Simple Summary
Amphistomes, also known as rumen flukes, are parasites of domestic and wild ruminants and occur globally. Adult parasites are found in the forestomach, and the young flukes are found in the small intestines, causing severe damage. The flukes are transmitted by various species of freshwater snails. While the disease is well documented in domestic ruminants, there are gaps in knowledge pertaining to wild ruminants with regard to the fluke species as well as the snail species which transmit them. Therefore, freshwater snails were surveyed from 19 water points that are frequented by wild ruminants in the Matebeleland region, Zimbabwe. Snails were found at nine sites, and eight species were identified and screened for rumen fluke DNA to determine the fluke species and prevalence of infection. Rumen fluke DNA was detected in 11.9% of the screened snails. Prevalence was high in the West Nicholson locality and in Bulinus globosus snail species. One rumen fluke species, i.e., Calicophoron microbothrium, was confirmed in one snail species and there were also mixed infections with lung fluke parasite, Paragonimus spp., in two snail species. This was the first study documenting the presence of this lung fluke in Zimbabwe.
Abstract
This study aimed at determining the identity of freshwater snails collected from selected water habitats frequented by wildlife as source of drinking water in the Matebeleland region of Zimbabwe and further screening the identified snails for natural infections with amphistomes using PCR. A total of 487 freshwater snails were collected from six areas in the Matebeleland region of Zimbabwe for identification and screening of amphistome infection. Eight freshwater snail species were morphologically identified and Biomphalaria pfeifferi, Bul. tropicus, Bul. truncatus, Bul. globosus, and L. (R.) natalensis were confirmed using the COI gene. Bulinus tropicus and Phy. acuta were the most abundant species at 33.9% (165/487) and 31.2% (155/487), respectively. DNA of amphistome was detected in 11.9% (58/487) of the collected snails. The highest infection rate was detected in Bul. globosus (44.4%). West Nicholson recorded the highest infection rate (33.9%), and infection was not detected in L. (R.) natalensis, Phy. acuta, and Bellamya spp. Amphistome DNA from M. tuberculata was successfully sequenced and identified as Calicophoron microbothrium. An additional band was detected in M. tuberculata, Bul. tropicus, and Bul. trancatus, which showed a 96.42% similarity to Paragonimus sp. sequence in the GenBank.
1. Introduction
Amphistomes are digenetic trematodes that exhibit a heteroxenous life cycle that includes an intermediate and definitive host [1]. Although over 70 amphistomes have been recorded globally to date [2,3], the intermediate host snails have only been described for a few amphistomes species in sub-Saharan Africa [4]. Amphistome species use several freshwater snail species as intermediate hosts (IHs) for transmission, and these include species from the genus Bulinus [5,6,7], Galba [8,9], Lymnaea (Radix) [10,11], Biomphalaria [12], and Segmentorbis [12]. According to Laidemitt et al. [12], Bulinus species, which are the most widely distributed snails in sub-Saharan Africa, accounts for the wide distribution of Calicophoron spp. in Africa. Furthermore, several Bulinus species have been implicated in the transmission of Calicophoron microbothrium, an amphistome responsible for most cases of amphistomosis in both wild and domestic ruminants [4].
Like other trematodes, both domestic and wild ruminants get infected with amphistomes by grazing in pasture around drinking water points contaminated with metacercariae [13]. Infection in wild ruminants can be confirmed at post-mortem by the presence of immature stages in the duodenum, and adult amphistomes in the forestomachs of ruminants, and at ante-mortem through the detection of eggs in feces [14]. However, surveys and monitoring of these infections in free-ranging wild ruminants can be challenging [15] due to factors such as accessibility and ethical issues associated with capturing, handling, and releasing wild animals to collect non-invasive fecal samples [16]. Moreover, identification up to the species level using morphological characters is a challenge, and at ante-mortem, the egg morphology is not easily distinguishable from that of other trematodes such as Fasciola spp. [17].
Data from previous research in sub-Saharan Africa have indicated that majority of amphistomes species occurring in wild ruminants are also common in domestic ruminants, except Bilatorchis papillogenitalis, Carmyerius bubalis, and Cotylophoron macrosphinctris, which have only been documented in wild ruminants to date [4,18]. These three species are amongst those with unknown intermediate snail hosts. Amphistome infection in the intermediate snail hosts can be detected through shedding of cercariae or squashing of the snail soft tissue and visualizing the developmental stages using a light microscope and the subsequent morphological characterization of the cercariae/rediae/sporocyst [19,20,21]. However, the morphological characterization of these larval stages has limitations as most studies could only discriminate up to the genus level [21,22,23], with only a few studies able to distinguish up to the species level [24,25,26,27]. Nonetheless, Kane et al. [28] indicated that morphological identification of larval trematodes is error-prone and hard or impossible for the genus or species level resolution.
Although some recent studies still utilize shedding of cercariae as sole detection method of infection [21], most recent studies couple the use of shedding cercariae with molecular techniques such as Restriction Fragment Length Polymorphism (RFLP) of Polymerase Chain Reaction (PCR) products [29] or sequencing of the PCR products for identification up to the species level [6,12,29]. Some studies also amplified the larval DNA directly from the snails, followed by sequencing [12,30] or RFLP of the products [29]. However, Schols [30] showed that detection of larval infection using PCR was better than cercariae shedding.
According to Carolus et al. [31], knowledge on the prevalence, diversity, and ecology of both intermediate host freshwater snails and trematodes is key to understanding disease transmission dynamics and the possible control of trematodes of economic significance. Although there are several ways of identifying freshwater snails and trematode larval stages based on morphological characters, molecular methods are known to provide better resolution for identifying snails and trematodes at the species level [32]. Hence, the aim of this study was to determine the identity of freshwater snails collected from selected water habitats serving as water sources for wildlife in selected game ranches, conservancies, and game parks located in the Matebeleland region of Zimbabwe and the amphistome species they may transmit in these habitats.
