then used to search for specific information in titles, abstracts, keywords, and substance names (where applicable) in Medline and titles, abstracts, and keywords in Scopus (Appendix A). The resulting citations were exported from Medline and Scopus into a reference manager software (EndNote version X9) to remove any duplicates, and then in a dedicated systematic literature reviews system (https://rayyan.ai, accessed on 2 February 2023) for the selection process. The researchers worked simultaneously by composing two pairs (ACA and CJ, TB and MB) that ran the initial article selection independently. In the first step, articles were selected if deemed eligible based on the title and abstract content. The final selection was performed after reading the full-text article. The selections of both groups of reviewers were compared and the mutually selected papers represented the total eligible papers to be analyzed. Any discrepancies between the two groups of researchers were resolved after a consensual discussion. The selected articles’ references were examined to detect any missing potentially eligible publications.
The inclusion criteria were the following:
Only articles in English, French, or Dutch were considered. There was no selection based on the year of publication.
- (1)
- Phase II/III clinical trials and retrospective series (>14 patients according to Simon’s design) [34,35] assessing ICI in TET and reporting at least one of the following clinical outcomes:
- progression-free survival (PFS), defined as the time from randomization to disease progression or death from any cause;
- overall survival (OS), defined as the time from randomization until death from any cause;
- objective response rate (ORR), defined as the proportion of patients who achieved an objective response (partial or complete according to the Response Evaluation Criteria in Solid Tumors (RECIST));
- all grade or grade ≥ 3 treatment-related adverse events.
- (2)
- Phase I/II/III clinical trials and retrospective series (>14 patients according to Simon’s design) assessing targeted therapies against an oncogenic driver mutation or translocation (EGFR, cKIT, KRAS, ALK, BRAF, PDGFR, HER2, MET etc.).
- (3)
- Experimental cohort studies investigating any of the following:
- −
- TME of TET, % of PD-L1 expression in TET or tumor mutational burden (TMB) AND prediction of ICI efficacy.
Phase I trials concerning different types of tumors, even including TET, were not considered.
The following data were extracted: study characteristics (design, patient selection), patients’ characteristics (gender, age, previous treatments), stage and histology, treatments and clinical outcomes (number of arms, hazard ratio (HR), and 95% confidence interval (CI) for PFS and OS, overall response rate (ORR), number of patients with grade 3 or greater adverse events (AEs).
The main judgment criterion was ORR. Secondary judgment criteria were PFS, OS (median and at specific time-point: 1–2–5 years), and grade 3–5 AEs.
Data Synthesis
Given the high heterogeneity in the selected studies in terms of inclusion criteria, treatments, and data presentation, a quantitative analysis was not performed.
3. Results
A total of 2944 abstracts were retrieved through the search equation, of which 31 were eligible for the systematic review. The PRISMA flowchart is depicted in Figure 1.
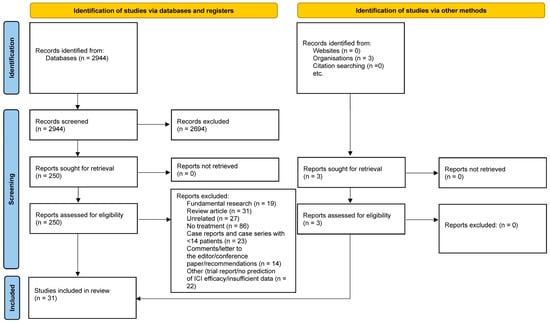
Figure 1.
PRISMA flowchart demonstrating the search strategy.
Six trials assessed ICI efficacy in TET [9,13,36,37,38,39,40,41] (Table 1 and Table 2). Five were phase II trials whereas the last was a retrospective cohort with 77 patients enrolled. All were recently published, from 2018 to 2023. Four studies were multicentric and two were elaborated in a single center. The administered drug was pembrolizumab in two studies and nivolumab in another two studies. In one study the ICI avelumab was combined with the anti-angiogenic agent axitinib. In the last study, different PD-1 inhibitors (anti-PD-1: nivolumab, pembrolizumab, sintilimab, camrelizumab, tislelizumab, and toripalimab) were used. The median number of patients was 37 (range 15–77). ORR was the primary endpoint of four phase II trials and the PFS rate at six months of the fifth one. In half of the studies, only patients with TC were included whereas patients with thymomas and TC were assessed in the remaining studies. The Masaoka–Koga classification was used throughout the trials. All patients presented with stage III (which were not candidates for curative surgical resection) or IV (IVa and/or IVb). The median follow-up duration was 14.9 months (range 13.3–22.4 months). The ORR varied from 0% to 34%. In trials exclusively enrolling patients with TC, the ORR was 0% to 22.5%. The mPFS ranged from 3.8 to 8.6 months overall, being 3.8 to 4.2 months in TC. The mOS ranged from 14.1 to 35.4 months. Treatment-related AE occurred in 6.6% to 27.3% of patients (Table 2).

Table 1.
List of the studies assessing ICI in TET (study design and patient enrollment).

Table 2.
List of the studies assessing ICI in TET (objectives and outcomes).
Sixteen studies testing targeted therapies were deemed eligible for further analysis [23,25,42,43,44,45,46,47,48,49,50,51,52,53,54]. There were thirteen phase II trials, two retrospective studies, and one prospective cohort (Table 3 and Table 4). There were four single-center studies, five studies were conducted in two centers and the remaining seven studies were multicenter trials. Different molecules with various actions were evaluated. The following molecules were assessed:

Table 3.
List of the studies assessing targeted therapies in TET (study design and patient enrollment).

Table 4.
List of the studies assessing targeted therapies in TET (objectives and outcomes).
Regorafenib, a VEGFR-PDGFR-FGFR inhibitor;
Apatinib and anlotinib that have a function of VEGFRs, KIT, PDGFRs TKI;
Sunitinib, a VEGFRs, KIT, PDGFRs TKI;
Buparlisib, a pan-PI3K inhibitor;
Saracatinib, a Src inhibitor;
Cixutumumab, an IGF-1R inhibitor;
Everolimus, an mTOR inhibitor;
Belinostat, a pan-HDAC inhibitor;
Gefitinib and erlotinib with a function of EGFR inhibitors;
Imatinib which is a BCR-ABL TKI;
Milciclib, a pan-cyclin d-dependent kinase inhibitor;
Lenvatinib, a multi-targeted inhibitor of VEGFR, FGFR, RET, c-Kit, and other kinases. Publication years ranged from 2008 to 2023, and the number of enrolled patients varied from 14 to 72. The median follow-up duration ranged from 15.5 to 46 months. Most of the studies (13 out of 16) enrolled patients with both thymomas and TC. In all studies, patients were treated with at least one prior chemotherapy scheme treatment. In seven studies, the ORR was the primary endpoint and ranged from 0% to 38%. The highest ORR (38%) was observed in patients with TC treated with lenvatinib, and mOS was not reached. In three studies with imatinib, a combination of erlotinib and bevacizumab, and saracatinib, the ORR was 0%. Grade 3–4 treatment-related AEs varied according to the molecule and are reported in Table 4.
