Abstract
The aim of this retrospective exploratory study was to investigate the prevalence of unfavorable findings during video-urodynamic studies (VUDS) in patients with minimally conscious state (MCS)/unresponsive wakefulness syndrome (UWS) and whether management of the lower urinary tract (LUT) was adjusted accordingly. A retrospective chart review was conducted to screen for patients diagnosed with MCS/UWS at our rehabilitation center between 2011 and 2020. Patients 18 years or older were included and underwent baseline VUDS after being diagnosed with MCS/UWS. We analyzed urodynamic parameters and subsequent changes in LUT management in this cohort. In total, 32 patients (7 females, 25 males, median age 37 years) with MCS/UWS were included for analysis. While at least one unfavorable VUDS finding (i.e., neurogenic detrusor overactivity [NDO], detrusor sphincter dyssynergia {DSD, high maximum detrusor pressure during storage phase [>40 cmH2O], low-compliance bladder [<20 mL/cmH2O], and vesico–uretero–renal reflux [VUR]) was found in each patient, NDO (78.1%, 25/32) and DSD (68.8%, 22/32) were the two most frequent unfavorable VUDS findings. Following baseline VUDS, new LUT treatment options were established in 56.3% (18/32) of all patients. In addition, bladder-emptying methods were changed in 46.9% (15/32) of all patients, resulting in fewer patients relying on indwelling catheters. Our retrospective exploratory study revealed a high prevalence of NDO and DSD in patients with MCS/UWS, illustrating the importance of VUDS to adapt LUT management in this cohort accordingly.
1. Introduction
Initially coined as a “vegetative state” (VS) by Jennet and Plum in 1972 [1], the unresponsive wakefulness syndrome (UWS), as proposed by the European Task Force on Disorders of Consciousness in 2010, “refers to patients showing several clinical signs (hence syndrome) of unresponsiveness (that is, without response to commands) in the presence of wakefulness (that is, eye opening)” [2]. While the American Academy of Neurology [3] refers to this disorder of consciousness as VS/UWS, the Royal College of Physicians [4] decided to remain with the term VS. In contrast, patients with a minimally conscious state (MCS) are characterized by “the presence of minimal but clearly discernible behavioral evidence of self or environmental awareness”. Further, MCS was defined by the Aspen Neurobehavioral Conference Workgroup in 2002 as “a condition of severely altered consciousness in which minimal but clearly discernible behavioral evidence of self or environmental awareness is demonstrated” [3]. While UWS and MCS are both hallmarked by severe impairments of consciousness and associated with a substantial social and economic burden [5], MCS differs considerably from VS/UWS, which is supported by recent evidence from neurobehavioral and neuroimaging studies [6]. However, beneficial evidence of neurological rehabilitation in MCS and VS/UWS has been demonstrated previously [7,8,9]. Furthermore, early rehabilitation has been reported to be associated with better outcomes in severely traumatic brain-injured patients [10,11].
However, little is known about the impact of MCS and VS/UWS on a patient’s lower urinary tract (LUT) function, in particular on urine storage and voiding. To our knowledge, only a few studies have conducted urodynamic studies (UDS) to assess LUT function in patients with VS or MCS/UWS [12,13,14,15,16]. In line with other neurological disorders, LUT function should be assessed and aided appropriately to protect or at least minimize the risk of structural and functional changes of the upper urinary tract (UUT) and LUT in the future [17]. Well-known complications and long-term consequences include recurrent urinary tract infection, stone formation (urinary bladder and kidney), and vesico–uretero–renal reflux (VUR), as well as renal insufficiency and renal failure [18].
Although patients with MCS/UWS can be assessed clinically, verbal assessments are mostly impossible given the patients’ impaired cognitive function. Complaints such as “I am wetting” or “I feel a burning sensation” cannot be expected from any individual in this cohort. However, objective assessments, such as sonography, cystoscopy, and UDS, can be performed to evaluate LUT and UUT. However, UDS is the only method to objectively assess LUT function (level of evidence [LE] 2a) [19], and Video-UDS (VUDS) is considered the preferred option to conduct UDS in individuals with underlying neurological disorders (LE 4) [19]. However, VUDS are not routinely performed in clinical practice.
