3.1. Modification of Nanosilica
Silica with abundant silanol exposed on the surface was synthesized using the vapor phase method, which can provide functionalization sites for surface modification. Some silane coupling agents such as KH–570 can easily react with silanol to form a stable hydrophobic tail. The modified silica has a specific hydrophile–lipophile balance (HLB) [
41]. When adjusting the quantity ratio of hydrophobic agents and raw silica, the modified silica showed various dispersion characteristics under different solutions. With the hydrophobic groups partly modified on the surface of the silica, dispersion characteristics were significantly improved, promoting the subsequent synthesis of TiO
2−SiO
2 nanoparticles.
The modified nanosilica was characterized by Fourier transform infrared spectroscopy with the Thermofisher Nicolet iS5, which is from the Thermo Fisher Scientific (Shanghai, China). The experimental results are shown in
Figure 2.
Figure 2 displays the infrared spectra of nanosilica modified with KH–570, SS, and TMCS. Clearly, the characteristic peaks of modified hydrophobic nanosilica were slightly weakened with the shift in the silanol absorption peak (with a wave number of 2800 cm
−1), except for the TMCS-modified nanoparticles [
42]. There are significant shape differences between the modified and unmodified nanosilica at the range of 1750 to 500 cm
−1. SS has a characteristic peak at 720 cm
−1, which indicates more than four adjacent –CH
2– groups [
34,
43], and a stretching vibration peak at 1300 cm
−1 belongs to a carbon skeleton of C–C. The more complex circumstance shown in the KH–570 spectrogram has a miscellaneous peak, resulting in a great contrast with the raw silica.
A surface morphology study was carried out with a Zeiss Merlin Compact scanning electron microscope and an Oxford AztecX–Max80 energy-dispersive spectrometer, both are supplied by Beijing Opton Optical Technology Co., Ltd. (Beijing, China), as depicted in
Figure 3. The electronic images of raw silica as well as the modified hydrophobic nanosilica were analyzed. Obviously, the raw nanosilica is liable to crosslinking, forming a large and continuous area. The distribution of modified nanosilica is significantly inconsistent. Hydrophobic modified nanosilica with high porosity shows a relatively longer distance between clusters.
The hydrophobicity of a modified material depends on the hydrophobic properties of the functional groups and the degree of functionalization (
Figure 4). TMCS, SS, and KH–570 have different chemical structures, which decide the degree of functionalization efficiency during the surface modification process with nanosilica. TMCS mainly undergoes a substitution reaction between Si–Cl and Silanol, generating volatile HCl. This reaction occurs more readily compared to the forward direction, making it easier to functionalize the surface of nanosilica. The long octadecyl hydrophobic chain of SS leads to significant hydrophobic modification. However, during surface modification, the chain tends to curl, wrapping the COO
− at the end of the tail and reducing functionalization efficiency. The Si(OH)
3 group on the modified end enables a single KH–570 molecule to simultaneously replace three silanol groups on the nanosilica surface with a higher substitution efficiency.
