Dynamic In Vitro Gastric Digestion of Sheep Milk: Influence of Homogenization and Heat Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Milk Supply and Processing Treatments
2.2. Chemicals for In Vitro Gastric Digestion
2.3. In Vitro Gastric Digestion
2.4. pH Measurement
2.5. Weight of Curds
2.6. Calcium, Protein, and Fat Content Analysis
2.7. Protein Hydrolysis
2.8. Microstructure of Curds and Digesta
2.9. Statistical Analysis
3. Results and Discussion
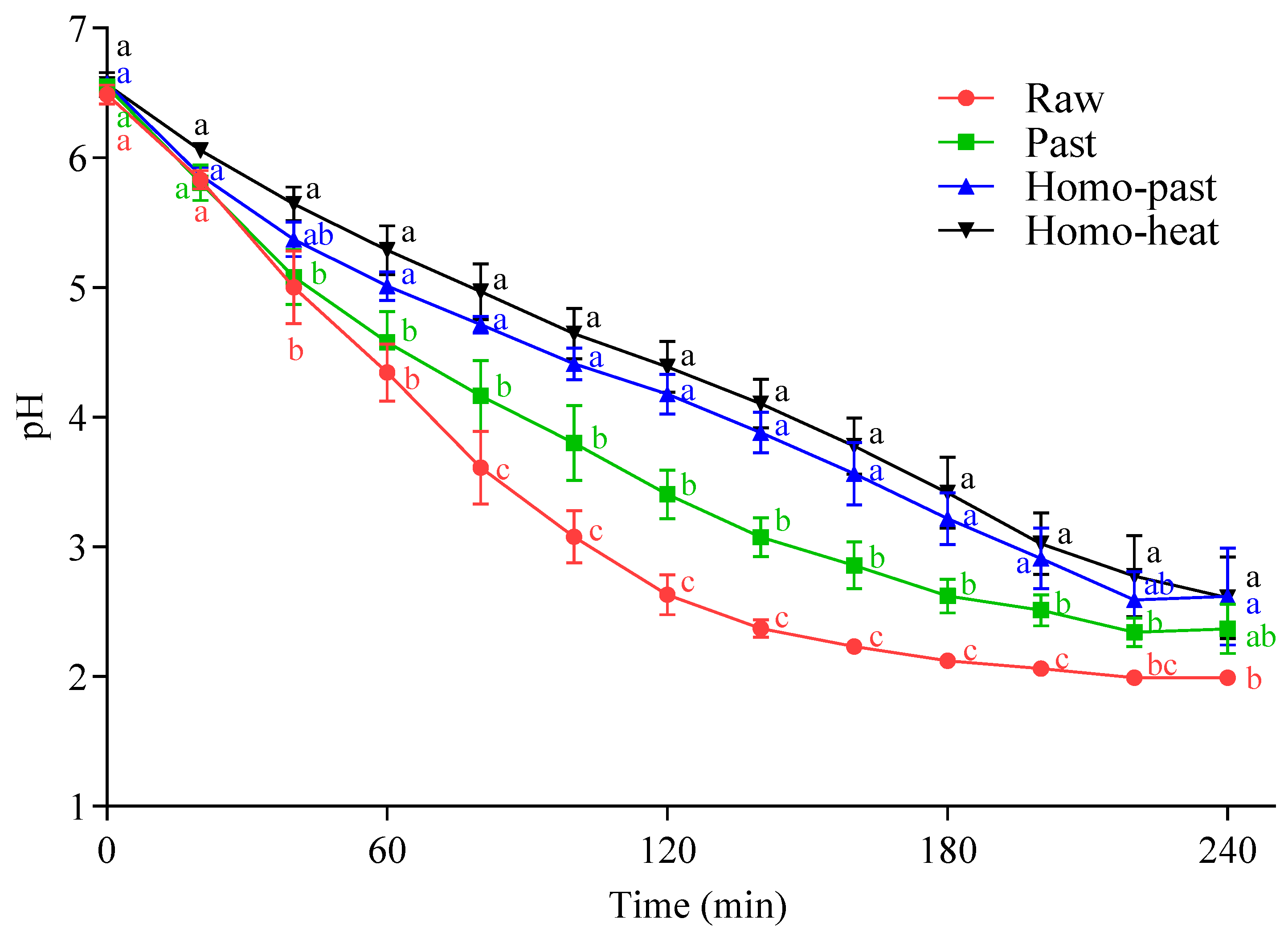
3.1. Gastric pH Profile
3.2. Gastric Coagulation of Sheep Milk
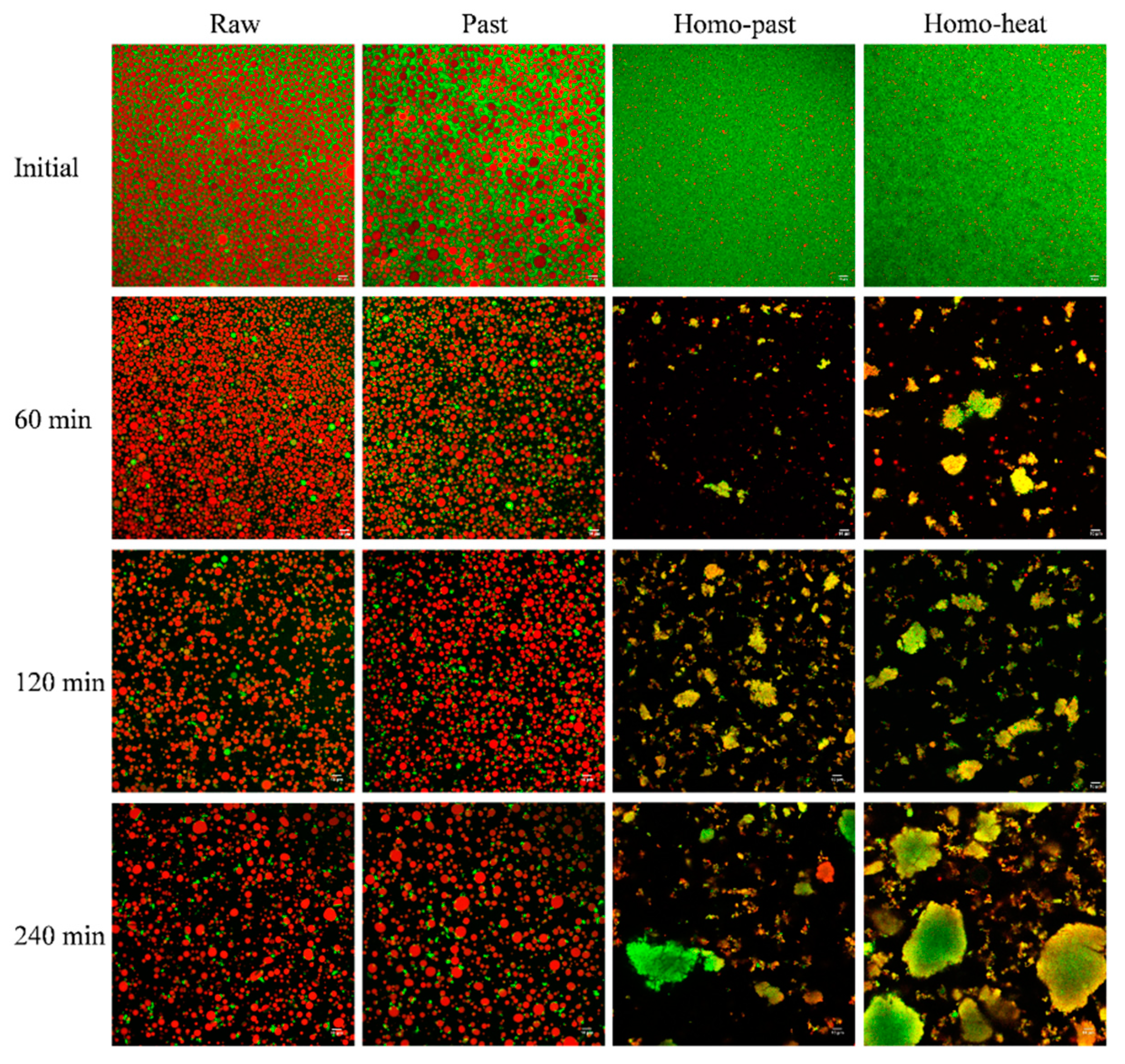
3.3. Microstructure of Curds
3.4. Disintegration of Curds
3.4.1. Weight of Curds
3.4.2. Changes in Protein, Fat, and Calcium Contents in the Curd
3.5. SDS-PAGE Protein Patterns of Curds
3.6. Protein and Fat Contents of Emptied Digesta
3.7. SDS-PAGE Protein Patterns of Digesta
3.8. Microstructure of Digesta
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wendorff, W.; Haenlein, G.F. Sheep milk–composition and nutrition. In Handbook of Milk of Non-Bovine Mammals, 2nd ed.; Park, Y.W., Haenlein, G.F., Wendorff, W.L., Eds.; John Wiley & Sons: Chichester, UK, 2017; pp. 210–221. [Google Scholar]
- Gallier, S.; Zhu, X.Q.; Rutherfurd, S.M.; Ye, A.; Moughan, P.J.; Singh, H. In vivo digestion of bovine milk fat globules: Effect of processing and interfacial structural changes. II. Upper digestive tract digestion. Food Chem. 2013, 141, 3215–3223. [Google Scholar] [CrossRef]
- Ye, A.; Cui, J.; Dalgleish, D.; Singh, H. Formation of a structured clot during the gastric digestion of milk: Impact on the rate of protein hydrolysis. Food Hydrocoll. 2016, 52, 478–486. [Google Scholar] [CrossRef]
- Mulet-Cabero, A.-I.; Mackie, A.R.; Wilde, P.J.; Fenelon, M.A.; Brodkorb, A. Structural mechanism and kinetics of in vitro gastric digestion are affected by process-induced changes in bovine milk. Food Hydrocoll. 2019, 86, 172–183. [Google Scholar] [CrossRef]
- Jasińska, B. The comparison of pepsin and trypsin action on goat, cow, mare and human caseins. Rocz. Akad. Med. Bialymst. 1995, 40, 486–493. [Google Scholar]
