Abstract
Avocado oil is excellent functional oil. Effects of three extraction methods (squeezing extraction, supercritical carbon dioxide extraction, and aqueous extraction) on the species, composition, and contents of lipids in avocado oil were analyzed via ultra-performance liquid chromatography–time-of-flight tandem mass spectrometry (UPLC-TOF-MS/MS), and the differential components of lipids were revealed by OrthogonalPartialLeast Squares-DiscriminantAnalysis (OPLS-DA), S-plot combined with variable importance in the projection (VIP). The results showed that the fatty acid composition of avocado oil mainly consisted of oleic acid (36–42%), palmitic acid (25–26%), linoleic acid (14–18%), and palmitoleic acid (10–12%). A total of 134 lipids were identified first from avocado oil, including 122 glycerides and 12 phospholipids, and the total number of carbon atoms contained in the fatty acid side chains of the lipids was 32–68, and the number of double bonds was 0–9. Forty-eight differential lipid compounds with significant effects of the three extraction methods on the lipid composition of avocado oil were excavated, among which the differences in triglycerides (TG), phosphatidylethanol (PEtOH), and phosphatidylmethanol (PMeOH) contents were highly significant, which provided basic data to support the subsequent guidance of avocado oil processing, quality evaluation, and functional studies.
1. Introduction
Avocado is typical subtropical fruit, and “Hass”, “Choquette”, “Gwen”, “Lula” and “Maluma” are the main cultivated varieties, among which “Hass” has the largest planting area. Avocado is rich in nutrients, containing a variety of vitamins, tocopherols, and trace metal elements such as calcium, magnesium, and zinc, and the oil in the avocado pulp mainly consists of various monounsaturated fatty acids and polyunsaturated fatty acids, of which oleic acid accounts for 34% to 81%, 7.2–38.9% for palmitic acid, 6–26.6% for linoleic acid, 2.1–5.8% for linolenic acid, etc. [1,2,3]. Avocado oil has been applied to the development of products that aid in lowering blood pressure, are anti-inflammatory, and promote wound healing, with promising applications [4].
Fatty acids are very important components of avocado oil, and their mechanism of action enhances vascular function, reduces the deterioration of nephropathy, and improves nonalcoholic fatty liver in hypertensive rats by improving mitochondrial dysfunction, reducing mitochondrial oxidative stress, decreasing reactive nitrogen species (RNS) production and normalizing NOx activity [5,6,7]. Cristian et al. [8] used avocado oil instillation in hypertensive rats and reduced diastolic and systolic blood pressure by 21.2% and 15.5%, respectively. In addition, avocado oil not only increases collagen synthesis, reduced the number of inflammatory cells, accelerated the coagulation process and the regeneration of epithelial cells, thus accelerating wound healing [9], but also regulated brain-derived neurotrophic factor (BDNF), oxidative stress and apoptotic molecules, and protected SH–SY5Y cells against cortisol-induced cytotoxicity [10]. Omar et al. [11] used rats as a model for type 2 diabetes and confirmed that lipid components such as oleic acid in avocado oil delayed the development of diabetic nephropathy. Pham et al. [12] isolated DKB122 from avocado oil extract, which effectively inhibited TNF-α or LPS-induced p65 nuclear migration in HEI-OC1 cells and THP-1 cells and reduced TNF-α-induced expression of inflammatory chemokines and interleukin genes.
The functionality of avocado oil was closely related to its nutritional composition, while the nutritional quality of avocado oil was influenced by factors such as fruit variety [3,13], extraction method [14], and fruit storage method [15]. Lozano et al. [13] confirmed that total sterols were higher in immature fruits (1.1–6.2%) than in mature fruits (0.8–2.0%) in four avocado varieties, “Zutano”, “Bacon”, “Fuerte”, and “Lula”. Ultrasonic-assisted water extraction [16], mechanical pressing [17], and supercritical CO2 extraction [18,19,20] methods were commonly used to extract avocado, and the results of a comparative study by Tan et al. [18,19,20] showed that different extraction methods had an effect on physicochemical properties such as iodine value in avocado oil, but did not have a large effect on fatty acid composition such as oleic acid, which varied in content. Fernanda et al. [17] showed that drying of avocado at 60 °C combined with mechanical pressing resulted in better retention of the biological activity of avocado oil. The drying method and storage conditions had a greater influence on the quality of avocado oil. Chaiyavat et al. [21] showed that drying conditions at 80 °C and above had a significant effect on the stability of avocado oil and that light-free conditions helped to extend the shelf life of avocado oil. The stability and quality of avocado oil were susceptible to temperature effects and were not suitable for continuous heating processes [22,23]. However, little research had been reported on the effects of extraction methods on avocado lipid composition. In this study, ultra-performance liquid chromatography–time-of-flight tandem mass spectrometry (UPLC-TOF-MS/MS) was used to investigate the effects of extraction methods on the components of avocado oil quality, to explore the differential components of avocado oil quality by extraction methods, and to provide basic data support for avocado oil extraction methods, product development, and functional studies.
2. Materials and Methods
2.1. Preparation of Avocado Oil
The variety of avocado was “Hass”, purchased from Zhanjiang Chang-da-Chang Super Shopping Plaza Co., and the fruit was 80% mature (skin color changed from dark green to dark brown). Referring to the method of Liu et al. [1], three methods of squeezing extraction, supercritical carbon dioxide extraction, and aqueous extraction were used to extract the oil from avocado pulp, the crude oil was centrifuged in a centrifuge at 5000× g for 10 min, and the collected oil layer was stored at 4 °C. The parameters of that three methods were as follows:
Squeezing extraction: Avocado pulp dried at 55 °C for 24 h was squeezed by the sing screw expeller with normal temperature mode, and the crude oils were collected and centrifuged at 5000× g for 10 min, and the crude oil layer was collected.
Supercritical carbon dioxide extraction: Avocado pulp dried at 55 °C for 24 h was extracted in a supercritical carbon dioxide extractor. The extraction temperature grades I and II were 45 °C and 50 °C, respectively, and the extraction pressure grades I and II were 5 MPa and 6 MPa, respectively, and the crude oil was collected.
Aqueous extraction: A 1 kg sample of avocado oils and 2 kg distilled water were beaten and mixed evenly, and then colloid mill was used for 1 min to obtain slurry solution. Then, 2 kg distilled water was used to clean the machine, and cleaning solutions were collected. The slurry solution and cleaning solution, adjusted to 8.0 with a 1.00 mol/L sodium hydroxide solution, were combined and stirred for 1.5 h at 75 °C water bath, then the mixed solution was centrifuged at 25,000× g for 10 min, and the upper crude oil was collected.
