Abstract
The recent increase in the number of rare yeasts isolated from clinical specimens is a cause for concern, requiring accurate identification of these yeasts and assessment of their antifungal susceptibility to guide treatment. In this regard, we identified 196 rare yeasts isolated from various clinical specimens, mostly urine and respiratory tract specimens of patients hospitalized in intensive care unit and wards, using MALDI-TOF MS, and assessed their susceptibility to amphotericin B, fluconazole, voriconazole, posaconazole, itraconazole, isavuconazole and anidulafungin using the EUCAST broth microdilution method. Among the rare yeast species we isolated, Candida lusitaniae (13.8%) was the most common, followed by Magnusiomyces capitatus (13.3%), Candida fabianii (12.2%), and Trichosporon asahii (11.7%). Antifungal susceptibility testing revealed high echinocandin MIC values against Magnusiomyces spp., Trichosporon spp., and Rhodotorula mucilaginosa isolates. Similarly, we found high MIC values for fluconazole against the isolates of Magnusiomyces spp., T. asahii, R. mucilaginosa, and several Candida spp., including Candida guilliermondii, Candida pararugosa, Candida rugosa, Candida pelliculosa, Candida norvegensis, and Candida fabianii. We found similar MIC values across phylogenetically closely related species. In conclusion, prompt identification of rare yeasts and assessment of their antifungal susceptibilities are essential for effective treatment of the infections they cause.
1. Introduction
Rare yeasts that cause superficial infections in hosts with normal immune systems but can cause invasive fungal infections associated with high mortality and morbidity rates in immunosuppressed hosts or the presence of underlying risk factors are being isolated more commonly [1,2].
All phenotypic identification systems, matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS), and molecular approaches can contribute to the identification of rare yeasts [2]. However, traditional fungal identification methods based on examination of morphological and phenotypic characteristics are complicated by the diversity of organisms that can cause infections [3,4]. In MALDI-TOF MS systems, which are widely used in microbiology laboratories today, the difficulty of updating the nomenclature in the database prevents clinical microbiology laboratories from successfully adapting to new species names [4].
The widespread use of molecular approaches in fungal identification in recent years has led to radical changes in fungal nomenclature and taxonomy. Long-recognized species have been transposed to new genera based on genotypic comparisons, or new genera and species have been created that include new organisms identified by detailed phylogenetic analyses [3,4]. Recent changes to the Fungal Nomenclature Code have also led to dramatic changes in the nomenclature of medically important molds and yeasts [5]. The binomial nomenclature system, which stipulated giving separate names to anamorphic (asexual) and teleomorphic (sexual) forms of fungi, was abandoned as of 1 January 2013 [5,6,7].
To reduce the instability caused by unnecessary and temporary nomenclature in this process, working groups and committees established under the auspices of the International Commission on the Taxonomy of Fungi (ICTF) and the Nomenclature Committee for Fungi (NCF) have published lists of protected and rejected names of key species/genera for which definitive changes have been approved [3]. Information on the classical names most commonly used in clinical laboratories and the recommended names to be reported for clinical use can be accessed from the Atlas of Clinical Fungi, Index Fungorum, and MycoBank [8,9,10].
As with their nomenclature, antifungal susceptibility values for rare yeasts are also not well established. In fact, the European Committee on Antimicrobial Susceptibility Testing (EUCAST) has not reported clinical breakpoints for antifungal drugs against rare yeasts due to insufficient evidence, except for commonly isolated Candida species and Cryptococcus neoformans [11]. On the other hand, based on the study by Astvad et al., EUCAST developed a classification for the criteria for assessing antifungal drugs to be used against different fungal species based on two key assumptions [12]. The first assumption is that genetically related isolates would have similar pathogenicities and intrinsic susceptibility patterns. The second assumption is that rare yeasts, even those not phylogenetically related to other species, will likely respond to treatment, provided their minimum inhibitory concentration (MIC) values are similar to those observed in wild-type isolates of other, more commonly isolated species. Current treatment recommendations based on in vivo efficacy data and clinical experience were also taken into account in the EUCAST classification [11,13]. Accordingly, provisional MIC values were determined to guide amphotericin B, anidulafungin, fluconazole, and voriconazole treatments against thirty different yeasts until official clinical breakpoints are set by EUCAST [12].
