Abstract
The aim of this systematic review was to describe the clinical and genetic features of syndromes showing oligodontia as a sign. The review was performed according to the PRISMA 2020 checklist guidelines, and the search was conducted using PubMed, Scopus, Lilacs, Web of science, Livivo, and EMBASE and supplemented by a gray literature search on Google Scholar and ProQuest, applying key terms relevant to the research questions. The systematic review identified 47 types of syndromes in 83 studies, and the most common was hypohidrotic ectodermal dysplasia, which was reported in 24 patients in 22 studies. Other common syndromes that reported oligodontia included Axenfeld–Rieger syndrome, Witkop’s syndrome, Ellis–van Creveld syndrome, blepharocheilodontic syndrome, and oculofaciocardiodental syndrome. The X-linked mode of inheritance was the most reported (n = 13 studies), followed by the autosomal dominant (n = 13 studies). The review describes the main syndromes that may have oligodontia as a clinical sign and reinforces the need for orodental–facial examining for adequate diagnosis and treatment of the affected patients. Molecular analysis in order to better understand the occurrence of oligodontia is imperative.
1. Introduction
Tooth agenesis is defined as the absence of teeth from the normal series due to a failure to develop and encompasses hypodontia, oligodontia, and anodontia [1]. The absence of up to five teeth is classified as hypodontia, the congenital absence of six or more teeth is defined as oligodontia, and anodontia refers to the complete absence of all teeth from the normal series [1]. Tooth development is regulated by a series of signaling pathways, and genetic mutations in specific genes have been described as the causes of such defects [2,3]. Moreover, environmental factors such as trauma, infections, toxins, and dietary deficiencies have been implicated and could interact with the genetic factors as a complex and multifactorial disease [4,5].
The occurrence of oligodontia can be observed as an isolated trait (non-syndromic oligodontia) or accompanying other features as part of a syndrome [6,7,8,9]. In cases of non-syndromic oligodontia, the congenital absence of teeth stands as the sole discernible clinical characteristic [10]. These instances are relatively uncommon, with a prevalence that varies from 0.08% to 0.36%, contingent upon the specific population under study [11,12,13]. Nevertheless, the intricate interplay between oligodontia and syndromes has increasingly captured attention. The association of oligodontia with over 60 different syndromes, including but not limited to hypohidrotic ectodermal dysplasia (HED), Rieger’s syndrome, Down’s syndrome, and Van der Woude’s syndrome [14], underscores its significance as a phenotypic marker and a consequence of intricate genetic interactions. Notably, while oligodontia assumes a central role as a diagnostic hallmark for numerous syndromes, additional dental anomalies may co-occur. These anomalies encompass microdontia, shortened roots, dental impactions, delayed tooth formation, delayed eruption, canine and premolar transpositions, taurodontism, and enamel hypoplasia [2,15,16]. Tooth development is orchestrated by conserved signaling pathways that facilitate communication between ectodermal and mesenchymal tissues. In humans, mutations in WNT10A are the most commonly reported in the genetic etiology for syndromic oligodontia, and PAX9 mutations are the most commonly reported genetic etiology for isolated oligodontia [17].
Patients with oligodontia present serious deficiencies in their quality of life due to decreased masticatory function, phonetic ability, and maxillofacial aesthetics [18,19]. As oligodontia can be a clinical manifestation of a large and heterogenous group of syndromes with multiple signs and symptoms, this systematic review aims to summarize the available literature concerning the presence of oligodontia in syndromes, emphasizing the phenotype and the molecular etiology in order to assist in the diagnosis and management of patients.
2. Materials and Methods
2.1. Protocol and Registration
This systematic review was performed according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist [20], and its protocol was recorded in the International Prospective Register of Systematic Reviews (PROSPERO) database under registration number CRD42020190814.
2.2. Eligibility Criteria
The PICOS approach was used to formulate the question for this study: P—participants (syndromic patients with oligodontia); I—intervention (none), C—comparison (none); O—outcomes (frequency and types of syndromes associated with oligodontia, pattern of missing teeth, and frequency of pathogenic variants); S—study (case reports and case series).
The criteria for the exclusion of articles were as follows: (1) non-syndromic cases (n = 8); (2) studies that did not report (or reported unclear) dental X-rays (n = 80); (3) studies that did not report representative cases of oligodontia (n = 30); (4) studies that report cases of hypodontia or anodontia (n = 13); (5) studies that do not include pattern of tooth agenesis (n = 8); (6) reviews, letters, conference abstract, personal opinions, and in vitro or in vivo animal studies (n = 3); (7) full-text copy not available (n = 37); (8) articles that were not in Roman alphabet (n = 2); (9) studies with absence of clinical information (n = 23).
2.3. Study Selection
The selection process of the studies was performed using an individual search in each bibliographic database: PubMed, Scopus, Lilacs, Web of science, Livivo, and EMBASE. A gray literature search was conducted using Google Scholar and ProQuest. The research was performed in December 2022. However, a second literature search was performed using the same terms on 8th August 2021, retrieving articles published between January and August 2023. The search strategy can be assessed in Supplementary Table S1. The duplicate references were removed by reference manager software (EndNote X7, Thomson Reuters, Toronto, ON, Canada). All references were transferred and worked on the Rayyan (Rayyan, Qatar Computing Research Institute, Qatar Foundation, Doha, Qatar), developed specifically to expedite the initial screening of abstracts and titles [21].
The course of the research was established in two distinct phases. In the first phase, two authors (N.L.C. and K.K.M.R.) independently read all titles and abstracts, taking into account the eligibility criteria initially defined. In cases of no consensus, a third author (A.C.A.) was involved who determined which articles would be included in the second phase. The second phase was carried out by the same authors, who performed a full-text reading of the screened articles. Disagreements were solved via discussions involving the third author (A.C.A.).
2.4. Data Collection Process and Data Items
The data collection process was carried out by the two authors (N.L.C. and K.K.M.R.) who initially selected the articles in the two phases, in which the necessary and relevant information of each study was collected. The information was checked by a third author (A.C.A.).
2.5. Risk of Bias within Studies
The risk-of-bias was assessed by two authors (N.L.C. and K.K.M.R.) using the Joanna Briggs Institute Critical Appraisal Tools for Studies Reporting Prevalence Data for Use in Systematic Reviews—referred-to Case Reports [22]. The authors scored each item as “yes”, “no”, “unclear”, or “not applicable” when assessing the quality of each included study. Decisions about scoring were discussed by all reviewers. A study was characterized as having a high risk of bias when it reached a “yes” score of up to 49%, moderate when 50% to 69%, and low when >70%.
2.6. Interaction Analysis
The functional relevance of identified genes was further investigated with STRING, version 11.0 (a search tool for the retrieval of interacting genes, http://string-db.org, accessed on 1 November 2023), which provides a p-value after applying a false discovery rate for correction of multiple testing.
