Abstract
In the cardiovascular system, atherogenic low-density lipoproteins (LDL) and the protective hormone adiponectin bind to the same receptor, T-cadherin. In this study, we tested the hypothesis that the ratio of circulating LDL to high-molecular weight (HMW) adiponectin could predict the development of atherosclerosis. Using enzyme-linked immunosorbent assay, we measured the level of circulating HMW adiponectin in the blood of donors together with ultrasound measuring of intima-media thickness (IMT) of carotid arteries. Single-nucleotide polymorphisms in the T-cadherin gene were identified using polymerase chain reaction. We found that carotid artery IMT is inversely correlated with the level of HMW in male subjects. We also found that the G allele of rs12444338 SNP in the T-cadherin gene correlates with a lower level of circulating T-cadherin and thinner IMT and therefore could be considered as an atheroprotective genotype. Despite our data, we could not provide direct evidence for the initial study hypothesis. However, we did uncover an important correlation between circulating T-cadherin and thinner carotid IMT.
1. Introduction
The causes of the atherosclerotic lesions’ formation and methods for the prevention of atherosclerosis have been studied for more than 100 years. It was demonstrated that the main mechanism of atherogenesis is the accumulation of low-density lipoproteins (LDL) in the vessel wall with the subsequent development of inflammation when increasing the quantity of extracellular matrix in the affected area and the formation of a plaque. LDL influence the vascular cells not only due to the transport of cholesterol into the cells [1] but also due to their ability to induce intracellular signaling (which is necessary for the normal functioning of the cardiovascular system) [2]. LDL in smooth muscle (SMC), endothelial cells, and fibroblasts induce fast (seconds, minutes) hormone-like intracellular signaling: phosphoinositide turnover, increasing the concentration of intracellular calcium ions, and the activation of protein kinase C. This signaling leads to cell proliferation and migration [2,3,4,5,6,7,8,9,10,11,12]. LDL not only triggers the calcium signaling but also enhances the effect of calcium-mobilizing substances: angiotensin II, epinephrine, thrombin, fibroblast growth factor, platelet-derived growth factor, and both insulin-like and epidermal growth factors [6,7,10,11,13,14,15,16,17]. These effects of LDL, as with its accumulation within cells, are also pro-atherogenic.
Our research group identified glycosylphosphatidylinositol(GPI)-anchored T-cadherin protein as a receptor mediating the “hormone-like” effects of LDL [18]. Such intracellular signaling from LDL is induced in vitro at concentrations ten times lower than physiological signaling. This, for a long time, led to the question: is this signaling permanent as a result of the constant occupation of the T-cadherin by LDL?
In 2004, adiponectin, another ligand of T-cadherin with higher affinity, was discovered [19]. Unlike LDL, adiponectin has an anti-atherogenic effect. The concentration of this hormone of adipose tissue decreases in obesity [20]. In early studies, it was found that its concentration also significantly decreases in various cardiovascular diseases, metabolic syndromes (low adiponectin levels are associated with obesity and insulin resistance), and type 2 diabetes [20,21,22,23,24,25,26,27,28]. Moreover, various anti-atherogenic effects of adiponectin on macrophages, smooth muscle and endothelial cells, leukocytes, and platelets have been demonstrated in experiments (see [29] for review). It affects the migration of lymphocytes to atherosclerotic lesions, reduces the level of C-reactive protein, and inhibits TNF-α and NF-κB-mediated pro-inflammatory intracellular signaling [30]. In human studies, it was shown that low adiponectin levels are associated with an increased carotid intima-media thickness (IMT) (a sign of the initial stages of atherosclerosis) [31]. Even though these data are not reproduced in all studies, meta-analysis suggests that a low level of adiponectin is associated with a greater carotid IMT [32]. The most physiologically active circulating form of adiponectin is the high molecular weight (HMW) [33]. The inverse correlations of plasma concentrations with body mass index, cardiovascular diseases, insulin sensitivity, and diabetes mellitus were shown exactly for this form [20,33,34,35,36,37,38,39]. In vitro data suggests that high molecular weight adiponectin causes phosphorylation of adenosine monophosphate-dependent protein kinase (AMPK) and prevents apoptosis of endothelial [37].
It has been proven that T-cadherin, but not the AdipoR1 and AdipoR2 receptors, binds a significant portion of blood adiponectin on the cells’ surfaces in the cardiovascular system and mediates the action of this hormone on them [40,41,42]. In T-cadherin gene knockout mice, adiponectin does not bind to tissues, and its concentration in the blood increases fivefold [40,41,42]. The association between adiponectin and T-cadherin concentrations is also shown in humans; a number of single nucleotide polymorphisms (SNPs) in the T-cadherin gene affecting its expression level correlate with plasma adiponectin concentration [43,44,45,46,47,48,49,50] and cardiovascular diseases [43,47,51,52,53].
In Ranscht’s laboratory, it was shown that the knockout of the T-cadherin gene in mice leads to the abolition of the protective effect of adiponectin, which is manifested in the development of excessive cardiac hypertrophy and an increase in the myocardial infarction area [40]. Using the same knockout animal model, it was demonstrated that T-cadherin is essential for adiponectin-mediated revascularization [42]. Finally, in 2017, it was shown that the adiponectin-T-cadherin complex protects against neointima proliferation and the formation of atherosclerotic plaques [41]. Thus, it was demonstrated in the mouse model that, in the absence of T-cadherin, the concentration of adiponectin in the blood is high. At the same time, however, atherosclerotic lesions develop. It can be assumed that in cases of the “adiponectin paradox” (high mortality associated with a high concentration of adiponectin in the blood (data summarized in [54]), adiponectin does not have a protective effect, since its concentration in tissues is low, despite the high content in the blood plasma. We suggest that such a decrease in the concentration of adiponectin in tissues may occur as a result of the competitive displacement of adiponectin from the T-cadherin receptor by LDL in cases of dyslipidemia. The coefficients of dissociation were measured by different methods for LDL [12,55,56] and for adiponectin [19,57]. Previously, we supposed a possible competition between them [58,59].
The normal plasma concentration of adiponectin is 2–20 µg/mL, which is several orders of magnitude higher than the concentration of many cytokines and hormones [20]. A significant part of adiponectin is normally associated with T-cadherin in tissues [40,41,42,58,59,60].
The concentration of adiponectin in the blood, unlike other adipose-tissue-originating hormones, decreases with obesity (while the LDL level increases). Obesity and metabolic syndrome, known as pro-atherosclerotic conditions, lead to a significant change in the ratio of adiponectin to LDL in the blood, presumably resulting in increasing the probability of LDL binding to T-cadherin, which can trigger adverse effects. According to our new hypothesis about the development of atherosclerosis, for the onset of atherogenesis, it is not the absolute values of LDL or adiponectin in the blood are important but rather the ratio between them.
To test this hypothesis, we checked if the ratio between LDL (or apolipoprotein B (ApoB)) and HMW adiponectin concentrations are associated with the initial stages of atherosclerosis in humans. The measurement of carotid intima-media thickness (IMT) was used to evaluate the presence of early-stage atherosclerosis. Since the concentration of T-cadherin and adiponectin in advanced atherosclerotic lesions is high [41,61] and may distort the HMW adiponectin levels in plasma, all patients with developed atherosclerotic plaques were excluded from the study.
Adiponectin and T-cadherin levels are at least partly genetically determined. That is why we checked not only the influence of the LDL to HMW adiponectin ratio on the IMT but also the role of single nucleotide polymorphisms (SNPs) in the CDH13 gene (encoding T-cadherin) in early atherosclerosis formation. Since some genome-wide association studies demonstrated the link between SNPs in the CDH13 gene and arterial hypertension (AH) [49,53], we also compared patients with AH and with normal blood pressure.
2. Materials and Methods
2.1. Patients and Eligibility Criteria
A cross-sectional observational multicenter cohort study was approved by the Local Ethics Committee of the Medical Scientific and Educational Center, Lomonosov Moscow State University (protocol No. 2/20 dated 16 March 2020). The study was conducted in accordance with the guidelines of the Declaration of Helsinki. All participants gave written informed consent to take part in the study. Male and female patients >30 years old with moderate cardiovascular risk (SCORE 1–4%) were enrolled in the study. Patient information was obtained from medical charts including age, sex, smoking, history of cardiovascular diseases, and therapy. Physical examination with a measurement of blood pressure, heart rate, height, and weight were performed.