2. Materials and Methods
2.1. Study Areas and Sample Collection
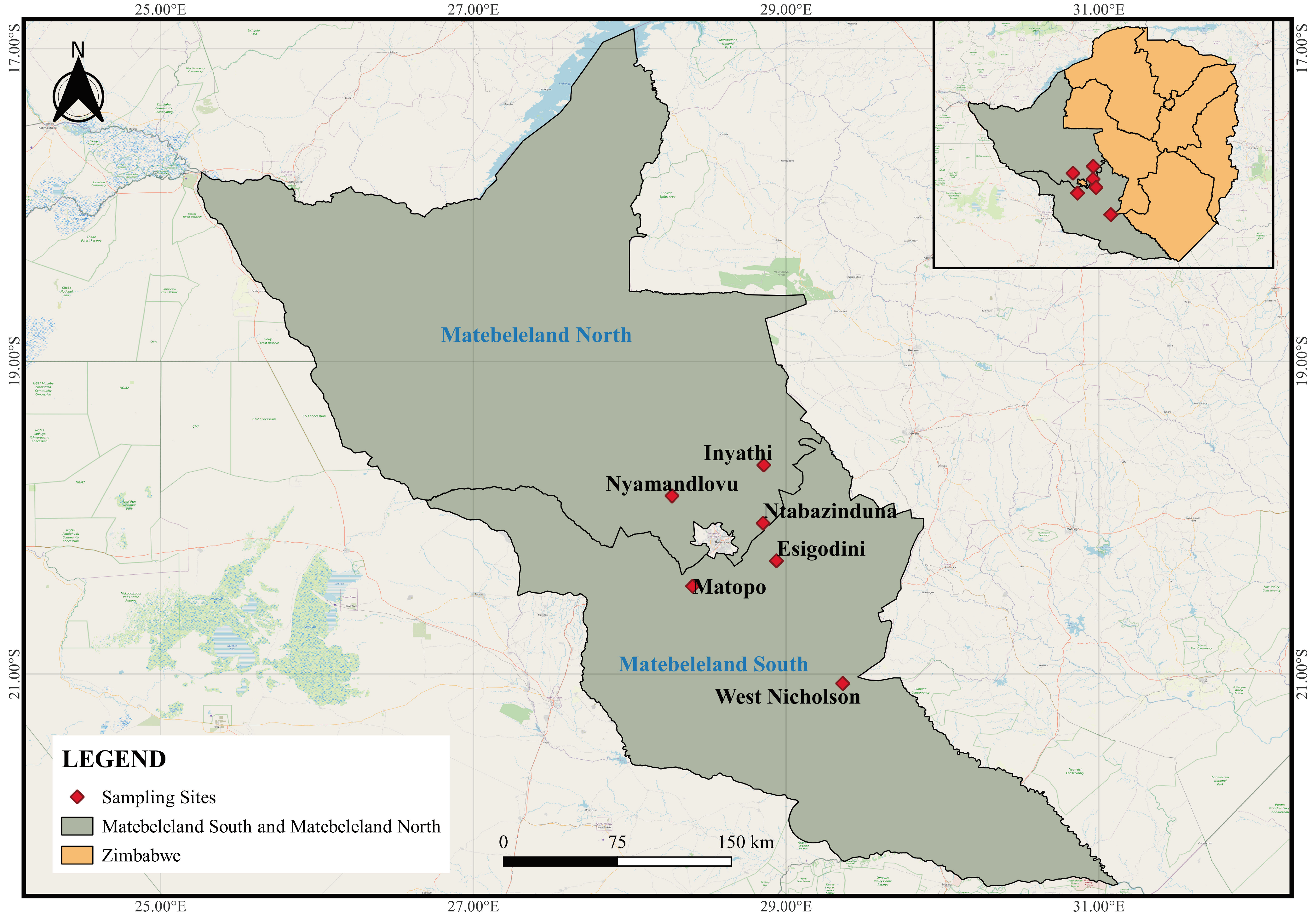
Freshwater gastropods were surveyed from water sources frequented by wildlife around game ranches, conservancies, and game parks located in Inyathi, Nyamandlovu, and Ntabazinduna, located in Matebeleland North and Esigodini, West Nicholson, and Matopos which are in Matebeleland South province of Zimbabwe (Figure 1). Sampling sites were pre-selected based on the following criteria: little or no human activity and locality is a drinking water point for wild ruminants. The habitat type and vegetation cover were recorded for each site (Table 1), and snails were collected using a scooping net and dredge for superficial and deep-water samples, respectively [31]. Collected snails were first washed with distilled water and thereafter preserved in 70% ethanol for morphological and molecular analyses.
Figure 1.
All sampling sites of the study located in the Matebeleland region of Zimbabwe.

Table 1.
Description of habitat type and animal/human activity at localities where freshwater snails were collected in the Matebeleland region of Zimbabwe.
2.2. Morphological and Molecular Identification of Snails
2.2.1. Morphological Classification
Morphological identification of snails was based on the classification keys as described by Brown [33]. Snails of the same species morphologically were grouped, and representative specimens were selected for confirmation using PCR.
2.2.2. DNA Extraction from Gastropods
Selected snail specimens were washed with sterile water to remove alcohol, and excess water was removed with sterile absorbent paper. Tissue from foot of each snail or whole snail tissue was harvested using either a sterile blade or the whole snail tissue was removed and then frozen at −20 °C for 2 h before use. DNA was extracted using a modified Quick-DNATM Tissue Miniprep Kit (Zymo Research Corporation, Irvine, CA 92164, USA) protocol.
2.2.3. Molecular Characterization of Gastropods
Extracted DNA were amplified based on the COI region using the primers COI (F) 5′-TAATGTWATTGTTACAGCACATG-3′ and COI (R) 5′-GTTGRTATAAAATAGCATCACCW-3′ [31]. PCR was performed in a total reaction volume of 25 µL, composing of 5 µL of 5X One Taq PCR buffer, 0.4 µL of 10 mM dNTPs, 0.5 µL forward primer (10 mM), 0.5 µL reverse primer (10 mM), 3 µL of 25 mM MgCl2, 2 µL DNA template, 0.125 µL of One Taq polymerase (NEB, Hitchin, UK), and nuclease-free water to make up the final volume. The cycling protocol for the reaction was as follows: initial denaturation at 94 °C for 3 min, followed by 30 cycles of denaturation at 94 °C for 30 s, annealing at 64 °C for 45 s, extension at 68 °C for 1 min, and a final extension of 68 °C for 15 min. The fragments were separated on 1% agarose stained with ethidium bromide, and successful amplicons were identified by a band at 630 base pairs. Amplicons were sent to Inqaba Biotech for Sanger sequencing.
