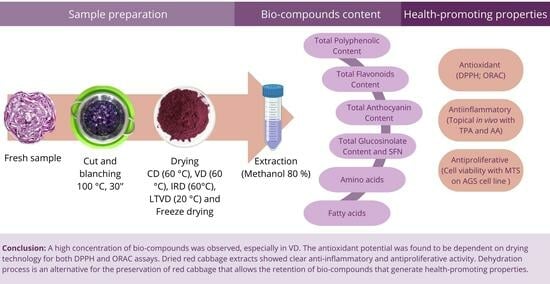
Health-Promoting Properties of Processed Red Cabbage (Brassica oleracea var. capitata f. rubra): Effects of Drying Methods on Bio-Compound Retention
Abstract
:1. Introduction
2. Materials and Methods
2.1. Solvents and Reagents
2.2. Raw Material and Drying Methods
2.2.1. Raw Material
2.2.2. Drying Procedure
2.3. Proximate Composition
2.4. Assessment of Bio-Compounds
2.4.1. Extraction Methods
2.4.2. Determination of Total Phenolics, Total Flavonoids and Total Anthocyanin Content
2.4.3. Determination of Total Glucosinolates
2.4.4. Determination of Phenolic Acids Profile
2.4.5. Determination of Amino Acid Content and Fatty Acids Profile
2.4.6. Determination of Sulforaphane (SFN) Content
2.5. Health-Promoting Properties
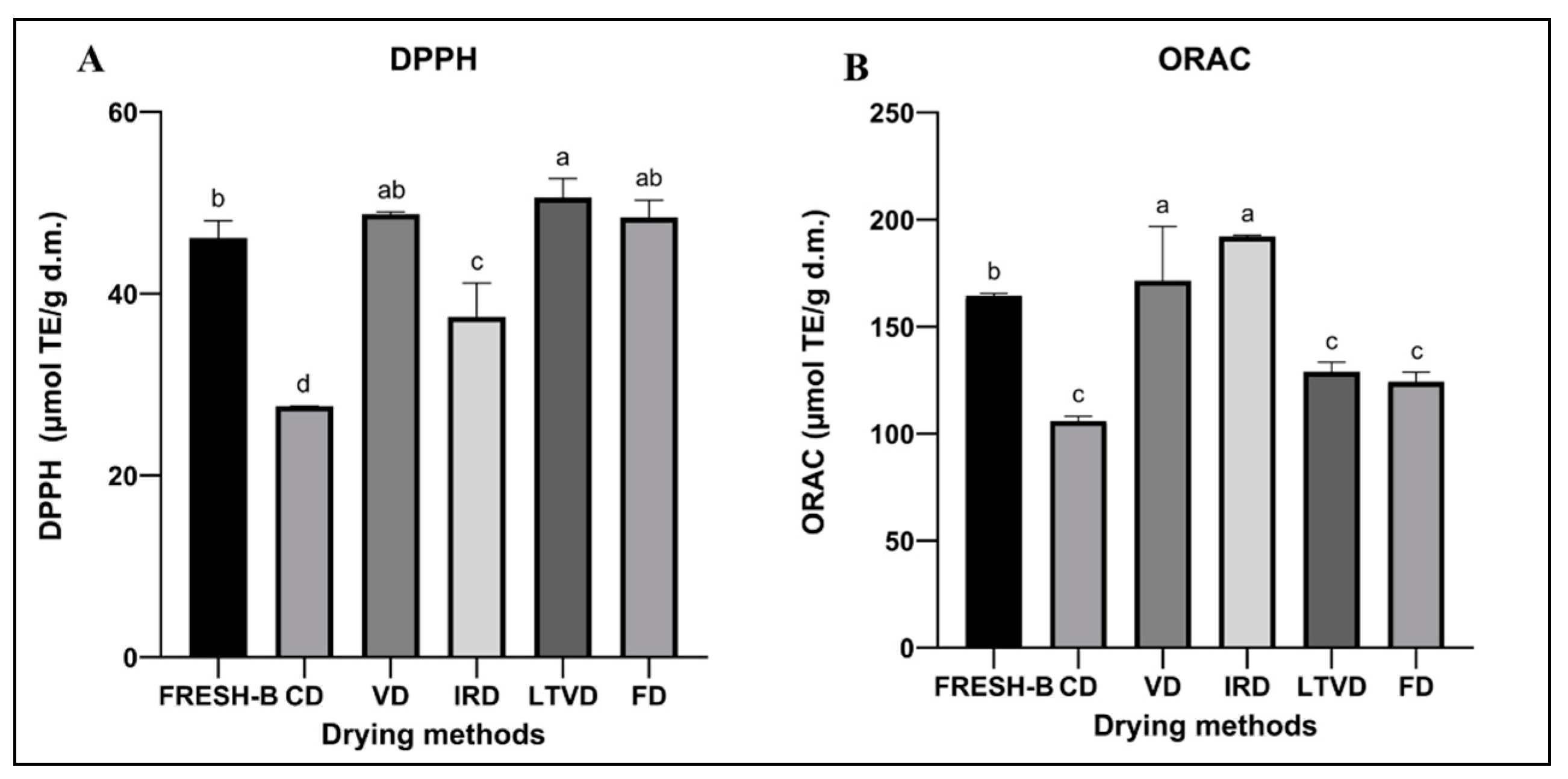
2.5.1. Antioxidant Potential by DPPH and ORAC Assays
2.5.2. Anti-Inflammatory Activity
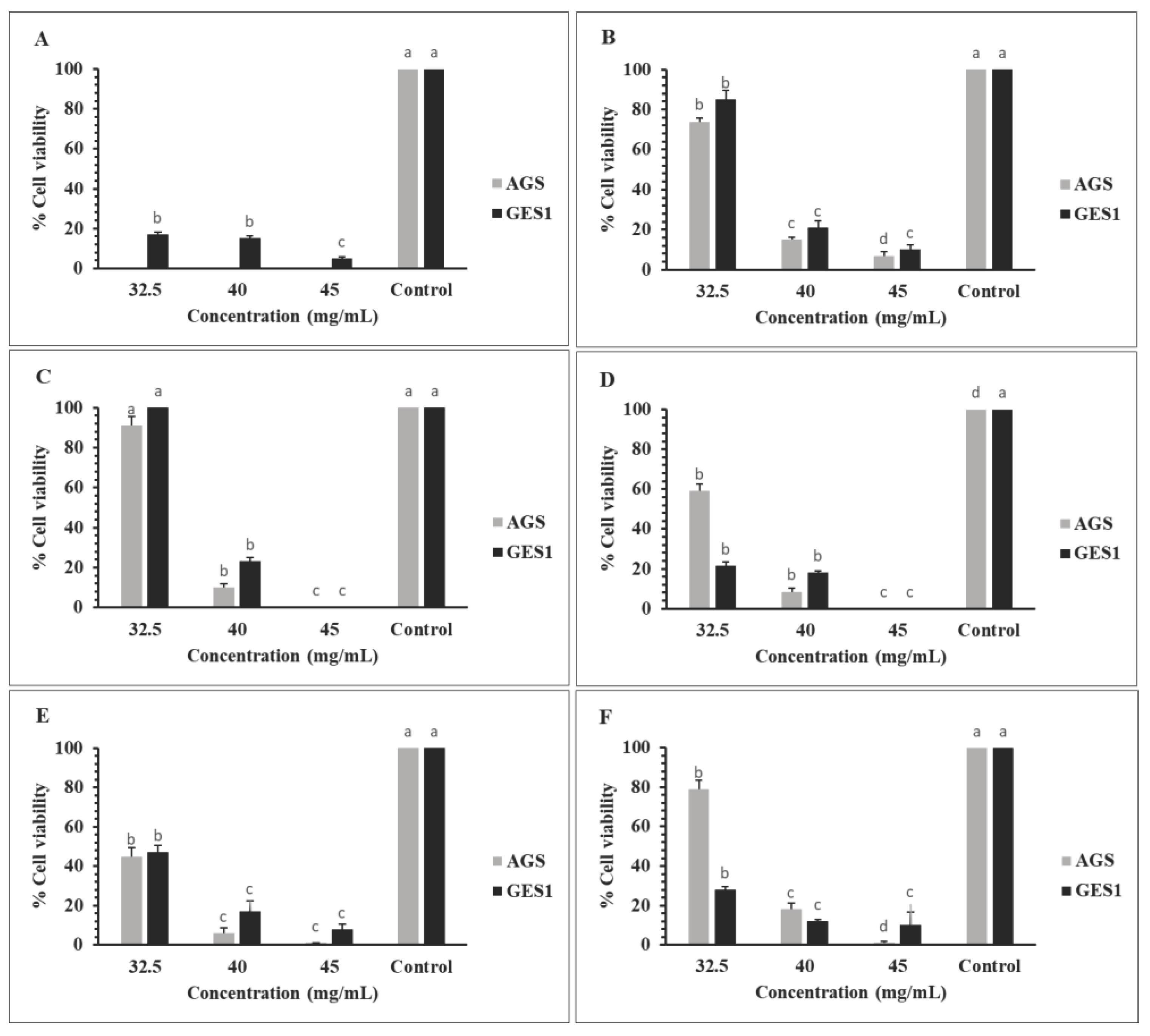
2.5.3. Antiproliferative Assay
2.6. Statistical Analysis
3. Results and Discussions
3.1. Proximate Composition
3.2. Determination of Total Phenolic, Total Flavonoid and Total Anthocyanin Content
3.3. Determination of Total Glucosinolate Content (TGC)
3.4. Determination of Phenolic Acid Profile
3.5. Determination of Sulforaphane Content
3.6. Determination of Amino Acid and Fatty Acid Profile
3.7. Health-Promoting Properties
3.7.1. Antioxidant Potential by DPPH and ORAC Assays
3.7.2. Anti-Inflammatory Activity
3.7.3. Antiproliferative Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garg, M.; Kaur, S.; Sharma, A.; Kumari, A.; Tiwari, V.; Sharma, S.; Kapoor, P.; Sheoran, B.; Goyal, A.; Krishania, M. Rising Demand for Healthy Foods-Anthocyanin Biofortified Colored Wheat Is a New Research Trend. Front. Nutr. 2022, 9, 878221. [Google Scholar] [CrossRef]
- Arumugam, T.; Sona, L.; Maheswari, M. Fruits and Vegetables as Superfoods: Scope and Demand. Pharma Innov. J. 2021, 10, 119–129. [Google Scholar]
- Arias, A.; Feijoo, G.; Moreira, M.T. Exploring the Potential of Antioxidants from Fruits and Vegetables and Strategies for Their Recovery. Innov. Food Sci. Emerg. Technol. 2022, 77, 102974. [Google Scholar] [CrossRef]
- Rakha, A.; Umar, N.; Rabail, R.; Butt, M.S.; Kieliszek, M.; Hassoun, A.; Aadil, R.M. Anti-Inflammatory and Anti-Allergic Potential of Dietary Flavonoids: A Review. Biomed. Pharmacother. 2022, 156, 113945. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Pu, Y.; Xu, Y.; He, X.; Cao, J.; Ma, Y.; Jiang, W. Anti-Diabetic and Anti-Obesity: Efficacy Evaluation and Exploitation of Polyphenols in Fruits and Vegetables. Food Res. Int. 2022, 157, 111202. [Google Scholar] [CrossRef] [PubMed]
- Alzate-Yepes, T.; Pérez-Palacio, L.; Martínez, E.; Osorio, M. Mechanisms of Action of Fruit and Vegetable Phytochemicals in Colorectal Cancer Prevention. Molecules 2023, 28, 4322. [Google Scholar] [CrossRef] [PubMed]
- Vega-Galvez, A.; Uribe, E.; Pasten, A.; Camus, J.; Gomez-Perez, L.S.; Mejias, N.; Vidal, R.L.; Grunenwald, F.; Aguilera, L.E.; Valenzuela-Barra, G. Comprehensive Evaluation of the Bioactive Composition and Neuroprotective and Antimicrobial Properties of Vacuum-Dried Broccoli (Brassica oleracea Var. Italica) Powder and Its Antioxidants. Molecules 2023, 28, 766. [Google Scholar] [CrossRef] [PubMed]
- Tapia-Hernández, J.A.; Rodríguez-Felix, F.; Juárez-Onofre, J.E.; Ruiz-Cruz, S.; Robles-García, M.A.; Borboa-Flores, J.; Wong-Corral, F.J.; Cinco-Moroyoqui, F.J.; Castro-Enríquez, D.D.; Del-Toro-Sánchez, C.L. Zein-Polysaccharide Nanoparticles as Matrices for Antioxidant Compounds: A Strategy for Prevention of Chronic Degenerative Diseases. Food Res. Int. 2018, 111, 451–471. [Google Scholar] [CrossRef]
- Ghareaghajlou, N.; Hallaj-Nezhadi, S.; Ghasempour, Z. Red Cabbage Anthocyanins: Stability, Extraction, Biological Activities and Applications in Food Systems. Food Chem. 2021, 365, 130482. [Google Scholar] [CrossRef]
- Rani, S.; Singh, A.; Kumar, P. Evaluation of Nutritional Facts and Health Benefits of Red Cabbage (Brassica). J. Rural Agric. Res. 2021, 21, 37–39. [Google Scholar]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Wang, Q. Assessment of Vitamin and Carotenoid Concentrations of Emerging Food Products: Edible Microgreens. J. Agric. Food Chem. 2012, 60, 7644–7651. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Lan, X.; Chen, S.; Zhong, X.; Li, W. Physical Character, Total Polyphenols, Anthocyanin Profile and Antioxidant Activity of Red Cabbage as Affected by Five Processing Methods. Food Res. Int. 2023, 169, 112929. [Google Scholar] [CrossRef] [PubMed]
