Abstract
The inoculation of Epichloë endophytes into modern cereals, resulting in systemic infection, depends on the genetics of both the host and the endophyte strain deployed. Until very recently, the only modern cereal to have been infected with Epichloë, in which normal phenotype seed-transmitted associations were achieved, is rye (Secale cereale). Whilst minor in-roads have been achieved in infecting hexaploid wheat (Triticum aestivum), the phenotypes of these associations have all been extremely poor, including host death and stunting. To identify host genetic factors that may impact the compatibility of Epichloë infection in wheat, wheat–alien chromosome addition/substitution lines were inoculated with Epichloë, and the phenotypes of infected plants were assessed. Symbioses were identified whereby infected wheat plants were phenotypically like uninfected controls. These plants completed their full lifecycle, including the vertical transmission of Epichloë into the next generation of grain, and represent the first ever compatible wheat–Epichloë associations to be created.
1. Introduction
Epichloë endophytes, particularly asexual forms, have important roles in pastoral agricultural systems in the Americas, Australia and New Zealand [1]. Selected strains add value to grass-based forage systems by providing both biotic and abiotic stress resistance. The symbioses of anamorph-typified Epichloë form are mutualistic in that both the fungus and the host grass benefit from the association. The fungus benefits from a biological niche with few if any competing organisms, a source of nutrients in the host’s apoplastic fluid and a mechanism for vicarious dispersal via the host seed. The host benefits from the range of secondary metabolites the fungus produces in the form of alkaloids, many of which have individual and/or multiple activities against different classes of organism [1].
Modern Hordeeae (Triticeae) cereal grasses such as wheat (Triticum aestivum), barley (Hordeum vulgare) and rye (Secale cereale) do not host Epichloë endophytes, although grasses of some genera within the tribe, such as Elymus, Hordeum and Leymus, do [2,3,4,5,6,7,8,9].
Both organism classes, Epichloë endophytes and cereal grasses, are of great importance in their own contexts, and the prospect of forming functioning symbioses offers the potential to improve cereal production systems. Previous research [10] demonstrated varying outcomes of symbiosis. Whereas symbioses established with rye (Secale cereale) showed phenotypes ranging from normal to compromised, those involving modern hexaploid wheat (Triticum aestivum) were always poor, exhibiting a dwarfed phenotype. This likely reflects that outcrossing species such as rye present a range of genotypes in an inoculated population, whereas inbred species such as wheat are a genetically narrow host population target.
Wild relatives of wheat are recognized as having potential as sources of genes for improving wheat performance; for example, the introduction of pathogen resistance using rye chromatin [11]. The best-known example of interspecific chromatin being transferred into a crop is the translocation of rye chromosome arm 1RS to wheat chromosome arm 1BL [12]. It is possible to add or substitute entire chromosomes, chromosome areas or chromatin segments. The rearrangement of wheat chromosomes in this way has constituted an important aspect of wheat improvement for over 50 years [13]. The transfer is effected by producing an amphidiploid, a hybrid between the two species with at least one complete diploid set of chromosomes from each species, or a partial amphidiploid, and then producing individual chromosome addition lines. Following this, the centric breakage fusion tendency can be exploited to transfer a whole alien chromosome arm. Strategies can also be deployed for transferring alien segments that are smaller than complete chromosome arms [14]. Where genetic diversity is limited, such as in highly selected and inbred wheat lines, the introduction of alien genetics from related species offers a means to broaden the genetic base of the population and increase the possibilities for the selection of desirable traits [15,16,17].
To improve the outcome of synthetic Epichloë symbioses with wheat, we utilized alien chromosome additions and substitutions from both rye and wild grass species from within the Hordeeae tribe (Supplementary Table S1).