2.2.4. Molecular Detection of Amphistomes from Snail Tissue Samples
Snails were assessed for amphistome infection using the primers GA1 (5′-AGAACATCGACATCTTGAAC-3′) and BD2 (5′-TATGCTTAAATTCAGCGGGT-3′) [12]. Calicophoron microbothrium DNA was used as a positive control/signal for amphistome DNA. The PCR mix was composed of 2 µL of 10X PCR buffer SuperTherm (Separation Scientific SA (Pty) Ltd., Roodepoort, South Africa), 0.8 µL of 10 mM dNTPs, 0.4 µL forward primer (10 µM/µL), 0.4 µL reverse primer (10 µM/µL), 1.6 µL 25 mM MgCl2, 1 µL DNA template, 0.2 µL Super Therm polymerase, and nuclease-free water to make up a total reaction volume of 20 µL. Amplification was performed under the following thermocycling conditions: 95 °C for 3 min, followed by 30 cycles of 95 °C for 30 s, 55 °C for 45 s, and 72 °C for 1 min, and a final extension of 72 °C for 15 min. Amplicons were separated on 1% agarose gels stained with ethidium bromide, and positive isolates were identified by a band at approximately 385 bp. Positive amplicons were sent to Inqaba Biotechnical Industries (Pretoria, South Africa) for Sanger sequencing.
2.2.5. Molecular Analysis of Trematode/Amphistome Isolates from Snails
Sequences were viewed, assembled, and manually edited using BioEdit version 7.2 (Sequence Alignment Editor) [34], and NCBI BLAST (Basic local alignment search tool) was used to identify the closest matches available on GenBank database. Sequences were trimmed to a common length of 455 nucleotides. Tamura 3-parameter (T92) was selected as the best model fit for the dataset, and the neighbour-joining (NJ) and maximum likelihood trees were generated on the MEGA 7 software [35]. The phylograms were 50% majority-rule, and the nodal support was estimated using 1000 bootstrap pseudo-replicates for both methods.
3. Results
3.1. Description of Snail Habitats
From the six localities visited in the Matebeleland region (Figure 1), 19 water sites were surveyed, and these were predominantly man-made waterholes in Nyamandlovu (n = 13), dams in West Nicholson (n = 2), Matopos National Park (n = 1), Ntabazinduna (n = 1), and Esigodini (n = 1), and lastly, a river in Inyathi (n = 1) (Table 1). All habitats had different types and levels of submerged vegetations, and only habitats from West Nicholson, Esigodini, and Inyathi had trees on their periphery. The surrounding areas were clear, with decaying matter found in dams in West Nicholson and Ntabazinduna. Results show that although all 13 sites from Nyamandlovu had solely wildlife activity, some sites showed a level of interaction or shared habitat with livestock (n = 4) and to a lesser extent humans (n = 1).
3.2. Morphologically Identified Snail Species and Their Abundance
Snails were found at 9 of 19 sites (47.37%), while none were found at 10 sites from Nyamandlovu (Table 1). A total of 487 gastropods were collected, and these were from Nyamandlovu (n = 212) and Ntabazinduna (n = 174), followed by West Nicholson (n = 59), Esigodini (n = 21), Matopos National Park (n = 18), and to a lesser extent Inyathi (n = 3) (Table 2). From these collections, eight species were morphologically identified as Melanoides (M.) tuberculata, Bulinus (Bul.) globosus, Bul. truncatus, Bul. tropicus, Biomphalaria (Bio.) pfeifferi, Physa (Phy.) acuta, Lymnaea (L.) natalensis, and Bellamya spp. (Table 2).

Table 2.
Morphological and molecular identities and number of snail species collected in the different localities surveyed in the Matebeleland region of Zimbabwe.
Bulinus tropicus was the most distributed species across sites and was found in four of the six surveyed areas. Furthermore, this species was the most abundant and contributed 33.9% (165/487) of the collected snail populations. This was followed by Phy. acuta that contributed to 31.2% (155/487) and was found in Esigodini and Nyamandlovu. Bulinus truncatus and M. tuberculata, which were each found in Ntabazinduna and West Nicholson, contributed to 12.5% (61/487) and 11.9% (58/487), respectively. Surprisingly, Bio. pfeifferi, Bul. globosus, and L. natalensis were found in multiple areas, though in low numbers of 20/487 (4.1%), 18/487 (3.7%), and 8/487(1.6%), respectively. The least collected snail species was Bellamya spp., which was found in Ntabazinduna and contributed to 0.4% (2/487) of the collected snail population.
