Abstract
Rhanterium epapposum, native to the Arabian Peninsula, is traditionally used to cure skin infections. The objective is to screen the phytochemical content and antimicrobial activity of aqueous, methanol and 80% methanol extracts of aerial parts of R. epapposum. The phytochemical screening of aqueous, methanolic, and 80% methanol extracts of R. epapposum was conducted using gas chromatographymass spectrometry. The antimicrobial activities of the extracts were assessed by well diffusion and microdilution methods. Qualitative phytochemical analysis revealed the presence of 2-methoxy-4-vinylphenol in all three extracts, whereas ethanol, 2-methoxy-, acetate; n-hexadecanoic acid; and 2,3-butanediol are present in higher amount exclusively in the methanol, 80% methanol and aqueous extracts of the aerial parts of R. epapposum, respectively. The highest antibacterial activity was shown by the aqueous extract S. aureus, P. aeruginosa, E. cloacae, and K. pneumoniae, methanolic extract against S. aureus, methicillin-resistant S. aureus, and E. coli, and 80% methanol extract against S. epidermidis, and S. paucimobilis. Interestingly, 80% methanol extracts showed the highest antifungal activity against C. albicans, C. guillermondii, C. vaginalis, C. utilis, and C. tropicalis. The aerial parts of R. epapposum showed broad-spread antimicrobial activity against bacteria and fungi. Especially, the 80% methanol extract showed potent antifungal activity against all the tested fungal strains.
1. Introduction
Microbial infections and the emergence of drug-resistance microbes have become one of the biggest challenges worldwide. Microbial infections cause millions of deaths every year because of the limited number of effective drugs in the market and the side effects of synthetic antibiotics [1]. For the management of microbial infections in humans, it is necessary to find new antimicrobial substances directed at new drug targets. Medicinal plants are rich in bioactive compounds, which have diverse phytochemicals and mechanisms of action. Several phytochemicals have exhibited potent activities in combating microbial infections [2,3,4]. Due to the extensive existence and minimum side effects, phytochemicals are of the utmost interest to pharmaceutical industries [5]. Already some plants or plant-derived chemicals are used commercially as antimicrobial drugs [1]. Although about 250,000 to 500,000 plant species exist on Earth [6], only a limited number of plants have been studied for their medicinal use. Thus, research focuses on identifying herbal-based antimicrobial agents, and its phytochemical screening has accelerated in recent years.
In the Arabian Peninsula, about 150 plants were used traditionally for medicinal treatments [7]. Rhanterium epapposum Oliv. (common name: Arfaj), a perennial dwarf shrub, is native to the Arabian Peninsula. It is used in folk medicine to treat microbial infections and gastrointestinal disturbances [8,9]. The essential oil of R. epapposum contains 107 volatile components, which are mostly terpenoids [10]. The essential oil is also reported to have antimicrobial, insect-repellent, and anticholinesterase activities [11]. The ethanolic extract is reported to have alkaloids, flavonoids, triterpenes, cumarins, and tannins [12]. Preliminary studies on the petroleum ether and methanol extracts of aerial parts of R. epapposum in Sudan showed antibacterial activity [13]. This study also analyzed the class of chemicals in the R. epapposum using colorimetric methods. The aerial parts of R. epapposum are reported to have flavonoids, tannins, sterols, triterpenes, and essential oils [14]. In this study, our aim is the in-depth analysis of the phytochemical content of the aerial parts of R. epapposum (in the Kingdom of Saudi Arabia) using GC–MS and evaluate the antibacterial and antifungal activities of aqueous, methanol, and 80% methanol extracts of R. epapposum in vitro.
2. Materials and Methods
2.1. Plant Collection
Aerial parts of Rhanterium epapposum (plant parts that are above the ground, including stems, leaves, and flowers) were collected from Ha′il region, Kingdom of Saudi Arabia, in March 2021. Botanical identification was made, and a voucher specimen was deposited at the herbarium in the Department of Biology, College of Science, University of Ha′il, Kingdom of Saudi Arabia. The plant was dried at room temperature and stored in closed containers in the dark until used.
2.2. Extract Preparation
For the preparation of extracts, the plant material was macerated in water, pure methanol, or 80% methanol (ratio: 1:10, w/v). The extracts were filtered, and the solvents were removed at 60 °C in the incubator chamber.