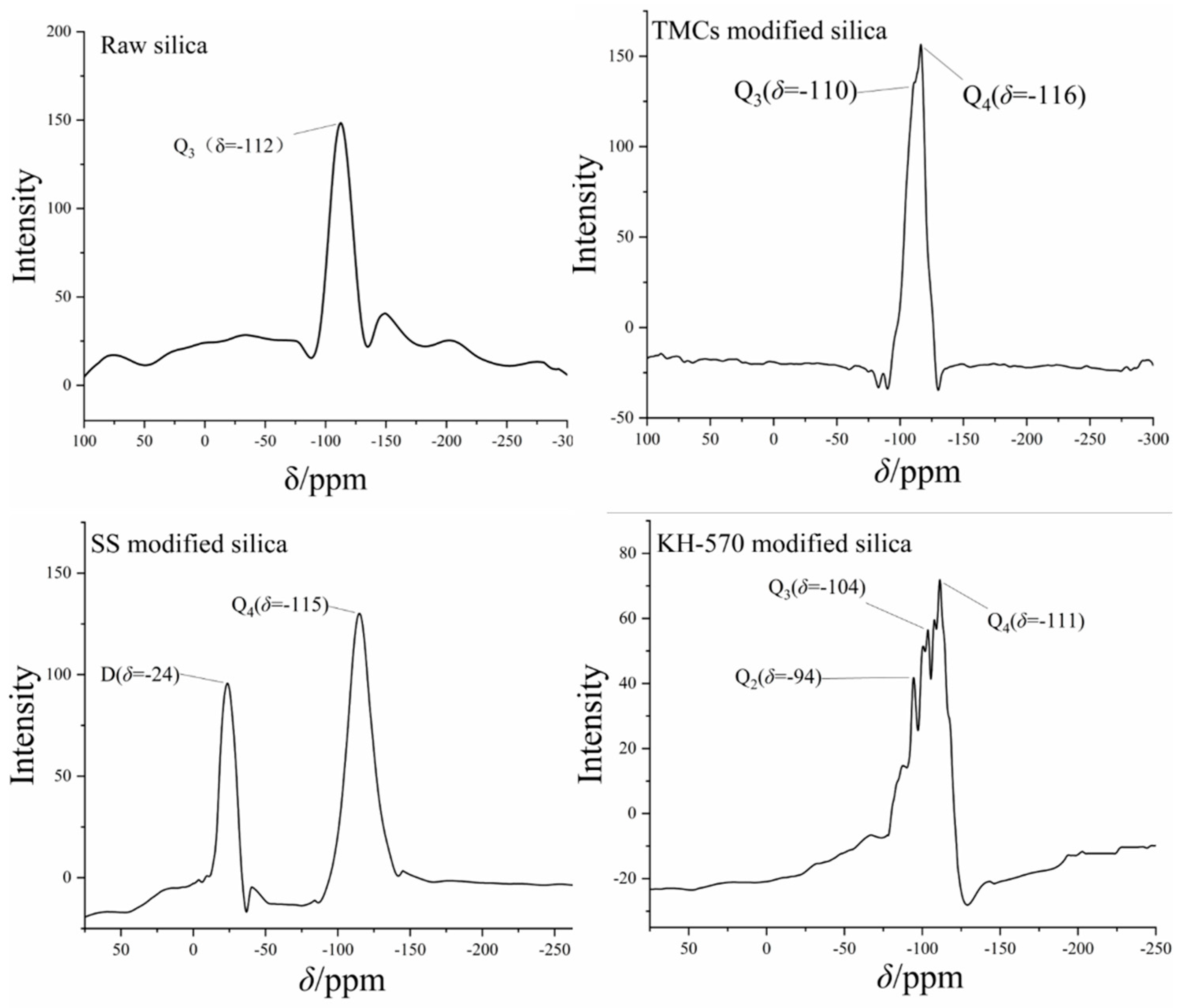
The silicon NMR spectra of three modified nanosilica materials, found by the Oxford MQR, are shown in
Figure 5. Chemical shifts in various nanosilica materials are decided by the number of bridging oxygen atoms in each material, which reflect the state of surface substituents [
11]. The appearance of Q2 and Q3 peaks indicates that the surface of silica has been modified with other functional groups. According to the spectrum results, KH–570 has a richer variety of peaks and has Q2, Q3, and Q4 peaks simultaneously, causing higher functionalization efficiency. Notably, TMCS shows Q3 and Q4 peaks, suggesting that the silica surface has only a single substitution structure, resulting in lower functionalization efficiency than KH–570. No substitution peak was detected in the SS silicon NMR spectrum, demonstrating that SS undergoes pore adsorption of nanosilica.
Further experiments were carried out to test the characteristic of hydrophobicity alongside the parameter of degree of lipophilicity. Methanol is a typical indicator used to evaluate lipophilicity. By counting different volumes of methanol consumption, the degree of lipophilicity can be calculated using the above formula. According to
Table 1, SS has the second-largest degree of lipophilicity at 11.50, while TMCS has the lowest degree of lipophilicity, with a value of 4.94. Thereby, KH–570-modified nanosilica shows the superior hydrophobic performance among the three materials, with a 13.64 degree of lipophilicity, which is consistent with the functionalization efficiency of the Si–NMR reaction.
In addition to maintaining the high specific surface area of nanoparticles, hydrophobic modification of nanosilica can also enhance the dispersion performance of particles in polar solvents. Since the surface of unmodified nanosilica is rich in silanol, the hydrogen bonding effect and the cross-linking effect between nanoparticles are markedly enhanced in polar solvents (such as water or alcohol solvents), affecting the dispersion of particles. Nanosilica with partial surface hydrophobic modification can use the remaining exposed silanol to form hydrogen bonds with solvent molecules, while the hydrophobic groups repel each other; thus, the particles are evenly dispersed in the solvent.
The same circumstance is shown in the sedimentation experiment. As listed in
Table 2, KH–570 has the best dispersion performance, with a sedimentation half-life of 13 min, and SS is the second best, with a sedimentation half-life of 10 min. TMCS has the lowest half-life, at 5 min. The half-lives of the three particles are, respectively, 2.5, 5, and 6.5 times that of the raw nanosilica, dramatically prolonging the duration of sedimentation.
3.2. SiO2–TiO2 Nanoparticle Characterization
Based on the above experimental results, KH–570 (15 wt%)-modified nanosilica possesses the best hydrophobic performance, high dispersity in solution, and can prevent aggregation. On the basis of this, a SiO2–TiO2 composite nanoparticle with a core−shell structure was prepared, and the particle’s characteristics were studied by IR, SEM, and a bench-scale UV degradation experimental test.