2.2. Instrumentations
Squeezer (OP101, Shenzhen Yimeikang Electronic Commerce Co., Ltd., Shenzhen, China), supercritical carbon dioxide extractor (HSFE-5 + 1, Jiangsu Gaoke Pharmaceutical Equipment Co., Ltd., Nantong, China), high-speed freezing centrifuge (CR22GIII, Hitachi Limited, Tokyo, Japan), juicer (JYL-C020E, Jiuyang Co., Ltd., Jinan, China), pipeline high shear colloid mill (ZVF300-G5R5/P7R5T4MD, Shanghai Qike Machinery Equipment Co., Ltd., Shanghai, China), ultraviolet–visible spectrophotometer (UV-1780, Shimadzu Corporation, Kyoto, Japan), gas chromatography–mass spectrometry (AOC5000-GC/MS-QP2010plus, Shimadzu Corporation, Kyoto, Japan), ultra-high performance liquid chromatograph–time-of-flight tandem mass spectrometer (LC-30A liquid chromatography, Shimadzu Corporation, Kyoto, Japan), ultra-pure water system (Milli-Q-Synthesis, Milli-pore Company, Boston, MA, USA), multi-tube vortex mixer (MTV-100, Hangzhou Aosheng Instrument Co., Ltd., Hangzhou, China), nitrogen blower (DC-24, Shanghai Ampu Experimental Technology Co., Ltd., Shanghai, China).
2.3. Determination of Fatty Acid Composition
The fatty acid composition in avocado oil was determined by potassium hydroxide methylation method with reference to the method of Liu et al. [24]. A sample of 1.0 μL passed through the chromatographic column (DB-FastFA, 30 m × 0.25 mm × 0.25 μm, Agilent, California, USA) in gas chromatography–mass spectrometry with the inlet temperature of 260 °C, nitrogen as the carrier gas, and the split ratio of 20:1. The initial temperature of the column was 150 °C, then it was raised below the program, and the speed of 10 C/min was raised to 210 °C and kept for 8 min, and the speed of 20 °C/min was raised to 230 °C and kept for 6 min. Finally, the sample passed through a detector with a temperature of 280 °C.
2.4. Determination of Lipid Composition
The lipid composition in avocado oil was determined equipped with a Phenomenex Kinete C18 column (100 × 2.1 mm, 2.6 µm, Phenomenex, Torrance, CA, USA) with reference to the method of Liu et al. [24,25]. One microliter of sample was pumped onto the C18 column at a rate of 0.4 mL/min. The column temperature and chamber temperature were 60 °C and 4 °C, respectively. The mobile phases A and B consisted of H2O–methanol–acetonitrile = 1:1:1 (containing 5 mmol/L ammonium acetate) and isopropanol-acetonitrile = 5:1 (containing 5 mmol/L ammonium acetate). The elution program of mobile phase was performed as 20% B for 0.5 min, 40% B for 1.5 min, 60% B for 3 min, 98% B for 13 min, 20% B for 13 min, and 20%B for 17 min.
2.5. Data Processing and Analysis
All samples were measured 3 times in parallel. The qualitative analysis of shotgun-MS data was treated by the LipidView software (v2.0, ABSciex, Concord, ON, Canada). In the process of data analysis, the analysis parameters were set according to the following figures: the mass tolerance was 0.5, the minimum signal-to-noise ratio was 10, the minimum% intensity was 1, the average flow injection spectrum from the top was 30% TIC, and the total double bonds were ≤12. OriginPro (2021, OriginLab Corporation, Northampton, UK) was used for plotting, thermal map analysis, and statistical analysis, and the SIMCA (14.1, Sartorius Lab Instruments GmbH & Co., KG, Goettingen, Germany) was used for PCA, OPLS-DA, VIP, and S-plot analysis, etc.