- Drozdowska, M.; Leszczyńska, T.; Koronowicz, A.; Piasna-Słupecka, E.; Domagała, D.; Kusznierewicz, B. Young Shoots of Red Cabbage Are a Better Source of Selected Nutrients and Glucosinolates in Comparison to the Vegetable at Full Maturity. Eur. Food Res. Technol. 2020, 246, 2505–2515. [Google Scholar] [CrossRef]
- Podsȩdek, A.; Majewska, I.; Kucharska, A.Z. Inhibitory Potential of Red Cabbage against Digestive Enzymes Linked to Obesity and Type 2 Diabetes. J. Agric. Food Chem. 2017, 65, 7192–7199. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Jonville, M.C.; Schneider, Y.J.; Hoffmann, L.; Bohn, T. Carotenoids, Polyphenols and Micronutrient Profiles of Brassica Oleraceae and Plum Varieties and Their Contribution to Measures of Total Antioxidant Capacity. Food Chem. 2014, 155, 240–250. [Google Scholar] [CrossRef]
- Koss-Mikołajczyk, I.; Kusznierewicz, B.; Wiczkowski, W.; Płatosz, N.; Bartoszek, A. Phytochemical Composition and Biological Activities of Differently Pigmented Cabbage (Brassica oleracea Var. Capitata) and Cauliflower (Brassica oleracea Var. Botrytis) Varieties. J. Sci. Food Agric. 2019, 99, 5499–5507. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Chen, J.; Yu, D.; Chen, S.; Ye, X.; Zhang, Z. Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by Lc-Ms/Ms in Multiple Reaction Monitoring Mode. Molecules 2021, 26, 5171. [Google Scholar] [CrossRef]
- Hanschen, F.S. Domestic Boiling and Salad Preparation Habits Affect Glucosinolate Degradation in Red Cabbage (Brassica oleracea Var. Capitata f. Rubra). Food Chem. 2020, 321, 126694. [Google Scholar] [CrossRef]
- Guan, Y.; Ji, Y.; Yang, X.; Pang, L.; Cheng, J.; Lu, X.; Zheng, J.; Yin, L.; Hu, W. Antioxidant Activity and Microbial Safety of Fresh-Cut Red Cabbage Stored in Different Packaging Films. LWT 2023, 175, 114478. [Google Scholar] [CrossRef]
- Tabart, J.; Pincemail, J.; Kevers, C.; Defraigne, J.O.; Dommes, J. Processing Effects on Antioxidant, Glucosinolate, and Sulforaphane Contents in Broccoli and Red Cabbage. Eur. Food Res. Technol. 2018, 244, 2085–2094. [Google Scholar] [CrossRef]
- Tajalli, F.; Saeedi, M.; Vafaei Malekabadi, A. Anticancer and Antioxidant Effects of Red Cabbage on Three Cancerous Cell Lines and Comparison with a Normal Cell Line (HFF-3). J. Genes Cells 2020, 6, 12. [Google Scholar] [CrossRef]
- Waghulde, S.; Khan, N.A.; Gorde, N.; Kale, M.; Naik, P.; Yewale, R.P. Comparative Antimicrobial Activity Study of Brassica Oleceracea. Proceedings 2019, 9, 64. [Google Scholar] [CrossRef]
- Cicio, A.; Serio, R.; Zizzo, M.G. Anti-Inflammatory Potential of Brassicaceae-Derived Phytochemicals: In Vitro and In Vivo Evidence for a Putative Role in the Prevention and Treatment of IBD. Nutrients 2023, 15, 31. [Google Scholar] [CrossRef]
- Zafar, I.; Hussain, A.I.; Fatima, T.; Alnasser, S.M.A.; Ahmad, A. Inter-Varietal Variation in Phenolic Profile, Sugar Contents, Antioxidant, Anti-Proliferative and Antibacterial Activities of Selected Brassica Species. Appl. Sci. 2022, 12, 5811. [Google Scholar] [CrossRef]
- Huang, H.; Jiang, X.; Xiao, Z.; Yu, L.; Pham, Q.; Sun, J.; Chen, P.; Yokoyama, W.; Yu, L.L.; Luo, Y.S.; et al. Red Cabbage Microgreens Lower Circulating Low-Density Lipoprotein (LDL), Liver Cholesterol, and Inflammatory Cytokines in Mice Fed a High-Fat Diet. J. Agric. Food Chem. 2016, 64, 9161–9171. [Google Scholar] [CrossRef] [PubMed]
- Calín-Sánchez, Á.; Lipan, L.; Cano-Lamadridm, M.; Kharaghani, A.; Masztalerz, K.; Carbonell-Barrachina, Á.; Fiegiel, A. Comparison of Traditional and Novel Drying Techniques and Its Effect on Quality of Fruits, Vegetables and Aromatic Herbs. Foods 2020, 9, 1261. [Google Scholar] [CrossRef] [PubMed]
- Inyang, U.; Oboh, I.; Etuk, B.R. Drying and the Different Technique. Int. J. Food Nutr. Saf. J. 2017, 8, 45–72. [Google Scholar]
- Vega-Gálvez, A.; Uribe, E.; Pastén, A.; Vega, M.; Poblete, J.; Bilbao-Sainz, C.; Chiou, B.S. Low-Temperature Vacuum Drying as Novel Process to Improve Papaya (Vasconcellea pubescens) Nutritional-Functional Properties. Future Foods 2022, 5, 100117. [Google Scholar] [CrossRef]
- Sakare, P.; Prasad, N.; Thombare, N.; Singh, R.; Sharma, S.C. Infrared Drying of Food Materials: Recent Advances. Food Eng. Rev. 2020, 12, 381–398. [Google Scholar] [CrossRef]
- Huang, D.; Yang, P.; Tang, X.; Luo, L.; Sunden, B. Application of Infrared Radiation in the Drying of Food Products. Trends Food Sci. Technol. 2021, 110, 765–777. [Google Scholar] [CrossRef]
- Managa, M.G.; Sultanbawa, Y.; Sivakumar, D. Effects of Different Drying Methods on Untargeted Phenolic Metabolites, and Antioxidant Activity in Chinese Cabbage (Brassica rapa L. Subsp. Chinensis) and Nightshade (Solanum retroflexum Dun.). Molecules 2020, 25, 1326. [Google Scholar] [CrossRef]
