Dietary DHA Enhanced the Textural Firmness of Common Carp (Cyprinus carpio L.) Fed Plant-Derived Diets through Restraining FoxO1 Pathways
Abstract
:Highlights
- Dietary DHA of 0.42% was beneficial to the growth of common carp fed plant-derived diets;
- Dietary DHA ameliorated the muscular fatty acid nutrition, and textural firmness of common carp fed plant-derived diets;
- Myogenic regulatory factors expression was significantly elevated, while mstna expression was decreased by dietary DHA;
- Dietary DHA modification to the muscular textural firmness of common carp may be attributed to the inhibition of FoxO1 pathways
Abstract
1. Introduction
2. Materials and Methods
2.1. Diets and Animal Management
2.2. Samples Collection and Growth Performance Evaluation
2.3. Proximal and FA Composition
2.4. Biochemical Analysis
2.5. Muscle Nutritional Indices and Texture Characteristics Test
2.6. Histological and Ultrastructural Observation
2.7. Transcriptome Analysis
2.8. RNA Extraction and Quantitative Real-Time PCR (qRT-PCR)
2.9. Statistical Analysis
3. Results
3.1. Growth and Proximate Composition of Fish
3.2. Biochemical Analysis
3.3. FA Compositions and Nutritional Indices of Muscle
3.4. Proximate Composition, Edible Quality, and Textural Properties of Muscle
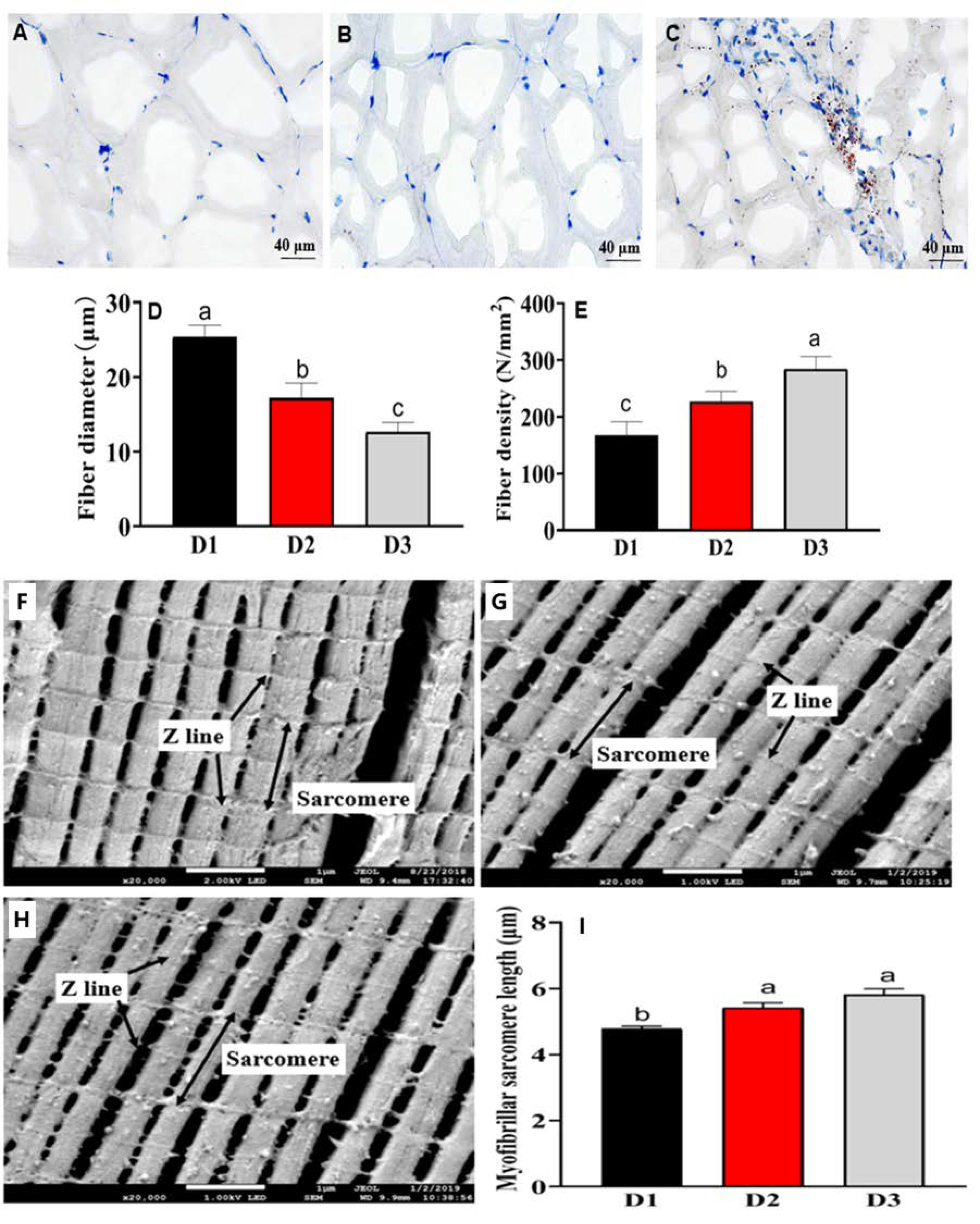
3.5. Histological Fiber and Ultrastructure Analysis
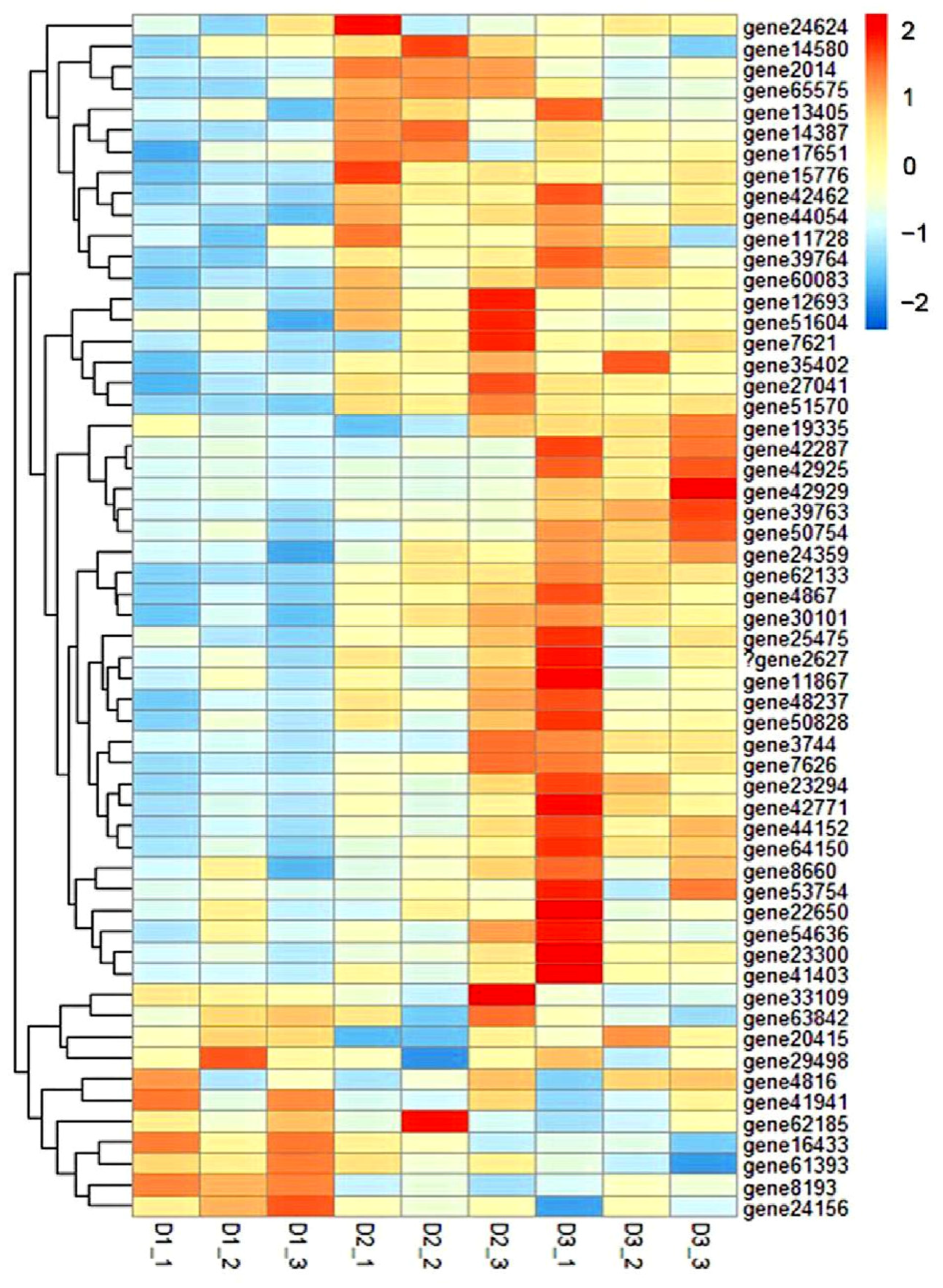
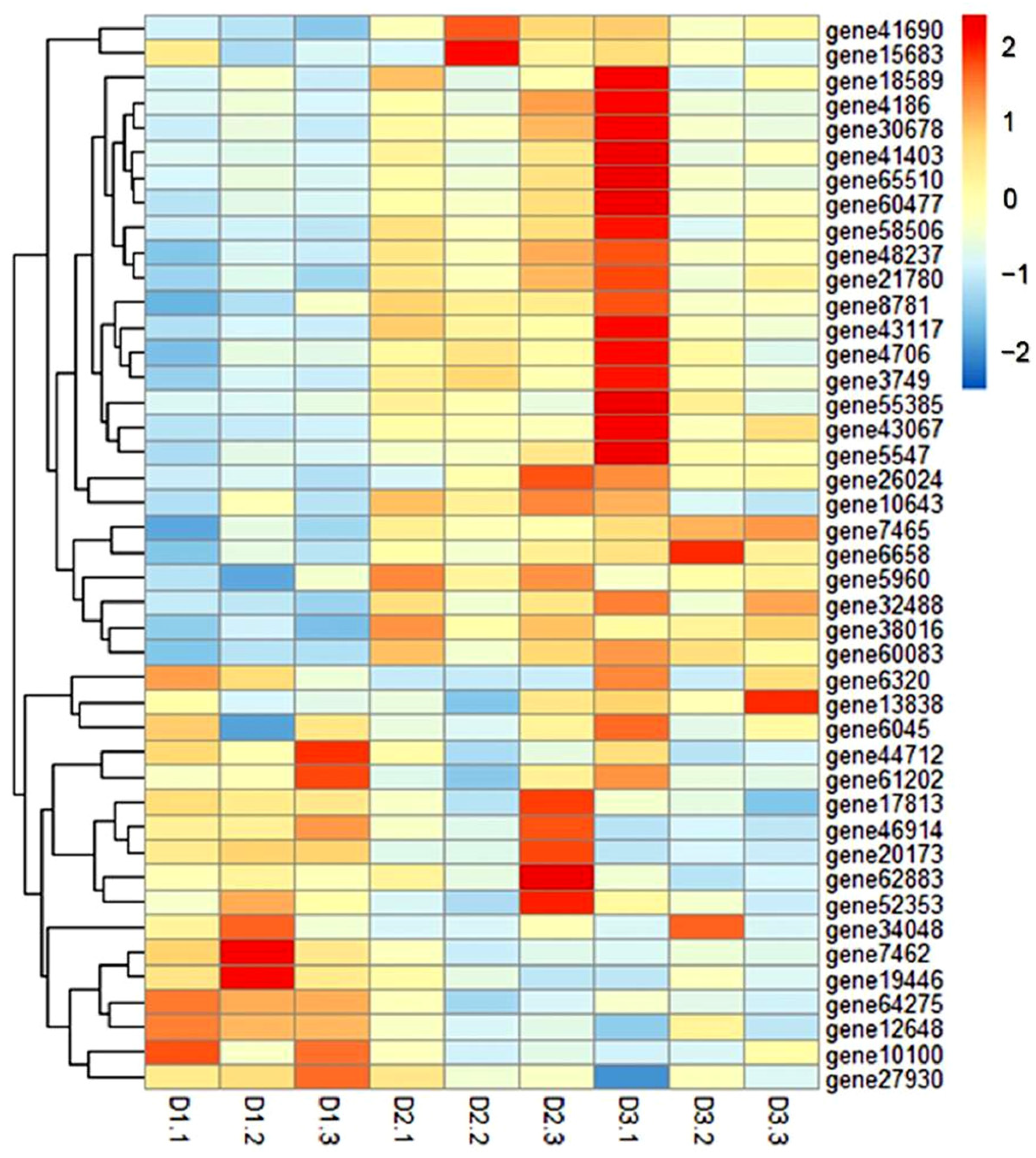
3.6. GO and KEGG Enrichment Analysis of DEGs
3.7. RNA-Seq Results Were Verified by qRT-PCR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Toyes-Vargas, E.A.; Parrish, C.C.; Viana, M.T.; Carreón-Palau, L.; Magallón-Servín, P.; Magallón-Barajas, F.J. Replacement of fish oil with camelina (Camelina sativa) oil in diets for juvenile tilapia (var. GIFT Oreochromis niloticus) and its effect on growth, feed utilization and muscle lipid composition. Aquaculture 2020, 523, 735235. [Google Scholar] [CrossRef]
- Song, D.; Yun, Y.; Mi, J.; Luo, J.; Zhou, Q. Effects of faba bean on growth performance and fillet texture of Yellow River carp, Cyprinus carpio haematopterus. Aquac. Rep. 2020, 17, 100379. [Google Scholar] [CrossRef]
- Tocher, D.R. Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 2015, 449, 94–107. [Google Scholar] [CrossRef]