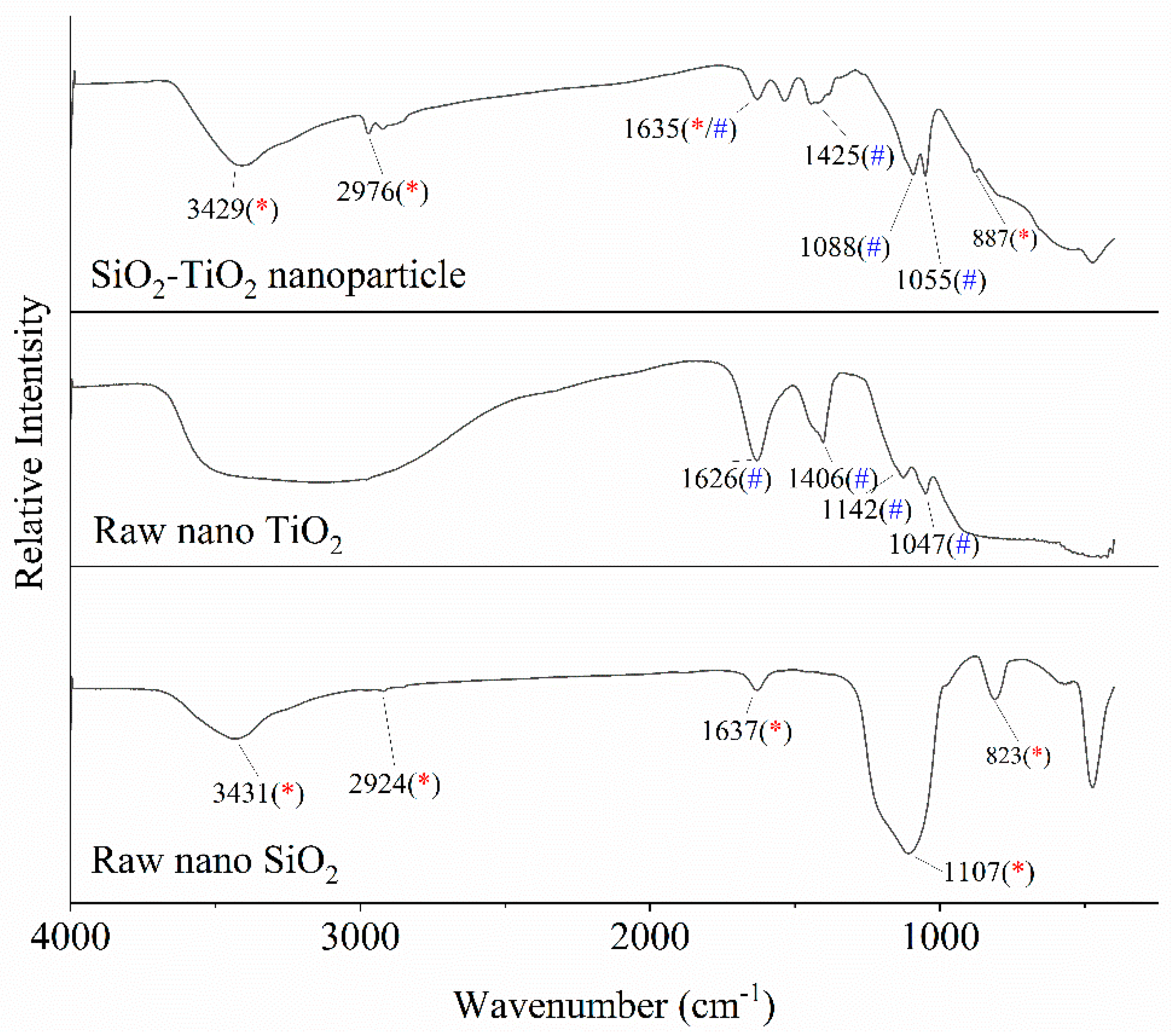
As the experimental results in
Figure 6 show, an overlap of peaks can be observed among the three spectrums in the wavelength range of 3500–2500, 1300–1000, and 850 cm
−1. It can be preliminarily judged that the TiO
2−SiO
2 nanoparticle contains characteristic groups of both SiO
2 and TiO
2 components [
34], which is consistent with the experimental results of SEM–EDS analysis shown in
Figure 7 and
Table 3.
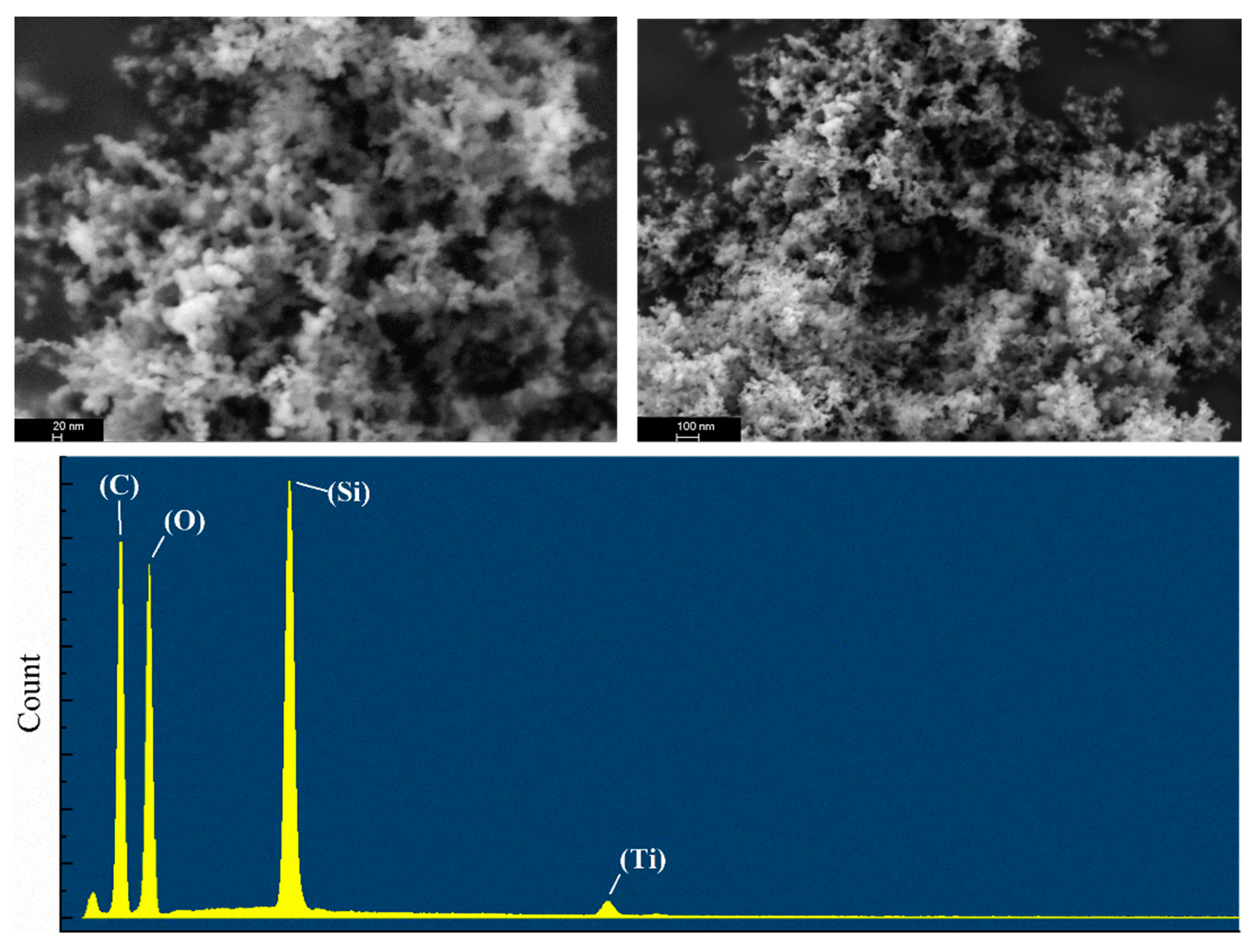
In contrast to raw silica, the SEM image in
Figure 7 shows that the SiO
2–TiO
2 nanoparticle has a porous structure, and the surface layer is covered with a continuously polished phase. According to EDS analysis, the surface layer of the particle was coated with silicon and uniformly trace amounts of titanium.
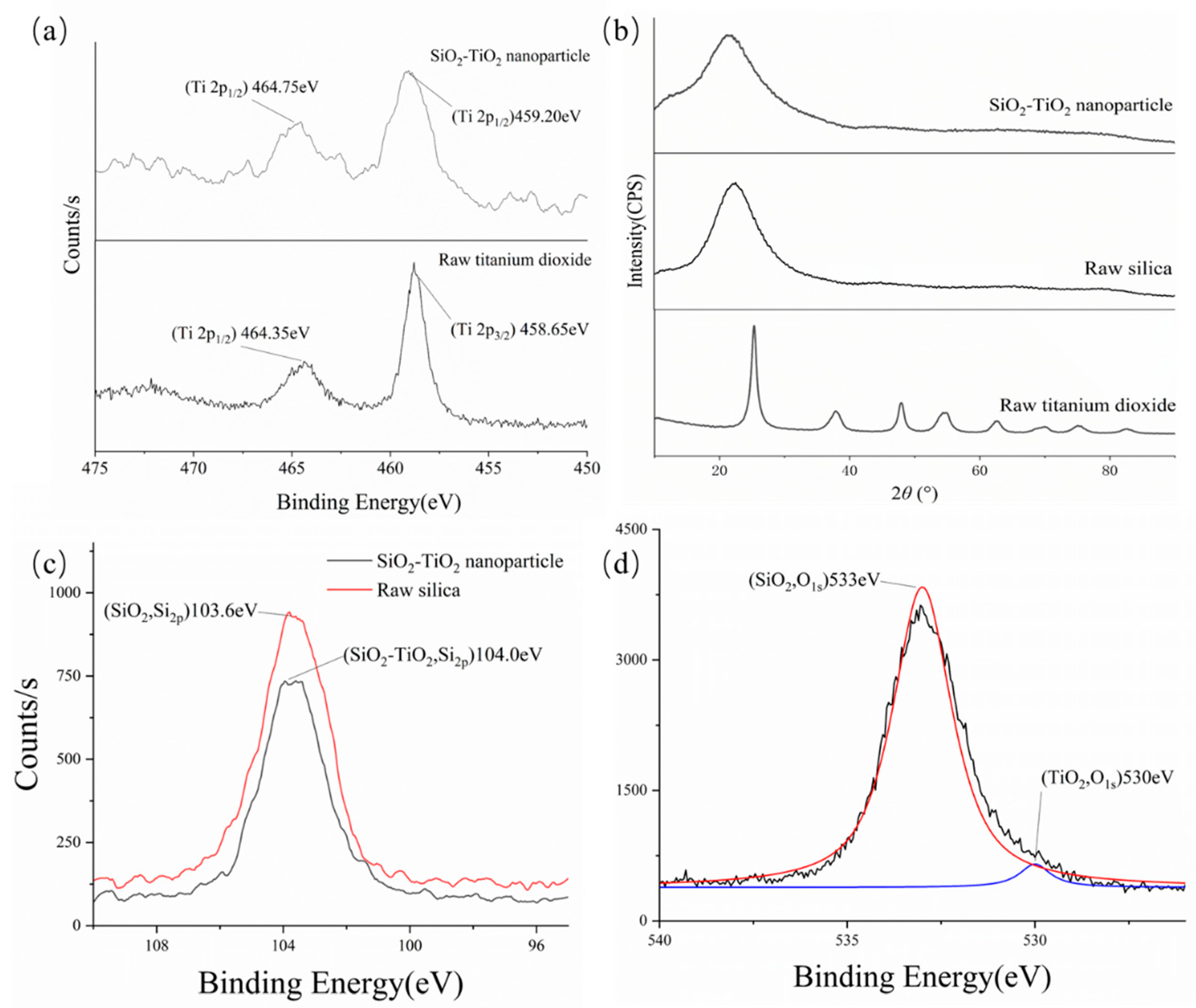
Figure 8b shows that no characteristic peak of TiO
2 occurred, which indicates that SiO
2 was still the dominant phase in the SiO
2–TiO
2 nanoparticles. From the XPS data in
Figure 8a,c,d, titanium and silicon can be simultaneously detected, but the positions of the binding energy peaks were slightly changed. In comparison with raw TiO
2, the electronic orbit of Ti 2p
3/2 was shifted by 0.55 eV. The increase in binding energy led to a higher bond dissociation energy in the Ti–O or Ti–Si covalent bonds, making the SiO
2–TiO
2 structure more stable; the photocatalytic process could also be carried out under higher-energy UV light instead of visible light, which improved the anti-interference ability of the ambient conditions. The binding energy of O
1s showed that most of the oxygen elements form covalent bonds with silicon in SiO
2–TiO
2 nanoparticles.
