Numerical Modeling of Ammonia-Fueled Protonic-Ion Conducting Electrolyte-Supported Solid Oxide Fuel Cell (H-SOFC): A Brief Review
Abstract
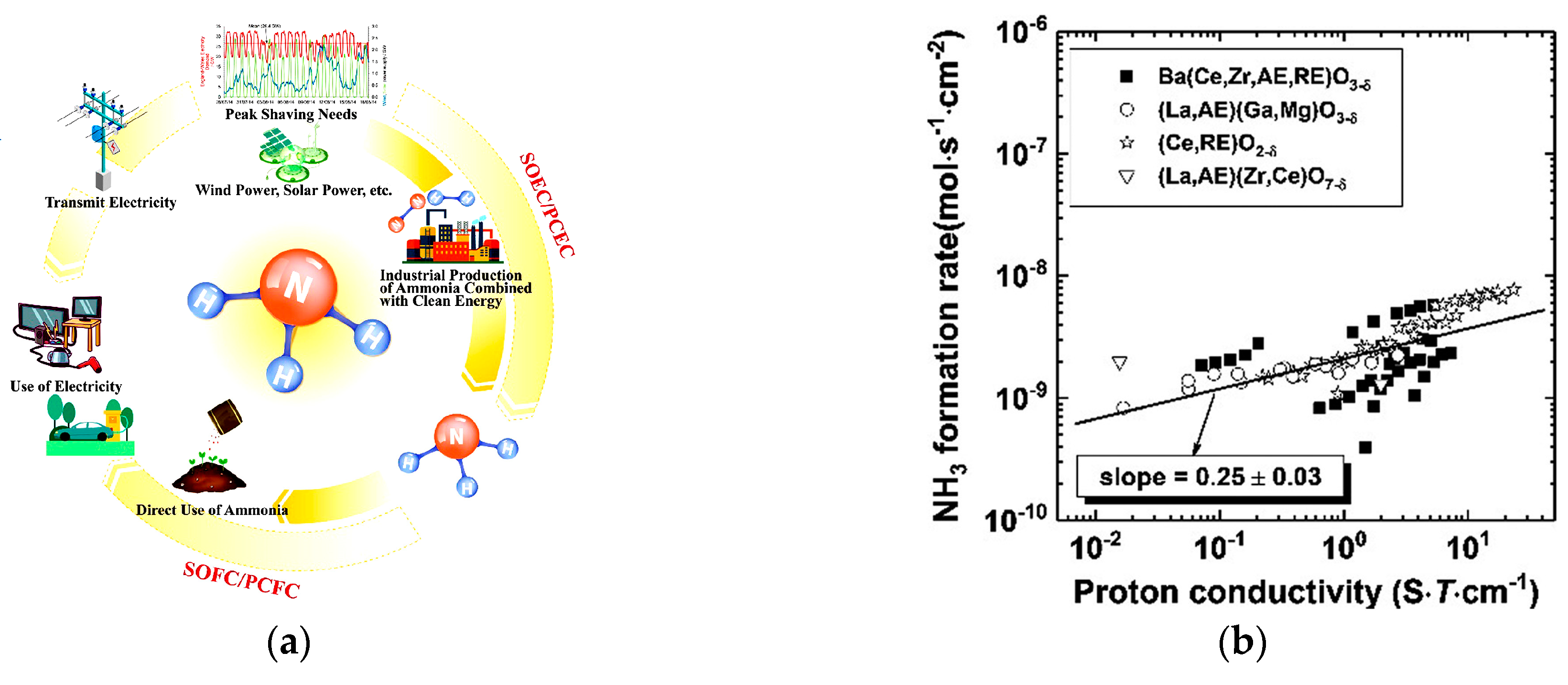
:1. Introduction
2. General Principle and Working Nature of Ammonia-Fueled H-SOFC
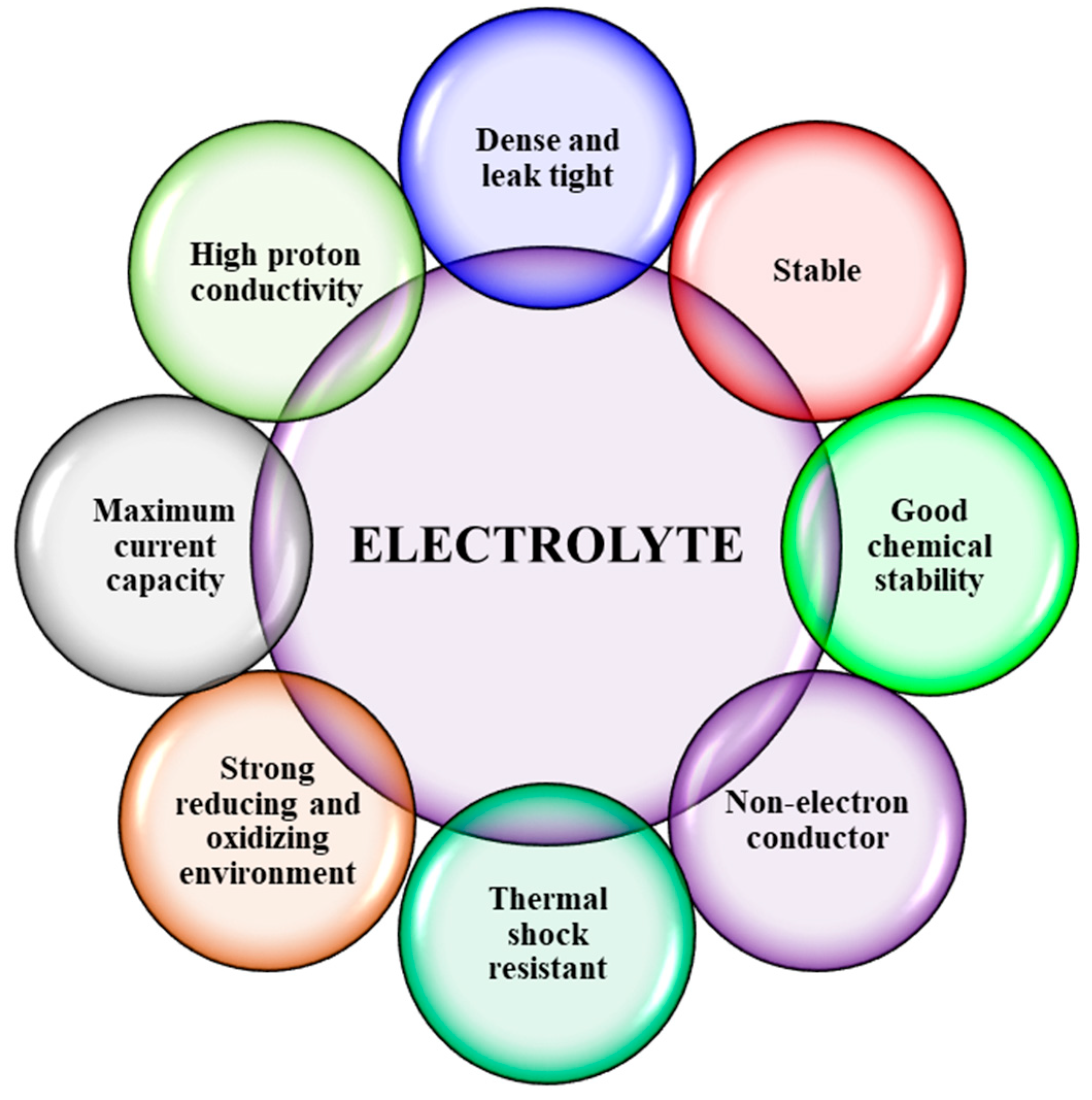
2.1. Electrolyte Materials
2.2. Cathode Materials
- High surface area and compatibility with the electrolyte;
- Fairly high electron-conducting activity;
- Adequate porosity to support oxygen diffusion;
- Oxide ions should have high ionic conductivity.