- Yue, T.; Xing, Y.; Xu, Q.; Yang, S.; Xu, L.; Wang, X.; Yang, P. Physical and Chemical Properties of Purple Cabbage as Affected by Drying Conditions. Int. J. Food Prop. 2021, 24, 997–1010. [Google Scholar] [CrossRef]
- Adelanwa, E.B.; Medugu, J.M. Variation in the Nutrient Composition of Red and Green Cabbage (Brassica oleracea) with Respect to Age at Harvest. J. Appl. Agric. Res. 2015, 7, 183–189. [Google Scholar]
- Liu, J.; Li, X.; Yang, Y.; Wei, H.; Xue, L.; Zhao, M.; Cai, J. Optimization of Combined Microwave and Hot Air Drying Technology for Purple Cabbage by Response Surface Methodology (RSM). Food Sci. Nutr. 2021, 9, 4568–4577. [Google Scholar] [CrossRef] [PubMed]
- Weligama Thuppahige, V.T.; Welsh, Z.G.; Joardder, M.; Karim, A. Recent Advances in Determining the Cellular-Level Property Evolutions of Plant-Based Food Materials during Drying. Trends Food Sci. Technol. 2024, 143, 104291. [Google Scholar] [CrossRef]
- Tao, Y.; Han, M.; Gao, X.; Han, Y.; Show, P.L.; Liu, C.; Ye, X.; Xie, G. Applications of Water Blanching, Surface Contacting Ultrasound-Assisted Air Drying, and Their Combination for Dehydration of White Cabbage: Drying Mechanism, Bioactive Profile, Color and Rehydration Property. Ultrason. Sonochem. 2019, 53, 192–201. [Google Scholar] [CrossRef] [PubMed]
- AOAC (Association of Official Analytical Chemists). Official Method of Analysis; Association of Official Analytical Chemists: Washington, DC, USA, 1990; Volume 1. [Google Scholar]
- Aghajanzadeh, T.; Hawkesford, M.J.; De Kok, L.J. The Significance of Glucosinolates for Sulfur Storage in Brassicaceae Seedlings. Front. Plant Sci. 2014, 5, 704. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Oh, I.N.; Kim, J.; Jung, D.; Cuong, N.P.; Kim, Y.; Lee, J.; Kwon, O.; Park, S.U.; Lim, Y.; et al. Phenolic Compound Profiles and Their Seasonal Variations in New Red-Phenotype Head-Forming Chinese Cabbages. LWT 2018, 90, 433–439. [Google Scholar] [CrossRef]
- Uribe, E.; Gómez-Pérez, L.S.; Pasten, A.; Pardo, C.; Puente, L.; Vega-Galvez, A. Assessment of Refractive Window Drying of Physalis (Physalis peruviana L.) Puree at Different Temperatures: Drying Kinetic Prediction and Retention of Bioactive Components. J. Food Meas. Charact. 2022, 16, 2605–2615. [Google Scholar] [CrossRef]
- Dini, I.; Tenore, G.C.; Dini, A. Antioxidant Compound Contents and Antioxidant Activity before and after Cooking in Sweet and Bitter Chenopodium Quinoa Seeds. LWT—Food Sci. Technol. 2010, 43, 447–451. [Google Scholar] [CrossRef]
- De Souza, V.R.; Pereira, P.A.P.; Da Silva, T.L.T.; De Oliveira Lima, L.C.; Pio, R.; Queiroz, F. Determination of the Bioactive Compounds, Antioxidant Activity and Chemical Composition of Brazilian Blackberry, Red Raspberry, Strawberry, Blueberry and Sweet Cherry Fruits. Food Chem. 2014, 156, 362–368. [Google Scholar] [CrossRef]
- López, J.; Vega-Gálvez, A.; Bilbao-Sainz, C.; Uribe, E.; Chiou, B.-S.; Quispe-Puentes, I. Influence of Vacuum Drying Temperature on: Physico-Chemical Composition and Antioxidant Properties of Murta Berries. J. Food Process Eng. 2017, 40, e12569. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane, G. A Simple Method for the Isolation and Purificationof Total Lipides from Animal Tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Araya, M.; García, S.; Rengel, J.; Pizarro, S.; Álvarez, G. Determination of Free and Protein Amino Acid Content in Microalgae by HPLC-DAD with Pre-Column Derivatization and Pressure Hydrolysis. Mar. Chem. 2021, 234, 103999. [Google Scholar] [CrossRef]
- Grajeda-Iglesias, C.; Salas, E.; Barouh, N.; Baréa, B.; Panya, A.; Figueroa-Espinoza, M.C. Antioxidant Activity of Protocatechuates Evaluated by DPPH, ORAC, and CAT Methods. Food Chem. 2016, 194, 749–757. [Google Scholar] [CrossRef]
- Uribe, E.; Lemus-Mondaca, R.; Vega-Gálvez, A.; Zamorano, M.; Quispe-Fuentes, I.; Pasten, A.; Di Scala, K. Influence of Process Temperature on Drying Kinetics, Physicochemical Properties and Antioxidant Capacity of the Olive-Waste Cake. Food Chem. 2014, 147, 170–176. [Google Scholar] [CrossRef]
- Valenzuela-Barra, G.; Castro, C.; Figueroa, C.; Barriga, A.; Silva, X.; De Las Heras, B.; Hortelano, S.; Delporte, C. Anti-Inflammatory Activity and Phenolic Profile of Propolis from Two Locations in Región Metropolitana de Santiago, Chile. J. Ethnopharmacol. 2015, 168, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Macuer-Guzmán, J.; Bernal, G.; Jamett-Díaz, F.; Ramírez-Rivera, S.; Ibáñez, C. Selective and Apoptotic Action of Ethanol Extract of Annona Cherimola Seeds against Human Stomach Gastric Adenocarcinoma Cell Line AGS. Plant Foods Hum. Nutr. 2019, 74, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Tapia, M.S.; Alzamora, S.M.; Chirife, J. Effects of Water Activity (Aw) on Microbial Stability as a Hurdle in Food Preservation. In Water Activity in Foods:Fundamentals and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2020; pp. 323–355. [Google Scholar]