3.3. Molecular Confirmation and Phylogenetic Relationship of Snail Species.
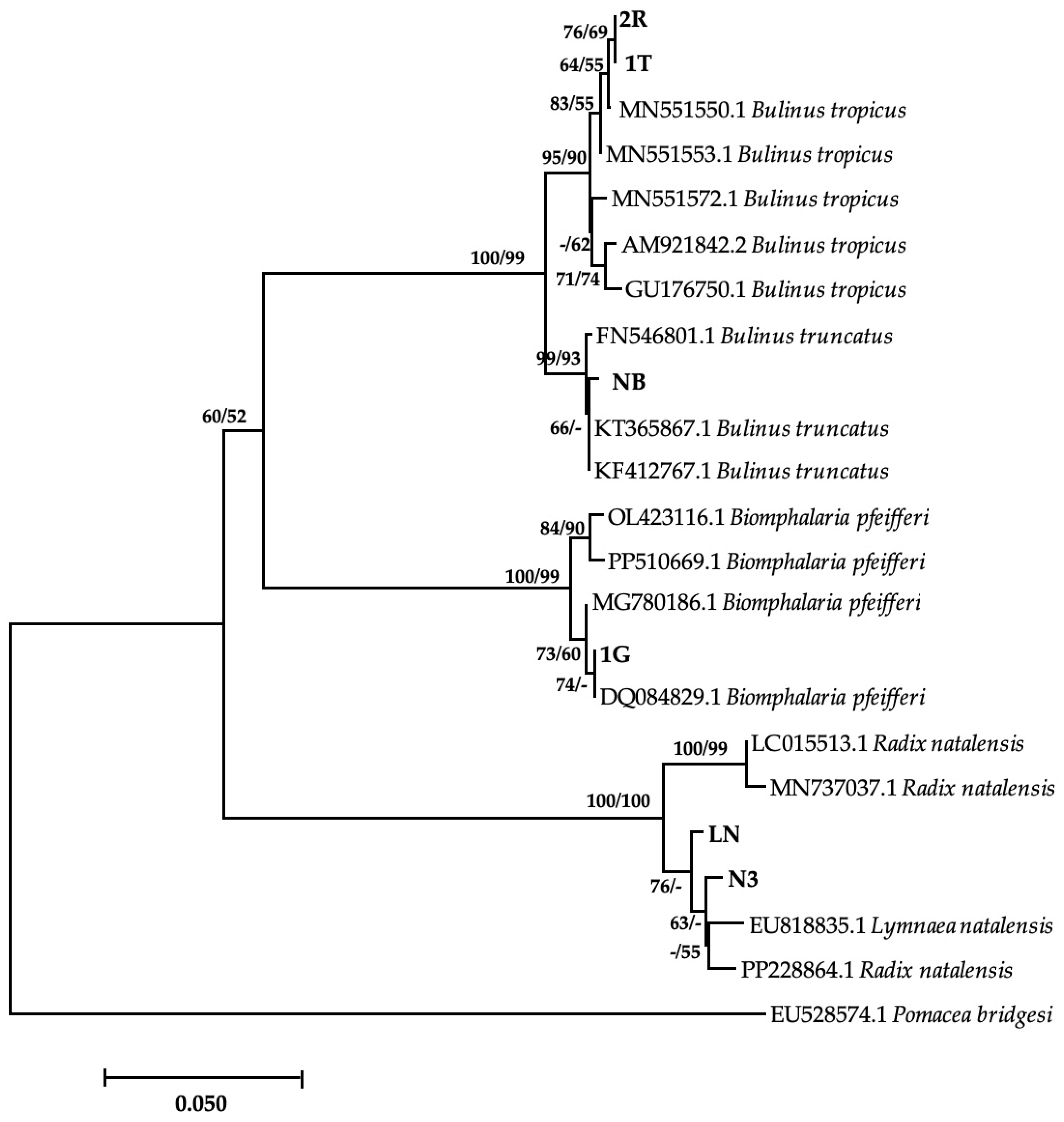
Of the eight morphologically identified species, four snail species were confirmed with a BLAST similarity index ranging between 98.24 and 100% (Table 2). Amongst these was 1G, which showed 100% homology with Bio. pfeifferi from Zimbabwe (DQ084829). However, the phylogenetic tree showed a moderate support between this isolate and other GenBank isolates (Figure 2). The isolated Bio. pfeifferi formed a well-supported sister clade to Bulinus species, which falls under the same family. Our isolate NB showed a 99.78% identity with Bul. truncatus isolate from Iran (KT365867) and formed a well-supported clade with other Bul. truncatus isolates. These isolates formed a strongly supported monophyletic sister clade with Bul. tropicus isolates, including those from this study (2R and IT) (Figure 2), which showed 99.78% homology with Bul. tropicus from Uganda (MN551550). Lastly, our L. natalensis isolates (LN and N3) (Figure 2) showed a homology of 98.68% with L. natalensis from Malawi (EU818835). These species formed a well-supported separate clade from the Bulinus and Biomphalaria clade. However, the relationship between the isolates was moderately to weakly supported. The sequences generated from this study were submitted to NCBI GenBank under the accession numbers PP389543–PP389548. Sequences for Bul. globosus failed quality control, while sequencing failed for M. tuberculata, Phy. acuta, and Bellamya spp. The time lapse between the PCR and the amplicons reaching South Africa from Zimbabwe for post clean-up and sequencing might have compromised the quality of the PCR products, and possibly causing their degradation.
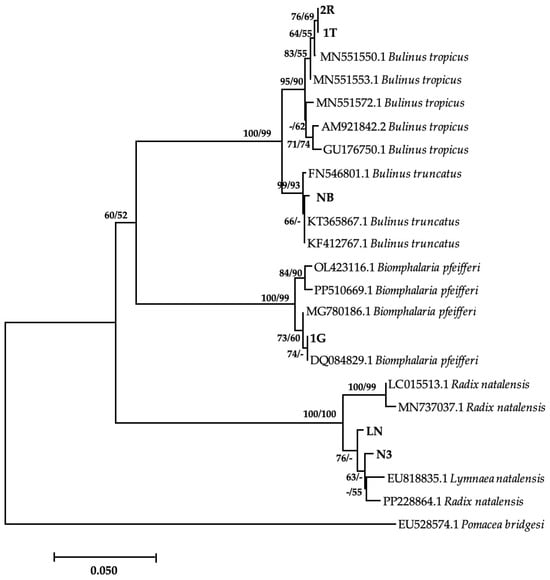
Figure 2.
Neighbor-joining tree-based COI gene illustrating the relationship between freshwater snails obtained from game ranches, conservancies, and game parks located in the Matebeleland region of Zimbabwe and the closest matches from the NCBI GenBank. The nodal support values (%) indicated in an order using neighbor-joining and maximum likelihood. 2R, IT, NB, 1G, LN, N3 = isolates from the study. The 50% majority-rule was applied, and any support value of lower than 50% was represented by a hyphen.
3.4. Molecular Detection of Amphistome DNA in Field-Collected Snails
All 487 gastropod isolates were individually screened for amphistome DNA based on the ITS-2 gene. A total of 58 of 487 (11.9%) snails showed a band at approximately 385 bp, which was consistent with a Calicophoron (Cal.) microbothrium isolate used as a control for amphistomes. Bulinus globosus recorded the highest prevalence of amphistome infections (44.4%), followed by M. tuberculata (33.9%) (Table 3). No amphistome DNA was detected in Phy. acuta, L. natalensis, and Bellamya spp. The highest incidence of amphistome infections in snails per locality was recorded in West Nicholson, with a prevalence of 33.9% in M. tuberculata. In contrast, snails from Inyathi did not harbor any amphistomes, and those from Nyamandlovu had a prevalence of 2.4% in Bul. tropicus despite the area recording the highest number of gastropod specimens collected. However, sequencing of amphistome amplicons was only successful for a sample from M. tuberculata, which was confirmed as Cal. microbothrium with a percentage similarity of 100% (Table 4) and submitted to GenBank under the accession number of PP392962. The remaining samples, which were the majority, failed quality control.

Table 3.
Prevalence per locality of amphistome infection in freshwater snails collected from the Matebeleland region of Zimbabwe as detected by PCR.

Table 4.
BLAST percentage similarity of trematode DNA obtained from snails collected in the Matebeleland region of Zimbabwe.