2.3. Anode Materials for SOFCs
- It should have sufficient porosity, conductivity, and chemical and mechanical stability properties;
- It should have electrochemical activity and fuel flexibility properties;
- It should have strong catalytic activity to promote the reaction of the fuel with oxide ions;
- It should have low polarization resistance of the electrode.
3. Role of Numerical Modeling
3.1. Application of Numerical Modeling in Ammonia-Fueled H-SOFC
3.2. Recent Ammonia-Fueled SOFC Models
3.3. Governing Mathematical Equations Used in Ammonia-Fueled H-SOFC
3.4. Comparative Model Analysis of Ammonia-Fueled H-SOFC with SOFC
3.5. Research Gap and Future Prospects
- Radiative heat transfer analysis is still needed to investigate the ammonia-fueled H-SOFC for temperature-dependent materials, reformations, transportations, and electrochemical reactions.
- One of the major challenges of ammonia-fueled H-SOFC modeling is to maintain low ohmic resistance, separate the higher ohmic overpotential, loss of polarization, and how other parameter affects them.
- Ammonia-fueled anode and cathode concentration overpotential for H-SOFCs is still a dilemma for SOFC research. A more precise dynamic numerical model should be introduced to mitigate the dilemma regarding the anode and cathode concentration of ammonia-fueled H-SOFCs.
- For fuel cell modeling, a fundamental challenge is to obtain the required input data for numerical modeling and validate them with experimental data. Despite the great potential of SOFC modeling, it is still difficult to include all modeling approaches that accurately describe the statistical analysis of fuel cells due to the lack of reliable modeling techniques and data. Today, obtaining data from experimental tests is challenging and sometimes severely constrained, making it more difficult to design fuel cells at an optimal scale. For an optimized fuel cell design, experimental data must be input for comparison with simulated data. This limitation should be removed because it brings difficulties in the fuel cell design analysis.
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hajimolana, S.A.; Hussain, M.A.; Daud, W.M.A.W.; Soroush, M.; Shamiri, A. Mathematical modeling of solid oxide fuel cells: A review. Renew. Sustain. Energy Rev. 2011, 15, 1893–1917. [Google Scholar] [CrossRef]
- Wang, B.; Li, T.; Gong, F.; Othman, M.H.D.; Xiao, R. Ammonia as a green energy carrier: Electrochemical synthesis and direct ammonia fuel cell—A comprehensive review. Fuel Process. Technol. 2022, 235, 107380. [Google Scholar] [CrossRef]
- Zainon, A.N.; Somalu, M.R.; Bahrain, A.M.K.; Muchtar, A.; Baharuddin, N.A.; SA, M.A.; Osman, N.; Samat, A.A.; Azad, A.K.; Brandon, N.P. Challenges in using perovskite-based anode materials for solid oxide fuel cells with various fuels: A review. Int. J. Hydrogen Energy 2023, 48, 20441–20464. [Google Scholar] [CrossRef]
- Azad, A.K.; Abdalla, A.M.; Afif, A.; Azad, A.; Afroze, S.; Idris, A.C.; Park, J.-Y.; Saqib, M.; Radenahmad, N.; Hossain, S.; et al. Improved mechanical strength, proton conductivity and power density in an ‘all-protonic’ ceramic fuel cell at intermediate temperature. Sci. Rep. 2021, 11, 19382. [Google Scholar] [CrossRef]
- Lee, T.H.; Park, K.Y.; Kim, N.I.; Song, S.J.; Hong, K.H.; Ahn, D.; Azad, A.K.; Hwang, J.; Bhattacharjee, S.; Lee, S.C.; et al. Robust NdBa0.5Sr0.5Co1.5Fe0.5O5+δ cathode material and its degradation prevention operating logic for intermediate temperature-solid oxide fuel cells. J. Power Sources 2016, 331, 495–506. [Google Scholar] [CrossRef]
- Abdalla, A.M.; Hossain, S.; Nisfindy, O.B.; Azad, A.T.; Dawood, M.; Azad, A.K. Hydrogen production, storage, transportation and key challenges with applications: A review. Energy Convers. Manag. 2018, 165, 602–627. [Google Scholar] [CrossRef]
- Radenahmad, N.; Azad, A.T.; Saghir, M.; Taweekun, J.; Bakar, M.S.A.; Reza, M.S.; Azad, A.K. A review on biomass derived syngas for SOFC based combined heat and power application. Renew. Sustain. Energy Rev. 2020, 119, 109560. [Google Scholar] [CrossRef]
- Afif, A.; Radenahmad, N.; Cheok, Q.; Shams, S.; Kim, J.H.; Azad, A.K. Ammonia-fed fuel cells: A comprehensive review. Renew. Sustain. Energy Rev. 2016, 60, 822–835. [Google Scholar] [CrossRef]
- Valera-Medina, A.; Amer-Hatem, F.; Azad, A.K.; Dedoussi, I.C.; de Joannon, M.; Fernandes, R.X.; Glarborg, P.; Hashemi, H.; He, X.; Mashruk, S.; et al. Review on Ammonia as a Potential Fuel: From Synthesis to Economics. Energy Fuels 2021, 35, 6964–7029. [Google Scholar] [CrossRef]
- Ilbas, M.; Alemu, M.A.; Cimen, F.M. Comparative performance analysis of a direct ammonia-fuelled anode supported flat tubular solid oxide fuel cell: A 3D numerical study. Int. J. Hydrogen Energy 2022, 47, 3416–3428. [Google Scholar] [CrossRef]
- Oh, S.; Oh, M.J.; Hong, J.; Yoon, K.J.; Ji, H.-I.; Lee, J.-H.; Kang, H.; Son, J.-W.; Yang, S. A comprehensive investigation of direct ammonia-fueled thin-film solid-oxide fuel cells: Performance, limitation, and prospects. iScience 2022, 25, 105009. [Google Scholar] [CrossRef] [PubMed]
- Asmare, M.; Ilbas, M.; Cimen, F.M.; Timurkutluk, C.; Onbilgin, S. Three-dimensional numerical simulation and experimental validation on ammonia and hydrogen fueled micro tubular solid oxide fuel cell performance. Int. J. Hydrogen Energy 2022, 47, 15865–15874. [Google Scholar] [CrossRef]