Ten studies assessed TME elements that could predict the efficacy of ICI [55,56,57,58,59,60,61,62,63,64]. One study was published in 2011 and the others from 2019 to 2022. Three studies were based on resected TC specimens (10, 32, and 39 patients). Four studies included mixed histologies (thymomas and TC) with 21, 22, 31, and 33 specimens. In three studies, TETs were included among different cancer types. The results of these different studies are detailed in Table 5. Four studies focused on the tumor-infiltrating immune cells suggesting improved outcomes in patients with TC and high tumor-infiltrating lymphocyte densities [55,57,59,60]. Another one showed that CD8+, CD20+, and CD204+ tumor-infiltrating immune cells in cancer stroma might be prognostic biomarkers in TC [58]. Another study identified the immune-related long non-coding RNAs as a predictor of response to immunotherapy [56]. A Chinese study identified TMB as a predictive factor of ICI efficacy [63]. Researchers from South Korea assessed the first-week proliferative response of PD-1+ CD8+ T cells as a predictive marker [61]. The immunological pathways predisposing to irAE are the subject of another study [62].

Table 5.
Studies investigating the elements of the TME that could be predictive factors of ICI efficacy.
4. Discussion
TETs are rare and histologically heterogeneous tumors. However, they represent the majority of neoplasms located in the anterior (prevascular) mediastinum. Surgical resection is the treatment of choice for early-stage and resectable tumors, whereas there is no consensus about the best systematic treatments for advanced diseases. This systematic review presents updated data on the clinical activity of ICI and targeted therapies in advanced and metastatic thymoma and TC. It provides a comprehensive review of the existing evidence, which has, until now, been represented mainly by anecdotal case reports and small case series.
4.1. Immune Checkpoint Inhibitors in TET
Immunotherapy has drawn the attention of researchers who are looking into its effectiveness in TET because it has demonstrated encouraging effects in other solid tumors. The crucial function of the thymus gland in the formation of adaptive immune responses makes the TET example fascinating [65]. ICI enhances the immune response against tumors but may also trigger immune-related adverse events (irAE). Accordingly, the activity and toxicity of ICI in clinical trials are emphasized in this systematic review. Preliminary encouraging clinical results are reported with ORR up to 34%, mPFS ranging from 3.8 to 8.6 months, and mOS between 14.1 and 35.4 months. A list of ongoing trials derived from clinicaltrials.gov is shown in Table 6.

Table 6.
List of ongoing trials assessing immune checkpoint inhibitors in thymic epithelial tumors.
However, substantial toxicity is observed in 6.6% to 27.3% of patients, which is a significant barrier to their routine and widespread use. IrAEs generally carry a tolerable level of morbidity but occasionally result in fatal outcomes (varying from 0.36% to 1.23%). Lethal toxicities are less frequent with anti-PD-1 and anti-PD-L1 antibodies, in comparison with anti-CTLA-4 antibodies and certainly for combined administrations (anti-PD-1/PD-L1 plus anti-CTLA-4) [36,66,67]. Currently, ICI (pembrolizumab, nivolumab, and avelumab) must only be used in clinical studies, as the most appropriate group of patients, those with reduced risk of irAEs and with the best therapeutic benefit, must yet be defined.
Immunotherapy agents, unlike those used in conventional cytotoxic therapy, work therapeutically by inducing the anti-tumor immune response, which is based on the immunoregulative process that takes place between cancer cells and the TME. In numerous cancer types, attempts to link PD-L1 expression in tumor cells and ICI effectiveness have shown inconsistent results [58]. PD-L1 expression in thymic epithelial malignancies has been documented in earlier research, but its application as a diagnostic biomarker in TET is not well understood. Therefore, more precise biomarkers and more pertinent predictive features for the identification of individuals who will potentially benefit from ICIs are needed to guide patients’ selection [55].
The thymus is an organ in charge of the evolution of adaptive immunity. Thymus cell lymphocytes or T cells, which are crucial components of adaptive immune function, mature in the thymus. More specifically, the complex thymic structure provides a special microscopic environment that directs thymocyte maturation and instructs T cells to develop self-tolerance [65]. More lymphocytes may boost the likelihood of a successful application of ICI because they fight cancer by increasing cytotoxic lymphocytes [59]. ICI effectiveness in TC patients is supported by improved outcomes in patients with high tumor-infiltrating lymphocyte densities [60]. Effector cells targeting cancer cells, including CD8+ cells, are a predictive marker for ICI activity [68]. The balance between effector and suppressor cells may be crucial for the TME function and might serve as a prognostic and predictive biomarker for ICI [69]. Previous studies have suggested that effector cells, such as CD8+ lymphocytes, are favorable prognostic indicators among patients with TC, but these data are inconsistent [57,70]. Sato et al. have demonstrated that CD8+, CD20+, and CD204+ tumor-infiltrating immune cells in cancer stroma might be prognostic biomarkers in TC. More specifically, high mean numbers of stromal CD8+, CD20+, and FOXP3+ cells have been shown to be significantly associated with favorable prognosis, whereas high CD204+ cell density tended to be correlated with poor prognosis [58]. Shim et al. have confirmed these findings, with a link between better TC prognosis and higher density of stromal CD20+ cells (B lymphocytes) [57]. These results suggest that thymic malignancy differs from other cancer types in the influence of CD20+ cells and that the density of CD20+ tumor-infiltrating immune cells in stromal lesions has to be examined. This opens the door to the investigation of immunotherapy approaches targeting B cell immunity in TC [57].
Other arguments suggest the important role of TME in predicting ICI activity or toxicity in TET. Su et al. have developed an immune-related long noncoding RNAs classifier to pinpoint the response in patients with TET. As the authors advocate, long noncoding RNAs can control the immune response by controlling homeostasis, TME, anti-inflammatory agents, and immune cell activity [56]. Six prognosis-related immune-related long noncoding RNAs (AC004466.3, AC138207.2, AC148477.2, AL450270.1, HOXB-AS1, and SNHG8) were selected to build an immune-related long noncoding RNAs classifier. According to these authors, their model can be used to forecast outcomes, the degree of immune infiltration, and the effectiveness of immunotherapy in patients diagnosed with TETs. It may also help with individualized immunotherapy counseling.
Kim et al. assessed the first-week proliferative response of PD-1+ CD8+ T cells as a predictive marker of tumor responses to anti-PD-1 therapy and clinical outcomes in patients with TETs. The proliferative response after anti-PD-1 therapy was evaluated by the fold-change in the percentage of Ki-67+ cells among PD-1+ CD8+ T cells on day 7 (Ki-67 D7/D0). In the cohort of patients with TETs, Ki-67 D7/D0 was found to be significantly higher in patients with durable clinical benefits than in those with no durable benefits [61]. However, Ki-67 D7/D0 significantly predicted OS in patients with non-small cell lung cancer, but not in patients with TETs.
Chen et al. investigated the association between protein kinase, DNA-activated, catalytic subunit (PRKDC) mutations and TMB, TME, and response to ICI on solid tumor samples collected from 3877 patients that underwent a panel-based next-generation sequencing assay [63]. PRKDC is an important gene for DNA double-strand break repair and central T-cell tolerance. PRKDC mutation is one of the significant factors linked to increased TMB, inflamed TME, and greater responsiveness to ICI. It frequently appears to co-exist with defects in other DNA damage repair pathways. However, their specificity in TET needs to be validated in larger-scale trials.