In view of the foregoing, this study was carried out to identify rare yeasts isolated from various clinical specimens and assess their antifungal susceptibilities to amphotericin B, fluconazole, voriconazole, posaconazole, itraconazole, isavuconazole, and anidulafungin.
2. Materials and Methods
2.1. Fungal Isolates
The study material consisted of 196 rare yeast collection isolates sent to the ISLAB-2 Central Mycology Laboratory for identification and antifungal susceptibility testing from hospitals affiliated with the 2nd Service Region of the Istanbul Provincial Health Directorate between 1 June 2023 and 30 October 2024, and identified by matrix-assisted laser desorption ionization–time of flight mass spectrometry (MALDI-TOF MS) (VITEK MS and VITEK MS PRIME, bioMérieux, Marcy l’Etoile, France). Identification of the isolates was supported by their macroscopic and microscopic appearances on Sabouroud Dextrose agar [(SDA), (RTA, İstanbul, Turkey)] and Candida chromogenic agar (RTA, İstanbul, Turkey). The isolates were stored at −80 °C until testing, revived by passage twice in SDA prior to testing, and incubated at 36 ± 1 °C for 18–24 h.
2.2. Antifungal Susceptibility Testing
Antifungal susceptibility tests were carried out using the broth microdilution method per EUCAST E.Def 7.4 guidelines [14]. The antifungal agents tested were obtained from Sigma–Aldrich Chemical Co., St. Louis, MO, USA. The MIC values of amphotericin B, fluconazole, posaconazole, itraconazole, voriconazole, isavuconazole, and anidulafungin against the isolates were determined. Growth was assessed visually after 24-h incubation at 37 °C and spectrophotometrically at 450 nm using a Multiskan Go (ThermoFisherScientific, Waltham, MA, USA) microplate reader. The MIC value was defined as the lowest concentration that caused a 90% reduction in growth for amphotericin B and a 50% reduction for other drugs compared to growth not involving an antifungal agent [15].
2.3. Statistical Analysis
SPSS 30.0 (Statistical Product and Service Solutions for Windows, Version 30.0, IBM Corp., Armonk, NY, USA, 2024) software package was used in the statistical analyses of the collected data. The results of the statistical analyses were expressed using descriptive statistics, i.e., mean ± standard deviation values and median with minimum and maximum values in the case of continuous variables, and frequencies (n) and percentage (%) values in the case of categorical variables. In comparing the differences in variables between the groups, the Mann–Whitney U test was used for numerical variables, and the chi-squared test was used for categorical variables. Probability (p) statistics of <0.05 were deemed to indicate statistical significance.