In our study, only patients who do not receive statins (to prevent impact on LDL levels) and do not have atherosclerotic plaques (to prevent impact on HMW adiponectin and T-cadherin levels) according to anamnesis and ultrasound data were included. The following exclusion criteria were also applied:
- Total cholesterol level >8 mmol/L (>310 mg/dL) or blood pressure >180/110 mm Hg
- Diabetes
- Established familial hypercholesterolemia
- The level of triglycerides is more than 5.7 mmol/L
- Established systemic inflammatory diseases
- Malignant neoplasm, or another disease (not related to the heart), limiting life expectancy to <three years
- Current abuse of alcohol and/or psychoactive drugs
- Established liver failure (total bilirubin >3 mg/dl) or a history of cirrhosis with signs of portal hypertension
- Pregnancy, lactation
2.2. Ultrasonography
All patients underwent Duplex Doppler ultrasonography of the brachiocephalic arteries and arteries of the lower extremities (Epiq 5, Philips Medical Systems B.V, Netherlands, ultrasound system with linear transducers 12-3 MHz and 18-5 MHz) by qualified researchers. In duplex scanning of the brachiocephalic branches of the aortic arch on the left and right, the following were investigated: the distal part of the brachiocephalic trunk, the proximal (before the divergence of the vertebral arteries) segments of the subclavian arteries, the common carotid arteries throughout, the external carotid arteries in the proximal segments, the internal carotid arteries in the extracranial parts, and the vertebral arteries (segments V1 and V2). During duplex scanning of the arteries of both lower extremities, the following were examined: common femoral, deep and superficial femoral arteries, popliteal artery, lower leg arteries (anterior and posterior tibial arteries, peroneal artery), arteries of the dorsum of the foot. During duplex scanning, the IMT was measured at a typical measurement site in the common carotid artery.
The detection of atherosclerotic plaque in any of the examined arteries was the criterion for excluding the patient from the study.
2.3. Laboratory Tests
All patients underwent laboratory tests. A blood count was performed using a Sysmex XN-1000 automated analyzer, and glycated hemoglobin levels were measured using a DCA Vantage analyzer. All biochemical blood tests, except for the analysis of HMW adiponectin and T-cadherin, were performed using a Beckman Coulter AU480 biochemical analyzer. HMW adiponectin was measured in serum using Human HMW Adiponectin/Acrp30 Quantikine ELISA Kit (DHWAD0, R&D Systems, Minneapolis, MN, USA). T-cadherin was measured in serum using Human Cadherin 13 ELISA Kit (RAB1029, Sigma-Aldrich, Burlington, MA, USA). DNA was isolated from whole venous blood using QIAamp DNA Blood Mini Kit (Qiagen, Venlo, The Netherlands) and QIACube automated station (Qiagen) according to manufacturer’s protocols. CDH13 single nucleotide polymorphisms (SNPs) genotyping was performed using corresponding TaqMan SNP Genotyping Assays (Applied Biosystems, Waltham, MA, USA) and 7500Fast real-time PCR machine (Applied Biosystems) according to manufacturer’s protocols.
2.4. Statistical Analysis
Continuous variables were expressed as median (interquartile range) and analyzed with the Mann–Whitney U test. For multiple comparisons, the Kruskal–Wallis test was used. The Spearman rank correlation coefficient test was used for the correlation of continuous variables. For binary variables, Yates corrected χ2 was used. SNP data were analyzed using the SNPStats online tool (version 0.96, Sole et. al., Barcelona, Spain) [62]. This tool uses logistic regression models and calculates the OR (odds ratio) for each genotype with respect to the reference genotype. According to the number of minor alleles needed in order to modify the risk, five inheritance models are estimated: codominant (every genotype gives a different and non-additive risk), dominant (a single copy of minor allele is enough to modify the risk), recessive (two copies of the minor allele are necessary to change the risk), overdominant (heterozygous are compared to a pool of both allele homozygous), and log-additive (each copy of the minor allele modifies the risk in an additive form). Akaike information criteria (AIC) was used to choose the inheritance model that best fits the data (lower AIC values indicate better models). In all figures, squares show medians; whiskers show upper and lower quartiles.
3. Results
3.1. Baseline Characteristics of the Subjects
Forty-two patients were enrolled in the study; seven of them were excluded after ultrasonography due to the presence of atherosclerotic plaques. The main characteristics of the 35 included patients are shown in Table 1.

Table 1.
Basic data of individuals included in the study.
3.2. Correlations between Laboratory Data and Carotid IMT
We tested correlations between parameters of interest (LDL, ApoB, HMW adiponectin, and T-cadherin levels, HMW adiponectin/LDL and HMW adiponectin/ApoB molar ratios) and carotid IMT and did not find significant correlations. All correlation coefficients are present in Table A1 (Appendix A). Despite age is considered as a cardiovascular risk factor, we did not find any significant correlations between age and IMT or laboratory data.
Since adiponectin levels are dependent on sex, we divided subjects into male and female subgroups. HMW adiponectin was higher in females (15.45 (11.71; 22.62) nM vs. 10.68 (7.81; 13.05) nM, p = 0.014), other differences in laboratory data were non-significant. It is known that adiponectin and particularly HMW adiponectin levels are lower in obesity; we also analyzed correlations in obese and non-obese patients (body mass index (BMI) ≥30 and <30 accordingly). In our dataset, there were no differences in laboratory data between obese and non-obese patients.
In male subjects, we found correlations between carotid IMT and HMW adiponectin. Some correlations between carotid IMT, HMW adiponectin/LDL, and HMW adiponectin/ApoB molar ratios were also present (Table 2).

Table 2.
Significant Spearman correlation coefficients between carotid IMT and laboratory data in male patients.
In female patients, no correlations between carotid IMT and laboratory data were found, but plasma T-cadherin levels were significantly (p < 0.05) correlated with HMW adiponectin levels (ρ = 0.397). The same correlation was found for the HMW adiponectin/ApoB molar ratio (ρ = 0.396).
No significant correlations between carotid IMT and laboratory data were demonstrated in subgroups divided by BMI (obese and non-obese patients).
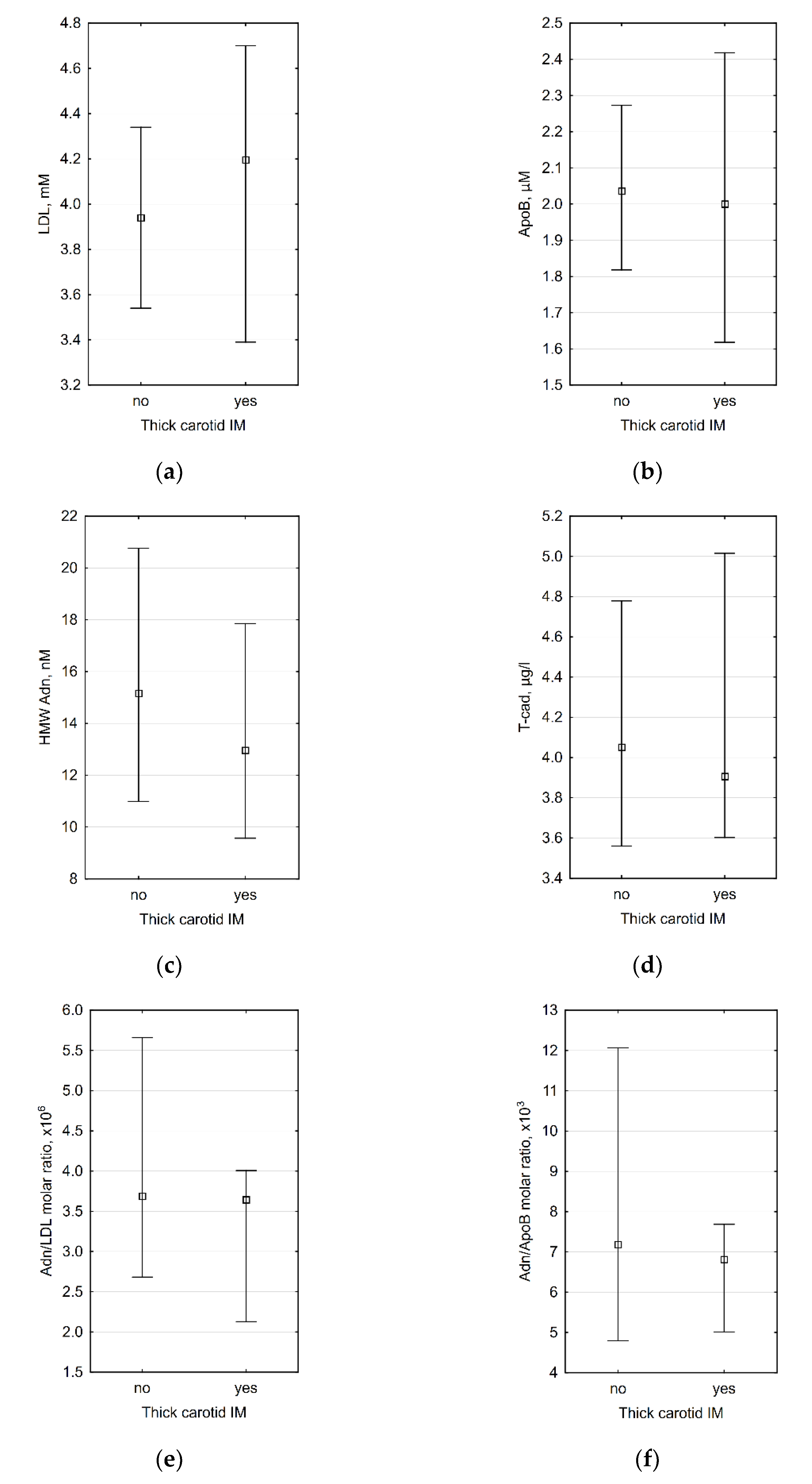
Since carotid IMT >0.9 mm is considered abnormal by the European Society of Cardiology [63,64], we also divided all patients into subgroups with thick (>0.9 mm) and thin (≤0.9 mm) carotid intima-media. The comparison of laboratory data between these subgroups did not reveal any significant differences (Figure 1).