Yip1 interacting factor homolog B (YIF1B) is a membrane protein that belongs to the FinGER protein family. It is involved in the endoplasmic reticulum (ER)-to-Golgi trafficking [71]. Recent research has demonstrated its role in serotonin-induced cancerogenesis. Liu et al. found a positive relationship between YIF1B expression and immune cell infiltration in several cancer types, and YIF1B expression was also found to be positively correlated with TMB, microsatellite instability, and methylation in some cancer types, linking its expression to a possible evaluation of therapy response [64].
In another study, peripheral blood T-cell characteristics are linked to the emergence of irAEs following anti-PD-1 medication and four different patient subgroups are defined: Th17-related, TNF-related, CD8-related Treg-compensated, and CD8-related Treg-uncompensated. Patients with severe irAEs presented a significantly lower fold increase in the frequency of effector regulatory T (eTreg) cells after anti-PD-1 treatment, a higher proportion of T helper-17 (Th17) and T helper-1 cells in the beginning, and an increased fraction of Ki-67+ cells among PD-1+ CD8+ T cells post treatment. Various irAE subtypes may have unique underlying immunological processes [62]. Early assessment of immune responses may also have clinical implications for irAE prediction.
4.2. Targeted Therapies in TET
Compared with thymomas, TC exhibits more somatic mutations in cancer-related genes [72]. Thus, it is reasonably expected that thymoma and TC may have distinct responses to targeted therapies. Different potential targets have been identified in thymoma and/or TC that are reported hereafter.
Except for a few isolated case reports in Asian individuals, somatic activating EGFR mutations are relatively uncommon in thymic malignancies [73,74]. EGFR protein overexpression is present in approximately 70% of thymomas and 50% of TC without any relationship with the histologic subtype [75,76]. About 20% of thymic malignancies exhibit EGFR gene amplification by fluorescence in situ hybridization (FISH), most frequently in type B3 thymoma and TC, related to more advanced stage and capsule invasion.
KIT immunohistochemical positivity can be seen in up to 73–86% of TC but only in 2% of thymomas [76,77]. As KIT is a target in other tumor forms, most notably in gastrointestinal stromal tumors, this variation in tumor biology results in a definite difference in therapeutic approaches between TCs and thymomas. Unfortunately, the rate of KIT mutations is still only 7 to 9%, despite the high frequency of KIT expression in TC. Four mutations have been described to date: the V560 deletion and L576P substitution found in exon 11, the D820E mutation in exon 17, and the H697Y mutation found in exon 14 [20,22,78].
Angiogenesis significantly influences TET carcinogenesis. Both thymomas and TC overexpress vascular endothelial growth factor (VEGF)-A and VEGFR-1 and -2, although there is little information on the effectiveness of angiogenesis inhibitors in thymic malignancies [79,80]. Low response rates have been observed with bevacizumab [21]. The activity of multikinase inhibitors, particularly sorafenib, and sunitinib, has been emphasized in case reports involving TC [21]. While multikinase inhibitors may have some impact on TC, angiogenesis inhibitors by themselves do not seem to have an effect on either thymomas or TC.
The overexpression of insulin-like growth factor-1 (IGF-1)/IGF-1 receptor (IGF-1R) is a poor prognostic factor in TET. Expression of IGF-1R varies between thymomas (4%) and TC (37%), implying different tumor biologies that might be the subject of targeted therapies [81]. In a retrospective analysis, IGF-1R expression was decreased in types A, AB, and B1 thymomas in comparison with types B2, B3, and TC [82]. A phase II study of cixutumumab, an IGF-1R monoclonal antibody, in 49 patients with previously treated advanced thymic tumors showed limited activity in thymoma (ORR 14%, 95% CI 5–29%) and no effectiveness in TC (ORR 0%, 95% CI 0–26%) [23].
Histone deacetylase (HDAC) inhibitors, in particular the pan-HDAC inhibitor belinostat, were also assessed in TET. A patient with thymoma who participated in a phase I study with belinostat experienced a mild response that lasted for 17 months [83]. A phase II trial with intravenous infusion of belinostat showed only two partial responses in thymomas (ORR 8%, 95% CI 2.3–25.9%) and no responses in TC [25].
The present systematic review has pointed out that the most active agent is lenvatinib, as demonstrated in the REMORA phase II trial. Lenvatinib is a multi-targeted inhibitor of VEGFR, FGFR, RET, c-kit, and other kinases. Further, we can suggest sunitinib as an acceptable second-line therapy for TC [44]. Imatinib demonstrated limited activity in chemotherapy-pretreated patients with TC harboring KIT mutations [27,84,85,86]. Everolimus is a potential treatment option for pre-treated patients with TETs when considering durable disease control in a significant proportion of patients with thymomas or TC [49]. Further investigations are underway, including antiangiogenic combinations, for example, ramucirumab with carboplatin and paclitaxel in a first-line setting [87].
5. Conclusions
Thymic malignancies are a heterogeneous group of cancers. Heterogeneity and rarity hinder the elaboration of large-scale randomized trials. This systematic review focusing on ICI and targeted therapies shows that ICI and some targeted therapies could be pertinent options for patients with TET not amenable to curative radical treatment when first-line chemotherapy fails. However, it is necessary to be able to define the group of patients most likely to benefit from these molecules by taking into account the benefit/toxicity ratio. Continuous research, not only towards the development of new drugs but also at the microscopic level, should define new targets, and better underline predictors of treatment efficacy and toxicity.
Author Contributions
Conceptualization, A.C.A. and T.B.; methodology, T.B. and V.D.; software, V.D.; validation, A.C.A., T.B. and V.D.; formal analysis, A.C.A., M.B. and C.J.; investigation, A.C.A., M.B. and C.J.; resources, V.D.; data curation, all authors.; writing—original draft preparation, all authors.; writing—review and editing, all authors; visualization, all authors.; supervision, T.B.; project administration, A.C.A. and T.B.; funding acquisition, n/a. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The following data are publicly available and can be retrieved upon demand: template data collection forms; data extracted from included studies; review protocol. The review is registered in INPLASY (INPLASY202380080).
Conflicts of Interest
Mariana Brandão has the following COIs but none are related to this work: travel grant: Sanofi, Takeda, AstraZeneca; Sanofi speaker fee: Janssen, Takeda, Pfizer, BMS; advisory board: Sanofi, Janssen; Amgen research grants (my institution): Roche/GNE, AstraZeneca, Merck, Boehringer, Merus, Sanofi, Oxford, and iTeos; PI in clinical trials: Roche/GNE, AstraZeneca, Boehringer, Merus, Sanofi. Thierry Berghmans has the following COIs but none are related to this work: Consultancy for InhaTarget; participation in advisory board for Bayer, Janssen, Merck, BMS, Daiichi-Sankyo, Roche; investigator for Pfizer, Merck, Astra Zeneca, Novartis, Peregrine, Amgen, Novocure; travel grant (Takeda). Apostolos C. Agrafiotis, Valérie Durieux and Christiane Jungels do not have COIs to declare.
