Chlorine-Resistant Hollow Nanosphere-Like VOx/CeO2 Catalysts for Highly Selective and Stable Destruction of 1,2-Dichloroethane: Byproduct Inhibition and Reaction Mechanism
Abstract
1. Introduction
2. Materials and Methods
2.1. Catalyst Preparation
2.2. Catalyst Characterizations
2.3. Catalytic Activity
3. Results
3.1. Structural and Textural Properties
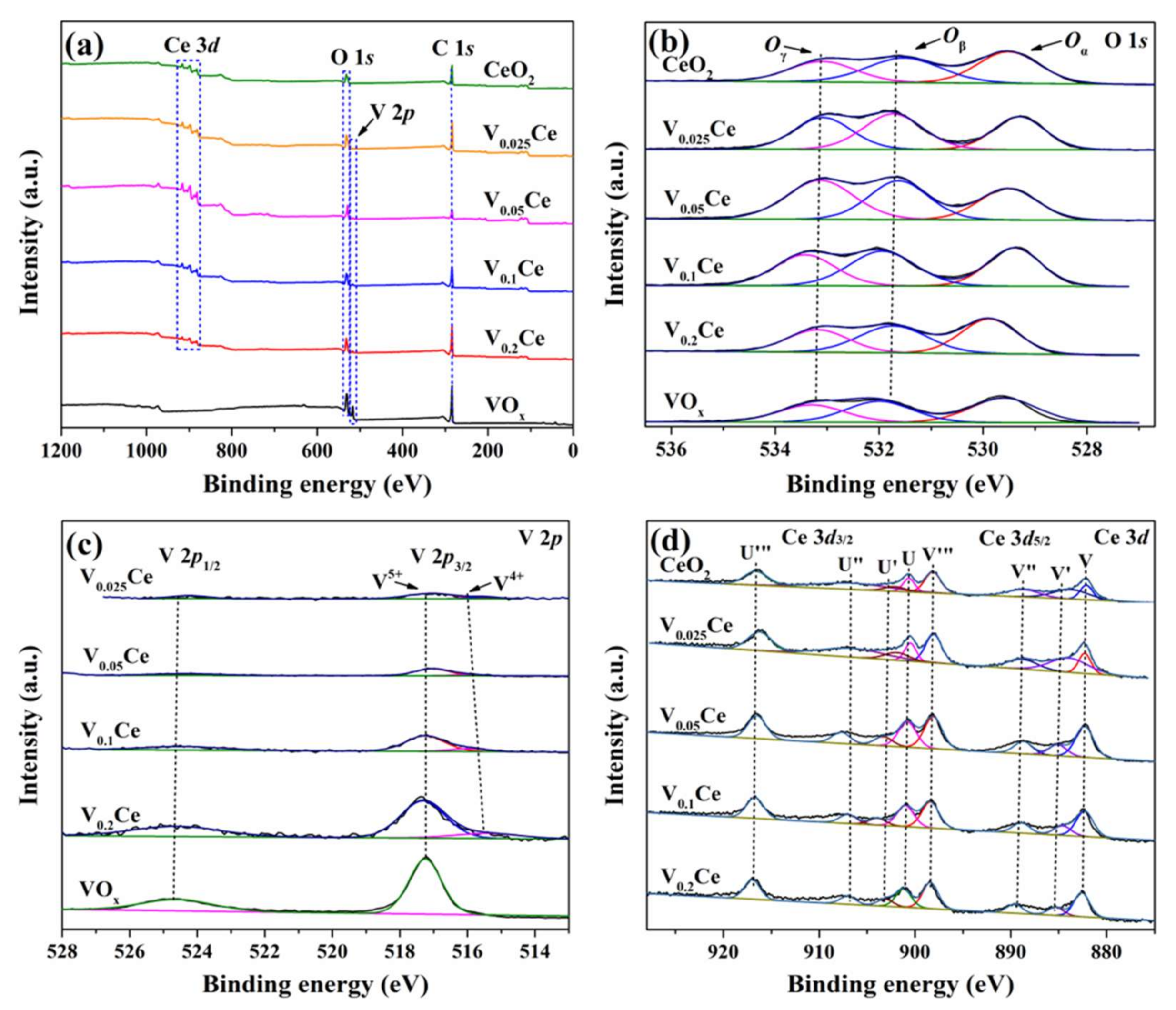
3.2. Surface Composition and Element Status
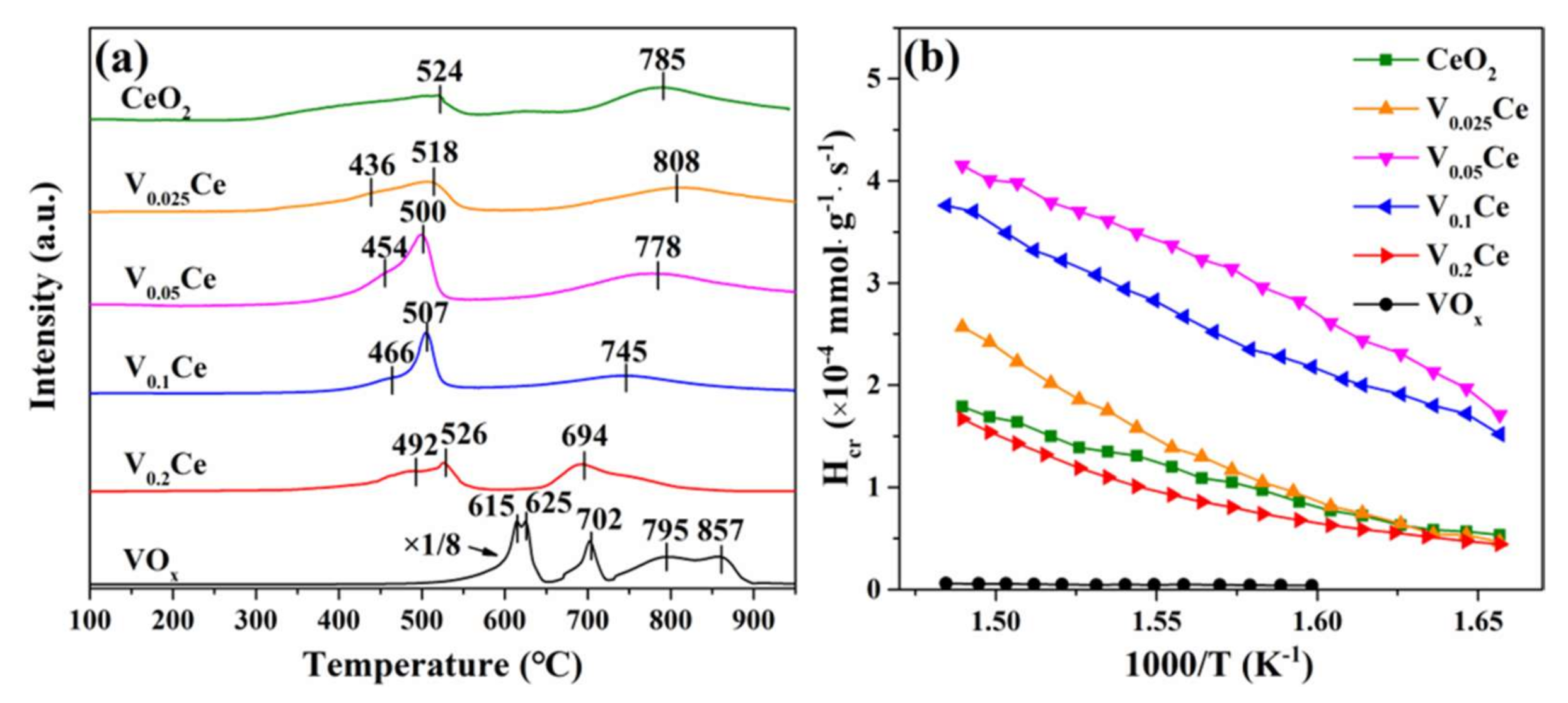
3.3. Reducibility and Oxygen Species Mobility
3.4. Surface Acidity
4. Discussion
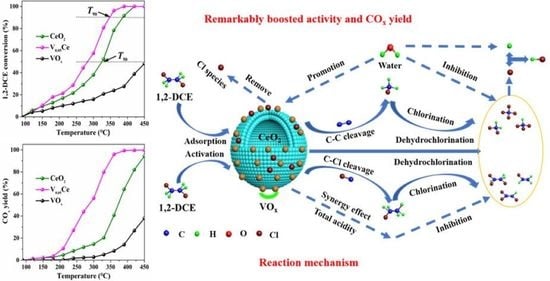
4.1. Catalytic Activity and Stability
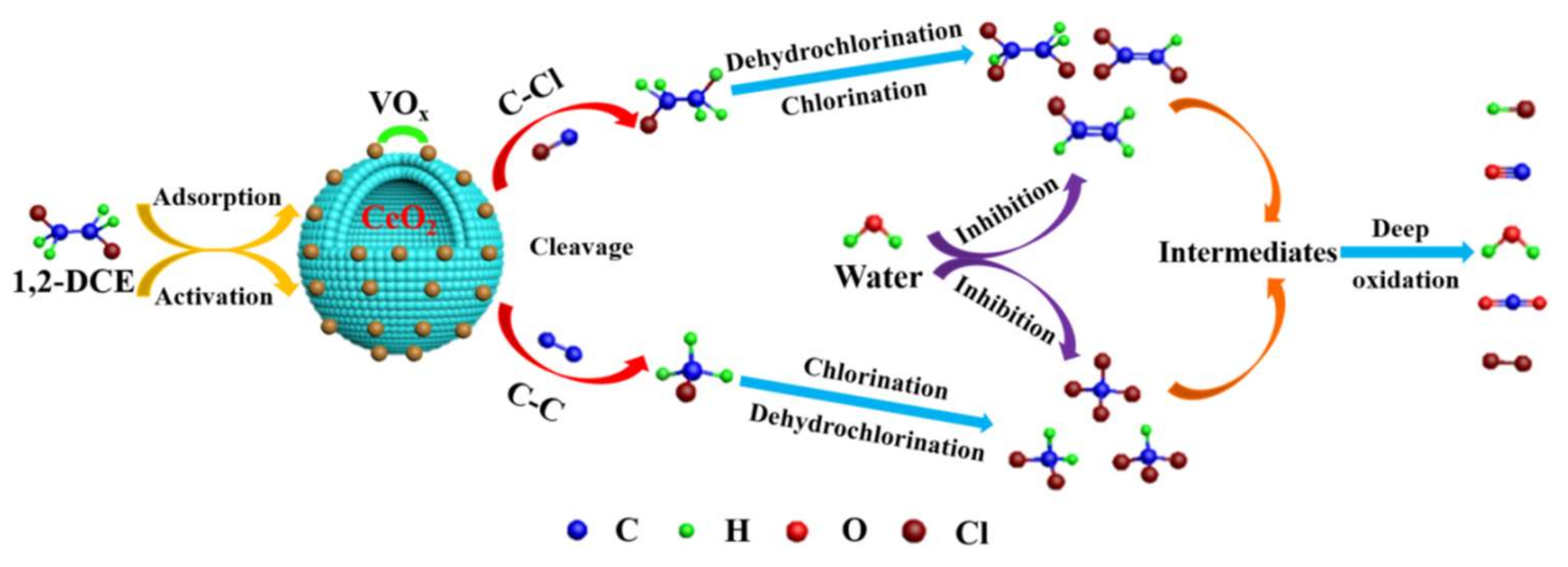
4.2. Chlorinated Intermediate Species Distribution and Proposed Reaction Mechanism
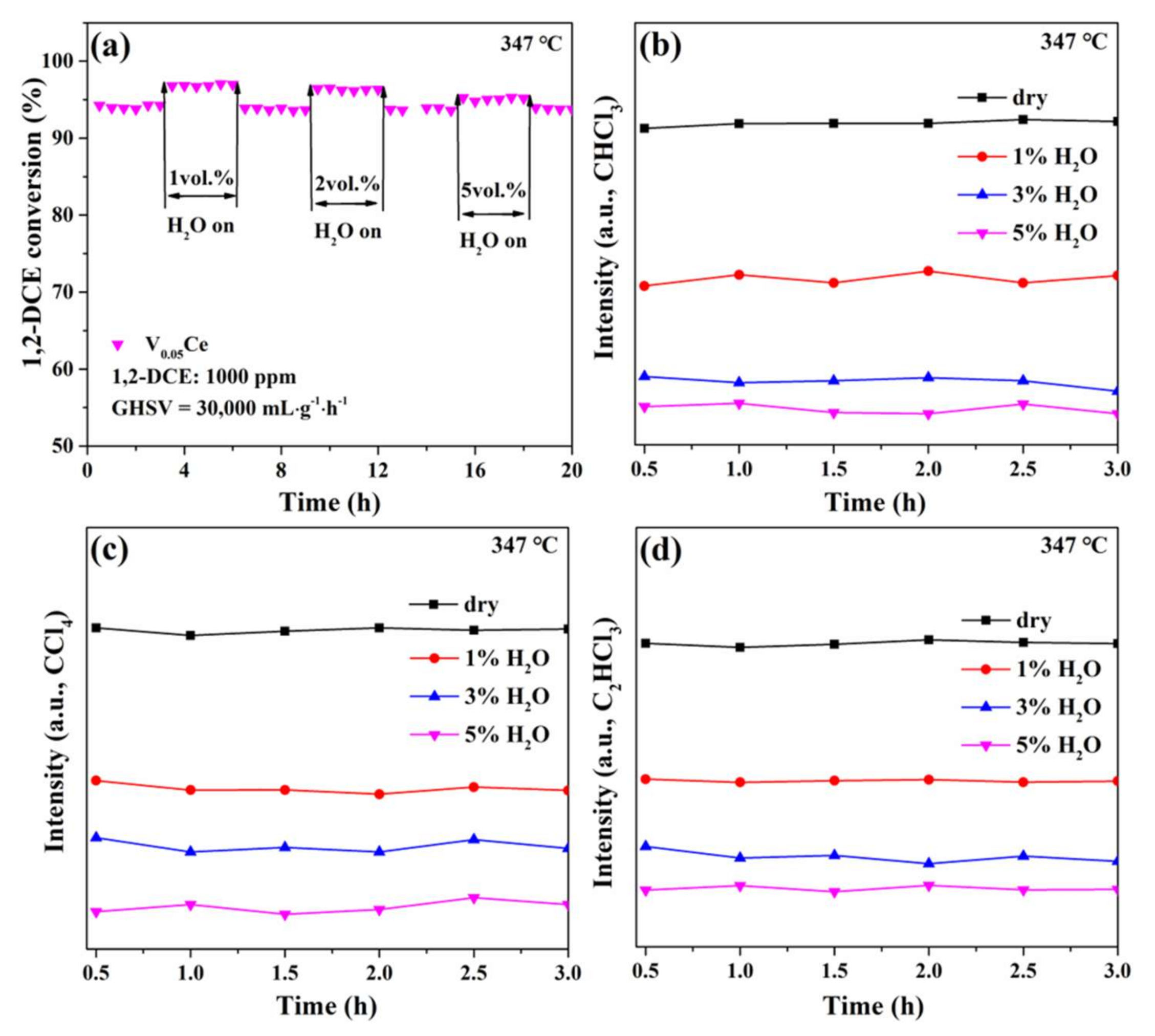
4.3. Effect of Water Vapor
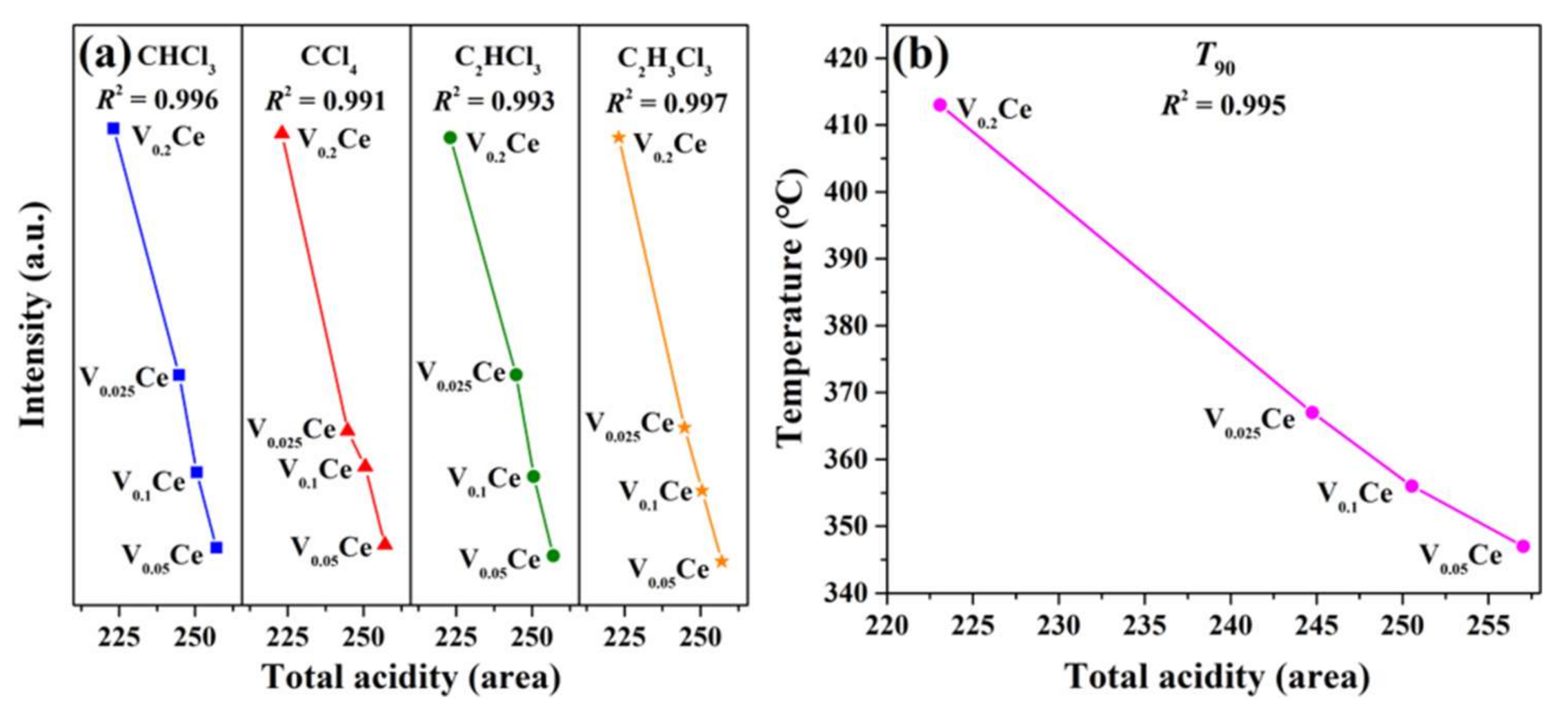
4.4. Key Factors Inhibiting the Formation of Chlorinated Intermediate Species
4.5. Characterization of the Used Catalysts
4.6. Key Factors for 1,2-DCE Oxidation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, P.; Yang, S.S.; Shi, Z.N.; Tao, F.; Guo, X.L.; Zhou, R.X. Accelerating effect of ZrO2 doping on catalytic performance and thermal stability of CeO2–CrOx mixed oxide for 1,2-dichloroethane elimination. Chem. Eng. J. 2016, 285, 544–553. [Google Scholar] [CrossRef]
- Gu, Y.F.; Cai, T.; Gao, X.H.; Xia, H.Q.; Sun, W.; Zhao, J.; Dai, Q.G.; Wang, X.Y. Catalytic combustion of chlorinated aromatics over WOx/CeO2 catalysts at low temperature. Appl. Catal. B Environ. 2019, 248, 264–276. [Google Scholar] [CrossRef]
- Lin, F.W.; Zhang, Z.M.; Li, N.; Yan, B.B.; He, C.; Hao, Z.P.; Chen, G.Y. How to achieve complete elimination of Cl-VOCs: A critical review on byproducts formation and inhibition strategies during catalytic oxidation. Chem. Eng. J. 2021, 404, 126534. [Google Scholar] [CrossRef]
- Lin, F.W.; Xiang, L.; Zhang, Z.M.; Li, N.; Yan, B.B.; He, C.; Hao, Z.P.; Chen, G.Y. Comprehensive review on catalytic degradation of Cl-VOCs under the practical application conditions. Crit. Rev. Environ. Sci. Technol. 2020, 1–45. [Google Scholar] [CrossRef]