- Kumar, Y.; Singh, L.; Sharanagat, V.S.; Mani, S.; Kumar, S.; Kumar, A. Quality Attributes of Convective Hot Air Dried Spine Gourd (Momordica dioica Roxb. Ex Willd) Slices. Food Chem. 2021, 347, 129041. [Google Scholar] [CrossRef] [PubMed]
- Guldhe, A.; Singh, B.; Rawat, I.; Ramluckan, K.; Bux, F. Efficacy of Drying and Cell Disruption Techniques on Lipid Recovery from Microalgae for Biodiesel Production. Fuel 2014, 128, 46–52. [Google Scholar] [CrossRef]
- Zainuddin, M.F.; Fai, C.K.; Ariff, A.B.; Rios-Solis, L.; Halim, M. Current Pretreatment/Cell Disruption and Extraction Methods Used to Improve Intracellular Lipid Recovery from Oleaginous Yeasts. Microorganisms 2021, 9, 251. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Lagunas, L.; Rodríguez-Ramírez, J.; Cruz-Gracida, M.; Sandoval-Torres, S.; Barriada-Bernal, G. Convective Drying Kinetics of Strawberry (Fragaria ananassa): Effects on Antioxidant Activity, Anthocyanins and Total Phenolic Content. Food Chem. 2017, 230, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, B.R.; Heleno, S.A.; Oliveira, M.B.P.P.; Barros, L.; Ferreira, I.C.F.R. Phenolic Compounds: Current Industrial Applications, Limitations and Future Challenges. Food Funct. 2021, 12, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Terahara, N. Flavonoids in Foods: A Review. Nat. Prod. Commun. 2015, 10, 521–528. [Google Scholar] [CrossRef]
- Dias, M.C.; Pinto, D.C.G.A.; Silva, A.M.S. Plant Flavonoids: Chemical Characteristics and Biological Activity. Molecules 2021, 26, 5377. [Google Scholar] [CrossRef]
- Jucá, M.M.; Cysne Filho, F.M.S.; de Almeida, J.C.; da Mesquita, D.S.; de Barriga, J.R.M.; Dias, K.C.F.; Barbosa, T.M.; Vasconcelos, L.C.; Leal, L.K.A.M.; Ribeiro, J.E.; et al. Flavonoids: Biological Activities and Therapeutic Potential. Nat. Prod. Res. 2020, 34, 692–705. [Google Scholar] [CrossRef]
- Gaafar, A.; Aly, H.; Salama, Z.; Mahmoud, K. Characterizing the Antioxidant and Anticancer Properties of Secondary Metabolites From Red and White Cabbage Brassica oleracea L. Var. Capitata. World J. Pharm. Res. 2014, 3, 171–186. [Google Scholar]
- ElGamal, R.; Song, C.; Rayan, A.M.; Liu, C.; Al-Rejaie, S.; ElMasry, G. Thermal Degradation of Bioactive Compounds during Drying Process of Horticultural and Agronomic Products: A Comprehensive Overview. Agronomy 2023, 13, 1580. [Google Scholar] [CrossRef]
- Oladzadabbasabadi, N.; Mohammadi Nafchi, A.; Ghasemlou, M.; Ariffin, F.; Singh, Z.; Al-Hassan, A.A. Natural Anthocyanins: Sources, Extraction, Characterization, and Suitability for Smart Packaging. Food Packag. Shelf Life 2022, 33, 100872. [Google Scholar] [CrossRef]
- Mattioli, R.; Francioso, A.; Mosca, L.; Silva, P. Anthocyanins: A Comprehensive Review of Their Chemical Properties and Health Effects on Cardiovascular and Neurodegenerative Diseases. Molecules 2020, 25, 3809. [Google Scholar] [CrossRef]
- Bernstein, A.; Noreña, C.P.Z. Kinetics of Enzymatic Inactivation and Loss of Anthocyanins and Antioxidant Activity in Red Cabbage Blanched under Different Conditions. J. Food Biochem. 2017, 41, e12340. [Google Scholar] [CrossRef]
- Sakulnarmrat, K.; Wongsrikaew, D.; Konczak, I. Microencapsulation of Red Cabbage Anthocyanin-Rich Extract by Drum Drying Technique. LWT 2021, 137, 110473. [Google Scholar] [CrossRef]
- Athira, V.A.; Gokulvel, E.; Nandhu Lal, A.M.; Venugopalan, V.V.; Rajkumar; Venkatesh, T. Advances in Drying Techniques for Retention of Antioxidants in Agro Produces. Crit. Rev. Food Sci. Nutr. 2022, 63, 10849–10865. [Google Scholar] [CrossRef] [PubMed]
- Sikorska-Zimny, K.; Beneduce, L. The Metabolism of Glucosinolates by Gut Microbiota. Nutrients 2021, 13, 2750. [Google Scholar] [CrossRef] [PubMed]
- Kamal, R.M.; Razis, A.F.A.; Sukri, N.S.M.; Perimal, E.K.; Ahmad, H.; Patrick, R.; Djedaini-Pilard, F.; Mazzon, E.; Rigaud, S. Beneficial Health Effects of Glucosinolates-Derived Isothiocyanates on Cardiovascular and Neurodegenerative Diseases. Molecules 2022, 27, 624. [Google Scholar] [CrossRef] [PubMed]
- Vega-Galvez, A.; Gomez-Perez, L.; Zepeda, F.; Vidal, R.; Grunenwald, F.; Mej, N.; Pasten, A.; Araya, M.; Ah-Hen, K. Assessment of Bio-Compounds Content, Antioxidant Activity, and Neuroprotective Effect of Red Cabbage ( Brassica oleracea Var. Capitata rubra ) Processed by Convective Drying at Different Temperatures. Antioxidants 2023, 12, 1789. [Google Scholar] [CrossRef] [PubMed]
- Barba, F.J.; Nikmaram, N.; Roohinejad, S.; Khelfa, A.; Zhu, Z.; Koubaa, M. Bioavailability of Glucosinolates and Their Breakdown Products: Impact of Processing. Front. Nutr. 2016, 3, 24. [Google Scholar] [CrossRef] [PubMed]