2. Materials and Methods
2.1. Endophyte Strain
Epichloë bromicola AR3060 (AgResearch strain 3060) was isolated from an Elymus dahuricus subspecies excelsus plant and used in inoculation experiments in Japan following isolation from the parent plant as previously described [18]. Epichloë bromicola AR3002 was isolated from an Elymus dahuricus plant and used in inoculation experiments in New Zealand. Fungal cultures were grown on potato dextrose agar as described by Fleetwood et al., 2007 [19].
2.2. Inoculation of Wheat Alien Chromosome Substitution and Addition Lines
Experiments involving the inoculation of wheat lines sourced from the Tottori Alien Chromosome Bank Of Wheat (TACBOW) at the National BioResource Project (NBRP) Komugiwere performed as follows.
Up to five seeds from each of 155 wheat lines with various alien chromosome introductions from species of Aegilops, Agropyron, Elymus, Hordeum, Leymus, Psathyrostachys, Haynaldia and Secale, along with the wheat cultivars ‘Monad’ and ‘Chinese Spring’ as controls (Supplementary Table S1), were inoculated, as described by Latch and Christensen (1985) [20], using strain AR3060, which had previously been demonstrated to infect the wheat cultivar “Monad”, albeit with a compromised host phenotype [10]. Plants were grown for ca. 6 weeks in commercial potting mix before identifying infected individuals through tissue-print immunoblotting as previously described [18]. Plants that were demonstrated to be infected with Epichloë by immunoblot were grown for a further 3 months under glasshouse conditions to complete their full life cycle. Endophyte-free plants of the same lines were also grown under equivalent conditions to serve as direct comparisons to the infected plants.
In subsequent experiments performed in Palmerston North, New Zealand, a subset of lines (Supplementary Table S2) that supported compatible AR3060 infection were inoculated with E. bromicola strain AR3002, as, unlike AR3060, this strain does not produce the mammalian toxin ergovaline.
2.3. Phenotyping
Plants were phenotyped at the completion of their life cycle and those that were originally infected, based upon earlier immunoblotting results, were confirmed as remaining Epichloë infected by microscopy and/or fungal isolation, as described below. Infected individuals were phenotypically compared to plants of the same line that did not become infected.
2.4. Epidermal Leaf Peel
Tillers were selected from mature plants for endophyte detection. Clean, live sheath tissue was placed under a dissecting microscope at 16× magnification with the adaxial epidermis facing up. A shallow transverse cut was made with a scalpel and the epidermis was gently lifted, separated, and pulled off the sheath. Tissue was mounted in a drop of aniline blue dye (glycerol 50%; lactic acid 25%; water 24.95%; aniline blue 0.05%), heated over a naked flame and examined at 100× and 400× using a compound microscope.
2.5. Seed Squash
Grain was covered with a 5% sodium hydroxide solution and left to imbibe overnight. The following day, the solution was decanted and the samples thoroughly rinsed with tap water. Samples were covered with Garner’s solution (0.325 g aniline blue, 100 mL water and 50 mL 85% lactic acid) and heated to boiling on a hot plate. After cooling, the softened grain was mounted on a microscope slide, a cover slip was placed over the grain and gentle, even pressure was applied, squashing the preparation. The preparations were then examined under a compound light microscope at 100× and 400× magnification.
2.6. Fungal Isolation
Fungus was isolated from plants following surface sterilization of plant tissue, as described by Christensen, et al. (2002) [21]. Blade tissue was surface sterilized by quick rinse with 96% ethanol and a 1 min soak in a sodium hypochlorite solution (10% Janola: 42 g/L NaOCl domestic bleach), followed by rinsing twice in sterile water. Tissue was plated on to potato dextrose agar containing 5 µg/mL tetracycline. Plates were incubated at 22–25 °C for 3–5 days.
