Antihyperglycemic, Antiaging, and L. brevis Growth-Promoting Activities of an Exopolysaccharide from Agrobacterium sp. FN01 (Galacan) Evaluated in a Zebrafish (Danio rerio) Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Exopolysaccharides from Agrobacterium sp. FN01
2.3. Total Sugar and Protein Content Determination
2.4. Fourier Transform Infrared Spectroscopy (FT-IR)
2.5. Monosaccharide Composition, Molecular Weight (Mw), and Element Determination
2.6. Periodate Oxidation, Smith Degradation, and NMR Analysis
2.6.1. Periodate Oxidation
2.6.2. Smith Degradation
2.6.3. NMR Analysis
2.7. Regulation of Galacan on the Growth of L. brevis in Intestinal Flora of Zebrafish
2.8. Antihyperglycemic Evaluation of Galacan in Zebrafish
2.9. Antiaging Effect of Galacan on Zebrafish
2.10. Statistical Analysis
3. Results and Discussion
3.1. Total Sugar and Protein Content of Galacan
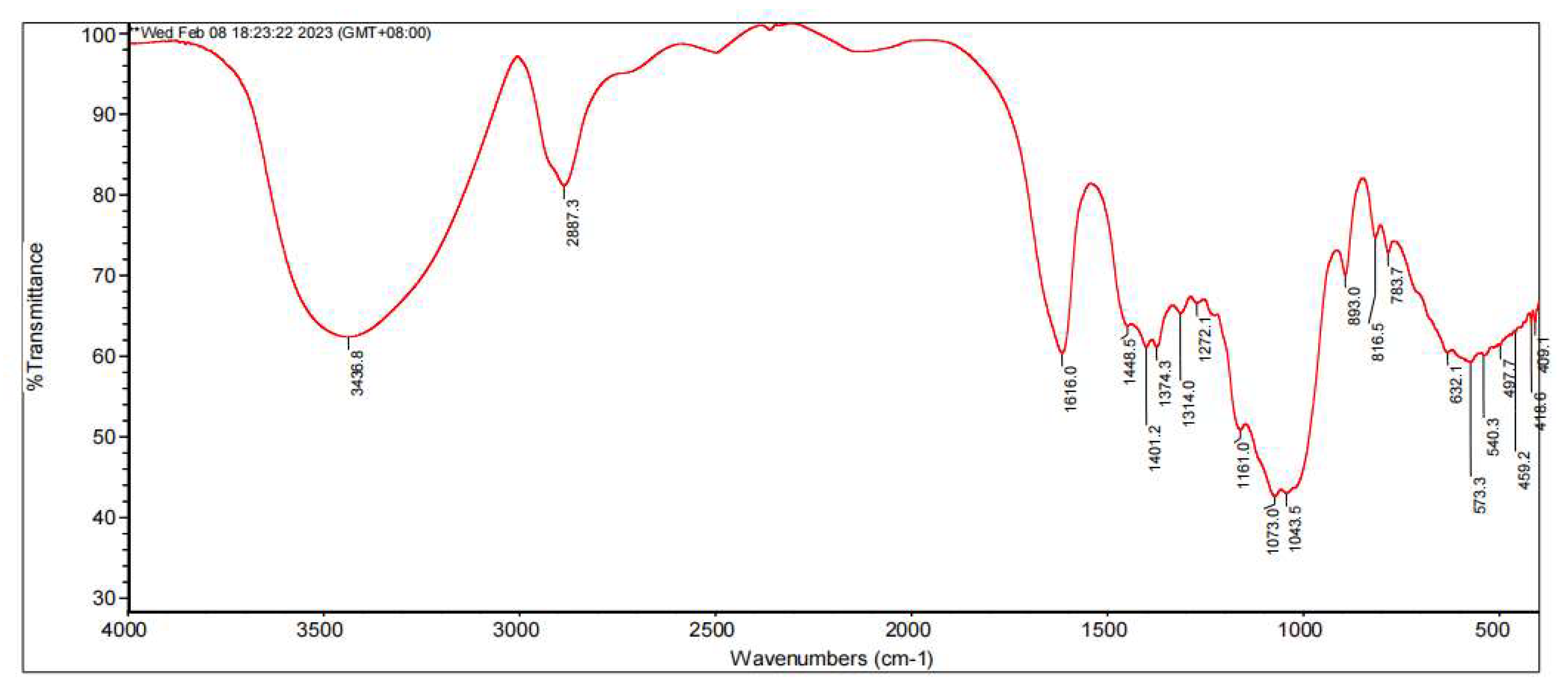
3.2. FT-IR Analysis
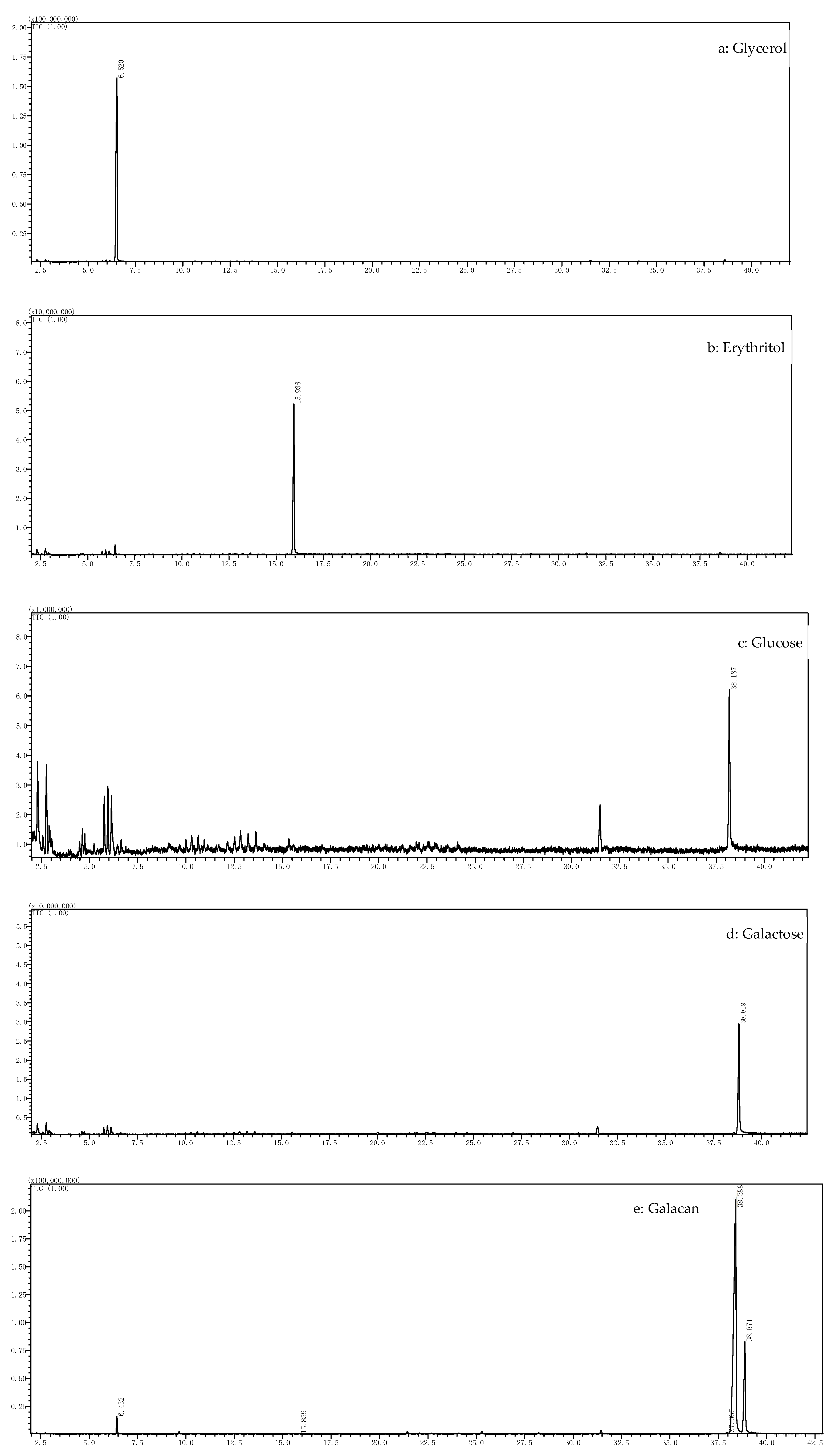
3.3. Results of Monosaccharide Composition, Mw, and Elemental Analysis of Galacan
3.4. Structural Characterization of Galacan
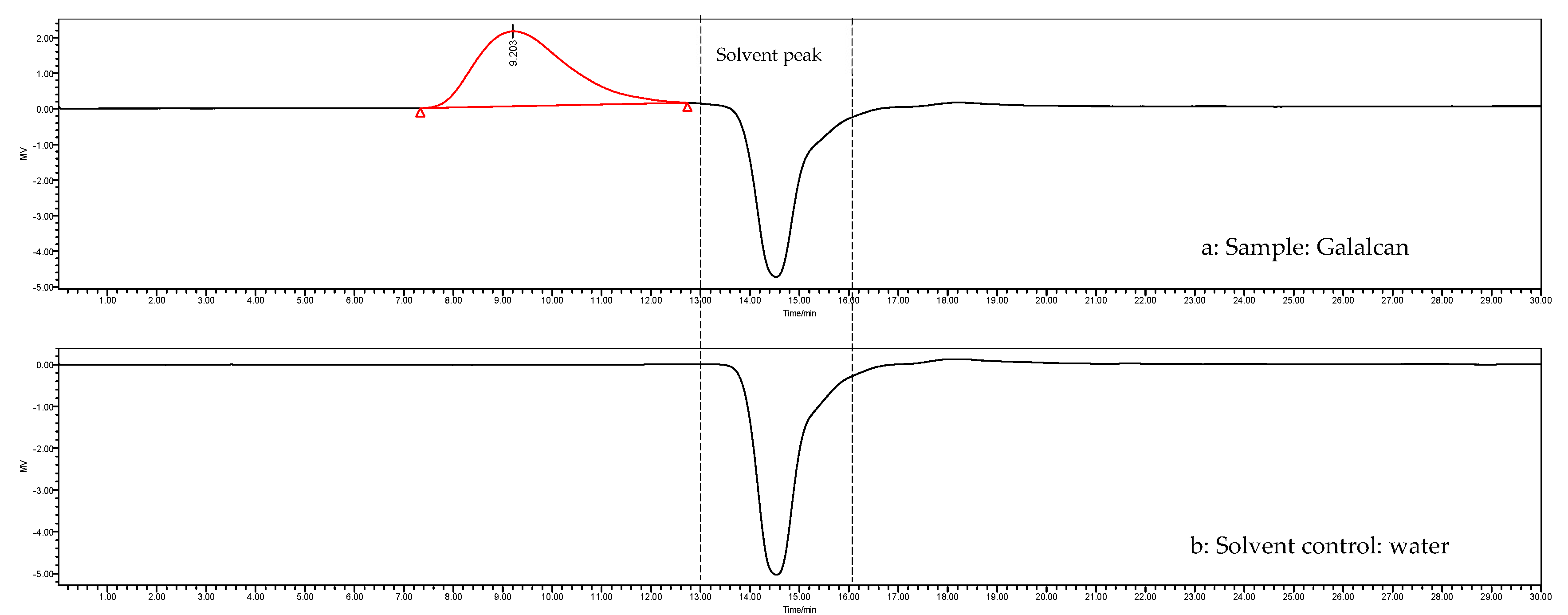
3.4.1. Analysis of Periodate Oxidation and Smith Degradation
3.4.2. NMR Results
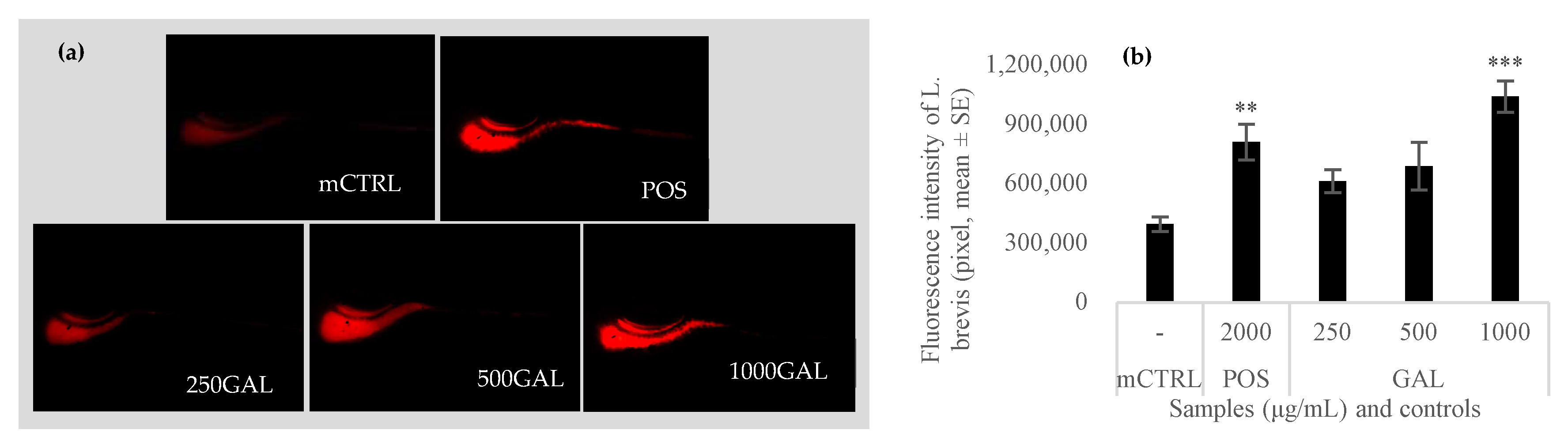
3.5. Effect of Galacan on the Growth of L. brevis
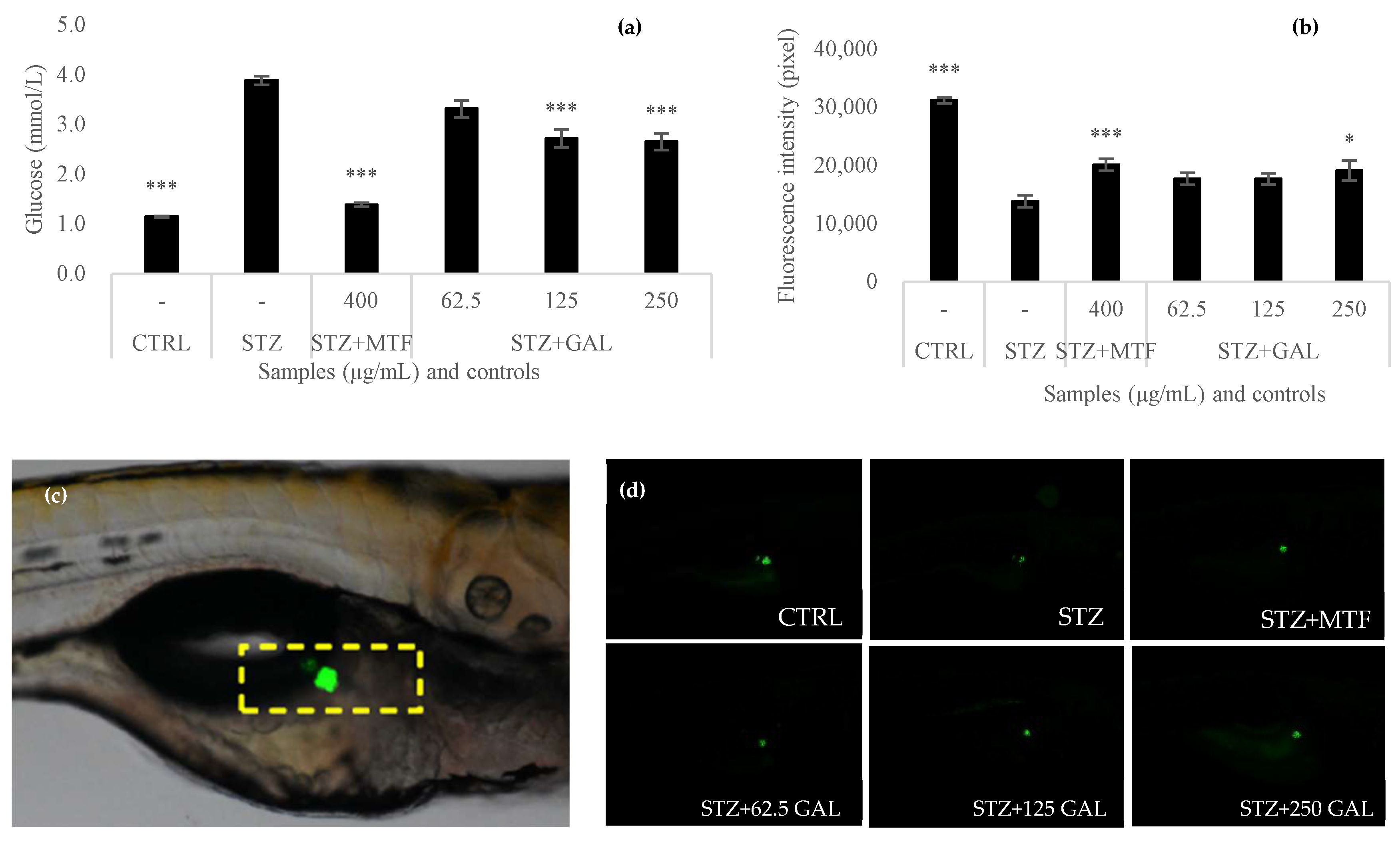
3.6. Antihyperglycemic Activity of Galacan
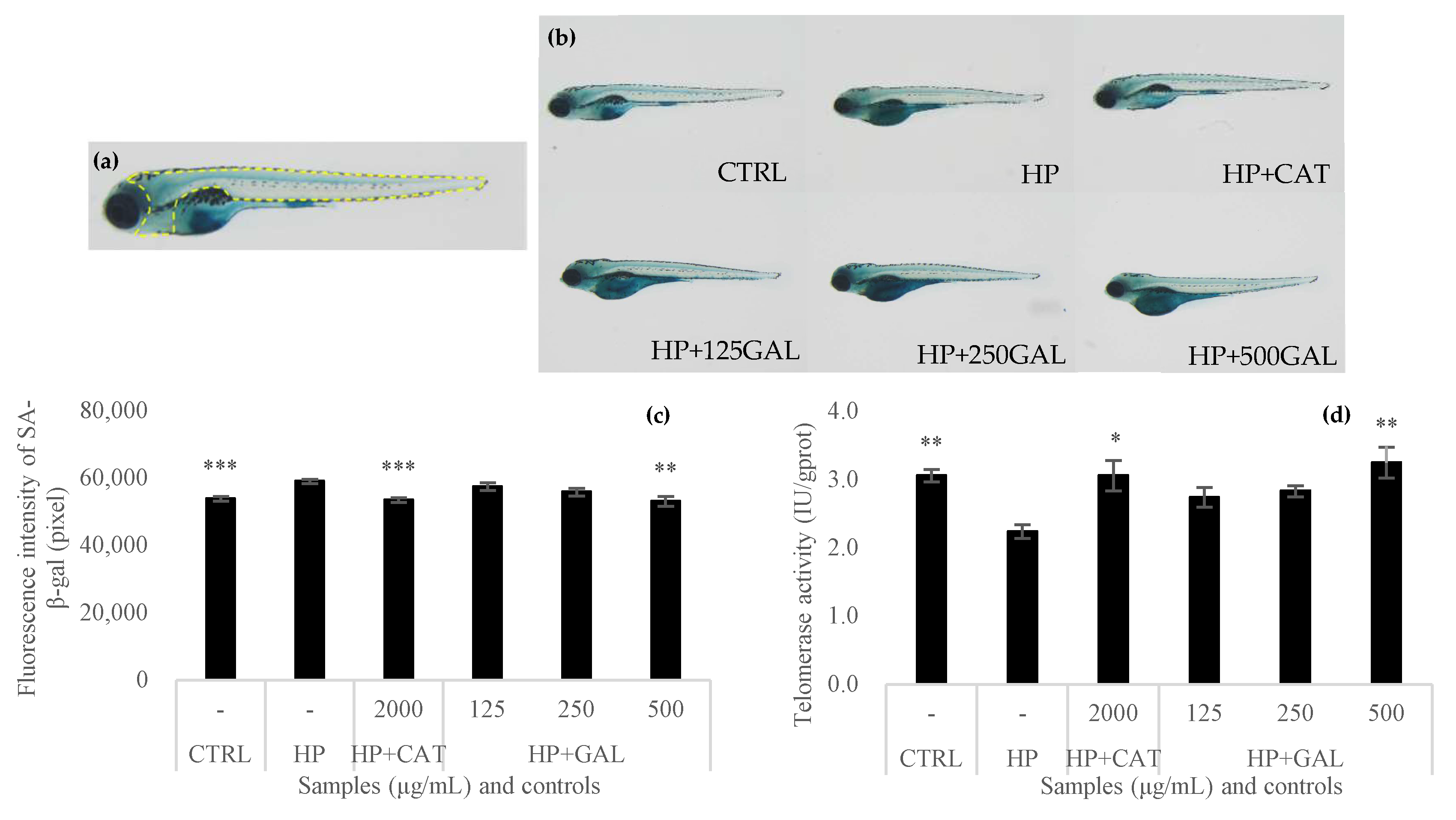
3.7. Antiaging Activity of Galacan
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Verma, D.K.; Niamah, A.K.; Patel, A.R.; Thakur, M.; Singh Sandhu, K.; Chávez-González, M.L.; Shah, N.; Noe Aguilar, C. Chemistry and microbial sources of curdlan with potential application and safety regulations as prebiotic in food and health. Food Res. Int. 2020, 133, 109136. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, I.W. Microbial Polysaccharide Products. Biotechnol. Genet. Eng. Rev. 1999, 16, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Liu, Z.; Zhao, Z.; Wang, Z.; Hu, X.; Jiang, Y.; Yan, J.; Li, Z.; Zheng, Z.; Zhan, X. Exopolysaccharide synthesis repressor genes (exoR and exoX) related to curdlan biosynthesis by Agrobacterium sp. Int. J. Biol. Macromol. 