2.3. Phytochemical Screening
The chemical contents of the extracts were screened using gas chromatography and mass spectrometery (GC–MS). The different parameters involved in the operation of the Clarus 500 MS were standardized as follows: inlet line temperature 200 °C; ion source temperature 230 °C. Mass spectra were taken at 70 eV; solvent delay time 5 min, acquisition mode- scan 40-550 amu. The GC used in the analysis employed a fused silica column packed with Elite-1 (100% dimethyl poly siloxane, 30 nm × 0.25 nm ID × 1µm df), and the components were separated using helium as carrier gas at a constant flow of 1 mL/min. The 2 µL sample extract injected into the instrument was detected by the Turbo gold mass detector (Perkin Elmer) with the aid of the Turbo mass 5.1 software. The oven temperature program was 2 min at 45 °C, 1.5 °C/min to 100 °C, 2 °C /min to 200 °C during the GC extraction process; split ratio 25:1. The injector temperature was set at 250 °C (mass analyzer). The GC run time was 90 min. The interpretation of mass spectrum and phytochemical identifications were carried out with the aid of a standard database of the National Institute of Standards and Technology.
2.4. Microorganisms
For assessing the antibacterial activity of extracts, eight Gram-positive or Gram-negative bacterial strains were used: Staphylococcus aureus (ATCC 29213), Staphylococcus epidermidis (ATCC 12228), methicillin-resistant Staphylococcus aureus (MRSA-136), Pseudomonas aeruginosa (ATCC 27853), Sphingomonas paucimobilis (clinical strain, 144), Enterobacter cloacae (clinical strain, 155), Klebsiella pneumoniae (clinical strain, 220), and Escherichia coli (ATCC 10536). For assessing the antifungal activity of the extract, five yeast strains were used: Candida albicans (ATCC 20402), Candida guilliermondii (ATCC 6260), Gardnerella vaginalis (clinical strain, 136), Candida utilis (ATCC 9255), and Candida tropicalis (ATCC 1362). The strains were maintained on Tryptone Soya agar (Oxoid, Milan, Italy).
2.5. Well Diffusion Assay
The antimicrobial activities of various extracts were evaluated by the well diffusion method. The agar plates were uniformly spread with 1 mL of the microbial strain (bacteria/fungi). Wells were made in the agar plate by puncturing the gel. A 100 μL aliquot of the extract or standard drug was added into the respective wells. After incubation for 24 h at 37 °C, zones of inhibition were measured. For positive control, standard antibiotics (ampicillin against bacteria and amphotericin B against fungus) were purchased from Sigma-Aldrich, St. Louis, MO, USA and used.
2.6. Microdilution Assay
The microtiter broth dilution method was carried out to evaluate the minimum inhibitory concentrations (MICs) and minimum bactericidal concentration (MBCs) or minimum fungicidal concentrations (MFCs) of various extracts [15]. Mueller–Hinton broth and Sabouraud dextrose broth were used as the test medium for bacterial and fungal strains, respectively. In a 96-well microtiter plate, 100 μL of microbial suspensions (bacteria/fungi) were inoculated with various concentrations of the extracts (0.039 to 50 mg mL−1). After incubation for 24 h at 37 °C, the lowest concentration of the extract, which did not show any visual growth of tested organisms, was recorded as the MIC value (mg mL−1). The MBCs/MFCs values are determined by streaking all wells after the MICs values on the correspondent agar media of the tested microorganisms. The mechanism of action of the extracts was interpreted by calculating MBC/MIC or MFC/MIC ratios [16].
2.7. Statistical Analysis
All the experiments were performed in triplicate to get statistically significant data. The results were expressed as mean (average value) ± standard deviation (SD).