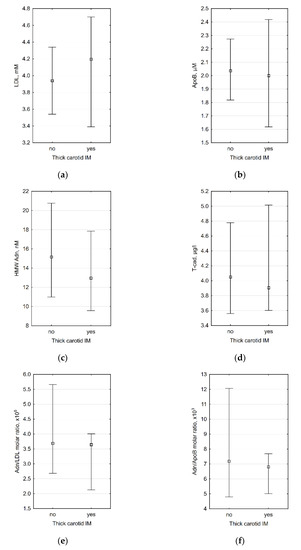
Figure 1.
Association between presence of thick (>0.9 mm) carotid intima-media complex and (a) plasma LDL level (p = 0.661), (b) plasma ApoB level (p = 0.711), (c) plasma HMW adiponectin level (p = 0.469), (d) plasma T-cadherin level (0.853), (e) HMW adiponectin/LDL molar ratio (0.578), and (f) HMW adiponectin/ApoB molar ratio (p = 0.625). n = 35 for all diagrams.
3.3. Association between Laboratory Data and the Presence of Arterial Hypertension (AH)
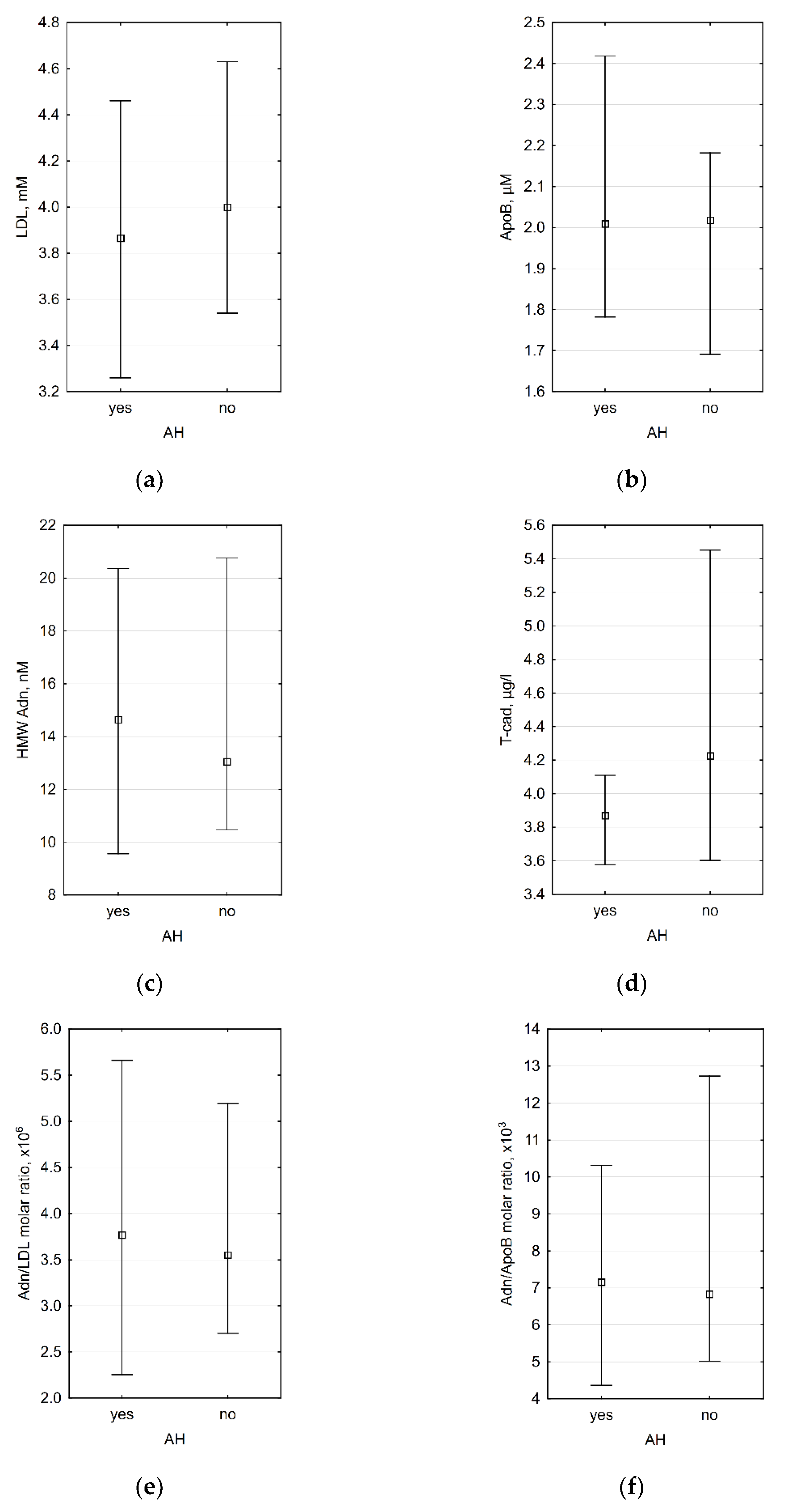
According to genome-wide association studies, genetically determined T-cadherin and adiponectin levels may correlate with blood pressure and the presence of AH [49,53]. We did not discover any significant differences between patients with AH and those with normal blood pressure (Figure 2).
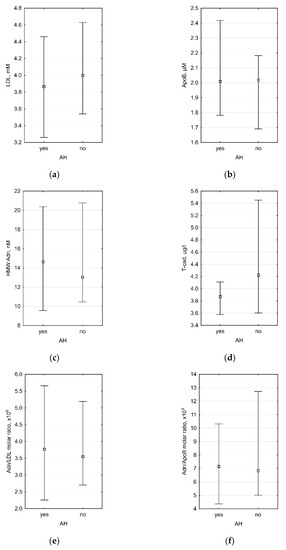
Figure 2.
Association between presence of AH and (a) plasma LDL level (p = 0.609), (b) plasma ApoB level (p = 0.668), (c) plasma HMW adiponectin level (p = 0.779), (d) plasma T-cadherin level (0.181), (e) HMW adiponectin/LDL molar ratio (0.961), and (f) HMW adiponectin/ApoB molar ratio (p = 0.779). n = 35 for all diagrams.
3.4. CDH13 SNP Data
We studied three SNPs in CDH13 (T-cadherin) gene: rs4783244, rs12051272, and rs12444338. According to the literature data, all these SNPs are linked to adiponectin levels [43,46,48,65,66,67], and rs12444338 is also linked to T-cadherin levels [46].
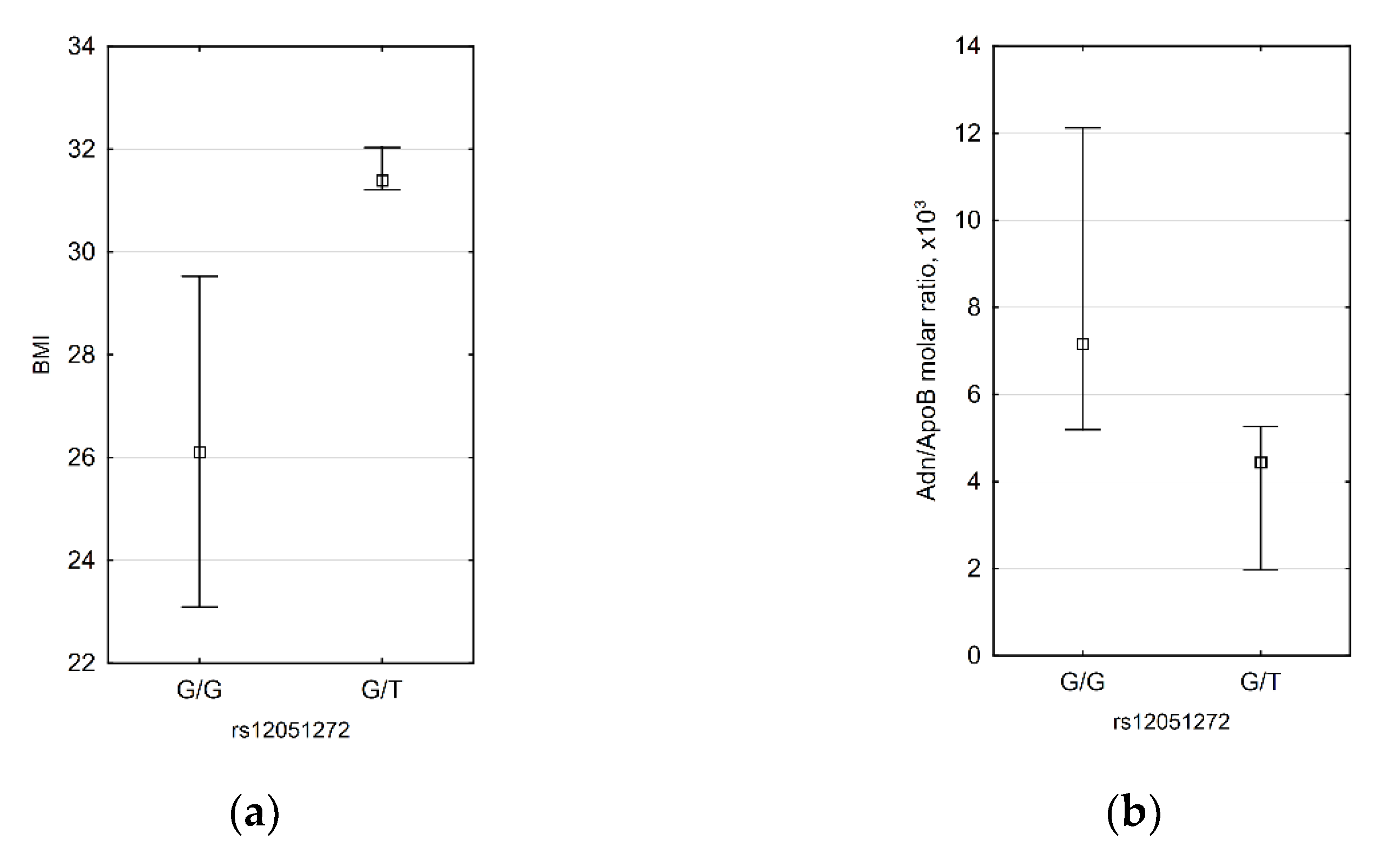
For rs12051272, we demonstrated that subjects with G/T genotype have higher BMI (31.4 (31.2; 32.0) vs. 26.1 (23.1; 29.5), p = 0.0311) and lower HMW adiponectin/ApoB molar ratio (4.44 × 103 (1.97 × 103; 5.26 × 103) vs. 7.15 × 103 (5.19 × 103; 12.12 × 103) (Figure 3). Inverse correlations between BMI and HMW adiponectin/ApoB molar ratio were demonstrated by us for men, but since in male patients there was only one subject with G/T genotype, it is not possible to say if rs12051272 has an effect only in men.