2022, 205, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Fu, G.; Sun, Y.; Zhang, D. Biosynthesis and applications of curdlan. Carbohydr. Polym. 2021, 273, 118597. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, Y.; Wang, Z.; Zhu, L.; Li, Z.; Jiang, Y.; Zhan, X.; Gao, M. Promoting substrates uptake and curdlan synthesis of Agrobacterium sp. by attenuating the exopolysaccharide encapsulation. Carbohydr. Polym. 2023, 315, 120941. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Gao, H.; Huang, Y.; Wu, S.; Tian, J.; Niu, Y.; Zou, C.; Jia, C.; Jin, M.; Huang, J.; et al. Glutamine synthetase gene glnA plays a vital role in curdlan biosynthesis of Agrobacterium sp. CGMCC 11546. Int. J. Biol. Macromol. 2020, 165, 222–230. [Google Scholar] [CrossRef]
- Gao, H.; Xie, F.; Zhang, W.; Tian, J.; Zou, C.; Jia, C.; Jin, M.; Huang, J.; Chang, Z.; Yang, X.; et al. Characterization and improvement of curdlan produced by a high-yield mutant of Agrobacterium sp. ATCC 31749 based on whole-genome analysis. Carbohydr. Polym. 2020, 245, 116486. [Google Scholar] [CrossRef]
- Gao, M.; Yang, G.; Li, F.; Wang, Z.; Hu, X.; Jiang, Y.; Yan, J.; Li, Z.; Zhan, X. Efficient endo-β-1,3-glucanase expression in Pichia pastoris for co-culture with Agrobacterium sp. for direct curdlan oligosaccharide production. Int. J. Biol. Macromol. 2021, 182, 1611–1617. [Google Scholar] [CrossRef]
- Han, X.; Ding, S.; Ma, Y.; Fang, J.; Jiang, H.; Li, Y.; Liu, G. Lactobacillus plantarum and Lactobacillus brevis Alleviate Intestinal Inflammation and Microbial Disorder Induced by ETEC in a Murine Model. Oxid. Med. Cell. Longev. 2021, 2021, 6867962. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, M.; Zhang, H.; Zhou, W.; Liu, W.; Pi, X.; Xing, J. An exopolysaccharide from Lactobacillus pentosus YY-112: Structure and effect on the human intestinal microbiota. Food Funct. 2023, 14, 7718–7726. [Google Scholar] [CrossRef]
- Nathan, N.N.; Philpott, D.J.; Girardin, S.E. The intestinal microbiota: From health to disease, and back. Microbes Infect. 2021, 23, 104849. [Google Scholar] [CrossRef] [PubMed]
- Association, A.D. Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2012, 36, S67–S74. [Google Scholar] [CrossRef]