3. Results
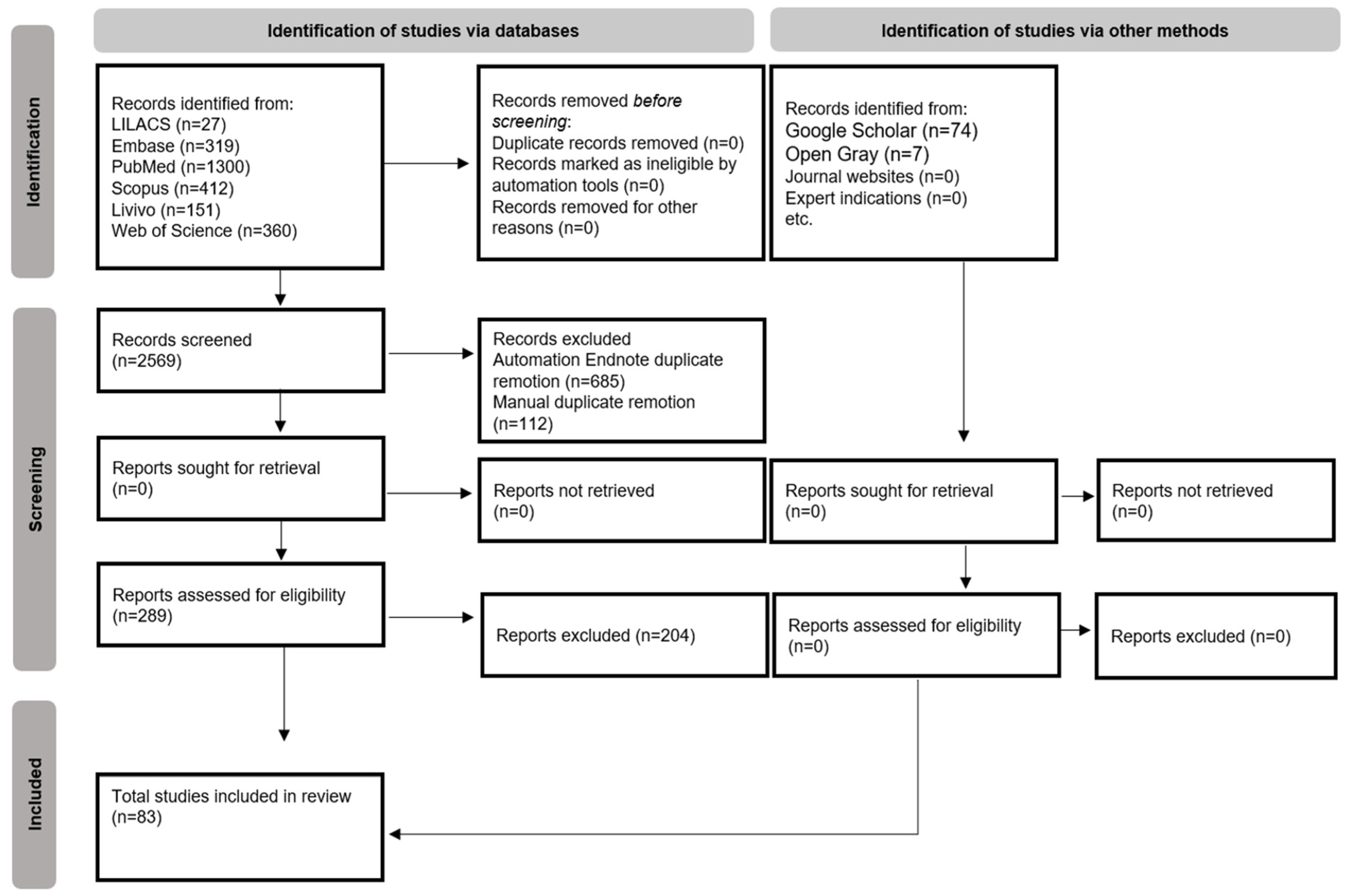
The searches conducted in the six databases resulted in 2569 scientific articles. After removal of duplicates, 1772 articles were totalled. The gray literature search resulted in 81 articles. After reading the titles and abstracts in the first phase, 288 articles were selected for the next phase. At the end of reading the full articles (second phase), 83 articles were included for the qualitative synthesis [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105]. The review process is schematized in a flowchart depicted on Figure 1.
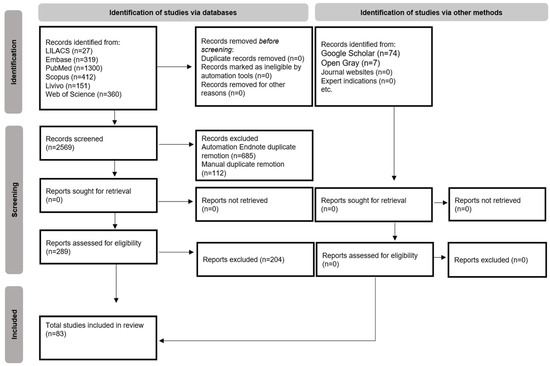
Figure 1.
Flow diagram of literature search and selection criteria adapted via the preferred reporting items for systematic reviews and meta-analyses (PRISMA) [20].
The main data of each selected article are summarized in Table 1. Together, the studies reported 97 patients, ages ranging from 3 to 75 years old, with oligodontia as a clinical feature of different types of syndromes. The risk-of-bias assessment in each study is reported in Supplementary Table S2. All included articles were classified as having a low risk of bias, except for eight articles. These articles had unclear information regarding the demographics of the patients, the timeline of diagnosis, and, most importantly, the diagnostic tests used for the final diagnosis.

Table 1.
Manifestations of oligodontia in primary and permanent dentition in the included articles (n = 83).
The syndromes more frequently identified, in decreasing order, were HED (24 patients in 22 studies), Axenfeld–Rieger syndrome (ARS) (9 patients in 5 studies), blepharocheilodontic syndrome (BCDS) (7 patients in 2 studies), Witkop’s syndrome (4 patients in 4 studies), oculofaciocardiodental syndrome (3 patients in 3 studies), incontinentia pigmenti (3 patients in 3 studies), Hallermann–Streiff syndrome (3 patients in 3 studies), polycistic ovarian syndrome (3 patients in 2 studies), Ellis–van Creveld syndrome (EVCS) (2 patients in 2 studies), Down’s syndrome (2 patients in 2 studies), Carvajal syndrome (2 patients in 2 studies), Carpenter syndrome (2 patients in 2 studies), and Kabuki syndrome (2 patients in 2 studies). Other uncommon syndromes listed in Table 1 were reported in one patient each.
Out of 97 patients, 10 patients were affected by oligodontia in both primary and permanent dentitions; in 2 cases, oligodontia was observed in the primary dentition; and in the other cases, oligodontia was observed in the permanent dentition (Table 1). The number of missing teeth ranged from 6 to 13 in the primary dentition and from 6 to 27 in the permanent dentition. In the deciduous dentition, the absence of the first molars and lateral and central incisors was observed in 76.9% of patients, and the second molars and canines were absent in 61.5% of patients (Table 1). In the permanent dentition, lateral incisors (76.2% of patients), first molars (75.5% of patients), central incisors (69.7% of patients), premolars (66% of patients), and second molars (40% of patients) were the more-affected teeth (Table 1).
The genetic profile of the different studies is summarized in Table 1. HED was the most-found syndrome, and the X-linked mode of inheritance was the most common for this syndrome. The most reported mode of inheritance for oculofaciocardiodental syndrome, incontinentia pigmenti and Christ–Siemens–Touraine syndrome was X-linked. The mode of inheritance for Carvajal syndrome, Noonan syndrome, ARS, Witkop’s syndrome, Apert syndrome, BCDS, and Kabuki syndrome was the autosomal dominant, whereas microcephalic osteodyplastic primordial dwarfism type II, tricho-odonto-onychodermal dysplasia, and EVCS were reported under autosomal recessive (Table 1).
