Bioactive Potential of Olive Mill Waste Obtained from Cultivars Grown in the Island of Malta
Abstract
:1. Introduction
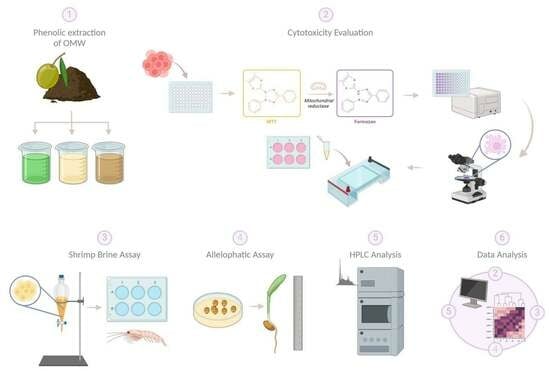
2. Materials and Methods
2.1. Collection and Extraction of Raw Materials
2.2. Cells
2.3. MTT Assay
2.4. Cell Staining
2.5. Preparation of Cells for Protein Assays
2.6. Apoptosis DNA
Gel Electrophoresis
2.7. Protein Content
Caspase-3 Assay
2.8. BCL-2 and BAX Protein Assays
2.9. Brine Shrimp Lethality Assay
2.10. Allelopathic Assay
Calculations
2.11. HPLC Profiling
2.12. Data Analysis
3. Results
3.1. Non-Adherent Cell Lines–Add Values
3.1.1. Effect of Bioactivity on Cancer Cell Lines
3.1.2. Effect of Cultivars on Cell Lines to Different Extraction Methods
3.2. Cell Staining
3.3. Apoptotic DNA Assay
3.4. Caspase-3 Activation
3.5. BAX and BCL-2
3.6. Allelopathic Assay
3.7. Brine Shrimp Lethality Assay
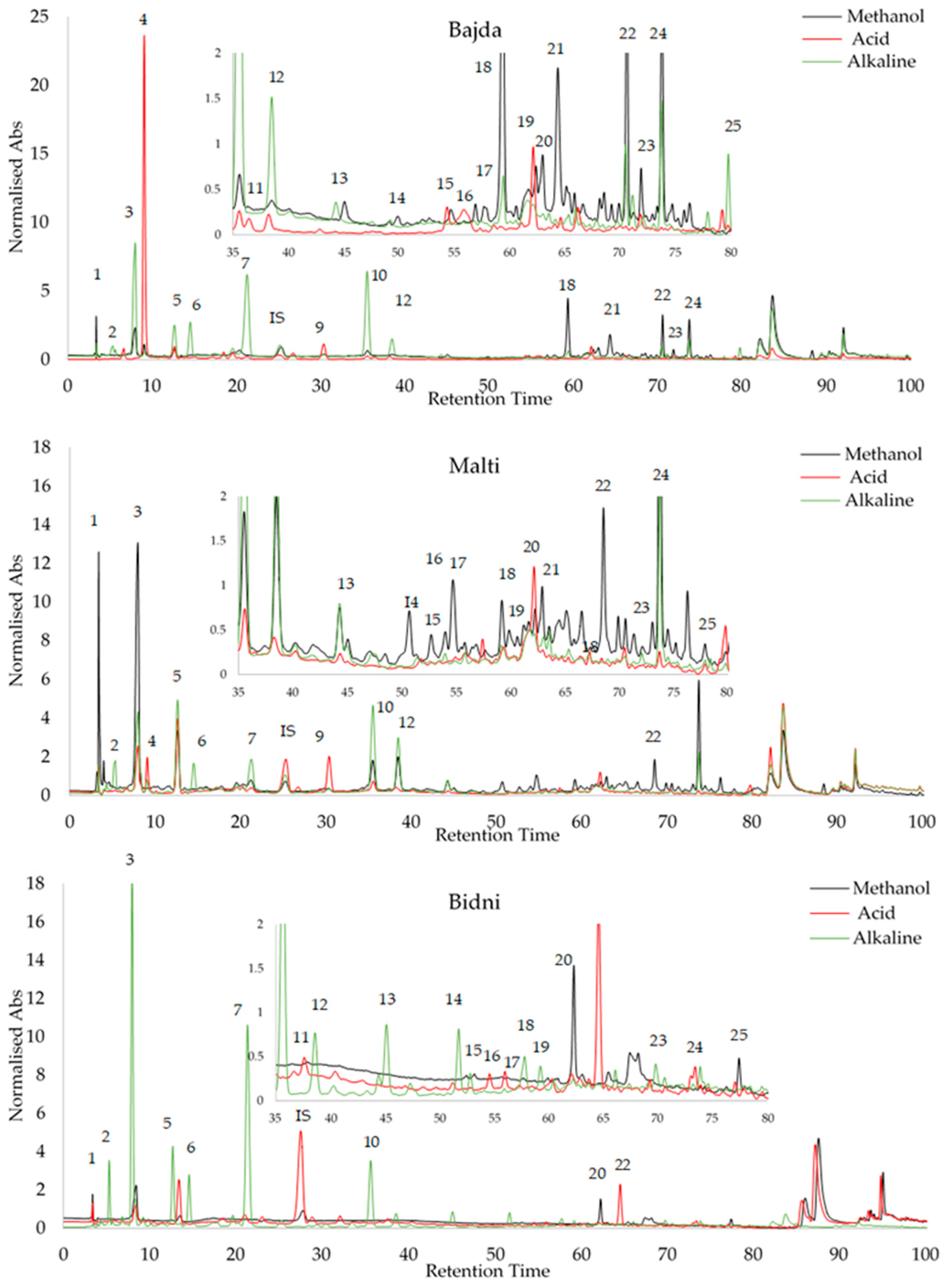
3.8. HPLC Analysis
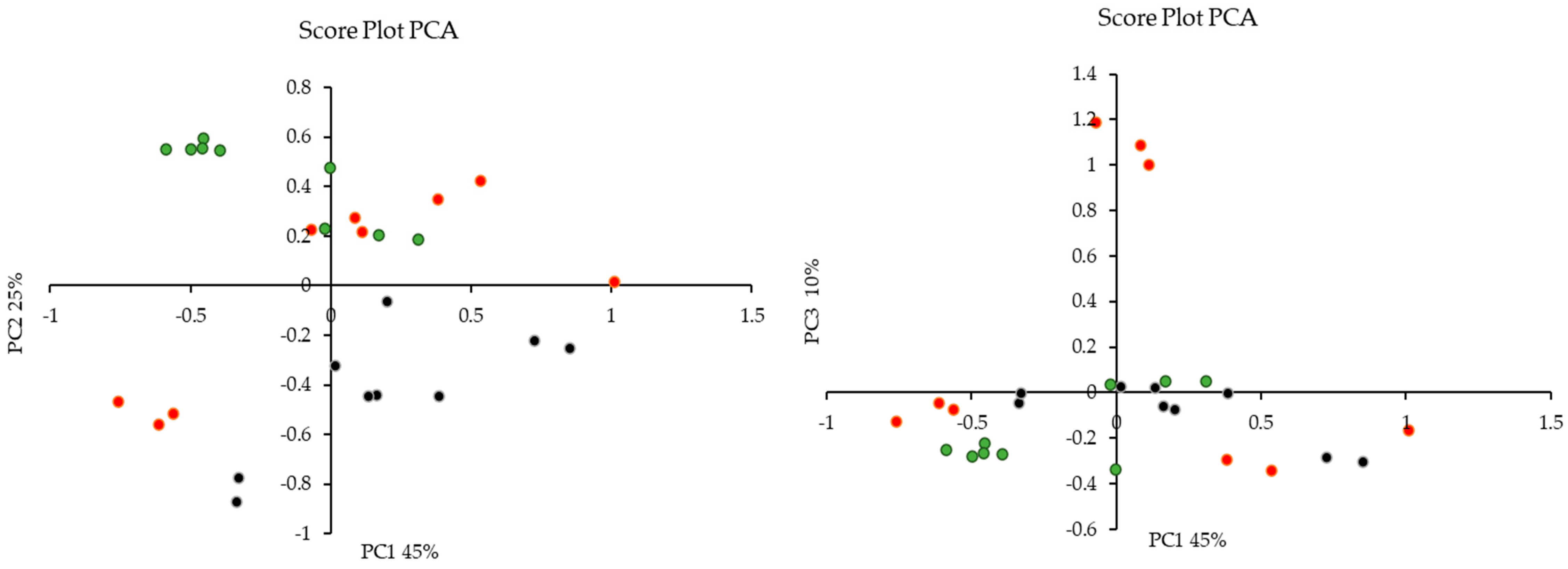
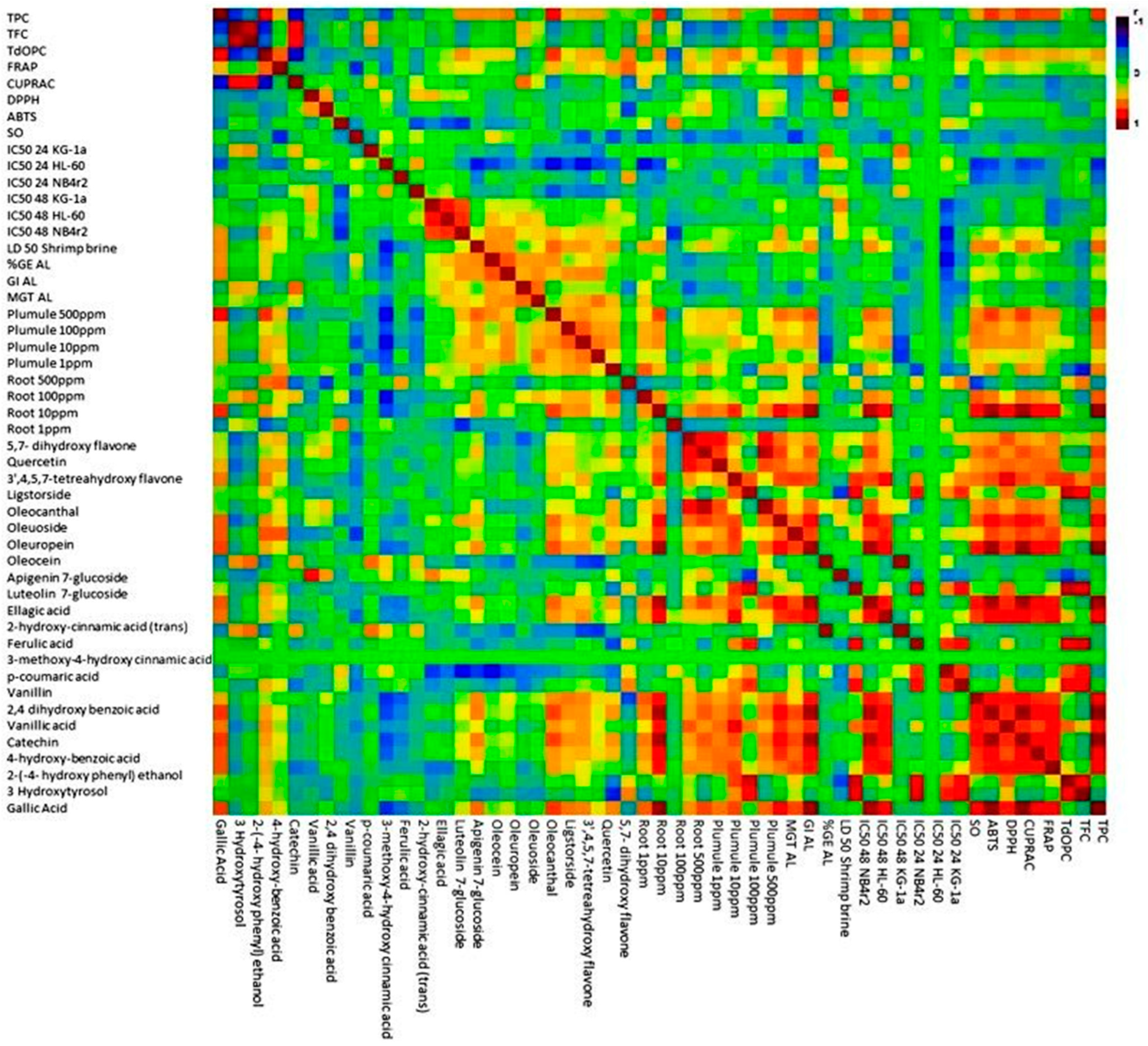
3.9. Multivariate Data Analysis
3.9.1. Principal Component Analysis (PCA)
3.9.2. Two-Way Cluster Analysis
3.9.3. Matrix Correlation Analysis
4. Discussion
4.1. Non-Adherent Cell Lines
4.2. Staining of Cells
4.3. DNA Laddering
4.4. Caspase-3 Activation
4.5. Apoptotic Regulation
4.6. Effect of Seed Germination
Root and Plumule Length
4.7. Shrimp Brine Assay
4.8. OMW HPLC Profiling
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bala, S.; Garg, D.; Sridhar, K.; Inbaraj, B.S.; Singh, R.; Kamma, S.; Tripathi, M.; Sharma, M. Transformation of Agro-Waste into Value-Added Bioproducts and Bioactive Compounds: Micro/Nano Formulations and Application in the Agri-Food-Pharma Sector. Bioengineering 2023, 10, 152. [Google Scholar] [CrossRef]
- Shabir, S.; Ilyas, N.; Saeed, M.; Bibi, F.; Sayyed, R.Z.; Almalki, W.H. Treatment technologies for olive mill wastewater with impacts on plants. Environ. Res. 2023, 216, 114399. [Google Scholar] [CrossRef]
- Rodis, P.S.; Karathanos, V.T.; Mantzavinou, A. Partitioning of olive oil antioxidants between oil and water phases. J. Agric. Food Chem. 2002, 50, 596–601. [Google Scholar] [CrossRef]
- Artajo, L.-S.; Romero, M.P.; Sua’rez, M.; Motilva, M.J. Partition of phenolic compounds during the virgin olive oil industrial extraction process. Eur. Food Res. Technol. 2007, 225, 617–625. [Google Scholar] [CrossRef]
- Li, A.-N.; Zhang, Y.-J.; Xu, X.-R.; Chen, Y.-M.; Li, H.-B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, Y.; Xiang, J.; Wang, C.; Jonhson, J.B.; Beta, T. Diverse polyphenol components contribute to antioxidant activity and hypoglycemic potential of mulberry varieties. LWT 2023, 173, 114308. [Google Scholar] [CrossRef]
- Fantini, M.; Benvenuto, M.; Masuelli, L.; Frajese, G.; Tresoldi, I.; Modesti, A.; Bei, R. In vitro and in vivo antitumoral effects of combinations of polyphenols, or polyphenols and anticancer drugs: Perspectives on cancer treatment. Int. J. Mol. Sci. 2015, 16, 9236–9282. [Google Scholar] [CrossRef] [PubMed]
- Manach, G.; Williamson, C.; Morand, A.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in human and mammalians. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2015, 81, 230S–242S. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, D.; Vauzour, C.G.; Krueger, D.; Shanmuganayagam, J.; Reed, L.; Calani, P.; Mena, D.; Del Rio, C.; Croziern, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef]
- Wagner, H. Synergy research: Approaching a new generation of phytopharmaceuticals. Fitoterapia 2011, 82, 34–37. [Google Scholar] [CrossRef]
- Nurgali, R.T.; Jagoe, R.T.; Abalo, R. Editorial: Adverse effects of cancer chemotherapy: Anything new to improve tolerance and reduce sequelae? Front. Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Rawal, C.; Jain, N. A brief review on plant-derived natural compounds as an anticancer agents. Int. J. Herb. Med. 2018, 6, 28–36. [Google Scholar]