- Darenskaya, M.A.; Kolesnikova, L.I.; Kolesnikov, S.I. Oxidative Stress: Pathogenetic Role in Diabetes Mellitus and Its Complications and Therapeutic Approaches to Correction. Bull. Exp. Biol. Med. 2021, 171, 179–189. [Google Scholar] [CrossRef]
- Brody, H. Diabetes. Nature 2012, 485, S1. [Google Scholar] [CrossRef] [PubMed]
- Toren, E.; Kepple, J.D.; Coutinho, K.V.; Poole, S.O.; Deeba, I.M.; Pierre, T.H.; Liu, Y.; Bethea, M.M.; Hunter, C.S. The SSBP3 co-regulator is required for glucose homeostasis, pancreatic islet architecture, and beta-cell identity. Mol. Metab. 2023, 76, 101785. [Google Scholar] [CrossRef] [PubMed]
- Turnheim, K. When drug therapy gets old: Pharmacokinetics and pharmacodynamics in the elderly. Exp. Gerontol. 2003, 38, 843–853. [Google Scholar] [CrossRef]
- Du, Y.; Gao, Y.; Zeng, B.; Fan, X.; Yang, D.; Yang, M. Effects of anti-aging interventions on intestinal microbiota. Gut Microbes 2021, 13, 1994835. [Google Scholar] [CrossRef]
- Donnini, S.; Giachetti, A.; Ziche, M. Assessing Vascular Senescence in Zebrafish. In Cell Senescence: Methods and Protocols; Galluzzi, L., Vitale, I., Kepp, O., Kroemer, G., Eds.; Humana Press: Totowa, NJ, USA, 2013; pp. 517–531. [Google Scholar] [CrossRef]
- Sun, C.; Chen, Y.; Li, X.; Tai, G.; Fan, Y.; Zhou, Y. Anti-hyperglycemic and anti-oxidative activities of ginseng polysaccharides in STZ-induced diabetic mice. Food Funct. 2014, 5, 845–848. [Google Scholar] [CrossRef]
- Hassan, I.; Gani, A.; Ahmad, M.; Banday, J. Extraction of polysaccharide from Althea rosea and its physicochemical, anti-diabetic, anti-hypertensive and antioxidant properties. Sci. Rep. 2022, 12, 17116. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, M.; Hong, R.; Chen, H. Preparation of a Momordica charantia L. polysaccharide-chromium (III) complex and its anti-hyperglycemic activity in mice with streptozotocin-induced diabetes. Int. J. Biol. Macromol. 2019, 122, 619–627. [Google Scholar] [CrossRef]
- Zhang, F.; Ren, T.; Gao, P.; Li, N.; Wu, Z.; Xia, J.; Jia, X.; Yuan, L.; Jiang, P. Characterization and anti-aging effects of polysaccharide from Gomphus clavatus Gray. Int. J. Biol. Macromol. 2023, 246, 125706. [Google Scholar] [CrossRef]