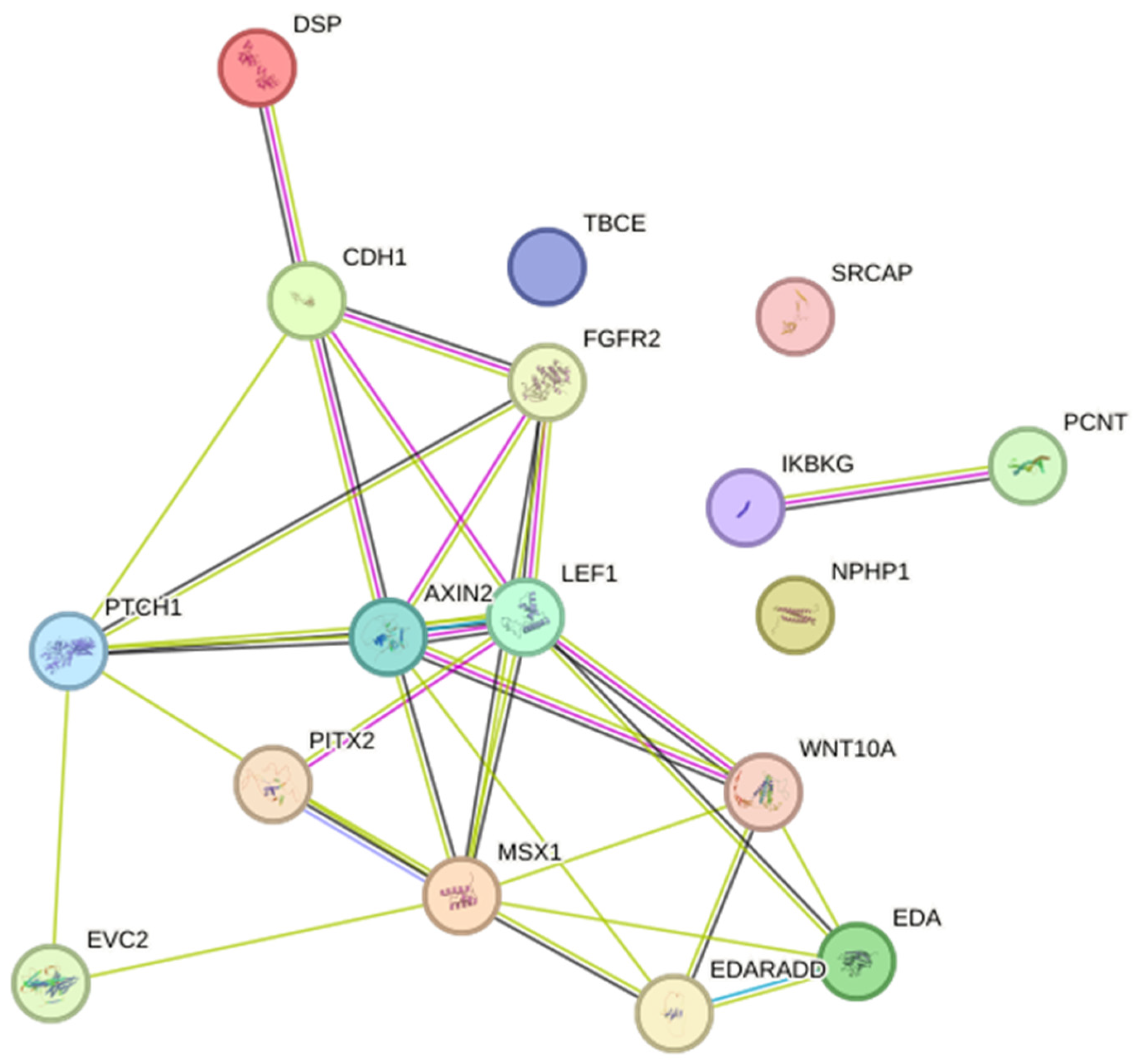
Seven studies reported mutations in EDA or WNT10A in HED, Christ–Siemens–Touraine syndrome, tricho-odonto-onychodermal dysplasia, and odonto-onychodermal dysplasia. Others studies have reported mutations in AXIN2 (HED), CDH1 (blepharocheilodontic syndrome), DSP (Carvajal/Naxos syndrome), EDARADD (HED), EVC2 (EVCS), FGFR2 (Beare–Stevenson syndrome), PCNT (microcephalic osteofysplastic dwarfism type 2), PITX2 (ARS), PTCH1 (basal cell nevus syndrome), IKBKG (incontinentia pigmenti), LEF1 (HED), MSX1 (Witkop’s syndrome), NPHP1 (juvenile nephronophtisis), SRCAP (Floating-Harbor syndrome), and TBCE (Sanjad–Sakati syndrome). Together, these genes participate in 80 biological processes and 19 pathways. The most significant biological processes were odontogenesis (GO:0042476; p = 1.56 × 10−7), gland development (GO:0048732; p = 7.29 × 10−6) and epithelium development (GO:0060429; p = 1.65 × 10−5), and the pathways were of pathways in cancer (hsa05200; p = 4.55 × 10−5), basal cell carcinoma (hsa05217; p = 4.55 × 10−5), and pathways of the gastric cancer (hsa05226; p = 4.55 × 10−5) (Supplementary Tables S3 and S4). The networks included 17 predicted interactions (Figure 2).
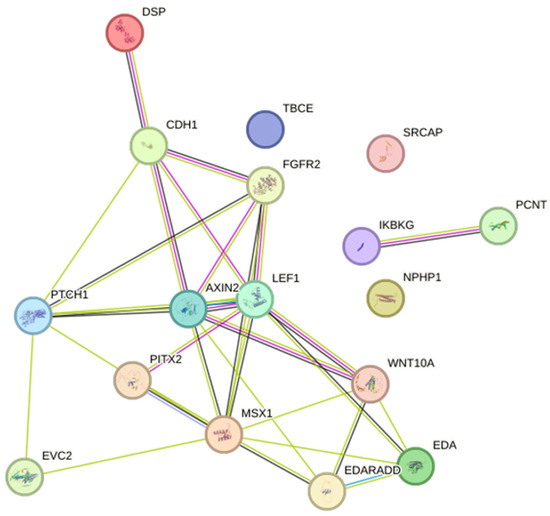
Figure 2.
Protein–protein interaction network with the genes associated with syndromes with oligodontia. Two nodes of interactions, involving AXIN2, CDH1, DSP, EDA, EDARADD, EVC2, FGFR2, LEF1, MSX1, PTCH1, PITX2, and WNT10A (p < 1.0 × 10−16) and between PCNT and IKBKG (p = 0.04) were identified. Different colors represent different levels of evidence of connection between proteins; light blue represents curated databases, purple experimental evidence, green gene neighborhood, light green evidence from text mining, black co-expression, and violet protein homology. This analysis had an average confidence score of 0.657, suggesting a low rate for false-positive interactions.
4. Discussion
Within the realm of syndromes characterized by oligodontia, questions arise regarding the consistency of this phenotype across cases, its varying expressiveness, its diagnostic utility, and the specific teeth most affected. This review sought to comprehensively address these queries by collating pertinent information from a diverse array of syndromes exhibiting oligodontia within their clinical spectrum. The exploration commenced by surveying the literature and transcending temporal constraints, leading to the identification of 47 distinct syndromes cataloged within the Online Mendelian Inheritance in Man. Among these, HED emerged as the most frequent. The hallmark trifecta of HED, involving hair, teeth, and sweat gland anomalies [106], was evident in the affected patients. The spectrum of dental agenesis in HED spans mainly oligodontia, but reports of hypodontia and even anodontia are found in the literature, with a predilection for the mandible. This remarkable variability necessitates close attention for correct diagnosis [107,108]. Notably, the distinctive conical shape of the anterior teeth, when present, offers a diagnostic clue. Furthermore, the potential confluence of maxillary retrusion, sagittal jaw underdevelopment, jaw displacement, and craniofacial alterations underlines the complex interplay of factors characterizing this syndrome [107,108,109].
Within our systematic review, several syndromes stood out for their prevalence and diverse oral manifestations. One of the noteworthy conditions identified was Ellis–van Creveld syndrome (EVCS), an autosomal recessive skeletal dysplasia. EVCS presents a range of oral phenotypes alongside limb abnormalities, including occlusion irregularities, labiogingival adhesions, hypertrophied labiogingival frenulum, accessory frenula, serrated incisal margins, dental transposition, diastemas, conical teeth, enamel hypoplasia, and congenital absence of multiple teeth [110,111]. The propensity for premature eruption or exfoliation further complicates the dental anomalies associated with EVCS. In our study, we observed an average of 6 deciduous teeth (one case) and 8.5 permanent teeth (two cases) missing in individuals with reported EVC2 mutations. Another syndrome we examined was oculofaciocardiodental syndrome, a rare multi-systemic anomaly, with a higher prevalence in females. This syndrome showcases the intricate interplay between congenital cataracts, facial dysmorphisms, and dental anomalies, including radiculomegaly and oligodontia, in addition to congenital heart defects [77,112]. In our study, we noted an average of nine permanent teeth (three cases) missing in individuals with reported WNT10A mutations. Witkop syndrome, an uncommon autosomal dominant genetic disorder attributed to mutations in MSX1, held our attention due to its distinct dental and nail dysplasia characteristics. This syndrome is primarily marked by dental agenesis, primarily oligodontia, although the absence of up to five teeth (hypodontia) is also reported. Dental features often include conical-shaped teeth and teeth with narrow crowns [113]. In our study, we observed an average of 16.75 permanent teeth (four cases) missing in individuals with reported MSX1 mutations. Axenfeld–Rieger syndrome (ARS), a condition characterized by ocular dysgenesis and systemic anomalies affecting the teeth, heart, craniofacial structure, and abdominal wall, was another significant focus. ARS is frequently associated with a 6p25 distal microdeletion but may also manifest in connection with other genetic loci such as 4q25 or 13q14. Several genes, including FOXC1, FOXC2, and FKHL7, have been implicated in the context of ARS [29,114]. In our study, we found an average of 9 primary teeth (one case) and 14.3 permanent teeth (nine cases) missing in individuals with reported PITX2 mutations. Lastly, we explored branchio-oculofacial syndrome (BCDS), an autosomal dominant disorder known for congenital facial clefting, oligodontia, euryblepharon, lagophthalmos, and ectropion. While the extent of its expression can vary, the common features often encompass cleft lip and/or palate, ectropion, and lagophthalmos [76,115]. In our study, we observed an average of 8.5 primary teeth (four cases) and 15.4 permanent teeth (seven cases) missing in individuals with reported CDH1 mutations.