- Menendez, A.; Vazquez-Martin, R.; Colomer, J.; Brunet, A.; CarrascoPancorbo, R.; GarciaVillalba, A.; Fernandez-Gutierrez, R.; Segura-Carretero, A. Olive oil’s bitter principle reverses acquired autoresistance to trastuzumab (HerceptinTM) in HER2-overexpressing breast cancer cells. BMC Cancer 2007, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, I.; Soerjomataram, R.; Dikshit, S.; Eser, C.; Mathers, M.; Rebelo, D.M.; Parkin, D.; Forman, F.; Bray, D. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN Globocan. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Zubair, A.; Bhardwaj, A.; Ahmad, S.K.; Srivastava, M.A.; Khan, G.K.; Patel, S.; Singh, A.; Singh, P. Hydroxytyrosol induces apoptosis and cell cycle arrest and suppresses multiple oncogenic signaling pathways in prostate cancer cells. Nutrit. Cancer 2017, 69, 932–942. [Google Scholar] [CrossRef] [PubMed]
- Calahorra, E.; Martínez-Lara, C.; De Dios, E.; Siles, S. Hypoxia modulates the antioxidant effect of hydroxytyrosol in MCF-7 breast cancer cells. PLoS ONE 2018, 13, e0203892. [Google Scholar] [CrossRef] [PubMed]
- Hashim, J.; Worthington, P.; Allsopp, N.G.; Ternan, E.M.; Brown, M.J.; McCann, I.R.; Rowland, S.; Esposto, M.; Servili, C.I.R.; Gil, G. Virgin olive oil phenolics extract inhibit invasion of HT115 human and mammalian colon cancer cells in vitro and in vivo. Food Funct. 2014, 5, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R.; Rosignoli, P.; De Bartolomeo, A.; Fuccelli, R.; Servili, M.; Montedoro, G.F.; Morozzi, G. Oxidative DNA Damage Is Prevented by Extracts of Olive Oil, Hydroxytyrosol, and Other Olive Phenolic Compounds in Human Blood Mononuclear Cells and HL-60 Cells. J. Nutr. 2008, 138, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Song, D.Y.; Lim, J.I.; Jung, H.J.; Cho, S.Y.; Park, G.T.; Kwon, Y.H.; Kang, K.W.; Lee, M.S.; Choi, J.H.; Park, Y. Dietary oleuropein inhibits tumor angiogenesis and lymphangiogenesis in the B16F10 melanoma allograft model: A mechanism for the suppression of high-fat diet-induced solid tumor growth and lymph node metastasis. Oncotarget 2017, 8, 32027–32042. [Google Scholar] [CrossRef]
- Ghosh, A.; Banik, S.; Islam, M. In vitro thrombolytic, anthelmintic, anti-oxidant and cytotoxic activity with phytochemical screening of methanolic extract of Xanthium indicum leaves. Bangladesh J. Pharmacol. 2015, 10, 854–859. [Google Scholar] [CrossRef]
- Kibiti, C.; Afolayan, A. Antifungal activity and brine shrimp toxicity assessment of Bulbine abyssinica used in the folk medicine in the Eastern Cape Province, South Africa. Bangladesh J. Pharmacol. 2016, 11, 469–477. [Google Scholar] [CrossRef]
- Saliba, L.J.; Krzyz, R.M. Effect of heavy metals on hatching of brine-shrimp eggs. Marine Poll. Bull. 1976, 7, 181–182. [Google Scholar] [CrossRef]
- Harwing, J.; Scott, P.M. Brine shrimp (Artemia salina L.) larvae as a screening system for fungal toxins. Appl. Microbiol. 1971, 21, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.S.; Thampson, C.G.; Abramoritz, M. Artemia salina as a test organism for bioassay. Science 1956, 123, 464. [Google Scholar] [CrossRef] [PubMed]
- Hisem, D.; Hrouzek, P.; Tomek, P.; Tomšíčková, J.; Zapomělová, E.; Skácelová, K.; Lukešová, A.; Kopecký, J. Cyanobacterial cytotoxicity versus toxicity to brine shrimp Artemia salina. Toxicon 2011, 57, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Gueboudji, Z.; Addad, D.; Kadi, K.; Nagaz, K.; Secrafi, M.; Yahya, L.B.; Lachehib, B.; Abdelmalek, A. Biological activities and phenolic compounds of olive oil mill wastewater from Abani, endemic Algerian variety. Sci. Rep. 2022, 12, 6042. [Google Scholar] [CrossRef] [PubMed]