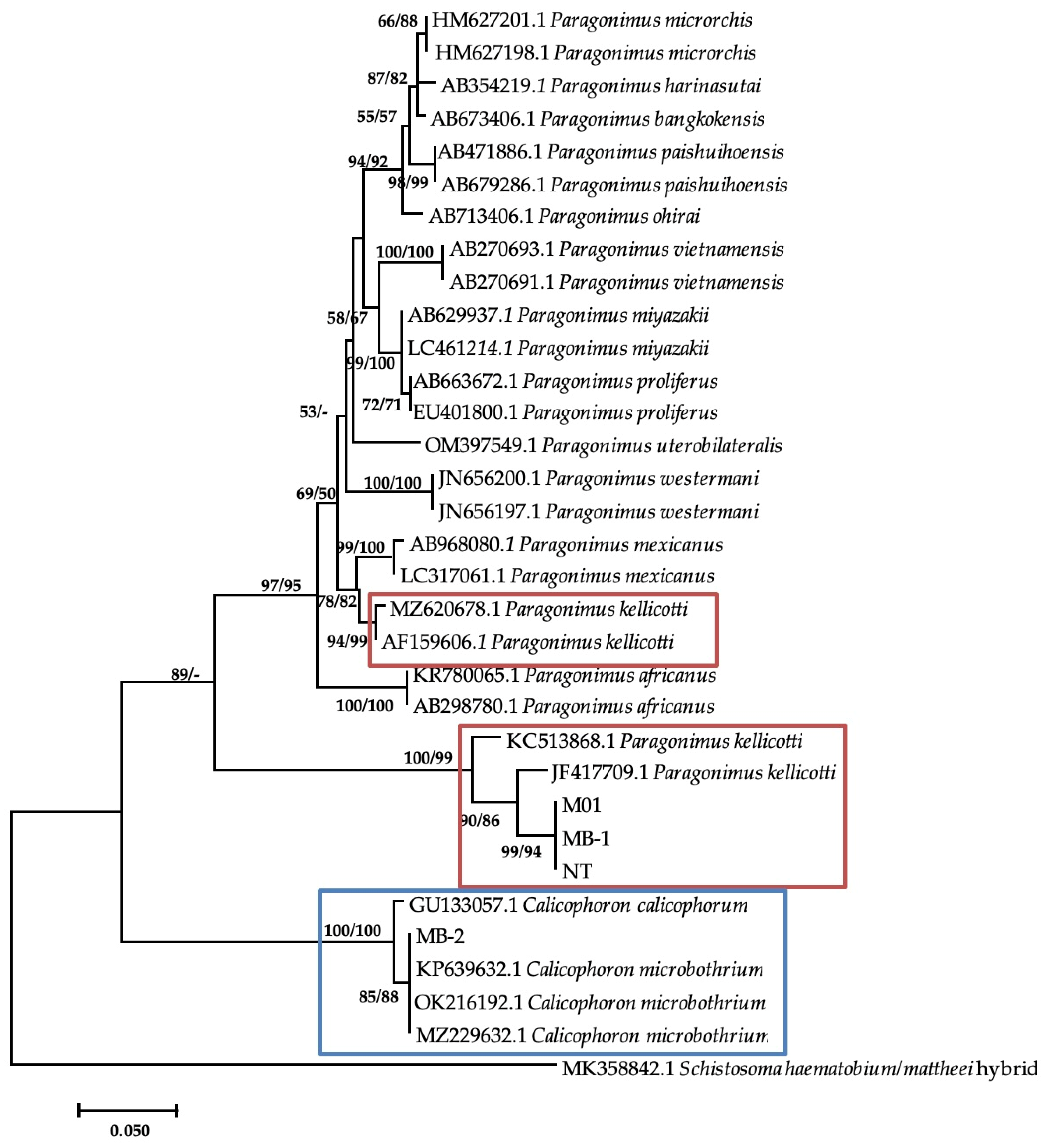
Additional bands were observed at approximately 280–290 base pairs from M. tuberculata (n = 9) and Bul. tropicus (n = 1), from Esigodini and Bul. truncatus (n = 1), and from Ntabazinduna, indicating a mixed infection. Sequencing of three of the additional bands from M. tuberculata and BLAST analysis showed a similarity index of 96.42% to Paragonimus kellicotti (Table 4). The sequences were submitted to GenBank as Paragonimus sp. under the accession numbers of PP392960, PP392961, and PP392963. Phylogenetic analysis showed that these sequences formed a clade with two P. kellicotti isolates from India (KC523868.1) and the United States (JF4177709.1) from Indoplarnobis exustus and Orconectes virilis, respectively. The genetic distance within this clade was 3%. This clade also formed a monophyletic sister clade with various Paragonimus species, including two more P. kellicotti (Figure 3). The genetic p-distance between these two clades was 28%.
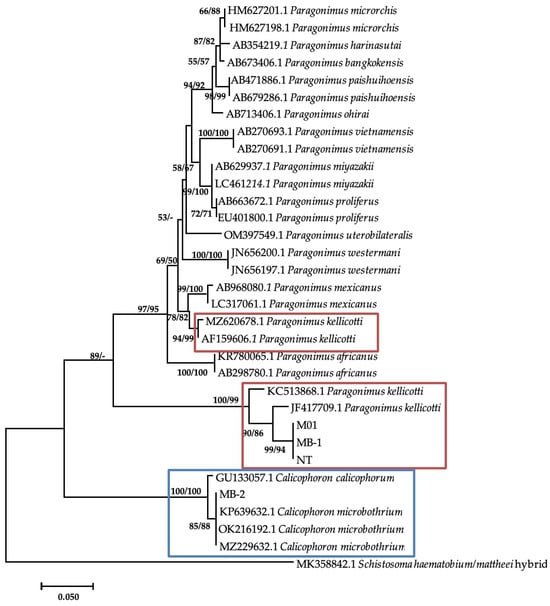
Figure 3.
Neighbor-joining tree-based ITS-2 gene illustrating the relationship between trematodes isolates from freshwater snails from the Matebeleland in Zimbabwe and the closest matches from the NCBI GenBank. The nodal support values (%) indicated in an order using neighbor-joining and maximum likelihood.
4. Discussion
Snail survey conducted in this study was from three different habitat types that include dams, waterholes, and a river. All the habitats had macrophytes (submerged vegetation), with Ntabazinduna sampling site having an abundance of brown algae macrophytes. According to Min et al. [36], physico-chemical properties and macrophyte abundance together with other factors influence the diversity of freshwater snails. The most abundant snail species were collected from Ntabazinduna, accounting for 36% of the total number of snails. Middelboe and Markager [37] suggested that the presence of submerged macrophytes increase periphyton, the food source of Bulinus spp. and Bio. pfeifferi. In this study, a total of eight snail species collected from the Matebeleland region of Zimbabwe in selected areas included Bul. globosus, Bul. tropicus, Bul. truncatus, M. tuberculata, Bio. pfeifferi, L. natalensis, and Phy. Acuta, and these have been implicated in the transmission of amphistome species in wildlife and domestic ruminants [4] except Phy. acuta and Bellamya spp.