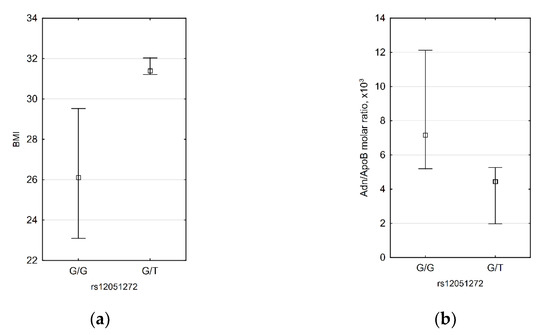
Figure 3.
Significant differences between rs12051272 T allele carriers and non-carriers: subjects with G/T genotype have higher BMI (a) and lower HMW adiponectin/ApoB molar ratio (b). n = 35 for both diagrams.
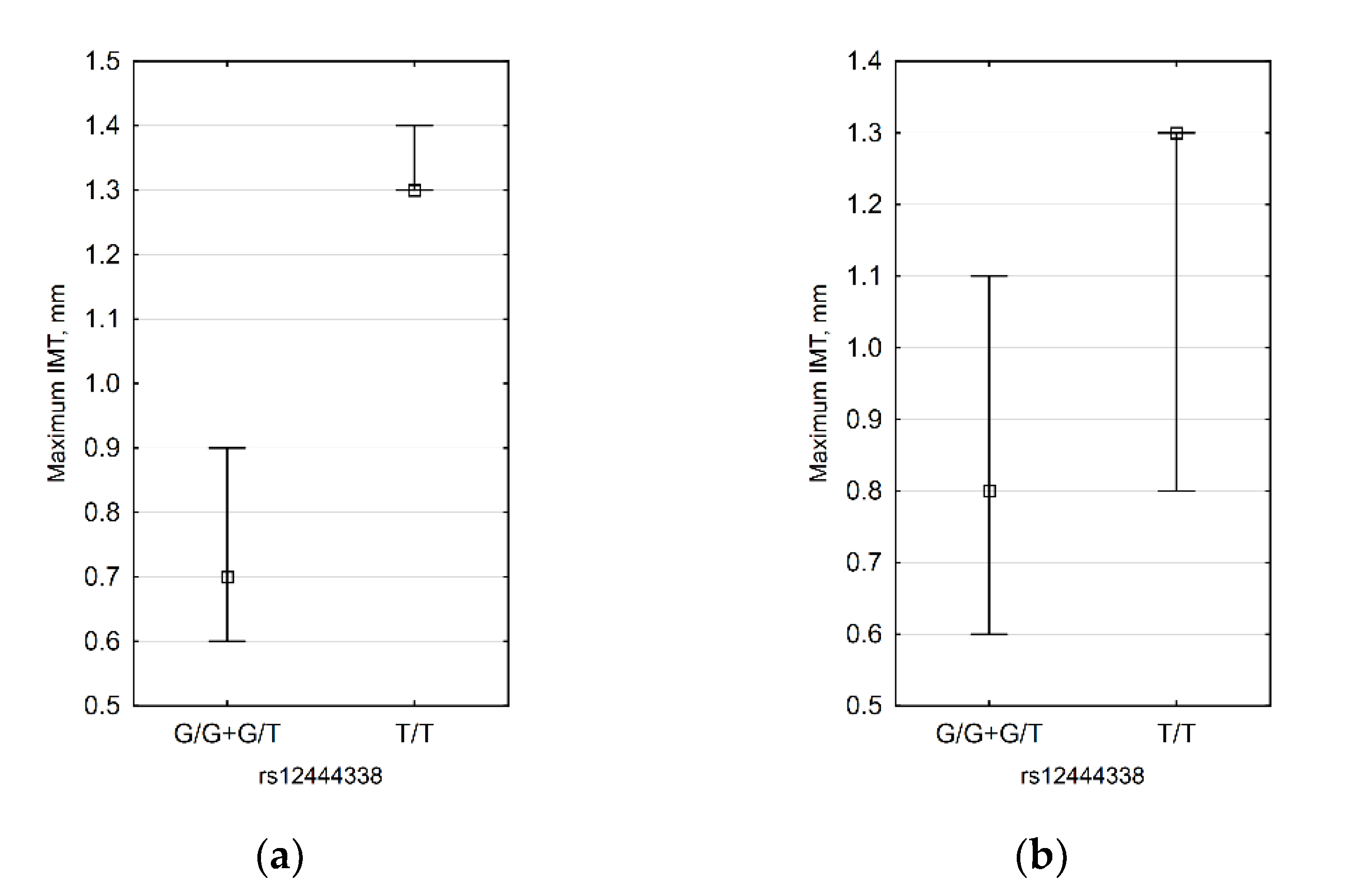
For rs12444338, we demonstrated the association of the G allele with thin IMT (Table 3). When IMT values were compared between G allele carriers and non-carriers, we found that the association is present in obese and female patients (Figure 4). The subgroup of obese female patients contained only nine subjects, so only a difference in the right carotid IMT was observed (0.6 mm vs. 1.3 mm).

Table 3.
Association between rs12444338 and the presence of thick intima-media.
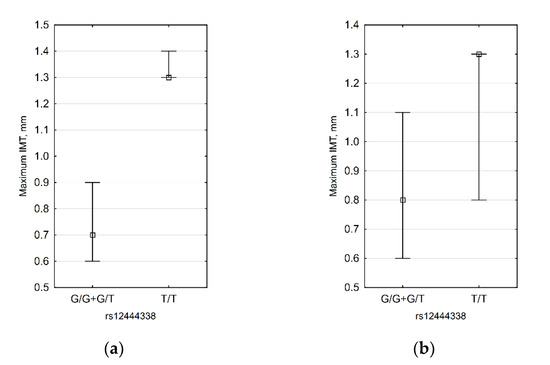
Figure 4.
Significant differences in IMT between rs12444338 G allele carriers and non-carriers: (a) obese patients (n = 10, p = 0.020) and (b) females (n = 25, p = 0.047).