Appendix A. The Literature Search Strategy
- Subject: immunotherapy and targeted therapies for thymic epithelial tumors
- Medline via l’interface OvidSP (Epub Ahead of Print, In-Process & Other Non-Indexed Citations, Ovid Medline® Daily and Ovid Medline® 1946-present)
- P = exp Thymus Neoplasms/ OR thymus neoplasm*.ti,ab,kw OR thymus cancer*.ti,ab,kw OR Thymus Carcinoma*.ti,ab,kw OR thymus tumour*.ti,ab,kw OR thymus tumor*.ti,ab,kw OR Thymoma*.ti,ab,kw OR thymic neoplasm*.ti,ab,kw OR thymic cancer*.ti,ab,kw OR thymic tumour*.ti,ab,kw OR thymic tumor*.ti,ab,kw OR Thymic Carcinoma*.ti,ab,kw OR Thymic Epithelial Tumor*.ti,ab,kw OR Thymic Epithelial Tumour*.ti,ab,kw
- I = exp Angiogenesis Modulating Agents/ OR exp Immunotherapy/ OR immunotherap*.ti,ab,kw OR immunization*.ti,ab,kw OR immunosuppression*.ti,ab,kw OR radioimmunotherap*.ti,ab,kw OR vaccin*.ti,ab,kw OR Cixutumumab.ti,ab,kw,nm OR anti-IGF-1R antibody A12.ti,ab,kw,nm OR Tivantinib.ti,ab,kw,nm OR ARQ 197.ti,ab,kw,nm OR Vorinostat/ OR Vorinostat.ti,ab,kw,nm OR Zolinza.ti,ab,kw,nm OR Ramucirumab.ti,ab,kw,nm OR Ipilimumab/ OR Ipilimumab.ti,ab,kw,nm OR Yervoy.ti,ab,kw,nm OR Anti-CTLA-4.ti,ab,kw,nm OR Tremelimumab.ti,ab,kw,nm OR ticilimumab.ti,ab,kw,nm OR Nivolumab/ OR Nivolumab.ti,ab,kw,nm OR Opdivo.ti,ab,kw,nm OR Pembrolizumab.ti,ab,kw,nm OR lambrolizumab.ti,ab,kw,nm OR Keytruda.ti,ab,kw,nm OR Atezolizumab.ti,ab,kw,nm OR MPDL3280A.ti,ab,kw,nm OR Durvalumab.ti,ab,kw,nm OR Avelumab.ti,ab,kw,nm OR Amatuximab.ti,ab,kw,nm OR MORAb-009.ti,ab,kw,nm OR SS1P.ti,ab,kw,nm OR anetumab ravtansine.ti,ab,kw,nm OR BAY 94-9343.ti,ab,kw,nm OR BNC105P.ti,ab,kw,nm OR BNC-105P.ti,ab,kw,nm OR ADI-PEG20.ti,ab,kw,nm OR pegylated arginine deiminase.ti,ab,kw,nm OR interleukin*.ti,ab,kw,nm OR interferon*.ti,ab,kw,nm OR EZH.ti,ab,kw,nm OR enhancer of zeste homolog.ti,ab,kw,nm OR Immune Checkpoint Inhibitors/ OR Immune Checkpoint Inhibitor*.ti,ab,kw,nm OR Immune Checkpoint Blockade.ti,ab,kw,nm OR PD L1.ti,ab,kw,nm OR PD 1 Inhibitor*.ti,ab,kw,nm OR Tumor Microenvironment/ OR Tumor Microenvironment*.ti,ab,kw OR Tumour Microenvironment*.ti,ab,kw OR Cancer Microenvironment*.ti,ab,kw OR Epidermal Growth Factor/ OR Epidermal Growth Factor.ti,ab,kw OR "HER2/Neu".ti,ab,kw,nm OR "anti-HER-2/neu".ti,ab,kw,nm OR exp Vascular Endothelial Growth Factors/ OR Vascular Endothelial Growth Factor*.ti,ab,kw,nm OR VEGFs.ti,ab,kw,nm OR exp Fibroblast Growth Factors/ OR Fibroblast Growth Factor*.ti,ab,kw,nm OR bFGF.ti,ab,kw,nm OR Tyrosine Protein Kinase Inhibitors/ OR TKI.ti,ab,kw,nm OR Tyrosine kinase inhibitor*.ti,ab,kw,nm OR CTLA-4 Antigen/ OR CTLA 4.ti,ab,kw,nm OR druggable molecular anomal*.ti,ab,kw OR immune-mediated adverse effect*.ti,ab,kw OR genetic marker*.ti,ab,kw OR immune microenvironment.ti,ab,kw OR PI3K.ti,ab,kw,nm
- = 1724 (6/02/2023) – 1715 (after duplicate removal)
- SciVerse Scopus
- P = TITLE-ABS-KEY(“thymus neoplasm*” OR “thymus cancer*” OR “Thymus Carcinoma*” OR “thymus tumour*” OR “thymus tumor*” OR Thymoma* OR “thymic neoplasm*” OR "thymic cancer*” OR “thymic tumour*” OR “thymic tumor*” OR “Thymic Carcinoma*” OR “Thymic Epithelial Tumor*” OR “Thymic Epithelial Tumour*”)
- I = TITLE-ABS-KEY(immunotherap* OR immunization* OR immunosuppression* OR radioimmunotherap* OR vaccin* OR Cixutumumab OR “anti-IGF-1R antibody A12” OR Tivantinib OR “ARQ 197” OR Vorinostat OR Zolinza OR Ramucirumab OR Ipilimumab OR Yervoy OR “Anti-CTLA-4” OR Tremelimumab OR ticilimumab OR Nivolumab OR Opdivo OR Pembrolizumab OR lambrolizumab OR Keytruda OR Atezolizumab OR “MPDL3280A” OR Durvalumab OR Avelumab OR Amatuximab OR MORAb-009 OR SS1P OR “anetumab ravtansine” OR “BAY 94-9343” OR BNC105P OR BNC-105P OR ADI-PEG20 OR “pegylated arginine deiminase” OR interleukin* OR interferon* OR EZH OR “enhancer of zeste homolog” OR “Immune Checkpoint Inhibitor*” OR “Immune Checkpoint Blockade” OR “PD L1” OR “PD 1 Inhibitor*” OR “Tumor Microenvironment*” OR “Tumour Microenvironment*” OR “Cancer Microenvironment*” OR “Epidermal Growth Factor” OR “HER2/Neu” OR “anti-HER-2/neu” OR “Vascular Endothelial Growth Factor*” OR VEGFs OR “Fibroblast Growth Factor*” OR bFGF OR TKI OR “Tyrosine kinase inhibitor*” OR “CTLA 4” OR “druggable molecular anomal*” OR “immune-mediated adverse effect*” OR “genetic marker*” OR “immune microenvironment” OR PI3K)
- = 2747 (6/2/2023)
- = 2944 (6/2/2023) merged
References
- Wright, C.D. Management of thymomas. Crit Rev. Oncol. Hematol. 2008, 65, 109–120. [Google Scholar]
- Marx, A.; Chan, J.K.; Coindre, J.-M.; Detterbeck, F.; Girard, N.; Harris, N.L.; Jaffe, E.S.; Kurrer, M.O.; Marom, E.M.; Moreira, A.L.; et al. The 2015 World Health Organization classification of tumors of the thymus: Continuity and changes. J. Thorac. Oncol. 2015, 10, 1383–1395. [Google Scholar]
- Ko, R.; Shukuya, T.; Okuma, Y.; Tateishi, K.; Imai, H.; Iwasawa, S.; Miyauchi, E.; Fujiwara, A.; Sugiyama, T.; Azuma, K.; et al. Prognostic factors and efficacy of first-line chemotherapy in patients with advanced thymic carcinoma: A retrospective analysis of 286 patients from NEJ023 study. Oncologist 2018, 23, 1210–1217. [Google Scholar]
- Berghmans, T.; Durieux, V.; Holbrechts, S.; Jungels, C.; Lafitte, J.-J.; Meert, A.-P.; Moretti, L.; Ocak, S.; Roelandts, M.; Girard, N. Systemic treatments for thymoma and thymic carcinoma: A systematic review. Lung Cancer 2018, 126, 25–31. [Google Scholar]