3. Results and Analysis
3.1. Analysis of Fatty Acid Composition and Lipid Composition in Avocado Oil
The fatty acid composition in avocado oil was determined by gas chromatography-mass spectrometer (GC-MS), and the retention time of each fatty acid standard was characterized with reference to the retention time of each fatty acid standard, and the relative percentage content was calculated according to the normalization method of chromatographic peak area. The fatty acids of avocado oil mainly consisted of oleic acid (36–42%), palmitic acid (25–26%), linoleic acid (14–18%), palmitoleic acid (10–12%), isoleic acid (6–7%), linolenic acid (0.5–0.8%) and stearic acid (0.5–0.6%). The content of saturated fatty acids and unsaturated fatty acids in avocado oil obtained by three extraction methods was about 26% and 73%, among which the content of monounsaturated fatty acids ranged from 54 to 60%.
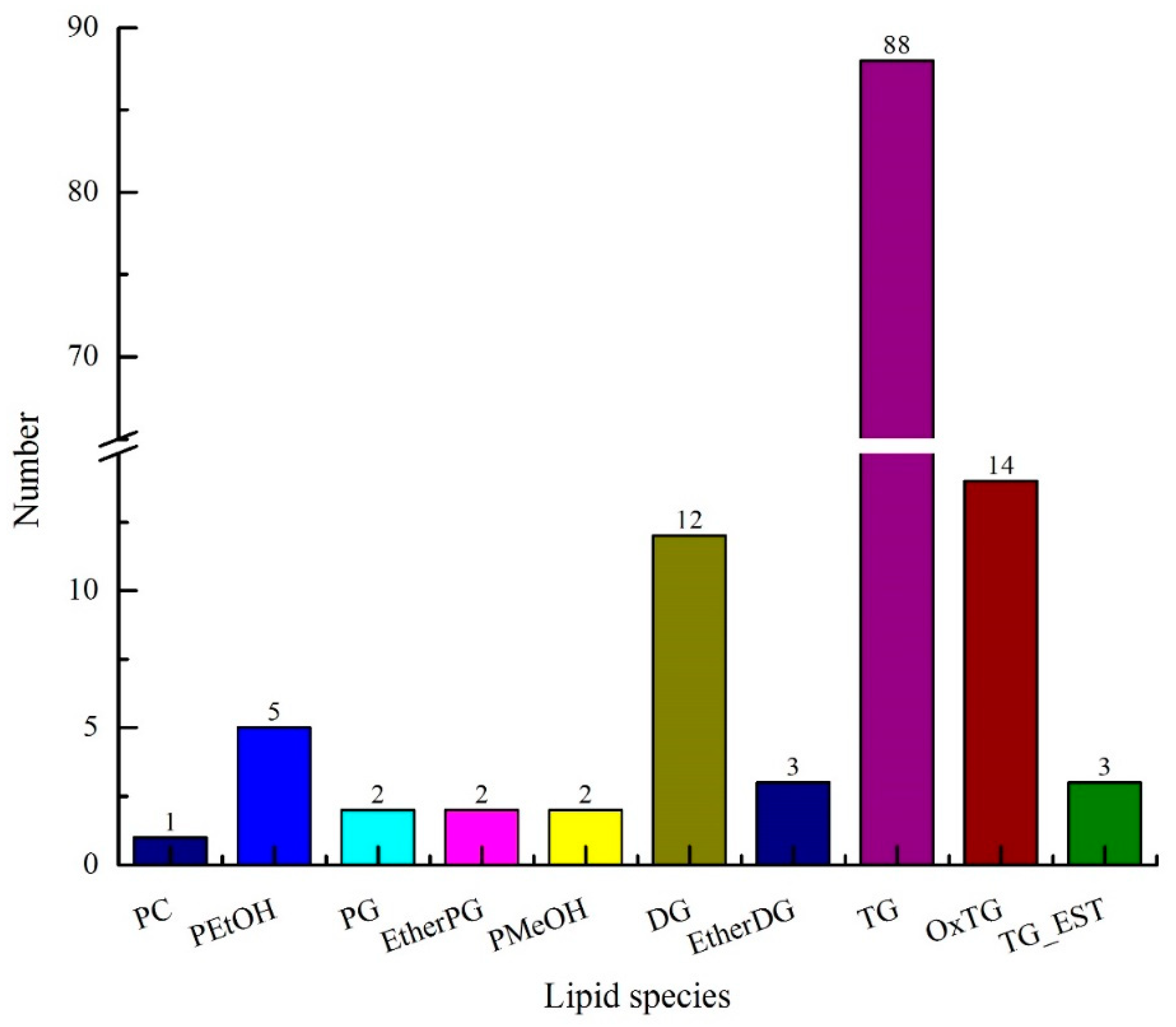
UPLC-TOF-MS/MS combined with composite scanning mode was used to analyze the lipid composition in avocado oil, as well as the accurate relative molecular weight, isotope distribution, and secondary mass spectrometry fragmentation information. As shown in Figure 1, a total of 134 lipids were identified in avocado oil, including 122 glycerides and 12 phospholipids. Glycerides were composed of diacylglycerol (DG), ether-linked diacylglycerol (EtherDG), triglycerides (TG), oxidized triglycerides (OxTG), triglycerides (TG_EST), and ether-linked triglycerides (EtherTG), and among of which type numbers were 12, 3, 88, 14, 3, and 2, respectively. Phospholipids were composed of phosphatidylcholine (PC), phosphatidylethanol (PEtOH), phosphatidylglycerol (PG), ether-linked phosphatidylglycerol (EtherPG), and phosphatidylmethanol (PMeOH), and among of which, type numbers were 1, 5, 2, 2, and 2, respectively.
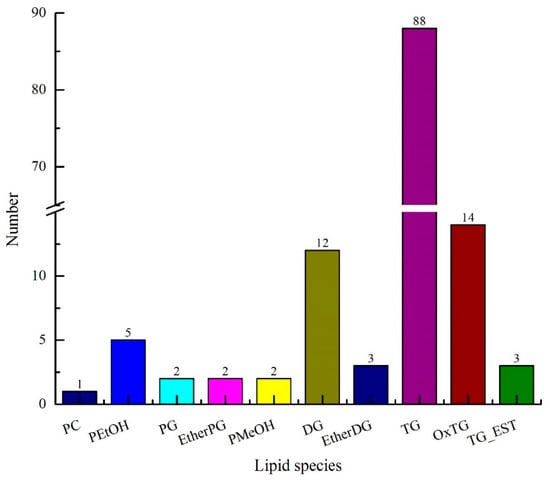
Figure 1.
Lipids of composition of avocado oil.
As can be seen from Table 1, the total number of carbon atoms in the fatty acid side chains of lipids in avocado oil was 32–68, and the number of double bonds was 0–9. The carbon atoms and double bonds number of DG, EtherDG, TG, OxTG, TG_EST, and EtherTG in glycerides were (32–42, 0–5), (34–36, 2–4), (34–64, 0–9), (50–54, 2–5), (66–68, 3–4), and (53–55, 2–5), respectively. The side chain of glycerides was mainly composed of C15, C16, C17, C18, and C19. The carbon atoms and double bonds number of PC, PEtOH, PG, EtherPG, and PMeOH in glycerides were (34, 2), (34–36, 1–4), (32–34, 0–1), (34–37, 3–5), and (34, 0), respectively.

Table 1.
Composition of the 134 lipids in avocado oil.
3.2. Analysis of Lipid Content in Avocado Oil
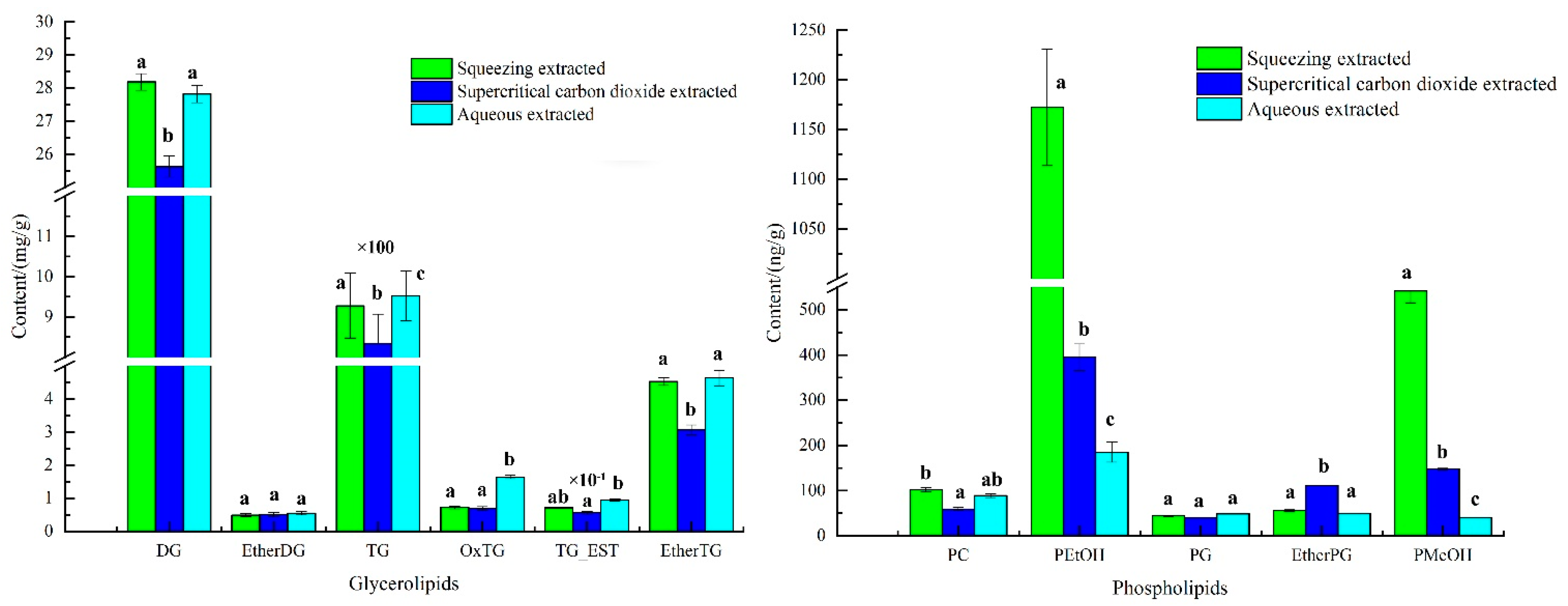
The lipid content of avocado oil obtained by three extraction methods was shown in Figure 2. As shown in Figure 2, the highest TG content in glycerides of avocado oil was (830–960) mg/g, followed by DG at (25–30) mg/g, and the highest PEtOH content in phospholipids was (180–1200) ng/g, followed by PMeOH at (40–545) ng/g. The significant difference results showed that the three extraction methods had the highest effect on the TG, PEtOH and PMeOH contents were highly significant, and the differences for EtherDG and PG contents were not significant.