3. Results
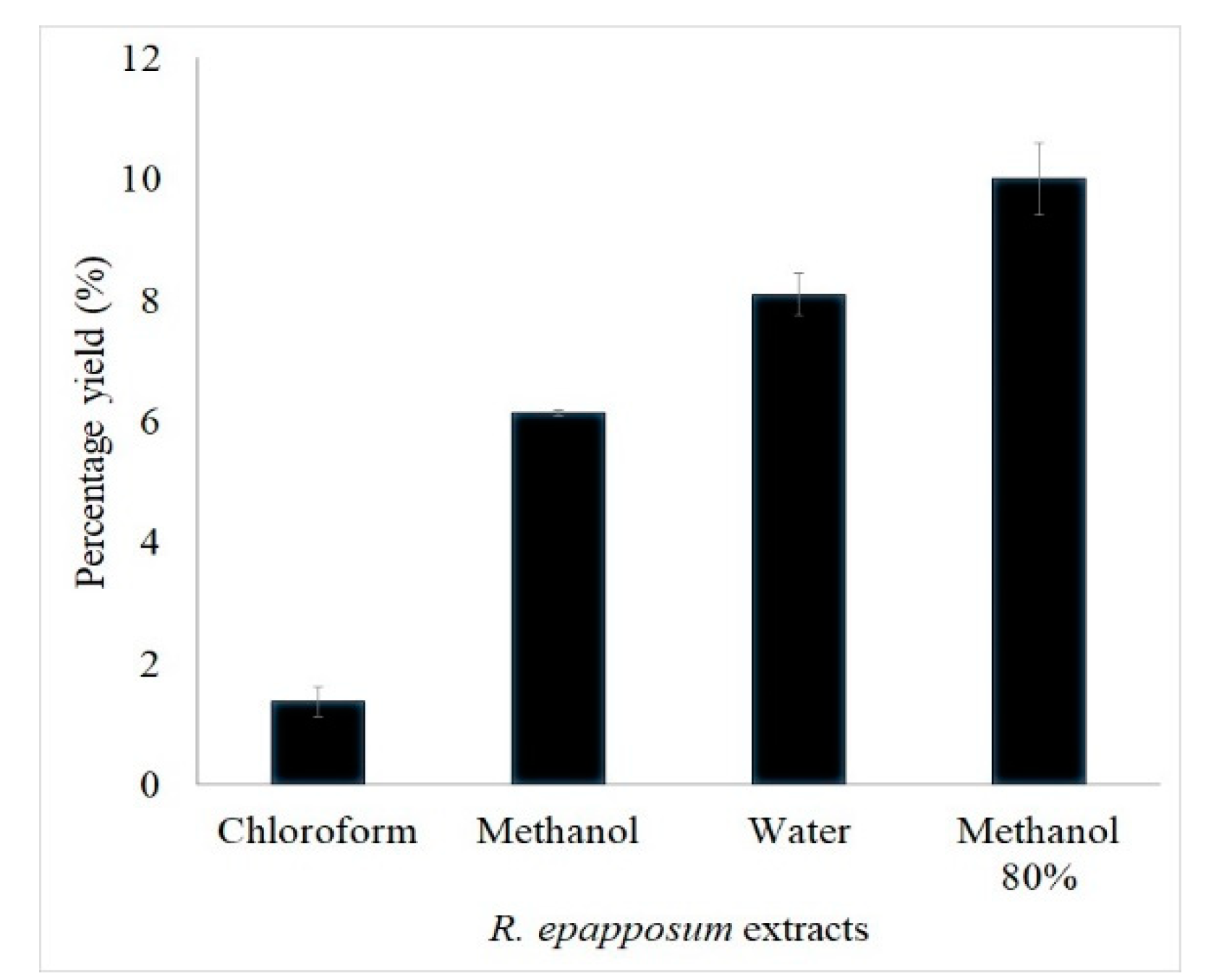
The yields of extraction from the aerial parts of R. epapposum using chloroform, water, methanol, and 80% methanol are 1.4%, 8.1%, 6.2%, and 10%, respectively (Figure 1). Due to the low yield of chloroform extract, only the remaining three extracts were used for phytochemical screening and antimicrobial experiments.
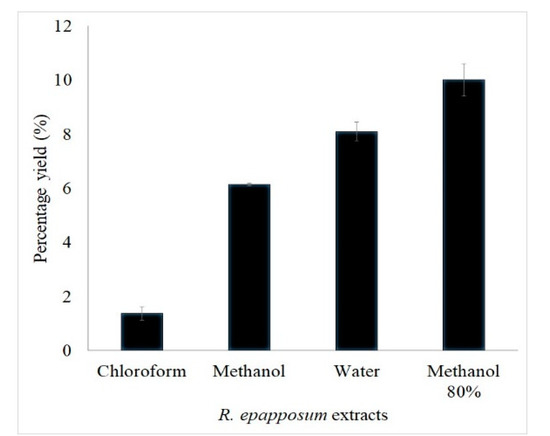
Figure 1.
Percent yield of chloroform, methanol, aqueous, and 80% methanol extracts of R. epapposum.
3.1. Phytochemical Composition
GC–MS analysis of R. epapposum extracts identified 16, 23, and 9 phytochemicals present exclusively only in in the methanol extract (Table 1), 80% methanol extract (Table 2), and aqueous extract (Table 3), respectively. Thirteen phytochemicals were present in at least two extracts (Table 4). All the identified compounds were listed along with their chemical formula, common name, and molecular weight.

Table 1.
Chemical composition of the compounds that are present only in the methanolic R. epapposum extracts.

Table 2.
Chemical composition of the compounds that are present only in the methanol 80 % R. epapposum extracts.