- Girard, N.; Ruffini, E.; Marx, A.; Faivre-Finn, C.; Peters, S.; ESMO Guidelines Committee. Thymic epithelial tumours: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26 (Suppl. S6), v40–v55. [Google Scholar]
- Ak, N.; Aydiner, A. Nivolumab treatment for metastatic thymic epithelial tumors. J. Oncol. Pharm. Pract. 2021, 27, 1710–1715. [Google Scholar] [CrossRef]
- Zander, T.; Aebi, S.; Rast, A.C.; Zander, A.; Winterhalder, R.; Brand, C.; Diebold, J.; Gautschi, O. Response to Pembrolizumab in a patient with relapsing thymoma. J. Thorac. Oncol. 2016, 11, e147–e149. [Google Scholar] [CrossRef]
- Uchida, N.; Fujita, K.; Okamura, M.; Nakatani, K.; Mio, T. The clinical benefits of immune checkpoint inhibitor for thymic carcinomas approximately experience of single public hospital in Japan approximately. Respir. Med. Case Rep. 2019, 26, 39–41. [Google Scholar]
- Katsuya, Y.; Horinouchi, H.; Seto, T.; Umemura, S.; Hosomi, Y.; Satouchi, M.; Nishio, M.; Kozuki, T.; Hida, T.; Sukigara, T.; et al. Single-arm, multicentre, phase II trial of nivolumab for unresectable or recurrent thymic carcinoma: PRIMER study. Eur. J. Cancer 2019, 113, 78–86. [Google Scholar]
- Yang, Y.; Ding, L.; Wang, P. Dramatic response to anti-PD-1 therapy in a patient of squamous cell carcinoma of thymus with multiple lung metastases. J. Thorac. Dis. 2016, 8, E535–E537. [Google Scholar] [CrossRef][Green Version]
- Yang, P.-C.; Guo, J.-C.; Hsieh, M.-S.; Lin, C.-C.; Hsu, C.-H. Response to nivolumab as salvage therapy in a patient with thymic carcinoma. J. Thorac. Oncol. 2018, 13, e36–e39. [Google Scholar] [CrossRef] [PubMed]
- Girard, N. Immune checkpoints in thymic epithelial tumors: Challenges and opportunities. Immuno-Oncol. Technol. 2019, 3, 8–14. [Google Scholar]
- Nivolumab in Patients with Type B3 Thymoma and Thymic Carcinoma (NIVOTHYM). Available online: https://clinicaltrials.gov/ct2/show/NCT03134118?term=NCT03134118&draw=2&rank=1 (accessed on 15 May 2023).
- Palmieri, G.; Marino, M.; Salvatore, M.; Budillon, A.; Meo, G.; Caraglia, M.; Montella, L. Cetuximab is an active treatment of metastatic and chemorefractory thymoma. Front. Biosci. 2007, 12, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Farina, G.; Garassino, M.C.; Gambacorta, M.; La Verde, N.; Gherardi, G.; Scanni, A. Response of thymoma to cetuximab. Lancet Oncol. 2007, 8, 449–450. [Google Scholar] [CrossRef]
- Christodoulou, C.; Murray, S.; Dahabreh, J.; Petraki, K.; Nikolakopoulou, A.; Mavri, A.; Skarlos, D. Response of malignant thymoma to erlotinib. Ann. Oncol. 2008, 19, 1361–1362. [Google Scholar] [CrossRef]
- Pedersini, R.; Vattemi, E.; Lusso, M.R.; Mazzoleni, G.; Ebner, H.; Graiff, C. Erlotinib in advanced well-differentiated thymic carcinoma with overexpression of EGFR: A case report. Tumori J. 2008, 94, 849–852. [Google Scholar] [CrossRef]
- Nakagiri, T.; Funaki, S.; Kadota, Y.; Takeuchi, Y.; Shiono, H.; Akashi, A.; Okumura, M. Does gefitinib have effects on EGFR mutation-positive thymoma? Case report of thymoma recurrence. Ann. Thorac. Cardiovasc. Surg. 2014, 20, 674–676. [Google Scholar] [CrossRef]
- Giaccone, G.; Rajan, A.; Ruijter, R.; Smit, E.; van Groeningen, C.; Hogendoorn, P.C. Imatinib mesylate in patients with WHO B3 thymomas and thymic carcinomas. J. Thorac. Oncol. 2009, 4, 1270–1273. [Google Scholar] [CrossRef]
- Ströbel, P.; Hartmann, M.; Jakob, A.; Mikesch, K.; Brink, I.; Dirnhofer, S.; Marx, A. Thymic carcinoma with overexpression of mutated KIT and the response to imatinib. N. Engl. J. Med. 2004, 350, 2625–2626. [Google Scholar] [CrossRef]
- Buti, S.; Donini, M.; Sergio, P.; Garagnani, L.; Schirosi, L.; Passalacqua, R.; Rossi, G. Impressive response with imatinib in a heavily pretreated patient with metastatic c-KIT mutated thymic carcinoma. J. Clin. Oncol. 2011, 29, e803–e805. [Google Scholar] [CrossRef]
- Bisagni, G.; Rossi, G.; Cavazza, A.; Sartori, G.; Gardini, G.; Boni, C. Long lasting response to the multikinase inhibitor bay 43–9006 (Sorafenib) in a heavily pretreated metastatic thymic carcinoma. J. Thorac. Oncol. 2009, 4, 773–775. [Google Scholar] [CrossRef] [PubMed]
- Rajan, A.; Carter, C.A.; Berman, A.; Cao, L.; Kelly, R.J.; Thomas, A.; Khozin, S.; Chavez, A.L.; Bergagnini, I.; Scepura, B.; et al. Cixutumumab for patients with recurrent or refractory advanced thymic epithelial tumours: A multicentre, open-label, phase 2 trial. Lancet Oncol. 2014, 15, 191–200. [Google Scholar] [PubMed]
- Wheler, J.J.; Hong, D.; Swisher, S.G.; Falchook, G.S.; Tsimberidou, A.M.; Helgason, T.; Naing, A.; Stephen, B.; Janku, F.; Stephens, P.J.; et al. Thymoma patients treated in a phase I clinic at MD Anderson Cancer Center: Responses to mTOR inhibitors and molecular analyses. Oncotarget 2013, 4, 890–898. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Giaccone, G.; Rajan, A.; Berman, A.; Kelly, R.J.; Szabo, E.; Lopez-Chavez, A.; Trepel, J.; Lee, M.-J.; Cao, L.; Espinoza-Delgado, I.; et al. Phase II study of belinostat in patients with recurrent or refractory advanced thymic epithelial tumors. J. Clin. Oncol. 2011, 29, 2052–2059. [Google Scholar] [CrossRef]
- Azad, A.; Herbertson, R.A.; Pook, D.; White, S.; Mitchell, P.L.; Tebbutt, N.C. Motesanib diphosphate (AMG 706), an oral angiogenesis inhibitor, demonstrates clinical efficacy in advanced thymoma. Acta Oncol. 2009, 48, 619–621. [Google Scholar] [CrossRef]