Interrogating the consistency of oligodontia across syndromic cases is of paramount importance. The range of dental presentations, spanning from hypodontia to anodontia, underscores the variable expressiveness within these syndromes. Consequently, the manifestation of oligodontia should be viewed as a continuum instead of an absolute trait. This variable expressivity poses challenges in diagnosis and underscores the importance of considering broader phenotypic traits in conjunction with dental anomalies. Regarding the most-affected teeth, patterns emerged from the collated data. In deciduous dentition, absence of first molars and lateral and central incisors was observed in the majority of patients, with second molars and canines affected in a significant proportion. In permanent dentition, lateral incisors, first molars, central incisors, premolars, and second molars exhibited the highest susceptibility to oligodontia. HED is the most frequently mentioned syndrome with oligodontia, while EDA and WNT10A mutations constitute the most frequently determined genetic cause: 30.4% of the syndromic oligodontia. These findings are consistent with previous reports which established WNT10A variants accounting for up to 50% of various HED syndromes with missing teeth [17,116]. These patterns may offer clues for diagnosis and genetic assessment. However, as previously reported, there are several mechanisms that are involved in tooth development and other tissues of the body, establishing very heterogeneous phenotypes in affected individuals. Thus, radiographic and molecular diagnoses may be necessary. The analysis of dental radiographs is an important part of the diagnostic process in daily clinical practice, and interpretation by an expert includes teeth detection and numbering [117,118,119]. In the reading of the articles included, panoramic radiographs with poor quality were evidenced, which can affect the diagnosis and consequent interpretation of the case report. The detailed radiographic report of the observed alterations was also absent in most studies.
In some situations, in the differential diagnosis process, sequencing analysis is useful, and further exploration of the identified mutations can assist in the interpretation of the phenotypes (genotype–phenotype correlation). The important role of genetics has been increasingly recognized in recent years with regard to the understanding of dental anomalies such as tooth agenesis [120]. However, many of the included studies in this systematic review did not perform molecular analysis. Only 23 studies [41,43,52,53,56,63,66,67,68,70,73,74,76,78,79,87,88,89,90,92,94,98,105] performed genetic analysis and reported the genetic variants associated with the syndromes. These genes can be grouped into two major groups: one with crucial roles at multiple stages of tooth development, also involving skin and sweat glands (AXIN2, CDH1, DSP, EDA, EDARADD, EVC2, FGFR2, LEF1, MSX1, PITX2, and WNT10A), which are involved in the signal pathway essential for ectodermal structure development [121,122]; the other with genes that intermediate cellular function and development (NPHP1, PCNT, PTCH1, IKBKG, SRCAP, and TBCE). Interestingly, some of the identified genes are also associated with non-syndromic oligodontia, but in these cases, the mutations cause reduced expression, decreased receptor-binding affinity, or altered signaling-intensity of the mutated protein, whereas the mutations associated with syndromic oligodontia are characterized by a more intense impact on protein function [123].
The teeth affected by oligodontia often exhibit various dental anomalies, such as reduced size, a conical shape, and delayed eruption [12,121,122,123,124,125,126]. These shared features suggest the influence of similar genetic mechanisms [124]. Our findings align with previous research [120], indicating that permanent dentition is more frequently affected by oligodontia compared to primary dentition. In our study of 97 patients, we observed bilateral agenesis of maxillary lateral incisors in 64 patients, while 13 patients displayed unilateral absence of the second mandibular premolar. It is worth noting that clinical studies have reported that bilateral agenesis of the maxillary lateral incisors is more common than unilateral agenesis, and unilateral agenesis of the second mandibular premolar is more prevalent than bilateral cases [127].
However, it is important to acknowledge the limitations of this study. One significant limitation arises from the varied terminology used to define oligodontia, including terms such as severe hypodontia or partial anodontia. This inconsistency in nomenclature can lead to challenges in accurately classifying cases. Additionally, the absence of radiographic data for correct oligodontia diagnosis was a noteworthy limitation that impacted our sample size. Many studies included in this review did not perform genetic tests, which precluded a more comprehensive phenotype–genotype correlation analysis. Furthermore, some very rare disorders may not have been reported in the literature, potentially limiting the scope of our review with regard to syndromes associated with oligodontia.
In addition to its academic significance, the findings from this review hold substantial implications for clinical practice. Identifying specific dental anomalies and their association with syndromes can play a pivotal role in the diagnostic process. When assessing patients with oligodontia, it is essential for clinicians to consider the potential presence of an underlying syndrome. The number and type of missing or affected teeth can serve as valuable diagnostic clues, but a holistic evaluation of the patient’s overall clinical presentation, including physical and developmental features, is indispensable. Furthermore, the genetic insights garnered from this review offer the potential for more precise and personalized patient care. Understanding the genetic underpinnings of syndromic oligodontia can facilitate early diagnosis, inform genetic counseling, and guide treatment planning. Genetic testing, where applicable, can provide confirmation and assist in tailoring therapeutic strategies. This knowledge can also pave the way for the development of targeted therapies or interventions with which to address the dental and craniofacial challenges associated with syndromic oligodontia.
Looking ahead, there is a compelling need for further research in this field. Delving deeper into the genetic underpinnings of syndromic oligodontia and exploring the phenotypic heterogeneity in greater detail are paramount. Studies that employ consistent terminology and diagnostic methods will contribute to a more comprehensive understanding of this complex condition. Additionally, further investigations into genotype–phenotype correlations and the development of standardized diagnostic criteria for syndromic oligodontia are warranted. Comprehensive radiographic assessment and molecular analysis will be instrumental in elucidating the underlying mechanisms and genetic factors associated with these syndromes.
5. Conclusions
The identification of specific phenotypes associated with oligodontia can significantly reduce diagnostic uncertainty, particularly when these phenotypes are highly predictive of a specific syndrome. Based on the insights gained from this review, it is advisable that clinicians and geneticists, during the diagnosis process of patients with oligodontia, remain vigilant for the most common syndromes with oligodontia as a clinical sign. These syndromes include hypohidrotic ectodermal dysplasia (HED), Axenfeld–Rieger Syndrome (ARS), Witkop’s syndrome, Ellis–van Creveld syndrome (EVCS), Branchio-oculofacial syndrome (BCDS), and oculofaciocardiodental syndrome. These findings contribute to a more informed and precise diagnostic approach, ultimately enhancing patient care and management.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/dj11120279/s1: Supplementary Table S1. Search strategies with appropriate key words and MeSH terms. Supplementary Table S2. Risk of bias assessment of the individual articles included (n:83). Risk of bias was categorized as High when the study reaches up to 49% score “yes”, Moderate when the study reached 50% to 69% score “yes”, and Low when the study reached more than 70% score “yes”. Supplementary Table S3. Biological processes characterized by the list of altered genes in syndromes with oligodontia. Supplementary Table S4. Activated pathways characterized with syndromes with oligodontia.