Table 3.
Chemical composition of the compounds that are present only in the water R. epapposum extracts.

Table 4.
Chemical composition of the compounds that are present in two or three methanolic, methanol 80%, and water R. epapposum extracts.
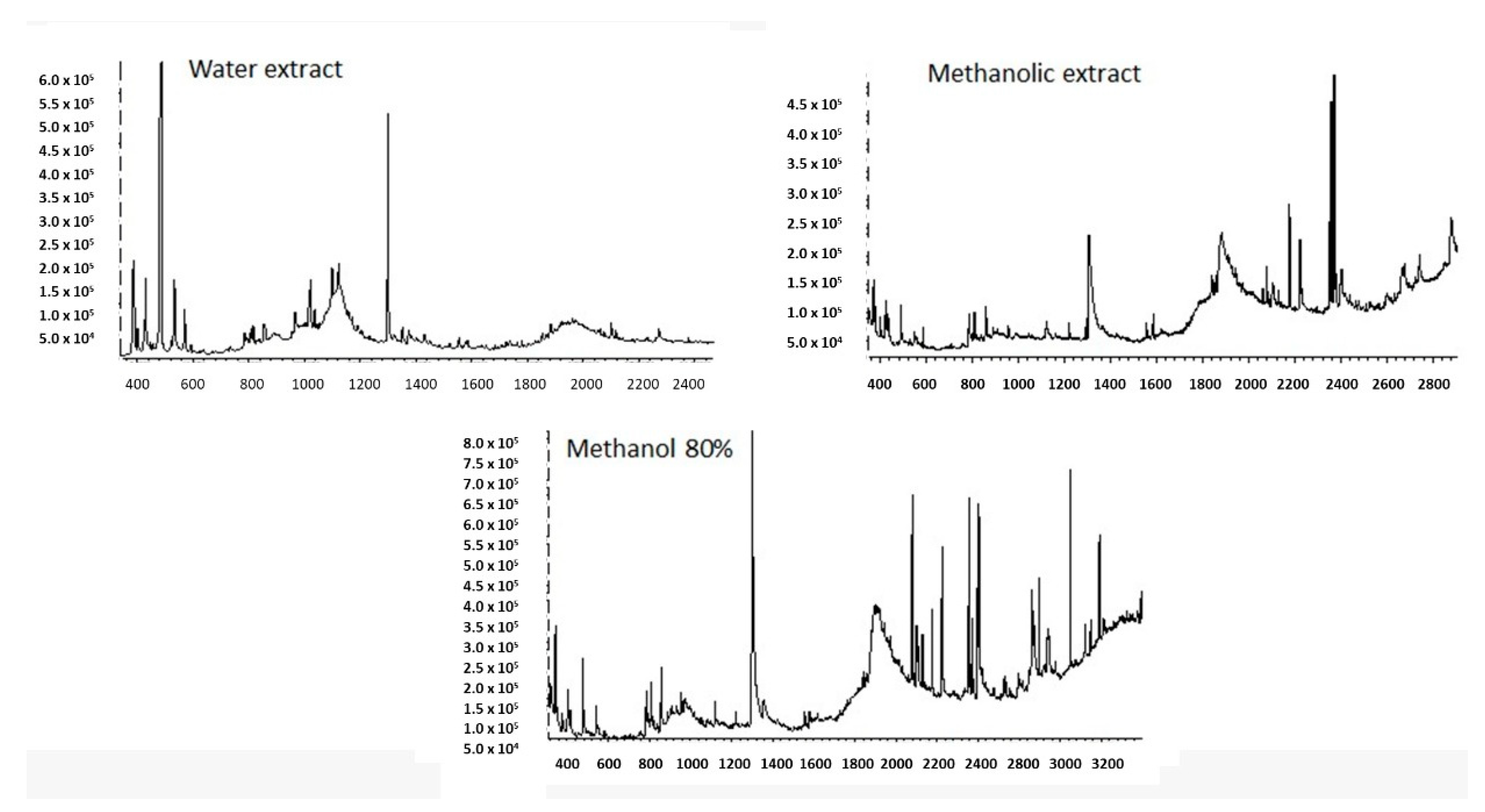
All the tested R. epapposum extracts contain 2-Methoxy-4-vinylphenol at a higher percentage (29–40%). The methanol extract is dominated (>5%) by 2-Methoxy-4-vinylphenol; phytol; 9,12,15-Octadecatrienoic acid, methyl ester and Hexadecanoic acid, methyl ester. The 80% methanol extract is dominated by 2-methoxy-4-vinylphenol; 1,4-Dioxane-2,5-dione, 3,6-dimethyl-; bicyclo[3.1.1]heptane, 2,6,6-trimethyl-, (1.alpha.,2.beta.,5.alpha.)-; n-Hexadecanoic acid; 9,12,15-Octadecatrienoic acid, methyl ester, (Z,Z,Z)-; Methyl 8,11,14-heptadecatrienoate; Carbamic acid, N-phenyl-, 1-methyl-1-(4-methylcyclohex-3-enyl)ethyl ester and heptacosane. The aqueous extract is dominated by 2-Methoxy-4-vinylphenol, 2,3-Butanediol; 1,4-Dioxane-2,5-dione, 3,6-dimethyl-; Glycine, N,N-dimethyl-, methyl ester and Dimethylaminomethyl-isopropyl-sulfide. The chromatograms (Figure 2) showed respectively major peaks indicating the presence of various phytochemical constituents. Figure 3 shows the chemical structure of all the identified chemicals.