Owing to the lack of evidence, we aimed to identify unfavorable VUDS findings in patients with MCS/UWS and whether clinical treatment was adjusted accordingly to improve LUT function in this cohort.
2. Materials and Methods
2.1. Population
Patients were eligible when meeting the following inclusion criteria: aged 18 years or older, diagnosis of MCS or UWS at our rehabilitation center between 2011 and 2020, and documented baseline VUDS for assessment of LUT function. No limits were set in terms of the time point of baseline VUDS following MCS/UWS diagnosis. Further, patients must not have had any spinal fractures or spinal cord injury (SCI), which were accounted for using computed tomography or magnetic resonance imaging.
2.2. Objectives
The primary outcome was to obtain the prevalence of unfavorable urodynamic findings [20] (i.e., high maximum detrusor pressure [Pdet] during the storage phase [>40 cmH2O], low-compliance bladder [<20 mL/cmH2O], neurogenic detrusor overactivity (NDO), detrusor sphincter dyssynergia (DSD), and VUR) during VUDS in patients with MCS/UWS. The secondary outcome was to compare patients who underwent VUDS “Early” (i.e., ≤12 months) versus “Late” (i.e., ≥13 months) after diagnosis and whether LUT management was changed with respect to VUDS findings.
2.3. Outcome Variables
We chose the following categorical outcome variables: sex (female and male), type of underlying brain injury resulting in MCS/UWS (traumatic, hypoxic, or hemorrhagic), presence of NDO (yes or no), presence of Pdetmax during storage ([>40 cmH2O], yes or no), presence of low-compliance bladder during filling cystometry ([<20 mL/cmH2O] yes or no), presence of DSD (yes or no), presence of VUR (yes or no), method of bladder emptying (indwelling catheter (transurethral/suprapubic), condom catheter, intermittent catheterization (i.e., self or assisted), or volitional voiding), and specific pharmaceutical LUT therapies, i.e., antimuscarinics (yes or no), alpha-blockers (yes or no), mirabegron (yes or no), intrasphincteric botulinum toxin-A injections (yes or no), intradetrusor botulinum toxin-A injections (yes or no).
We chose the following continuous outcome variables: age [years], time from diagnosis to VUDS (months), neurological/cognitive function prior to VUDS, i.e., coma recovery scale—revised (CRS-R) [21], Glasgow Coma Scale (GCS) [22], and/or Bavesta Score [23], VUDS parameters, i.e., maximum cystometric capacity (MCC, [mL]), Pdetmax during storage [cmH2O], bladder compliance during filling cystometry [mL/cmH2O], Pdetmax during voiding [cmH2O], and detrusor overactivity leak point pressure (DOLPP, [cmH2O]), detrusor overactivity leak volume (DOLV, [mL]), and post-void residual (PVR) urine [mL].
2.4. Data Source and Collection
Data were collected between 2011 and 2020 (Figure 1). We retrospectively obtained data from medical charts (i.e., categorical and continuous outcome variables). VUDS were conducted using the urodynamic system (Uromic Quickstep, Medkonsult Medical Technology, MMT; Olomouc, Czech Republic) in combination with the fluoroscopy system (Artis zee multi-purpose, Siemens Healthineers International AG; Erlangen, Germany). All urodynamic assessments were performed in accordance with the International Continence Society’s “Good Urodynamic Practices” [24] to evaluate LUT function and quantify the current extent of neurogenic lower urinary tract dysfunction (NLUTD).
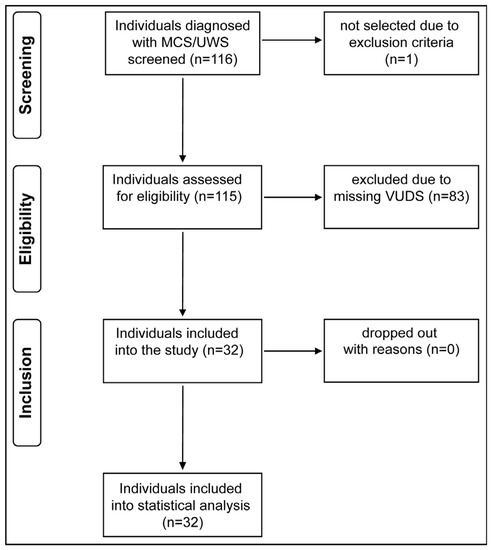
Figure 1.