3. Results
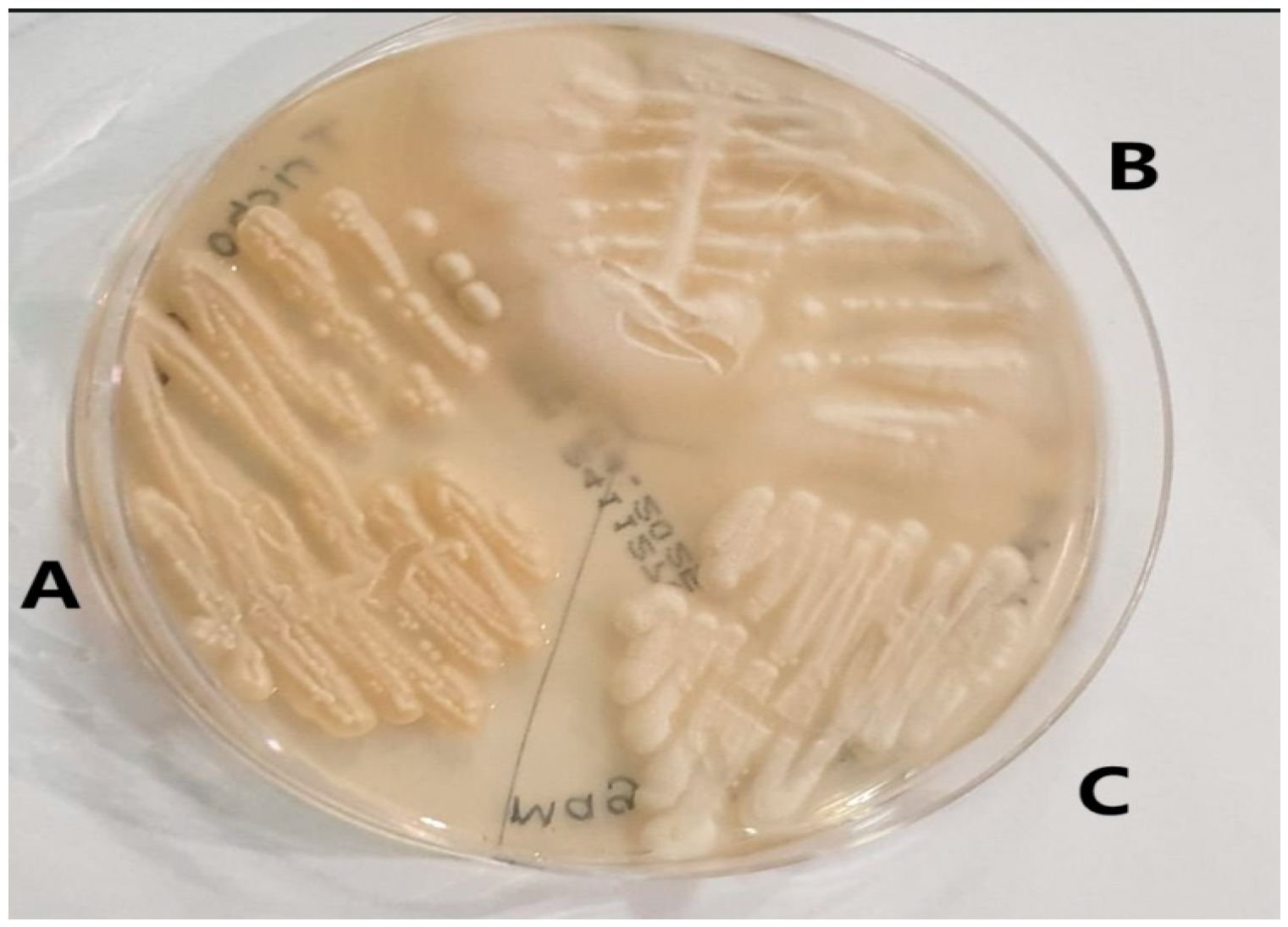
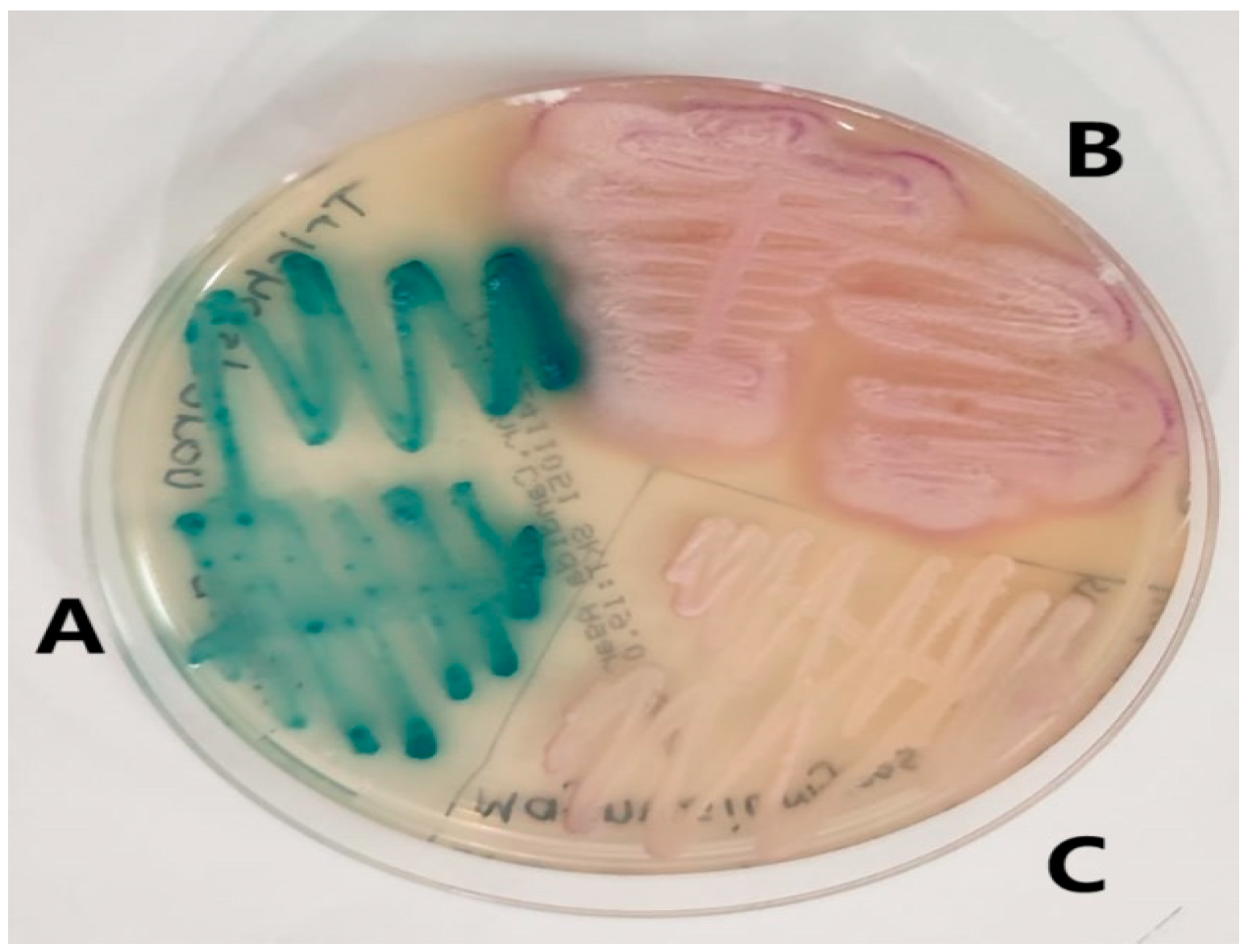
We identified 196 rare yeasts and studied their antifungal drug susceptibility during the study period. Compared to other common Candida species, i.e., Candida albicans complex (1685), Candida glabrata complex (Nakaseomyces glabratus) (653), Candida tropicalis (441), Candida auris (Candidozyma auris) (417), Candida parapsilosis (307), Candida kefyr (Kluvyeromyces marxianus) (243), and Candida krusei (Issatchenkia orientalis) (76), rare yeasts accounted for 4.9% of all growth samples. The names and isolation rates of the 196 rare yeasts that make up the study material are shown in Table 1. The appearance of some yeasts isolated in the study on SDA is shown in Figure 1, and their appearance on Candida chromogenic agar is shown in Figure 2.

Table 1.
Rare yeasts isolated in the study.
Figure 1.
Images of rare yeasts on Sabouraud Dextrose agar. (A) Trichosporon asahii. (B) Geotrichum capitatum. (C) Magsusiomyces capitatus (From the study).
Figure 2.
Images of rare yeasts on Candida chromogenic agar. (A) Trichosporon asahii. (B) Geotrichum capitatum. (C) Magsusiomyces capitatus (From the study).