- Lee, K.; Kim, H.J.; Kim, S.A.; Park, S.-D.; Shim, J.-J.; Lee, J.-L. Exopolysaccharide from Lactobacillus plantarum HY7714 Protects against Skin Aging through Skin–Gut Axis Communication. Molecules 2021, 26, 1651. [Google Scholar] [CrossRef] [PubMed]
- Kavitake, D.; Veerabhadrappa, B.; Sudharshan, S.J.; Kandasamy, S.; Devi, P.B.; Dyavaiah, M.; Shetty, P.H. Oxidative stress alleviating potential of galactan exopolysaccharide from Weissella confusa KR780676 in yeast model system. Sci. Rep. 2022, 12, 1089. [Google Scholar] [CrossRef]
- Olsen, A.S. A Zebrafish Model of Diabetes Mellitus and Metabolic Memory Reveals Epigenetic Alterations. Ph.D. Thesis, Rosalind Franklin University of Medicine and Science, North Chicago, IL, USA, 2011. [Google Scholar]
- Kim, M.-K.; Ryu, K.-E.; Choi, W.-A.; Rhee, Y.-H.; Lee, I.-Y. Enhanced production of (1 → 3)-β-d-glucan by a mutant strain of Agrobacterium species. Biochem. Eng. J. 2003, 16, 163–168. [Google Scholar] [CrossRef]
- Trabelsi, I.; Slima, S.B.; Chaabane, H.; Riadh, B.S. Purification and characterization of a novel exopolysaccharides produced by Lactobacillus sp. Ca6. Int. J. Biol. Macromol. 2015, 74, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Peng, Q.; Zhang, Y.; Tian, D.; Zhang, P.; Huang, Y.; Ma, L.; Dia, V.P.; Qiao, Y.; Shi, B. Antibacterial potential of a novel Lactobacillus caseistrain isolated from Chinese northeast sauerkraut and the antibiofilm activity of its exopolysaccharides. Food Funct. 2020, 11, 4697–4706. [Google Scholar] [CrossRef]
- Chen, S.; Han, P.; Zhang, Q.; Liu, P.; Liu, J.; Zhao, L.; Guo, L.; Li, J. Lactobacillus brevis alleviates the progress of hepatocellular carcinoma and type 2 diabetes in mice model via interplay of gut microflora, bile acid and NOTCH 1 signaling. Front. Immunol. 2023, 14, 1179014. [Google Scholar] [CrossRef]
- Kumar, S.; Praneet, N.S.; Suchiang, K. Lactobacillus brevis MTCC 1750 enhances oxidative stress resistance and lifespan extension with improved physiological and functional capacity in Caenorhabditis elegans via the DAF-16 pathway. Free. Radic. Res. 2022, 56, 555–571. [Google Scholar] [CrossRef]