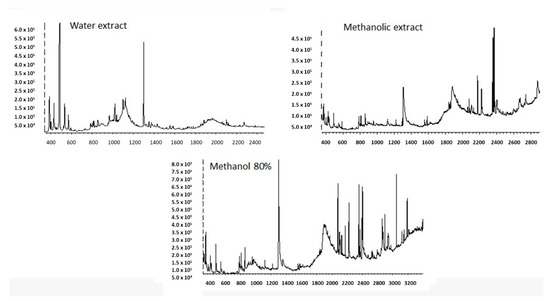
Figure 2.
GC-MS chromatograms of aqueous, methanol and 80% methanol extracts of R. epapposum.
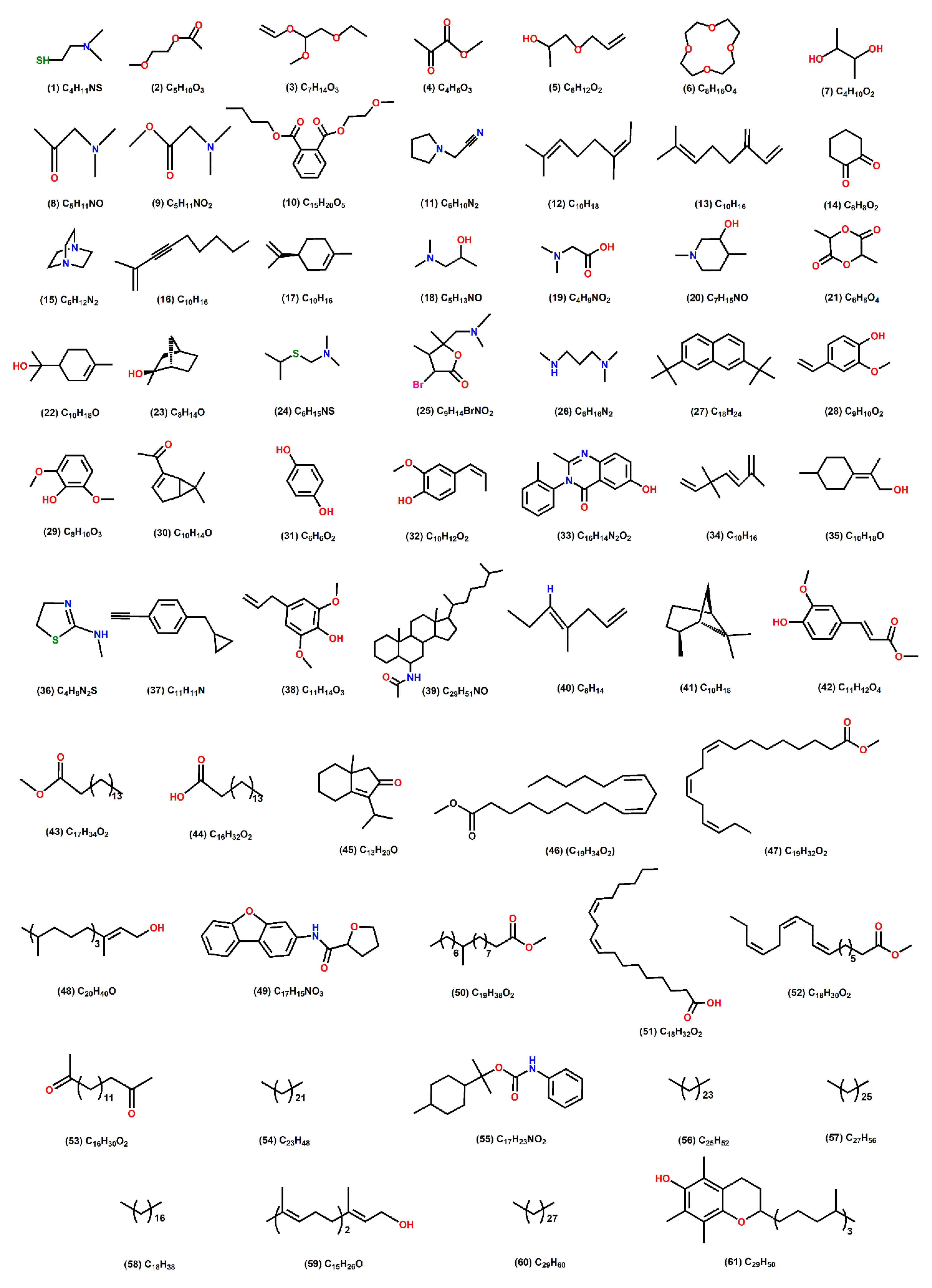
Figure 3.