- Roy, D.; Ye, A.; Moughan, P.J.; Singh, H. Structural changes in cow, goat, and sheep skim milk during dynamic in vitro gastric digestion. J. Dairy Sci. 2021, 104, 1394–1411. [Google Scholar] [CrossRef]
- Ye, A.; Cui, J.; Carpenter, E.; Prosser, C.; Singh, H. Dynamic in vitro gastric digestion of infant formulae made with goat milk and cow milk: Influence of protein composition. Int. Dairy J. 2019, 97, 76–85. [Google Scholar] [CrossRef]
- Shen, L.; Dael, P.V.; Luten, L.; Deelstra, H. Estimation of selenium bioavailability from human, cow’s, goat and sheep milk by an in vitro method. Int. J. Food Sci. Nutr. 1996, 47, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Claeys, W.L.; Verraes, C.; Cardoen, S.; De Block, J.; Huyghebaert, A.; Raes, K.; Dewettinck, K.; Herman, L. Consumption of raw or heated milk from different species: An evaluation of the nutritional and potential health benefits. Food Control 2014, 42, 188–201. [Google Scholar] [CrossRef]
- Balthazar, C.F.; Pimentel, T.C.; Ferrão, L.L.; Almada, C.N.; Santillo, A.; Albenzio, M.; Mollakhalili, N.; Mortazavian, A.M.; Nascimento, J.S.; Silva, M.C.; et al. Sheep milk: Physicochemical characteristics and relevance for functional food development. Compr. Rev. Food Sci. Food Saf. 2017, 16, 247–262. [Google Scholar] [CrossRef]
- Remeuf, F.; Lenoir, J.; Duby, C.; Letilly, M.-T.; Normand, A. Etude des relations entre les caractéristiques physico-chimiques des laits de chèvre et leur aptitude à la coagulation par la présure. Lait 1989, 69, 499–518. [Google Scholar] [CrossRef] [Green Version]
- Anema, S.G.; Li, Y. Association of denatured whey proteins with casein micelles in heated reconstituted skim milk and its effect on casein micelle size. J. Dairy Res. 2003, 70, 73–83. [Google Scholar] [CrossRef]
- Anema, S.G. Role of κ-casein in the association of denatured whey proteins with casein micelles in heated reconstituted skim milk. J. Agric. Food Chem. 2007, 55, 3635–3642. [Google Scholar] [CrossRef] [PubMed]