We isolated the rare yeasts mostly from the urine and respiratory tract specimens of patients hospitalized in the intensive care unit (ICU) and wards. No significant difference was found between the urine samples of patients hospitalized in the ICU and wards in terms of the isolation rates of rare yeasts compared to other common Candida species (p = 0.403). On the other hand, a significant difference was found between the respiratory tract samples of patients hospitalized in the ICU and wards in terms of the isolation rates of rare yeasts compared to other common Candida species (p < 0.001). The isolation rates of other Candida species were significantly higher than those of rare yeasts in the blood samples of patients hospitalized in the ICU and wards (p = 0.004). Among the rare yeast species we isolated, Candida lusitaniae (Clavispora lusitaniae) (13.8%) was the most common, followed by Saprochaete capitata (Magnusiomyces capitatus) (13.3%), Candida fabianii (Cyberlindnera fabianii). (12.2%), and Trichosporon asahii (11.7%). Demographic characteristics of patients from whom rare yeasts were isolated and isolation rates by sample type are shown in Table 2.

Table 2.
Demographic Distribution of Rare Yeasts.
EUCAST has not provided clinical breakpoints for antifungal drugs against rare yeasts. Therefore, it is recommended that evaluations of clinical breakpoints for antifungal medicines against rare yeasts should be made according to clinical breakpoints specified for either species to which rare yeasts are phylogenetically similar or wild isolates of other common species. We found high MIC values against Magnusiomyces spp., Trichosporon spp., and Rhodotorula mucilaginosa, which reportedly have intrinsic echinocandin resistance. Similarly, we found high MIC values for fluconazole against the isolates of M. capitatus, Saprochaete clavata (Magnusiomyces clavatus), T. asahii, and R. mucilaginosa, as well as Candida guilliermondii (Meyerozyma guilliermondii), Candida pararugosa (Diutina pararugosa), Candida rugosa (Diutina rugosa), Candida pelliculosa (Wickerhamomyces anomalus), Candida norvegensis (Pichia norvegensis), and C. fabianii, which were formerly included in Candida species. Antifungal susceptibilities of yeasts isolated from more than one sample are shown in Table 3, whereas antifungal susceptibilities of rare yeasts grown in only one sample are shown in Table 4.

Table 3.
Distribution of Antifungal Susceptibilities of Rare Yeasts Growing in Multiple Samples.

Table 4.
Distribution of Antifungal Susceptibilities of Rare Yeasts Growing in a Single Sample.
4. Discussion
The accurate identification of rare yeasts and determination of their antifungal susceptibilities are crucial for guiding treatment and vaccination. Some antifungals have limited spectra of activity in rare yeast species due to intrinsic resistance. As with fluconazole, the antifungal efficacies of other antifungal drugs also vary [16]. Among the 196 rare yeast species we isolated, C. lusitaniae (13.8%) was the most common, followed by M. capitatus (13.3%), C. fabianii (12.2%), and T. asahii (11.7%). We found high MIC values of anidulafungin against isolates of Magnusiomyces spp., Trichosporon spp., and R. mucilaginosa, and of fluconazole against isolates of M. capitatus, M. clavatus, T. asahii, and R. mucilaginosa, as well as isolates of other species such as M. guilliermondii, D. pararugosa, D. rugosa, W. anomalus, P. norvegensis, and C. fabianii.
EUCAST has not reported clinical breakpoints for antifungal drugs against rare yeasts due to insufficient evidence. Then again, it has been recommended that antifungal drugs used in the treatment of isolates to which rare yeasts are genetically related be also used in the treatment of rare yeast infections, based on the assumption that their pathogenicity and intrinsic susceptibility patterns would be similar, as well as antifungal drugs that exhibit MIC values against wild-type isolates of other frequently isolated species that are not phylogenetically related to rare yeasts similar to those against isolates genetically related to rare yeasts [12,16].