- Wang, C.; Liu, W.; Huang, Y.; Wang, X.; Li, X.; Zhang, D.; Jiang, G. Dietary DHA affects muscle fiber development by activating AMPK/Sirt1 pathway in blunt snout bream (Megalobrama amblycephala). Aquaculture 2019, 518, 734835. [Google Scholar] [CrossRef]
- Li, M.; Zhang, M.; Ma, Y.; Ye, R.; Wang, M.; Chen, H.; Xie, D.; Dong, Y.; Ning, L.; You, C.; et al. Dietary supplementation with n-3 high unsaturated fatty acids decreases serum lipid levels and improves flesh quality in the marine teleost golden pompano Trachinotus ovatus. Aquaculture 2020, 516, 734632. [Google Scholar] [CrossRef]
- Song, D.; Yun, Y.; He, Z.; Mi, J.; Luo, J.; Jin, M.; Zhou, Q.; Nie, G. Effects of Faba bean (Vicia faba L.) on fillet quality of Yellow River carp (Cyprinus carpio) via the oxidative stress response. Food Chem. 2022, 388, 132953. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Tan, Q.; Kong, F.; Yu, H.; Zhu, Y.; Yao, J.; Azm, F.R.A. Fish growth in response to different feeding regimes and the related molecular mechanism on the changes in skeletal muscle growth in grass carp (Ctenopharyngodon idellus). Aquaculture 2019, 512, 734295. [Google Scholar] [CrossRef]
- Castillero, E.; Martín, A.; López-Menduiña, M.; Villanúa, M.; López-Calderón, A. Eicosapentaenoic acid attenuates arthritis-induced muscle wasting acting on atrogin-1 and on myogenic regulatory factors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 297, R1322. [Google Scholar] [CrossRef]
- Bryner, R.W.; Woodworth-Hobbs, M.E.; Williamson, D.L.; Alway, S.E. Docosahexaenoic Acid Protects Muscle Cells from Palmitate-Induced Atrophy. ISRN. Obesity 2012, 10, 647348. [Google Scholar] [CrossRef] [Green Version]
- Tachtsis, B.; Camera, D.; Lacham-Kaplan, O. Potential roles of n-3 PUFAs during skeletal muscle growth and regeneration. Nutrients 2018, 10, 309. [Google Scholar] [CrossRef]
- Harland, J. The origins of aquaculture. Nat. Ecol. Evol. 2019, 3, 1378–1379. [Google Scholar] [CrossRef] [PubMed]
- Liu, X. China Fishery Statistical Yearbook; China Agriculture Press: Beijing, China, 2022; pp. 24–25. [Google Scholar]