- Ye, A.; Singh, H.; Taylor, M.W.; Anema, S. Interactions of whey proteins with milk fat globule membrane proteins during heat treatment of whole milk. Lait 2004, 84, 269–283. [Google Scholar] [CrossRef] [Green Version]
- Ye, A.; Anema, S.G.; Singh, H. Changes in the surface protein of the fat globules during homogenization and heat treatment of concentrated milk. J. Dairy Res. 2008, 75, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Michalski, M.-C.; Januel, C. Does homogenization affect the human health properties of cow’s milk? Trends Food Sci. Technol. 2006, 17, 423–437. [Google Scholar] [CrossRef]
- Roy, D.; Ye, A.; Moughan, P.J.; Singh, H. Impact of gastric coagulation on the kinetics of release of fat globules from milk of different species. Food Funct. 2021, 12, 1783–1802. [Google Scholar] [CrossRef]
- Ye, A.; Liu, W.; Cui, J.; Kong, X.; Roy, D.; Kong, Y.; Han, J.; Singh, H. Coagulation behaviour of milk under gastric digestion: Effect of pasteurization and ultra-high temperature treatment. Food Chem. 2019, 286, 216–225. [Google Scholar] [CrossRef]
- Ye, A.; Cui, J.; Dalgleish, D.; Singh, H. Effect of homogenization and heat treatment on the behavior of protein and fat globules during gastric digestion of milk. J. Dairy Sci. 2017, 100, 36–47. [Google Scholar] [CrossRef]
- Mulet-Cabero, A.-I.; Mackie, A.R.; Brodkorb, A.; Wilde, P.J. Dairy structures and physiological responses: A matter of gastric digestion. Crit. Rev. Food Sci. Nutr. 2020, 60, 3737–3752. [Google Scholar] [CrossRef]
- Egger, L.; Ménard, O.; Baumann, C.; Duerr, D.; Schlegel, P.; Stoll, P.; Vergères, G.; Dupont, D.; Portmann, R. Digestion of milk proteins: Comparing static and dynamic in vitro digestion systems with in vivo data. Food Res. Int. 2019, 118, 32–39. [Google Scholar] [CrossRef]
- Egger, L.; Schlegel, P.; Baumann, C.; Stoffers, H.; Guggisberg, D.; Brügger, C.; Dürr, D.; Stoll, P.; Vergères, G.; Portmann, R. Physiological comparability of the harmonized INFOGEST in vitro digestion method to in vivo pig digestion. Food Res. Int. 2017, 102, 567–574. [Google Scholar] [CrossRef]