Study flow diagram. From a pool of 116 screened individuals, 32 fulfilled all inclusion criteria and were included in our analysis. MCS/UWS = minimally conscious state/unresponsive wakefulness syndrome, VUDS = video-urodynamic studies.
A semi-quantitative assessment of the level of consciousness was performed using three different scales to establish diagnosis and monitor behavioral recovery. Thus, CRS-R [21] and GCS [22] are two internationally well-established scores to assess the level of consciousness. The BAVESTA score [23] is an interprofessional observation tool to measure convalescence in MCS/UWS, which uses relevant functions of patients suffering from severe consciousness impairment, such as gazing, muscle tone, and hygiene. Furthermore, it has been validated and is commonly used in our institution for follow-up in MCS/UWS [25].
2.5. Bias
We encountered two kinds of bias: (1) selection bias—all patients were retrieved retrospectively during a pre-defined 10-year period, and (2) temporal bias—the time point of VUDS across all patients was not standardized and, therefore, comprised a significant variance.
2.6. Statistical Methods
Statistical analyses were conducted using R (Version 4.0.5 for Mac Os). Non-parametric statistics were applied. A Wilcoxon signed rank test (i.e., age, time from diagnosis to VUDS, CRS-R, GCS, Bavesta score, Pdetmax storage, DOLPP, DOLV, Compliance, MCC and PVR), Fisher exact test (i.e., sex, NDO, Pdetmax storage > 40 cmH2O, low-compliance bladder and DSD), and Kruskal–Wallis Test (i.e., type of brain injury) were used to compare between “Early” and “Late” VUDS. Data are presented as raw values and percentages, medians with lower (Q1) and upper quartiles (Q3), as well as minimum and maximum, wherever indicated. The threshold for a statistically significant difference was p < 0.05.
3. Results
Overall, 32 patients (7 females, 25 males, median age 37 years, median time from diagnosis to VUDS 4 months) diagnosed with MCS/UWS were included in this study. Demographics and injury characteristics are highlighted in Table 1.

Table 1.
Patient demographics and injury characteristics.
Prior to baseline VUDS, the minority of patients had at least one pharmaceutical LUT treatment (21.9%) and utilized one method of assisted urinary bladder emptying (78.1%) (see Table 2). Overall, pharmaceutical LUT treatment changed as a direct consequence of the VUDS in 56.3% (18/32) of patients (see Table 2).

Table 2.
Lower urinary tract management before and after baseline urodynamic studies.
Furthermore, 46.9% (15/32) of all patients had experienced at least one LUT complication since admission, comprising UTI/CA-UTI (defined as the presence of symptoms or signs compatible with UTI with no other identified source of infection along with 103 colony-forming units) [26] in 31.3% (10/32) and catheter occlusion in 21.9% (7/32). While all patients demonstrated at least one unfavorable urodynamic parameter, NDO and DSD were the two most frequent (see Table 3).

Table 3.
Video-urodynamic parameters and distribution of unfavorable findings.
4. Discussion
4.1. Main Findings
Our retrospective exploratory study revealed novel findings highlighting the extent of NLUTD in patients with MCS/UWS. We found a high prevalence in three of the five unfavorable urodynamic findings (i.e., NDO, Pdetmax during storage > 40 cmH2O, and DSD), which led to subsequent therapeutic changes in our cohort of 32 patients with MCS/UWS.
4.2. Findings of Unfavorable Urodynamic Parameters
At least one unfavorable urodynamic finding was detected in each patient of our cohort—most commonly NDO and DSD. A similar high prevalence of these two unfavorable urodynamic parameters has been recently reported in individuals with SCI [27].