- Pierantozzi, P.; Zampini, C.; Torres, M.; Isla, M.I.; Verdenelli, R.A.; Meriles, J.M.; Maestri, D. Physico-chemical and toxicological assessment of liquid wastes from olive processing-related industries. J. Sci. Food Agric. 2011, 92, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Zhu, S.; Li, J.; Hui, X.; Wang, G.X. The developmental toxicity, bioaccumulation and distribution of oxidized single walled carbon nanotubes in Artemia salina. Toxicol. Res. 2018, 7, 897–906. [Google Scholar] [CrossRef]
- Trompeta, A.F.; Preiss, I.; Ben-Ami, F.; Benayahu, Y.; Charitidis, C.A. Toxicity testing of MWCNTs to aquatic organisms. RSC Adv. 2019, 9, 36707–36716. [Google Scholar] [CrossRef]
- Ates, M.; Daniels, J.; Arslan, Z.; Farah, I.O.; Rivera, H.F. Comparative evaluation of impact of Zn and ZnO nanoparticles on brine shrimp (Artemia salina) larvae: Effects of particle size and solubility on toxicity. Environ. Sci. Process Impacts 2013, 15, 225–233. [Google Scholar] [CrossRef]
- Da Silveira Carvalho, J.M.; De Morais Batista, A.H.; Nogueira, N.A.P.; Holanda, A.K.M.; De Sousa, J.R.; Zampieri, D.; Bezerra, M.J.B.; Stefânio Barreto, F.; De Moraes, M.O.; Batista, A.A.; et al. A biphosphinic ruthenium complex with potent anti-bacterial and anti-cancer activity. New J. Chem. 2017, 41, 13085–13095. [Google Scholar] [CrossRef]
- Roig, A.; Cayuela, M.L.; Sanchez-Monedero, M.A. An overview of olive mill wastes and their valorisation methods. Waste Manag. 2006, 26, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Reddy, G.V.P.; Shi, P.; Huang, J.; Hu, H.; Hu, T. Allelopathic effects of Cinnamomum septentrionale leaf litter on Eucalyptus grandis saplings. Glob. Ecol. Conserv. 2020, 21, e00872. [Google Scholar] [CrossRef]
- Rice, E.L. Allelopathy, 2nd ed.; Academic Press: New York, NY, USA, 1984. [Google Scholar]
- Turk, M.A.; Tawaha, A.M. Allelopathic effect of black mustard (Brassica nigra L.) on germination and growth of wild oat (Avena fatua L.). Crop Prot. 2003, 22, 673–677. [Google Scholar] [CrossRef]
- Kong, C.H.; Li, H.B.; Hu, F.; Xu, X.H.; Wang, P. Allelochemicals released by rice roots and residues in soil. Plant Soil 2006, 288, 47–56. [Google Scholar] [CrossRef]
- Duke, S.O. Weeding with allelochemicals and allelopathy-a commentary. Pest Manag. Sci. 2007, 63, 307. [Google Scholar] [CrossRef]
- Braine, J.W.; Curcio, G.R.; Wachowicz, C.M.; Hansel, F.A. Allelopathic effects of Araucaria angustifolia needle extracts in the growth of Lactuca sativa seeds. J. For. Res. 2012, 17, 440–445. [Google Scholar] [CrossRef]
- Soltys, D.; Krasuska, U.; Bogatek, R.; Gniazdowska, A. Allelochemicals as Bioherbicides-Present and Perspectives; Intechopen: London, UK, 2013. [Google Scholar]
- Attard, K.; Oztop, M.H.; Lia, F. The Effect of Hydrolysis on the Antioxidant Activity of Olive Mill Waste. Appl. Sci. 2022, 12, 12187. [Google Scholar] [CrossRef]
- Riss, T.L.; Moravec, R.A.; Niles, A.L.; Duellman, S.; Benink, H.A.; Worzella, T.J.; Minor, L. Cell Viability Assays. In Assay Guidance Manual [Internet]; Eli Lilly & Company and the National Center for Advancing Translational Sciences: Bethesda, MD, USA, 2016. [Google Scholar]
- Salah, B.A. Staining HAEMATOXYLIN & EOSIN; Research Gate: Berlin, Germany, 2020. [Google Scholar]