Author Contributions
N.L.C.: data extraction, methodology, writing—original draft; K.K.M.R.: data extraction, methodology, writing—original draft; J.A.d.S.: data extraction, methodology; R.A.M.: data extraction, methodology, supervision, writing—original draft and editing; R.D.C.: conceptualization, supervision, writing—review and editing; E.N.S.G.: conceptualization, methodology; A.C.A.: conceptualization, data extraction, methodology; H.M.-J.: conceptualization, project administration, validation, data curation, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
The study was supported by the Minas Gerais State Research Foundation-FAPEMIG, Brazil, National Council for Scientific and Technological Development—CNPq, Brazill and the Coordination for the Improvement of Higher Education Personnel, CAPES, Brazil.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- de La Dure-Molla, M.; Fournier, B.P.; Manzanares, M.C.; Acevedo, A.C.; Hennekam, R.C.; Friedlander, L.; Boy-Lefèvre, M.L.; Kerner, S.; Toupenay, S.; Garrec, P.; et al. Elements of morphology: Standard terminology for the teeth and classifying genetic dental disorders. Am. J. Med. Genet. Part A 2019, 179, 1913–1981. [Google Scholar] [CrossRef] [PubMed]
- Weide, Y.S.; Beemer, F.A.; Faber, J.A.; Bosman, F. Symptomatology of patients with oligodontia. J. Oral Rehabil. 1994, 21, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Zu, X.; Shen, Y.; Zheng, J. [Progress in genetic research on tooth agenesis associated with Wnt/beta-catenin signaling pathway]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2021, 38, 506–509. [Google Scholar]
- Al-Ani, A.H.; Antoun, J.S.; Thomson, W.M.; Merriman, T.R.; Farella, M. Hypodontia: An Update on Its Etiology, Classification, and Clinical Management. BioMed Res. Int. 2017, 2017, 9378325. [Google Scholar] [CrossRef]
- Brook, H. Multilevel complex interactions between genetic, epigenetic and environmental factors in the aetiology of anomalies of dental development. Arch. Oral Biol. 2009, 54, S3–S17. [Google Scholar] [CrossRef]
- Nieminen, P.; Arte, S.; Pirinen, S.; Peltonen, L.; Thesleff, I. Gene defect in hypodontia: Exclusion of MSX1 and MSX2 as candidate genes. Hum. Genet. 1995, 96, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Gorlin, R.J. Syndromes of Head and Neck, 2nd ed.; Oxford University Press: Oxford, MS, USA, 2001. [Google Scholar]
- Vieira, A.R. Oral clefts and syndromic forms of tooth agenesis as model for genetics of isolated tooth agenesis. J. Dent. Res. 2003, 82, 162–165. [Google Scholar] [CrossRef]
- Baba, R.; Sato, A.; Arai, K. Consecutive tooth agenesis patterns in non-syndromic oligodontia. Odontology 2022, 110, 183–192. [Google Scholar] [CrossRef]
- Ritwik, P.; Partsson, K.K. Diagnosis of Tooth Agenesis in Childhood and Risk for Neoplasms in Adulthood. Ochsner J. 2018, 18, 345–350. [Google Scholar] [CrossRef]
- Rolling, S.; Poulsen, S. Oligodontia in Danish schoolchildren. Acta Odontol. Scand. 2001, 59, 111–112. [Google Scholar] [CrossRef]
- Nieminen, P. Genetic basis of tooth agenesis. J. Exp. Zool. Part B Mol. Dev. Evol. 2009, 312, 320–342. [Google Scholar] [CrossRef] [PubMed]
- Laganà, G.; Venza, N.; Borzabadi-Farahani, A.; Fabi, F.; Danesi, C.; Cozza, P. Dental anomalies: Prevalence and associations between them in a large sample of non-orthodontic subjects, a cross-sectional study. BMC Oral Health 2017, 17, 62. [Google Scholar] [CrossRef] [PubMed]
- Klein, O.D.; Oberoi, S.; Huysseune, A.; Hovorakova, M.; Peterka, M.; Peterkova, R. Developmental disorders of the dentition: An update. Am. J. Med. Genet. Part C Semin. Med. Genet. 2013, 163, 318–332. [Google Scholar] [CrossRef]
- Adaimy, L.; Chouery, E.; Mégarbané, H.; Mroueh, S.; Delague, V.; Nicolas, E.; Belguith, H.; de Mazancourt, P.; Mégarbané, A. Mutation in WNT10A Is Associated with an Autosomal Recessive Ectodermal Dysplasia: The odonto-onycho-dermal dysplasia. Am. J. Hum. Genet. 2007, 81, 821–828. [Google Scholar] [CrossRef] [PubMed]
- De Coster, P.J.; Marks, L.A.; Martens, L.C.; Huysseune, A. Dental agenesis: Genetic and clinical perspectives. J. Oral Pathol. Med. 2009, 38, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Zhang, H.; Camhi, H.; Seymen, F.; Koruyucu, M.; Kasimoglu, Y.; Kim, J.W.; Kim-Berman, H.; Yuson, N.M.; Benke, P.J.; et al. Analyses of oligodontia phenotypes and genetic etiologies. Int. J. Oral Sci. 2021, 13, 32. [Google Scholar] [CrossRef] [PubMed]
- Nordgarden, H.; Jensen, J.; Storhaug, K. Oligodontia is associated with extra-oral ectodermal symptoms and low whole salivary flow rates. Oral Dis. 2001, 7, 226–232. [Google Scholar] [CrossRef]
- Dhamo, B.; Kuijpers, M.A.R.; Balk-Leurs, I.; Boxum, C.; Wolvius, E.B.; Ongkosuwito, E.M. Disturbances of dental development distinguish patients with oligodontia-ectodermal dysplasia from isolated oligodontia. Orthod. Craniofacial Res. 2018, 21, 48–56. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetc, R.; Currie, M.; Lisy, K.; Qureshi, R.; Mattis, P.; et al. Systematic reviews of etiology and risk. In Joanna Briggs Institute Reviewer’s Manual; Aromataris, E., Munn, Z., Eds.; Joanna Briggs Institute: Adelaide, Australia, 2017; Chapter 7. [Google Scholar]
- Abdulla, A.M.; Almaliki, A.Y.; Shakeela, N.V.; Alkahtani, Z.; Alqahtani, M.A.; Sainudeen, S.; Shamsuddin, S.V. Prosthodontic Management of a Pediatric Patient with Christ-Siemens-Touraine Syndrome: A Case Report. Int. J. Clin. Pediatr. Dent. 2019, 12, 569–572. [Google Scholar]
- Abs, R.; Raes, D.; Vercruysse, H. Association of isolated hypogonadotropic hypogonadism, pronounced hypodontia and the Wolff-Parkinson-White syndrome. J. Int. Med. 1994, 236, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Aditya, A.; Lele, S.; Aditya, P. Fahr’s disease with oral manifestations: Report of a rare case. Med. Princ. Pract. 2012, 21, 395–397. [Google Scholar] [CrossRef]
- Agarwal, S.S.; Nehra, K.; Jayan, B.; Singh, N.; Thapa, A. Multidisciplinary management of oligodontia polycystic ovarian syndrome: A rare case report. Int. J. Oral Health Sci. Adv. 2014, 2, 33–41. [Google Scholar]
- Aminabadi, N.A.; Ebrahimi, A.; Oskouei, S.G. Chondroectodermal dysplasia (Ellis-van Creveld syndrome): A case report. J. Oral Sci. 2010, 52, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Ann Drum, M.; Kaiser-Kupfer, M.I.; Guckes, A.D.; Roberts, M.W. Oral manifestations of the Rieger syndrome: Report of case. J. Am. Dent. Assoc. (1939) 1985, 110, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Ardila, C.M.; Álvarez-Martínez, E. Dental and Maxillofacial Manifestations of Axenfeld-Rieger Syndrome: Presentation of a Case in a 5-Year-Old Girl. Case Rep. Dent. 2022, 2022, 4348264. [Google Scholar] [CrossRef]
- Arora, V.; Agrawal, K.K.; Mishra, A.; Chandra, A. Witkop’s syndrome: A case report. J. Oral Biol. Craniofacial Res. 2016, 6, 79–81. [Google Scholar] [CrossRef]
- Awadh, W.; Kiukkonen, A.; Nieminen, P.; Arte, S.; Hurmerinta, K.; Rice, D.P. Blepharocheilodontic (BCD) syndrome: New insights on craniofacial and dental features. Am. J. Med. Genet. A 2017, 173, 905–913. [Google Scholar] [CrossRef]