- Asmare, M.; Ilbas, M.; Yalcin, S. Numerical modelling and comparative analysis of direct ammonia fuelled protonic and oxygen- ion conducting tubular solid oxide fuel cell. Int. J. Hydrogen Energy 2021, 46, 36878–36889. [Google Scholar] [CrossRef]
- Radenahmad, N.; Afif, A.; Petra, P.I.; Rahman, S.M.H.; Eriksson, S.-G.; Azad, A.K. Proton-conducting electrolytes for direct methanol and direct urea fuel cells—A state-of-the-art review. Renew. Sustain. Energy Rev. 2016, 57, 1347–1358. [Google Scholar] [CrossRef]
- Hossain, S.; Abdalla, A.M.; Suhaili, S.B.H.; Kamal, I.; Shaikh, S.P.S.; Dawood, M.K.; Azad, A.K. Nanostructured graphene materials utilization in fuel cells and batteries: A review. J. Energy Storage 2020, 29, 101386. [Google Scholar] [CrossRef]
- Dhanasekaran, A.; Subramanian, Y.; Omeiza, L.A.; Raj, V.; Yassin, H.P.H.M.; SA, M.A.; Azad, A.K. Computational Fluid Dynamics for Protonic Ceramic Fuel Cell Stack Modeling: A Brief Review. Energies 2022, 16, 208. [Google Scholar] [CrossRef]
- Hossain, S.; Abdalla, A.M.; Zaini, J.H.; Savaniu, C.D.; Irvine, J.T.S.; Azad, A.K. Highly dense and novel proton conducting materials for SOFC electrolyte. Int. J. Hydrogen Energy 2017, 42, 27308–27322. [Google Scholar] [CrossRef]
- Hossain, S.; Abdalla, A.M.; Jamain, S.N.B.; Zaini, J.H.; Azad, A.K. A review on proton conducting electrolytes for clean energy and intermediate temperature-solid oxide fuel cells. Renew. Sustain. Energy Rev. 2017, 79, 750–764. [Google Scholar] [CrossRef]
- Afroze, S.; Karim, A.; Cheok, Q.; Eriksson, S.; Azad, A.K. Latest development of double perovskite electrode materials for solid oxide fuel cells: A review. Front. Energy 2019, 13, 770–797. [Google Scholar] [CrossRef]
- Hussain, S.; Li, Y. Review of solid oxide fuel cell materials: Cathode, anode, and electrolyte. Energy Transit. 2020, 4, 113–126. [Google Scholar] [CrossRef]
- Fergus, J.W. Electrolytes for solid oxide fuel cells. J. Power Sources 2006, 162, 30–40. [Google Scholar] [CrossRef]
- Dalslet, B.; Blennow, P.; Hendriksen, P.V.; Bonanos, N.; Lybye, D.; Mogensen, M. Assessment of doped ceria as electrolyte. J. Solid State Electrochem. 2006, 10, 547–561. [Google Scholar] [CrossRef]
- Shi, H.; Su, C.; Ran, R.; Cao, J.; Shao, Z. Electrolyte materials for intermediate-temperature solid oxide fuel cells. Prog. Nat. Sci. Mater. Int. 2020, 30, 764–774. [Google Scholar] [CrossRef]
- Biswas, S.; Kaur, G.; Paul, G.; Giddey, S. A critical review on cathode materials for steam electrolysis in solid oxide electrolysis. Int. J. Hydrogen Energy. 2023, 48, 12541–12570. [Google Scholar] [CrossRef]
- Xu, H.; Dang, L.; Yan, J.; Wan, F.; Gong, W. Preparation of a nano-size (La0.2Nd0.2Sm0.2Sr0.2Ba0.2)Co0.2Fe0.8O3−δ/SDC high-entropy oxide composite cathode. Mater. Lett. 2023, 338, 134029. [Google Scholar] [CrossRef]
- Azad, A.K.; Irvine, J.T.S. Synthesis, chemical stability and proton conductivity of the perovksites Ba(Ce,Zr)1−x Scx O3−δ. Solid State Ion 2007, 178, 635–640. [Google Scholar] [CrossRef]
- Radenahmad, N.; Afif, A.; Petra, M.I.; Rahman, S.M.H.; Eriksson, S.; Azad, A.K. High conductivity and high density proton conducting Ba1−xSrxCe0.5Zr0.35Y0.1Sm0.05O3−δ(x = 0.5, 0.7, 0.9, 1.0) perovskites for IT-SOFC. Int. J. Hydrogen Energy 2016, 41, 11832–11841. [Google Scholar] [CrossRef]
- Afif, A.; Radenahmad, N.; Lim, C.M.; Petra, M.I.; Islam, M.A.; Rahman, S.M.H.; Eriksson, S.; Azad, A.K. Structural study and proton conductivity in BaCe0.7Zr0.25−xYxZn0.05O3(x = 0.05, 0.1, 0.15, 0.2 & 0.25). Int. J. Hydrogen Energy 2016, 41, 11823–11831. [Google Scholar] [CrossRef]
- Rathore, S.S.; Biswas, S.; Fini, D.; Kulkarni, A.P.; Giddey, S. Direct ammonia solid-oxide fuel cells: A review of progress and prospects. Int. J. Hydrogen Energy 2021, 46, 35365–35384. [Google Scholar] [CrossRef]
- Kaur, P.; Singh, K. Review of perovskite-structure related cathode materials for solid oxide fuel cells. Ceram. Int. 2020, 46, 5521–5535. [Google Scholar] [CrossRef]
- Aziz, A.J.A.; Baharuddin, N.A.; Somalu, M.R.; Muchtar, A. Review of composite cathodes for intermediate-temperature solid oxide fuel cell applications. Ceram. Int. 2020, 46, 23314–23325. [Google Scholar] [CrossRef]
- Gao, Y.; Huang, X.; Yuan, M.; Gao, J.; Wang, Z.; Abdalla, A.M.; Azad, A.K.; Xu, L.; Lv, Z.; Wei, B. A SrCo0.9Ta0.1O3-δ derived medium-entropy cathode with superior CO2 poisoning tolerance for solid oxide fuel cells. J. Power Sources 2022, 540, 231661. [Google Scholar] [CrossRef]
- Cowin, P.I.; Petit, C.T.G.; Lan, R.; Irvine, J.T.S.; Tao, S. Recent Progress in the Development of Anode Materials for Solid Oxide Fuel Cells. Adv. Energy Mater. 2011, 1, 314–332. [Google Scholar] [CrossRef]
- Hua, B.; Yan, N.; Li, M.; Sun, Y.F.; Zhang, Y.Q.; Li, J.; Etsell, T.; Sarkar, P.; Luo, J.L. Anode-Engineered Protonic Ceramic Fuel Cell with Excellent Performance and Fuel Compatibility. Adv. Mater. 2016, 28, 8922–8926. [Google Scholar] [CrossRef] [PubMed]
- Boukamp, B.A. Fuel cells: The amazing perovskite anode. Nat. Mater. 2003, 2, 294–296. [Google Scholar] [CrossRef]
- Prakash, B.S.; Kumar, S.S.; Aruna, S.T. Properties and development of Ni/YSZ as an anode material in solid oxide fuel cell: A review. Renew. Sustain. Energy Rev. 2014, 36, 149–179. [Google Scholar] [CrossRef]
- Pihlatie, M.; Kaiser, A. Mogensen, Redox stability of SOFC: Thermal analysis of Ni–YSZ composites. Solid State Ion. 2009, 180, 1100–1112. [Google Scholar] [CrossRef]