- Shettles, L.B. Quinacrine Fluorescence of the Human Y Chromosome. Nature 1971, 230, 52. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.L.B.; Lima, M.R.F.; Conserva, L.M.; Andrade, V.S.; Rocha, E.M.M.; Lemos, R.P.L. Studies on the antimicrobial activity and brine shrimp toxicity of Zeyheria tuberculosa (Vell.) Bur. (Bignoniaceae) extracts and their main constituents. Ann. Clin. Microbiol. Antimicrob. 2009, 8, 6. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Z.; Zou, J.; Li, Q. Allelopathic efects of sesame extracts on seed germination of moso bamboo and identification of potential allelochemicals. Sci. Rep. 2022, 12, 6661. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.P.; Wei, W.; Zhai, W.T.; Chu, H.; Yin, J.L.; Bai, X.F.; Bu, Q.M. Effects of litter from dominant tree species on seed germination and seedling growth of exotic plant Rhus typhina in hilly areas in Shandong peninsula. Sci. Silvae Sin. 2016, 52, 28–34. [Google Scholar]
- Jiang, Z.; Yu, Q.; Qiao, M.; Xiao, J.; Zhang, Z.; Yin, H. Effects of root exudates from Picea asperata seedlings on the seed germination and seedling growth of two herb species. Sci. Silvae Sin. 2019, 55, 160–166. [Google Scholar]
- Williamson, G.B.; Richardson, D. Bioassays for allelopathy: Measuring treatment responses with independent controls. J. Chem. Ecol. 1998, 14, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Anderle, P.; Niederer, E.; Rubas, W.; Hilgendorf, C.; Spahn-Langguth, H.; Wunderli-Allenspach, H.; Merkle, H.P.; Langguth, P. P-Glycoprotein (P-gp) mediated efflux in Caco-2 cell monolayers: The influence of culturing conditions and drug exposure on P-gp expression levels. J. Pharm. Sci. 1998, 87, 757–762. [Google Scholar] [CrossRef] [PubMed]
- Tajudin, T.; Mat, N.; Siti-Aishah, A.; Yusran, A.; Alwi, A.; Ali, A. Cytotoxicity, Antiproliferative Effects, and Apoptosis Induction of Methanolic Extract of Cynometra cauliflora Linn. Whole Fruit on Human Promyelocytic Leukemia HL-60 Cells. Evid.-Based Complement. Altern. Med. 2012, 2012, 127373. [Google Scholar] [CrossRef] [PubMed]
- Wyllie, A.H. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 1980, 284, 555–556. [Google Scholar] [CrossRef] [PubMed]
- Nousis, L.; Doulias, P.-T.; Aligiannis, N.; Bazios, D.; Agalias, A.; Galaris, D.; Mitakou, S. DNA protecting and genotoxic effects ofolive oil related components in cells exposed to hydrogen peroxide. Free Radic. Res. 2005, 39, 787–795. [Google Scholar] [CrossRef]
- Rusnak, J.M.; Calmels, T.P.; Hoyt, D.G.; Kondo, Y.; Yalowich, J.C.; Lazo, J.S. Genesis of discrete higher order DNA fragments in apoptotic human prostatic carcinoma cells. Mol. Pharmacol. 1996, 49, 244–252. [Google Scholar]
- Mahassni, S.H.; Al-Reemi, R.M. Apoptosis and necrosis of human breast cancer cells by an aqueous extract of garden cress (Lepidium sativum) seeds. Saudi J. Biol. Sci. 2013, 20, 131–139. [Google Scholar] [CrossRef]
- Eskandari, E.; Eaves, C.J. Paradoxical roles of caspase-3 in regulating cell survival, proliferation, and tumorigenesis. J. Cell Biol. 2022, 221, e202201159. [Google Scholar] [CrossRef] [PubMed]
- Julien, O.; Wells, J.A. Caspases and their substrates. Cell Death Differ. 2017, 24, 1380–1389. [Google Scholar] [CrossRef] [PubMed]
- Sakamaki, K.; Satou, Y. Caspases: Evolutionary aspects of their functions in vertebrates. J. Fish. Biol. 2009, 74, 727–753. [Google Scholar] [CrossRef] [PubMed]
- Enari, M.; Sakahira, H.; Yokoyama, H.; Okawa, K.; Iwamatsu, A.; Nagata, S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 1998, 391, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Jänicke, R.U.; Sprengart, M.L.; Wati, M.R.; Porter, A.G. Caspase-3 Is Required for DNA Fragmentation and Morphological Changes Associated with Apoptosis. J. Biol. Chem. 1998, 273, 9357–9360. [Google Scholar] [CrossRef] [PubMed]
- Hampton, M.B.; Fadeel, B.; Orrenius, S. Redox Regulation of the Caspases during Apoptosis. N. Y. Acad. Sci. 2006, 854, 328–335. [Google Scholar] [CrossRef]
- Murphy, K.M.; Ranganathan, V.; Farnsworth, M.L.; Kavallaris, M.; Lock, R.B. Bcl-2 inhibits Bax translocation from cytosol to mitochondria during drug-induced apoptosis of human tumor cells. Cell Death Differ. 2000, 7, 102–111. [Google Scholar] [CrossRef]