- Jiao, Y.M.; Chen, X.; He, F.; Liu, S.T. Simple preparation of uniformly distributed mesoporous Cr/TiO2 microspheres for low-temperature catalytic combustion of chlorobenzene. Chem. Eng. J. 2019, 372, 107–117. [Google Scholar] [CrossRef]
- Ye, N.; Li, Y.; Yang, Z.; Zheng, J.; Zuo, S.F. Rare earth modified kaolin-based Cr2O3 catalysts for catalytic combustion of chlorobenzene. Appl. Catal. A Gen. 2019, 579, 44–51. [Google Scholar] [CrossRef]
- Dai, Q.G.; Bai, S.X.; Wang, Z.Y.; Wang, X.Y.; Lu, G.Z. Catalytic combustion of chlorobenzene over Ru-doped ceria catalysts. Appl. Catal. B Environ. 2012, 126, 64–75. [Google Scholar] [CrossRef]
- Topka, P.; Delaigle, R.; Kaluža, L.; Gaigneaux, E.M. Performance of platinum and gold catalysts supported on ceria–zirconia mixed oxide in the oxidation of chlorobenzene. Catal. Today 2015, 253, 172–177. [Google Scholar] [CrossRef]
- Romero-Sáez, M.; Divakar, D.; Aranzabal, A.; González-Velasco, J.R.; González-Marcos, J.A. Catalytic oxidation of trichloroethylene over Fe-ZSM-5: Influence of the preparation method on the iron species and the catalytic behavior. Appl. Catal. B Environ. 2016, 180, 210–218. [Google Scholar] [CrossRef]
- Su, J.; Liu, Y.; Yao, W.Y.; Wu, Z.B. Catalytic Combustion of Dichloromethane over HZSM-5-Supported Typical Transition Metal (Cr, Fe, and Cu) Oxide Catalysts: A Stability Study. J. Phys. Chem. C 2016, 120, 18046–18054. [Google Scholar] [CrossRef]
- Wang, X.Y.; Kang, Q.; Li, D. Catalytic combustion of chlorobenzene over MnOx–CeO2 mixed oxide catalysts. Appl. Catal. B Environ. 2009, 86, 166–175. [Google Scholar]
- Yang, Z.Y.; Yi, H.H.; Tang, X.L.; Zhao, S.Z.; Huang, Y.H.; Xie, X.Z.; Song, L.L.; Zhang, Y.Y. Study of reaction mechanism based on further promotion of low temperature degradation of toluene using nano-CeO2/Co3O4 under microwave radiation for cleaner production in spraying processing. J. Hazard. Mater. 2019, 373, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Gannoun, C.; Turki, A.; Kochkar, H.; Delaigle, R.; Eloy, P.; Ghorbel, A.; Gaigneaux, E.M. Elaboration and characterization of sulfated and unsulfated V2O5/TiO2 nanotubes catalysts for chlorobenzene total oxidation. Appl. Catal. B Environ. 2014, 147, 58–64. [Google Scholar] [CrossRef]