Chemical structure of the different compounds present in the aqueous, methanol and 80% methanol extracts of R. epapposum.
3.2. Antimicrobial Activities
The antimicrobial activities of various R. epapposum extracts against Gram-positive/Gram-negative bacteria and yeast strains are shown in Table 5.

Table 5.
Growth inhibition zone obtained for bacterial and fungal strains tested using well diffusion assay from aqueous, methanolic and 80% methanol extracts of R. epapposum.
The results revealed that the R. epapposum extracts exhibited a wide antimicrobial spectrum, against tested bacteria and fungi with the inhibition zone diameter fluctuant from 14.67 to 44.67 mm. Methanol, 80% methanol, and aqueous extracts of R. epapposum showed various degrees of inhibition. In comparison, the aqueous extract showed the highest antibacterial activity against S. aureus, P. aeruginosa, E. cloacae, and K. pneumoniae. The methanolic extract showed the highest antibacterial activity against S. aureus, methicillin-resistant S. aureus, and E. coli. The 80% methanol extract showed the highest antibacterial activity against S. epidermidis, and S. paucimobilis. The 80% methanol extract also showed the highest antifungal activity than methanolic and aqueous extracts against all the tested fungal strains.
The MBC/MIC ratios in Table 6 showed that aqueous extract of R. epapposum acts as a bactericidal agent (MBC/MIC ≤ 4) [17] against five tested bacterial strains (S. epidermidis, methicillin-resistant S. aureus, P. aeruginosa, K. pneumoniae, and E. coli), whereas methanol and 80% methanol extracts act as bactericidal agents against two (S. epidermidis and E. coli) and four (S. aureus, S. epidermidis, K. pneumoniae, and E. coli) bacterial strains, respectively. In addition, the aqueous, methanol, and 80% methanol extracts act as bacteriostatic agents (MBC/MIC > 4) against three, six, and four tested bacterial strains, respectively. Against the five tested fungi strains, the methanolic extract acts as a fungicidal agent (MFC/MIC ≤ 4) against all the fungi strains. The aqueous extract acts as a fungicidal agent against four (all except C. albicans) fungal strains and a fungistatic agent (MFC/MIC > 4) against C. albicans. The aqueous extracts act as a fungicidal agent against C. vaginalis and C. tropicalis, whereas it acts as a fungistatic agent against C. albicans, C. guillermondii, and C. utilis.

Table 6.
MIC, MBC, and MFC values obtained for bacterial and yeast strains tested using microdilution assay.
When comparing the effect of ampicillin, we observe that all three extracts showed better antibacterial activity against S. aureus, methicillin-resistant S. aureus, P. aeruginosa, E. cloacae, and E. coli. At least two extracts showed better activity than ampicillin against S. epidermidis and K. pneumoniae. Concerning the antifungal activity, all three extracts showed better activity than the standard antifungal antibiotic, amphotericin B, against all the tested fungal strains.
4. Discussion
In the present work, we identified 61 chemical compounds in R. epapposum aerial parts using the GC–MS technique. When comparing the phytochemicals in all three extracts, 16 chemicals were present only in the methanol extract, 23 compounds were present only in 80% methanol extract, and 9 chemicals were present only in aqueous extracts. The extraction of specific compounds by methanol, 80% methanol, and water are responsible for the difference in the biological activity of various extracts. The essential oil of R. epapposum is reported to contain five major chemicals (55.6%), namely, α-phellandrene, linalol, geraniol, bulnesol, and β-phellandrene [10]. However, none of these chemicals are present in the aqueous, methanol, or 80% methanol extracts.