- Shinoura, N.; Yoshida, Y.; Nishimura, M.; Muramatsu, Y.; Asai, A.; Kirino, T.; Hamada, H. Expression level of Bcl-2 determines anti- or proapoptotic function. Cancer Res. 1999, 59, 4119–4128. [Google Scholar] [PubMed]
- Annis, M.G.; Soucie, E.L.; Dlugosz, P.J.; Cruz-Aguado, J.A.; Penn, L.Z.; Leber, B.; Andrews, D.V. Bax forms multispanning monomers that oligomerize to permeabilize membranes during apoptosis. EMBO J. 2005, 24, 2096–2103. [Google Scholar] [CrossRef]
- Wood, D.; Newcomb, E. Cleavage of Bax enhances its cell death function. Exp. Cell Res. 2000, 256, 375–382. [Google Scholar] [CrossRef]
- Wei, M.; Zong, W.; Cheng, E.; Lindsten, T.; Panoutsakopoulou, V.; Ross, A.; Roth, K.; MacGregor, G.; Thompson, C.; Korsmeyer, S. Proapoptotic BAX and BAK: A Requisite Gateway to Mitochondrial Dysfunction and Death. Science 2001, 292, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; Jockel, J.; Wei, M.; Korsmeyer, S. Enforced dimerization of BAX results in its translocation, mitochondrial dysfunction and apoptosis. EMBO J. 1998, 17, 3878–3885. [Google Scholar] [CrossRef] [PubMed]
- Selvakumaran, M.; Lin, H.; Miyashita, T.; Wang, H.G.; Krajewski, S.; Reed, J.C.; Hoffman, B.; Liebermann, D. Immediate early up-regulation of bax expression by p53 but not TGF beta 1: A paradigm for distinct apoptotic pathways. Oncogene 1994, 9, 1791–1798. [Google Scholar] [PubMed]
- Zeng, R.S.; Mallik, A.U.; Luo, S.M. Allelopathy in Sustainable Agriculture and Forestry; Springer Science Business Media: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A. In vitro assessment of allelopathic potential of olive processing waste on maize (Zea mays L.). Egypt. Soc. Exp. Biol. 2013, 9, 35–39. [Google Scholar]
- Li, Z.-H.; Wang, Q.; Ruan, X.; Pan, C.-D.; Jiang, D.-A. Phenolics and Plant Allelopathy. Molecules 2010, 15, 8933–8952. [Google Scholar] [CrossRef]
- Kelepesi, S.; Tzortzakis, N.G. Olive Mill Wastes—A Growing Medium Component for Seedling and Crop Production of Lettuce and Chicory. Int. J. Veg. Sci. 2009, 15, 325–339. [Google Scholar] [CrossRef]
- Naman, M. Effects of raw and treated olive mill wastewater (OMW) by coagulation-flocculation, on the germination and the growth of three plant species (wheat, white beans, lettuce). Moroc. J. Chem. 2019, 7, 111–122. [Google Scholar]
- Ouzounidou, G.; Ntougias, S.; Asfi, M.; Zervakis, G. Raw and fungal-treated olive-mill wastewater effects on selected parameters of lettuce (Lactuca sativa L.) growth—The role of proline. J. Environ. Sci. Health 2012, 47, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Masoko, P. Chapter 9—In Vitro Cytotoxicity Tests of the Developed Extracts; University of Pretoria: Hatfield, UK, 2007. [Google Scholar]
- Geran, R.I.; Greenberg, H.M.; McDonald, M.; Abbott, B.J. Protocols for screening chemical agents and natural products against animal tumors and other biological systems. Cancer Chemoth Rep. 1972, 33, 1–17. [Google Scholar]
- Márquez, K.; Márquez, N.; Ávila, F.; Cruz, N.; Burgos-Edwards, A.; Pardo, X.; Carrasco, B. Oleuropein-Enriched Extract From Olive Mill Leaves by Homogenizer-Assisted Extraction and Its Antioxidant and Antiglycating Activities. Front. Nutr. 2022, 9, 895070. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Guyot, S.; Marnet, N.; Lopes-da-Silva, J.A.; Silva, A.M.; Renard, C.M.; Coimbra, M.A. Identification of oleuropein oligomers in olive pulp and pomace. J. Sci. Food Agric. 2006, 86, 1495–1502. [Google Scholar] [CrossRef]
- Sengling Cebin Coppa, C.F.; Rosim, R.E.; Cardim de Oliveira, C.A.; Rodrigues, E.D.C.R.; Gonçalves, C.B. Extraction of oleuropein from olive leaves using a hydroalcoholic solvent. Braz. J. Food Technol. 2017, 20, e2016169. [Google Scholar]