- Oerlemans, K.; Barrett, D.M.; Suades, C.B.; Verkerk, R.; Dekker, M. Thermal Degradation of Glucosinolates in Red Cabbage. Food Chem. 2006, 95, 19–29. [Google Scholar] [CrossRef]
- Favela-González, K.M.; Hernández-Almanza, A.Y.; De la Fuente-Salcido, N.M. The Value of Bioactive Compounds of Cruciferous Vegetables (Brassica) as Antimicrobials and Antioxidants: A Review. J. Food Biochem. 2020, 44, e13414. [Google Scholar] [CrossRef]
- Li, Z.; Lee, H.W.; Liang, X.; Liang, D.; Wang, Q.; Huang, D.; Ong, C.N. Profiling of Phenolic Compounds and Antioxidant Activity of 12 Cruciferous Vegetables. Molecules 2018, 23, 1139. [Google Scholar] [CrossRef]
- Coman, V.; Vodnar, D.C. Hydroxycinnamic Acids and Human Health: Recent Advances. J. Sci. Food Agric. 2020, 100, 483–499. [Google Scholar] [CrossRef]
- Xiao, Z.; Rausch, S.R.; Luo, Y.; Sun, J.; Yu, L.; Wang, Q.; Chen, P.; Yu, L.; Stommel, J.R. Microgreens of Brassicaceae: Genetic Diversity of Phytochemical Concentrations and Antioxidant Capacity. LWT 2019, 101, 731–737. [Google Scholar] [CrossRef]
- Park, S.; Arasu, M.V.; Jiang, N.; Choi, S.H.; Lim, Y.P.; Park, J.T.; Al-Dhabi, N.A.; Kim, S.J. Metabolite Profiling of Phenolics, Anthocyanins and Flavonols in Cabbage (Brassica oleracea Var. Capitata). Ind. Crops Prod. 2014, 60, 8–14. [Google Scholar] [CrossRef]
- Wojdyło, A.; Figiel, A.; Lech, K.; Nowicka, P.; Oszmiański, J. Effect of Convective and Vacuum-Microwave Drying on the Bioactive Compounds, Color, and Antioxidant Capacity of Sour Cherries. Food Bioprocess Technol. 2014, 7, 829–841. [Google Scholar] [CrossRef]
- Mangla, B.; Javed, S.; Sultan, M.H.; Kumar, P.; Kohli, K.; Najmi, A.; Alhazmi, H.A.; Al Bratty, M.; Ahsan, W. Sulforaphane: A Review of Its Therapeutic Potentials, Advances in Its Nanodelivery, Recent Patents, and Clinical Trials. Phyther. Res. 2021, 35, 5440–5458. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.A.; Motaal, A.A. Sulforaphane Composition, Cytotoxic and Antioxidant Activity of Crucifer Vegetables. J. Adv. Res. 2010, 1, 65–70. [Google Scholar] [CrossRef]
- Park, S.; Arasu, M.; Lee, M.; Chun, J.; Seo, J.; Al-Dhabi, N.; Kim, S. Analysis and Metabolite Profiling of Glucosinolates, Anthocyanins and Free Amino Acids in Inbred Lines of Green and Red Cabbage (Brassica oleracea L.). LWT 2014, 58, 203–213. [Google Scholar] [CrossRef]
- Lin, X.; Wu, L.; Wang, X.; Yao, L.; Wang, L. Ultrasonic-Assisted Extraction for Flavonoid Compounds Content and Antioxidant Activities of India Moringa oleifera L. Leaves: Simultaneous Optimization, HPLC Characterization and Comparison with Other Methods. J. Appl. Res. Med. Aromat. Plants 2021, 20, 100284. [Google Scholar] [CrossRef]
- Jin, N.; Jin, L.; Luo, S.; Tang, Z.; Liu, Z.; Wei, S.; Liu, F.; Zhao, X. Comprehensive Evaluation of Amino Acids and Polyphenols in 69 Varieties of Green Cabbage (Brassica oleracea L. Var. Capitata L.) Based on Multivariate Statisticl Analysis. Molecules 2021, 26, 5355. [Google Scholar] [CrossRef]
- Mohanty, B.; Mahanty, A.; Ganguly, S.; Sankar, T.V.; Chakraborty, K.; Rangasamy, A.; Paul, B.; Sarma, D.; Mathew, S.; Asha, K.K.; et al. Amino Acid Compositions of 27 Food Fishes and Their Importance in Clinical Nutrition. J. Amino Acids 2014, 2014, 269797. [Google Scholar] [CrossRef]
- Volden, J.; Borge, G.I.A.; Bengtsson, G.B.; Hansen, M.; Thygesen, I.E.; Wicklund, T. Effect of Thermal Treatment on Glucosinolates and Antioxidant-Related Parameters in Red Cabbage (Brassica oleracea L. Ssp. Capitata f. Rubra). Food Chem. 2008, 109, 595–605. [Google Scholar] [CrossRef]
- Bi, Y.; Ni, J.; Xue, X.; Zhou, Z.; Tian, W.; Orsat, V.; Yan, S.; Peng, W.; Fang, X. Effect of Different Drying Methods on the Amino Acids, α-Dicarbonyls and Volatile Compounds of Rape Bee Pollen. Food Sci. Hum. Wellness 2024, 13, 517–527. [Google Scholar] [CrossRef]
- Calder, P.C. Functional Roles of Fatty Acids and Their Effects on Human Health. J. Parenter. Enter. Nutr. 2015, 39, 18S–32S. [Google Scholar] [CrossRef]
- Mancini, A.; Imperlini, E.; Nigro, E.; Montagnese, C.; Daniele, A.; Orrù, S.; Buono, P. Biological and Nutritional Properties of Palm Oil and Palmitic Acid: Effects on Health. Molecules 2015, 20, 17339–17361. [Google Scholar] [CrossRef] [PubMed]
- Białek, M.; Rutkowska, J. The Importance of γ-Linolenic Acid in the Prevention and Treatment. Postepy Hig. Med. Dosw. 2015, 69, 892–904. [Google Scholar] [CrossRef]
- Prabhu, Y.; Gopalakrishnan, A. γ-Linolenic Acid Ameliorates DHEA Induced pro-Inflammatory Response in Polycystic Ovary Syndrome via PPAR-γ Signaling in Rats. Reprod. Biol. 2020, 20, 348–356. [Google Scholar] [CrossRef]