NDO is highly prevalent (i.e., up to 95%) in individuals with suprasacral SCI after the initial spinal shock phase [28] and to a lesser degree in individuals with multiple sclerosis (i.e., ~43%) [29] or myelomeningocele (i.e., ~44%) [30]. In our cohort, NDO was detected in 78% of all patients, which is in line with Benecchi et al. [14], who reported a prevalence of 79% in a cohort of 20 patients with VS. In contrast, both Girando et al. [13] (i.e., 63%, 10/16) and Windaele [16] (i.e., 33%, 5/15) reported a lower prevalence of NDO in patients with MCS/UWS. In our cohort, 85% (21/25) of all patients with NDO had a Pdetmax during storage > 40 cmH2O.
DSD is highly prevalent (i.e., >70%) in individuals with suprasacral SCI after the initial spinal shock phase [31] and to a lesser degree in individuals with multiple sclerosis (i.e., ~36%) [29] or myelomeningocele (i.e., ~56%) [30]. In our cohort, the prevalence of DSD was close to 70%. This finding contrasts the reports from Krimchansky et al. [12], who could not find any DSD in a cohort of 17 patients in a post-traumatic VS, as well as from Windaele [16], who reported only one of fifteen patients with DSD, in whom a spinal cord lesion was discovered in the course of the disease. Thus, none of the patients with VS/coma due to brain injury presented with DSD. Further, neither Benecchi et al. [14] nor Girando et al. [13] reported on the presence of DSD. Considering that DSD, defined as a detrusor contraction concurrent with an involuntary contraction of the urethral and/or periurethral striated muscle [32], is a consequence of disruption of central nervous system regulation of the micturition reflex and is usually seen in patients with SCI. In our cohort, neuro-imaging was used to screen for SCI. However, none of the 32 patients presented a SCI, which raises the question of what the underlying mechanism for DSD in our cohort might be.
While none of the other studies reported on the presence of VUR, in our cohort, fortunately, only one patient had a VUR (i.e., grade 1). While VUR can be a complication after a prolonged period of high intravesical pressure in individuals with NLUTD [33], recent studies reported a similar low prevalence of VUR in individuals with acute SCI (i.e., within the first year after injury) [27,34]. Given the paucity of longitudinal urodynamic data in patients with MCS/UWS, at the present time, one can only refer to other entities of NLUTD, such as SCI and MS, regarding the risk of developing VUR long-term. While Sirasaporn and Saengsuwan [35] reported an overall low incidence (i.e., 7.5 cases per 100 person-years) in a historical study comprising individuals with chronic SCI, Piquet et al. [36] also reported a low prevalence (i.e., 5.5%) in individuals with MS.
While the prevalence of a low-compliance bladder was approximately one in six in patients with SCI (i.e., 17%) [37], we found that one in four patients in our cohort had a low-compliance bladder. This surprising finding emphasizes the need for conducting VUDS early in patients with MCS/UWS, as half the patients in the “Late VUDS” group presented with a compliance of less than 20 cmH2O compared to one in five patients in the “Early VUDS” group.
4.3. LUT Management before and after VUDS
Prior to conducting VUDS, most patients (i.e., >75%) utilized assisted urinary bladder emptying by means of indwelling catheterization. To avoid catheter-associated complications such as CA-UTI, one should refrain from using indwelling catheters to manage urinary incontinence whenever possible [26].
VUDS can help to identify potential patients eligible to switch bladder emptying methods, i.e., condom catheter or intermittent catheterization, as alternatives to indwelling catheters. A variety of indwelling catheter-associated complications, such as CA-UTI, urethral trauma, urethritis, fistula, bladder neck incompetence, sphincter erosion, bladder stones, bladder cancer, and allergies, have been described extensively [38]. We recorded complications in about half of our cohort (i.e., 47%), mostly associated with indwelling catheterization, of which a third were UTIs/CAUTIs [26]. Although no data from randomized controlled trials on bladder emptying methods in patients with MCS/UWS are available, previous studies showed the benefit of intermittent catheterization or urinal condoms vs. indwelling catheters in patients with NLUTD [39].