- Boldrin, P.; Ruiz-Trejo, E.; Mermelstein, J.; Menéndez, J.M.B.; Ramı, T.; Brandon, N.P. Strategies for Carbon and Sulfur Tolerant Solid Oxide Fuel Cell Materials, Incorporating Lessons from Heterogeneous Catalysis. Chem. Rev. 2016, 116, 13633–13684. [Google Scholar] [CrossRef]
- Rafique, M.; Nawaz, H.; Rafique, M.S.; Tahir, M.B.; Nabi, G.; Khalid, N.R. Material and method selection for efficient solid oxide fuel cell anode: Recent advancements and reviews. Int. J. Energy Res. 2019, 43, 2423–2446. [Google Scholar] [CrossRef]
- Tao, S.; Irvine, J.T.S. A redox-stable efficient anode for solid-oxide fuel cells. Nat. Mater. 2003, 2, 320–323. [Google Scholar] [CrossRef]
- Ghosh, A.; Azad, A.; Irvine, J.T. Study of Ga Doped LSCM as an Anode for SOFC. ECS Trans. 2011, 35, 1337–1343. [Google Scholar] [CrossRef]
- Yang, J.; Akagi, T.; Okanishi, T.; Muroyama, H.; Matsui, T.; Eguchi, K. Catalytic Influence of Oxide Component in Ni-Based Cermet Anodes for Ammonia-Fueled Solid Oxide Fuel Cells. Fuel Cells 2015, 15, 390–397. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C. Ammonia-fed solid oxide fuel cells for power generation-A review. Int. J. Energy Res. 2009, 33, 943–959. [Google Scholar] [CrossRef]
- Ghorbani, B.; Vijayaraghavan, K. A review study on software-based modeling of hydrogen-fueled solid oxide fuel cells. Int. J. Hydrogen Energy 2019, 44, 13700–13727. [Google Scholar] [CrossRef]
- Faheem, H.H.; Abbas, S.Z.; Tabish, A.N.; Fan, L.; Maqbool, F. A review on mathematical modelling of Direct Internal Reforming- Solid Oxide Fuel Cells. J. Power Sources 2022, 520, 230857. [Google Scholar] [CrossRef]
- Ni, M. Thermo-electrochemical modeling of ammonia-fueled solid oxide fuel cells considering ammonia thermal decomposition in the anode. Int. J. Hydrogen Energy 2011, 36, 3153–3166. [Google Scholar] [CrossRef]
- Wojcik, A.; Middleton, H.; Damopoulos, I.; Van Herle, J. Ammonia as a fuel in solid oxide fuel cells. J. Power Sources 2003, 118, 342–348. [Google Scholar] [CrossRef]
- Ilbas, M.; Kumuk, B.; Alemu, M.A.; Arslan, B. Numerical investigation of a direct ammonia tubular solid oxide fuel cell in comparison with hydrogen. Int. J. Hydrogen Energy 2020, 45, 35108–35117. [Google Scholar] [CrossRef]
- Gould, H.; Tobochnik, J.; Christian, W. An Introduction to Computer Simulation Methods: Applications to Physical Systems (third edition). Am. J. Phys. 2006, 74, 652–653. [Google Scholar] [CrossRef]
- Chen, Y.; Qin, W. A variable range step technique for propagation predictions over large irregular terrain using the Fourier split-step parabolic equation. In Proceedings of the 2015 Asia-Pacific Microwave Conference (APMC), Nanjing, China, 6–9 December 2015; Volume 2, pp. 1–3. [Google Scholar] [CrossRef]
- Li, C.Z.; Wang, S. The finite volume method and application in combinations. J. Comput. Appl. Math. 1999, 106, 21–53. [Google Scholar] [CrossRef]
- Yahya, A.; Naji, H.; Dhahri, H. A lattice Boltzmann analysis of the performance and mass transport of a solid oxide fuel cell with a partially obstructed anode flow channel. Fuel 2023, 334, 126537. [Google Scholar] [CrossRef]
- Adami, S.; Hu, Y.X.; Adams, A.N. A generalized wall boundary condition for smoothed particle hydrodynamics. J. Comput. Phys. 2012, 231, 7057–7075. [Google Scholar] [CrossRef]
- Fahmy, A.M. A new boundary element algorithm for modeling and simulation of nonlinear thermal stresses in micropolar FGA composites with temperature-dependent properties. Adv. Model. Simul. Eng. Sci 2021, 8, 6. [Google Scholar] [CrossRef]
- Pazirofteh, M.; Dehghani, M.; Niazi, S.; Mohammadi, H.A.; Asghari, M. Molecular dynamics simulation and Monte Carlo study of transport and structural properties of PEBA 1657 and 2533 membranes modified by functionalized POSS-PEG material. J. Mol. Liq. 2017, 241, 646–653. [Google Scholar] [CrossRef]
- Yu, P.; Ren, Z.; Chen, Z.; Bordas, A.P.S. A multiscale finite element model for prediction of tensile strength of concrete. Finite Elem. Anal. Des. 2023, 215, 103877. [Google Scholar] [CrossRef]
- Gooneie, A.; Schuschnigg, S.; Holzer, C. A Review of Multiscale Computational Methods in Polymeric Materials. Polymers 2017, 9, 16. [Google Scholar] [CrossRef]
- Ezzat, M.F.; Dincer, I. Energy and exergy analyses of a novel ammonia combined power plant operating with gas turbine and solid oxide fuel cell systems. Energy 2020, 194, 116750. [Google Scholar] [CrossRef]
- Osman, N.; Mazlan, N.A.; Affandi, N.S.M.; Mazlan, N.W.; Jani, A.M.M. Optimization of electrolyte performance by tailoring the structure and morphology of Ba(Ce,Zr)O3 ceramics with different types of surfactants. Ceram. Int. 2020, 46, 27401–27409. [Google Scholar] [CrossRef]
- Ni, M.; Leung, D.; Leung, M. Mathematical modeling of ammonia-fed solid oxide fuel cells with different electrolytes. Int. J. Hydrogen Energy 2008, 33, 5765–5772. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, W. Direct ammonia solid oxide fuel cell based on thin proton-conducting electrolyte. J. Power Sources 2008, 179, 92–95. [Google Scholar] [CrossRef]
- Ni, M.; Shao, Z.; Chan, K. Modeling of Proton-Conducting Solid Oxide Fuel Cells Fueled with Syngas. Energies 2014, 7, 4381–4396. [Google Scholar] [CrossRef]
- Kalinci, Y.; Dincer, I. Analysis and performance assessment of NH3 and H2 fed SOFC with proton-conducting electrolyte. Int. J. Hydrogen Energy 2018, 43, 5795–5807. [Google Scholar] [CrossRef]
- Kumuk, B.; Alemu, M.A.; Ilbas, M. Investigation of the effect of ion transition type on performance in solid oxide fuel cells fueled hydrogen and coal gas. Int. J. Hydrogen Energy 2022, 47, 3409–3415. [Google Scholar] [CrossRef]