- Barber, S.; Day, P.; Judge, M.; Toole, E.O.; Fayle, S. Variant Carvajal syndrome with additional dental anomalies. Int. J. Paediatr. Dent. 2012, 22, 390–396. [Google Scholar] [CrossRef]
- Bekiesinska-Figatowska, M.; Mierzewska, H.; Kuczynska-Zardzewialy, A.; Szczepanik, E.; Obersztyn, E. Hypomyelination, hypogonadotropic hypogonadism, hypodontia—First Polish patient. Brain Dev. 2010, 32, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Bergendal, B.; Bjerklin, K.; Bergendal, T.; Koch, G. Dental Implant Therapy for a Child with X-linked Hypohidrotic Ectodermal Dysplasia—Three Decades of Managed Care. Int. J. Prosthodont. 2015, 28, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Bergendal, B. Prosthetic habilitation of a young patient with hypohidrotic ectodermal dysplasia and oligodontia: A case report of 20 years of treatment. Int. J. Prosthodont. 2001, 14, 471–479. [Google Scholar]
- Bildik, T.; Ozbaran, B.; Kose, S.; Koturoglu, G.; Gokce, B.; Gunaydin, A.; Altintas, I. Hypohidrotic ectodermal dysplasia: A multidisciplinary approach. Int. J. Psychiatry Med. 2012, 44, 225–240. [Google Scholar] [CrossRef] [PubMed]
- Blankenstein, R.; Brook, A.H.; Smith, R.N.; Patrick, D.; Russell, J.M. Oral findings in Carpenter syndrome. Int. J. Paediatr. Dent. 2001, 11, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Cagetti, M.G.; Camoni, N.; Cetraro, F.; Scanferla, M.; Moretti, G.M. Special-needs patients in pediatric dentistry: Progeroid syndrome. A case of dental management and oral rehabilitation. Pediatr. Rep. 2019, 11, 7951. [Google Scholar] [CrossRef]
- Callanan, A.P.; Anand, P.; Sheehy, E.C. Sotos syndrome with hypodontia. Int. J. Paediatr. Dent. 2006, 16, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, V.M.; Romero, J.S.S.; Paiva Correa de Melo, F.; Fonseca Faro, T.; Nunes Santos, A.C.; Carvalho, S.; Veras Sobral, A.P. Hypohidrotic and hidrotic ectodermal dysplasia: A report of two cases. Dermatol. Online J. 2013, 19, 18985. [Google Scholar]
- Chalabreysse, L.; Senni, F.; Bruyère, P.; Aime, B.; Ollagnier, C.; Bozio, A.; Bouvagnet, P. A new hypo/oligodontia syndrome: Carvajal/Naxos syndrome secondary to desmoplakin-dominant mutations. J. Dent. Res. 2011, 90, 58–64. [Google Scholar] [CrossRef]
- Cho, S.Y.; Lee, C.K.; Drummond, B.K. Surviving male with incontinentia pigmenti: A case report. Int. J. Paediatr. Dent. 2004, 14, 69–72. [Google Scholar] [CrossRef]
- Clauss, F.; Waltmann, E.; Barriere, P.; Hadj-Rabia, S.; Manière, M.C.; Schmittbuhl, M. Dento-maxillo-facial phenotype and implants-based oral rehabilitation in Ectodermal Dysplasia with WNT10A gene mutation: Report of a case and literature review. J. Cranio-Maxillofac. Surg. 2014, 42, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Cogulu, D.; Ertugrul, F. Dental management of a patient with oculo-facio-cardio-dental syndrome. J. Dent. Child. 2008, 75, 306–308. [Google Scholar]
- Cogulu, D.; Oncag, O.; Celen, E.; Ozkinay, F. Kabuki Syndrome with additional dental findings: A case report. J. Dent. Child. 2008, 75, 185–187. [Google Scholar]
- Costa, F.; Sarno, M.; Khouri, R. Emergence of Congenital Zika Syndrome: Viewpoint from the Front Lines. Ann. Intern. Med. 2016, 164, 689–691. [Google Scholar] [CrossRef]
- Dall’Oca, S.; Ceppi, E.; Pompa, G.; Polimeni, A. X-linked hypohidrotic ectodermal dysplasia: A ten-year case report and clinical considerations. Eur. J. Paediatr. Dent. 2008, 9, 14–18. [Google Scholar] [PubMed]
- Devadas, S.; Varma, B.; Mungara, J.; Joseph, T.; Saraswathi, T.R. Witkop tooth and nail syndrome: A case report. Int. J. Paediatr. Dent. 2005, 15, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Downing, A.; Welbury, R.R. The tricho-rhino-phalangeal syndrome—A case report. Int. J. Paediatr Dent. 1992, 2, 35–40. [Google Scholar] [CrossRef]
- Dunbar, A.C.; McIntyre, G.T.; Laverick, S.; Stevenson, B. Axenfeld-Rieger syndrome: A case report. J. Orthod. 2015, 42, 324–330. [Google Scholar] [CrossRef]
- Emral, M.E.; Akcam, M.O. Noonan syndrome: A case report. J. Oral Sci. 2009, 51, 301–306. [Google Scholar] [CrossRef][Green Version]
- Fan, Z.; Sun, S.; Liu, H.; Yu, M.; Liu, Z.; Wong, S.W.; Liu, Y.; Han, D.; Feng, H. Novel PITX2 mutations identified in Axenfeld-Rieger syndrome and the pattern of PITX2-related tooth agenesis. Oral Dis. 2019, 25, 2010–2019. [Google Scholar] [CrossRef]
- Ghosh, S.; Garg, M.; Gupta, S.; Choudhary, M.; Chandra, M. Microcephalic osteodyplastic primordial dwarfism type II: Case report with unique oral findings and a new mutation in the pericentrin gene. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 129, e204–e211. [Google Scholar] [CrossRef]
- Gupta, S.R. Isolated aglossia congenita: A rare case of oromandibular limb hypogenesis syndrome type IB. J. Oral Maxillofac. Pathol. 2012, 16, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Govind, M.; Sawai, M.A.; Ansari, M.D. Hypohidrotic ectodermal dysplasia with autosomal recessive inheritance pattern: Report of a rare and unusual case with a brief review of literature. J. Oral Maxillofac. Pathol. 2019, 23, 479. [Google Scholar] [PubMed]
- Hassona, Y.; Rajab, L.; Taimeh, D.; Scully, C. Sanjad-Sakati Syndrome: Oral Health Care. Med. Princ. Pract. 2018, 27, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Hattab, F.N.; Angmar-Månsson, B. Oligodontia of the permanent dentition in two sisters with polycystic ovarian syndrome: Case reports. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1997, 84, 368–371. [Google Scholar] [CrossRef]
- Hattab, F.N.; al-Khateeb, T.; Mansour, M. Oral manifestations of severe short-limb dwarfism resembling Grebe chondrodysplasia: Report of a case. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1996, 81, 550–555. [Google Scholar] [CrossRef]
- Hingston, E.J.; Hunter, M.L.; Hunter, B.; Drage, N. Hurler’s syndrome: Dental findings in a case treated with bone marrow transplantation in infancy. Int. J. Paediatr. Dent. 2006, 16, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Jain, S.; Kumar, A.; Rahangdale, T. Hypohidrotic Ectodermal Dysplasia: Prosthetic and Endodontic Management. Int. J. Clin. Pediatr. Dent. 2010, 3, 63–67. [Google Scholar] [CrossRef]
- Jain, N.; Naitam, D.; Wadka, A.; Nemane, A.; Katoch, S.; Dewangan, A. Prosthodontic rehabilitation of hereditary ectodermal dysplasia in an 11-year-old patient with flexible denture: A case report. Case Rep. Dent. 2012, 2012, 489769. [Google Scholar] [CrossRef]
- Kale, L.; Khambete, N.; Sodhi, S.; Kumar, R. Achondroplasia with oligodontia: Report of a rare case. J. Oral Maxillofac. Pathol. 2013, 17, 451–454. [Google Scholar]
- Kantaputra, P.; Kaewgahya, M.; Jotikasthira, D.; Kantaputra, W. Tricho-odonto-onycho-dermal dysplasia and WNT10A mutations. Am. J. Med. Genet. Part A 2014, 164, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Reddy, R. Prosthetic rehabilitation of an adolescent with hypohidrotic ectodermal dysplasia with partial anodontia: Case report. J. Indian Soc. Pedod. Prev. Dent. 2008, 26, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, T.; Motohashi, N.; Ohyama, K. A case of oculo-facio-cardio-dental syndrome with integrated orthodontic-prosthodontic treatment. Cleft Palate-Craniofacial J. Off. Publ. Am. Cleft Palate-Craniofacial Assoc. 2004, 41, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Khabour, O.F.; Mesmar, F.S.; Al-Tamimi, F.; Al-Batayneh, O.B.; Owais, A.I. Missense mutation of the EDA gene in a Jordanian family with X-linked hypohidrotic ectodermal dysplasia: Phenotypic appearance and speech problems. Genet. Mol. Res. 2010, 9, 941–948. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khurana, V.K.; Gupta, R.K.; Kumar, L.P. Witkop syndrome: A case report of an affected family. Dermatol. Online J. 2012, 18, 2. [Google Scholar] [CrossRef] [PubMed]