- Authority, N.Z.F.S. Heat Treatment Code of Practice. 2009. Available online: https://www.mpi.govt.nz/dmsdocument/46228-Dairy-Heat-treatment-Code-of-practice (accessed on 1 August 2019).
- Jørgensen, C.E.; Abrahamsen, R.K.; Rukke, E.-O.; Johansen, A.-G.; Schüller, R.B.; Skeie, S.B. Improving the structure and rheology of high protein, low fat yoghurt with undenatured whey proteins. Int. Dairy J. 2015, 47, 6–18. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assuncao, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carriere, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Singh, R.P. A human gastric simulator (HGS) to study food digestion in human stomach. J. Food Sci. 2010, 75, E627–E635. [Google Scholar] [CrossRef]
- Lydon, A.; Murray, C.; McGinley, J.; Plant, R.; Duggan, F.; Shorten, G. Cisapride does not alter gastric volume or pH in patients undergoing ambulatory surgery. Can. J. Anesth. 1999, 46, 1181–1184. [Google Scholar] [CrossRef] [PubMed]
- Vertzoni, M.; Dressman, J.; Butler, J.; Hempenstall, J.; Reppas, C. Simulation of fasting gastric conditions and its importance for the in vivo dissolution of lipophilic compounds. Eur. J. Pharm. Biopharm. 2005, 60, 413–417. [Google Scholar] [CrossRef] [PubMed]
- International Dairy Federation. IDF Standard? IDF: Brussels, Belgium, 1987. [Google Scholar]
- Wang, X.; Lin, Q.; Ye, A.; Han, J.; Singh, H. Flocculation of oil-in-water emulsions stabilised by milk protein ingredients under gastric conditions: Impact on in vitro intestinal lipid digestion. Food Hydrocoll. 2019, 88, 272–282. [Google Scholar] [CrossRef]
- Delfour, A.; Alais, C.; Jollès, P. Structure of cows kappa-caseino-glycopeptide-n-terminal octadecapeptide. Chimia 1966, 20, 148–150. [Google Scholar]
- Mulvihill, D.M.; Fox, P.F. Proteolytic Specificity of Chymosins and Pepsins on Beta-Caseins. Irish J. Food Sci. Technol. 1979, 2, 135–139. Available online: http://www.jstor.org/stable/25557963 (accessed on 18 February 2021).