- Cai, W.; Yuan, J.; Zheng, Q.; Yu, W.; Yin, Z.; Zhang, Z.; Pei, Y.; Li, S. Numerical Investigation of Heat/Flow Transfer and Thermal Stress in an Anode-Supported Planar SOFC. Crystals 2022, 12, 1697. [Google Scholar] [CrossRef]
- Kümük, B.; İlbaş, M. Coal gas fuel utilization effects on electrolyte supported solide oxide fuel cell performance. Int. J. Hydrogen Energy 2021, 46, 29523–29528. [Google Scholar] [CrossRef]
- Luo, Y.; Shi, Y.; Liao, S.; Chen, C.; Zhan, Y.; Au, C.-T.; Jiang, L. Coupling ammonia catalytic decomposition and electrochemical oxidation for solid oxide fuel cells: A model based on elementary reaction kinetics. J. Power Sources 2019, 423, 125–136. [Google Scholar] [CrossRef]
- Cimen, F.M.; Kumuk, B.; Ilbas, M. Simulation of hydrogen and coal gas fueled flat-tubular solid oxide fuel cell (FT-SOFC). Int. J. Hydrogen Energy 2022, 47, 3429–3436. [Google Scholar] [CrossRef]
- Ilbas, M.; Karyeyen, S.; Cimen, F.M. Numerical investigation of combustion and flame characteristics for a model solid oxide fuel cell performance improvement. Fuel 2022, 322, 124188. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, L.; Espinoza, M.; Li, T.; Andersson, M. Numerical simulation of solid oxide fuel cells comparing different electrochemical kinetics. Int. J. Energy Res. 2021, 45, 12980–12995. [Google Scholar] [CrossRef]
- Hajimolana, S.A.; Hussain, M.A.; WanDaud, W.M.A. Comparative study on the performance of a tubular solid oxide fuel cell fuelled by ammonia and hydrogen. In Proceedings of the CHEMECA Annual Conference 2011, Sydney, Australia, 18–21 September 2011. [Google Scholar]
- Milewski, J.; Szczęśniak, A.; Szabłowski, Ł. A proton conducting solid oxide fuel cell—Implementation of the reduced order model in available software and verification based on experimental data. J. Power Sources 2021, 502, 229948. [Google Scholar] [CrossRef]
- Ishak, F.; Dincer, I.; Zamfirescu, C. Energy and exergy analyses of direct ammonia solid oxide fuel cell integrated with gas turbine power cycle. J. Power Sources 2012, 212, 73–85. [Google Scholar] [CrossRef]
- Alsarraf, J.; Alnaqi, A.A.; Al-Rashed, A.A.A.A. Thermodynamic modeling and exergy investigation of a hydrogen-based integrated system consisting of SOFC and CO2 capture option. Int. J. Hydrogen Energy 2022, 47, 26654–26664. [Google Scholar] [CrossRef]
- Huang, Y.; Lin, Q.; Liu, H.; Ni, M.; Zhang, X. Evaluation of the waste heat and residual fuel from the solid oxide fuel cell and system power optimization. Int. J. Heat Mass Transf. 2017, 115, 1166–1173. [Google Scholar] [CrossRef]
- Perna, A.; Minutillo, M.; Jannelli, E.; Cigolotti, V.; Nam, S.W.; Han, J. Design and performance assessment of a combined heat, hydrogen and power (CHHP) system based on ammonia-fueled SOFC. Appl. Energy 2018, 231, 1216–1229. [Google Scholar] [CrossRef]
- Zitouni, B.; Andreadis, G.M.; Hocine, B.M.; Hafsia, A.; Djamel, H.; Mostefa, Z. Two-dimensional numerical study of temperature field in an anode supported planar SOFC: Effect of the chemical reaction. Int. J. Hydrogen Energy 2011, 36, 4228–4235. [Google Scholar] [CrossRef]
- Al-Khori, K.; Bicer, Y.; Boulfrad, S.; Koç, M. Techno-economic and environmental assessment of integrating SOFC with a conventional steam and power system in a natural gas processing plant. Int. J. Hydrogen Energy 2019, 44, 29604–29617. [Google Scholar] [CrossRef]






| Fuel Cell | Temperature (°C) | Charge Carrier | Efficiency (%) | Competent Power | Application | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|
| Alkaline fuel cell (AFC) | 50–90 | OH− | 50–70 | 10–100 KW | Aerospace applications | Less operating temperature and high efficiency | Toxic byproduct, illiberal to CO2 in impure H2 and air, corrosion, and expensive |
| Phosphoric acid fuel cell (PAFC) | 175–220 | H+ | 40–45 | 10 MW | Energy needed in hospitals, schools, and offices | High efficiency and liberal to impure H2 | Corrosion and sulfur poisoning, low power density |
| Molten carbonate fuel cell (MCFC) | 600–650 | CO3 2− | 50–60 | 100 MW | Large-scale distributed commercial generation | High efficiency | Sulfur poisoning, electrolyte instability, and corrosion |
| Direct methanol fuel cell (DMFC) | 50–120 | H+ | 25–40 | 100 KW to 1 MW | Mobile phones, laptops, and battery chargers | High power density, low temperature, and no reforming | Methanol crossover and byproduct poisoning, low efficiency |
| Polymer electrolyte membrane fuel cell (PEMFC) | 60–100 | H+ | 40–50 | 100 W to 500 KW | Vehicles | Low temperature and high power density | Illiberal to CO in impure H2 and expensive |
| Solid oxide fuel cell (SOFC) | 800–1000 | O2− | 50–60 | 100 MW | Residential and Factory | Direct fossil fuel and high efficiency | Thermal stress failure, high temperature, and sulfur poisoning |
| Protonic ceramic fuel cell (PCFC) | 450–750 | H+ | >50 | 25 KW | Heavy-duty trucking and remote power applications | Lower degradation, high conductivity, and less working temperature | More appropriate electrolyte and electrode materials needed and complex fabrication |
| Description of Model | Analysis of Model |
|---|---|
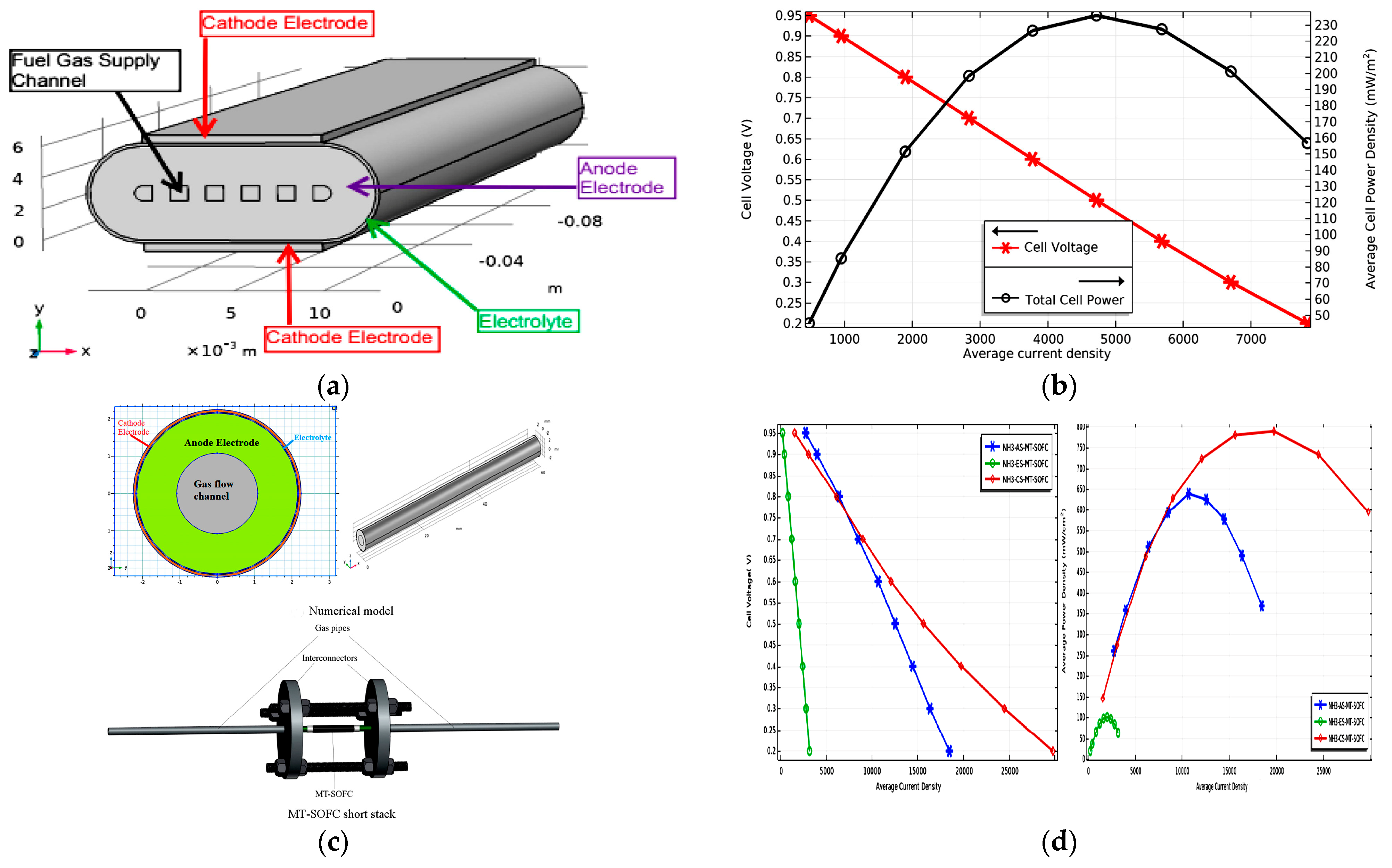