- Thambi, A.; Chakraborty, K. A novel anti-hyperglycemic sulfated pyruvylated polysaccharide from marine macroalga Hydropuntia edulis. Nat. Prod. Res. 2022, 37, 2987–2999. [Google Scholar] [CrossRef]
- Park, S.; Saravanakumar, K.; Sathiyaseelan, A.; Park, S.; Hu, X.; Wang, M.-H. Cellular antioxidant properties of nontoxic exopolysaccharide extracted from Lactobacillales (Weissella cibaria) isolated from Korean kimchi. LWT 2022, 154, 112727. [Google Scholar] [CrossRef]
- Saad, M.H.; Sidkey, N.M.; El-Fakharany, E.M. Characterization and optimization of exopolysaccharide extracted from a newly isolated halotolerant cyanobacterium, Acaryochloris Al-Azhar MNE ON864448.1 with antiviral activity. Microb. Cell Factories 2024, 23, 117. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ahmed, Z.; Feng, W.; Li, C.; Song, S. Physicochemical properties of exopolysaccharide produced by Lactobacillus kefiranofaciens ZW3 isolated from Tibet kefir. Int. J. Biol. Macromol. 2008, 43, 283–288. [Google Scholar] [CrossRef]
- Heperkan, Z.D.; Bolluk, M.; Bulbul, S. Structural analysis and properties of dextran produced by Weissella confusa and the effect of different cereals on its rheological characteristics. Int. J. Biol. Macromol. 2020, 143, 305–313. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.; Yang, S.; Lu, Z.; Li, G.; Liu, J.; Zhou, B.; Wu, D.; Wang, L. Isolation, Purification, Characterization, and Immunomodulatory Activity Analysis of alpha-Glucans from Spirulina platensis. ACS Omega 2021, 6, 21384–21394. [Google Scholar] [CrossRef]
- Haxaire, K.; Maréchal, Y.; Milas, M.; Rinaudo, M. Hydration of hyaluronan polysaccharide observed by IR spectrometry. II: Definition and quantitative analysis of elementary hydration specra and water uptake. Biopolymers 2003, 72, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Bremer, P.J.; Geesey, G.G. An evaluation of biofilm development utilizing non-destructive attenuated total reflectance Fourier transform infrared spectroscopy. Biofouling 1991, 3, 89–100. [Google Scholar] [CrossRef]