- Volf, I.; Ignat, I.; Neamtu, M.; Popa, V.I. Thermal Stability, Antioxidant Activity, and Photo-Oxidation of Natural Polyphenols. Chem. Pap. 2014, 68, 121–129. [Google Scholar] [CrossRef]
- Poprac, P.; Jomova, K.; Simunkova, M.; Kollar, V.; Rhodes, C.J.; Valko, M. Targeting Free Radicals in Oxidative Stress-Related Human Diseases. Trends Pharmacol. Sci. 2017, 38, 592–607. [Google Scholar] [CrossRef] [PubMed]
- Jomova, K.; Raptova, R.; Alomar, S.Y.; Alwasel, S.H.; Nepovimova, E.; Kuca, K.; Valko, M. Reactive Oxygen Species, Toxicity, Oxidative Stress, and Antioxidants: Chronic Diseases and Aging; Springer: Berlin/Heidelberg, Germany, 2023; Volume 97, ISBN 0123456789. [Google Scholar]
- El-Lateef, H.M.A.; El-Dabea, T.; Khalaf, M.M.; Abu-Dief, A.M. Recent Overview of Potent Antioxidant Activity of Coordination Compounds. Antioxidants 2023, 12, 213. [Google Scholar] [CrossRef]
- Rahaman, M.M.; Hossain, R.; Herrera-Bravo, J.; Islam, M.T.; Atolani, O.; Adeyemi, O.S.; Owolodun, O.A.; Kambizi, L.; Daştan, S.D.; Calina, D.; et al. Natural Antioxidants from Some Fruits, Seeds, Foods, Natural Products, and Associated Health Benefits: An Update. Food Sci. Nutr. 2023, 11, 1657–1670. [Google Scholar] [CrossRef] [PubMed]
- Wiczkowski, W.; Szawara-Nowak, D.; Topolska, J. Changes in the Content and Composition of Anthocyanins in Red Cabbage and Its Antioxidant Capacity during Fermentation, Storage and Stewing. Food Chem. 2015, 167, 115–123. [Google Scholar] [CrossRef]
- Wojdyło, A.; Lech, K.; Nowicka, P. Effects of Different Drying Methods on the Retention of Bioactive Compounds, on-Ine Antioxidant Capacity and Color of the Novel Snack from Red-Fleshed Apples. Molecules 2020, 25, 5521. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Y.; Corke, H.; Zhu, H. Dynamic Changes in Flavonoids Content during Congou Black Tea Processing. LWT 2022, 170, 114073. [Google Scholar] [CrossRef]
- Karam, M.C.; Petit, J.; Zimmer, D.; Baudelaire Djantou, E.; Scher, J. Effects of Drying and Grinding in Production of Fruit and Vegetable Powders: A Review. J. Food Eng. 2016, 188, 32–49. [Google Scholar] [CrossRef]
- How, Y.K.; Siow, L.F. Effects of Convection-, Vacuum-and Freeze-Drying on Antioxidant, Physicochemical Properties, Functional Properties and Storage Stability of Stink Bean (Parkia speciosa) Powder. J. Food Sci. Technol. 2020, 57, 4637–4648. [Google Scholar] [CrossRef]
- Chandrakanthan, M.; Handunnetti, S.M.; Premakumara, G.S.A.; Kathirgamanathar, S. Topical Anti-Inflammatory Activity of Essential Oils of Alpinia Calcarata Rosc., Its Main Constituents, and Possible Mechanism of Action. Evid.-Based Complement Altern. Med. 2020, 2020, 2035671. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary Polyphenols, Oxidative Stress and Antioxidant and Anti-Inflammatory Effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Callaway, J.B.; Ting, J.P.Y. Inflammasomes: Mechanism of Action, Role in Disease, and Therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef]
- Theoduloz, C.; Delporte, C.; Valenzuela-Barra, G.; Silva, X.; Cádiz, S.; Bustamante, F.; Pertino, M.W.; Schmeda-Hirschmann, G. Topical Anti-Inflammatory Activity of New Hybrid Molecules of Terpenes and Synthetic Drugs. Molecules 2015, 20, 11219–11235. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, A.; Jaswal, V.S.; Choudhary, S.; Sonika; Sharma, A.; Beniwal, V.; Tuli, H.S.; Sharma, S. Ferulic Acid: A Promising Therapeutic Phytochemical and Recent Patents Advances. Recent Pat. Inflamm. Allergy Drug Discov. 2019, 13, 115–123. [Google Scholar] [CrossRef]
- Castro, J.; Rivera, D.; Franco, L.A. Topical Anti-Inflammatory Activity in TPA-Induced Mouse Ear Edema Model and in Vitro Antibacterial Properties of Cordia Alba Flowers. J. Pharm. Investig. 2019, 49, 331–336. [Google Scholar] [CrossRef]
- Liu, Y.; Shen, J.; Xu, L.; Li, H.; Li, Y.; Yi, L. Ferulic Acid Inhibits Neuro-Inflammation in Mice Exposed to Chronic Unpredictable Mild Stress. Int. Immunopharmacol. 2017, 45, 128–134. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, B.; Berrada, H.; Barba, F.J. Marine Resources and Cancer Therapy: From Current Evidence to Challenges for Functional Foods Development. Curr. Opin. Food Sci. 2022, 44, 100805. [Google Scholar] [CrossRef]
- Cömert, E.D.; Gökmen, V. Physiological Relevance of Food Antioxidants. Adv. Food Nutr. Res. 2020, 93, 205–250. [Google Scholar] [CrossRef]
- Toydemir, G.; Gultekin Subasi, B.; Hall, R.D.; Beekwilder, J.; Boyacioglu, D.; Capanoglu, E. Effect of Food Processing on Antioxidants, Their Bioavailability and Potential Relevance to Human Health. Food Chem. X 2022, 14, 100334. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, B.; Sun, J.; Jiang, X.; Bai, W. Anthocyanin Supplement as a Dietary Strategy in Cancer Prevention and Management: A Comprehensive Review. Crit. Rev. Food Sci. Nutr. 2021, 62, 7242–7254. [Google Scholar] [CrossRef] [PubMed]