3. Results and Discussion
Epichloë Infection of Wheat Alien Chromosome Addition/Substitution Lines
Of the 157 lines inoculated, immunoblot results showed that 97 lines had one or more Epichloë-infected plants (Table 1), with the remaining lines being uninfected. All lines were subsequently phenotyped at maturity. Uninfected lines were universally tall and floral with fully developed grain and, significantly, wheat lines with no alien chromosome introductions (Chinese Spring and Monad) were dwarfed (Figure 1) in a similar manner to that previously reported [10]. Infected plants of chromosome addition/substitution lines included tall floral individuals that were similar to uninfected plants of the same line and abnormal phenotype plants that were short in stature and often failed to produce inflorescences. Additionally, tall floral plants delayed in their maturation that were exemplified by a ‘stay green’ phenotype and a protracted elaboration and maturation of the floral spike were identified. Additional evidence for endophyte infection was obtained for a sub-set of plants from each of the three infection phenotypes using a combination of fungal isolation and microscopy of leaf and seed material. Although not all the plants that had a delayed maturation were confirmed to be infected in this way, this phenotype was indicative of endophyte infection and plants were classified as such for further analysis.

Table 1.
Wheat lines, alien introgressions and Epichloë-infection phenotypes.

Figure 1.
Wheat cultivars (a) Chinese Spring and (b) Monad, infected (E+) or uninfected (E−) with Epichloë bromicola strain AR3060.
An analysis of the alien genetics associated with TACBOW lines displaying tall floral infection phenotypes (Table 1) was performed in terms of the donor species and which of the seven homoeologous groups (based upon wheat) the chromosome belonged to, aiming to reveal patterns in host genetics associated with the phenotypic outcomes. We initially looked specifically at the full wheat–rye chromosome addition lines comprising seven “Chinese Spring”-based lines with 1R-7R additions from S. cereale. Tall floral infected plants were obtained for wheat–rye 1R, 3R and 7R addition lines, with abnormal phenotypes or no infection occurring with the remaining four addition lines (2R, 4R, 5R, 6R). This result suggests that there may be genetic factors associated with alien chromosomes from homoeologous groups 1, 3 and 7 and, indeed, chromosome additions or substitutions from other grasses, distinct from rye, with these homoeologous groups were also enriched for tall floral infection phenotypes (Table 1). For example, the inoculation of the line TACBOW11, a line with alien genetics representing homoeologous group 3, produced tall floral infected plants. The underlying genetics of this line is T. aestivum cv. Chinese Spring Leymus racemosus H substitution (20″ + 1″ [H]). Line TACBOW5 was another example of tall floral infected plants involving the L. racemosus H chromosome, but as an addition rather than a substitution (21″ + 1″ [H]). Line TACBOW7 also gave rise to infected large phenotype plants but with a delayed maturation. Despite this, grain was fully filled and was confirmed to contain endophyte by microscopy (Figure 2). The underlying genetics of this line is T. aestivum cv. Chinese Spring L. racemosus J addition (20″ + 1″ [J]). Leymus J and Leymus H chromosomes have been shown, using RFLP markers, to share some syntenic regions [17,22], which also have homology with wheat chromosomes belonging to homoeologous groups 1, 3 and 7. Interestingly, chromosomes 1, 3 and 7 have been described as sharing a “close genetic relationship” [23] (pp. 29–45) and [24] (p. 73).
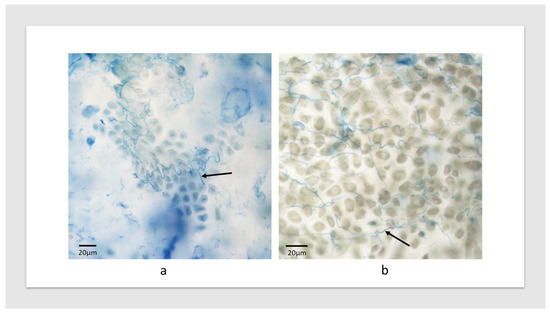
Figure 2.
Seed of (a) TACBOW11 and (b) TACBOW67 infected with Epichloë bromicola strain AR3060. Arrows indicate stained hyphae.