- Gallier, S.; Ye, A.; Singh, H. Structural changes of bovine milk fat globules during in vitro digestion. J. Dairy Sci. 2012, 95, 3579–3592. [Google Scholar] [CrossRef] [Green Version]
- Ye, A.; Cui, J.; Singh, H. Proteolysis of milk fat globule membrane proteins during in vitro gastric digestion of milk. J. Dairy Sci. 2011, 94, 2762–2770. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.L.; Huppertz, T.; Sheehan, J.J. Pre-treatment of cheese milk: Principles and developments. Dairy Sci. Technol. 2008, 88, 549–572. [Google Scholar] [CrossRef] [Green Version]
- Ye, A.; Cui, J.; Dalgleish, D.; Singh, H. The formation and breakdown of structured clots from whole milk during gastric digestion. Food Funct. 2016, 7, 4259–4266. [Google Scholar] [CrossRef]
- Lopez, C. Focus on the supramolecular structure of milk fat in dairy products. Reprod. Nutr. Dev. 2005, 45, 497–511. [Google Scholar] [CrossRef] [Green Version]
- Ong, L.; Dagastine, R.R.; Kentish, S.E.; Gras, S.L. Microstructure of milk gel and cheese curd observed using cryo scanning electron microscopy and confocal microscopy. LWT 2011, 44, 1291–1302. [Google Scholar] [CrossRef]
- Guinee, T.; O’Brien, B. The quality of milk for cheese manufacture. In Technology of Cheesemaking, 2nd ed.; Law, B.A., Tamime, A.Y., Eds.; John Wiley & Sons: Chichester, UK, 2010; Volume 67, pp. 1–67. [Google Scholar]
- Everett, D.W.; Auty, M.A.E. Cheese structure and current methods of analysis. Int. Dairy J. 2008, 18, 759–773. [Google Scholar] [CrossRef]
- Awad, S.; Lüthi-Peng, Q.-Q.; Puhan, Z. Proteolytic activities of chymosin and porcine pepsin on buffalo, cow, and goat whole and β-casein fractions. J. Agric. Food Chem. 1998, 46, 4997–5007. [Google Scholar] [CrossRef]
- Anema, S.G. The whey proteins in milk: Thermal denaturation, physical interactions, and effects on the functional properties of milk. In Milk Proteins, 3rd ed.; Boland, M., Singh, H., Eds.; Academic Press: London, UK, 2020; pp. 325–384. [Google Scholar]
- Wang, X.; Ye, A.; Lin, Q.; Han, J.; Singh, H. Gastric digestion of milk protein ingredients: Study using an in vitro dynamic model. J. Dairy Sci. 2018, 101, 6842–6852. [Google Scholar] [CrossRef] [PubMed] [Green Version]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, Z.; Ye, A.; Li, S.; Dave, A.; Fraser, K.; Singh, H. Dynamic In Vitro Gastric Digestion of Sheep Milk: Influence of Homogenization and Heat Treatment. Foods 2021, 10, 1938. https://doi.org/10.3390/foods10081938
Pan Z, Ye A, Li S, Dave A, Fraser K, Singh H. Dynamic In Vitro Gastric Digestion of Sheep Milk: Influence of Homogenization and Heat Treatment. Foods. 2021; 10(8):1938. https://doi.org/10.3390/foods10081938
Chicago/Turabian StylePan, Zheng, Aiqian Ye, Siqi Li, Anant Dave, Karl Fraser, and Harjinder Singh. 2021. "Dynamic In Vitro Gastric Digestion of Sheep Milk: Influence of Homogenization and Heat Treatment" Foods 10, no. 8: 1938. https://doi.org/10.3390/foods10081938
APA StylePan, Z., Ye, A., Li, S., Dave, A., Fraser, K., & Singh, H. (2021). Dynamic In Vitro Gastric Digestion of Sheep Milk: Influence of Homogenization and Heat Treatment. Foods, 10(8), 1938. https://doi.org/10.3390/foods10081938