Prior to VUDS, more than one in five patients received voiding-altering medications. There is evidence that routine administration of anticholinergic drugs may benefit patients with MCS/UWS. Krimanchsky et al. showed that while decreasing detrusor pressure and increasing bladder capacity with an anticholinergic agent, external signs of pain on emptying disappeared [12]. Considering our high number of NDOs with considerably high intravesical pressure, routine application of anticholinergic agents could be considered.
Following VUDS, LUT management with respect to bladder emptying methods was changed in half of our cohort. In patients with sufficient reflex voiding, condom catheters were used to successfully eliminate indwelling catheters and avoid urinary incontinence. Intermittent catheterization was not performed in our cohort and appears to be not a practical option in MCS/UWS patients due to the limited level of consciousness.
On account of our urodynamic findings, we commenced pharmacological treatment in 75% of patients to aid their NLUTD, mainly antimuscarinics and alpha-blockers. We recommended intradetrusor botulinum toxin-A injections in two cases to lower elevated detrusor pressure and intrasphincteric botulinum toxin-A injections in six patients to reduce subvesical resistance and allow condom catheters to collect urine without endangering the UUT, respectively. With respect to the aforementioned literature in patients with MCS/UWS [12,13,14,15,16], only Girando et al. [13] reported on the effect of pharmacological treatment on urodynamic parameters (i.e., before and after continuous intrathecal baclofen, ITB). While MCC and PVR urine were increased after ITB, there was a smaller number of patients with NDO and a lower detrusor leak point pressure [13].
The best time point for using VUDS to investigate LUT function in patients with MCS/UWS has yet to be determined. Jiang et al. [40] showed in an experimental trial in rats that there was a transient reduction in bladder contractility immediately after traumatic brain injury (TBI). However, one month after TBI, they showed an increase in overactive bladder or urinary urge incontinence [40]. This suggests that urodynamic evaluation/VUDS should not only be performed during the acute phase of TBI but repeated 4–6 weeks after TBI to surpass the initial shock phase in order to avoid relying on false-negative findings from the very first assessment after an acquired brain injury [40]. Furthermore, regular follow-up urodynamic evaluations/VUDS should be conducted to monitor LUT function, detect its decline, and adapt LUT management accordingly in MCS/UWS patients, as recommended in other patient groups with NLUTD [41].
4.4. Limitations
The retrospective nature of our study and the lack of performing VUDS in all MCS/UWS patients systematically at our institute led to a selection bias. In the past, whether to perform VUDS and when was not predetermined. A number of VUDS have been performed because of underlying urological complications, leading to a selection bias, i.e., patients with apparent LUT dysfunction. This might contribute to the high prevalence in three of the five unfavorable urodynamic parameters [20].
Furthermore, after adjusting LUT management, we did not perform follow-up VUDS systematically. Thus, we cannot report on the long-term efficacy of the adjusted LUT management. Despite the recommendation of long-term UDS surveillance in other entities with NLUTD [19], the value of follow-up VUDS in patients with UWS/MCS still needs to be determined. To answer this, our institute has started to systematically perform UDS at the following time points: (1) between 2 and 6 months, (2) at 12 months, and (3) at 24 months following a brain injury/diagnosis of MCS/UWS. Depending on the extent of NLUTD, additional VUDS will be performed individually to monitor LUT function as well as treatment efficacy.
5. Conclusions
This retrospective cohort study revealed a high prevalence of unfavorable urodynamic findings in patients with MCS/UWS, which was very similar to individuals with SCI. These findings illustrate the importance of early VUDS in order to adapt LUT management in this cohort accordingly. Considering the paucity of evidence in this cohort, further research is needed to track how urinary tract function is developing long-term and whether additional unfavorable urodynamic parameters will arise in the course of the disease while patients are aging. Lastly, given the aforementioned, clinicians taking care of patients with MCS/UWS need to strive for the implementation of follow-up strategies rigorously to minimize the risk of deterioration of the entire urinary tract function long-term.