In a study on rare yeasts isolated from fungemia cases, Tepe et al. [17] found that Trichosporon spp., Saprochaete spp., Cryptococcus spp., and Rhodotorula spp. had intrinsic resistance to echinocandin. We also isolated yeasts with intrinsic echinocandin resistance, such as Trichosporon spp. and Saprochaete spp., although rare (7.7%), among the rare yeasts we isolated from fungemia cases. Therefore, in patients with predisposing risk factors for the development of fungal infections, rare yeasts as well as Candida species should be kept in mind when deciding on empirical treatment. In a study on antifungal susceptibilities of rare yeasts according to EUCAST standards, Stavru et al. categorized the MIC values of antifungal drugs against rare yeasts for which there are no epidemiological breakpoints as susceptible/reduced susceptible in comparison with the epidemiological breakpoints of C. albicans. Accordingly, they reported that species of Meyerozyma, Diutina/Kodamaea, and Pichia clades showed reduced susceptibility to azoles, and Candida haemulonii, Candida pseudohaemulonii, and Candida duobushaemulonii species, which are closely related to C. auris, also showed reduced susceptibility to some antifungal drugs, mainly azoles [16]. In parallel, high MIC values were reported for azoles against Candida inconspicua, C. pararugosa, and P. norvegensis isolates [18]. We also found high MIC values of fluconazole against M. guilliermondii, P. norvegensis, D. pararugosa, D. rugosa, Candida palmophilia, C. haemuli, M. capitatus, T. asahii, and R. mucilaginosa.
Echinocandins are recommended as first-line treatment for invasive candidiasis [19]. In a study investigating echinocandin resistance in yeasts using the broth microdilution method based on a MIC threshold value of ≥0.5 mg/L, Ollivier et al. reported that echinocandins had low MIC values against isolates of C. lusitaniae, Candida dubliniensis, M. guilliermondii, P. inconspicua, Saccharomyces cerevisiae, C. haemuli, and W. anomalus, and high MIC values against isolates of M.capitatus, M. clavatus, T. asahii, R. mucilaginosa, and Yarrowia lypolitica. We found MIC values generally similar to those reported in the above studies, except against isolates of some specific genera and species, which acquired resistance due to previous exposure to antifungal drugs [19].
In a study investigating the antifungal susceptibilities of rare yeasts isolated from various clinical samples, it was reported that amphotericin B, fluconazole and itraconazole showed high MIC values against C. haemuli isolates, fluconazole and caspofungin against M. capitatus isolates, fluconazole against K. ohmeri isolates, and fluconazole, posaconazole and caspofungin against R. mucilaginosa isolates [20]. We also found similar MIC values against these yeasts.
Onychomycosis is another fungal infection in which rare yeasts have been isolated as causative agents. A study reported Candida orthopsilosis (n = 9) as the most commonly isolated rare yeasts causing onychomycosis, followed by C. lusitaniae (n = 2), W. pararugosa (n = 2), Naganishia diffluens (n = 2), W. anomalus (n = 2), C.fabianii (n = 1), and Meyerozyma caribbica (n = 1) [21]. In the same study, fluconazole was found to have high MIC values against most of these species. Of the rare yeast isolates we identified, 5.1% were causative agents of onychomycosis. Of these isolates, R. mucilaginosa and T. asahii were isolated in three cases each, C. fermentati and M. guilliermondii in two cases each, and C. orthopsilosis and Zygosaccharomyces rouxii in one case each. Fluconazole exhibited high MIC values against all these species.