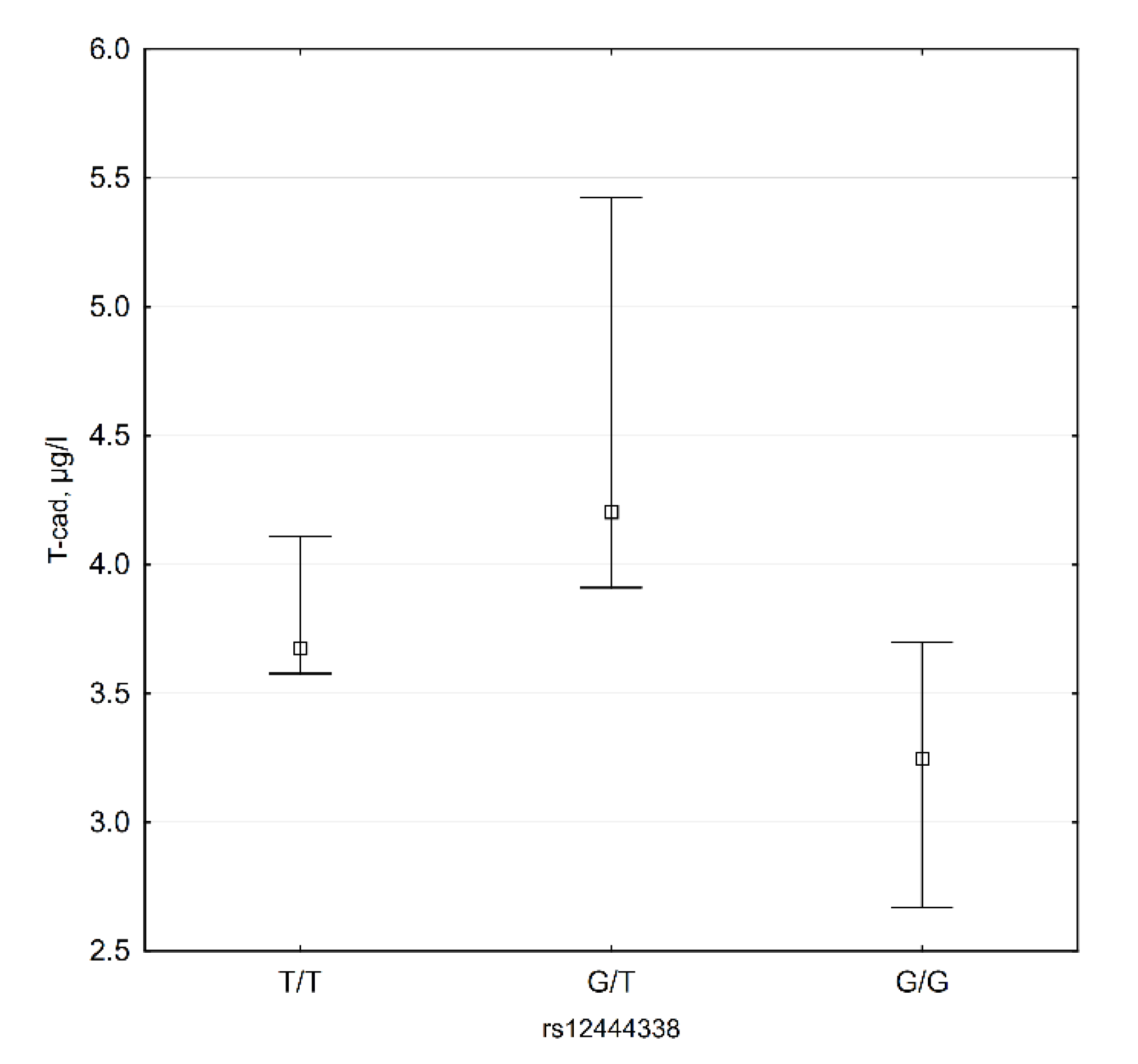
Our data show that plasma T-cadherin levels are lower in G/G subjects (Figure 5). Median T-cadherin plasma level in these subjects is 3.25 (2.67; 3.70) µg/L, while in other genotypes this level is 4.061 (3.62; 5.07) µg/L (p = 0.018).
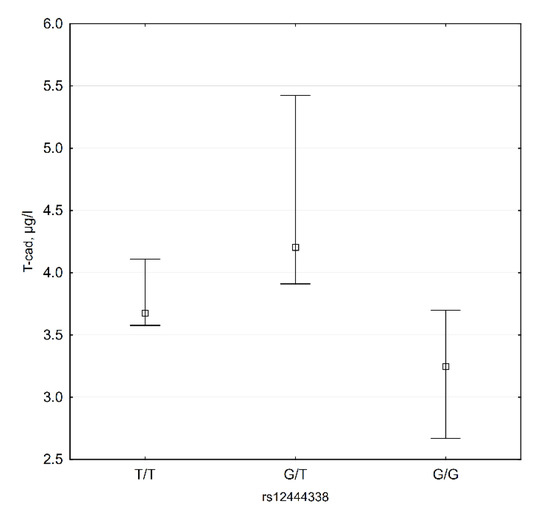
Figure 5.
CDH13 rs12444338 genotypes and T-cadherin plasma levels (n = 35).
Additionally, we demonstrated that rs12444338 G allele carriers have higher ApoB and LDL levels comparing to T/T genotype carriers—2.15 (1.98; 2.42) µM vs. 1.82 (1.62; 2.02) µM (p = 0.023) and 4.33 (3.62; 4.70) mM vs. 3.78 (3.12; 4.04) mM (p = 0.017) accordingly.
We showed no significant correlations for rs4783244.
4. Discussion
This study allowed us to test the hypothesis that the ratio of HMW adiponectin plasma level to LDL plasma level is a better predictor for early atherosclerosis than LDL or HMW adiponectin levels alone. Carotid IMT was chosen as a marker of early atherosclerosis. Our research has some limitations because data concern only early atherosclerotic changes. Nevertheless, we believe that biochemical markers of early atherosclerosis are very important from the clinical point of view. Early discovery of atherosclerotic lesions by biochemical screening methods may prevent the development of atherosclerosis complications.
The significance of LDL for atherosclerosis development is well established and demonstrated in many studies. Many anti-atherogenic effects were demonstrated in vitro for various cells (data reviewed in [29]). Human studies are also demonstrating an influence of low adiponectin levels on IMT (data reviewed in [32]). HMW adiponectin is considered as most active, and its levels inversely correlate with different pro-atherogenic conditions and cardiovascular diseases [20,33,34,35,36,37,38,39]. Nevertheless, a few data about the link between HMW adiponectin and carotid IMT are available. Several studies demonstrated no correlations between HMW adiponectin and carotid IMT in type 2 diabetic patients [68], obstructive sleep apnea patients [69], obese/overweight children [70], and in hemodialysis patients [71]. Some authors showed correlations between HMW adiponectin and carotid [72] or coronary [73] atherosclerotic plaques, but not IMT. In obese adolescents, the strong correlation between HMW adiponectin and carotid IMT was demonstrated, and after multiple testing, the HMW subfraction showed a better correlation to IMT compared with total adiponectin [74]. As far we know, there are no studies investigating the effects of HMW adiponectin on carotid IMT in persons with low or moderate cardiovascular risk. Our data demonstrate the link between HMW adiponectin and carotid IMT in both obese and non-obese male patients. It is known that total adiponectin levels are lower in men than in women [20], and the percentage of HMW adiponectin is lower in men, too [33]. This is consistent with our data demonstrating lower HMW adiponectin in men. In our study, we did not find the effect of HMW adiponectin on IMT in women.
In this paper, we studied the effects of the ratio of T-cadherin ligands for the first time. Previously it was only shown that HMW to total adiponectin ratio is related to the stable state of IMT in hemodialysis patients [71]. Takamura with colleagues also demonstrated that leptin to high-molecular-weight adiponectin ratio is independently correlated with carotid intima-media thickness in men but not in women [75]. We found some correlations between carotid IMT, HMW adiponectin/LDL, and HMW adiponectin/ApoB molar ratios, but they were obviously attributed to HMW adiponectin only (see Table 3).
Data from genetic studies demonstrate a possible link between SNPs in the CDH13 gene and blood pressure. Thus, we checked if levels T-cadherin and their ligands are correlated with AH, but no correlations were found.
It is also known that some SNPs in the CDH13 gene are associated with plasma adiponectin levels. For rs12051272, the T allele is associated with lower adiponectin levels. We discovered the correlation between the presence of this allele and the lower HMW adiponectin/ApoB molar ratio. T-allele was also associated with higher BMI. It is well known that adiponectin level is lower in obese patients [20]. Our data raise the question about the causal relationship between body mass and adiponectin level that is genetically determined, at least partly. We also can suppose that the ratio of T-cadherin ligands better correlates with genetically altered T-cadherin levels than levels of its ligands alone.
For rs12444338, we demonstrated that G allele carriers have lower IMT. Our data do not support previous research by Lee et al., who demonstrated lower IMT in GG vs. TT genotype carriers [47]. This inconsistency may be due to different populations studied and low sample sizes in both studies. In our case, maximal IMT measurements were used, while Lee et al. used mean IMT. We believe that maximal IMT is the better marker for early atherosclerosis, but our dataset showed similar results for maximal and mean IMT (data not shown). We also demonstrate lower plasma levels of T-cadherin in G/G subjects. It was unexpected after the demonstration of a 2.2-times increase of CDH13 promoter activity for the G allele by luciferase assay [46]. Higher T-cadherin expression may does not mean its higher plasma level, and this question needs further research. However, it needs to be noted that we showed higher ApoB and LDL levels in G allele carriers. It is possible to speculate that the G allele is linked to a lower tissue level of the receptor (T-cadherin), and this may lead to a higher level of the ligand (LDL) in plasma.
The correlation between rs12444338 G allele and lower IMT was confirmed in obese patients and females. Female sex and obesity have opposite effects on levels of T-cadherin ligands and, as far we know, have no effect on the level of T-cadherin itself. Females and persons with normal BMI have lower LDL and higher adiponectin levels comparing to males and obese persons accordingly [20,76]. Further investigations are needed to clarify the relationship between SNP in the T-cadherin gene, levels of its ligands, sex, BMI, and IMT.
5. Conclusions
This is the first study of the T-cadherin ligands (HMW adiponectin and LDL) ratio and its effect on early atherosclerosis. Despite the initial study hypothesis was not proven directly, we demonstrate a possible link between T-cadherin, its ligands levels, and carotid IMT. The inverse correlation between carotid artery IMT and HMW adiponectin levels in male subjects with moderate cardiovascular risk was demonstrated. We discovered an unexpected link between the G allele of rs12444338 SNP in the T-cadherin gene and two parameters, the lower level of circulating T-cadherin and the thinner intima-media.