Figure 8d displays that the energy of O
1s (TiO
2) in the nanoparticles increased by circa 1.0 eV compared to raw TiO
2 (529.1 eV) [
44], and the enhanced Ti–O bonds may create more oxygen vacancies for electron transfer, thus improving photocatalytic efficiency [
45]. In summary, compared to raw TiO
2, SiO
2–TiO
2 needs UV light instead of visible light or simulated solar light. This change causes the materials to undergo photocatalysis under an external trigger, rather than unstable conditions. This characteristic also means that catalysis of foam constituents under solar irradiation can be avoided. Moreover, SiO
2–TiO
2 frameworks can provide more abundant active sites on the surface of silica for VOCs’ degradation, because of the stronger binding energy of Ti–O [
46,
47].
3.3. Aqueous Foam Stability
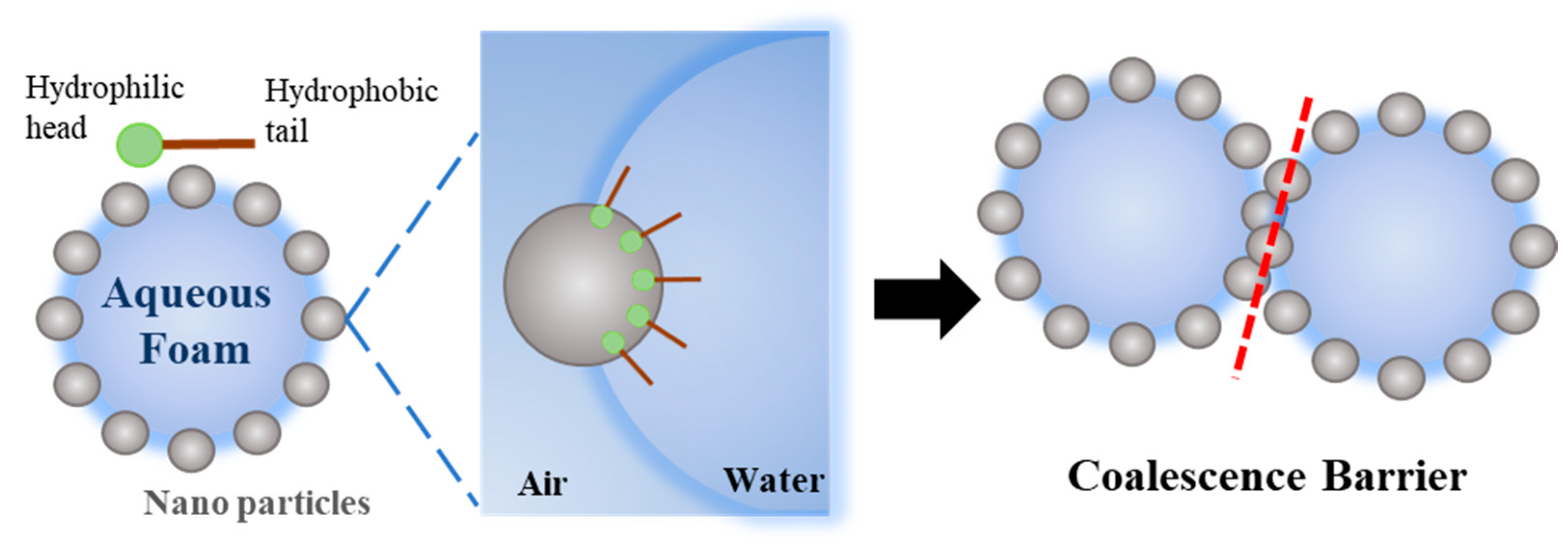
When modified nanoparticles are introduced into the stable foam stock solution, the particles will spontaneously move to the air-liquid interface, and the semisphere of particles will be exposed under the air phase due to the hydrophobic groups on the surface of the particles. The exposure area is related to the degree of hydrophobicity. This kind of self-assembled nanoparticle on the surface of the liquid film can form an agglomeration barrier to further prevent the liquid film between foams from coalescing (
Figure 9). As a result, the foam continues to maintain a fine size and restrain the Ostwald ripening process.