- Xie, D.; Guan, J.; Huang, X.; Xu, C.; Pan, Q.; Li, Y. Tilapia can be a beneficial n-3 LC-PUFA source due to its high biosynthetic capacity in the liver and intestine. J. Agric. Food Chem. 2022, 70, 2701–2711. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Chen, F.; Guan, J.; Xu, C.; Li, Y.; Xie, D. Beneficial effects of re-feeding high α-linolenic acid diets on the muscle quality, cold temperature and disease resistance of tilapia. Fish Shellfish Immun. 2022, 126, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, M.; Xie, D.; Chen, S.; Dong, Y.; Wang, M.; Zhang, G.; Zhang, M.; Chen, H.; Ye, R.; et al. Fishmeal can be replaced with a high proportion of terrestrial protein in the diet of the carnivorous marine teleost (Trachinotus ovatus). Aquaculture 2020, 519, 734910. [Google Scholar] [CrossRef]
- Song, Y.; Yan, G.; Christer, H.; Li, D.; Pan, Y.; Luo, Z. Upstream regulators of apoptosis mediates methionine-induced changes of lipid metabolism. Cell. Signal. 2018, 51, 176–190. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT. Method 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Yang, G.; Jiang, W.; Chen, Y.; Vikas, K. Effect of oil source on growth performance, antioxidant capacity, fatty acid composition and fillet quality of juvenile grass carp (Ctenopharyngodon idella). Aquac. Nutr. 2020, 26, 1186–1197. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, L.; Gao, J. Effects of dietary highly unsaturated fatty acid levels on growth, fatty acid profiles, antioxidant activities, mucus immune responses and hepatic lipid metabolism related gene expressions in loach (Misgurnus anguillicaudatus) juveniles. Aquac. Res. 2020, 50, 2486–2495. [Google Scholar] [CrossRef]
- Ji, H.; Li, J.; Liu, P. Regulation of growth performance and lipid metabolism by dietary n-3 highly unsaturated fatty acids in juvenile grass carp, Ctenopharyngodon idellus. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2011, 159, 49–56. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, X.; Han, D.; Yang, Y.; Lei, W.; Xie, S. Effect of dietary n-3 LC-PUFA on growth performance and tissue fatty acid composition of gibel carp carassius auratus gibelio. Aquac. Nutr. 2011, 17, 476–485. [Google Scholar] [CrossRef]
- Nobrega, R.O.; Batista, R.O.; Corrêa, C.F.; Mattioni, B.; Filer, K.; Pettigrew, J.E.; Fracalossi, D.M. Dietary supplementation of Aurantiochytrium sp. meal, a docosahexaenoic-acid source, promotes growth of Nile tilapia at a suboptimal low temperature. Aquaculture 2019, 507, 500–509. [Google Scholar] [CrossRef]
