1. Introduction
The assessment of toxicity in water by simple, reliable and rapid test systems has been a crucial issue for environmental safety and protection. Due to their high sensitivity for toxicants, various bioassays based on bacteria, protozoa and algae are particularly suitable for rapid analyses. In practice, these bioassays are based on complex metabolic processes such as respiration, nitrification [
1,
2,
3,
4,
5,
6,
7,
8] and bioluminescence [
3,
9,
10,
11]. In addition, enzymatic processes such as the estimation of dehydrogenase activity [
12,
13] and the growth of bacteria in a pure or in mixed culture [
14,
15,
16,
17] have been used. As eukaryotic organisms, pure cultures of algae have been serving as reliable test organisms for bioassays [
18,
19]. All these tests aim at the determination of the toxicity data of pure chemicals and chemical mixtures in an aqueous environment. The luminescent bacteria test using
Vibrio fischeri, which is now designated as
Aliivibrio fischeri, is a widely accepted bacterial toxicity test and has been normalized by the ISO (International Organisation for Standardization) [
11]. It is predominantly used to determine the toxicity of wastewaters, aqueous eluates and leachates from waste deposits [
20,
21]. Furthermore,
Vibrio fischeri can also be used for the determination of toxicity of gases such as benzene, trichloroethylene and acetone when the luminous bacteria are entrapped in a complex membrane [
22].
Additionally, the luminescent bacteria test proved to be a reliable test for the determination of toxicity for pure chemicals, chemical mixtures and wastewaters [
3,
20,
23,
24,
25,
26,
27,
28]. For this purpose, often test kits using conserved, freeze-dried
Vibrio fischeri cells are used, but it is also possible to use freshly prepared cells or cells which have been frozen [
11]. On the other hand, certain problems have often arisen when using frozen cells. When reactivating these cells, there can be variations in viability which can cause unreliable results in a toxicity test.
For this reason, attempts were made to further improve the cultivation of Vibrio fischeri, as well as the freezing and reactivation process, in order to create an improved ISO 11348 test method. In detail, the cultivation medium and the reactivation medium should be optimized. Additionally, the duration of the conservation period for the frozen bacteria should be enhanced. Moreover, the reliability of the overall improved procedure should be evaluated with reference compounds like 3.5- dichlorophenol, as well as zinc (II) and chromium (VI) ions. A new modified method would enable more laboratories to use the Vibrio fischeri test as an important screening test without being dependent on commercially available test kits.
Besides the luminescence of
Vibrio fischeri, the respiration activity of activated sludge bacteria can be used for setting up a reliable test system for measuring bacterial toxicity. The respiration inhibition test with activated sludge is another widely used and reliable test system for the evaluation of toxicity, and it has been internationally standardized [
7]. This test is generally performed at incubation times of 30 and 180 min in order to determine acute toxicity. However, there is also a prolonged test version with an incubation time of 27 h, intended to give a hint to the subchronic or chronic inhibitory effects of toxicants. This prolonged test system also proved to show a higher sensitivity for 3.5-dichlorophenol [
1].
In this study, the prolonged test system should be examined in further detail to evaluate the toxicity of further different reference compounds, such as dichlorophenols and also 3.4-dichloroaniline, to give more insight into the usability of this test system modification. The principal aim of this investigation was to establish the prolonged test system as an equivalent method to the established short-term methods to achieve a higher versatility of testing. This modification could also be integrated in the appropriate ISO and OECD (Organization for Economic Cooperation and Development) protocols to further update standardized test methods.
In general, ISO and OECD test methods play a crucial role when results from toxicity tests are taken to make hazard or risk assessments for certain chemicals or chemical mixtures. Therefore, a regular update of these test methods and research in this field are of great importance.