| Anode-supported flat tubular SOFC using ammonia fuel [10] | A three-dimensional modeling approach was explored for an anode-supported flat tubular SOFC. Comparative analysis revealed that the performance of the anode-supported flat tubular SOFC exceeded that of the tubular SOFC at high temperatures. However, no simulations were conducted for intermediate temperatures, which are of utmost importance for minimizing cell degradation. Moreover, no findings are available regarding cell performance when cathode concentration remains constant. |
| A thin film SOFC performance using ammonia fuel [11] | An intermediate temperature (923 K) was considered for this experiment, which found the highest density (1330 mW/cm2). However, the influence of active catalysts with suitable diffusion channels within the SOFC was not taken into consideration. The optimization of the diffusion process has the potential to substantially enhance the performance of ammonia-fueled SOFCs, enabling them to attain a performance level comparable to that of hydrogen-fueled cells. |
| A thermo-electrochemical model was developed for ammonia-fueled planar SOFC [46] | SOFCs typically exhibit higher electric potential. However, in this specific model, achieving higher electric potential at intermediate temperatures was not possible due to the requirement for a higher temperature gradient. Higher temperature also causes thermal stress in SOFCs. Furthermore, the model did not definitively determine whether a low inlet velocity or a high inlet velocity is more appropriate for the ammonia-fueled SOFC. |
| Electrolyte and electrode-supported tubular SOFC was discussed for fuel comparison [48] | Cathode-supported ammonia-fueled SOFCs revealed better performance than anode and electrolyte-supported ones in high temperatures. But, an intermediate temperature was not considered for the modeling. Furthermore, in cases when the anode is positioned within the inner part of the tube, there is no conclusive evidence regarding high concentration losses due to low diffusion at the cathode. |
| Method | Description | Advantages | Disadvantages |
|---|---|---|---|
| Finite Difference Method (FDM) | The FDM is a straightforward approach to discretizing the governing equations on a grid and approximate derivatives using finite differences. It is relatively simple to implement and suitable for regular geometries. The FDM is often used for steady-state and transient simulations of SOFCs. | The notable strength of the finite difference method lies in its adaptability to represent intricate geometries and diverse material characteristics. This adaptability renders it applicable across a broad spectrum of scenarios, enabling the treatment of varied problems and systems. Furthermore, the finite difference method facilitates the computation of transfer functions and supports acoustic analysis pertaining to intricate structural configurations [50]. | The major drawback is the computational strength of the calculations, especially when fine sampling is required on both horizontal and vertical grids. This can lead to enhanced computational time and resource requirements, particularly for large-scale problems. As a consequence, the finite difference method might not be the most efficient choice for certain applications where computational efficiency is more important [50]. |
| Finite Volume Method (FVM) | The FVM involves discretizing the domain into control volumes and solving the conservation equations for mass, momentum, energy, and species within each volume. The FVM is known for its conservation properties and is widely used for modeling fluid flow and heat transfer in SOFCs. | The Finite Volume Method inherently conserves quantities within control volumes, making it suitable for problems involving conservation laws. This ensures an accurate representation of physical processes. It naturally handles source terms within control volumes, ensuring proper incorporation of external influences or internal generation of quantities. For example, the FVM has become very promising for solving elliptic boundary value problems where it needs to follow the conservation of law principle [51]. | Implementing the FVM can be complex, especially for problems involving non-orthogonal grids or complex boundary conditions. Proper intervention of boundary conditions can be challenging and may require specialized techniques [51]. |
| Finite Element Method (FEM) | The FEM is versatile and can handle complex geometries with ease. It subdivides the domain into finite elements, allowing for an accurate representation of irregular shapes. The FEM is often used for structural analysis and can also be extended to solve the fluid flow and heat transfer equations. | The FEM is well suited for handling complex geometries and irregular domains. It can efficiently model structures with intricate shapes or irregular boundaries. It can achieve high accuracy by using higher-order elements, allowing for better representation of curved or intricate solutions. The FEM naturally integrates boundary conditions, including essential (Dirichlet) and natural (Neumann) boundary conditions, making it suitable for problems involving external constraints [12]. | Solution accuracy can be sensitive to the quality and density of the mesh. Fine meshes can improve accuracy but may lead to higher computational costs. Some problems can result in ill-conditioned matrices, affecting the stability and convergence of the numerical solution. For certain time-dependent problems, the FEM might require substantial computational resources due to the transient nature of the solution [48]. |