- Shan, J.; Sun, G.; Ren, J.; Zhu, T.; Jia, P.; Qu, W.; Li, Q.; Wu, J.; Ma, H.; Wen, S.; et al. An alpha-glucan isolated from root of Isatis Indigotica, its structure and adjuvant activity. Glycoconj. J. 2014, 31, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Ai, L.; Zhang, H.; Guo, B.; Chen, W.; Wu, Z.; Wu, Y. Preparation, partial characterization and bioactivity of exopolysaccharides from Lactobacillus casei LC2W. Carbohydr. Polym. 2008, 74, 353–357. [Google Scholar] [CrossRef]
- Behare, P.; Singh, R.; Kumar, M.; Prajapati, J.B.; Singh, R.P. Exopolysaccharides of Lactic Acid Bacteria—A review. J. Food Sci. Technol. 2009, 46, 1–11. [Google Scholar]
- De Vuyst, L.; Degeest, B. Heteropolysaccharides from lactic acid bacteria. FEMS Microbiol. Rev. 1999, 23, 153–177. [Google Scholar] [CrossRef] [PubMed]
- Şanlıbaba, P.; Çakmak, G. Exopolysaccharides Production by Lactic Acid Bacteria. Appl. Microbiol. 2016, 2. [Google Scholar] [CrossRef]
- Wang, Z.; Luo, D.; Liang, Z. Structure of polysaccharides from the fruiting body of Hericium erinaceus Pers. Carbohydr. Polym. 2004, 57, 241–247. [Google Scholar] [CrossRef]
- Asker, M.M.S.; El Sayed, O.H.; Mahmoud, M.G.; Ramadan, M.F. Chemical structure and antioxidant activity of a new exopolysaccharide produced from Micrococcus luteus. J. Genet. Eng. Biotechnol. 2014, 12, 121–126. [Google Scholar] [CrossRef]
- Li, H.; Mi, Y.; Duan, Z.; Ma, P.; Fan, D. Structural characterization and immunomodulatory activity of a polysaccharide from Eurotium cristatum. Int. J. Biol. Macromol. 2020, 162, 609–617. [Google Scholar] [CrossRef]
- Yan, M.; Mao, W.; Chen, C.; Kong, X.; Gu, Q.; Li, N.; Liu, X.; Wang, B.; Wang, S.; Xiao, B. Structural elucidation of the exopolysaccharide produced by the mangrove fungus Penicillium solitum. Carbohydr. Polym. 2014, 111, 485–491. [Google Scholar] [CrossRef]
- Ibrahem, M.D. Evolution of probiotics in aquatic world: Potential effects, the current status in Egypt and recent prospectives. J. Adv. Res. 2015, 6, 765–791. [Google Scholar] [CrossRef]
- Maldonado Galdeano, C.; Cazorla, S.I.; Lemme Dumit, J.M.; Velez, E.; Perdigon, G. Beneficial Effects of Probiotic Consumption on the Immune System. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Takauji, S.; Konishi, H.; Fujiya, M.; Ueno, N.; Tanaka, H.; Sato, H.; Isozaki, S.; Kashima, S.; Moriichi, K.; Mizukami, Y.; et al. Polyphosphate, Derived from Lactobacillus brevis, Modulates the Intestinal Microbiome and Attenuates Acute Pancreatitis. Dig. Dis. Sci. 2021, 66, 3872–3884. [Google Scholar] [CrossRef]