This study has some limitations. First, the epidemiology of rare yeasts varies across regions, countries, and even institutions. Although the participating centers had a large patient population and diverse populations, this study only presents data on strains identified in the ISLAB-2 service area. Therefore, our findings may not reflect species distribution or resistance patterns in other regions with different climates or patient demographics. Second is the lack of clinical data. The study included only in vitro antifungal susceptibility results. Clinical characteristics such as patient risk factors and thirty-day mortality rates could not be examined in detail due to limited access to clinical data. This situation caused insufficient confirmation of the clinical validity of the obtained MIC values. Another limitation is that molecular methods cannot be used in the identification of rare yeasts or in the search for antifungal resistance genes. In our study, yeasts isolated from axenic cultures, which were thought to have preserved their morphological, physiological and genetic characteristics, were identified by MALDI-TOF MS and supported by conventional methods. It has been concluded that identification should be supported by additional conventional methods to avoid possible misidentifications, especially in the identification of phylogenetically similar species. Finally, in this study, susceptibility results for seven traditional antifungal drugs were investigated. This presents a limited antifungal panel. In particular, in rare yeasts where antifungal resistance is observed, the inclusion of new antifungal drugs with different mechanisms of action, such as ibrexafungerp, rezafungin, fosmanogepix or oteseconazole, will allow the expansion of treatment alternatives and provide information for resistance monitoring programs.
5. Conclusions
EUCAST guidelines currently do not provide established clinical breakpoints for antifungal drugs against rare yeasts, yet recommend the use of antifungal drugs that exhibit MIC values against these rare yeasts similar to those against more common and possibly more pathogenic Candida species in the treatment of invasive infections in which rare yeasts have been isolated as causative agents. In this context, the identification and antifungal susceptibility results we obtained may contribute to epidemiological studies and guidelines on antifungal management of rare yeasts.
Author Contributions
Conceptualization, D.T. and S.A.; methodology, D.T., Z.H. and H.O.; software, M.Ö. and D.T.; validation, D.T., H.O., N.G. and E.D.; formal analysis, Z.H., M.Ö. and D.T.; investigation, D.T., H.O., N.G. and E.D.; resources, Z.H. and M.Ö.; data curation, Z.H., M.Ö. and D.T.; writing—original draft preparation, D.T. and S.A.; writing—review and editing, D.T. and S.A.; visualization, D.T.; supervision, H.O., Z.H. and D.T.; project administration, D.T. and H.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by Haydarpasa Numune Training and Research Hospital Ethics Committee (No: 2024/147–4645, Date: 19 November 2024).
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Xiao, M.; Chen, S.C.A.; Kong, F.; Fan, X.; Cheng, J.W.; Hou, X.; Zhou, M.L.; Wang, H.; Xu, Y.C. Five-year China Hospital Invasive Fungal Surveillance Net (CHIF-NET) study of invasive fungal infections caused by noncandidal yeasts: Species distribution and azole susceptibility. Infect. Drug. Resist. 2018, 11, 1659–1667. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Perfect, J.; Colombo, A.L.; Cornely, O.A.; Groll, A.H.; Seidel, D.; Albus, K.; de Almedia, J.N.; Garcia-Effron, G.; Gilroy, N. Global guideline for the diagnosis and management of rare yeast infections: An initiative of the ECMM in cooperation with ISHAM and ASM. Lancet Infect. Dis. 2021, 21, e375–e386. [Google Scholar] [CrossRef] [PubMed]
- Borman, A.M.; Johnson, E.M. Name Changes for Fungi of Medical Importance, 2018 to 2019. J. Clin. Microbiol. 2021, 59, e01811-20. [Google Scholar] [CrossRef] [PubMed]
- Kidd, S.E.; Abdolrasouli, A.; Hagen, F. Fungal Nomenclature: Managing Change is the Name of the Game. Open Forum. Infect. Dis. 2023, 10, ofac559. [Google Scholar] [CrossRef] [PubMed]
- de Hoog, G.S.; Chaturvedi, V.; Denning, D.W.; Dyer, P.S.; Frisvad, J.C.; Gräser, Y.; Guarro, J.; Haase, G.; Kwon-Chung, K.; Meis, J.F.; et al. Name Changes in Medically Important Fungi and Their Implications for Clinical Practice. J. Clin. Microbiol. 2015, 53, 1056–1062. [Google Scholar] [CrossRef] [PubMed]
- Warnock, D.W. Name Changes for Fungi of Medical Importance, 2012 to 2015. J. Clin. Microbiol. 2016, 55, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Blyth, C.C.; Chen, S.C.A.; Khanina, A.; Morrissey, O.; Roberts, J.A.; Thursky, K.A.; Worth, L.J.; Slavin, M.A. Introduction to the updated Australasian consensus guidelines for the management of invasive fungal disease and use of antifungal agents in the haematology/oncology setting. Intern. Med. J. 2021, 51, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.atlasclinicalfungi.org (accessed on 1 July 2025).