- Calderón-Reyes, C.; Pezoa, R.S.; Leal, P.; Ribera-Fonseca, A.; Cáceres, C.; Riquelme, I.; Zambrano, T.; Peña, D.; Alberdi, M.; Reyes-Díaz, M. Anthocyanin-Rich Extracts of Calafate (Berberis microphylla G. Forst.) Fruits Decrease In Vitro Viability and Migration of Human Gastric and Gallbladder Cancer Cell Lines. J. Soil Sci. Plant Nutr. 2020, 20, 1891–1903. [Google Scholar] [CrossRef]
- Kaiser, A.E.; Baniasadi, M.; Giansiracusa, D.; Giansiracusa, M.; Garcia, M.; Fryda, Z.; Wong, T.L.; Bishayee, A. Sulforaphane: A Broccoli Bioactive Phytocompound with Cancer Preventive Potential. Cancers 2021, 13, 4796. [Google Scholar] [CrossRef]
- Iahtisham-Ul-Haq; Khan, S.; Awan, K.A.; Iqbal, M.J. Sulforaphane as a Potential Remedy against Cancer: Comprehensive Mechanistic Review. J. Food Biochem. 2021, 46, e13886. [Google Scholar] [CrossRef]

| Drying Process | Equipment | Conditions |
|---|---|---|
| CD | Hot air dryer designed and constructed at the Department of Food Engineering, Universidad de La Serena, La Serena, Chile. | 60 °C and set at 1.5 m/s air speed for 5.5 h. |
| VD | Vacuum oven (Memmert, model VO 400, Schwabach, Germany). | 10 kPa and 60 °C for 10 h. |
| IRD | Infrared drying oven designed and constructed at the Department of Food Engineering, Universidad de La Serena, La Serena, Chile. | Two 175-Watt lamps as radiant source for 8.5 h at 60 °C. |
| LTVD | Vacuum oven (Memmert, model VOcool 400, Schwabach, Germany). | 1 kPa and 20 °C for 35.6 h. |
| FD | Freeze-dryer (Virtis, AdVantage Plus, Gardiner, NY, USA). | Pre-frozen at −80 °C and then freeze-dried for 24 h. Condenser temperature: −60 °C and chamber pressure: 0.027 kPa. |
| Parameters g/100 g d.m. | Fresh-b | CD | VD | IRD | LTVD | FD |
|---|---|---|---|---|---|---|
| Water * | 92.59 ± 0.12 a | 14.05 ± 0.09 c | 11.31 ± 0.08 d | 13.99 ± 0.04 c | 15.06 ± 0.56 b | 10.95 ± 0.23 d |
| Lipid | 0.21 ± 0.01 e | 1.04 ± 0.03 b | 0.53 ± 0.07 d | 0.90 ± 0.04 c | 0.96 ± 0.09 bc | 1.15 ± 0.02 a |
| Ash | 9.99 ± 0.33 b | 8.02 ± 0.06 e | 8.22 ± 0.06 de | 8.36 ± 0.09 d | 10.27 ± 0.14 a | 9.00 ± 0.05 c |
| Protein | 19.19 ± 3.10 a | 17.87 ± 0.36 ab | 17.14 ± 0.27 ab | 17.32 ± 0.28 ab | 15.76 ± 0.30 b | 17.02 ± 0.16 ab |
| Crude fiber | 13.31 ± 0.61 a | 10.95 ± 1.19 b | 9.91 ± 0.95 b | 12.35 ± 0.41 ab | 13.15 ± 1.32 a | 11.53 ± 0.96 ab |
| Total carbohydrates (difference) | 72.01 ± 1.88 c | 62.80 ± 0.41 ab | 65.73 ± 0.23 ab | 63.15 ± 0.32 ab | 62.02 ± 0.81 abc | 64.86 ± 0.09 bc |
| Water activity ** | 0.993 ± 0.004 a | 0.467 ± 0.002 b | 0.339 ± 0.003 e | 0.452 ± 0.002 c | 0.427 ± 0.001 d | 0.335 ± 0.01 e |
| Parameters | Drying Methods | |||||
|---|---|---|---|---|---|---|
| Fresh-b | CD | VD | IRD | LTVD | FD | |
| TPC (mg GAE/g d.m.) | 11.53 ± 0.39 ab | 9.81 ± 0.28 d | 11.89 ± 0.28 a | 9.93 ± 0.27 d | 10.91 ± 0.51 bc | 10.80 ± 0.34 c |
| TFC (mg QE/g d.m.) | 10.56 ± 0.75 b | 6.66 ± 0.59 d | 11.30 ± 0.90 a | 9.26 ± 0.39 c | 8.77 ± 0.67 c | 10.15 ± 0.62 b |
| TAC (mg Cya3 glu/g d.m.) | 0.391 ± 0.004 a | 0.196 ± 0.025 d | 0.265 ± 0.011 b | 0.122 ± 0.009 e | 0.257 ± 0.019 b | 0.223 ± 0.005 c |
| Caffeic acid (µg/g d.m.) | ND | 479 ± 20.08 a | 427 ± 10.80 ab | 388 ± 29.04 b | 412 ± 28.72 b | 290 ± 44.38 c |
| Ferulic acid (µg/g d.m.) | 1520 ± 69.24 d | 3173 ± 11.40 a | 3073 ± 70.21 a | 3118 ± 242.66 a | 2568 ± 153.86 b | 2151 ± 334.86 c |
| Sinapic acid (µg/g d.m.) | 1107 ± 85.60 c | 959 ± 10.03 bc | 915 ± 19.06 cd | 1075 ± 74.62 ab | 795 ± 44.43 c | 785 ± 102.57 c |
| TGC (µmol SE/g d.m.) | 47.38 ± 2.32 b | 43.26 ± 3.76 c | 51.15 ± 3.31 a | 42.17 ± 1.95 c | 41.30 ± 3.37 c | 47.05 ± 3.05 b |
| SFN (mg/g d.m.) | ND | 0.004 ± 0.002 b | 0.039 ± 0.004 b | 0.001 d | 0.070 ± 0.004 a | 0.032 ± 0.001 c |
| Amino Acids (g/100 g) d.m. | CD | VD | IRD | LTVD | FD |
|---|---|---|---|---|---|
| Aspartic acid | 0.83 ± 0.08 b | 0.64 ± 0.11 bc | 1.06 ± 0.04 a | 0.47 ± 0.14 c | 1.19 ± 0.18 a |
| Glutamic acid | 2.99 ± 0.29 a | 2.10 ± 0.04 b | 3.37 ± 0.10 a | 1.13 ± 0.34 c | 3.58 ± 0.51 a |
| Serine | 0.59 ± 0.03 ab | 0.47 ± 0.09 b | 0.69 ± 0.03 a | 0.26 ± 0.08 c | 0.67 ± 0.11 a |
| Glycine | 0.46 ± 0.06 b | 0.40 ± 0.11 b | 0.60 ± 0.01 ab | 0.17 ± 0.07 c | 0.47 ± 0.07 a |
| Threonine 1 | 0.29 ± 0.05 bc | 0.24 ± 0.07 b | 0.44 ± 0.03 a | 0.15 ± 0.05 c | 0.47 ± 0.09 c |
| Arginine | 0.72 ± 0.04 ab | 0.58 ± 0.16 b | 0.84 ± 0.01 a | 0.30 ± 0.08 c | 0.74 ± 0.12 ab |
| Alanine | 0.47 ± 0.04 a | 0.35 ± 0.08 b | 0.52 ± 0.01 a | 0.21 ± 0.06 c | 0.54 ± 0.08 a |
| Tyrosine | 0.24 ± 0.00 b | 0.29 ± 0.02 ab | 0.32 ± 0.04 a | 0.04 ± 0.02 c | 0.26 ± 0.07 ab |
| Valine 1 | 0.31 ± 0.04 b | 0.28 ± 0.05 b | 0.45 ± 0.02 a | 0.17 ± 0.05 c | 0.45 ± 0.06 a |