- Obied, H.; Allen, M.; Bedgood, P.; Prenzler, P.; Robards, K. Investigation of Australian olive mill waste for recovery of biophenols. J. Agric. Food Chem. 2005, 53, 9911–9920. [Google Scholar] [CrossRef] [PubMed]
- Feki, M.; Allouche, N.; Bouazi, M.; Gargoubi, A.; Sayadi, S. Effect of storage of olive mill wastewaters on hydroxytyrosol concentration. Eur. J. Lipid Sci. Technol. 2006, 108, 1021–1027. [Google Scholar] [CrossRef]
- Leouifoudi, I.; Harnafi, H.; Zyad, A. Olive Mill Waste Extracts: Polyphenols Content, Antioxidant, and Antimicrobial Activities. Adv. Pharmacol. Sci. 2015, 2015, 714138. [Google Scholar] [CrossRef]
- Tzathas, K.; Chrysagi, E.; Lyberatos, G.; Vlyssides, A.; Vlysidis, A. Pretreatment of Olive Mill Wastes for the Extraction of Residual Oil and High Added Value Compounds. Waste Biomass Valorization 2019, 11, 4025–4034. [Google Scholar] [CrossRef]
- Lafka, T.-L.; Lazou, A.; Sinanoglou, V.J.; Lazos, E.S. Phenolic and antioxidant potential of olive oil mill wastes. Food Chem. 2011, 125, 92–98. [Google Scholar] [CrossRef]






| Allelopathy | |||||||
|---|---|---|---|---|---|---|---|
| Sample | Concentration (μg/mL) | Germination Rate (%) | Germination Energy (%) | Germination Index | Plumule (mm) | Root (mm) | Ratio |
| Control | / | 90.00 ± 0.36 | 43.33 ± 2.87 | 13.72 ± 0.35 | 14.20 ± 4.0 | 40.95 ± 8.72 | 3.01 ± 0.79 |
| Bidni Methanol | 500 | 72.08 ± 6.29 | 50.00 ± 13.23 | 8.31 ± 1.93 | 13.41 ± 3.66 | 35.84 ± 9.04 | 2.86 ± 1.00 |
| 100 | 84.17 ± 2.59 | 66.67 ± 2.89 | 10.44 ± 0.93 | 12.90 ± 2.29 | 32.57 ± 8.23 | 2.61 ± 0.84 | |
| 10 | 84.38 ± 2.61 | 73.33 ± 2.89 | 10.38 ± 1.40 | 10.40 ± 1.65 | 37.22 ± 7.69 | 3.63 ± 0.90 | |
| 1 | 84.58 ± 2.77 | 68.33 ± 17.56 | 10.69 ± 2.15 | 10.34 ± 3.80 | 31.76 ± 7.08 | 3.45 ± 1.41 | |
| Bidni Acid | 500 | 64.47 ± 2.28 | 11.67 ± 2.89 | 6.13 ± 0.72 | 13.72 ± 7.44 | 31.80 ± 19.97 | 2.30 ± 0.99 |
| 100 | 81.67 ± 2.04 | 56.67 ± 7.64 | 9.80 ± 0.64 | 16.18 ± 6.70 | 37.39 ± 20.25 | 2.38 ± 1.12 | |
| 10 | 86.25 ± 1.66 | 71.67 ± 11.55 | 10.73 ± 0.74 | 13.58 ± 4.34 | 26.06 ± 8.52 | 2.14 ± 1.06 | |
| 1 | 86.67 ± 1.98 | 80.00 ± 10.00 | 10.78 ± 1.30 | 9.97 ± 3.65 | 33.55 ± 8.38 | 3.83 ± 1.83 | |
| Bidni Alkaline | 500 | 70.21 ± 3.16 | 21.67 ± 16.07 | 7.65 ± 0.50 | 10.12 ± 2.01 | 29.32 ± 11.15 | 5.75 ± 4.39 |
| 100 | 90.21 ± 1.51 | 51.67 ± 7.64 | 13.23 ± 1.50 | 11.58 ± 4.13 | 41.08 ± 17.72 | 3.72 ± 1.54 | |
| 10 | 95.42 ± 0.81 | 23.33 ± 7.64 | 16.78 ± 1.08 | 10.31 ± 4.06 | 54.04 ± 13.47 | 5.61 ± 1.49 | |
| 1 | 93.75 ± 1.30 | 23.33 ± 12.58 | 16.06 ± 1.54 | 9.89 ± 3.98 | 41.19 ± 19.36 | 4.90 ± 3.33 | |
| Bajda Methanol | 500 | 69.79 ± 2.85 | 31.67 ± 7.64 | 8.15 ± 1.24 | 5.00 ± 0.84 | 16.26 ± 6.68 | 3.26 ± 1.22 |
| 100 | 81.46 ± 3.30 | 56.67 ± 11.55 | 10.92 ± 0.61 | 9.47 ± 4.12 | 36.86 ± 12.52 | 4.46 ± 2.05 | |
| 10 | 82.71 ± 3.12 | 55.00 ± 8.66 | 11.00 ± 1.46 | 11.47 ± 2.68 | 35.00 ± 10.49 | 3.11 ± 0.89 | |
| 1 | 86.04 ± 1.15 | 65.00 ± 10.00 | 11.08 ± 0.13 | 9.64 ± 3.74 | 30.83 ± 6.93 | 3.54 ± 1.25 | |
| Bajda Acid | 500 | 59.58 ± 6.93 | 11.67 ± 7.64 | 5.76 ± 1.35 | 8.15 ± 1.63 | 45.38 ± 14.27 | 5.69 ± 1.81 |
| 100 | 76.88 ± 7.26 | 58.33 ± 15.28 | 9.79 ± 2.46 | 9.85 ± 4.49 | 46.60 ± 16.25 | 5.44 ± 2.31 | |
| 10 | 83.54 ± 1.87 | 53.33 ± 10.41 | 10.95 ± 1.04 | 7.88 ± 2.09 | 42.56 ± 9.37 | 5.79 ± 2.19 | |
| 1 | 84.79 ± 2.76 | 50.00 ± 15.00 | 11.77 ± 1.04 | 7.58 ± 2.69 | 29.87 ± 11.78 | 4.29 ± 2.22 | |
| Bajda Alkaline | 500 | 49.79 ± 3.41 | 3.33 ± 2.89 | 4.92 ± 1.11 | 5.03 ± 2.77 | 33.79 ± 23.61 | 8.25 ± 8.97 |
| 100 | 86.04 ± 3.05 | 36.67 ± 12.58 | 12.90 ± 1.46 | 8.57 ± 4.05 | 57.72 ± 26.17 | 7.16 ± 2.95 | |
| 10 | 93.75 ± 2.46 | 31.67 ± 16.07 | 15.87 ± 2.68 | 7.47 ± 2.38 | 41.53 ± 9.83 | 6.00 ± 1.96 | |
| 1 | 94.38 ± 2.00 | 28.33 ± 18.93 | 16.41 ± 1.42 | 11.95 ± 4.43 | 43.35 ± 13.30 | 4.02 ± 1.49 | |