- Finocchio, E.; Ramis, G.; Busca, G. A study on catalytic combustion of chlorobenzenes. Catal. Today 2011, 169, 3–9. [Google Scholar] [CrossRef]
- Dai, Q.G.; Bai, S.X.; Li, H.; Liu, W.; Wang, X.Y.; Lu, G.Z. Catalytic total oxidation of 1,2-dichloroethane over highly dispersed vanadia supported on CeO2 nanobelts. Appl. Catal. B Environ. 2015, 168–169, 141–155. [Google Scholar] [CrossRef]
- Huang, H.; Gu, Y.F.; Zhao, J.; Wang, X.Y. Catalytic combustion of chlorobenzene over VOx/CeO2 catalysts. J. Catal. 2015, 326, 54–68. [Google Scholar] [CrossRef]
- Feng, Z.T.; Ren, Q.M.; Peng, R.S.; Mo, S.P.; Zhang, M.Y.; Fu, M.L.; Chen, L.M.; Ye, D.Q. Effect of CeO2 morphologies on toluene catalytic combustion. Catal. Today 2019, 332, 177–182. [Google Scholar] [CrossRef]
- Jiang, X.L.; Chen, R.Q.; Zhang, J.; Yu, L.; Xu, X. Synthesis of mono-dispersed ceria hollow nanospheres by a hydrothermal method. Micro Nano Lett. 2016, 11, 137–141. [Google Scholar] [CrossRef]
- Le, T.K.; Kang, M.; Kim, S.W. Morphology engineering, room-temperature photoluminescence behavior, and sunlight photocatalytic activity of V2O5nanostructures. Mater. Charact. 2019, 153, 52–59. [Google Scholar] [CrossRef]
- Khan, M.E.; Khan, M.M.; Cho, M.H. Ce3+-ion, Surface Oxygen Vacancy, and Visible Light-induced Photocatalytic Dye Degradation and Photocapacitive Performance of CeO2-Graphene Nanostructures. Sci. Rep. 2017, 7, 5928. [Google Scholar] [CrossRef]
- Le, T.K.; Kang, M.; Tran, V.T.; Kim, S.W. Relation of photoluminescence and sunlight photocatalytic activities of pure V2O5nanohollows and V2O5/RGO nanocomposites. Mat. Sci. Semico. Proc. 2019, 100, 159–166. [Google Scholar] [CrossRef]
- Li, Y.; Wei, Z.H.; Gao, F.; Kovarik, L.; Peden, C.H.F.; Wang, Y. Effects of CeO2 support facets on VOx/CeO2 catalysts in oxidative dehydrogenation of methanol. J. Catal. 2014, 315, 15–24. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, C.Z.; Li, J.H. Structure–activity relationship of VOx/CeO2 nanorod for NO removal with ammonia. Appl. Catal. B Environ. 2014, 144, 538–546. [Google Scholar] [CrossRef]