Author Contributions
Conceptualization, methods, and investigation, F.L., C.E., L.Z., C.A.R., H.S., R.A., M.H.-G., S.M. and M.W.; formal analysis, F.L., S.M. and M.W.; writing—original draft preparation, F.L., S.M. and M.W.; writing—review and editing, F.L., C.E., L.Z., C.A.R., H.S., R.A., M.H.-G., S.M. and M.W.; visualization, F.L. and M.W.; supervision, F.L., S.M. and M.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This retrospective cohort study was conducted in accordance with the Declaration of Helsinki and approved by our local ethics committee (Ethikkommission Nordwest- und Zentralschweiz, EKNZ: 2019–02394). Further, this study is reported in accordance with the STROBE statement guideline for cohort studies.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of this study. Further, given the provided information, patients cannot be identified (including by the patients themselves).
Data Availability Statement
De-identified data are available upon request from the authors.
Conflicts of Interest
M.W. receives or has received research support from the Rick Hansen Institute and Foundation, the Michael Smith Foundation for Health Research, Pfizer Canada, Coloplast, and Wellspect. M.W. serves on the advisory board for Coloplast. All other authors declare no conflicts of interest.
References
- Jennett, B.; Plum, F. Persistent vegetative state after brain damage: A syndrome in search of a name. Lancet 1972, 299, 734–737. [Google Scholar] [CrossRef]
- Laureys, S.; Celesia, G.G.; Cohadon, F.; Lavrijsen, J.; León-Carrión, J.; Sannita, W.G.; Sazbon, L.; Schmutzhard, E.; Von Wild, K.R.; Zeman, A.; et al. Unresponsive wakefulness syndrome: A new name for the vegetative state or apallic syndrome. BMC Med. 2010, 8, 68. [Google Scholar] [CrossRef]
- Giacino, J.T.; Katz, D.I.; Schiff, N.D.; Whyte, J.; Ashman, E.J.; Ashwal, S.; Barbano, R.; Hammond, F.M.; Laureys, S.; Ling, G.S.F.; et al. Practice guideline update recommendations summary: Disorders of consciousness. Neurology 2018, 91, 450–460. [Google Scholar] [CrossRef]
- College of Physicians, R. Prolonged Disorders of Consciousness Following Sudden Onset Brain Injury: National Clinical Guidelines. 2009. Available online: https://www.rcplondon.ac.uk/guidelines-policy/prolonged-disorders-consciousness-following-sudden-onset-brain-injury-national-clinical-guidelines (accessed on 27 July 2023).
- Magnani, F.G.; Leonardi, M.; Sattin, D. Caregivers of people with disorders of consciousness: Which burden predictors? Neurol. Sci. 2020, 41, 2773–2779. [Google Scholar] [CrossRef] [PubMed]
- Naro, A.; Calabrò, R.S. Towards New Diagnostic Approaches in Disorders of Consciousness: A Proof of Concept Study on the Promising Use of Imagery Visuomotor Task. Brain Sci. 2020, 10, 746. [Google Scholar] [CrossRef] [PubMed]
- Berrol, S. Evolution and the persistent vegetative state. J. Head Trauma Rehabil. 1986, 1, 7–13. [Google Scholar] [CrossRef]
- Levin, H.S.; Saydjari, C.; Eisenberg, H.M.; Foulkes, M.; Marshall, L.F.; Ruff, R.M.; Jane, J.A.; Marmarou, A. Vegetative State after Closed-Head Injury. A Traumatic Coma Data Bank Report. Arch. Neurol. 1991, 48, 580–585. [Google Scholar] [CrossRef]
- Wales, L.R.; Bernhardt, J.A. A case for slow to recover rehabilitation services following severe acquired brain injury. Aust. J. Physiother. 2000, 46, 143–146. [Google Scholar] [CrossRef]
- Cope, D.N.; Hall, K. Head injury rehabilitation: Benefit of early intervention. Arch. Phys. Med. Rehabil. 1982, 63, 433–437. [Google Scholar]