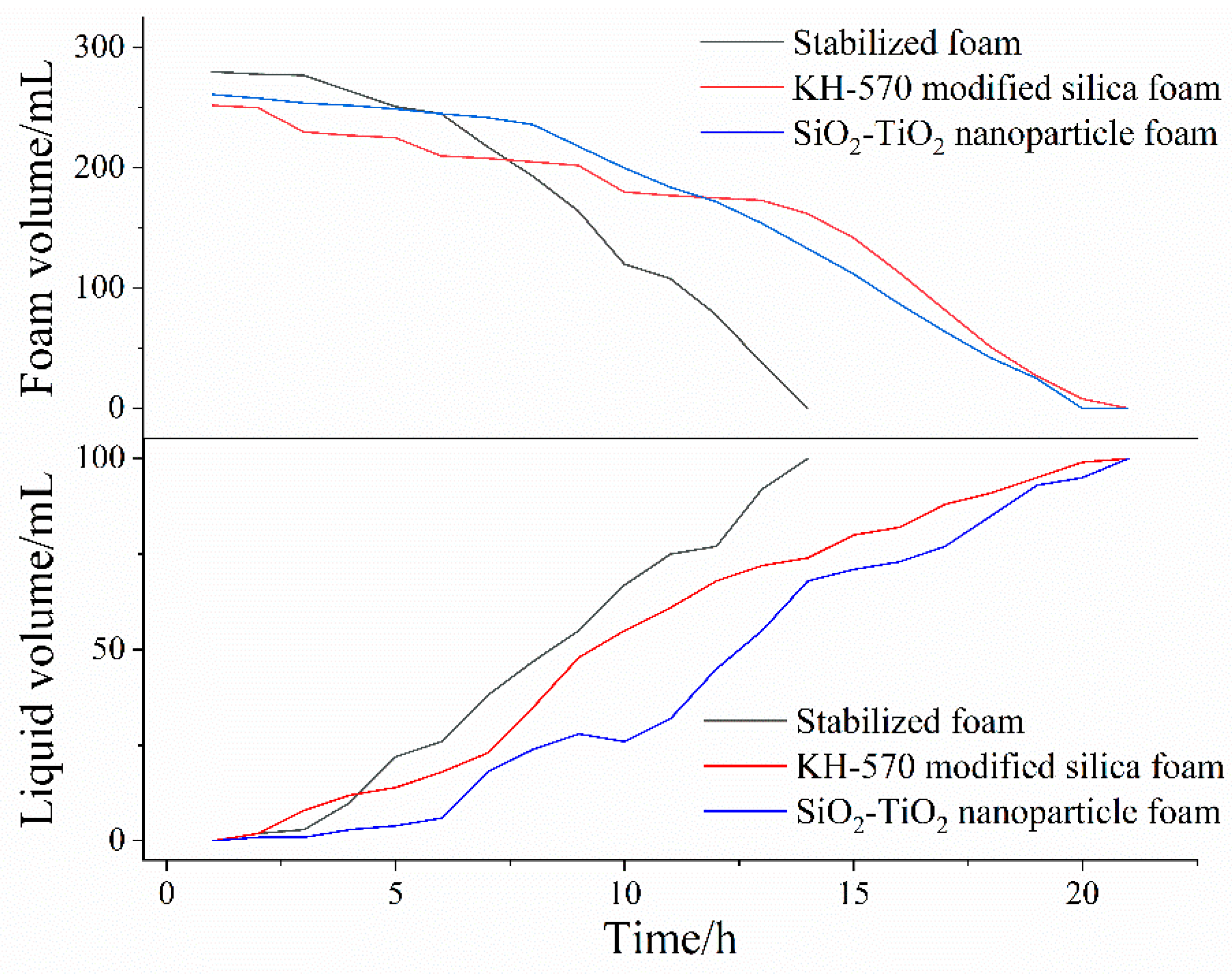
A Krüss DFA100–FSM dynamic foam analyzer provided by Krüss Scientific Instruments Co., Ltd. (Shanghai, China) was used to observe the morphology of aqueous foam as a function of time. In the results of foam dynamic analysis test, there were great morphological differences between unmodified and hydrophobically modified particle foams. As shown in
Figure 10, the liquid volume half-life and foam volume half-life of each column were measured, as shown in
Table 4. Looking at the experimental results, after the addition of modified nanosilica particles, the liquid half-life and foam half-life were significantly improved, indicating enhanced stability. Foam structures with different stages were obtained from the dynamic analysis images in
Figure 11. When the SiO
2–TiO
2 nanoparticles were added into the stable bulk foam solution, the average size of the bubbles at the foam volume half-life obviously reduced, thus effectively preventing the foam aggregation process, as well as enhancing the foam’s duration. This is consistent with the mechanism of the coalescence barrier.
3.4. Aqueous Foam Suppressing VOCs
Aqueous foam can significantly suppress most VOCs by forming a circuitous route for the harmful vapor. The lamellae between the adjacent foam is capable of providing a mass transfer barrier to retard the vapor transition through the vapor solution–volatilization process on the foam membrane surface. However, the suppression ability is completely dependent on the lamellae thickness, which is directly decided by the amount of liquid that the foam membrane can carry. In addition, vapor leakage happens when the adjacent foam coalesces or rearranges, promoting the gas transfer rate and the formation of vacant sites. By adding photocatalytic nanoparticles to the foam solution, the foam membrane was mechanically strengthened and endowed with photocatalytic function [
34,
48]. Bi-functional foam can make up for the lack of barrier capacity in single liquid film.