| Malti Methanol | 500 | 48.75 ± 2.45 | 18.33 ± 2.89 | 4.87 ± 0.49 | 4.46 ± 1.35 | 10.75 ± 8.79 | 2.26 ± 1.58 |
| 100 | 71.04 ± 4.56 | 41.67 ± 7.64 | 8.22 ± 1.49 | 7.79 ± 3.42 | 39.28 ± 19.77 | 5.29 ± 3.02 | |
| 10 | 84.79 ± 3.85 | 60.00 ± 13.23 | 11.46 ± 2.24 | 15.01 ± 8.33 | 29.63 ± 12.66 | 2.46 ± 1.69 | |
| 1 | 85.42 ± 0.79 | 66.67 ± 2.89 | 10.53 ± 0.93 | 10.11 ± 2.08 | 25.34 ± 5.11 | 2.62 ± 0.80 | |
| Malti Acid | 500 | 61.04 ± 3.28 | 26.67 ± 18.93 | 6.07 ± 0.98 | 11.44 ± 4.20 | 22.94 ± 11.17 | 2.08 ± 0.86 |
| 100 | 79.17 ± 3.41 | 61.67 ± 12.58 | 9.38 ± 1.51 | 12.64 ± 3.51 | 41.24 ± 14.60 | 3.41 ± 1.24 | |
| 10 | 86.88 ± 1.82 | 68.33 ± 7.64 | 11.44 ± 1.17 | 17.32 ± 6.24 | 41.29 ± 10.40 | 2.81 ± 1.53 | |
| 1 | 85.21 ± 1.27 | 73.33 ± 12.58 | 10.87 ± 0.14 | 13.33 ± 4.79 | 36.02 ± 11.14 | 2.89 ± 1.14 | |
| Malti Alkaline | 500 | 26.67 ± 5.68 | 0.01 ± 0.00 | 2.32 ± 0.90 | 4.97 ± 1.98 | 13.57 ± 8.12 | 3.22 ± 1.96 |
| 100 | 75.83 ± 1.69 | 38.33 ± 5.77 | 8.64 ± 0.67 | 8.12 ± 3.95 | 43.88 ± 19.57 | 5.97 ± 2.90 | |
| 10 | 88.75 ± 2.31 | 35.00 ± 13.23 | 13.64 ± 1.14 | 12.13 ± 2.17 | 46.26 ± 9.64 | 3.98 ± 1.34 | |
| 1 | 92.71 ± 1.49 | 38.33 ± 5.77 | 15.01 ± 1.29 | 12.12 ± 3.23 | 30.79 ± 8.67 | 2.70 ± 1.05 | |
| % Mortality of Shrimp Nauplii | |||||||
|---|---|---|---|---|---|---|---|
| Sample | Concentration (μg/mL) | Number of Surviving Nauplii (After 24 h) | Total Number of Nauplius Survivors | % Mortality | LD50 (μg/mL) | ||
| T1 | T2 | T3 | |||||
| Bidni Methanol | 500 | 2 | 3 | 3 | 8 | 73.9 ± 6.8 | 14. 6 ± 6.6 |
| 50 | 6 | 4 | 3 | 13 | 59.7 ± 12.7 | ||
| 10 | 4 | 5 | 4 | 13 | 56.7 ± 5.8 | ||
| 0.1 | 7 | 8 | 8 | 23 | 25.5 ± 9.4 | ||
| Bidni Acid | 500 | 0 | 0 | 0 | 0 | 100.0 ± 0.0 | 2.9 ± 1.6 |
| 50 | 2 | 3 | 3 | 8 | 74.2 ± 5.2 | ||
| 10 | 4 | 5 | 5 | 14 | 53.4 ± 3.0 | ||
| 0.1 | 7 | 8 | 6 | 21 | 32.1 ± 10.7 | ||
| Bidni Alkaline | 500 | 0 | 0 | 0 | 0 | 100.0 ± 0.0 | 3.6 ± 1.6 |
| 50 | 2 | 2 | 4 | 8 | 75.2 ± 10.0 | ||
| 10 | 6 | 7 | 6 | 19 | 38.5 ± 7.8 | ||
| 0.1 | 6 | 8 | 6 | 20 | 28.1 ± 15.1 | ||
| Bajda Methanol | 500 | 3 | 2 | 2 | 7 | 76.7 ± 5.8 | 13.0 ± 1.3 |
| 50 | 3 | 3 | 3 | 9 | 70.0 ± 0.0 | ||
| 10 | 4 | 6 | 4 | 14 | 51.9 ± 10.5 | ||
| 0.1 | 7 | 8 | 8 | 23 | 25.8 ± 5.2 | ||
| Bajda Acid | 500 | 0 | 0 | 0 | 0 | 100.0 ± 0.0 | 4.4 ± 0.6 |
| 50 | 3 | 5 | 4 | 12 | 60.0 ± 10.0 | ||
| 10 | 8 | 6 | 7 | 21 | 34.5 ± 6.6 | ||
| 0.1 | 7 | 7 | 8 | 22 | 26.7 ± 5.8 | ||
| Bajda Alkaline | 500 | 0 | 1 | 0 | 1 | 96.7 ± 5.8 | 2.2 ± 0.8 |
| 50 | 2 | 1 | 1 | 4 | 87.3 ± 4.7 | ||
| 10 | 3 | 4 | 3 | 10 | 68.8 ± 4.7 | ||
| 0.1 | 7 | 5 | 7 | 19 | 36.7 ± 11.5 | ||
| Malti Methanol | 500 | 1 | 0 | 0 | 1 | 97.2 ± 4.8 | 3.8 ± 1.6 |
| 50 | 4 | 3 | 2 | 9 | 72.9 ± 8.4 | ||
| 10 | 5 | 4 | 6 | 15 | 52.7 ± 11.9 | ||
| 0.1 | 5 | 8 | 8 | 21 | 25.3 ± 4.6 | ||
| Malti Acid | 500 | 1 | 0 | 0 | 1 | 96.7 ± 5.8 | 6.4 ± 4.6 |
| 50 | 5 | 6 | 5 | 16 | 46.7 ± 5.8 | ||
| 10 | 4 | 5 | 6 | 15 | 42.9 ± 15.9 | ||
| 0.1 | 8 | 6 | 5 | 19 | 36.7 ± 15.3 | ||
| Malti Alkaline | 500 | 0 | 0 | 0 | 0 | 100.0 ± 0.0 | 2.7 ± 1.6 |
| 50 | 2 | 1 | 0 | 3 | 90.6 ± 9.1 | ||
| 10 | 6 | 7 | 6 | 19 | 36.7 ± 5.8 | ||
| 0.1 | 6 | 8 | 6 | 20 | 35.2 ± 13.4 | ||
| Compound | RRT | Malti Methanol | Malti Acid | Malti Alkaline | Bajda Methanol | Bajda Acid | Bajda Alkaline | Bidni Methanol | Bidni Acid | Bidni Alkaline | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 25 | 5,7-dihydroxy flavone | 3.78 | 10.091 ± 0.048 | 9.631 ± 0.060 | 9.678 ± 0.044 | <LOD | 9.955 ± 0.164 | 6.916 ± 0.547 | 1.641 ± 0.030 | < LOD | < LOD |
| 24 | Quercetin | 3.05 | 2.225 ± 0.005 | <LOD | <LOD | 2.064 ± 0.001 | 2.032 ± 0.007 | 3.003 ± 0.018 | <LOD | <LOD | 2.157 ± 0.005 |