| Lattice Boltzmann Method (LBM) | The LBM is a mesoscopic method based on the kinetic theory of gases. It is particularly suitable for simulating complex fluid flows with porous media, such as the electrodes in SOFCs. The LBM can handle multiphase and multiscale phenomena effectively. | The LBM is well suited for high-performance computing architectures, allowing for efficient simulations of large-scale problems. Boundary conditions can be implemented naturally by incorporating bounce-back schemes, making it straightforward to handle various flow conditions and geometries. It can handle irregular and complex geometries with relative ease, as the method’s discrete lattice structure simplifies boundary treatment and mesh generation [52]. | While the LBM has seen significant development and application, it might not be as mature as other established methods like finite volume or finite element methods, particularly for certain types of problems. It might face challenges in accurately simulating flows with high Reynolds numbers or strong turbulence, as the lattice structure and inherent viscosity can impose limitations on turbulent flow modeling. Achieving high accuracy in LBM simulations often requires fine spatial and temporal resolutions, which can lead to increased computational costs [52]. |
| Smoothed Particle Hydrodynamics (SPH) | SPH is a Lagrangian particle-based method used for simulating fluid dynamics. It is well suited for problems involving large deformations and fluid–structure interactions. SPH can be applied to simulate fluid flow and heat transfer within SOFCs. | SPH is well suited for simulations of complex geometries, as it does not require structured grids. This makes it particularly useful for problems like fluid–structure interactions and multiphase flows, where the boundaries are complex and may change over time. It can handle free surface simulations, making it useful for simulating flows with distinct interfaces between different phases, such as water and air or liquid droplets in a gas environment. Since SPH is a Lagrangian method, meaning that each particle moves with the fluid, and its motion is tracked individually. This is favorable for capturing fluid movement and deformation accurately, especially in cases where Eulerian methods might struggle, such as highly turbulent or chaotic flows [53]. | Properly imposing boundary conditions in SPH simulations can be challenging. The smoothing kernels used in SPH can lead to inaccuracies near boundaries, which can affect the simulation results since SPH does not rely on structured grids, which can lead to some difficulties in interpolating properties between particles [53]. |
| Boundary Element Method (BEM) | The BEM is useful for problems with boundary-dominated phenomena, such as heat conduction through thin structures like SOFC electrolytes. It discretizes only the boundary of the domain, reducing the computational effort compared to volume-based methods. | The BEM solves problems on the boundary, resulting in lower-dimensional integrals compared to volume-based methods like the Finite Element Method (FEM). It is well equipped to handle potential singularities in the governing equations, such as when dealing with Laplace or Helmholtz problems, and is suitable for problems involving unbounded or semi-infinite domains, such as acoustic or electromagnetic scattering problems. It only requires discretizing the boundary, making it advantageous for such scenarios [54]. | The BEM lacks a traditional mass matrix like the FEM, making it less suitable for transient or dynamic problems where mass effects are substantial. Time-domain analysis can be more involved in the BEM. Mesh adaptation and refinement can also be challenging [54]. |
| Molecular Dynamics (MD) and Monte Carlo (MC) Methods | MD and MC methods are used at the molecular scale to study interactions between atoms and molecules. They can be employed to analyze surface reactions, adsorption, and diffusion of species on electrode surfaces and within the electrolyte. | MD simulates the time evolution of a system’s particles, providing insights into the dynamic behavior of molecules and their interactions. It captures temporal evolution, equilibration, and dynamic processes. It accurately conserves energy and momentum, obeying the laws of mechanics. This allows for the study of energy fluctuations and transport properties, which are essential for understanding thermodynamics. MC methods provide statistically accurate sampling of configuration space, enabling the calculation of thermodynamic properties. It does not have inherent time-step limitations, making it suitable for simulating processes with a wide range of timescales [55]. | MD simulations are typically limited to picoseconds to microseconds, restricting the study of slower processes or long-term phenomena. It depends on empirical force fields, which may have limitations in accurately describing certain molecular interactions. MC simulations are subject to systematic errors, including finite-size effects and discretization errors. It uses discrete moves to explore configuration space, which can limit the accuracy of sampling, particularly in high-dimensional spaces [55]. |