- Kochetkov, N.; Smorodinskaya, S.; Vatlin, A.; Nikiforov-Nikishin, D.; Nikiforov-Nikishin, A.; Danilenko, V.; Anastasia, K.; Reznikova, D.; Grishina, Y.; Antipov, S.; et al. Ability of Lactobacillus brevis 47f to Alleviate the Toxic Effects of Imidacloprid Low Concentration on the Histological Parameters and Cytokine Profile of Zebrafish (Danio rerio). Int. J. Mol. Sci. 2023, 24, 12290. [Google Scholar] [CrossRef]
- Eames, S.C.; Philipson, L.H.; Prince, V.E.; Kinkel, M.D. Blood sugar measurement in zebrafish reveals dynamics of glucose homeostasis. Zebrafish 2010, 7, 205–213. [Google Scholar] [CrossRef]
- Kishi, S.; Uchiyama, J.; Baughman, A.M.; Goto, T.; Lin, M.C.; Tsai, S.B. The zebrafish as a vertebrate model of functional aging and very gradual senescence. Exp. Gerontol. 2003, 38, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, A.G.; Ouellette, M.; Frolkis, M.; Holt, S.E.; Chiu, C.-P.; Morin, G.B.; Harley, C.B.; Shay, J.W.; Lichtsteiner, S.; Wright, W.E. Extension of Life-Span by Introduction of Telomerase into Normal Human Cells. Science 1998, 279, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Yang, D.; Zhang, W.; Lu, Y.; Zhang, M.; Wang, L.; Li, X.; Zhou, L.; Wu, Q.; Pan, W.; et al. Antioxidant and anti-aging activities of the polysaccharide TLH-3 from Tricholoma lobayense. Int. J. Biol. Macromol. 2016, 85, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, J.; Zhang, J.; Wang, X.; Hao, L.; Jia, L. Purification, in vitro antioxidant and in vivo anti-aging activities of exopolysaccharides by Agrocybe cylindracea. Int. J. Biol. Macromol. 2017, 102, 351–357. [Google Scholar] [CrossRef] [PubMed]


| Time (h) | 0 | 24 | 48 | 72 | 96 | 120 | 144 | 168 | 180 | 192 |
|---|---|---|---|---|---|---|---|---|---|---|
| OD223 | 0.567 | 0.543 | 0.533 | 0.526 | 0.520 | 0.510 | 0.506 | 0.496 | 0.490 | 0.490 |
| Sample | Weight (mg) | NaIO4 Consumption (mmol) | Formic Acid Production (mmol) | NaIO4 Consumption/ Formic Acid Production | NaIO4 Consumption/ Hexose Value |