- Feng, X.B.; Tian, M.J.; He, C.; Li, L.; Shi, J.-W.; Yu, Y.K.; Cheng, J. Yolk-shell-like mesoporous CoCrOx with superior activity and chlorine resistance in dichloromethane destruction. Appl. Catal. B Environ. 2020, 264, 118493. [Google Scholar] [CrossRef]
- Kang, D.J.; Yu, X.L.; Ge, M.F. Morphology-dependent properties and adsorption performance of CeO2 for fluoride removal. Chem. Eng. J. 2017, 330, 36–43. [Google Scholar] [CrossRef]
- Jing, X.Y.; Zhang, Y.F.; Jiang, H.M.; Cheng, Y.; Xing, N.; Meng, C.G. Facile template-free fabrication of hierarchical V2O5 hollow spheres with excellent charge storage performance for symmetric and hybrid supercapacitor devices. J. Alloys Compd. 2018, 763, 180–191. [Google Scholar] [CrossRef]
- Ji, F.R.; Li, C.X.; Wang, J.Q.; Wang, J.; Shen, M.Q. New insights into the role of vanadia species as active sites for selective catalytic reduction of NO with ammonia over VOx/CeO2 catalysts. J. Rare Earths 2020, 38, 719–724. [Google Scholar] [CrossRef]
- Zhang, T.; Chang, H.Z.; Li, K.Z.; Peng, Y.; Li, X.; Li, J.H. Different exposed facets VOx/CeO2 catalysts for the selective catalytic reduction of NO with NH3. Chem. Eng. J. 2018, 349, 184–191. [Google Scholar] [CrossRef]
- Zeleke, M.A.; Kuo, D.-H. Synthesis and application of V2O5-CeO2 nanocomposite catalyst for enhanced degradation of methylene blue under visible light illumination. Chemosphere 2019, 235, 935–944. [Google Scholar] [CrossRef]
- Tian, M.J.; Guo, X.; Dong, R.; Guo, Z.; Shi, J.W.; Yu, Y.K.; Cheng, M.X.; Albilali, R.; He, C. Insight into the boosted catalytic performance and chlorine resistance of nanosphere-like meso-macroporous CrOx/MnCo3Ox for 1,2-dichloroethane destruction. Appl. Catal. B Environ. 2019, 259, 118018. [Google Scholar] [CrossRef]
- Xiao, P.; Zhu, J.J.; Li, H.L.; Jiang, W.; Wang, T.; Zhu, Y.J.; Zhao, Y.X.; Li, J.L. Effect of Textural Structure on the Catalytic Performance of LaCoO3 for CO Oxidation. ChemCatChem 2014, 6, 1774–1781. [Google Scholar] [CrossRef]
