Abstract
Candida haemulonii complex (C. haemulonii, C. duobushaemulonii and C. haemulonii var. vulnera) is well-known for its resistance profile to different available antifungal drugs. Although echinocandins are the most effective class of antifungal compounds against the C. haemulonii species complex, clinical isolates resistant to caspofungin, micafungin and anidulafungin have already been reported. In this work, we present a literature review regarding the effects of echinocandins on this emergent fungal complex. Published data has revealed that micafungin and anidulafungin were more effective than caspofungin against the species forming the C. haemulonii complex. Subsequently, we investigated the susceptibilities of both planktonic and biofilm forms of 12 Brazilian clinical isolates of the C. haemulonii complex towards caspofungin and micafungin (anidulafungin was unavailable). The planktonic cells of all the fungal isolates were susceptible to both of the test echinocandins. Interestingly, echinocandins caused a significant reduction in the biofilm metabolic activity (viability) of almost all fungal isolates (11/12, 91.7%). Generally, the biofilm biomasses were also affected (reduction range 20–60%) upon exposure to caspofungin and micafungin. This is the first report of the anti-biofilm action of echinocandins against the multidrug-resistant opportunistic pathogens comprising the C. haemulonii complex, and unveils the therapeutic potential of these compounds.
1. Introduction
The members of the Candida haemulonii species complex (C. haemulonii, C. duobushaemulonii and C. haemulonii var. vulnera) are well-known for their (multi)drug-resistance towards several antifungal agents available in clinical practice. Resistance of the C. haemulonii complex to azoles (e.g., fluconazole, itraconazole and voriconazole) and polyenes (e.g., amphotericin B) has been documented extensively [1,2,3,4,5,6,7]. On the other hand, susceptibility to prescribed echinocandins (anidulafungin, caspofungin and micafungin) is commonly observed [7,8,9,10,11], although there have been some reports of clinical isolates being resistant to these compounds [5,12].
Echinocandins are the newest class of antifungal agents to be used in clinical practice, exhibiting fungicidal activity against yeasts as well as having a good safety profile [8]. In this sense, the guidelines of the Centers for Disease Control and Prevention (CDC, USA) strongly recommend that echinocandins should be the first choice for the treatment of candidemia in both neutropenic and non-neutropenic patients [9]. The mechanism of action of the echinocandins involves the noncompetitive inhibition of the enzyme β-(1,3)-d-glucan synthase, which is involved in the synthesis of the polysaccharide glucan, resulting in the loss of cell wall integrity and severe stress in the fungal wall [8].
The three clinically available echinocandins usually exhibit both in vitro and in vivo fungicidal activity against a variety of Candida species, including those that are intrinsically resistant to azoles or amphotericin B (e.g., C. krusei, C. glabrata and C. lusitaniae), and also emerging species (e.g., C. famata and C. rugosa) [10]. Additionally, the antifungal activity of echinocandins against Candida biofilms represents an aspect that should be highlighted, since microbial biofilm is considered a resistance structure that precludes efficient antimicrobial treatment [10]. For instance, both caspofungin and micafungin, at concentrations attainable in clinical treatments, were able to kill fungal cells in preformed biofilms of either C. albicans or C. parapsilosis [11]. Therapeutic concentrations of caspofungin and micafungin were active against the biofilms formed by isolates of C. albicans and C. glabrata recovered from cases of bloodstream infections, but not against C. tropicalis, demonstrating that species-specific differences can influence the outcome [12]. Corroborating these findings, caspofungin was also shown to be effective in the treatment and prevention of C. albicans biofilms in an in vivo murine model of central venous catheter-associated candidiasis [13].
Considering the aforementioned aspects, the aim of the present study was to evaluate the antifungal susceptibility of both planktonic- and biofilm-forming cells from 12 Brazilian clinical isolates comprising the C. haemulonii complex towards caspofungin and micafungin. Furthermore, we have performed a literature review concerning the susceptibility of the C. haemulonii species complex towards echinocandins in order to present a comprehensive summary of this field.
2. Materials and Methods
2.1. Microorganisms and Growth Conditions
Twelve clinical fungal isolates, previously identified by molecular methods [6], belonging to the C. haemulonii species complex were used in the present study: five isolates of C. haemulonii (LIPCh2 recovered from the sole of the foot, GenBank accession number KJ476194; LIPCh3 from a toe nail, KJ476195; LIPCh4 from a finger nail, KJ476196; LIPCh7 from a toe nail, KJ476199; LIPCh12 from blood, KJ476204), four isolates of C. duobushaemulonii (LIPCh1 from finger nail, KJ476193; LIPCh6 from a toe nail, KJ476198; LIPCh8 from blood, KJ476200 and LIPCh10 from bronchoalveolar lavage, KJ476202) and three isolates of C. haemulonii var. vulnera (LIPCh5 from a toe nail, KJ476197; LIPCh9 from urine, KJ476201 and LIPCh11 from blood, KJ476203) [6]. In all experiments, Sabouraud dextrose medium was used to culture the fungal isolates at 37 °C for 48 h under constant agitation (200 rpm). Yeasts were counted in a Neubauer chamber.