- Kinyó, A.; Vályi, P.; Farkas, K.; Nagy, N.; Gergely, B.; Tripolszki, K.; Torok, D.; Bata-Csorgo, Z.; Kemény, L.; Szell, M. A newly identified missense mutation of the EDA1 gene in a Hungarian patient with Christ-Siemens-Touraine syndrome. Arch. Dermatol. Res. 2014, 306, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Kishore, M.; Panat, S.R.; Aggarwal, A.; Agarwal, N.; Upadhyay, N.; Ajai, K.; Alok, A. Hypohidrotic Ectodermal Dysplasia (ED): A Case Series. J. Clin. Diagn. Res. 2014, 8, 273–275. [Google Scholar]
- Kobayashi, T.; Iida, A.; Narimatsu, N.; Shimomura, Y. A case of multiple oral cancers in the patient with hypohidrotic ectodermal dysplasia. J. Oral Maxillofac. Surg. Med. Pathol. 2022, 34, 759–763. [Google Scholar] [CrossRef]
- Kozma, C.; Chong, S.S.; Meck, J.M. Interstitial deletion of 4p15.32p16.3 in a boy with minor anomalies, hearing loss, borderline intelligence, and oligodontia. Am. J. Med. Genet. 1999, 86, 316–320. [Google Scholar] [CrossRef]
- Kramer, F.J.; Baethge, C.; Tschernitschek, H. Implants in children with ectodermal dysplasia: A case report and literature review. Clin. Oral Implant. Res. 2007, 18, 140–146. [Google Scholar] [CrossRef]
- Krøigård, A.B.; Thomassen, M.; Lænkholm, A.V.; Kruse, T.A.; Larsen, M.J. Evaluation of Nine Somatic Variant Callers for Detection of Somatic Mutations in Exome and Targeted Deep Sequencing Data. PLoS ONE 2016, 11, e0151664. [Google Scholar] [CrossRef] [PubMed]
- Lévy, J.; Capri, Y.; Rachid, M.; Dupont, C.; Vermeesch, J.R.; Devriendt, K.; Verloes, A.; Tabet, A.C.; Bailleul-Forestier, I. LEF1 haploinsufficiency causes ectodermal dysplasia. Clin. Genet. 2020, 97, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Liedén, A.; Kvarnung, M.; Nilssson, D.; Sahlin, E.; Lundberg, E.S. Intragenic duplication—A novel causative mechanism for SATB2-associated syndrome. Am. J. Med. Genet. Part A 2014, 164, 3083–3087. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Liu, Y.; Su, L.; Liu, H.; Feng, H.; Yu, M.; Liu, H. A Novel CDH1 Variant Identified in a Chinese Family with Blepharocheilodontic Syndrome. Diagnostics 2022, 12, 2936. [Google Scholar] [CrossRef] [PubMed]
- Martinho, J.; Ferreira, H.; Paulo, S.; Paula, A.; Marto, C.M.; Carrilho, E.; Marques-Ferreira, M. Oculo-Facio-Cardio-Dental Syndrome: A Case Report about a Rare Pathological Condition. Int. J. Environ. Res. Public Health 2019, 16, 928. [Google Scholar] [CrossRef] [PubMed]
- Marvin, M.L.; Mazzoni, S.M.; Herron, C.M.; Edwards, S.; Gruber, S.B.; Petty, E.M. AXIN2-associated autosomal dominant ectodermal dysplasia and neoplastic syndrome. Am. J. Med. Genet. Part A 2011, 155, 898–902. [Google Scholar] [CrossRef]
- Murata, Y.; Kurosaka, H.; Ohata, Y.; Aikawa, T.; Takahata, S.; Fujii, K.; Miyashita, T.; Morita, C.; Inubushi, T.; Kubota, T.; et al. A novel PTCH1 mutation in basal cell nevus syndrome with rare craniofacial features. Hum. Genome Var. 2019, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- O’Dwyer, E.M.; Jones, D.C. Dental anomalies in Axenfeld-Rieger syndrome. Int. J. Paediatr. Dent. 2005, 15, 459–463. [Google Scholar] [CrossRef]
- Pipa, A.V.; López, E.M.A.; González, M.G.; Martínez, M.F.; Blanco, F.M.A.B. Treatment with removable prosthesis in hypohidrotic ectodermal dysplasia: A clinical case. Med. Oral Patol. Oral Cir. Bucal 2008, 13, E119–E123. [Google Scholar]
- Reiche, W.; Parkanyi, L.; Braunitzer, G.; Nagy, K.; Karpati, K. Oligodontia in a teenager with Down syndrome—A case study. Klin. Padiatr. 2015, 227, 35–37. [Google Scholar] [CrossRef]
- Retna, R.N.; Sockalingam, S. Clinical manifestation of blepharocheilodontic (BCD) syndrome: A case review. Sains Malays. 2016, 42, 85–88. [Google Scholar]
- Richieri-Costa, A.; Pirolo Júnior, L.; Cohen, M.M., Jr. Carpenter syndrome with normal intelligence: Brazilian girl born to consanguineous parents. Am. J. Med. Genet. 1993, 47, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Rizos, M.; Negrón, R.J.; Serman, N. Möbius syndrome with dental involvement: A case report and literature review. Cleft Palate-Craniofacial J. Off. Publ. Am. Cleft Palate-Craniofacial Assoc. 1998, 35, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Rock, W.P.; McLellan, N.J. Severe hypodontia in association with Klinefelter (47 XXY) syndrome: A case report. Br. J. Orthod. 1990, 17, 321–323. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Han, D.; Zhang, J.; Zhao, H.; Feng, H. Two novel heterozygous mutations of EVC2 cause a mild phenotype of Ellis-van Creveld syndrome in a Chinese family. Am. J. Med. Genet. Part A 2011, 155, 2131–2136. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, H.P.; Sennimalai, K.; Samrit, V.D.; Duggal, R.; Yadav, R. Adjunctive orthodontic therapy for prosthetic rehabilitation in a growing child with Axenfeld-Rieger syndrome: A case report. Spec. Care Dent. 2021, 41, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Sikora, P.; Majewski, M.; Kandzierski, G.; Zajączkowska, M.; Borzęcka, H.; Korolczuk, A.; Kozlowski, K. Juvenile nephronophtisis, short stature, partial adontia and skeletal abnormalities—A new syndromic association. Nephrol. Dial. Transplant. 2006, 21, 2335–2336. [Google Scholar] [CrossRef][Green Version]
- Singh, A.; Bhatia, H.P.; Sood, S.; Sharma, N.; Mohan, A. A novel finding of oligodontia and ankyloglossia in a 14-year-old with Floating-Harbor syndrome. Spec. Care Dent. Off. Publ. Am. Assoc. Hosp. Dent. Acad. Dent. Handicap. Am. Soc. Geriatr. Dent. 2017, 37, 318–321. [Google Scholar] [CrossRef]
- Subramaniam, P.; Neeraja, G. Witkop’s tooth and nail syndrome: A multifaceted approach to dental management. J. Indian Soc. Pedod. Prev. Dent. 2008, 26, 22–25. [Google Scholar]
- Suda, N.; Bazar, A.; Jigjid, B.; Garidkhuu, A.; Ganburged, G.; Moriyama, K. A Mongolian patient with hypohidrotic ectodermal dysplasia with a novel P121S variant in EDARADD. Orthod. Craniofacial Res. 2010, 13, 114–117. [Google Scholar] [CrossRef]
- Sultan, A.; Juneja, A.; Bhaskar, S. Co-morbidity of down syndrome with autism spectrum disorder: Dental implications. J. Oral Biol. Craniofacial Res. 2020, 10, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Li, F.; Liu, Y.; Qu, H.; Wong, S.W.; Zeng, L.; Yu, M.; Feng, H.; Liu, H.; Han, D. A novel inhibitor of nuclear factor kappa-B kinase subunit gamma mutation identified in an incontinentia pigmenti patient with syndromic tooth agenesis. Arch. Oral Biol. 2019, 101, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Talasila, J.; Pachigolla, R.; Yarlagadda, K.V.S.N.; Vuppala, R.; Grzeschik, K.H.; Kiran, S.; Rose, C.M.; Gottesman, G.S.; Urban, Z. Acromelia-oligodontia syndrome. Clin. Case Rep. 2017, 5, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Tanboga, I.; Kargul, B.; Ergeneli, S.; Aydin, M.Y.; Atasu, M. Clinical features of incontinentia pigmenti with emphasis on dermatoglyphic findings. J. Clin. Pediatr. Dent. 2001, 25, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Tanboga, I.; Pinçe, S.; Düzdar, L. Dental management of a child with EEC syndrome. Int. J. Paediatr. Dent. 1992, 2, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.C.; Slavatinek, A.M.; Vargervik, K.; Oberoi, S. Hypodontia in Beare-Stevenson syndrome: An example of dental anomalies in FGFR-related craniosynostosis syndromes. Cleft Palate-Craniofacial J. 2010, 47, 253–258. [Google Scholar] [CrossRef]