- Yadav, A.K.; Rossi Jr, W.; Habte-Tsion, H.M.; Kumar, V. Impacts of dietary eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) level and ratio on the growth, fatty acids composition and hepatic-antioxidant status of largemouth bass (Micropterus salmoides). Aquaculture 2020, 529, 735683. [Google Scholar] [CrossRef]
- An, W.; Dong, X.; Tan, B.; Yang, Q.; Chi, S.; Zhang, S.; Liu, H.; Yang, Y. Dietary n-3 long chain polyunsaturated fatty acids affect the serum biochemical parameters, lipid-metabolism-related of gene expression and intestinal health of juvenile hybrid grouper (♀ Epinephelus fuscoguttatus×♂ Epinephelus lanceolatu). Aquac. Nutr. 2021, 27, 1383–1395. [Google Scholar] [CrossRef]
- Xu, X.; Ji, H.; Belghit, I.; Liland, N.S.; Wu, W.; Li, X. Effects of black soldier fly oil rich in n-3 HUFA on growth performance, metabolism and health response of juvenile mirror carp (Cyprinus carpio var. specularis). Aquaculture 2020, 533, 736144. [Google Scholar] [CrossRef]
- Mata-Sotres, J.A.; Marques, V.H.; Barba, D.; Braga, A.; Araújo, B.; Viana, M.T.; Rombenso, A.N. Increasing dietary SFA: MUFA ratio with low levels of LC-PUFA affected lipid metabolism, tissue fatty acid profile and growth of juvenile California Yellowtail (Seriola dorsalis). Aquaculture 2021, 543, 737011. [Google Scholar] [CrossRef]
- Hou, Y.; Hou, Y.; Yao, L.; Chen, S.; Fan, J.; Qian, L. Effects of chromium yeast, tributyrin and bile acid on growth performance, digestion and metabolism of Channa argus. Aquac. Res. 2019, 50, 836–846. [Google Scholar] [CrossRef]
- Luz, J.R.; Ramos, A.P.S.; Melo, J.F.B.; Braga, L.G.T. Use of sodium butyrate in the feeding of Arapaima gigas (Schinz, 1822) juvenile. Aquaculture 2019, 510, 248–255. [Google Scholar] [CrossRef]
- Liu, P.; Li, C.; Huang, J.; Ji, H. Regulation of adipocytes lipolysis by n-3 LC-PUFA in grass carp (Ctenopharyngodon idellus) in vitro and in vivo. Fish. Physiol. Biochem. 2014, 40, 1447–1460. [Google Scholar] [CrossRef]
- Jorge, T.; Moura, G.; Ribeiro-Junior, V.; Donzele, J.; Pedreira, M.; Sousa, T.; Lanna, E. Effects of dietary supplementation time with Schizochytrium microalgae meal on growth, meat quality and fatty acid composition of Nile tilapia. Aquac. Res. 2022, 53, 528–543. [Google Scholar] [CrossRef]