Author Contributions
Conceptualization, A.B., M.B., and V.T.; methodology, A.B., M.B., M.T., A.P., A.K., and S.B.; formal analysis, A.B.; investigation, A.B., M.T., A.P., and A.T.; resources, M.T., A.P., I.B., A.K., and L.S.; data curation, A.B. and I.B.; writing—original draft preparation, A.B. and N.K.; writing—review and editing, A.B., A.K., M.B., N.K., and V.T.; supervision, V.T.; project administration, A.B.; funding acquisition, L.S. and N.K. All authors have read and agreed to the published version of the manuscript.
Funding
The reported study was funded by Russian foundation of basic research (RFBR), project 19-29-04118. Genotyping was conducted under the state assignment for Lomonosov Moscow State University using the equipment acquired as part of the Scientific Development Program of Lomonosov Moscow State University.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Local Ethics Committee of the Medical Scientific and Educational Center, Lomonosov Moscow State University (protocol No. 2/20 dated 16 March 2020).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy reason.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Appendix A

Table A1.
Spearman correlation coefficients between carotid IMT and laboratory data.
Table A1.
Spearman correlation coefficients between carotid IMT and laboratory data.
| ApoB | HMW Adn 1 | LDL | Adn/ApoB Molar Ratio | Adn/LDL Molar Ratio | T-cad | Carotid IMT, Right | Carotid IMT, Left | Carotid IMT, Max. | |
|---|---|---|---|---|---|---|---|---|---|
| ApoB | −0.277 | 0.805 * | −0.534 * | −0.468 * | −0.098 | −0.058 | −0.025 | −0.140 | |
| HMW Adn | −0.277 | −0.145 | 0.945 * | 0.944 * | 0.303 | −0.075 | −0.145 | −0.104 | |
| LDL | 0.805 * | −0.145 | −0.332 | −0.395 * | 0.187 | 0.146 | 0.114 | 0.022 | |
| Adn/ApoB molar ratio | −0.534 * | 0.945 * | −0.332 | 0.963 * | 0.322 | −0.046 | −0.112 | −0.046 | |
| Adn/LDL molar ratio | −0.468 * | 0.944 * | −0.395 * | 0.963 * | 0.232 | −0.097 | −0.101 | −0.058 | |
| T-cad | −0.098 | 0.303 | 0.187 | 0.322 | 0.232 | 0.075 | 0.068 | −0.006 | |
| Carotid IMT, right | −0.058 | −0.075 | 0.146 | −0.046 | −0.097 | 0.075 | 0.801 * | 0.936 * | |
| Carotid IMT, left | −0.025 | −0.145 | 0.114 | −0.112 | −0.101 | 0.068 | 0.801 * | 0.899 * | |
| Carotid IMT, max. | −0.140 | −0.104 | 0.022 | −0.046 | −0.058 | −0.006 | 0.936 * | 0.899 * |
1 High molecular weight adiponectin. * Significant correlations at p < 0.05.
References
- Goldstein, J.L.; Anderson, R.G.; Brown, M.S. Coated pits, coated vesicles, and receptor-mediated endocytosis. Nature 1979, 279, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Buhler, F.R.; Tkachuk, V.A.; Hahn, A.W.; Resink, T.J. Low- and high-density lipoproteins as hormonal regulators of platelet, vascular endothelial and smooth muscle cell interactions: Relevance to hypertension. J. Hypertens. Suppl. 1991, 9, S28–S36. [Google Scholar] [CrossRef] [PubMed]
- Bjorkerud, S.; Bjorkerud, B. Lipoproteins are major and primary mitogens and growth promoters for human arterial smooth muscle cells and lung fibroblasts in vitro. Arterioscler. Thromb. 1994, 14, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, V.; Tkachuk, V.; Buhler, F.; Resink, T. Phosphoinositide and calcium signalling responses in smooth muscle cells: Comparison between lipoproteins, Ang II, and PDGF. Biochem. Biophys. Res. Commun. 1992, 188, 1295–1304. [Google Scholar] [CrossRef]
- Bochkov, V.N.; Kuz’menko, E.S.; Rezink, T.; Tkachuk, V.A. “Classical” apo B,E-receptor does not mediate the activating effect of low density lipoproteins on the second messenger system in human platelets and vascular smooth muscle cells. Biokhimiia 1994, 59, 1330–1339. [Google Scholar] [PubMed]
- Bochkov, V.N.; Matchin, Y.G.; Fuki, I.V.; Lyakishev, A.A.; Tkachuk, V.A. Platelets in patients with homozygous familial hypercholesterolemia are sensitive to Ca(2+)-mobilizing activity of low density lipoproteins. Atherosclerosis 1992, 96, 119–124. [Google Scholar] [CrossRef]
- Bochkov, V.N.; Tkachuk, V.A.; Hahn, A.W.; Bernhardt, J.; Buhler, F.R.; Resink, T.J. Concerted effects of lipoproteins and angiotensin II on signal transduction processes in vascular smooth muscle cells. Arterioscler. Thromb. 1993, 13, 1261–1269. [Google Scholar] [CrossRef]
- Drobnik, W.; Mollers, C.; Resink, T.; Schmitz, G. Activation of phosphatidylinositol-specific phospholipase C in response to HDL3 and LDL is markedly reduced in cultured fibroblasts from Tangier patients. Arterioscler. Thromb. Vasc. Biol. 1995, 15, 1369–1377. [Google Scholar] [CrossRef]
- Kipmen-Korgun, D.; Osibow, K.; Zoratti, C.; Schraml, E.; Greilberger, J.; Kostner, G.; Jürgens, G.; Graier, W. T-Cadherin Mediates Low-Density Lipoprotein–Initiated Cell Proliferation Via the Ca2+-Tyrosine Kinase-Erk 1/2 Phathway. J. Cardiovasc. Pharmacol. 2005, 45, 418–430. [Google Scholar] [CrossRef]
- Orlov, S.; Resink, T.J.; Bernhardt, J.; Ferracin, F.; Buhler, F.R. Vascular smooth muscle cell calcium fluxes. Regulation by angiotensin II and lipoproteins. Hypertension 1993, 21, 195–203. [Google Scholar] [CrossRef]
- Resink, T.J.; Bochkov, V.N.; Tkachuk, V.A.; Buhler, F.R.; Hahn, A.W. Lipoproteins and angiotensin II exert synergistic effects on signalling processes in vascular smooth muscle cells. J. Hypertens. Suppl. 1993, 11, S110–S111. [Google Scholar] [CrossRef] [PubMed]
- Rubina, K.; Talovskaya, E.; Cherenkov, V.; Ivanov, D.; Stambolsky, D.; Storozhevykh, T.; Pinelis, V.; Shevelev, A.; Parfyonova, Y.; Resink, T.; et al. LDL induces intracellular signalling and cell migration via atypical LDL-binding protein T-cadherin. Mol. Cell. Biochem. 2005, 273, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, V.N.; Rozhkova, T.A.; Matchin Yu, G.; Lyakishev, A.A.; Bochkova, N.A.; Borisova Yu, L.; Kukharchuk, V.V.; Tkachuk, V.A. LDL- and agonist-induced Ca(2+)-mobilization in platelets of healthy subjects and in patients with familial hyperlipoproteinemia type II. Thromb. Res. 1991, 61, 403–409. [Google Scholar] [CrossRef]
- Bochkov, V.N.; Voino-Yasenetskaya, T.A.; Tkachuk, V.A. Epinephrine potentiates activation of human platelets by low density lipoproteins. Biochim. Biophys. Acta 1991, 1097, 123–127. [Google Scholar] [CrossRef]
- González-Timón, B.; Gonzalez-Muñoz, M.; Zaragoza, C.; Lamas, S.; Melián, E. Native and oxidized low density lipoproteins oppositely modulate the effects of insulin-like growth factor I on VSMC. Cardiovasc. Res. 2004, 61, 247–255. [Google Scholar] [CrossRef][Green Version]
- Resink, T.J.; Bochkov, V.N.; Hahn, A.W.; Philippova, M.P.; Buhler, F.R.; Tkachuk, V.A. Low- and high-density lipoproteins as mitogenic factors for vascular smooth muscle cells: Individual, additive and synergistic effects. J. Vasc. Res. 1995, 32, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Seewald, S.; Nickenig, G.; Ko, Y.; Vetter, H.; Sachinidis, A. Low density lipoprotein enhances the thrombin-induced growth of vascular smooth muscle cells. Cardiovasc. Res. 1997, 36, 92–100. [Google Scholar] [CrossRef]
- Tkachuk, V.; Bochkov, V.; Philippova, M.; Stambolsky, D. Identification of atypical lipoprotein-binding protein from human aortic smooth muscle as T-cadherin. FEBS Lett. 1998, 421, 208–212. [Google Scholar] [CrossRef]
- Hug, C.; Wang, J.; Ahmad, N.S.; Bogan, J.S.; Tsao, T.S.; Lodish, H.F. T-cadherin is a receptor for hexameric and high-molecular-weight forms of Acrp30/adiponectin. Proc. Natl. Acad. Sci. USA 2004, 101, 10308–10313. [Google Scholar] [CrossRef]