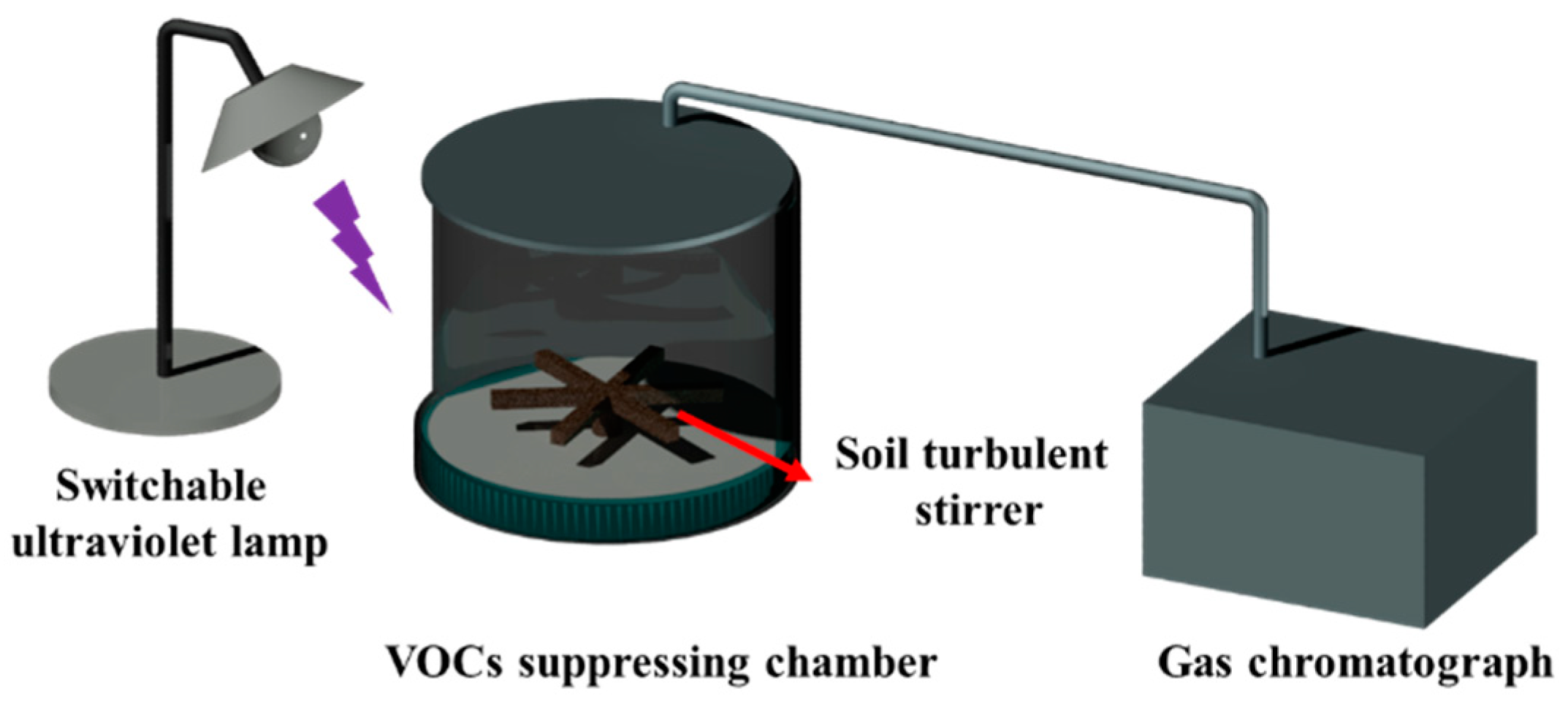
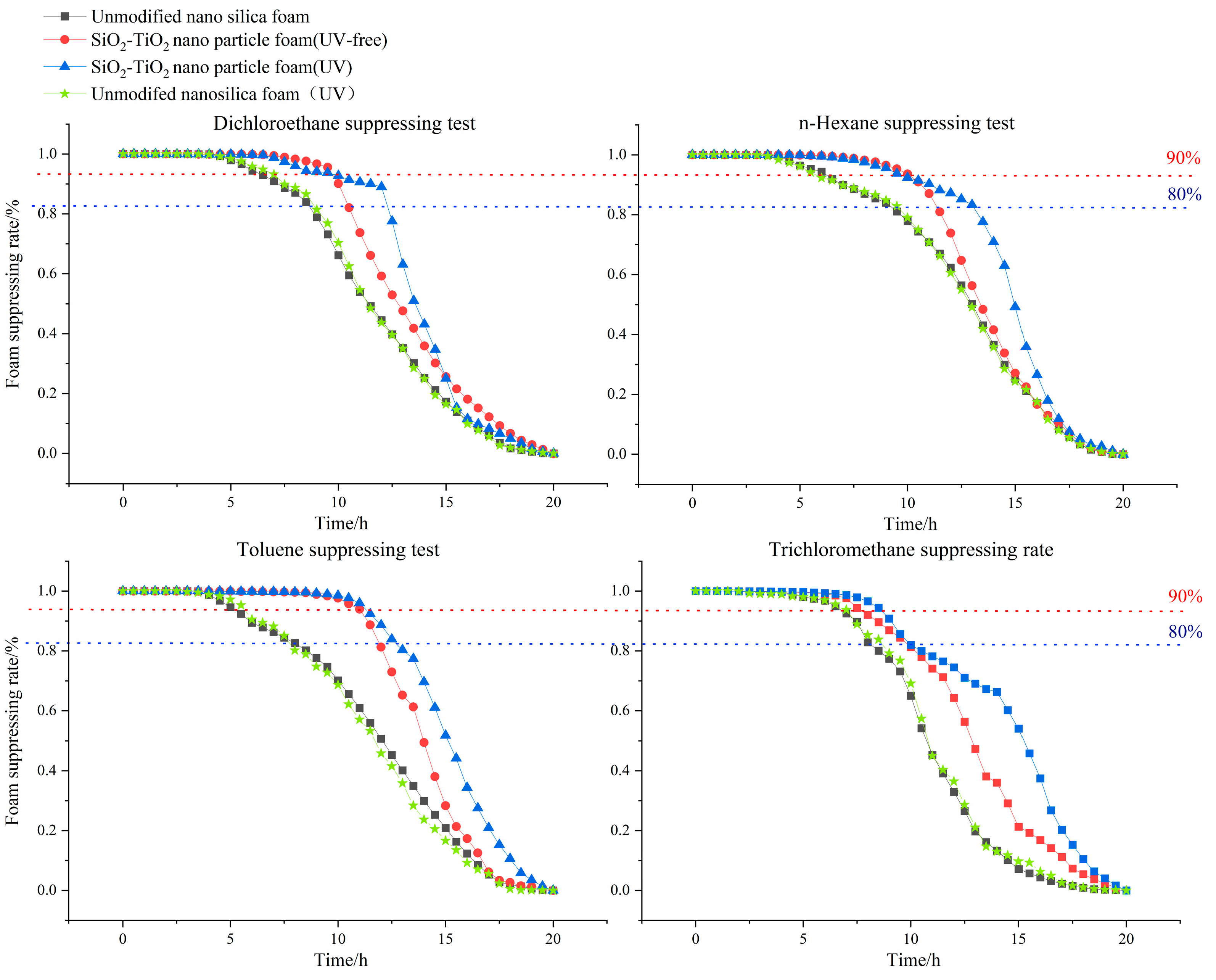
Experiments on foam’s suppressing performance were carried out using the assessment index of suppressing rate, calculated with Formula (2). Each stabilizing foam solution contains 0.5 wt% nanoparticles or nanosilica. Three stems of aqueous foam (unmodified nanosilica foam, SiO2–TiO2 nanoparticle foam in a UV-free environment, and SiO2–TiO2 nanoparticle foam under UV light) were used to suppress the following typical VOCs: a chlorohydrocarbon (dichloroethane), a petroleum hydrocarbon (n–hexane), a benzene derivative (toluene), and an emerging contaminant (trichloromethane). All the tests of VOCs were carried out by diluting standard laboratory agents.
In order to reflect the photocatalytic effect of the nanoparticles, a bidirectional experiment, including the conservation of the VOC suppressing ratio as a function of time, was carried out with modified nanoparticles and unmodified nanoparticles. The experimental results are as follows. From the dynamic curve, the t
80/t
90 index is presented in
Table 4. Compared with unmodified nanosilica, the t
80 and t
90 VOC suppressing times of the modified nanoparticle foam were longer than those of the nanoparticles-free stabilizing foam. The SiO
2–TiO
2 nanoparticle foam under the irradiation of UV light showed an excellent VOC-suppressing ability on four contaminants.
Table 5 shows that the suppression times of the four pollutants are obviously different, depending on the degree of oil resistance of foam materials to different pollutants. From
Table 6, all the E indexes of foam solution are greater than 0 in the presence of n–hexane, dichloroethane, and toluene, and S indexes are less than 0, indicating that the four pollutants have a defoaming effect. From the indexes of toluene, we observed that this contaminant is more easily adsorbed on the surface of the liquid film, and forms a refractory liquid droplet, which is consistent with the short duration of toluene’s blocking rate. The experimental data of n–hexane indicate that n–hexane is more easily adsorbed on the surface of the liquid membrane, affecting the effectiveness of the surface active agent. Therefore, the resistance of the foam to n–hexane is weaker than that of dichloroethane, toluene, and trichloromethane.