- Mozanzadeh, M.T.; Agh, N.; Yavari, V.; Marammazi, J.G.; Mohammadian, T.; Gisbert, E. Partial or total replacement of dietary fish oil with alternative lipid sources in silvery-black porgy (Sparidentex hasta). Aquaculture 2016, 451, 232–240. [Google Scholar] [CrossRef]
- Álvarez, A.; Fontanillas, R.; Hernández-Contreras, A.; Hernández, M.D. Partial replacement of fish oil with vegetal oils in commercial diets: The effect on the quality of gilthead seabream (Sparus aurata). Anim. Feed Sci. Tech. 2020, 265, 114504. [Google Scholar] [CrossRef]
- Lopes, G.; Castro, L.F.C.; Valente, L.M.P. Total substitution of dietary fish oil by vegetable oils stimulates muscle hypertrophic growth in Senegalese sole and the upregulation of fgf6. Food Funct. 2017, 8, 1869–1879. [Google Scholar] [CrossRef] [PubMed]
- Periago, M.J.; Ayala, M.D.; López-Albors, O.; Abdel, I.; Martinez, C.; García-Alcázar, A.; Ros, G.; Gil, F. Muscle cellularity and flesh quality of wild and farmed sea bass, Dicentrarchus labrax L. Aquaculture 2005, 249, 175–188. [Google Scholar] [CrossRef]
- Isesele, P.O.; Mazurak, V.C. Regulation of Skeletal Muscle Satellite Cell Differentiation by Omega-3 Polyunsaturated Fatty Acids: A Critical Review. Front. Physiol. 2021, 12, 682091. [Google Scholar] [CrossRef]
- Xu, M.; Chen, X.; Chen, D.; Yu, B.; Huang, Z. FoxO1: A novel insight into its molecular mechanisms in the regulation of skeletal muscle differentiation and fiber type specification. Oncotarget 2017, 8, 10662–10674. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.J.; Chen, C.C.; Li, T.K.; Wang, P.H.; Liu, L.R.; Chang, F.Y.; Wang, Y.C.; Yu, Y.H.; Lin, S.P.; Mersmann, H.J.; et al. Docosahexaenoic acid suppresses the expression of FoxO and its target genes. J. Nutr. Biochem. 2012, 23, 1609–1616. [Google Scholar] [CrossRef]
- Liu, Y.L.; Chen, F.; Odle, J.; Lin, X.; Zhu, H.L.; Shi, H.F.; Hou, Y.Q.; Yin, J.D. Fish oil increases muscle protein mass and modulates Akt/FOXO, TLR4, and NOD signaling in weanling piglets after lipopolysaccharide challenge. J. Nutr. 2013, 143, 1331–1339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Ju, S.; Fan, D.; Li, Y.; Guo, L. Orexin A stimulates Foxo1 phosphorylation via OX1R-induced PI3K/AKT and MAPK/ERK signaling pathways in hepatocytes. Int. J. Clin. Exp. Med. 2019, 12, 6757–6767. [Google Scholar]