| Phenylalanine | 0.44 ± 0.01 b | 0.37 ± 0.10 bc | 0.56 ± 0.01 a | 0.28 ± 0.10 c | 0.61 ± 0.08 a |
| Isoleucine 1 | 0.68 ± 0.04 bc | 0.58 ± 0.09 c | 0.90 ± 0.04 a | 0.36 ± 0.11 d | 0.83 ± 0.12 ab |
| Leucine 1 | 0.30 ± 0.00 b | 0.29 ± 0.08 b | 0.41 ± 0.01 a | 0.18 ± 0.04 c | 0.30 ± 0.06 b |
| Lysine 1 | 0.44 ± 0.01 bc | 0.37 ± 0.04 c | 0.52 ± 0.03 ab | 0.22 ± 0.07 d | 0.60 ± 0.07 a |
| Total EAAs | 2.02 ± 0.15 b | 1.76 ± 0.33 b | 2.73 ± 0.13 a | 1.08 ± 0.32 c | 2.65 ± 0.41 a |
| Total AA | 8.76 ± 0.69 ab | 6.9 ± 1.47 b | 10.70 ± 0.37 a | 4.0 ± 1.20 c | 10.70 ± 1.61 a |
| Fatty Acid (g/100 g FAMES) | Drying Methods | ||||
|---|---|---|---|---|---|
| CD | VD | IRD | LTVD | FD | |
| Saturated Fatty Acids | |||||
| C12:0 Lauric Acid | 0.07 ± 0.02 a | 0.17 ± 0.04 a | 0.07 ± 0.04 a | 0.13 ± 0.05 a | 0.03 ± 0.01 a |
| C14:0 Myristic Acid | 0.11 ± 0.01 b | 0.29 ± 0.03 a | 0.07 ± 0.02 b | 0.03 ± 0.01 b | 0.09 ± 0.04 b |
| C15:0 Pentadecanoic Acid | 0.40 ± 0.06 ab | 0.52 ± 0.06 a | 0.33 ± 0.01 b | 0.29 ± 0.04 b | 0.42 ± 0.06 ab |
| C16:0 Palmitic Acid | 15.95 ± 0.37 b | 15.90 ± 0.36 b | 17.44 ± 0.39 a | 16.92 ± 0.513 ab | 17.39 ± 0.15 a |
| C17:0 Heptadecanoic Acid | 0.57 ± 0.02 c | 0.87 ± 0.06 a | 0.79 ± 0.02 b | 0.65 ± 0.02 c | 0.61 ± 0.02 c |
| C18:0 Stearic Acid | 4.81 ± 0.17 a | 3.99 ± 0.14 b | 4.13 ± 0.11 b | 3.95 ± 0.14 b | 4.38 ± 0.25 ab |
| C20:0 Arachidic Acid | 0.59 ± 0.09 a | 0.59 ± 0.02 b | 0.40 ± 0.07 b | 0.35 ± 0.04 b | 0.56 ± 0.04 ab |
| C22:0 Behenic Acid | 0.28± 0.14 a | 0.56 ± 0.25 a | 0.19 ± 0.07 a | 0.21 ± 0.06 a | 0.35 ± 0.02 a |
| C23:0 Tricosanoic Acid | n.d. | n.d. | n.d. | n.d. | 0.69 ± 0.14 |
| Monounsaturated fatty acids | |||||
| C16:1 Palmitoleic Acid | 0.28 ± 0.07 ab | 0.43 ± 0.08 a | 0.21 ± 0.03 b | 0.17 ± 0.05 b | 0.25 ± 0.07 ab |
| C17:1 Cis-10-Heptadecenoic Acid | 0.32 ± 0.03 a | n.d. | 0.29 ± 0.01 a | 0.35 ± 0.02 a | 0.32 ± 0.07 a |
| C18:1n9c/C18:1n9t Oleic Acid/Elaidic Acid | 1.56 ± 0.01 a | 1.31 ± 0.09 b | 1.21 ± 0.03 bc | 1.06 ± 0.07 c | 1.56 ± 0.08 a |
| C20:1n9 Cis-11-Eicosenoic Acid | 0.31 ± 0.21 a | 0.16 ± 0.02 a | 0.11 ± 0.04 a | 0.16 ± 0.03 a | 0.29 ± 0.08 a |
| C24:1n9 Nervonic Acid | 0.43 ± 0.08 ab | 0.70 ± 0.24 a | 0.41 ± 0.03 ab | 0.34 ± 0.11 b | 0.47 ± 0.04 ab |
| Polyunsaturated Fatty Acids | |||||
| C18:2n6c Linoleic Acid | 23.43 ± 0.12 b | 21.69 ± 0.37 c | 21.69 ± 0.07 c | 22.19 ± 0.10 c | 24.46 ± 0.11 a |
| C18:3n6 γ-linolenic acid | 50.04 ± 0.17 ab | 51.61± 1.73 a | 51.97 ± 0.12 a | 52.33 ± 0.16 a | 47.51 ± 0.56 b |
| C20:2 Cis-11,14-Eicosadienoic Acid | 0.15 ± 0.05 a | 0.39 ± 0.21 a | 0.16 ± 0.09 a | 0.25 ± 0.08 a | 0.27 ± 0.06 a |
| C20:3n3 Cis-11,14,17-Eicosatrienoic Acid | 0.14 ± 0.03 a | 0.46 ± 0.24 a | 0.11 ± 0.07 a | 0.14 ± 0.01 a | 0.18 ± 0.02 a |
| C22:6n3 Cis-4,7,10,13,16,19-Docosahexaenoic Acid | 0.64 ± 0.26 a | 1.07 ± 0.55 a | 0.42 ± 0.10 a | 0.40 ± 0.05 a | n.d. |
| SFAs | 22.79 ± 0.23 bc | 22.90 ± 0.22 bc | 23.41 ± 0.22 b | 22.53 ± 0.34 c | 24.51 ± 0.29 a |
| MUFAs | 2.80 ± 0.192 a | 2.90 ± 0.52 a | 2.22 ± 0.12 a | 2.08 ± 0.21 a | 2.79 ± 0.14 a |
| PUFAs | 74.40 ± 0.11 a | 74.58 ± 0.90 a | 74.36 ± 0.13 a | 75.17 ± 0.35 a | 77.62 ± 0.43 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mejías, N.; Vega-Galvez, A.; Gomez-Perez, L.S.; Pasten, A.; Uribe, E.; Cortés, A.; Valenzuela-Barra, G.; Camus, J.; Delporte, C.; Bernal, G. Health-Promoting Properties of Processed Red Cabbage (Brassica oleracea var. capitata f. rubra): Effects of Drying Methods on Bio-Compound Retention. Foods 2024, 13, 830. https://doi.org/10.3390/foods13060830
Mejías N, Vega-Galvez A, Gomez-Perez LS, Pasten A, Uribe E, Cortés A, Valenzuela-Barra G, Camus J, Delporte C, Bernal G. Health-Promoting Properties of Processed Red Cabbage (Brassica oleracea var. capitata f. rubra): Effects of Drying Methods on Bio-Compound Retention. Foods. 2024; 13(6):830. https://doi.org/10.3390/foods13060830
Chicago/Turabian StyleMejías, Nicol, Antonio Vega-Galvez, Luis S. Gomez-Perez, Alexis Pasten, Elsa Uribe, Anielka Cortés, Gabriela Valenzuela-Barra, Javiera Camus, Carla Delporte, and Giuliano Bernal. 2024. "Health-Promoting Properties of Processed Red Cabbage (Brassica oleracea var. capitata f. rubra): Effects of Drying Methods on Bio-Compound Retention" Foods 13, no. 6: 830. https://doi.org/10.3390/foods13060830
APA StyleMejías, N., Vega-Galvez, A., Gomez-Perez, L. S., Pasten, A., Uribe, E., Cortés, A., Valenzuela-Barra, G., Camus, J., Delporte, C., & Bernal, G. (2024). Health-Promoting Properties of Processed Red Cabbage (Brassica oleracea var. capitata f. rubra): Effects of Drying Methods on Bio-Compound Retention. Foods, 13(6), 830. https://doi.org/10.3390/foods13060830