- Zhang, W.; Niu, X.Y.; Chen, L.Q.; Yuan, F.L.; Zhu, Y.J. Soot Combustion over Nanostructured Ceria with Different Morphologies. Sci. Rep. 2016, 6, 29062. [Google Scholar] [CrossRef]
- Feng, X.B.; Chen, C.W.; He, C.; Chai, S.N.; Yu, Y.K.; Cheng, J. Non-thermal plasma coupled with MOF-74 derived Mn-Co-Ni-O porous composite oxide for toluene efficient degradation. J. Hazard. Mater. 2020, 383, 121143. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.H.; Li, Y.L.; Zhang, L.Y.; Shen, L.J.; Xiao, Y.H.; Zhang, Y.F.; Au, C.; Jiang, L.L. Insight into the effect of morphology on catalytic performance of porous CeO2 nanocrystals for H2S selective oxidation. Appl. Catal. B Environ. 2019, 252, 98–110. [Google Scholar] [CrossRef]
- Dong, F.; Meng, Y.; Han, W.L.; Zhao, H.J.; Tang, Z.C. Morphology effects on surface chemical properties and lattice defects of Cu/CeO2 catalysts applied for low-temperature CO oxidation. Sci. Rep. 2019, 9, 12056. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.J.; Jian, Y.F.; Ma, M.D.; He, C.; Chen, C.W.; Liu, C.; Shi, J.-W. Rational design of CrOx/LaSrMnCoO6 composite catalysts with superior chlorine tolerance and stability for 1,2-dichloroethane deep destruction. Appl. Catal. A Gen. 2019, 570, 62–72. [Google Scholar] [CrossRef]
- Liang, H.; Hong, Y.X.; Zhu, C.Q.; Li, S.H.; Chen, Y.; Liu, Z.L.; Ye, D.Q. Influence of partial Mn-substitution on surface oxygen species of LaCoO3 catalysts. Catal. Today 2013, 201, 98–102. [Google Scholar] [CrossRef]
- Lin, X.T.; Li, S.J.; He, H.; Wu, Z.; Wu, J.L.; Chen, L.M.; Ye, D.Q.; Fu, M.L. Evolution of oxygen vacancies in MnOx-CeO2 mixed oxides for soot oxidation. Appl. Catal. B Environ. 2018, 223, 91–102. [Google Scholar] [CrossRef]
- Zhang, X.-m.; Deng, Y.-Q.; Tian, P.F.; Shang, H.-h.; Xu, J.; Han, Y.-F. Dynamic active sites over binary oxide catalysts: In situ/operando spectroscopic study of low-temperature CO oxidation over MnOx-CeO2 catalysts. Appl. Catal. B Environ. 2016, 191, 179–191. [Google Scholar] [CrossRef]