- Siraj, M.A.; Jacobs, A.T.; Tan, G.T. Altersolanol B, a fungal tetrahydroanthraquinone, inhibits the proliferation of estrogen receptor-expressing (ER+) human breast adenocarcinoma by modulating PI3K/AKT, p38/ERK MAPK and associated signaling pathways. Chem.-Biol. Interact. 2022, 359, 109916. [Google Scholar] [CrossRef]
- Matoba, A.; Matsuyama, N.; Masaki, E.; Emala, C.W.; Mizuta, K. The omega-3 free fatty acid receptor FFAR4/GPR120 contributes human airway smooth muscle cell proliferation through PI3K/Akt and MEK/ERK pathways. In Proceedings of the American Society of Anesthesiologists (ASA) Annual Meeting, Boston, MA, USA, 21–25 October 2017. [Google Scholar]

| Ingredients (% Dry Matter) | Experimental Diets | ||
|---|---|---|---|
| D1 | D2 | D3 | |
| Soybean meal | 40 | 40 | 40 |
| Rapeseed meal | 25 | 25 | 25 |
| Cottonseed meal | 10 | 10 | 10 |
| Soybean oil | 6 | 5.5 | 5 |
| DHA purified oil a | 0 | 0.5 | 1 |
| Proxim compound b | 1.0 | 1.0 | 1.0 |
| Others c | 18.1 | ||
| Proximate composition (% dry matter) | |||
| Dry matter | 90.48 | 90.54 | 90.78 |
| Crude protein | 31.43 | 32.23 | 31.45 |
| Crude lipid | 6.88 | 6.94 | 7.03 |
| Ash | 6.77 | 6.86 | 6.15 |
| Main FA composition (% total FAs) | |||
| 14:0 | 0.58 | 0.59 | 0.57 |
| 16:0 | 6.38 | 5.9 | 7.35 |
| 18:0 | 2.9 | 2.76 | 2.66 |
| 20:0 | 0.46 | 0.6 | 0.62 |
| 16:1 | 0.65 | 0.72 | 0.56 |
| 18:1n9 | 20.64 | 18.57 | 18.29 |
| 18:2n6 | 55.49 | 49.61 | 41.64 |
| 18:3n3 | 6.12 | 6.72 | 7.09 |
| 20:4n6 | / | 0.47 | 0.7 |
| 20:5n3 | / | 0.89 | 1.21 |
| 20:6n3 | / | 5.98 | 9.3 |
| Experimental Diets | |||
|---|---|---|---|
| D1 | D2 | D3 | |
| IBW (g) | 15.26 ± 0.06 | 15.21 ± 0.04 | 15.34 ± 0.05 |
| FBW (g) | 31.04 ± 1.27 | 32.92 ± 1.64 | 31.83 ± 1.42 |
| WGR (%) | 103.37 ± 4.16 b | 115.06 ± 3.88 a | 107.52 ± 4.09 a,b |
| SRG (%) | 1.27 ± 0.03 b | 1.37 ± 0.03 a | 1.30 ± 0.03 a,b |
| FCR | 1.38 ± 0.03 | 1.33 ± 0.02 | 1.29 ± 0.03 |
| Survival (%) | 100 | 100 | 100 |
| HSI | 5.16 ± 0.27 | 5.21 ± 0.26 | 5.18 ± 0.24 |
| VSI | 1.04 ± 0.06 | 1.08 ± 0.04 | 1.07 ± 0.12 |