| 23 | 3′,4,5,7-tetreahydroxy flavone | 2.96 | 3.572 ± 0.003 | <LOD | <LOD | 3.551 ± 0.001 | 3.782 ± 0.186 | 3.791 ± 0.002 | <LOD | <LOD | <LOD |
| 22 | Ligstorside | 2.92 | 1.002 ± 0.011 | <LOD | <LOD | 1.303 ± 0.013 | 2.254 ± 0.540 | 3.010 ± 0.011 | 0.286 ± 0.006 | <LOD | 1.619 ± 0.014 |
| 21 | Oleocanthal | 2.94 | 1.359 ± 0.022 | ND | 2.028 ± 0.089 | 1.284 ± 0.069 | 7.070 ± 0.235 | <LOD | <LOD | <LOD | 11.713 ± 0.368 |
| 20 | Oleuoside | 2.85 | 5.089 ± 0.046 | 5.081 ± 0.049 | 1.220 ± 0.034 | <LOD | 3.563 ± 0.622 | 1.668 ± 0.040 | <LOD | 1.990 ± 0.066 | 19.292 ± 0.122 |
| 19 | Oleuropein | 2.69 | 0.858 ± 0.010 | <LOD | <LOD | 6.265 ± 0.042 | 8.916 ± 0.837 | 1.137 ± 0.031 | 2.398 ± 0.240 | <LOD | 3.823 ± 0.021 |
| 18 | Oleacein | 2.54 | 13.518 ± 0.980 | <LOD | 12.199 ± 0.269 | 11.085 ± 0.021 | 18.345 ± 0.054 | 3.816 ± 0.027 | 5.651 ± 0.126 | <LOD | 9.828 ± 0.038 |
| 17 | Salicylic acid | 2.44 | 2.590 ± 0.019 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD |
| 16 | Apigenin 7-glucoside | 2.41 | 3.282 ± 0.009 | <LOD | >LOD–< LOQ | 8.109 ± 0.178 | 1.518 ± 0.036 | 2.145 ± 0.010 | 3.343 ± 0.129 | >LOD–< LOQ | <LOD |
| 15 | Luteolin 7-glucoside | 2.34 | <LOD | >LOD–< LOQ | <LOD | >LOD–< LOQ | 1.735 ± 0.066 | >LOD–< LOQ | <LOD | <LOD | 12.849 ± 0.046 |
| 14 | Ellagic acid | 2.22 | 0.699 ± 0.024 | <LOD | <LOD | 0.218 ± 0.001 | 1.971 ± 0.021 | <LOD | <LOD | <LOD | >LOD–< LOQ |
| 13 | 2-hydroxy-cinnamic acid (trans) | 2.19 | >LOD–< LOQ | >LOD–< LOQ | >LOD–< LOQ | >LOD–< LOQ | >LOD–< LOQ | >LOD -LOQ | >LOD–< LOQ | >LOD–< LOQ | >LOD–< LOQ |
| 12 | Ferulic acid | 1.91 | <LOD | <LOD | >LOD–< LOQ | <LOD | <LOD | <LOD | <LOD | <LOD | 2.394 ± 0.008 |
| 11 | 3-methoxy-4-hydroxy cinnamic acid | 1.78 | 1.055 ± 0.009 | <LOD | 0.974 ± 0.033 | <LOD | ND | >LOD -LOQ | <LOD | <LOD | 1.436 ± 0.068 |
| 10 | p-coumaric acid | 1.44 | 4.724 ± 0.042 | 2.188 ± 0.011 | 6.219 ± 0.018 | 2.348 ± 0.013 | 3.391 ± 0.018 | 11.358 ± 0.046 | 1.927 ± 0.007 | 2.067 ± 0.010 | 22.046 ± 0.056 |
| 9 | Vanillin | 1.22 | 4.103 ± 0.001 | 4.851 ± 0.007 | 4.106 ± 0.002 | 4.084 ± 0.001 | 7.641 ± 0.027 | 4.205 ± 0.012 | <LOD | 4.194 ± 0.005 | 4.655 ± 0.004 |
| 7 | 2,4 dihydroxy benzoic acid | 0.82 | <LOD | <LOD | <LOD | <LOD | 1.900 ± 0.005 | <LOD | <LOD | <LOD | <LOD |
| 6 | Vanillic acid | 0.78 | 1.688 ± 0.302 | 1.191 ± 0.010 | 1.570 ± 0.004 | 2.011 ± 0.013 | 4.153 ± 0.027 | 2.221 ± 0.023 | <LOD | 1.544 ± 0.002 | 5.513 ± 0.184 |
| 5 | Catechin | 0.66 | <LOD | <LOD | <LOD | <LOD | 10.332 ± 0.277 | <LOD | <LOD | <LOD | <LOD |
| 4 | 4-hydroxy-benzoic acid | 0.54 | 2.395 ± 0.009 | <LOD | 2.359 ± 0.004 | <LOD | 3.980 ± 0.731 | 2.458 ± 0.042 | <LOD | 2.317 ± 0.001 | 2.882 ± 0.011 |
| 3 | 2-(-4-hydroxy phenyl) ethanol | 0.51 | 11.159 ± 0.075 | 8.482 ± 0.011 | 11.305 ± 0.011 | 3.763 ± 0.010 | 8.959 ± 0.808 | 9.431 ± 0.124 | 3.608 ± 0.019 | 7.321 ± 0.081 | 43.837 ± 0.105 |
| 2 | 3 Hydroxytyrosol | 0.31 | 48.084 ± 0.035 | 4.699 ± 0.075 | 10.559 ± 0.032 | 6.034 ± 0.044 | 1.050 ± 0.063 | 32.838 ± 0.123 | 6.801 ± 0.312 | 2.565 ± 0.048 | 188.819 ± 0.341 |
| 1 | Gallic Acid | 0.26 | 0.480 ± 0.001 | 0.676 ± 0.008 | 0.785 ± 0.006 | 0.505 ± 0.006 | 7.136 ± 0.030 | 0.792 ± 0.053 | <LOD | 0.494 ± 0.001 | 0.484 ± 0.004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lia, F.; Attard, K. Bioactive Potential of Olive Mill Waste Obtained from Cultivars Grown in the Island of Malta. Foods 2024, 13, 1152. https://doi.org/10.3390/foods13081152
Lia F, Attard K. Bioactive Potential of Olive Mill Waste Obtained from Cultivars Grown in the Island of Malta. Foods. 2024; 13(8):1152. https://doi.org/10.3390/foods13081152
Chicago/Turabian StyleLia, Frederick, and Karen Attard. 2024. "Bioactive Potential of Olive Mill Waste Obtained from Cultivars Grown in the Island of Malta" Foods 13, no. 8: 1152. https://doi.org/10.3390/foods13081152