Several lines were identified in which, like the Leymus J addition line described above, maturation was somewhat delayed. Some of these lines were confirmed as endophytes infected by isolations, leaf sheath microscopy or seed squash (Table 1). The remainder are assumed to be infected, as uninfected lines were universally tall and floral, with fully developed grain. These delayed phenotype lines have certain genetics in common with each other and to those plants showing normal, mature seed-transmitting phenotypes. As described above, the Leymus H and J chromosomes share some synteny, and with respect to wheat, share homology with chromosomes belonging to homoeologous groups 1, 3 and 7. In addition to the Leymus H (substitution) and Leymus J (addition) lines, a Leymus N line was also confirmed to be infected (by isolation) and the Leymus N chromosome also shares homology with homoeologous groups 3 and 7. Further to this, we confirmed that many of the delayed maturation infection phenotypes also contain alien additions from homoeologous groups 1, 3 or 7 (Table 1). These include, in addition to the 1R, 3R and 7R additions from Secale described above, the 1U, 3U and 7U additions from Aegilops and 1H and 7H additions from Elymus. Whilst these lines are not exclusive and the evolution of the Hordeeae is complex, there seems to be some evidence that genes from wild species located on chromosomes belonging to homoeologous groups 1, 3 and 7 may be beneficial in terms of their compatibility with Epichloë.
The ability to infect rye with Epichloë and obtain normal phenotype plants [10], together with TACBOW lines involving rye 1R, 3R and 7R chromosome additions (this study), suggests that wheat–rye hybrids might also contain the genetics required to form floral associations with Epichloë. Whilst some limited attempts have been made to infect commercial Triticale (T. aestivum × S. cereale) lines (Simpson, unpublished), in the present study, two T. turgidum × S. cereale amphidiploid lines (TACBOW66 and TACBOW67) containing all seven rye chromosomes became infected and produced inflorescences.
In subsequent inoculations, performed in New Zealand, with the non-toxic strain AR3002 into a sub-set of TACBOW lines, we also saw a range of infection phenotypes. Examples of these are shown in (Figure 3) and plants were confirmed by seed squash to be transmitting Epichloë into the grain.

Figure 3.
(a) TACBOW232- and (b) TACBOW288-infected (E+) and uninfected (E−) with Epichloë bromicola strain AR3002.
Whether the host genetics required for Epichloë compatibility will apply to all E. bromicola strains beyond those described here remains to be tested, as functional differences based upon the Epichloë genotype have been observed (unpublished data). Regardless, the genetics underlying Epichloë compatibility needs to be introgressed into modern cultivars since Chinese Spring is not agronomically useful.
4. Conclusions
The inoculation of commercial hexaploid wheat with Epichloë has, to date, been unsuccessful, with the resulting infected plants having poor phenotypes. This result was confirmed here with inoculations of the wheat cultivars Chinese Spring and Monad (no chromosome additions or substitutions). However, in this study, the inoculation of Chinese Spring-based wheat containing certain alien chromosome additions or substitutions sourced from rye and wild grass species (many of which also host Epichloë) were successful. This suggests that lines containing chromosome additions/substitutions, where infection produced tall floral plants, may have a genetic profile underlying this. Our analysis of these lines indicates that alien chromosome introgressions from homoeologous groups 1, 3 and 7 may be important for the improved compatibility between wheat and Epichloë. However, whilst infected normal phenotype lines were enriched for 1, 3 and 7 chromosome additions/substitutions, some infected plants displaying normal phenotypes consisted of lines carrying chromosome additions that did not belong to homoeologous groups 1, 3 or 7. Further work will be required to prove the role of 1, 3, 7 homoeologous group genetics and to determine if there are other commonalties present on some of the non-1, -3 or -7 chromosome groups where infection phenotypes were not compromised.
5. Patents
A patent (International application number PCT/IB2019/051395) resulted from the work reported in this manuscript.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jof10060384/s1, Table S1. Wheat lines used in study; Table S2.