Results showed that Bul. tropicus was the most widely distributed and abundant species in the Matebeleland region of Zimbabwe. This was not surprising as earlier reports have indicated that this species is the most widely distributed freshwater snail in Zimbabwe [38] and is well adapted to a wide variety of environments [33]. However, Chingwena et al. [7] later reported that Bul. tropicus was the most abundant species in the lowveld and the third most abundant snail species following L. natalensis and Bul. globosus in the highveld. Although this still explains the abundance of this species in the Matebeleland region that falls within the middle to highveld, L. natalensis and Bul. Globosus were surprisingly collected in low numbers in this study. Physa acuta, the second most abundant species, is an invasive snail, which was previously reported in Zimbabwe [39]. Although this species showed a limitation in distribution, and the one locality where this snail species was collected had macrophytes in abundance compared to other species. This is not surprising as not only several authors have shown that this species is now widely distributed in Zimbabwe [40] but also that it is invasive [41].
Of the eight snail species screened for amphistome DNA, M. tuberculata, Bio. pfeifferi, Bul. tropicus, Bul. globosus, and Bul. truncatus were positive. Amphistome detection in the Bulinus and Biomphalaria species was not surprising as a wide range of these snail species have been reported to act as intermediate hosts for different amphistome species in sub-Saharan Africa [7,25,42,43,44]. Although Bul. tropicus was reported to act as an intermediate host of Cal. microbothrium, with high prevalence of experimental infections [7], our study reported a high prevalence of amphistome DNA in Bul. globosus. Melanoides tuberculata showed the second highest prevalence of amphistome infection in this study. Previous reports have already confirmed the susceptibility of this species through experimental infections with Cal. microbothrium in Zimbabwe [7] and South Africa [2], and to another trematode species, Gastrodiscus aegyptiacus, in Zimbabwe [45]. Based on the successful identification of Cal. microbothrium from M. tuberculata through sequencing, to the best of our knowledge, this is the first study to confirm natural infection of this Thiaridae species with Cal. microbothrium in Africa.
Results showed that amphistome DNA was not detected in three snail species, namely, Phy. acuta, Bellamya spp., and L. natalensis. Although earlier experimental studies have showed that L. natalensis was refractory to Cal. microbothrium [7], this species was found infected with Gastrothylax/Paramphistomum in Zimbabwe [30]. A similar pattern was observed with Phy. acuta, which was successfully infected with G. aegyptiacus during dissection after experimental exposure [45], but no amphistome DNA was detected from this species despite being collected in high numbers, indicating that there might be amphistome species IH specificity, and this may be confirmed through experimental studies like those conducted by Chingwena et al. [7].
Results from this study revealed cases of mixed infections of amphistome DNA with other DNA of a trematode species identified as Paragonimus-like species in Bul. truncatus and M. tuberculata. Paragonimus spp. are lung flukes that have been reported worldwide including Africa and mainly in West and Central African regions. Species reported in the West and Central Africa include P. africanus, P. gondwanensis, P. kerberti, and P. uterobilateralis [46]. According to Procop [47], Paragonimus species usually utilize snail species from families Pleuroceridae and Thiaridae as IHs. While results from this study and previous reports link paragonimiasis with presence of M. tuberculata in South Africa [48], which may explain the infections observed in M. tuberculata in this study, the phylogenetic positioning of these isolates and the high genetic distance with other reported Paragonimus spp. raise concerns of whether these isolates belong to the genus Paragonimus, or the sequences deposited on GenBank are misidentified under the genus Paragonimus.
5. Conclusions
Eight freshwater snail species common in drinking water sources frequented by wildlife from the Matebeleland region of Zimbabwe were identified in this study with Bul. tropicus and Phy. acuta as the predominant species, and the overall prevalence of amphistome DNA (Cal. microbothrium) was 11.9%. Prevalence rate was higher in Bul. globosus followed by M. tuberculata. The highest number of snails were collected from Nyamandlovu, meanwhile the highest prevalence of amphistome DNA was recorded in Beitbridge. A single sample was confirmed as Cal. microbothrium through DNA sequencing and a mixed infection with Paragonimus species-like trematode was also confirmed. Failure to sequence other amphistome samples shows the need to specifically use the larval stage from the infected snails (sporocysts, rediae, or cercariae), to increase the quantity and quality of DNA to be successful in identifying and characterizing the amphistome species found in freshwater snails. There is a need to also develop and apply the use of primers and protocols that do not require sequencing such as the LAMP protocols and PCR-RLFP to discriminate multiple amphistome species of wildlife in IH snails including mixed infections. We recommend future studies such as collecting amphistome specimens from culled wildlife ruminants from the study locations for morphological and molecular identification.
Author Contributions
Conceptualization, M.S.S. and S.M.; methodology, M.S.S., C.N. and M.P.M.; software, M.P.M.; validation, M.S.S. and M.P.M.; formal analysis, M.S.S.; investigation M.S.S. and C.N.; resources, M.S.S. and S.M.; data curation, S.M., M.S.S. and M.P.M.; writing—original draft preparation, M.S.S., M.P.M. and C.N. writing—review and editing, M.P.M. and S.M.; supervision, S.M. and M.P.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by SM’s research productivity funds (UKZN).
Institutional Review Board Statement
The protocols for this study were reviewed and approved by the Institutional Review Board (NUST/IRB/2021/17) of the National University of Science and Technology, Zimbabwe.
Informed Consent Statement
Not applicable.
Data Availability Statement
The sequences generated in this study were deposited to GenBank under accession numbers of PP389543-48 and PP392960-63. The alignments used for the phylogenetic analysis are available from the corresponding author on request.