- Ströbel, P.; Bargou, R.; Wolff, A.; Spitzer, D.; Manegold, C.; Dimitrakopoulou-Strauss, A.; Strauss, L.; Sauer, C.; Mayer, F.; Hohenberger, P.; et al. Sunitinib in metastatic thymic carcinomas: Laboratory findings and initial clinical experience. Br. J. Cancer 2010, 103, 196–200. [Google Scholar] [CrossRef]
- Chuah, C.; Lim, T.H.; Lim, A.S.T.; Tien, S.L.; Lim, C.H.; Soong, R.; Lee, F.; Linn, Y.C.; Goh, Y.T.; Cheah, F.K.; et al. Dasatinib induces a response in malignant thymoma. J. Clin. Oncol. 2006, 24, e56–e68. [Google Scholar] [CrossRef]
- Neuhaus, T.; Luyken, J. Long lasting efficacy of sorafenib in a heavily pretreated patient with thymic carcinoma. Target. Oncol. 2012, 7, 247–251. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar]
- Agrafiotis, A.C.; Siozopoulou, V.; Hendriks, J.M.H.; Pauwels, P.; Koljenovic, S.; Van Schil, P.E. Tumor Microenvironment in Thymic Epithelial Tumors: A Narrative Review. Cancers 2022, 14, 6082. [Google Scholar]
- Ohm, B.; Jungraithmayr, W. Balancing the Risk of Adverse Events against the Efficacy of Immunotherapy in Advanced Thymic Epithelial Tumors. Cancers 2023, 15, 289. [Google Scholar]
- Masaoutis, C.; Palamaris, K.; Kokkali, S.; Levidou, G.; Theocharis, S. Unraveling the Immune Microenvironment of Thymic Epithelial Tumors: Implications for Autoimmunity and Treatment. Int. J. Mol. Sci. 2022, 23, 7864. [Google Scholar] [CrossRef]
- Simon, R. Design, Analysis and Reporting of Cancer Clinical Trials. In Biopharmaceutical Statistics for Drug Development; Peace, K.E., Ed.; Marcel Dekker: New York, NY, USA, 1987. [Google Scholar]
- Simon, R. Optimal two-stage designs for phase II clinical trials. Control. Clin. Trials 1989, 10, 1–10. [Google Scholar] [PubMed]
- Giaccone, G.; Kim, C.; Thompson, J.; McGuire, C.; Kallakury, B.; Chahine, J.J.; Manning, M.; Mogg, R.; Blumenschein, W.M.; Tan, M.T.; et al. Pembrolizumab in patients with thymic carcinoma: A single-arm, single-centre, phase 2 study. Lancet Oncol. 2018, 19, 347–355. [Google Scholar] [PubMed]
- Giaccone, G.; Kim, C. Durable Response in Patients with Thymic Carcinoma Treated with Pembrolizumab After Prolonged Follow-Up. J. Thorac. Oncol. 2021, 16, 483–485. [Google Scholar] [CrossRef]
- Cho, J.; Kim, H.S.; Ku, B.M.; Choi, Y.-L.; Cristescu, R.; Han, J.; Sun, J.-M.; Lee, S.-H.; Ahn, J.S.; Park, K.; et al. Pembrolizumab for patients with refractory or relapsed thymic epithelial tumor: An open-label phase II trial. J. Clin. Oncol. 2019, 37, 2162–2170. [Google Scholar] [CrossRef]
- Wang, W.; Lin, G.; Hao, Y.; Guan, Y.; Zhang, Y.; Xu, C.; Wang, Q.; Wang, D.; Jiang, Z.; Cai, J.; et al. Treatment outcomes and prognosis of immune checkpoint inhibitors therapy in patients with advanced thymic carcinoma: A multicentre retrospective study. Eur. J. Cancer 2022, 174, 21–30. [Google Scholar]
- Conforti, F.; Zucali, P.A.; Pala, L.; Catania, C.; Bagnardi, V.; Sala, I.; Della Vigna, P.; Perrino, M.; Zagami, P.; Corti, C.; et al. Avelumab plus axitinib in unresectable or metastatic type B3 thymomas and thymic carcinomas (CAVEATT): A single-arm, multicentre, phase 2 trial. Lancet Oncol. 2022, 23, 1287–1296. [Google Scholar] [CrossRef]
- Girard, N.; Aix, S.P.; Cedres, S.; Berghmans, T.; Burgers, S.; Toffart, A.-C.; Popat, S.; Janssens, A.; Gervais, R.; Hochstenbag, M.; et al. Efficacy and safety of nivolumab for patients with pre-treated type B3 thymoma and thymic carcinoma: Results from the EORTC-ETOP NIVOTHYM phase II trial. ESMO Open 2023, 8, 101576. [Google Scholar] [CrossRef]
- Perrino, M.; De Pas, T.; Bozzarelli, S.; Giordano, L.; De Vincenzo, F.; Conforti, F.; Digiacomo, N.; Cordua, N.; D’Antonio, F.; Borea, F.; et al. Resound Trial: A phase 2 study of regorafenib in patients with thymoma (type B2-B3) and thymic carcinoma previously treated with chemotherapy. Cancer 2022, 128, 719–726. [Google Scholar] [CrossRef]
- Remon, J.; Girard, N.; Mazieres, J.; Dansin, E.; Pichon, E.; Greillier, L.; Dubos, C.; Lindsay, C.R.; Besse, B. Sunitinib in patients with advanced thymic malignancies: Cohort from the French RYTHMIC network. Lung Cancer 2016, 97, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Rajan, A.; Berman, A.; Tomita, Y.; Brzezniak, C.; Lee, M.-J.; Lee, S.; Ling, A.; Spittler, A.J.; Carter, C.A.; et al. Sunitinib in patients with chemotherapy-refractory thymoma and thymic carcinoma: An open-label phase 2 trial. Lancet Oncol. 2015, 16, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Antonarelli, G.; Corti, C.; Zucali, P.A.; Perrino, M.; Manglaviti, S.; Russo, G.L.; Varano, G.M.; Salvini, P.; Curigliano, G.; Catania, C.; et al. Continuous sunitinib schedule in advanced platinum refractory thymic epithelial neoplasms: A retrospective analysis from the ThYmic MalignanciEs (TYME) Italian collaborative group. Eur. J. Cancer 2022, 174, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Abu Zaid, M.I.; Radovich, M.; Althouse, S.; Liu, H.; Spittler, A.J.; Solzak, J.; Badve, S.; Loehrer, P.J., Sr. A phase II study of buparlisib in relapsed or refractory thymomas. Front. Oncol. 2022, 12, 891383. [Google Scholar] [CrossRef]
- Guan, Y.; Gu, X.; Si, J.; Xiang, J.; Wei, J.; Hao, Y.; Wang, W.; Sun, Y. The efficacy of small molecule anti-angiogenic drugs in previously treated Thymic carcinoma. BMC Cancer 2023, 23, 16. [Google Scholar]
- Gubens, M.A.; Burns, M.; Perkins, S.M.; Pedro-Salcedo, M.S.; Althouse, S.K.; Loehrer, P.J.; Wakelee, H.A. A phase II study of saracatinib (AZD0530), a Src inhibitor, administered orally daily to patients with advanced thymic malignancies. Lung Cancer 2015, 89, 57–60. [Google Scholar]