The water, methanol, and 80% methanol extracts of aerial parts of R. epapposum were effective against all the tested bacterial and fungal strains. It showed that all three extracts contain antimicrobial compounds. Previous reports also revealed the antibacterial efficacy of R. epapposum methanol extract and essential oils [11,13]. Interestingly, the aqueous extract of aerial parts of R. epapposum showed the highest antibacterial activity against many of the tested bacterial strains, whereas Adam et al. [13] reported that the aqueous extract of aerial parts of R. epapposum does not have any antibacterial activity. The discrepancy is due to the difference in the extraction method. Adam et al. prepared the aqueous extract using the residues after the sequential extraction using petroleum ether and methanol [13].
The water, methanol, and 80% methanol extracts showed the highest antibacterial activity against four (S. aureus, P. aeruginosa, E. cloacae, and K. pneumoniae), three (S. aureus, methicillin-resistant S. aureus, and E. coli), and two (S. epidermidis and S. paucimobilis) bacterial strains, respectively. During the screening of 21 plant species in the Alqassim region, Saudi Arabia for their antimicrobial activity, it was found that the crude extracts of R. epapposum have antibacterial activity [18]. The MIC values of the extracts ranging from 12.5 to 25 mg/dL. MBC/MIC ratio indicates that the extracts act as bactericidal or bacteriostatic. The reason for the selectivity and different mechanism of action of the extracts could be the difference in the specific antibacterial compounds in the extracts.
All the extracts (aqueous, methanol, and 80% methanol) of aerial parts of R. epapposum showed potent antifungal activities, which are higher than amphotericin B. Out of the three extracts, the 80% methanol extract showed the highest antifungal activity as compared to aqueous and methanol extracts. In line with our finding, a previous study also reports that n-hexane extract of R. epapposum showed antifungal effect against C. albicans [19]. The MIC of aqueous extract ranging from 3.125 to 25 mg/mL, whereas the MIC of methanolic and 80% methanol extracts ranging from 6.25 to 12.5 mg/mL. It is interesting to note that the highly active 80% methanolic extract act as a fungicidal agent (against C. vaginalis and C. tropicalis) or fungistatic agent (against C. albicans, C. guillermondii, and C. utilis), whereas both aqueous and methanolic extracts mostly act as a fungicidal agent (except the action of aqueous extract against C. albicans). As about 150 million people are affected by serious fungal diseases [20], the 80% methanol extract of R. epapposum aerial parts deserves further research to explore an effective antifungal drug.
5. Conclusions
This study demonstrates the antibacterial and antifungal activities of aerial parts of R. epapposum in vitro. The study also outlines the differences in the antibacterial activity of aqueous, methanolic, and 80% methanol extracts. In the antifungal experiments, 80% methanol extract showed potent and highest activity than the methanol and aqueous extracts. Testing the antimicrobial activities of purified phytochemicals would be a future perspective of this work in order to find the bio-active antimicrobial compound. In addition, testing the antimicrobial activities of mixtures of major compounds present in extracts (methanol, 80% methanol, and water) may reveal the synergistic effects of phytochemicals in R. epapposum.