Author Contributions
Conceptualization, R.D.J., W.R.S. and H.T.; methodology, R.D.J. and W.R.S.; formal analysis, R.D.J. and W.R.S.; investigation, R.D.J. and W.R.S.; resources, H.T. and D.E.H.; data curation, R.D.J. and W.R.S.; writing—original draft preparation, R.D.J. and W.R.S.; writing—review and editing, R.D.J., W.R.S., H.T. and D.E.H.; funding acquisition, R.D.J. and D.E.H. All authors have read and agreed to the published version of the manuscript.
Funding
Ministry of Business, Innovation and Employment, New Zealand (contract C10X0815), Grasslanz Technology Limited (PN23098), the Foundation for Arable Research and the Grains Research and Development Corporation (GTL1709-001RTX) for funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The TACBOW lines are available from the National BioResouce Project (NBRP)-Komugi: https://shigen.nig.ac.jp/wheat/komugi/. URL accessed on 27 May 2024.
Acknowledgments
We acknowledge the assistance of staff at Margot Forde Germplasm Centre, New Zealand and The Arid Land Research Center, Japan.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Johnson, L.J.; de Bonth, A.C.M.; Briggs, L.R.; Caradus, J.R.; Finch, S.C.; Fleetwood, D.J.; Fletcher, L.R.; Hume, D.E.; Johnson, R.D.; Popay, A.J.; et al. The exploitation of epichloae endophytes for agricultural benefit. Fungal Divers. 2013, 60, 171–188. [Google Scholar] [CrossRef]
- Wilson, A.D. First report of clavicipitaceous anamorphic endophytes in Hordeum species. Plant Dis. 1991, 75, 215D. [Google Scholar] [CrossRef]
- Wilson, A.D. Clavicipitaceous Anamorphic Endophytes in Hordeum germplasm. Plant Pathol. J. 2007, 6, 1–13. [Google Scholar] [CrossRef]
- Kuldau, G.; Bacon, C. Clavicipitaceous endophytes: Their ability to enhance resistance of grasses to multiple stresses. Biol. Control. 2008, 46, 57–71. [Google Scholar] [CrossRef]
- Li, W.; Ji, Y.-L.; Yu, H.-S.; Wang, Z.-W. A new species of Epichloe symbiotic with Chinese grasses. Mycologia 2006, 98, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.K.; Gao, Y.B.; Xu, H.; Su, D.; Zhang, X.; Wang, Y.H.; Lin, F.; Chen, L.; Nie, L.Y.; Ren, A.Z. Occurrence of endophytes in grasses native to northern China. Grass Forage Sci. 2006, 61, 422–429. [Google Scholar] [CrossRef]
- Yan, K.; Yanling, J.; Xianghui, S.; Lihui, Z.; Wei, L.; Hanshou, Y.; Zhiwei, W. Taxonomy of Neotyphodium endophytes of Chinese native Roegneria plants. Mycologia 2009, 101, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Ji, Y.-L.; Zhang, C.-W.; Wang, Z.-W. Neotyphodium sinicum, from several Roegneria species throughout China, provides insights into the evolution of asexual endophytes. Symbiosis 2011, 54, 37–45. [Google Scholar] [CrossRef]
- Zhu, M.-J.; Ren, A.-Z.; Wen, W.; Gao, Y.-B. Diversity and taxonomy of endophytes from Leymus chinensis in the Inner Mongolia steppe of China. FEMS Microbiol. Lett. 2013, 340, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Simpson, W.R.; Faville, M.J.; Moraga, R.A.; Williams, W.M.; McManus, M.T.; Johnson, R.D. Epichloë fungal endophytes and the formation of synthetic symbioses in Hordeeae (=Triticeae) grasses. J. Syst. Evol. 2014, 52, 794–806. [Google Scholar] [CrossRef]