- Mackay, L.E.; Bernstein, B.A.; Chapman, P.E.; Morgan, A.S.; Milazzo, L.S. Early intervention in severe head injury: Long-term benefits of a formalized program. Arch. Phys. Med. Rehabil. 1992, 73, 635–641. [Google Scholar]
- Krimchansky, B.-Z.; Sazbon, L.; Heller, L.; Kosteff, H.; ZLuttwak, Z. Bladder tone in patients in post-traumatic vegetative state. Brain Inj. 1999, 13, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Giraudo, D.; Gozzerino, F.; Lamberti, G.; Antoniono, E.; Califano, C.; Filocamo, M.; Massazza, G. Urodynamic evaluation before and after continuous intrathecal Baclofen infusion (ITB) in patients unresponsive wakefulness syndrome and minimally conscious state. Pelviperineology 2017, 36, 60–62. [Google Scholar]
- Benecchi, L.; Caldera, G.; Cavestro, C.; Bordinazzo, R.; Privitera, O.; Feller, S. Post-comatose cystomanometric peculiarities. Urol. J. 1996, 63 (Suppl. S1), 99–104. [Google Scholar] [CrossRef]
- Nardulli, R.; Monitillo, V.; Losavio, E.; Megna, M.; Megna, G. Urodynamic evaluation in the apallic syndrome. Urodinamica 1992, 2, 180–182. [Google Scholar]
- Wyndaele, J.J. Urodynamics in comatose patients. Neurourol. Urodyn. 1990, 9, 43–52. [Google Scholar] [CrossRef]
- Satar, N.; Bauer, S.B.; Shefner, J.; Kelly, M.D.; Darbey, M.M. The Effects of Delayed Diagnosis and Treatment in Patients with an Occult Spinal Dysraphism. J. Urol. 1995, 154 2 Pt 2, 754–758. [Google Scholar] [CrossRef]
- Çetinel, B.; Önal, B.; Can, G.; Talat, Z.; Erhan, B.; Gündüz, B. Risk factors predicting upper urinary tract deterioration in patients with spinal cord injury: A retrospective study. Neurourol. Urodyn. 2017, 36, 653–658. [Google Scholar] [CrossRef]
- Blok, B.; Castro-Diaz, D.; Del Popolo, G.; Groen, J.; Hamid, R.; Karsenty, G.; Kessler, T.M.; Pannek, J. EAU Guidelines on Neuro-Urology. 2023. Available online: https://uroweb.org/guidelines/neuro-urology (accessed on 27 July 2023).
- Welk, B.; Schneider, M.P.; Thavaseelan, J.; Traini, L.R.; Curt, A.; Kessler, T.M. Early urological care of patients with spinal cord injury. World J. Urol. 2018, 36, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Giacino, J.T.; Kalmar, K.; Whyte, J. The JFK Coma Recovery Scale-Revised: Measurement characteristics and diagnostic utility. Arch. Phys. Med. Rehabil. 2004, 85, 2020–2029. [Google Scholar] [CrossRef]
- Teasdale, G.; Jennett, B. Assessment of coma and impaired consciousness: A practical scale. Lancet 1974, 2, 81–84. [Google Scholar] [CrossRef]
- Huber, M.; Koch, S.; Hund-Georgiadis, M.; Mäder, M.; Borgwardt, S.; Stieglitz, R.-D. Diagnostic Validity of the Basler Vegetative State Assessment-BAVESTA/Diagnostische Validität des Basler Vegetative State Assessments-BAVESTA. Int. J. Health Prof. 2014, 1, 50–60. [Google Scholar] [CrossRef]
- Schäfer, W.; Abrams, P.; Liao, L.; Mattiasson, A.; Pesce, F.; Spangberg, A.; Sterling, A.M.; Zinner, N.R.; van Kerrebroeck, P. Good urodynamic practices: Uroflowmetry, filling cystometry, and pressure-flow studies. Neurourol. Urodyn. 2002, 21, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; Koch, S.; Mäder, M.; Hund-Georgiadis, M.; Borgwardt, S.; Stieglitz, R.D. Verlaufsbeobachtung von Wachkomapatienten-Validierung des Basler Vegetative State Assessments (BAVESTA). Neurol. Rehabil. 2014, 20, 123–132. [Google Scholar]