- Arita, Y.; Kihara, S.; Ouchi, N.; Takahashi, M.; Maeda, K.; Miyagawa, J.; Hotta, K.; Shimomura, I.; Nakamura, T.; Miyaoka, K.; et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem. Biophys. Res. Commun. 1999, 425, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Fruebis, J.; Tsao, T.S.; Javorschi, S.; Ebbets-Reed, D.; Erickson, M.R.; Yen, F.T.; Bihain, B.E.; Lodish, H.F. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc. Natl. Acad. Sci. USA 2001, 98, 2005–2010. [Google Scholar] [CrossRef]
- Hu, E.L.P.; Splegelman, B.M. AdipoQ Is a Novel Adipose-specific Gene Dysregulated in Obesity. J. Biol. Chem. 1996, 271, 10697–10703. [Google Scholar] [CrossRef]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef] [PubMed]
- Kojima, S.; Funahashi, T.; Sakamoto, T.; Miyamoto, S.; Soejima, H.; Hokamaki, J.; Kajiwara, I.; Sugiyama, S.; Yoshimura, M.; Fujimoto, K.; et al. The variation of plasma concentrations of a novel, adipocyte derived protein, adiponectin, in patients with acute myocardial infarction. Heart 2003, 89, 667. [Google Scholar] [CrossRef] [PubMed]
- Kumada, M.; Kihara, S.; Sumitsuji, S. Association of Hypoadiponectinemia With Coronary Artery Disease in Men. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Scherer, P.E.; Williams, S.; Fogliano, M.; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q, produced exclusively in adipocytes. J. Biol. Chem. 1995, 270, 26746–26749. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Imai, Y.; Shimozawa, N.; Hioki, K.; Uchida, S.; Ito, Y.; Takakuwa, K.; Matsui, J.; et al. Globular adiponectin protected ob/ob mice from diabetes and ApoE-deficient mice from atherosclerosis. J. Biol. Chem. 2003, 278, 2461–2468. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef]
- Zhu, W.; Cheng, K.K.; Vanhoutte, P.M.; Lam, K.S.; Xu, A. Vascular effects of adiponectin: Molecular mechanisms and potential therapeutic intervention. Clin. Sci. 2008, 114, 361–374. [Google Scholar] [CrossRef]
- Ebrahimi-Mamaeghani, M.; Mohammadi, S.; Arefhosseini, S.R.; Fallah, P.; Bazi, Z. Adiponectin as a potential biomarker of vascular disease. Vasc. Health Risk Manag. 2015, 11, 55–70. [Google Scholar] [CrossRef]
- Iglseder, B.; Mackevics, V.; Stadlmayer, A.; Tasch, G.; Ladurner, G.; Paulweber, B. Plasma adiponectin levels and sonographic phenotypes of subclinical carotid artery atherosclerosis: Data from the SAPHIR Study. Stroke 2005, 36, 2577–2582. [Google Scholar] [CrossRef] [PubMed]
- Gasbarrino, K.; Gorgui, J.; Nauche, B.; Cote, R.; Daskalopoulou, S.S. Circulating adiponectin and carotid intima-media thickness: A systematic review and meta-analysis. Metabolism 2016, 65, 968–986. [Google Scholar] [CrossRef]
- Waki, H.; Yamauchi, T.; Kamon, J.; Ito, Y.; Uchida, S.; Kita, S.; Hara, K.; Hada, Y.; Vasseur, F.; Froguel, P.; et al. Impaired multimerization of human adiponectin mutants associated with diabetes. Molecular structure and multimer formation of adiponectin. J. Biol. Chem. 2003, 278, 40352–40363. [Google Scholar] [CrossRef] [PubMed]
- Daniele, A.; De Rosa, A.; De Cristofaro, M.; Monaco, M.L.; Masullo, M.; Porcile, C.; Capasso, M.; Tedeschi, G.; Oriani, G.; Di Costanzo, A. Decreased concentration of adiponectin together with a selective reduction of its high molecular weight oligomers is involved in metabolic complications of myotonic dystrophy type 1. Eur. J. Endocrinol. 2011, 165, 969–975. [Google Scholar] [CrossRef]
- De Rosa, A.; Monaco, M.L.; Nigro, E.; Scudiero, O.; D’Andrea, M.; Pilla, F.; Oriani, G.; Daniele, A. Tissue-specific downregulation of the adiponectin "system": Possible implications for fat accumulation tendency in the pig. Domest. Anim. Endocrinol. 2013, 44, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Horakova, D.; Azeem, K.; Benesova, R.; Pastucha, D.; Horak, V.; Dumbrovska, L.; Martinek, A.; Novotny, D.; Svagera, Z.; Hobzova, M.; et al. Total and High Molecular Weight Adiponectin Levels and Prediction of Cardiovascular Risk in Diabetic Patients. Int. J. Endocrinol. 2015, 545068. [Google Scholar] [CrossRef]
- Kobayashi, H.; Ouchi, N.; Kihara, S.; Walsh, K.; Kumada, M.; Abe, Y.; Funahashi, T.; Matsuzawa, Y. Selective suppression of endothelial cell apoptosis by the high molecular weight form of adiponectin. Circ. Res. 2004, 94, e27-31. [Google Scholar] [CrossRef]
- Pajvani, U.B.; Hawkins, M.; Combs, T.P.; Rajala, M.W.; Doebber, T.; Berger, J.P.; Wagner, J.A.; Wu, M.; Knopps, A.; Xiang, A.H.; et al. Complex distribution, not absolute amount of adiponectin, correlates with thiazolidinedione-mediated improvement in insulin sensitivity. J. Biol. Chem. 2004, 279, 12152–12162. [Google Scholar] [CrossRef] [PubMed]
- Pischon, T.; Hu, F.B.; Girman, C.J.; Rifai, N.; Manson, J.E.; Rexrode, K.M.; Rimm, E.B. Plasma total and high molecular weight adiponectin levels and risk of coronary heart disease in women. Atherosclerosis 2011, 219, 322–329. [Google Scholar] [CrossRef]
- Denzel, M.S.; Scimia, M.C.; Zumstein, P.M.; Walsh, K.; Ruiz-Lozano, P.; Ranscht, B. T-cadherin is critical for adiponectin-mediated cardioprotection in mice. J. Clin. Investig. 2010, 120, 4342–4352. [Google Scholar] [CrossRef]
- Fujishima, Y.; Maeda, N.; Matsuda, K.; Masuda, S.; Mori, T.; Fukuda, S.; Sekimoto, R.; Yamaoka, M.; Obata, Y.; Kita, S.; et al. Adiponectin association with T-cadherin protects against neointima proliferation and atherosclerosis. FASEB J. 2017, 31, 1571–1583. [Google Scholar] [CrossRef] [PubMed]
- Parker-Duffen, J.L.; Nakamura, K.; Silver, M.; Kikuchi, R.; Tigges, U.; Yoshida, S.; Denzel, M.S.; Ranscht, B.; Walsh, K. T-cadherin Is Essential for Adiponectin-mediated Revascularization. J. Biol. Chem. 2013, 288, 24886–24897. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.M.; Lin, T.H.; Chen, J.W.; Leu, H.B.; Yang, H.C.; Ho, H.Y.; Ting, C.T.; Sheu, S.H.; Tsai, W.C.; Chen, J.H.; et al. A genome-wide association study reveals a quantitative trait locus of adiponectin on CDH13 that predicts cardiometabolic outcomes. Diabetes 2011, 60, 2417–2423. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Danese, E.; Montagnana, M.; Sjogren, M.; Almgren, P.; Guidi, G.C.; Hedblad, B.; Engstrom, G.; Lechi, A.; Minuz, P.; et al. A variant upstream of the CDH13 adiponectin receptor gene and metabolic syndrome in Swedes. Am. J. Cardiol. 2011, 108, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Kim, Y.M.; Chen, P.; Igase, M.; Kawamoto, R.; Kim, M.K.; Kohara, K.; Lee, J.; Miki, T.; Ong, R.T.; et al. Genetic variation in CDH13 is associated with lower plasma adiponectin levels but greater adiponectin sensitivity in East Asian populations. Diabetes 2013, 62, 4277–4283. [Google Scholar] [CrossRef] [PubMed]
- Jee, S.H.; Sull, J.W.; Lee, J.E.; Shin, C.; Park, J.; Kimm, H.; Cho, E.Y.; Shin, E.S.; Yun, J.E.; Park, J.W.; et al. Adiponectin concentrations: A genome-wide association study. Am. J. Hum. Genet. 2010, 87, 545–552. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, J.H.; Shin, D.J.; Park, S.; Kang, S.M.; Jang, Y.; Lee, S.H. Association between CDH13 variants and cardiometabolic and vascular phenotypes in a Korean population. Yonsei Med. J. 2013, 54, 1305–1312. [Google Scholar] [CrossRef]
- Morisaki, H.; Yamanaka, I.; Iwai, N.; Miyamoto, Y.; Kokubo, Y.; Okamura, T.; Okayama, A.; Morisaki, T. CDH13 gene coding T-cadherin influences variations in plasma adiponectin levels in the Japanese population. Hum. Mutat. 2012, 33, 402–410. [Google Scholar] [CrossRef]