- Crespo-Herrera, L.A.; Garkava-Gustavsson, L.; Åhman, I. A systematic review of rye (Secale cereale L.) as a source of resistance to pathogens and pests in wheat (Triticum aestivum L.). Hereditas 2017, 154, 14. [Google Scholar] [CrossRef] [PubMed]
- Lukaszewski, A.J. Introgressions between wheat and rye. In Alien Introgression in Wheat: Cytogenetics, Molecular Biology, and Genomics; Springer: Berlin/Heidelberg, Germany, 2015; pp. 163–189. [Google Scholar] [CrossRef]
- Graybosch, R. Mini Review: Uneasy Unions: Quality Effects of Rye Chromatin Transfers to Wheat. J. Cereal Sci. 2001, 33, 3–16. [Google Scholar] [CrossRef]
- Qi, L.; Friebe, B.; Zhang, P.; Gill, B.S. Homoeologous recombination, chromosome engineering and crop improvement. Chromosom. Res. 2007, 15, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Shi, Q.; Liu, Y.; Su, H.; Zhang, J.; Wang, M.; Wang, C.; Wang, J.; Zhang, K.; Fu, S.; et al. Systemic development of wheat-Thinopyrum elongatum translocation lines and their deployment in wheat breeding for Fusarium head blight resistance. Plant J. 2023, 114, 1475–1489. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, G.V.; Tomohiro, B.; Masahiro, K.; Osamu, I.; Samejima, H.; Wang, H.Y.; Pearse, S.J.; Gopalakrishnan, S.; Nakahara, K.; Hossain, A.K.M.Z.; et al. Can biological nitrification inhibition (BNI) genes from perennial Leymus racemosus (Triticeae) combat nitrification in wheat farming? Plant Soil 2007, 299, 55–64. [Google Scholar] [CrossRef]
- Kishii, M.; Yamada, T.; Sasakuma, T.; Tsujimoto, H. Production of wheat–Leymus racemosus chromosome addition lines. Theor. Appl. Genet. 2004, 109, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Simpson, W.R.; Schmid, J.; Singh, J.; Faville, M.J.; Johnson, R.D. A morphological change in the fungal symbiont Neotyphodium lolii induces dwarfing in its host plant Lolium perenne. Fungal Biol. 2012, 116, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Fleetwood, D.J.; Scott, B.; Lane, G.A.; Tanaka, A.; Johnson, R.D. A complex ergovaline gene cluster in Epichloë endophytes of grasses. Appl. Environ. Microbiol. 2007, 73, 2571–2579. [Google Scholar] [CrossRef]
- Latch, G.C.M.; Christensen, M.J. Artificial infection of grasses with endophytes. Ann. Appl. Biol. 1985, 107, 17–24. [Google Scholar] [CrossRef]
- Christensen, M.J.; Bennett, R.J.; Schmid, J. Growth of Epichloë/Neotyphodium and p-endophytes in leaves of Lolium and Festuca grasses. Mycol. Res. 2002, 106, 93–106. [Google Scholar] [CrossRef]
- Edet, O.U.; Kim, J.-S.; Okamoto, M.; Hanada, K.; Takeda, T.; Kishii, M.; Gorafi, Y.S.A.; Tsujimoto, H. Efficient anchoring of alien chromosome segments introgressed into bread wheat by new Leymus racemosus genome-based markers. BMC Genet. 2018, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Sears, E. Nullisomic-tetrasomic combinations in hexaploid wheat. In Chromosome Manipulations and Plant Genetics: The Contributions to a Symposium Held during the Tenth International Botanical Congress Edinburgh 1987; Springer Science+Business Media: Dordrecht, The Netherlands; Berlin/Heidelberg, Germany, 1966. [Google Scholar] [CrossRef]
- Lupton, F. Wheat Breeding-Its Scientific Basis; Springer: Berlin/Heidelberg, Germany, 1987. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).