Convergence barriers were formed during the transfer of nanoparticles to the air-liquid interface of bubble film, which obstructed the gas penetration between foam units. The barriers are also able to strengthen the foam structure, enabling more flexibility to confront outer vapor pressures such as wind. The prolonged retention time increases the suppression time accordingly. At the same time, the titanium in the SiO
2–TiO
2 composite nanoparticles plays a role in photocatalysis, causing the amounts of VOCs to be further effectively suppressed. The photocatalytic property of the nanoparticles was characterized by the degradation test. Experiments were performed by headspace gas chromatography–mass spectrometry, and results are shown in
Figure 12. Apart from the first three typical VOCs, trichloromethane is easily affected by photochemical reactions and can be degraded without the addition of catalysts, producing hydrogen chloride and phosgene [
49]. The introduction of nanoparticles may weaken the degradation caused by the photochemical reaction due to the decrease in light transmittance. Furthermore, as the foam lamellae become thinner because of the film liquid drainage, the foam skeleton keeps its initial frame structure, which gradually emerges from top to bottom; it has then lost the function of mass transfer across the barrier. By adding the photocatalytic nanoparticles in the foam solution, the post-forming skeleton of aqueous foam will be endowed with a function of VOCs’ adsorption-degradation, meaning the dry foam is still able to suppress the VOCs. This theory can be testified by the experimental solution shown in
Figure 13, in which the subsequent suppression time t
80 of UV-irradiated foam is slightly delayed by about 1 h compared to the UV-free case.