Author Contributions
Conceptualization, S.R., H.E.M., M.S., R.M.R., E.A.S., K.M.Y. and H.S.E.-B.; methodology and software, S.R., H.E.M. and M.S.; validation, S.R., H.E.M., M.S. and R.M.R.; formal analysis and data curation, S.R., H.E.M., M.S., R.M.R., E.A.S., K.M.Y. and H.S.E.-B.; writing—original draft preparation, S.R.; writing—review and editing, H.E.M., M.S., R.M.R., E.A.S., K.M.Y. and H.S.E.-B.; visualization, H.E.M.; project administration and supervision, H.E.M. and R.M.R.; funding acquisition, R.M.R. and E.A.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Deanship of Research, University of Ha′il, Ha′il, Saudi Arabia; grant number RG-191256.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Khameneh, B.; Iranshahy, M.; Soheili, V.; Bazzaz, B.S.F. Review on plant antimicrobials: A mechanistic viewpoint. Antimicrob. Resist. Infect. Control. 2019, 8, 1–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gyawali, R.; Ibrahim, S.A. Natural products as antimicrobial agents. Food Control. 2014, 46, 412–429. [Google Scholar] [CrossRef]
- Narayana, D.V. A critical review on natural antimicrobial agents. World J. Pharm. Pharm. Sci. 2017, 329–341. [Google Scholar] [CrossRef]
- Anand, U.; Nandy, S.; Mundhra, A.; Das, N.; Pandey, D.K.; Dey, A. A review on antimicrobial botanicals, phytochemicals and natural resistance modifying agents from Apocynaceae family: Possible therapeutic approaches against multidrug resistance in pathogenic microorganisms. Drug Resist. Updat. 2020, 51, 100695. [Google Scholar] [CrossRef]
- Borris, R.P. Natural products research: Perspectives from a major pharmaceutical company. J. Ethnopharmacol. 1996, 51, 29–38. [Google Scholar] [CrossRef]
- Saganuwan, S.A. Some medicinal plants of Arabian Pennisula. J. Med. Plant Res. 2010, 4, 766–788. [Google Scholar] [CrossRef]
- El-shanawany, M.A.A. Plants Used in Saudi Folk Medicine; King Abdul-Aziz City for Science and Technology: Riyadh, Kingdom of Saudi Arabia, 1996. [Google Scholar]
- Phondani, P.C.; Bhatt, A.; Elsarrag, E.; Horr, Y.A. Ethnobotanical magnitude towards sustainable utilization of wild foliage in Arabian Desert. J. Tradit. Complement. Med. 2016, 6, 209–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yaghmai, M.S.; Kolbadipour, S. Volatile components ofRhanterium epapposum oliv. Flavour Fragr. J. 1987, 2, 29–32. [Google Scholar] [CrossRef]
- Demirci, B.; Yusufoglu, H.S.; Tabanca, N.; Temel, H.E.; Bernier, U.; Agramonte, N.; Alqasoumi, S.I.; Al-Rehaily, A.J.; Başer, K.H.C.; Demirci, F. Rhanterium epapposum Oliv. essential oil: Chemical composition and antimicrobial, insect-repellent and anticholinesterase activities. Saudi Pharm. J. 2017, 25, 703–708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeinab, E.B.E.; Abdelhafiz, A.D. Effectiveness of using oils extracts of Peganum harmala and Rhanterium epapposum against Khapra beetle (Coleoptera: Dermestidae) and their chemical compositions. Sci. Res. Essays 2019, 14, 68–73. [Google Scholar] [CrossRef]
- Adam, S.I.Y.; EL-Kamali, H.; Adama, S.E.I. Phytochemical screening and antibacterial activity of two Sudanese wild plants, Rhanterium epapposum and Trichodesma africanum. J. Fac. Sci. Tech. 2011, 2, 83–96. [Google Scholar]
- Al-yahya, M.A.; Al-meshal, I.A.; Mossa, J.S.; Al-badr, A.A.; Tariq, M. Saudi Plants, A Phytochemical and Biological Approach; King Saud University: Riyadh, Saudi Arabia, 1990; pp. 75–80. [Google Scholar]
- Snoussi, M.; Dehmani, A.; Noumi, E.; Flamini, G.; Papetti, A. Chemical composition and antibiofilm activity of Petroselinum crispum and Ocimum basilicum essential oils against Vibrio spp. strains. Microb. Pathog. 2016, 90, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Gatsing, D.; Tchakoute, V.; Ngamga, D.; Kuiate, J.R.; Tamokou, J.D.D.; Nji-nkah, B.F.; Tchouanguep, F.M.; Fodouop, C. In Vitro antibacterial activity of Crinum purpurascens Herb. leaf extract against the Salmonella species causing typhoid fever and its toxicological evaluation. Iran. J. Med. Sci. 2009, 34, 126–136. [Google Scholar]
- Hazen, K.C. Fungicidal versus fungistatic activity of terbinafine and itraconazole: An In Vitro comparison. J. Am. Acad. Dermatol. 1998, 38, S37–S41. [Google Scholar] [CrossRef]
- Aldoweriej, A.M.; Alharbi, K.B.; Saeed, E.M.A.; El-ashmawy, I.M. Antimicrobial activity of various extracts from some plants native to Alqassim region, Saudi Arabia. J. Food Agric. Environ. 2016, 14, 14–19. [Google Scholar]
- Mohammed, H.; Al-Omer, M.S.; Ahmed, A.M.; Hashish, N.E.; Alsaedi, H.M.; Alghazy, S.A.; Abdellatif, A.A.H. Comparative Study for the Volatile Oil Constituents and Antimicrobial Activity of Rhanterium epapposum Oliv. Growing in Qassim, Saudi Arabia. Pharmacogn. J. 2019, 11, 195–199. [Google Scholar] [CrossRef] [Green Version]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J. Fungi. 2017, 3, 57. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).