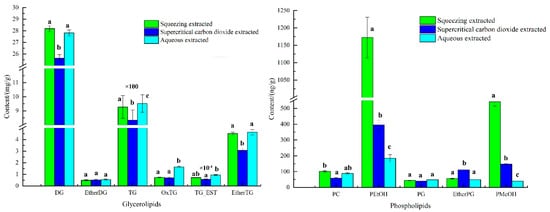
Figure 2.
The composed of phospholipids and glycerides in avocado oil from three extraction methods. Note: Different letters a, b and c represented significant difference.
3.3. Modeling and Evaluation of Differential Metabolites of Lipids in Avocado Oil
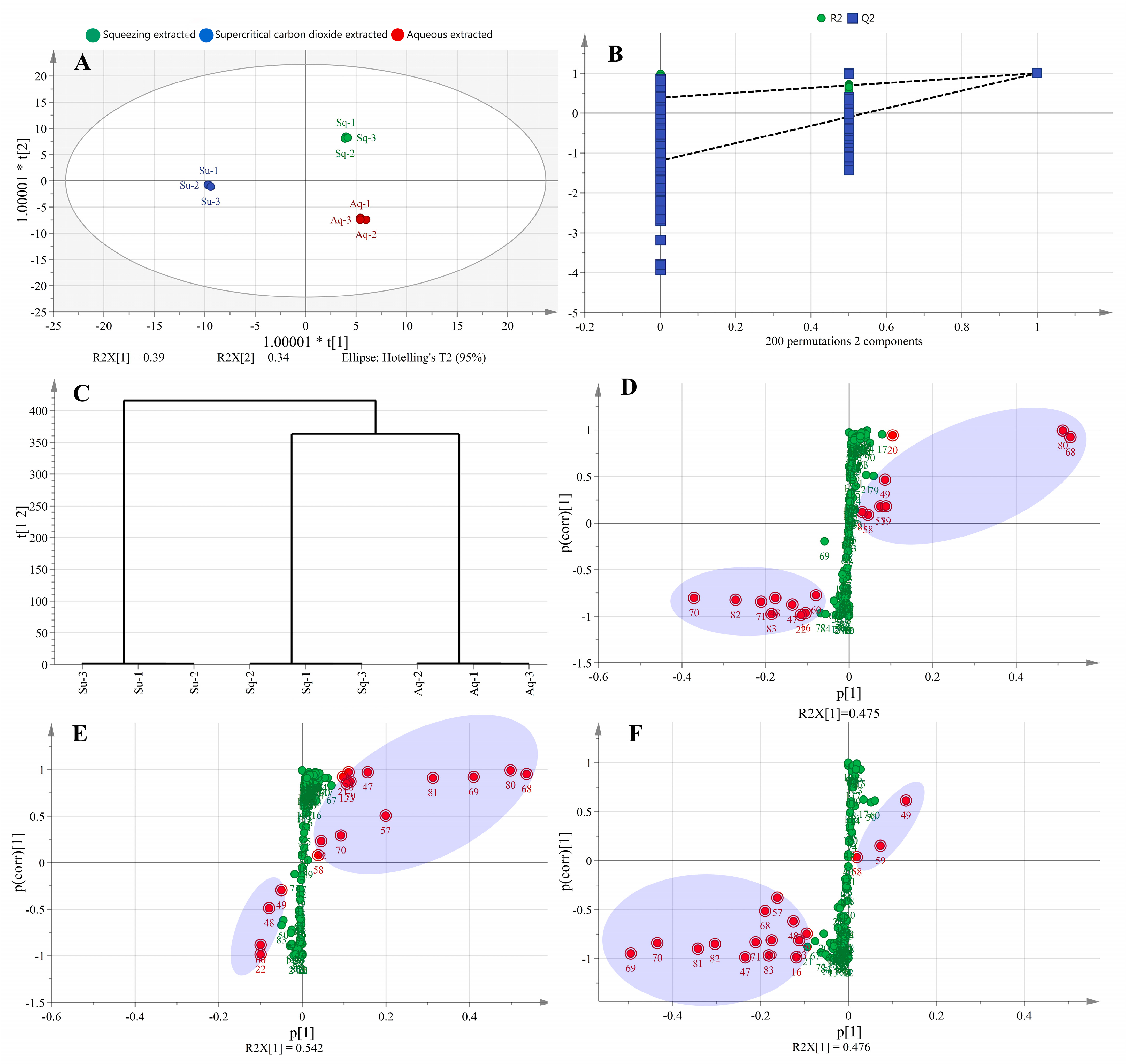
From Figure 3A, it can be seen that the avocado oil samples obtained by the three extraction methods could be better distinguished in the OPLS–DA model, and the three oil samples were distributed in the first, third, and fourth quadrants, indicating that they differed from each other. From Figure 3B, it can be seen that the lipid composition data obtained by the three extraction methods were subjected to permutation test and cross–validation analysis (CV–ANOVA), the intercepts of R2 and Q2 curves with vertical coordinates were less than one, and the intercept of Q2 in vertical coordinates was less than zero, indicating that the established OPLS–DA model did not show any overfitting phenomenon. In addition, the significance probability value p < 0.05 in CV–ANOVA analysis indicated that the established OPLS–DA model was stable, reliable, and statistically significant [26]. As shown in Figure 3C, the avocado oil obtained from the three extraction methods was well clustered.
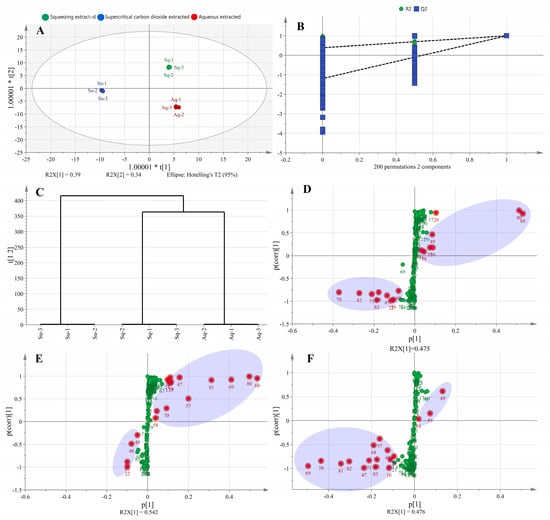
Figure 3.