- Org, E.; Eyheramendy, S.; Juhanson, P.; Gieger, C.; Lichtner, P.; Klopp, N.; Veldre, G.; Doring, A.; Viigimaa, M.; Sober, S.; et al. Genome-wide scan identifies CDH13 as a novel susceptibility locus contributing to blood pressure determination in two European populations. Hum. Mol. Genet. 2009, 18, 2288–2296. [Google Scholar] [CrossRef]
- Teng, M.S.; Hsu, L.A.; Wu, S.; Sun, Y.C.; Juan, S.H.; Ko, Y.L. Association of CDH13 genotypes/haplotypes with circulating adiponectin levels, metabolic syndrome, and related metabolic phenotypes: The role of the suppression effect. PLoS ONE 2015, 10, e0122664. [Google Scholar] [CrossRef]
- Balatsky, A.V.; Konovalov, D.Y.; Samokhodskaya, L.M.; Kochegura, T.N.; Rubina, K.A.; Tkachuk, V.A. Single Nucleotide Polymorphisms in T-Cadherin Gene (CDH13) Have Cumulative Effect on Body Mass in Patients With Ischemic Heart Disease. Kardiologiia 2015, 55, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Consortium, W.T.C.C. Genome-wide association study of 14,000 cases of seven common diseases and 3000 shared controls. Nature 2007, 447, 661–678. [Google Scholar] [CrossRef]
- Levy, D.; Larson, M.G.; Benjamin, E.J.; Newton-Cheh, C.; Wang, T.J.; Hwang, S.J.; Vasan, R.S.; Mitchell, G.F. Framingham Heart Study 100K Project: Genome-wide associations for blood pressure and arterial stiffness. BMC Med. Genet. 2007, 8, 1–11. [Google Scholar] [CrossRef]
- Menzaghi, C.; Trischitta, V. The Adiponectin Paradox for All-Cause and Cardiovascular Mortality. Diabetes 2018, 67, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, V.N.; Tkachuk, V.A.; Kuzmenko, Y.S.; Borisova, Y.L.; Buhler, F.R.; Resink, T.J. Characteristics of low and high density lipoprotein binding and lipoprotein-induced signaling in quiescent human vascular smooth muscle cells. Mol. Pharmacol. 1994, 45, 262–270. [Google Scholar] [PubMed]
- Tkachuk, V.A.; Kuzmenko, Y.S.; Resink, T.J.; Stambolsky, D.V.; Bochkov, V.N. Atypical Low-Density-Lipoprotein Binding-Site That May Mediate Lipoprotein-Induced Signal-Transduction. Mol. Pharmacol. 1994, 46, 1129–1137. [Google Scholar]
- Fukuda, S.; Kita, S.; Obata, Y.; Fujishima, Y.; Nagao, H.; Masuda, S.; Tanaka, Y.; Nishizawa, H.; Funahashi, T.; Takagi, J.; et al. The unique prodomain of T-cadherin plays a key role in adiponectin binding with the essential extracellular cadherin repeats 1 and 2. J. Biol. Chem. 2017. [Google Scholar] [CrossRef]
- Balatskaya, M.N.; Balatskii, A.V.; Sharonov, G.V.; Tkachuk, V.A. T-cadherin as a novel receptor regulating metabolism in the blood vessel and heart cells: From structure to function. J Evol Biochem Phys. 2016, 52, 103–118. [Google Scholar] [CrossRef]
- Rubina, K.A.; Semina, E.A.; Balatskaya, M.N.; Plekhanova, O.S.; Tkachuk, V.A. Mechanisms of regulation of the directed growth of vessels and nerves by the fibrinolytic system components and GPI-anchored navigation receptors. Neurosci. Behav. Physiol. 2018, 1001–1026. [Google Scholar] [CrossRef]
- Matsuda, K.; Fujishima, Y.; Maeda, N.; Mori, T.; Hirata, A.; Sekimoto, R.; Tsushima, Y.; Masuda, S.; Yamaoka, M.; Inoue, K.; et al. Positive feedback regulation between adiponectin and T-cadherin impacts adiponectin levels in tissue and plasma of male mice. Endocrinology 2014, en20141618. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, D.; Philippova, M.; Antropova, J.; Gubaeva, F.; Iljinskaya, O.; Tararak, E.; Bochkov, V.; Erne, P.; Resink, T.; Tkachuk, V. Expression of cell adhesion molecule T-cadherin in the human vasculature. Histochem. Cell Biol. 2001, 115, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Sole, X.; Guino, E.; Valls, J.; Iniesta, R.; Moreno, V. SNPStats: A web tool for the analysis of association studies. Bioinformatics 2006, 22, 1928–1929. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corra, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts)Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Putku, M.; Kals, M.; Inno, R.; Kasela, S.; Org, E.; Kozich, V.; Milani, L.; Laan, M. CDH13 promoter SNPs with pleiotropic effect on cardiometabolic parameters represent methylation QTLs. Hum. Genet. 2015, 134, 291–303. [Google Scholar] [CrossRef]
- Wu, Y.; Li, Y.; Lange, E.M.; Croteau-Chonka, D.C.; Kuzawa, C.W.; McDade, T.W.; Qin, L.; Curocichin, G.; Borja, J.B.; Lange, L.A.; et al. Genome-wide association study for adiponectin levels in Filipino women identifies CDH13 and a novel uncommon haplotype at KNG1-ADIPOQ. Hum. Mol. Genet. 2010, 19, 4955–4964. [Google Scholar] [CrossRef]
- Uetani, E.; Tabara, Y.; Kawamoto, R.; Onuma, H.; Kohara, K.; Osawa, H.; Miki, T. CDH13 genotype-dependent association of high-molecular weight adiponectin with all-cause mortality: The J-SHIPP study. Diabetes Care 2014, 37, 396–401. [Google Scholar] [CrossRef][Green Version]
- Mansouri, M.; Heshmat, R.; Tabatabaei-Malazy, O.; Sharifi, F.; Badamchizadeh, Z.; Alatab, S.; Omidfar, K.; Fakhrzadeh, H.; Larijani, B. The association of carotid intima media thickness with retinol binding protein-4 and total and high molecular weight adiponectin in type 2 diabetic patients. J. Diabetes Metab. Disord. 2012, 11, 2. [Google Scholar] [CrossRef][Green Version]
- Song, F.; Zou, J.; Song, Z.; Xu, H.; Qian, Y.; Zhu, H.; Liu, S.; Guan, J.; Chen, J.; Yi, H. Association of Adipocytokines With Carotid Intima Media Thickness and Arterial Stiffness in Obstructive Sleep Apnea Patients. Front. Endocrinol. 2020, 11, 177. [Google Scholar] [CrossRef] [PubMed]
- Ciccone, M.M.; Faienza, M.F.; Altomare, M.; Nacci, C.; Montagnani, M.; Valente, F.; Cortese, F.; Gesualdo, M.; Zito, A.; Mancarella, R.; et al. Endothelial and Metabolic Function Interactions in Overweight/Obese Children. J. Atheroscler. Thromb. 2016, 23, 950–959. [Google Scholar] [CrossRef]
- Tsushima, M.; Terayama, Y.; Momose, A.; Funyu, T.; Ohyama, C. Progression of atherosclerosis in hemodialysis patients: Effect of adiponectin on carotid intima media thickness. J. Atheroscler. Thromb. 2008, 15, 213–218. [Google Scholar] [CrossRef]
- Brovin, D.L.; Belyaeva, O.D.; Pchelina, S.N.; Berezina, A.V.; Karonova, T.L.; Bazhenova, E.A.; Kolodina, D.A.; Bakulina, A.S.; Polyakova, E.A.; Listopad, O.V.; et al. Common Carotid Intima-Media Thickness, Levels of Total and High-Molecular Weight Adiponectin in Women With Abdominal Obesity. Kardiologiia 2018, 58, 29–36. [Google Scholar] [CrossRef] [PubMed]
- von Eynatten, M.; Humpert, P.M.; Bluemm, A.; Lepper, P.M.; Hamann, A.; Allolio, B.; Nawroth, P.P.; Bierhaus, A.; Dugi, K.A. High-molecular weight adiponectin is independently associated with the extent of coronary artery disease in men. Atherosclerosis 2008, 199, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Mangge, H.; Almer, G.; Haj-Yahya, S.; Pilz, S.; Gasser, R.; Moller, R.; Horejsi, R. Preatherosclerosis and adiponectin subfractions in obese adolescents. Obesity (Silver Spring) 2008, 16, 2578–2584. [Google Scholar] [CrossRef]
- Takamura, N.; Hayashida, N.; Hagane, K.; Kadota, K.; Yamasaki, H.; Abiru, N.; Ozono, Y.; Kamihira, S.; Aoyagi, K.; Ishibashi, K.; et al. Leptin to high-molecular-weight adiponectin ratio is independently correlated with carotid intima-media thickness in men, but not in women. Biomarkers 2010, 15, 340–344. [Google Scholar] [CrossRef]
- Swiger, K.J.; Martin, S.S.; Blaha, M.J.; Toth, P.P.; Nasir, K.; Michos, E.D.; Gerstenblith, G.; Blumenthal, R.S.; Jones, S.R. Narrowing sex differences in lipoprotein cholesterol subclasses following mid-life: The very large database of lipids (VLDL-10B). J. Am. Heart Assoc. 2014, 3, e000851. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).