Acknowledgments
The authors acknowledge Upenyu Makombo for technical assistance rendered during sample collection.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Huson, K.M.; Oliver, N.A.; Robinson, M.W. Paramphistomosis of ruminants: An emerging parasitic disease in Europe. Trends Parasitol. 2017, 33, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Malatji, M.P.; Myende, N.; Mukaratirwa, S. Are Freshwater Snails, Melanoides sp. and Invasive Tarebia granifera (Gastropoda: Thiaridae) Suitable Intermediate Hosts for Calicophoron microbothrium (Trematoda: Paramphistomoidea)? An experimental study. Front. Vet. Sci. 2021, 8, 1–8. [Google Scholar] [CrossRef]
- Ghatani, S.; Shylla, J.A.; Tandon, V.; Chatterjee, A.; Roy, B. Molecular characterization of pouched amphistome parasites (Trematoda: Gastrothylacidae) using ribosomal ITS2 sequence and secondary structures. J. Helminthol. 2012, 86, 117–124. [Google Scholar] [CrossRef]
- Pfukenyi, D.M.; Mukaratirwa, S. Amphistome infections in domestic and wild ruminants in East and Southern Africa: A review. Onderstepoort J. Vet. Res. 2018, 85, 1–13. [Google Scholar] [CrossRef]
- De Kock, K.N.; Wolmarans, C.T. Distribution, habitats and role as intermediate host of the freshwater snail, Bulinus forskalii, in South Africa. Onderstepoort J. Vet. Res. 2005, 72, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Lotfy, W.M.; Brant, S.V.; Ashmawy, K.I.; Devkota, R.; Mkoji, G.M.; Loker, E.S. A molecular approach for identification of paramphistomes from Africa and Asia. Vet. Parasitol. 2010, 174, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Chingwena, G.; Mukaratirwa, S.; Chimbari, M.; Kristensen, T.K.; Madsen, H. Population dynamics and ecology of freshwater gastropods in the highveld and lowveld regions of Zimbabwe, with emphasis on schistosome and amphistome intermediate hosts. Afr. Zool. 2004, 39, 55–62. [Google Scholar] [CrossRef]
- Dinnik, J. Paramphistomum daubneyi sp. nov. from cattle and its snail host in the Kenya Highlands. Parasitology 1962, 52, 143–151. [Google Scholar] [CrossRef]
- Eduardo, S.L. Zoogeographical affinities of paramphistomids of ruminants. Trans. Natl. Acad. Sci. Technol. (Philipp.) 1987, 9, 229–242. [Google Scholar]
- Sanabria, R.; Titi, A.; Mekroud, A.; Vignoles, P.; Dreyfuss, G.; Rondelaud, D.; Romero, J. Paramphistomum daubneyi: Characteristics of infection in three lymnaeid species. Parasite 2012, 19, 445–449. [Google Scholar] [CrossRef]
- Rafiq, N.; Ayaz, S.; Niaz, S.; Haleem, S.; Ullah, R.; Bari, A.; Bourhia, M.; Ali, E.A. Changes in the prevalence of natural Paramphistomum cercariae infection in Indoplanorbis and Lymnaea intermediate hosts influenced by meteorological factors. J. Trop. Med. 2022, 2022, 8719834. [Google Scholar] [CrossRef]
- Laidemitt, M.R.; Zawadzki, E.T.; Brant, S.V.; Mutuku, M.W.; Mkoji, G.M.; Loker, E.S. Loads of trematodes: Discovering hidden diversity of paramphistomoids in Kenyan ruminants. Parasitology 2012, 144, 131–147. [Google Scholar] [CrossRef]
- Phiri, A.M.; Chota, A.; Muma, J.B.; Munyeme, M.; Sikasunge, C.S. Helminth parasites of the Kafue lechwe antelope (Kobus leche kafuensis): A potential source of infection to domestic animals in the Kafue wetlands of Zambia. J. Helminthol. 2011, 85, 20–27. [Google Scholar] [CrossRef]
- Zintl, A.; Garcia-Campos, A.; Trudgett, A.; Chryssafidis, A.L.; Talavera-Arce, S.; Fu, Y.; Egan, S.; Lawlor, A.; Negredo, C.; Brennan, G.; et al. Bovine paramphistomes in Ireland. Vet. Parasitol. 2014, 204, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Ryser-Degiorgis, M.P. Wildlife health investigations: Needs, challenges and recommendations. BMC Vet. Res. 2013, 9, 223. [Google Scholar] [CrossRef]
- Fontoura-Gonçalves, C.; Portocarrero, É.; Oliveira, A.; Lozano, J.; Rinaldi, L.; Cringoli, G.; Madeira de Carvalho, L.; Santos, N. Challenges of copro-parasitological surveys in wild Iberian ibex (Capra pyrenaica) populations addressed through a combination of molecular and statistical tools. Ecol. Evol. 2023, 13, e10172. [Google Scholar] [CrossRef] [PubMed]
- González-Warleta, M.; Lladosa, S.; Castro-Hermida, J.A.; Martínez-Ibeas, A.M.; Conesa, D.; Munoz, F.; López-Quílez, A.; Manga-González, Y.; Mezo, M. Bovine paramphistomosis in Galicia (Spain): Prevalence, intensity, aetiology and geospatial distribution of the infection. Vet. Parasitol. 2013, 191, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Sibula, M.S.; Nyagura, I.; Malatji, M.P.; Mukaratirwa, S. Prevalence and geographical distribution of amphistomes of African wild ruminants: A scoping review. Int. J. Parasitol. Parasites Wildl. 2024, 23, 100906. [Google Scholar] [CrossRef] [PubMed]
- King, P.H.; Van As, J.G. Cercariae shed by Bulinus tropicus (Krauss, 1848) in the Free State, South Africa. Afr. Zool. 2001, 36, 95–105. [Google Scholar] [CrossRef]
- Chingwena, G.; Mukaratirwa, S.; Kristensen, T.K.; Chimbari, M. Susceptibility of freshwater snails to the amphistome Calicophoron microbothrium and the influence of the species on susceptibility of Bulinus tropicus to Schistosoma haematobium and Schistosoma mattheei infections. J. Parasitol. 2002, 88, 880–883. [Google Scholar] [CrossRef]
- Mereta, S.T.; Bedewi, J.; Yewhalaw, D.; Mandefro, B.; Abdie, Y.; Tegegne, D.; Birke, W.; Mulat, W.L.; Kloos, H. Environmental determinants of distribution of freshwater snails and trematode infection in the Omo Gibe River Basin, southwest Ethiopia. Infect. Dis. Poverty 2019, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Nzalawahe, J.; Kassuku, A.A.; Stothard, J.R.; Coles, G.C.; Eisler, M.C. Associations between trematode infections in cattle and freshwater snails in highland and lowland areas of Iringa Rural District, Tanzania. Parasitology 2015, 142, 1430–1439. [Google Scholar] [CrossRef] [PubMed]