- Zucali, P.A.; De Pas, T.; Palmieri, G.; Favaretto, A.; Chella, A.; Tiseo, M.; Caruso, M.; Simonelli, M.; Perrino, M.; De Vincenzo, F.; et al. Phase II Study of Everolimus in Patients with Thymoma and Thymic Carcinoma Previously Treated with Cisplatin-Based Chemotherapy. J. Clin. Oncol. 2018, 36, 342–349. [Google Scholar] [CrossRef]
- Kurup, A.; Burns, M.; Dropcho, S.; Pao, W.; Loehrer, P.J. Phase II study of gefitinib treatment in advanced thymic malignancies. J. Clin. Oncol. 2005, 23, 7068. [Google Scholar] [CrossRef]
- Palmieri, G.; Marino, M.; Buonerba, C.; Federico, P.; Conti, S.; Milella, M.; Petillo, L.; Evoli, A.; Lalle, M.; Ceribelli, A.; et al. Imatinib mesylate in thymic epithelial malignancies. Cancer Chemother. Pharmacol. 2012, 69, 309–315. [Google Scholar] [CrossRef]
- Besse, B.; Garassino, M.C.; Rajan, A.; Novello, S.; Mazieres, J.; Weiss, G.J.; Kocs, D.M.; Barnett, J.M.; Davite, C.; Crivori, P.; et al. Efficacy of milciclib (PHA-848125AC), a pan-cyclin d-dependent kinase inhibitor, in two phase II studies with thymic carcinoma (TC) and B3 thymoma (B3T) patients. J. Clin. Oncol. 2018, 36, 8519. [Google Scholar] [CrossRef]
- Sato, J.; Satouchi, M.; Itoh, S.; Okuma, Y.; Niho, S.; Mizugaki, H.; Murakami, H.; Fujisaka, Y.; Kozuki, T.; Nakamura, K.; et al. Lenvatinib in patients with advanced or metastatic thymic carcinoma (REMORA): A multicentre, phase 2 trial. Lancet Oncol. 2020, 21, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Bedano, P.M.; Perkins, S.; Burns, M.; Kessler, K.; Nelson, R.; Schneider, B.P.; Risley, L.; Dropcho, S.; Loehrer, P.J. A phase II trial of erlotinib plus bevacizumab in patients with recurrent thymoma or thymic carcinoma. J. Clin. Oncol. 2008, 26, 19087. [Google Scholar] [CrossRef]
- Hou, X.; Lin, S.; Liu, Y.; Wang, K.; Yu, Z.; Jia, J.; Yu, J.; Zheng, W.; Bai, J.; Chang, L.; et al. Analysis of the tumor microenvironment and mutation burden identifies prognostic features in thymic epithelial tumors. Am. J. Cancer Res. 2022, 12, 2387–2396. [Google Scholar] [PubMed]
- Su, Y.; Ou, Y.; Chen, Y.; Ma, X. Construction of immune-related LncRNAs classifier to predict prognosis and immunotherapy response in thymic epithelial tumors. Biosci. Rep. 2022, 42, BSR20220317. [Google Scholar] [CrossRef] [PubMed]
- Shim, H.S.; Byun, C.S.; Bae, M.K.; Lee, C.Y.; Park, I.K.; Kim, D.J.; Chung, K.Y.; Lee, J.G. Prognostic effect of stromal lymphocyte infiltration in thymic carcinoma. Lung Cancer 2011, 74, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Sato, J.; Kitano, S.; Motoi, N.; Ino, Y.; Yamamoto, N.; Watanabe, S.; Ohe, Y.; Hiraoka, N. CD20+ tumor-infiltrating immune cells and CD204+ M2 macrophages are associated with prognosis in thymic carcinoma. Cancer Sci. 2020, 111, 1921–1932. [Google Scholar] [CrossRef]
- Blessin, N.C.; Spriestersbach, P.; Li, W.; Mandelkow, T.; Dum, D.; Simon, R.; Hube-Magg, C.; Lutz, F.; Viehweger, F.; Lennartz, M.; et al. Prevalence of CD8+ cytotoxic lymphocytes in human neoplasms. Cell. Oncol. 2020, 43, 421–430. [Google Scholar]
- Bocchialini, G.; Schiefer, A.I.; Müllauer, L.; Thanner, J.; Bauer, J.; Thaler, F.; Laggner, M.; Veraar, C.; Klepetko, W.; Hötzenecker, K.; et al. Tumour immune microenvironment in resected thymic carcinomas as a predictor of clinical outcome. Br. J. Cancer 2022, 127, 1162–1171. [Google Scholar]
- Kim, K.H.; Cho, J.; Ku, B.M.; Koh, J.; Sun, J.-M.; Lee, S.-H.; Ahn, J.S.; Cheon, J.; Min, Y.J.; Park, S.-H.; et al. The First-week Proliferative Response of Peripheral Blood PD-1+CD8+ T Cells Predicts the Response to Anti-PD-1 Therapy in Solid Tumors. Clin. Cancer Res. 2019, 25, 2144–2154. [Google Scholar]
- Kim, K.H.; Hur, J.Y.; Cho, J.; Ku, B.M.; Koh, J.; Koh, J.Y.; Sun, J.-M.; Lee, S.-H.; Ahn, J.S.; Park, K.; et al. Immune-related adverse events are clustered into distinct subtypes by T-cell profiling before and early after anti-PD-1 treatment. OncoImmunology 2020, 9, 1722023. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Y.; Guan, Y.; Huang, Y.; Lin, J.; Chen, L.; Li, J.; Chen, G.; Pan, L.K.; Xia, X.; et al. Prevalence of PRKDC mutations and association with response to immune checkpoint inhibitors in solid tumors. Mol. Oncol. 2020, 14, 2096–2110. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, Z.; Zhao, P.; Li, W. Prognostic and immune regulating roles of YIF1B in Pan-Cancer: A potential target for both survival and therapy response evaluation. Biosci. Rep. 2020, 40, BSR20201384. [Google Scholar] [CrossRef] [PubMed]
- Thapa, P.; Farber, D.L. The Role of the Thymus in the Immune Response. Thorac. Surg. Clin. 2019, 29, 123–131. [Google Scholar] [PubMed]
- Arbour, K.C.; Naidoo, J.; Steele, K.E.; Ni, A.; Moreira, A.L.; Rekhtman, N.; Robbins, P.B.; Karakunnel, J.; Rimner, A.; Huang, J.; et al. Expression of PD-L1 and other immunotherapeutic targets in thymic epithelial tumors. PLoS ONE 2017, 12, e0182665. [Google Scholar]
- Tateo, V.; Manuzzi, L.; De Giglio, A.; Parisi, C.; Lamberti, G.; Campana, D.; Pantaleo, M. Immunobiology of Thymic Epithelial Tumors: Implications for Immunotherapy with Immune Checkpoint Inhibitors. Int. J. Mol. Sci. 2020, 21, 9056. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti–PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Mazzaschi, G.; Madeddu, D.; Falco, A.; Bocchialini, G.; Goldoni, M.; Sogni, F.; Armani, G.; Lagrasta, C.A.; Lorusso, B.; Mangiaracina, C.; et al. Low PD-1 expression in cytotoxic CD8 + tumor-infiltrating lymphocytes confers an immune-privileged tissue microenvironment in NSCLC with a prognostic and predictive value. Clin. Cancer Res. 2018, 24, 407–420. [Google Scholar]