- Available online: https://www.indexfungorum.org (accessed on 1 July 2025).
- Available online: https://www.mycobank.org (accessed on 1 August 2025).
- Astvad, K.M.T.; Arikan-Akdagli, S.; Arendrup, M.C. A Pragmatic Approach to Susceptibility Classification of Yeasts without EUCAST Clinical Breakpoints. J. Fungi 2022, 8, 141. [Google Scholar] [CrossRef] [PubMed]
- EUCAST Guidance on Interpretation of MICs for Rare Yeast Without Breakpoints in Breakpoint Tables. 2024. Available online: https://www.eucast.org/astoffungi/clinicalbreakpointsforantifungals (accessed on 1 August 2025).
- Arendrup, M.C.; Boekhout, T.; Akova, M.; Meis, J.F.; Cornely, O.A.; Lortholary, O. ESCMID and ECMM joint clinical guidelines for the diagnosis and management of rare invasive yeast infections. Clin. Microbiol. Infect. 2014, 20, 76–98. [Google Scholar] [CrossRef] [PubMed]
- EUCAST method for susceptibility testing of yeasts. 2023. Available online: https://www.eucast.org/astoffungi/methodsinantifungalsusceptibilitytesting/susceptibility_testing_of_yeasts (accessed on 1 August 2025).
- John, L.L.H.; Thomson, D.D.; Bicanic, T.; Hoenigl, M.; Brown, A.J.P.; Harrison, T.S.; Bignell, E.M. Heightened Efficacy of Anidulafungin When Used in Combination with Manogepix or 5-Flucytosine against Candida auris In Vitro. Antimicrob. Agents Chemother. 2023, 67, e0164522. [Google Scholar] [CrossRef] [PubMed]
- Stavrou, A.A.; Lackner, M.; Lass-Flörl, C.; Boekhout, T. The changing spectrum of Saccharomycotina yeasts causing candidemia: Phylogeny mirrors antifungal susceptibility patterns for azole drugs and amphothericin B. FEMS Yeast Res. 2019, 19, foz037. [Google Scholar] [CrossRef] [PubMed]
- Tepe, D.; Aksoy, F.; Yılmaz, G.; Tosun, İ.; Özkaya, E.; Kaya, S. Fungemia due to Rare Yeasts Other Than Candida: 10 Years of Single-center Experience. Mediterr. J. Infect. Microb. Antimicrob. 2023, 12, 9. [Google Scholar] [CrossRef]
- Hansen, A.P.; Lass-Flörl, C.; Lackner, M.; Rare Yeast Study Group. Antifungal susceptibility profiles of rare ascomycetous yeasts. J. Antimicrob. Chemother. 2019, 74, 2649–2656. [Google Scholar] [CrossRef] [PubMed]
- Desnos-Ollivier, M.; Bretagne, S.; Lortholary, O.; Dromer, F. Echinocandins Susceptibility Patterns of 2,787 Yeast Isolates: Importance of the Thresholds for the Detection of FKS Mutations. Antimicrob. Agents Chemother. 2022, 66, e0172521. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Asadzadeh, M.; Al-Sweih, N.; Khan, Z. Spectrum and management of rare Candida/yeast infections in Kuwait in the Middle East. Ther. Adv. Infect. Dis. 2024, 11, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Jabrodini, A.; Eghtedarnejad, E.; Ghanbarzadeh, A.; Motamedi, M.; Jafari, M.; Kharazi, M.; Yazdanpanah, S.; Khodadadi, H. Molecular identification and antifungal susceptibility profile of rare and emerging yeast species causing onychomycosis. BMC Res. Notes. 2025, 18, 167. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).