Scores plot of OPLS-DA model (A), cross-validation of OPLS-DA model (B), HCA (C), and S-plot of lipids with different extraction methods. Sq, Su, Aq in Figure 3A represent squeezing extraction, supercritical carbon dioxide extraction, and aqueous extraction, respectively. (D) Represents S–plot of squeezing extracted and aqueous extracted, (E) represents S–plot of supercritical carbon dioxide extracted and aqueous extracted, and (F) represents S–plot of supercritical carbon dioxide extracted and squeezing extracted. The red dots and green dots in Figure 3D–F indicate metabolites with VIP values > 1, and VIP values < 1, respectively.
The S–plot was used to identify significant differential metabolites between the two samples, and metabolites with large contributions were concentrated at two ends of the S–plot, while those with small contributions were concentrated around the origin [27]. The abscissa and ordinate represented the co-correlation coefficient and correlation coefficient of the principal component and metabolite, respectively. The red dots in Figure 3D–F indicate metabolites with VIP values >1. From Figure 3D, seventeen significantly different components were analyzed between the squeezing extraction and aqueous extraction methods, including eight metabolites with VIP values >2, namely TG 52:2|TG 16:0_18:1_18:1 (68), TG 54:3|TG 18:1_18:1_18:1 (80), TG 52:4|TG 16:1_18:1_18:2 (70), TG 50:2|TG 16:0_16:1_18:1 (58), TG 54:5|TG 18:1_18:2_18:2 (82), TG 50:3|TG 16:0_16:1_18:2 (59), TG 50:1|TG 16:0_16:0_18:1 (57), TG 52:5|TG 16:1_18:2_18:2 (71). From Figure 3 (E), eighteen significantly different components were analyzed between the supercritical carbon dioxide extraction and aqueous extraction methods, including eight metabolites with VIP values >2, namely TG 52:2|TG 16:0_18:1_18:1 (68), TG 54:3|TG 18:1_18:1_18:1 (80), TG 52:3|TG 16:0_18:1_18:2 (69), TG 54:4|TG 18:1_18:1_18:2 (81), TG 50:2|TG 16:0_16:1_18:1 (58), TG 50:1|TG 16:0_16:0_18:1 (57), TG 50:3|TG 16:0_16:1_18:2 (59), TG 52:4|TG 16:1_18:1_18:2 (70). From Figure 3F, seventeen significantly different components were analyzed between the supercritical carbon dioxide extraction and squeezing extraction methods, including ten metabolites with VIP values >2, namely TG 52:3|TG 16:0_18:1_18:2 (69), TG 52:4|TG 16:1_18:1_18:2 (70), TG 54:4|TG 18:1_18:1_18:2 (81), TG 54:5|TG 18:1_18:2_18:2 (82), TG 50:3|TG 16:0_16:1_18:2 (59), TG 50:1|TG 16:0_16:0_18:1 (57), TG 48:0|TG 16:0_16:0_16:0 (47), TG 50:2|TG 16:0_16:1_18:1 (58), TG 52:2|TG 16:0_18:1_18:1 (68), TG 52:5|TG 16:1_18:2_18:2 (71).
3.4. Differential Metabolite Differential Analysis and Mass Spectrometry of Lipids in Avocado Oil
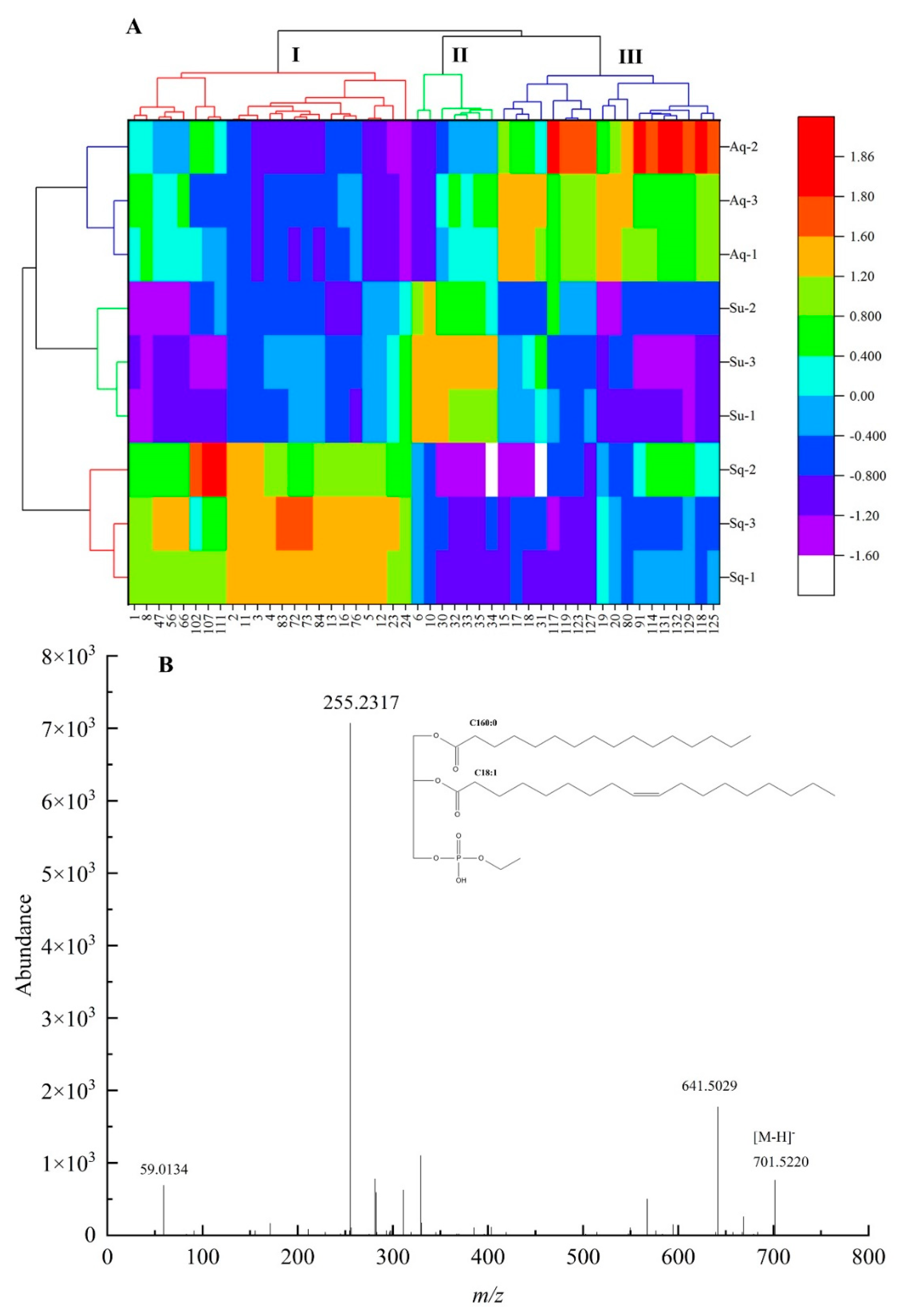
VIP analysis of lipid components in avocado oil obtained by the three extraction methods was performed on the OPLS–DA model, 77 differential metabolites with VIP value > 1 were obtained, and Krural–Walli’s significance test was performed on the 77 metabolites, and 48 significantly different metabolites were obtained. The differential metabolites were subjected to Z–score transformation to standardize the data, Z–score = (original data – mean)/standard deviation, and the standardized data were produced as a heat map, as shown in Figure 4A.