|---|---|---|---|---|---|
| Galacan | 24.7 | 0.0582 | 0.0215 | 2.7068 | 0.3819 |
| Glycosidic Linkages | Number of 13C | 13C δ(ppm) | HSQC (13C × 1H) | COSY (1H × 1H) | HMBC (13C→1H) |
|---|---|---|---|---|---|
| a: β-D-Galp(1→ | 1 | 102.66 | 4.38 | 4.38 (H1) | 3.32 (c: H3) |
| 2 | 72.85 | 3.36 | 3.36 (H2) | ||
| 3 | 3.58 (H3) | ||||
| 4 | ND | ||||
| 5 | (-) | ||||
| 6 | (-) | ||||
| b: →3)β-D-Galp(1→ | 1 | 102.35 | 4.39 | 4.39 (H1) | 3.39 (d: H2) |
| 2 | 73.23 | 3.19 | 3.19 (H2) | ||
| 3 | 68.00 | 3.35 | 3.35 (H3) | ||
| 4 | ND | ||||
| 5 | (-) | ||||
| 6 | (-) | ||||
| c: →2,3)β-D-Glcp(1→ | 1 | 102.53 | 4.58 | 4.58 (H1) | 3.35 (b: H3) |
| 2 | 73.38 | 3.21 | 3.21 (H2) | 3.35 (b: H3) | |
| 3 | 75.50 | 3.32 | 3.32 (H3) | 3.35 (b: H3) | |
| 4 | ND | ||||
| 5 | (-) | ||||
| 6 | (-) | ||||
| d: β-D-Glcp(1→ | 1 | 101.37 | 4.60 | 4.60 (H1) | |
| 2 | 72.92 | 3.39 | 3.39 (H2) (+) | ||
| 3 | 3.61 (H3) (+) | ||||
| 4 | ND | ||||
| 5 | (-) | ||||
| 6 | (-) | ||||
| e: →2)β-D-Glcp(1→ | 1 | 102.77 | 4.64 | 4.64 (H1) | 3.21 (c: H2) 3.35 (b: H3) |
| 2 | 69.50 | 3.24 | 3.24 (H2) | 3.35 (b: H3) | |
| 3 | ND | ||||
| 4 | ND | ||||
| 5 | + | (-) | |||
| 6 | (-) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.; Du, L.; Wang, M.; Zhang, R.; Shan, J.; Qiao, Y.; Peng, Q.; Shi, B. Antihyperglycemic, Antiaging, and L. brevis Growth-Promoting Activities of an Exopolysaccharide from Agrobacterium sp. FN01 (Galacan) Evaluated in a Zebrafish (Danio rerio) Model. Foods 2024, 13, 2729. https://doi.org/10.3390/foods13172729
Xu X, Du L, Wang M, Zhang R, Shan J, Qiao Y, Peng Q, Shi B. Antihyperglycemic, Antiaging, and L. brevis Growth-Promoting Activities of an Exopolysaccharide from Agrobacterium sp. FN01 (Galacan) Evaluated in a Zebrafish (Danio rerio) Model. Foods. 2024; 13(17):2729. https://doi.org/10.3390/foods13172729
Chicago/Turabian StyleXu, Xiaoqing, Lingling Du, Meng Wang, Ran Zhang, Junjie Shan, Yu Qiao, Qing Peng, and Bo Shi. 2024. "Antihyperglycemic, Antiaging, and L. brevis Growth-Promoting Activities of an Exopolysaccharide from Agrobacterium sp. FN01 (Galacan) Evaluated in a Zebrafish (Danio rerio) Model" Foods 13, no. 17: 2729. https://doi.org/10.3390/foods13172729