- Casapu, M.; Kroecher, O.; Mehring, M.; Nachtegaal, M.; Borca, C.; Harfouche, M.; Grolimund, D. Characterization of Nb-Containing MnOx-CeO2 Catalyst for Low-Temperature Selective Catalytic Reduction of NO with NH3. J. Phys. Chem. C 2010, 114, 9791–9801. [Google Scholar] [CrossRef]
- Su, J.; Yao, W.Y.; Liu, Y.; Wu, Z.B. The impact of CrOx loading on reaction behaviors of dichloromethane (DCM) catalytic combustion over Cr-O/HZSM-5 catalysts. Appl. Surf. Sci. 2017, 396, 1026–1033. [Google Scholar] [CrossRef]
- Dai, Y.; Wang, X.Y.; Dai, Q.G.; Li, D. Effect of Ce and La on the structure and activity of MnOx catalyst in catalytic combustion of chlorobenzene. Appl. Catal. B Environ. 2012, 111–112, 141–149. [Google Scholar] [CrossRef]
- Wang, W.; Zhu, Q.; Dai, Q.G.; Wang, X.Y. Fe doped CeO2 nanosheets for catalytic oxidation of 1,2-dichloroethane: Effect of preparation method. Chem. Eng. J. 2017, 307, 1037–1046. [Google Scholar] [CrossRef]
- Gannoun, C.; Delaigle, R.; Debecker, D.P.; Eloy, P.; Ghorbel, A.; Gaigneaux, E.M. Effect of support on V2O5 catalytic activity in chlorobenzene oxidation. Appl. Catal. A Gen. 2012, 447–448, 1–6. [Google Scholar] [CrossRef]
- Mei, J.; Ke, Y.; Yu, Z.J.; Hu, X.F.; Qu, Z.; Yan, N.Q. Morphology-dependent properties of Co3O4/CeO2 catalysts for low temperature dibromomethane (CH2Br2) oxidation. Chem. Eng. J. 2017, 320, 124–134. [Google Scholar] [CrossRef]
- Yang, P.; Meng, Z.H.; Yang, S.S.; Shi, Z.N.; Zhou, R.X. Highly active behaviors of CeO2–CrOx mixed oxide catalysts in deep oxidation of 1,2-dichloroethane. J. Mol. Catal. A-Chem. 2014, 393, 75–83. [Google Scholar] [CrossRef]
- Cai, T.; Huang, H.; Deng, W.; Dai, Q.G.; Liu, W.; Wang, X.Y. Catalytic combustion of 1,2-dichlorobenzene at low temperature over Mn-modified Co3O4 catalysts. Appl. Catal. B Environ. 2015, 166–167, 393–405. [Google Scholar] [CrossRef]
- Tian, M.J.; He, C.; Yu, Y.K.; Pan, H.; Smith, L.; Jiang, Z.Y.; Gao, N.B.; Jian, Y.F.; Hao, Z.P.; Zhu, Q. Catalytic oxidation of 1,2-dichloroethane over three-dimensional ordered meso-macroporous Co3O4/La0.7Sr0.3Fe0.5Co0.5O3: Destruction route and mechanism. Appl. Catal. A Gen. 2018, 553, 1–14. [Google Scholar] [CrossRef]