- Hooton, T.M.; Bradley, S.F.; Cardenas, D.D.; Colgan, R.; Geerlings, S.E.; Rice, J.C.; Saint, S.; Schaeffer, A.J.; Tambayh, P.A.; Tenke, P.; et al. Diagnosis, Prevention, and Treatment of Catheter-Associated Urinary Tract Infection in Adults: 2009 International Clinical Practice Guidelines from the Infectious Diseases Society of America. Clin. Infect. Dis. 2010, 50, 625–663. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, M.; Birkhäuser, V.; Anderson, C.E.; Bywater, M.; Gross, O.; Kiss, S.; Knüpfer, S.C.; Koschorke, M.; Leitner, L.; Mehnert, U.; et al. Neurogenic Lower Urinary Tract Dysfunction in the First Year After Spinal Cord Injury: A Descriptive Study of Urodynamic Findings. J. Urol. 2023, 209, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Weld, K.J.; Dmochowski, R.R. Association of level of injury and bladder behavior in patients with post-traumatic spinal cord injury. Urology 2000, 55, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Al Dandan, H.B.; Coote, S.; McClurg, D. Prevalence of Lower Urinary Tract Symptoms in People with Multiple Sclerosis: A systematic review and meta-analysis. Int. J. MS Care 2020, 22, 91–99. [Google Scholar] [CrossRef]
- Yu, W.-R.; Kuo, H.-C. Usefulness of videourodynamic study in the decision-making of surgical intervention and bladder management for neurogenic lower urinary tract dysfunction among patients with myelomeningocele. Int. Urol. Nephrol. 2022, 54, 1815–1824. [Google Scholar] [CrossRef]
- Blaivas, J.G.; Sinha, H.P.; Zayed, A.A.H.; Labib, K.B. Detrusor-external Sphincter Dyssynergia: A Detailed Electromyographic Study. J. Urol. 1981, 125, 545–548. [Google Scholar] [CrossRef]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; Van Kerrebroeck, P.; Victor, A.; Wein, A.; Standardisation Sub-committee of the International Continence Society. The standardisation of terminology of lower urinary tract function: Report from the standardisation sub-committee of the International Continence Society. Neurourol. Urodyn. 2002, 21, 167–178. [Google Scholar]
- Gormley, E.A. Urologic Complications of the Neurogenic Bladder. Urol. Clin. N. Am. 2010, 37, 601–607. [Google Scholar] [CrossRef]
- Bywater, M.; Tornic, J.; Mehnert, U.; Kessler, T.M. Detrusor Acontractility after Acute Spinal Cord Injury—Myth or Reality? J. Urol. 2018, 199, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Sirasaporn, P.; Saengsuwan, J. Incidence and predictive factors for developing vesicouretericreflux in individuals with suprasarcral spinal cord injury: A historical cohort study. Spinal Cord 2021, 59, 753–760. [Google Scholar]
- Piquet, V.; Turmel, N.; Chesnel, C.; Haddad, R.; Lebreton, F.; Amarenco, G.; Hentzen, C. Upper urinary tract function of patients with multiple sclerosis. Neurourol. Urodyn. 2022, 41, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Hackler, R.H.; Hall, M.K.; Zampieri, T.A. Bladder Hypocompliance in the Spinal Cord Injury Population. J. Urol. 1989, 141, 1390–1393. [Google Scholar] [CrossRef]
- Igawa, Y.; Wyndaele, J.-J.; Nishizawa, O. Catheterization: Possible complications and their prevention and treatment. Int. J. Urol. 2008, 15, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Jamison, J.; Maguire, S.; McCann, J. Catheter policies for management of long term voiding problems in adults with neurogenic bladder disorders. Cochrane Database Syst. Rev. 2013, 11, CD004375. [Google Scholar] [CrossRef]
- Jiang, H.-H.; Kokiko-Cochran, O.N.; Li, K.; Balog, B.; Lin, C.-Y.; Damaser, M.S.; Lin, V.; Cheng, J.Y.; Lee, Y.-S. Bladder dysfunction changes from underactive to overactive after experimental traumatic brain injury. Exp. Neurol. 2013, 240, 57–63. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kavanagh, A.; Akhavizadegan, H.; Walter, M.; Stothers, L.; Welk, B.; Boone, T.B. Surveillance urodynamics for neurogenic lower urinary tract dysfunction: A systematic review. Can. Urol. Assoc. J. 2019, 13, 133–141. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).