- Yokoyama, S.; Miyoshi, H.; Nakashima, K.; Shimono, J.; Hashiguchi, T.; Mitsuoka, M.; Takamori, S.; Akagi, Y.; Ohshima, K. Prognostic value of programmed death ligand 1 and programmed death 1 expression in thymic carcinoma. Clin. Cancer Res. 2016, 22, 4727–4734. [Google Scholar] [CrossRef]
- Graab, P.; Bock, C.; Weiss, K.; Hirth, A.; Koller, N.; Braner, M.; Jung, J.; Loehr, F.; Tampé, R.; Behrends, C.; et al. Lysosomal targeting of the ABC transporter TAPL is determined by membrane-localized charged residues. J. Biol. Chem. 2019, 294, 7308–7323. [Google Scholar] [PubMed]
- Petrini, I.; Meltzer, P.S.; Kim, I.-K.; Lucchi, M.; Park, K.-S.; Fontanini, G.; Gao, J.; A Zucali, P.; Calabrese, F.; Favaretto, A.; et al. A specific missense mutation in GTF2I occurs at high frequency in thymic epithelial tumors. Nat. Genet. 2014, 46, 844–849. [Google Scholar]
- Yoh, K.; Nishiwaki, Y.; Ishii, G.; Goto, K.; Kubota, K.; Ohmatsu, H.; Niho, S.; Nagai, K.; Saijo, N. Mutational status of EGFR and KIT in thymoma and thymic carcinoma. Lung Cancer 2008, 62, 316–320. [Google Scholar] [PubMed]
- Yamaguchi, H.; Soda, H.; Kitazaki, T.; Tsukamoto, K.; Hayashi, T.; Kohno, S. Thymic carcinoma with epidermal growth factor receptor gene mutations. Lung Cancer 2006, 52, 261–262. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, E.; Sasaki, H.; Kawano, O.; Endo, K.; Haneda, H.; Yukiue, H.; Kobayashi, Y.; Yano, M.; Fujii, Y. Expression and mutation statuses of epidermal growth factor receptor in thymic epithelial tumors. Jpn. J. Clin. Oncol. 2006, 36, 351–356. [Google Scholar] [PubMed]
- Henley, J.D.; Cummings, O.W.; Loehrer, P.J., Sr. Tyrosine kinase receptor expression in thymomas. J. Cancer Res. Clin. Oncol. 2004, 130, 222–224. [Google Scholar] [CrossRef]
- Pan, C.C.; Chen, P.C.; Chiang, H. KIT (CD117) is frequently overexpressed in thymic carcinomas but is absent in thymomas. J. Pathol. 2004, 202, 375–381. [Google Scholar] [CrossRef]
- Girard, N.; Shen, R.; Guo, T.; Zakowski, M.F.; Heguy, A.; Riely, G.J.; Huang, J.; Lau, C.; Lash, A.E.; Ladanyi, M.; et al. Comprehensive genomic analysis reveals clinically relevant molecular distinctions between thymic carcinomas and thymomas. Clin. Cancer Res. 2009, 15, 6790–6799. [Google Scholar]
- Cimpean, A.M.; Raica, M.; Encica, S.; Cornea, R.; Bocan, V. Immunohistochemical expression of vascular endothelial growth factor A (VEGF), and its receptors (VEGFR1, 2) in normal and pathologic conditions of the human thymus. Ann. Anat. Anat. Anz. 2008, 190, 238–245. [Google Scholar]
- Sasaki, H.; Yukiue, H.; Kobayashi, Y.; Nakashima, Y.; Moriyama, S.; Kaji, M.; Kiriyama, M.; Fukai, I.; Yamakawa, Y.; Fujii, Y. Elevated serum vascular endothelial growth factor and basic fıbroblast growth factor levels in patients with thymic epithelial neoplasms. Surg. Today 2001, 31, 1038–1040. [Google Scholar] [CrossRef]
- Girard, N.; Teruya-Feldstein, J.; Payabyab, E.C.; Riely, G.J.; Rusch, V.W.; Kris, M.G.; Zakowski, M.F. Insulin-like growth factor-1 receptor expression in thymic malignancies. J. Thorac. Oncol. 2010, 5, 1439–1446. [Google Scholar]
- Zucali, P.A.; Petrini, I.; Lorenzi, E.; Merino, M.; Cao, L.; Di Tommaso, L.; Lee, H.S.; Incarbone, M.; Walter, B.A.; Simonelli, M.; et al. Insulin-like growth factor-1 receptor and phosphorylated AKT-serine 473 expression in 132 resected thymomas and thymic carcinomas. Cancer 2010, 116, 4686–4695. [Google Scholar]
- Steele, N.L.; Plumb, J.A.; Vidal, L.; Tjørnelund, J.; Knoblauch, P.; Rasmussen, A.; Ooi, C.E.; Buhl-Jensen, P.; Brown, R.; Evans, T.R.J.; et al. A phase 1 pharmacokinetic and pharmacodynamic study of the histone deacetylase inhibitor belinostat in patients with advanced solid tumors. Clin. Cancer Res. 2008, 14, 804–810. [Google Scholar] [PubMed]
- Petrini, I.; Zucali, P.A.; Lee, H.S.; Pineda, M.A.; Meltzer, P.S.; Walter-Rodriguez, B.; Roncalli, M.; Santoro, A.; Wang, Y.; Giaccone, G. Expression and mutational status of c-kit in thymic epithelial tumors. J. Thorac. Oncol. 2010, 5, 1447–1453. [Google Scholar] [PubMed]
- Hirai, F.; Edagawa, M.; Shimamatsu, S.; Toyozawa, R.; Toyokawa, G.; Nosaki, K.; Yamaguchi, M.; Seto, T.; Twakenoyama, M.; Ichinose, Y. c-kit mutation-positive advanced thymic carcinoma successfully treated as a mediastinal gastrointestinal stromal tumor: A case report. Mol. Clin. Oncol. 2016, 4, 527–529. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hagemann, I.S.; Govindan, R.; Javidan-Nejad, C.; Pfeifer, J.D.; Cottrell, C.E. Stabilization of disease after targeted therapy in a thymic carcinoma with KIT mutation detected by clinical next-generation sequencing. J. Thorac. Oncol. 2014, 9, e12–e16. [Google Scholar] [PubMed]
- Imbimbo, M.; Vitali, M.; Fabbri, A.; Ottaviano, M.; Pasello, G.; Petrini, I.; Palmieri, G.; Berardi, R.; Zucali, P.; Ganzinelli, M.; et al. RELEVENT Trial: Phase II Trial of Ramucirumab, Carboplatin, and Paclitaxel in Previously Untreated Thymic Carcinoma/B3 Thymoma with Area of Carcinoma. Clin. Lung Cancer 2018, 19, e811–e814. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).