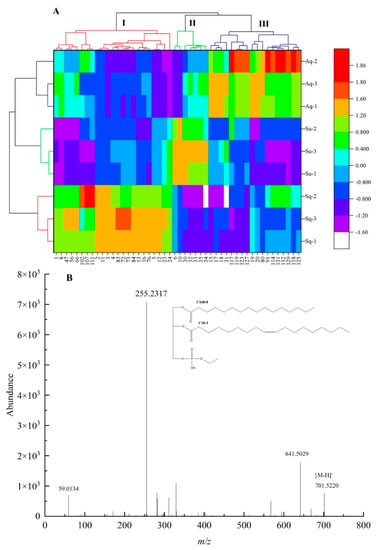
Figure 4.
(A) Represented the thermogram of lipid components with significant differences among the three extraction methods, and (B) represented the mass spectrogram of PETOH 34:1|PETOH 16:0 _18:1 in negativity mode.
From Figure 4A, it can be seen that the metabolites of the three extraction methods can be categorized into three groups. Groups I, II, and III were the groups with significant upregulation of differential lipid components obtained by the squeezing extraction method, supercritical carbon dioxide extraction method, and aqueous extraction method, respectively, in which 23 lipid components including PC 34:2 (1), PEtOH 34:1|PEtOH 16:0_18:1 (2), PEtOH 34:2|PEtOH 16:0_18:2 (3), PEtOH 36:2|PEtOH 18:1_18:1 (4), TG 60:3|TG 24:0_18:1_18:2/TG 26:0_16:1_18:2 (102), and TG 61:4|TG 25:0_18:2_18:2 (107) were upregulated in group I. Seven lipid components including PEtOH 36:4|PEtOH 18:2_18:2 (6), PG O–37:5|PG O–16:2_21:3 (10), TG 38:0|TG 8:0_14:0_16:0 (30), TG 40:0|TG 10:0_14:0_16:0 (32), TG 40:1|TG 10:0_12:0_18:1 (33), TG 42:1|TG 8:0_16:0_18:1 (35) were upregulated in group II. Twenty–three lipid components including TG 54:3|TG 18:1_18:1_18:1 (80), TG 59:1|TG 16:0_25:0_18:1 (97), TG 64:2|TG 28:0_18:1_18:1 (114), TG 50:3;1O|TG 16:0_18:2_16:1;1O (117), TG 52:2;1O|TG 16:0_18:1_18:1;1O (118), TG 52:3;1O|TG 16:0_18:1_18:2;1O (119), TG 52:2;2O|TG 16:0_19:1_17:1;2O (123), TG 52:4;2O|TG 16:0_18:2_18:2;2O (125), TG 54:3;2O|TG 18:1_18:1_18:1;2O (127), TG 54:5;2O|TG 18:2_19:2_17:1;2O (129) were upregulated in group III.
After the precursor ions of selected lipid molecules enter the mass spectrometry Q2, collision–induced dissociation (CID) occurs at a certain collision energy (CE), resulting in fragment ions, and the neutral loss of specific fragment ions or specific functional groups from lipid molecules lead to diagnostic ions. In this study, the differential metabolite PEtOH 34:1|PEtOH 16:0_18:1 in phosphatidylethanol (PEtOH) was used as an example to analyze its mass spectrometric behavior and fracture mechanism in detail, as shown in Figure 4B. From Figure 4B, m/z 701.5220 corresponded to the mass spectrum information of [M − H]− parent ion of PEtOH 34:1, m/z 125.0009 was phosphoethanol, and m/z 255.2327 and m/z 281.2475 represented the mass spectrum information of [FA 16:0–H]− and [FA 18:1–H]−, respectively.
4. Discussion
There were few studies on the lipid composition in avocado oil, but there were more studies on pitaya seed oil, coffee bean oil, canola oil, and soybean oil. The present study showed that avocado oil was mainly composed of oleic acid (36–42%), palmitic acid (25–26%), linoleic acid (14–18%), and palmitoleic acid (10–12%), similar to the fatty acid composition in avocado reported by Fernandes et al. [28], but there was variability in the fatty acid content, such as low oleic acid content of 10–20%, palmitic acid content was 10–15% higher, and linoleic acid was about 5% higher, with differences in the variety and origin of avocado leading to differences between the two.
In this study, a total of 134 lipid molecules were identified from different extraction methods, which was less than that of pitaya seed oil (152) [24] and cycad oil (169) [29]. Avocado oil was similar to pitaya seed oil in that it consists mainly of glycerides and phospholipids and had the highest content of TG in glycerides and PEtOH in phospholipids, but some variability exists in that avocado oil contained phosphatidylcholine PC, which was lacking in dragon fruit seed oil [24]. Additionally, it was based on the variability of glycerides and phospholipid species in oils and fats that much research work had been completed to identify the source, quality, and variety of oils and fats. Tian et al. [30] analyzed and identified 24 triglycerides, mainly OOO (triglyceride of trioleic acid), OOL (triglyceride of 1,2–dioleic acid–3–linoleic acid), OOP (triglyceride of 1,2–dioleic acid–3–palmitic acid), and other unsaturated triglycerides from six different oil tea species and nine different common oil tea varieties and constructed a fingerprint profile of triglycerides in oil tea seeds. The fingerprint profiles of triglycerides in oil tea seeds were also constructed to identify different varieties of oil tea seed oil. The results of Zhao et al. [31] showed that LL and OO in DAGs and OLLn and LLL in TAGs were important indicators for the grade identification of olive oil, and these indicators could be used for the quality identification of different grades of olive oil. Therefore, the information on the type and content of microscopic lipid components in oils and fats by profiling could provide new ideas and more accurate analysis for the source, type, and quality identification of oils and fats.