| Coupled Methods | Combining different numerical methods can provide more accurate and efficient solutions. For instance, coupling FEM with FVM or FDM can be used to model the coupled fluid flow, heat transfer, and electrochemical reactions in SOFCs. | Coupled methods enable multiscale simulations, where different simulation techniques are applied to different lengths or timescales. This is important for studying systems with multiple interacting processes occurring at different scales. It can enhance accuracy by incorporating higher-fidelity simulations for specific regions or components of a system and help to identify potential limitations in the individual methods [56]. | Coupled simulations can encounter numerical stability and convergence issues, especially when transferring information between different simulation domains or grids. Developing and maintaining coupled software can be complex and time-consuming [56]. |
| Hybrid Methods | Hybrid methods combine continuum-based approaches (such as FVM or FEM) with particle-based methods (such as SPH or LBM) to capture different lengths and timescales within the system. | Hybrid methods enable multiscale simulations, allowing the study of systems with multiple lengths and timescales. It can significantly reduce the overall computational cost compared to fully resolving the entire system with a single high-fidelity method [57]. | Hybrid methods frequently involve assumptions and approximations when transitioning between different methods. It is difficult to ensure accuracy at the interfaces and minimize errors [57]. |
| Software Package | Ammonia-Fueled SOFC | Hydrogen-Fueled SOFC | Hydrocarbon-Fueled SOFC | Advantages and Limitations of Software |
|---|---|---|---|---|
| COMSOL Multiphysics, version 5.3 | Anode-supported flat tubular 3D model analysis [10] | Thermal stress analysis on SOFC [65] | Inlet fuel flow analysis in SOFC [66] | COMSOL offers a wide range of benefits, including multiphysics simulation capabilities, a user-friendly interface, flexibility, and customization options. However, it also has limitations related to computational resources, learning curve, cost, model complexity, and post-processing challenges. |
| Ammonia usage analysis in the electrolyte-supported SOFC [48] | Ion transition analysis in SOFC [64] | Ion transition analysis in SOFC [64] | ||
| Analyzing the mechanistic model [67] | Flat tubular SOFC modeling [68] | Flat tubular SOFC modeling [68] | ||
| Analysis of the flame characteristics for performance improvement of the SOFC model [69] | ||||
| Analysis of the electrochemistry and electrochemical models for SOFC [70] | ||||
| MATLAB | Tubular SOFC performance analysis [71] | Tubular SOFC performance analysis [71] | Analysis of the reduced order H-SOFC [72] | MATLAB offers various numerical computing and data visualization capabilities, supported by an extensive library of functions and toolboxes. It has a user-friendly interface and a strong presence in academia and industry. However, it comes with limitations such as cost, performance for certain tasks, memory usage, its closed-source nature, and potential complexities in advanced topics and deployment. |
| Analysis of the integration of NH3-fueled SOFC with gas turbine [73] | Plant-based thermodynamic SOFC modeling analysis [74] | Waste heat and residual fuel composition analysis [75] | ||
| ANSYS | Torsion test, crack installation test, and creep behavior analysis [16] | ANSYS provides a wide range of simulation capabilities, accuracy, physics integration, customization options, and community support. However, it comes with limitations such as cost, learning curve, resource-intensive computations, model complexity, and potential deployment challenges. | ||
| FORTRAN | Analysis of the combined hydrogen, heat, and power system in SOFC [76] | Chemical reaction analysis on the SOFC temperature field [77] | Transport and reaction investigations in a 2D SOFC [62] | FORTRAN’s strengths lie in numerical performance, legacy code, efficient array operations, and parallel computing. However, it comes with limitations related to syntax complexity, standard libraries, modern language features, and memory management. |
| Engineering Equation Solver (EES) | One-dimensional H-SOFC analysis [63] | One-dimensional H-SOFC analysis [63] | Integrating SOFC with power and traditional steam [78] | The EES provides valuable capabilities for equation-based engineering problem solving, symbolic manipulation, and parametric studies. Its broad applicability to various engineering fields makes it a useful tool for engineers and scientists. However, it also comes with limitations related to its specific domain, advanced features, customization, and cost considerations. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, M.M.; Abdalla, A.M.; Omeiza, L.A.; Raj, V.; Afroze, S.; Reza, M.S.; Somalu, M.R.; Azad, A.K. Numerical Modeling of Ammonia-Fueled Protonic-Ion Conducting Electrolyte-Supported Solid Oxide Fuel Cell (H-SOFC): A Brief Review. Processes 2023, 11, 2728. https://doi.org/10.3390/pr11092728
Rahman MM, Abdalla AM, Omeiza LA, Raj V, Afroze S, Reza MS, Somalu MR, Azad AK. Numerical Modeling of Ammonia-Fueled Protonic-Ion Conducting Electrolyte-Supported Solid Oxide Fuel Cell (H-SOFC): A Brief Review. Processes. 2023; 11(9):2728. https://doi.org/10.3390/pr11092728
Chicago/Turabian StyleRahman, Md. Mosfiqur, Abdalla M. Abdalla, Lukman Ahmed Omeiza, Veena Raj, Shammya Afroze, Md. Sumon Reza, Mahendra Rao Somalu, and Abul K. Azad. 2023. "Numerical Modeling of Ammonia-Fueled Protonic-Ion Conducting Electrolyte-Supported Solid Oxide Fuel Cell (H-SOFC): A Brief Review" Processes 11, no. 9: 2728. https://doi.org/10.3390/pr11092728