- Tosun, G.; Sener, Y. Apert syndrome with glucose-6-phosphate dehydrogenase deficiency: A case report. Int. J. Paediatr. Dent. 2006, 16, 218–221. [Google Scholar] [CrossRef]
- Tuna, E.B.; Sulun, T.; Rosti, O.; El Abdallah, F.; Kayserili, H.; Aktoren, O. Craniodentofacial manifestations in Hallermann-Streiff syndrome. Cranio J. Craniomandib. Pract. 2009, 27, 33–38. [Google Scholar]
- Tuna, E.B.; Marşan, G.; Gençay, K.; Seymen, F. Craniofacial and dental characteristics of Kabuki syndrome: Nine years cephalometric follow-up. J. Clin. Pediatr. Dent. 2012, 36, 393–400. [Google Scholar] [CrossRef]
- Vasudevan, B.; Sinha, A. A Rare Case of Odonto-Onycho-Dermal-Dysplasia with WNT10a Mutation. Indian J. Dermatol. 2023, 68, 355. [Google Scholar]
- Waldron, J.M.; McNamara, C.; Hewson, A.R.; McNamara, C.M. Axenfeld-Rieger syndrome (ARS): A review and case report. Spec. Care Dent. Off. Publ. Am. Assoc. Hosp. Dent. Acad. Dent. Handicap. Am. Soc. Geriatr. Dent. 2010, 30, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Ye, X.; Bian, Z. The second deletion mutation in exon 8 of EDA gene in an XLHED pedigree. Dermatology 2013, 226, 105–110. [Google Scholar] [CrossRef]
- Zidane, F.E.; El Alloussi, M. Oligodontia and Facial Phenotype Associated with a Rare Syndrome. Case Rep. Dent. 2022, 2022, 1045327. [Google Scholar] [CrossRef] [PubMed]
- Galluccio, G.; Castellano, M.; La Monaca, C. Genetic basis of non-syndromic anomalies of human tooth number. Arch. Oral Biol. 2012, 57, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Ogaard, B.; Krogstad, O. Craniofacial structure and soft tissue profile in patients with severe hypodontia. Am. J. Orthod. Dentofac. Orthop. 1995, 108, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Lu, H.; Xiao, X.; Zhou, L.; Lu, J.; Zhu, L.; Yu, D.; Zhao, W. Novel EDA mutation in X-linked hypohidrotic ectodermal dysplasia and genotype-phenotype correlation. Oral Dis. 2015, 21, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Moura, E.; Rotenberg, I.S.; Pimpão, C.T. X-Linked Hypohidrotic Ectodermal Dysplasia-General Features and Dental Abnormalities in Affected Dogs Compared With Human Dental Abnormalities. Top. Companion Anim. Med. 2019, 35, 11–17. [Google Scholar] [CrossRef]
- Reyes-Reali, J.; Mendoza-Ramos, M.I.; Garrido-Guerrero, E.; Méndez-Catalá, C.F.; Méndez-Cruz, A.R.; Pozo-Molina, G. Hypohidrotic ectodermal dysplasia: Clinical and molecular review. Int. J. Dermatol. 2018, 57, 965–972. [Google Scholar] [CrossRef]
- Aubert-Mucca, M.; Huber, C.; Baujat, G.; Michot, C.; Zarhrate, M.; Bras, M.; Boutaud, L.; Malan, V.; Attie-Bitach, T.; Cormier-Daire, V. Ellis-Van Creveld Syndrome: Clinical and Molecular Analysis of 50 Individuals. J. Med. Genet. 2023, 60, 337–345. [Google Scholar] [CrossRef]
- Hu, Q.; Mai, J.; Xiang, Q.; Zhou, B.; Liu, S.; Wang, J. A novel deletion mutation in the BCOR gene is associated with oculo-facio-cardio-dental syndrome: A case report. BMC Pediatr. 2022, 22, 82. [Google Scholar] [CrossRef]
- McMillan, K.B.; McMillan, D.C.; Arce, K.; Salinas, T. Surgical and prosthetic rehabilitation of siblings with Witkop tooth and nail syndrome using zygomatic implants: A familial case series of 3 patients with up to 15-year follow-up. Oral Maxillofac. Surg. 2023, 27, 711–719. [Google Scholar] [CrossRef]
- Reis, L.M.; Maheshwari, M.; Capasso, J.; Atilla, H.; Dudakova, L.; Thompson, S.; Zitano, L.; Lay-Son, G.; Lowry, R.B.; Black, J.; et al. Axenfeld-Rieger syndrome: More than meets the eye. J. Med. Genet. 2023, 60, 368–379. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, S.; Naveen, D.; Haan, E.; Barnett, C. CDH1-related blepharocheilodontic syndrome is associated with diffuse gastric cancer risk. Am. J. Med. Genet. A 2020, 182, 1780–1784. [Google Scholar] [CrossRef] [PubMed]
- Arzoo, P.S.; Klar, J.; Bergendal, B.; Norderyd, J.; Dahl, N. WNT10A mutations account for (1/4) of population-based isolated oligodontia and show phenotypic correlations. Am. J. Med. Genet. A 2014, 164, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Tuzoff, D.V.; Tuzova, L.N.; Bornstein, M.M.; Krasnov, A.S.; Kharchenko, M.A.; Nikolenko, S.I.; Sveshnikov, M.M.; Bednenko, G.B. Tooth detection and numbering in panoramic radiographs using convolutional neural networks. Dentomaxillofacial Radiol. 2019, 48, 20180051. [Google Scholar] [CrossRef] [PubMed]
- Román, J.C.M.; Fretes, V.R.; Adorno, C.G.; Silva, R.G.; Noguera, J.L.V.; Legal-Ayala, H.; Mello-Román, J.D.; Torres, R.D.E.; Facon, J. Panoramic Dental Radiography Image Enhancement Using Multiscale Mathematical Morphology. Sensors 2021, 21, 3110. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.C.; Line, S.R. The genetics of amelogenesis imperfecta: A review of the literature. J. Appl. Oral Sci. 2005, 13, 212–217. [Google Scholar] [CrossRef]
- Vastardis, H. The genetics of human tooth agenesis: New discoveries for understanding dental anomalies. Am. J. Orthod. Dentofac. Orthop. 2000, 117, 650–655. [Google Scholar] [CrossRef]
- Zeng, B.; Zhao, Q.; Li, S.; Lu, H.; Lu, J.; Ma, L.; Zhao, W.; Yu, D. Novel EDA or EDAR Mutations Identified in Patients with X-Linked Hypohidrotic Ectodermal Dysplasia or Non-Syndromic Tooth Agenesis. Genes 2017, 8, 259. [Google Scholar] [CrossRef]
- Andreoni, F.; Sgattoni, C.; Bencardino, D.; Simonetti, O.; Forabosco, A.; Magnani, M. Missense mutations in EDA and EDAR genes cause dominant syndromic tooth agenesis. Mol. Genet. Genom. Med. 2020, 9, 1555. [Google Scholar] [CrossRef]
- Mues, G.; Tardivel, A.; Willen, L.; Kapadia, H.; Seaman, R.; Frazier-Bowers, S.; Schneider, P.; D’souza, R.N. Functional analysis of ectodysplasin—A mutations causing selective tooth agenesis. Eur. J. Hum. Genet. 2010, 18, 19–25. [Google Scholar] [CrossRef]
- Peck, J.; Douglas, G.; Wu, C.H.; Burbelo, P.D. Human RhoGAP domain-containing proteins: Structure, function and evolutionary relationships. FEBS Lett. 2002, 528, 27–34. [Google Scholar] [CrossRef]
- Schalk-van der Weide, Y.; Steen, W.H.; Bosman, F. Distribution of missing teeth and tooth morphology in patients with oligodontia. ASDC J. Dent. Child. 1992, 59, 133–140. [Google Scholar]
- Schalk-van der Weide, Y.; Prahl-Andersen, B.; Bosman, F. Tooth formation in patients with oligodontia. Angle Orthod. 1993, 63, 31–37. [Google Scholar]
- Polder, B.J.; Van’t Hof, M.A.; Van der Linden, F.P.; Kuijpers-Jagtman, A.M. A meta-analysis of the prevalence of dental agenesis of permanent teeth. Community Dent. Oral Epidemiol. 2004, 32, 217–226. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).