The fatty acid content and composition of avocado oil varied depending on the variety, origin [32], and extraction method [1,19,33], with the differences existing in extraction methods being particularly pronounced, yet there were few comparative studies from a microscopic perspective. In this study, 48 differential metabolites were identified from 134 lipid components using OPLS–DA combined with VIP and other methods, among which 23, 7, and 23 differential metabolites were upregulated by the squeezing extraction, supercritical carbon dioxide extraction, and aqueous extraction, respectively, while phospholipids were more abundant in avocado oil obtained by supercritical carbon dioxide extraction, which was in accordance with the principle of similar compatibility. The long extraction process by the aqueous extraction method and the long air contact time resulted in higher OxTG content. In addition, the principles of the pressing method and extraction method were different, resulting in differences in both PMeOH and glycerol ester compounds. Therefore, revealing the differences among the oils and fats obtained by the three extraction methods from the perspective of lipid molecules could provide basic data to support the study of the transformation mechanism of lipid molecules during processing.
5. Conclusions
In this study, the UPLC–TOF–MS/MS was used to profile the lipid profile of avocado oil first, and 134 lipid components were identified, including 122 glycerides and 12 phospholipids. The total number of carbon atoms contained in the fatty acid side chains of the lipids ranged from 32 to 68, and the number of double bonds ranged from 0 to 9. The differences between the three extraction methods were highly significant for the contents of TG, PEtOH, and PMeOH, and not significant for the contents of EtherDG and PG. The analysis by OPLS–DA, S–plot, and VIP identified 44 differential metabolic components, which provided theoretical data for guiding the avocado oil’s processing, quality evaluation, and in–depth functional research.
Author Contributions
Conceptualization, Y.L. and Q.X.; methodology, Y.L., Y.Q. and Y.K.; software, J.L. and Y.K.; formation analysis, Y.L., L.L. and Y.Q.; investigation, Y.L., Y.Q. and Q.X.; resources, L.L. and Q.X.; writing—original draft preparation, Y.L., Y.Q. and Q.X.; writing—review and editing, L.L.; supervision, Y.K. and J.L.; project administration, Y.L. and Q.X.; funding acquisition, Y.L. and Q.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Hainan Provincial Natural Science Foundation of China (No. 320QN326), the 2021 Guangdong Science and Technology Innovation Strategy Special Fund (No. 2021A101 and 2021A05217), and the Basic and Applied Basic Research Foundation of Guangdong Province of China (No. 2021A1515010538), and the Key Laboratory of Food Nutrition and Functional Food of Hainan Province (KF202007).
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Liu, Y.J.; Bu, M.T.; Tan, G.; Chen, W.T.; Chen, X.Y.; Zhang, L.; Li, J.H. Comparative study on physicochemical properties, antioxidant activity and fatty acid composition of avocado oil by different extraction methods. J. Sichuan Agric. Univ. 2020, 38, 161–167. [Google Scholar] [CrossRef]
- Pérez-Saucedo, M.R.; Jiménez-Ruiz, E.I.; Rodríguez-Carpena, J.G.; Ragazzo-Sánchez, J.A.; Ulloa, J.A.; Ramírez-Ramírez, J.C.; Gastón-Peña, C.R.; Bautista-Rosales, P.U. Properties of the avocado oil extracted using centrifugation and ultrasound-assisted methods. Food Sci. Biotechnol. 2021, 30, 1051–1061. [Google Scholar] [CrossRef]
- Elosaily, A.H.; Mahrous, E.A.; Salama, A.A.; Salama, A.M.; Elzalabani, S.M. Composition, anti-inflammatory, and antioxidant activities of avocado oil obtained from Duke and Fuerte cultivars. J. Am. Oil Chem. Soc. 2021, 99, 181–186. [Google Scholar] [CrossRef]
- Aktar, T.; Adal, E. Determining the Arrhenius Kinetics of Avocado Oil: Oxidative Stability under Rancimat Test Conditions. Foods 2019, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Adrián, M.C.; Eridani, O.B.; Isabel, G.C.; Elizabeth, C.; Rocío, M.; Alfredo, S.; Raimundo, R.A.; Christian, C. Avocado oil prevents kidney injury and normalizes renal vasodilation after adrenergic stimulation in hypertensive rats: Probable role of improvement in mitochondrial dysfunction and oxidative stress. Life 2021, 11, 1122. [Google Scholar] [CrossRef]
- Olmos-Orizaba, B.E.; Márquez-Ramírez, C.A.; Garcia-Berumen, C.I.; Villagómez, A.V.H.; Calderón-Cortés, E.; Saavedra-Molina, A.; Montoya-Pérez, R. Avocado Oil Alleviates Renal Damage and decreases NADPH Oxidase Activity, Peroxynitrite Production and Mitochondrial Calcium Uptake in Hypertension Rats. FASEB J. 2019, 33 (Suppl. S1), 660. [Google Scholar] [CrossRef]
- Garcia-Berumen, C.I.; Olmos-Orizaba, B.E.; Márquez-Ramírez, C.A.; Orozco, A.R.R.; González-Cortez, A.; Saavedra-Molina, A.; Montoya-Pérez, R.; Cortés-Rojo, C. Avocado oil ameliorates non-alcoholic fatty liver disease by down-regulating inflammatory cytokines and improving mitochondrial dynamics. FASEB J. 2019, 33 (Suppl. S1), 660. [Google Scholar] [CrossRef]
- Márquez-Ramírez, C.A.; Paz, J.L.H.d.l.; Ortiz-Avila, O.; Raya-Farias, A.; González-Hernández, J.C.; Rodríguez-Orozco, A.R.; Salgado-Garciglia, R.; Saavedra-Molina, A.; Godínez-Hernández, D.; Cortés-Rojo, C. Comparative effects of avocado oil and losartan on blood pressure, renal vascular function, and mitochondrial oxidative stress in hypertensive rats. Nutrition 2018, 54, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, R.E.S.; Mehdi, F.; Elham, M.K. Histomorphological examination of skin wound healing under the effect of avocado oil in wistar rats. Acta Vet. Eurasia 2021, 47, 121–128. [Google Scholar] [CrossRef]
- Rosso, M.J.; Cruz, J.I.E.D.; Farina, A.V.; Ferreira, T.C.; Elizabete, B.L.; Augusto, D.O.N.D.; Echart, M.M.A.; Frescura, D.M.M.M.; Aguiar, M.E.; Mânica, C.I.B.; et al. Avocado oil (Persea americana) protects SH-SY5Y cells against cytotoxicity triggered by cortisol by the modulation of BDNF, oxidative stress, and apoptosis molecules. J. Food Biochem. 2021, 45, e13596. [Google Scholar] [CrossRef]