- Dai, Q.G.; Yin, L.-L.; Bai, S.X.; Wang, W.; Wang, X.Y.; Gong, X.-Q.; Lu, G.Z. Catalytic total oxidation of 1,2-dichloroethane over VOx/CeO2 catalysts: Further insights via isotopic tracer techniques. Appl. Catal. B Environ. 2016, 182, 598–610. [Google Scholar] [CrossRef]
- Dai, Q.G.; Wu, J.Y.; Deng, W.; Hu, J.S.; Wu, Q.Q.; Guo, L.M.; Sun, W.; Zhan, W.C.; Wang, X.Y. Comparative studies of P/CeO2 and Ru/CeO2 catalysts for catalytic combustion of dichloromethane: From effects of H2O to distribution of chlorinated by-products. Appl. Catal. B Environ. 2019, 249, 9–18. [Google Scholar] [CrossRef]
- Deng, W.; Dai, Q.G.; Lao, Y.J.; Shi, B.B.; Wang, X.Y. Low temperature catalytic combustion of 1,2-dichlorobenzene over CeO2–TiO2 mixed oxide catalysts. Appl. Catal. B Environ. 2016, 181, 848–861. [Google Scholar] [CrossRef]
- Hetrick, C.E.; Patcas, F.; Amiridis, M.D. Effect of water on the oxidation of dichlorobenzene over V2O5/TiO2 catalysts. Appl. Catal. B Environ. 2011, 101, 622–628. [Google Scholar] [CrossRef]
- Li, N.; Cheng, J.; Xing, X.; Sun, Y.G.; Hao, Z.P. Distribution and formation mechanisms of polychlorinated organic by-products upon the catalytic oxidation of 1,2-dichlorobenzene with palladium-loaded catalysts. J. Hazard. Mater. 2020, 393, 122412. [Google Scholar] [CrossRef] [PubMed]
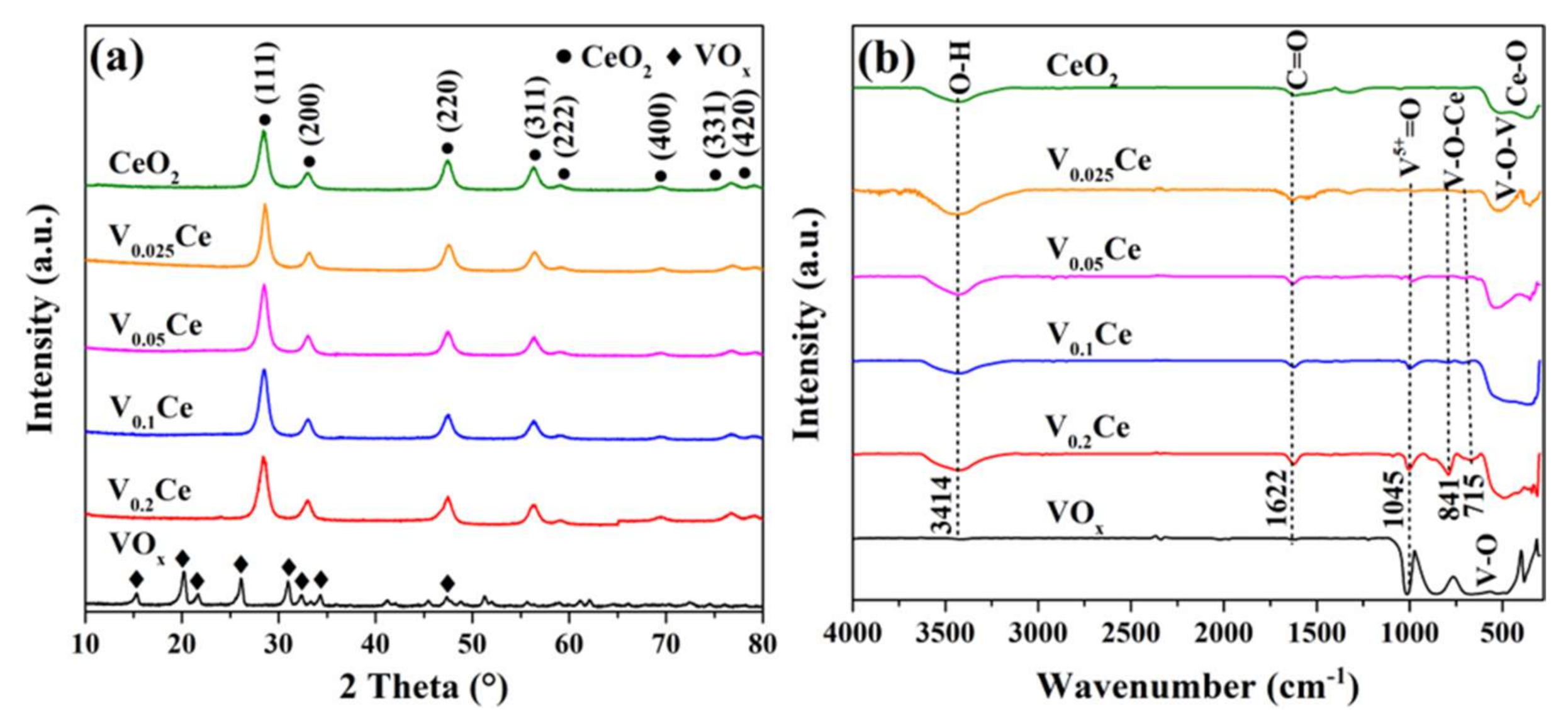


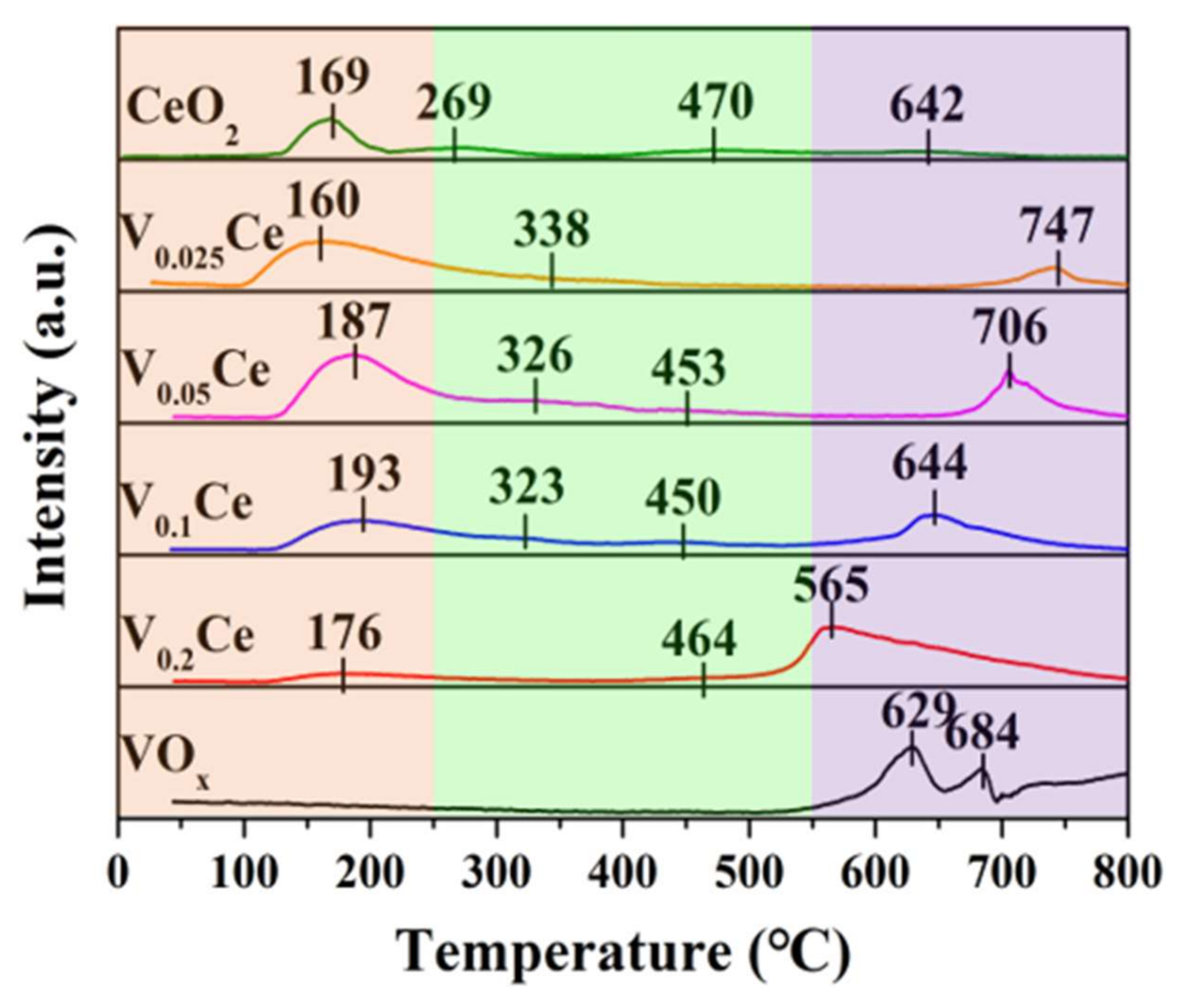
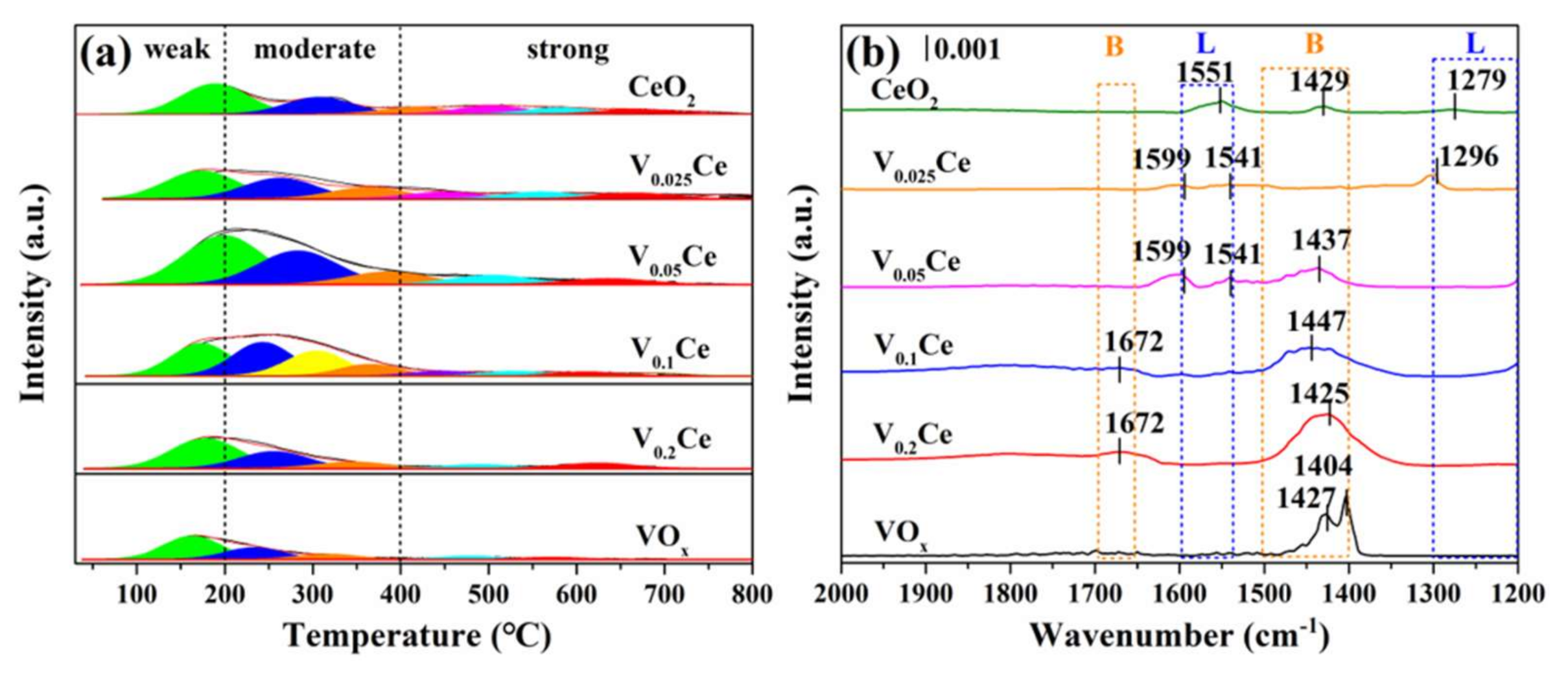

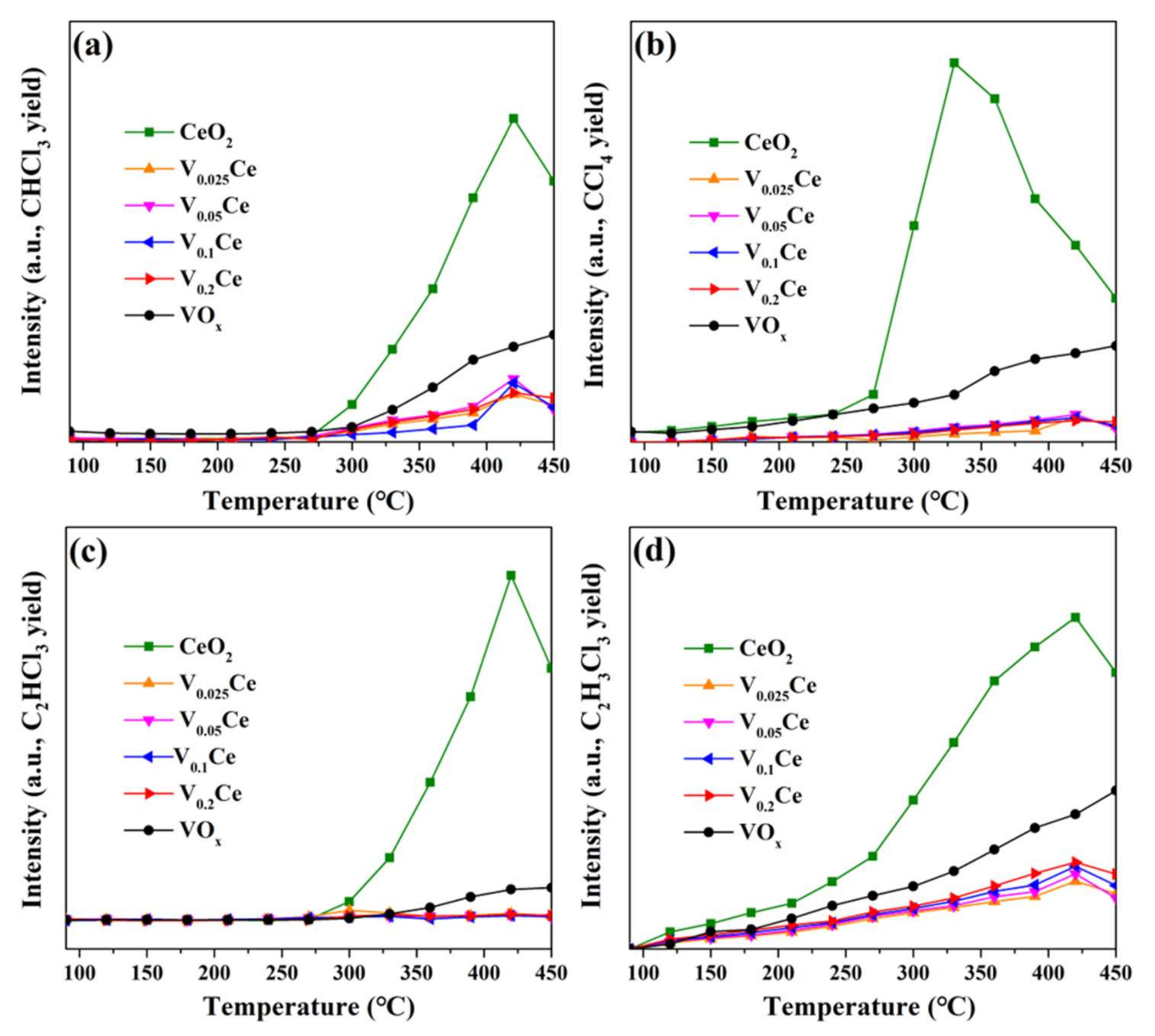
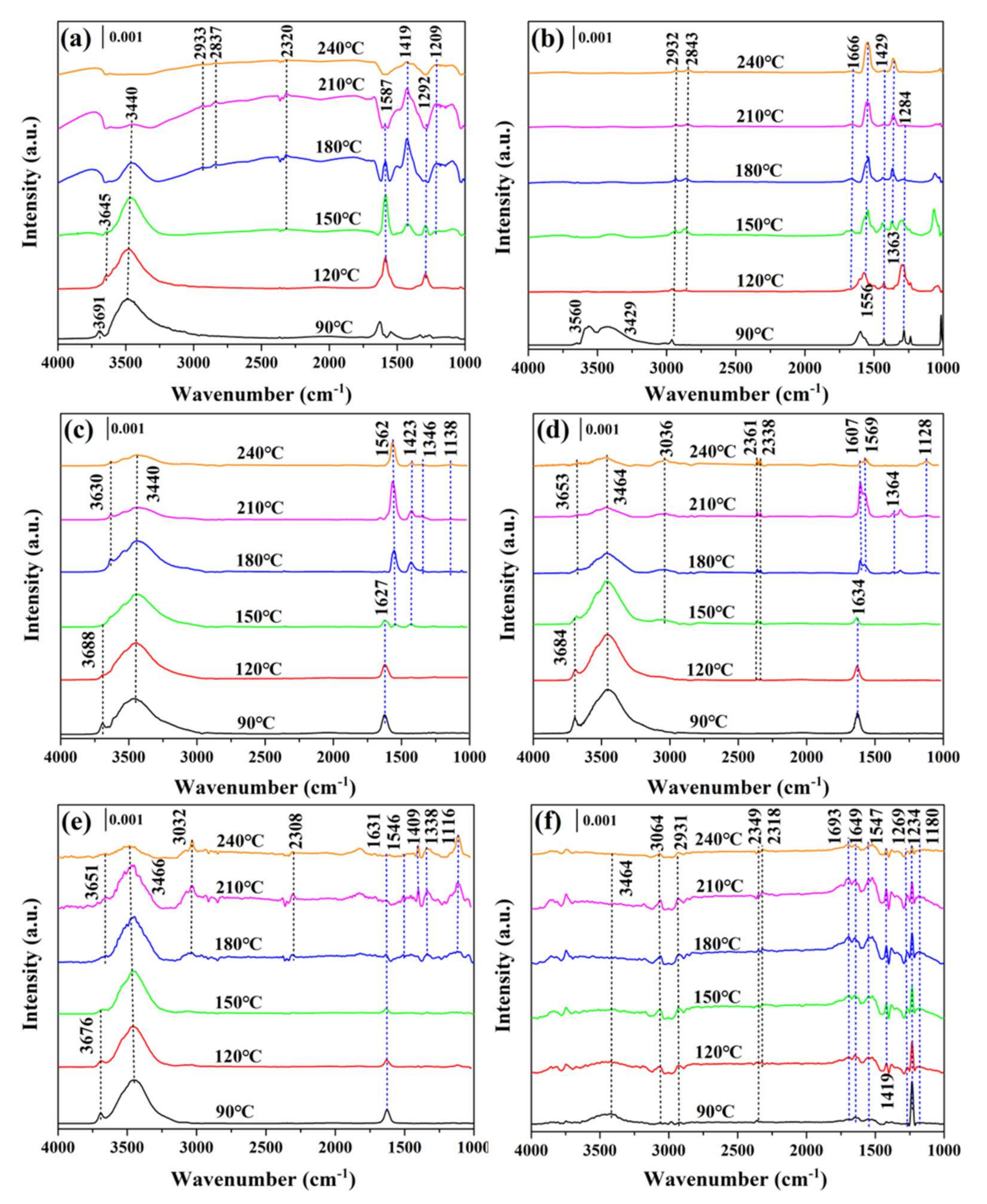

| Sample | SBETa (m2·g−1) | Vtb (×10−2, cm3·g−1) | Dpc (nm) | T50d (°C) | T90d (°C) |
|---|---|---|---|---|---|
| CeO2 | 110.9 | 36.54 | 13.2 | 328 | 381 |
| V0.025Ce | 101.0 | 45. 46 | 18.0 | 299 | 367 |
| V0.05Ce | 98.6 | 26. 43 | 10.7 | 281 | 347 |
| V0.1Ce | 93.4 | 30.46 | 30.1 | 294 | 356 |
| V0.2Ce | 76.1 | 28.18 | 14.8 | 307 | 413 |
| VOx | 11.3 | 17.92 | 63.1 | / | / |
| Sample | Binding Energy (eV) | Molar Ratio | ||||||
|---|---|---|---|---|---|---|---|---|
| V4+ | V5+ | Oαa | Oβb | Oγc | Oβ/(Oα + Oβ) | V5+/(V5+ + V4+) | Ce3+/(Ce3+ + Ce4+) | |