4. Discussion
Concerning the luminescent bacteria test,
Vibrio fischeri was cultivated in the modified growth medium in which high cell concentrations with high luminescence could be achieved. These cells were stored by freezing at −80 °C for up to seven weeks and, after reactivation, the EC50 values for 3.5-dichlorophenol were determined. The EC50 values obtained were in the range of 3.5–3.6 mgL
−1, even at different incubation times. These data are in good accordance with data formerly published [
3,
11,
38]. The toxicity of chlorinated phenolic compounds has been studied before [
3], and the EC50 data all lie in the range of 1–7 mL
−1, therefore being suitable reference compounds for this test system. In contrast, the EC50 values for Cr (VI) cations were clearly dependent on the incubation time and were in the range of 2.65 mgL
−1 (30 min incubation time). Therefore, they were in good accordance to data already published [
11,
38]. Also, the toxicity data for Zn (II) cations were comparable to formerly published data [
11,
38]. On the other hand, toxicity data can also vary in a wide range, as indicated in the literature [
38]. Influencing factors can be the composition of the growth medium, the cultivation and the storage method used. It is also possible that ionic compounds like chromium (VI) can form complexes, precipitate or be reduced [
39,
40,
41,
42]. These chemical reactions can influence the bioavailability of the compound and therefore have an effect on determined EC values. For this reason, organic compounds like chlorinated phenols, which show a high chemical stability impaired with a defined toxicity, seem to be ideal reference substances.
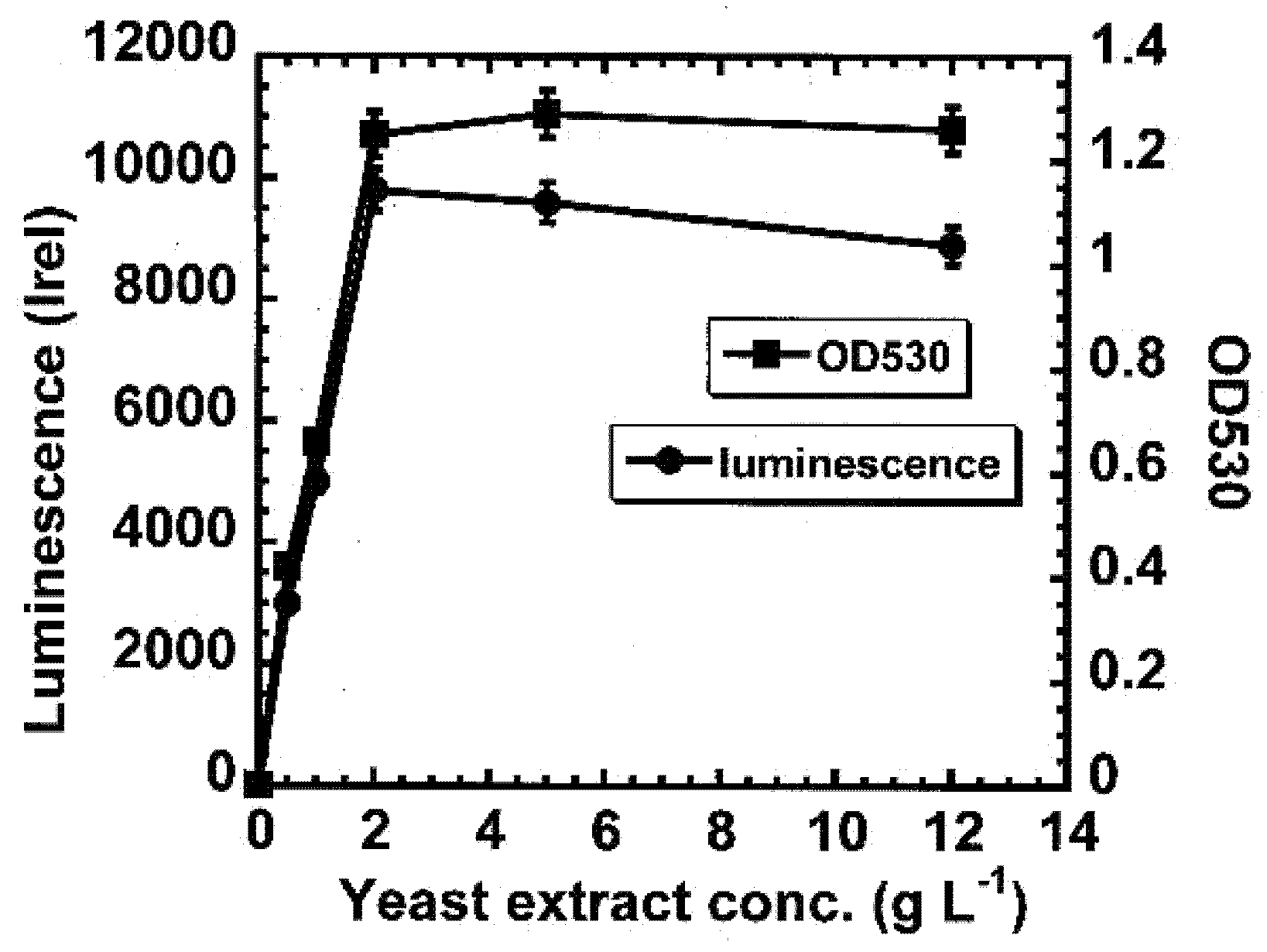
The new cultivation medium with an optimized yeast extract, magnesium and calcium content was very reliable for cultivation processes. The modified reactivation medium was suitable when testing organic test substances but is not recommended for testing inorganic compounds, which can precipitate with phosphate. The storage of Vibrio fischeri bacteria is now possible up to periods of seven weeks at −80 °C, whereas ISO 11348 Part 1 strictly recommends using only fresh bacteria for regular tests and thawed bacteria only for investigative and preliminary experiments. In our opinion, a prolonged storage period does not cause any loss of activity at all, in cases where it occurs at a temperature of −80 °C. Therefore, a modification of the ISO protocol concerning the cultivation and reactivation medium, as well as the storage period, should be taken into consideration.