| CF (g/cm3) | 2.57 ± 0.08 | 2.73 ± 0.16 | 2.66 ± 0.12 |
| Proximate composition (% wet matter) | |||
| Moisture | 69.40 ± 0.53 | 70.25 ± 0.74 | 68.02 ± 0.72 |
| Crude protein | 17.31 ± 0.03 | 17.81 ± 0.34 | 17.53 ± 0.28 |
| Crude lipid | 8.17 ± 0.37 | 7.94 ± 0.44 | 7.50 ± 0.61 |
| Ash | 3.05 ± 0.04 | 3.56 ± 0.13 | 3.10 ± 0.09 |
| Index | Experimental Diets | ||
|---|---|---|---|
| D1 | D2 | D3 | |
| TP (mmol/L) | 23.27 ± 0.99 | 22.67 ± 1.49 | 22.07 ± 0.55 |
| GLU (mmol/L) | 4.74 ± 0.87 b | 5.17 ± 0.60 a,b | 6.28 ± 0.87 a |
| TG (mmol/L) | 1.05 ± 0.05 a | 0.72 ±0.08 b | 0.81 ± 0.05 b |
| NEFA (mmol/L) | 1.92 ± 0.36 | 2.05 ± 0.46 | 2.04 ± 0.44 |
| CHO (mmol/L) | 2.50 ± 0.13 a | 1.85 ± 0.17 b | 2.55 ± 0.19 a |
| LDL (mmol/L) | 1.00 ± 0.09 a | 0.65 ± 0.11 b | 0.82 ± 0.08 b |
| HDL (mmol/L) | 1.27 ± 0.05 b | 1.51 ±0.07 a,b | 1.90 ± 0.41 a |
| ALB (mmol/L) | 10.87 ± 0.75 a | 8.78 ±0.71 b | 12.58 ± 0.65 a |
| ACP (U/mL) | 5.24 ± 0.35 b | 7.59 ± 0.10 a | 7.68 ± 0.60 a |
| AKP (U/mL) | 5.97 ± 0.40 b | 7.01 ± 0.64 a | 7.86 ± 0.86 a |
| SOD (U/mL) | 136.72 ± 1.74 | 137.83 ±2.96 | 140.17 ± 3.80 |
| GSH-Px (U/mL) | 542.73 ± 33.78 | 492.45 ± 44.74 | 602.12 ± 15.28 |
| CAT (U/mL) | 94.68 ± 2.57 | 104.33 ± 3.35 | 107.65 ± 1.77 |
| MDA (nmol/mL) | 0.91 ± 0.04 | 0.72 ± 0.14 | 0.84 ± 0.25 |
| Index | Experimental Diets | ||
|---|---|---|---|
| D1 | D2 | D3 | |
| Main FAs (% total FAs) | |||
| 12:0 | / | / | / |
| 14:0 | 0.44 ± 0.02 | 0.48 ± 0.03 | 0.42 ± 0.02 |
| 16:0 | 16.48 ± 1.86 | 14.90 ± 1.47 | 15.30 ± 1.34 |
| 18:0 | 5.01 ± 0.36 a | 3.70 ± 0.11 b | 2.18 ± 0.46 b |
| 20:0 | 0.45 ± 0.04 a | 0.26 ± 0.01 b | 0.23 ± 0.02 b |
| SFA | 23.08 ± 2.06 | 21.04 ± 1.84 | 21.15 ± 1.76 |
| 16:1 | 6.80 ± 0.91 | 6.82 ± 0.42 | 6.73 ± 0.44 |
| 18:1 | 32.04 ± 1.83 a | 29.13 ± 1.12 a,b | 27.50 ± 1.06 b |
| MUFA | 40.02 ± 3.83 a | 37.12 ± 2.82 a,b | 36.45 ± 1.88 b |
| 18:2n-6 | 34.04 ± 3.58 a | 30.66 ± 0.99 a,b | 28.09 ± 3.32 b |
| 18:3n-6 | 1.72 ± 0.02 a | 1.33 ± 0.03 b | 1.26 ± 0.59 b |
| 20:4n-6 | 0.73 ± 0.06 b | 0.78 ± 0.03 b | 0.91 ± 0.05 a |
| n-6 PUFA | 38.57 ± 3.55 a | 34.28 ± 1.54 a,b | 32.11 ± 3.04 b |
| 18:3n-3 | 1.87 ± 0.20 b | 2.16 ± 0.07 a | 2.09 ± 0.29 a |
| 18:4n-3 | 0.24 ± 0.08 | 0.25 ± 0.03 | 0.20 ± 0.02 |
| 20:5n-3 | 0.40 ± 0.01c | 0.68 ± 0.02 b | 0.89 ± 0.03 a |