| CeO2 | / | / | 529.5 | 531.5 | 533.2 | 0.48 | / | 0.15 |
| V0.025Ce | 515.2 | 517.1 | 529.3 | 531.7 | 533.0 | 0.54 | 0.84 | 0.17 |
| V0.05Ce | 515.4 | 517.2 | 529.5 | 531.6 | 533.1 | 0.61 | 0.87 | 0.19 |
| V0.1Ce | 515.0 | 517.2 | 529.4 | 531.9 | 533.4 | 0.57 | 0.85 | 0.18 |
| V0.2Ce | 515.4 | 517.4 | 529.9 | 531.8 | 533.3 | 0.46 | 0.83 | 0.13 |
| VOx | / | 517.2 | 529.6 | 531.9 | 533.3 | 0.45 | / | / |
| Sample | H2 Consumption (mmol·g−1) | O2 Desorption Peaks | NH3 Desorption Peaks | ||
|---|---|---|---|---|---|
| Pa | P1b | P2c | P3d | Pe | |
| CeO2 | 8.02 | 25,200 | 7190 | 5029 | 231.78 |
| V0.025Ce | 8.64 | 40,000 | 9423 | 11,700 | 250.55 |
| V0.05Ce | 9.86 | 30,700 | 13,700 | 26,900 | 257.03 |
| V0.1Ce | 9.53 | 27,800 | 12,256 | 56,600 | 244.76 |
| V0.2Ce | 7.63 | 8536 | 2551 | 59,100 | 223.09 |
| VOx | / | / | / | 26,700 | 83.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Fang, S.; Tian, M.; Jiang, Z.; Wu, Y.; He, C. Chlorine-Resistant Hollow Nanosphere-Like VOx/CeO2 Catalysts for Highly Selective and Stable Destruction of 1,2-Dichloroethane: Byproduct Inhibition and Reaction Mechanism. Processes 2021, 9, 119. https://doi.org/10.3390/pr9010119
Huang Y, Fang S, Tian M, Jiang Z, Wu Y, He C. Chlorine-Resistant Hollow Nanosphere-Like VOx/CeO2 Catalysts for Highly Selective and Stable Destruction of 1,2-Dichloroethane: Byproduct Inhibition and Reaction Mechanism. Processes. 2021; 9(1):119. https://doi.org/10.3390/pr9010119
Chicago/Turabian StyleHuang, Yu, Shiyue Fang, Mingjiao Tian, Zeyu Jiang, Yani Wu, and Chi He. 2021. "Chlorine-Resistant Hollow Nanosphere-Like VOx/CeO2 Catalysts for Highly Selective and Stable Destruction of 1,2-Dichloroethane: Byproduct Inhibition and Reaction Mechanism" Processes 9, no. 1: 119. https://doi.org/10.3390/pr9010119
APA StyleHuang, Y., Fang, S., Tian, M., Jiang, Z., Wu, Y., & He, C. (2021). Chlorine-Resistant Hollow Nanosphere-Like VOx/CeO2 Catalysts for Highly Selective and Stable Destruction of 1,2-Dichloroethane: Byproduct Inhibition and Reaction Mechanism. Processes, 9(1), 119. https://doi.org/10.3390/pr9010119