Concerning the respiration inhibition test, the EC50 values obtained for 3.5-dichlorophenol in the long-term respiration inhibition test with an incubation time of 27 h are lower than data in other studies, with shorter incubation times of 0.5 to 3 h. Here, an EC50 of 6.35 mg L
−1 was measured, whereas an EC50 in the range of 10–60 mg L
−1 was reported in former publications [
43,
44,
45,
46,
47]. In the ISO 8192 protocol, a ring test is mentioned in which EC50 values of 9.8 mg L
−1 and 9.3 mgL
−1, with incubation times of 30 min and 180 min, respectively, were obtained. According to the ISO 8192 protocol, the EC50 should lie in the range of 2–25 mg L
−1. In a previous study, an EC50 of 6.3 m L
−1 in a long-term test system was published, whereas in 30 and 180 min tests, the EC50 values ranged from 5–9 mg L
−1 [
1], which is in good accordance with the data presented in this study.
The suitability of the long-term incubation test was also demonstrated, especially for 2.3-dichlorophenol, where the test system showed the highest sensitivity at an elevated incubation time of 27 h. For this reason, a modification of the OECD 209 and ISO 8192 protocols should be considered. In cases where a chemical compound has an EC50 value lower than 50 mg L−1 at an incubation time of 30 or 180 min, a retest at a prolonged incubation time of 27 h should be recommended in order to approve or reevaluate the data.
In general, a prolongation of the incubation time of bacteria with a toxicant should result in a higher toxicity, represented by lower EC values. In this study, this assumption could be stated for 2.3-dichlorophenol, 3.4-dichlorophenol and 3.4-dichloroaniline in the respiration inhibition test and for Cr (VI) in the luminescence bacteria test. In contrast, a prolonged incubation time had no effect on the EC50 values for 3.5-dichlorphenol in the respiration inhibition test. This may be due to the mode of action of the toxicant and the kinetics of the inhibition process. To summarize, tests with a prolonged incubation time provide data with a higher safety level, which might be useful for the risk assessment of chemicals and chemical mixtures.
From the data presented, it seems to be obvious that different dichlorophenols are appropriate reference compounds for bacterial toxicity tests. In
Table 6, EC50 values from different ISO and OECD normalized tests systems are summarized.
It seems to be obvious that the nitrification inhibition test is the most sensitive test system, followed by the luminescent bacteria test. The sewage bacteria growth inhibition test proved to be the most insensitive test system in several cases. The respiration inhibition test is located between the luminescent bacteria test and the sewage bacteria growth inhibition test.
Concerning the mode of an inhibitory action of dichlorophenols, the pH proved to have a severe influence, as shown in a dehydrogenase assay with activated sludge bacteria [
13]. There in tests at pH values in the range of 5.5 and 6.0 lower EC50 values were obtained compared to tests performed at pH values of 7.0 and 7.5, thus indicating a clear pH effect on toxicity.
It also has to be taken into account that toxicity data can be dependent on the source of the inoculum used [
14,
15]. A high variability of EC50 values can be explained by the choice of the inoculum used. It is obvious that sludge from a municipal wastewater treatment plant is adapted to easily biodegradable compounds, whereas sludge from an industrial wastewater treatment plant can often degrade even poorly biodegradable chemical compounds [
48]. Therefore, the sludge of an industrial wastewater treatment plant is adapted to a variety of chemical compounds and can more easily tolerate potential inhibitors.