| 22:6n-3 | 2.28 ± 0.32c | 4.21 ± 0.12 b | 7.68 ± 0.28 a |
| n-3 PUFA | 5.85 ± 0.40c | 8.02 ± 0.54 b | 11.63 ± 0.66 a |
| Experimental Diets | |||
|---|---|---|---|
| D1 | D2 | D3 | |
| Proximate composition (% wet matter) | |||
| Moisture | 76.20 ± 1.45 | 77.28 ± 2.34 | 77.02 ± 1.66 |
| Crude protein | 18.21 ± 0.23 | 17.89 ± 0.44 | 17.93 ± 0.55 |
| Crude lipid | 5.57 ± 0.47 | 4.94 ± 0.55 | 5.50 ± 0.63 |
| Nutritional indices of FAs | |||
| PUFA/SFA | 1.93 ± 0.16 | 2.01 ± 0.13 | 2.07 ± 0.15 |
| n-6/n-3 PUFA | 6.57 ± 0.21 a | 4.26 ± 0.37 b | 2.87 ± 0.15 c |
| FLQ | 2.69 ± 0.18 c | 4.89 ± 0.11 b | 8.57 ± 0.27 a |
| IA | 0.22 ± 0.01 | 0.21 ± 0.02 | 0.21 ± 0.02 |
| IT | 0.38 ± 0.02 a | 0.32 ± 0.02 a | 0.26 ± 0.03 b |
| HH | 4.52 ± 0.10 | 4.64 ± 0.08 | 4.53 ± 0.05 |
| Muscular edible quality | |||
| pH | 6.54 ± 0.02 | 6.53 ± 0.06 | 6.58 ± 0.03 |
| COP/% | 84.64 ± 2.43 | 88.32 ± 1.24 | 85.41 ± 1.60 |
| WHC/% | 6.14 ± 0.63 a | 6.84 ± 0.14 a | 7.61 ± 0.73 b |
| Muscular textural properties | |||
| Tenderness (gf) | 11.04 ± 0.59 | 11.78 ± 0.37 | 11.12 ± 0.84 |
| Hardness (gf) | 297.67 ± 24.31 | 349.04 ± 18.29 | 398.12 ± 26.36 |
| Adhesiveness (mJ) | 4.09 ± 0.51 | 4.87 ± 0.51 | 4.94 ± 0.60 |
| Springiness (mm) | 0.47 ± 0.02 | 0.46 ± 0.02 | 0.44 ± 0.02 |
| Chewiness (mJ) | 73.39 ± 7.37 | 71.89 ± 8.66 | 72.04 ± 7.01 |
| Gumminess (mJ) | 108.75 ± 10.08 a | 147.35 ± 7.53 b | 144.08 ± 8.37 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Z.; Xu, C.; Chen, F.; Lou, Y.; Nie, G.; Xie, D. Dietary DHA Enhanced the Textural Firmness of Common Carp (Cyprinus carpio L.) Fed Plant-Derived Diets through Restraining FoxO1 Pathways. Foods 2022, 11, 3600. https://doi.org/10.3390/foods11223600
He Z, Xu C, Chen F, Lou Y, Nie G, Xie D. Dietary DHA Enhanced the Textural Firmness of Common Carp (Cyprinus carpio L.) Fed Plant-Derived Diets through Restraining FoxO1 Pathways. Foods. 2022; 11(22):3600. https://doi.org/10.3390/foods11223600
Chicago/Turabian StyleHe, Zijie, Chao Xu, Fang Chen, Yunkun Lou, Guoxing Nie, and Dizhi Xie. 2022. "Dietary DHA Enhanced the Textural Firmness of Common Carp (Cyprinus carpio L.) Fed Plant-Derived Diets through Restraining FoxO1 Pathways" Foods 11, no. 22: 3600. https://doi.org/10.3390/foods11223600