- Ortiz-Avila, O.; Saavedra-Molina, A.; Cortés-Rojo, C. Effect of avocado oil on metabolic profile and development of diabetic nephropathy in goto-kakizaki rats. FASEB J. 2019, 33 (Suppl. S1), 487. [Google Scholar] [CrossRef]
- Pham, T.N.M.; Jeong, S.Y.; Kim, D.H.; Park, Y.H.; Lee, J.S.; Lee, K.W.; Moon, I.S.; Choung, S.Y.; Kim, S.H.; Kang, T.H.; et al. Protective mechanisms of avocado oil extract against ototoxicity. Nutrients 2020, 12, 947. [Google Scholar] [CrossRef] [PubMed]
- Lozano, Y.F.; Mayer, C.D.; Bannon, C.; Gaydou, E.M. Unsaponifiable matter, total sterol and tocopherol contents of avocado oil varieties. J. Am. Oil Chem. Soc. 1993, 70, 561–565. [Google Scholar] [CrossRef]
- Costagli, G.; Betti, M. Avocado oil extraction processes: Method for cold-pressed high-quality edible oil production versus traditional production. J. Agric. Eng. 2015, 46, 115–122. [Google Scholar] [CrossRef]
- Liu, Y.J.; Bu, M.T.; He, J.N.; Zhan, Y. Characterization of the volatile organic compounds produced from avocado during ripening by gas chromatography ion mobility spectrometry. J. Sci. Food Agric. 2020, 101, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Du, C.; Luo, Y.H. Ultrasonic-assisted extraction technology of oil from avocado. Food Ind. 2020, 41, 104–107. [Google Scholar]
- Krumreich, F.D.; Borges, C.D.; Mendonça, C.R.B.; Jansen-Alves, C.; Zambiazi, R.C. Bioactive compounds and quality parameters of avocado oil obtained by different processes. Food Chem. 2018, 257, 376–381. [Google Scholar] [CrossRef]
- Tan, C.X.; Hean, C.G.; Hamzah, H.; Ghazali, H.M. Optimization of ultrasound-assisted aqueous extraction to produce virgin avocado oil with low free fatty acids. J. Food Process Eng. 2018, 41, 9. [Google Scholar] [CrossRef]
- Tan, C.X.; Chong, G.H.; Hamzah, H.; Ghazali, H.M. Comparison of subcritical CO2 and ultrasound-assisted aqueous methods with the conventional solvent method in the extraction of avocado oil. J. Supercrit. Fluids 2018, 135, 45–51. [Google Scholar] [CrossRef]
- Tan, C.X.; Chong, G.H.; Hamzah, H.; Ghazali, H.M. Hypocholesterolaemic and hepatoprotective effects of virgin avocado oil in diet-induced hypercholesterolaemia rats. Int. J. Food Sci. Technol. 2018, 53, 2706–2713. [Google Scholar] [CrossRef]
- Chaiyasut, C.; Kesika, P.; Sirilun, S.; Makhamrueang, N.; Peerajan, S.; Sivamaruthi, B.S. Influence of extraction process on yield, total phenolic content, and antioxidant properties of avocado (persea americana mill.) oil and stability assessment. Asian J. Pharm. Clin. Res. 2019, 12, 391–396. [Google Scholar] [CrossRef]
- Braga, R.L.M.; Rios, d.S.V.; Dias, F.G.M.; Antônio, N.C. Changes in quality and phytochemical contents of avocado oil under different temperatures. J. Food Sci. Technol. 2019, 56, 401–408. [Google Scholar] [CrossRef]
- Forero-Doria, O.; García, M.F.; Vergara, C.E.; Guzman, L. Thermal analysis and antioxidant activity of oil extracted from pulp of ripe avocados. J. Therm. Anal. Calorim. 2017, 130, 959–966. [Google Scholar] [CrossRef]
- Liu, Y.J.; Tu, X.H.; Lin, L.J.; Du, L.Q.; Feng, X.Q. Analysis of lipids in pitaya seed oil by ultra-performance liquid chromatography–time-of-flight tandem mass spectrometry. Foods 2022, 11, 2988. [Google Scholar] [CrossRef] [PubMed]
- Aipeng, H.; Fang, W.; Fenghong, H.; Ya, X.; Bangfu, W.; Xin, L.; Hong, C. Comprehensive and high-coverage lipidomic analysis of oilseeds based on ultrahigh-performance liquid chromatography coupled with electrospray ionization quadrupole time-of-flight mass spectrometry. J. Agric. Food Chem. 2021, 69, 8964–8980. [Google Scholar] [CrossRef]
- Liu, Y.J.; Qian, Y.Y.; Shu, B.; Liu, Y.Y.; Tu, X.H.; Ouyang, H.J.; Li, Y.; Tan, G.; Yu, Z.W.; Chen, F.; et al. Effects of four drying methods on Ganoderma lucidum volatile organic compounds analyzed via headspace solid-phase microextraction and comprehensive two-dimensional chromatography-time-of-flight mass spectrometry. Microchem. J. 2021, 166, 106258. [Google Scholar] [CrossRef]
- Xiangwu, H.; Lihong, Z.; Sheng, P.; Yijun, L.; Jianrong, L.; Meiqian, Z. Effects of varieties, cultivation methods, and origins of citrus sinensis ‘hongjiang’ on volatile organic compounds: HS-SPME-GC/MS analysis coupled with OPLS-DA. Agriculture 2022, 12, 1725. [Google Scholar] [CrossRef]
- Fernandes, G.; Gómez-Coca, R.; Camino, M.; Moreda, W.; Arellano, D. Chemical characterization of commercial and single-variety avocado oils. Grasas Y Aceites 2018, 69, 256. [Google Scholar] [CrossRef]
- Wei, H.L.; Lv, X.; Xie, Y.; Xu, S.L.; Chen, H.; Wei, F. Lipid of prinsepia utilis royle oil by utra-performance liquid chromatography-time of flight-tandem mass spectrometry. Chin. J. Oil Crop Sci. 2019, 41, 947–955. [Google Scholar] [CrossRef]
- Tian, X.X.; Fang, X.Z.; Sun, H.Z.; Du, M.H. Analysis of triacylglycerols in different oil-tea camellia cones. For. Res. 2018, 31, 41–47. [Google Scholar] [CrossRef]
- Zhao, S.Z.; Feng, Z.R.; Bao, M.; Yi, X.H.; Deng, X.J.; Guo, D.H.; Ding, T.; Liu, H. Grade identification of olive oil grade identification based on chemometrics combined with DAGs and TAGs analysis. J. Chin. Cereals Oils 2022, 37, 288–294. Available online: https://kns.cnki.net/kcms/detail/11.2864.TS.20211217.1306.002.html (accessed on 1 January 2020).
- Wang, J.Y.; Shang, Y.E.; Zhang, D.; Fang, O. Comparison of qualities of avocado and its oil from different origins and maturities. China Oils Fats 2018, 43, 94–97. [Google Scholar] [CrossRef]
- Liu, Y.J.; Gong, X.; Jing, W.; Lin, L.J.; Zhou, W.; He, J.N.H.; Li, J.H. Fast discrimination of avocado oil for different extracted methods using headspace-gas chromatography-ion mobility spectroscopy with PCA based on volatile organic compounds. Open Chem. 2021, 19, 367–376. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).