- Chibwana, F.; Nkwengulila, G. A faunistic survey of digenean larvae infecting freshwater snails Biomphalaria, Radix and Bulinus species in the Lake Victoria and Mindu dam, Morogoro in Tanzania. Tanzan. J. Sci. 2017, 43, 1–13. [Google Scholar]
- Wright, C.A.; Rollinson, D.; Goll, P.H. Parasites in Bulinus senegalensis (Mollusca: Planorbidae) and their detection. Parasitology 1979, 79, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.A.; Southgate, V.; Howard, G. A note on the life-cycles of some amphistome flukes in Zambia. J. Helminthol. 1979, 53, 251–252. [Google Scholar] [CrossRef]
- Southgate, V.R.; Brown, D.S.; Rollinson, D.; Ross, G.C.; Knowles, R.J. Bulinus tropicus from Central Kenya acting as a host for Schistosoma bovis. Z. Parasitenkd. 1985, 71, 61–69. [Google Scholar] [CrossRef]
- Southgate, V.R.; Brown, D.S.; Warlow, A.; Knowles, R.J.; Jones, A. The influence of Calicophoron microbothrium on the susceptibility of Bulinus tropicus to Schistosoma bovis. Parasitol. Res. 1989, 75, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Kane, R.A.; Stothard, J.R.; Emery, A.M.; Rollinson, D. Molecular characterization of freshwater snails in the genus Bulinus: A role for barcodes? Parasite Vectors 2008, 1, 15. [Google Scholar] [CrossRef]
- Molaba, G.G.; Molefe-Nyembe, N.I.; Taioe, O.M.; Mofokeng, L.S.; Thekisoe, O.M.; Mtshali, K. Molecular detection of Fasciola, Schistosoma and Paramphistomum species from freshwater snails occurring in Gauteng and Free State provinces, South Africa. Vet. Parasitol. 2023, 320, 109978. [Google Scholar] [CrossRef]
- Schols, R. Gastropod-Borne Trematode Communities of Man-Made Reservoirs in Zimbabwe, with a Special Focus on Fasciola and Schistosoma Helminth Parasites. Master of Science Thesis, KU Leuven, Leuven, Belgium, 2019. [Google Scholar]
- Carolus, H.; Muzarabani, K.C.; Hammoud, C.; Schols, R.; Volckaert, F.A.; Barson, M.; Huyse, T. A cascade of biological invasions and parasite spillback in man-made Lake Kariba. Sci. Total Environ. 2019, 659, 1283–1292. [Google Scholar] [CrossRef]
- Tookhy, N.A.; Isa, N.M.M.; Mansor, R.; Rahaman, Y.A.; Ahmad, N.I.; Bui, D.T.; Idris, L.H.; Hamzah, N.H.; Zulkifli, N. Morphological and molecular identification of lymnaeid snail and trematodes cercariae in different water bodies in Perak, Malaysia. Parasitol. Res. 2023, 122, 1475–1488. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.S. Freshwater Snails of Africa and Their Medical Importance, 2nd ed.; Taylor and Francis: London, UK, 1994. [Google Scholar]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. In Nucleic Acids Symposium Series; Information Retrieval Ltd.: London, UK, 1999; Volume 41, pp. c1979–c2000. [Google Scholar]
- Kumar, S.; Stecher, G.; Tumara, K. Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Min, F.; Wang, J.; Liu, X.; Yuan, Y.; Guo, Y.; Zhu, K.; Chai, Z.; Zhang, Y.; Li, S. Environmental Factors Affecting Freshwater Snail Intermediate Hosts in Shenzhen and Adjacent Region, South China. Trop. Med. Infect. 2022, 7, 426. [Google Scholar] [CrossRef]
- Middelboe, A.L.; Markager, S. Depth limits and minimum light requirements of freshwater macrophytes. Freshw. Biol. 1997, 37, 553–568. [Google Scholar] [CrossRef]
- Makura, O.; Kristensen, T.K. National freshwater survey of Zimbabwe. In Proceedings of the Tenth International Malacological Congress; Meier-Brook, C., Ed.; Institute of Tropical Medicine, University of Tübingen: Tübingen, Germany, 1991; Volume 47, pp. 227–232. [Google Scholar]
- Appleton, C.C. Alien and invasive fresh water Gastropoda in South Africa. Afr. J. Aquat. Sci. 2003, 28, 69–81. [Google Scholar] [CrossRef]
- Schols, R.; Carolus, H.; Hammoud, C.; Muzarabani, K.C.; Barson, M.; Huyse, T. Invasive snails, parasite spillback, and potential parasite spillover drive parasitic diseases of Hippopotamus amphibius in artificial lakes of Zimbabwe. BMC Biol. 2021, 19, 160. [Google Scholar] [CrossRef]
- De Kock, K.N.; Wolmarans, C.T. Distribution and habitats of the alien invader freshwater snail Physa acuta in South Africa. Water SA 2007, 33, 717–722. [Google Scholar] [CrossRef]
- Dinnik, J. Paramphistomum phillerouxi sp. nov.(Trematoda: Paramphistomatidae) and its development in Bulinus forskalii. J. Helminthol. 1961, 35, 69–90. [Google Scholar] [CrossRef]
- Dinnik, J. The snail hosts of certain Paramphistomatidae and Gastrothylacidae (Trematoda) discovered by the late Dr. PL LeRoux in Africa. J. Helminthol. 1965, 39, 141–150. [Google Scholar] [CrossRef]
- Graber, M.; Daynes, P. Mollusques vecteurs de trematodeses humanies et animals en Ethiopie. Rev. D’elevage Med. Vet. Pays Trop. 1974, 27, 307–322. [Google Scholar] [CrossRef]
- Mukaratirwa, S.; Munjere, I.F.; Takawira, M.; Chingwena, G. Susceptibility of 7 freshwater gastropod species in Zimbabwe to infection with Gastrodiscus aegyptiacus (Cobbold, 1876) Looss, 1896. J. S. Afr. Vet. Assoc. 2004, 75, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Cumberlidge, N.; Rollinson, D.; Vercruysse, J.; Tchuem Tchuenté, L.A.; Webster, B.; Clark, P.F. Paragonimus and paragonimiasis in West and Central Africa: Unresolved questions. Parasitology 2018, 145, 1748–1757. [Google Scholar] [CrossRef] [PubMed]
- Procop, G.W. North American paragonimiasis (caused by Paragonimus kellicotti) in the context of global paragonimiasis. Clin. Microbiol. Rev. 2009, 22, 415–446. [Google Scholar] [CrossRef] [PubMed]
- Appleton, C.C. Paragonimiasis in KwaZulu-Natal Province, South Africa. J. Helminthol. 2014, 88, 123–128. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).