2.2. Determination of Minimal Inhibitory Concentration (MIC)
Antifungal susceptibility testing, using the planktonic cells of C. haemulonii species complex, against caspofungin and micafungin (Sigma-Aldrich, St. Louis, MO, USA) was performed according to the broth microdilution technique standardized in the M27-Ed4 protocol [14] and interpreted according to the M27-S3 document published by the Clinical and Laboratory Standards Institute (CLSI) [15]. C. krusei (ATCC 6258) and C. parapsilosis (ATCC 22019) were used as quality control isolates in each test as directed by the CLSI. The clinical breakpoints to echinocandins are detailed below.
2.3. Echinocandins’ Breakpoints
Until now, there have been no established breakpoints for echinocandins (or any other antifungal class) regarding the species belonging to the C. haemulonii complex. To overcome this problem, researchers working with this fungal complex, as well as “newly identified” Candida species, have generally been using a comparative perspective in order to interpret and discuss antifungal susceptibilities. Results are normally presented as CLSI breakpoints which have been established for the Candida genus (CLSI document M27S3 [15]) in order to have a minimum (even if not precise) parameter to interpret this kind of experiment. Alternatively, a possible option is to compare the MIC values of C. haemulonii complex with the breakpoints established for non-albicans Candida species (e.g., C. glabrata, C. tropicalis, C. krusei, C. parapsilosis and C. guilliermondii) as recently suggested by the CLSI (document M27S4 [16] and protocol M60 [17]). However, this approach varies depending on the particular Candida species, since each presents its own breakpoint for each of the echinocandin drugs used. Moreover, the CDC (USA) recently published on its website (https://www.cdc.gov/fungal/candida-auris/c-auris-antifungal.html) a proposal of echinocandins’ breakpoints for C. auris, a phylogenetically related species to the C. haemulonii complex, as follows: resistant breakpoint for caspofungin is ≥2 mg/L and for micafungin and anidulafungin, ≥4 mg/L. After contemplating these various viewpoints, we chose to use, herein, the breakpoints available for Candida spp. in the CLSI document M27-S3 [15], which considers as susceptible the strains having MIC values ≤ 2 mg/L and non-susceptible those with MIC values > 2 mg/L for the three clinically available echinocandins; a MIC summary table was prepared.
2.4. Effects of Echinocandins on the Biofilm Formed by the C. haemulonii Species Complex
Fungal suspensions in Sabouraud broth (200 µL containing 106 yeast cells) were transferred into each well of a flat-bottom 96-well polystyrene microtiter plate and incubated without agitation at 37 °C for 48 h, which has been shown to be the best incubation time for biofilm formation by species belonging to the C. haemulonii complex [18]. Afterwards, the biofilm supernatant fluids were carefully removed, washed once with sterile phosphate-buffered saline (PBS; 10 mM NaH2PO4, 10 mM Na2HPO4, 150 mM NaCl, pH 7.2) and then 200 µL of Roswell Park Memorial Institute Medium (RPMI) 1640 medium containing different concentrations of echinocandins (range 0.25–8 mg/L) were added to each well. RPMI 1640 medium without echinocandins was used as a positive control and medium-only blanks were used as the negative control. The biofilms were then incubated at 37 °C for an additional 48 h. Afterwards, the supernatant fluids were carefully removed and the wells were washed twice with PBS to remove any non-adherent cells. Finally, two classic biofilm parameters (biomass and metabolic activity/viability) were measured as described below. The results were expressed as percentage of reduction of both viability and biomass. The minimal biofilm eradication concentration (MBEC) was achieved, considering the lowest concentration of each echinocandin capable of causing a 50% reduction in the biofilm viability [19].
2.4.1. Viability Assay
The viability of the fungal cells forming the biofilm was determined using a colorimetric assay that measures the metabolic reduction of 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino) carbonyl]-2H-tetrazolium hydroxide (XTT; Sigma-Aldrich) to a water-soluble brown formazan product [20,21]. A XTT/menadione solution was prepared as follows: 2 mg of XTT was dissolved in 10 mL of pre-warmed PBS solution supplemented with 100 μL of a menadione stock solution (made by dissolving 55 mg of menadione in 100 mL of acetone). The XTT/menadione solution (200 μL) was added to all wells containing the biofilms (see Section 2.4 above) and incubated in the dark at 37 °C for 3 h. One hundred microliters of the supernatant from each well were then transferred to a new microplate and the colorimetric readings were measured at 492 nm using a microplate reader (SpectraMax M3; Molecular Devices, Sunnyvale, CA, USA) [21].
2.4.2. Biomass Measurement
Biomass quantification was assessed as described by Peeters et al. [20]. Firstly, biofilms (see Section 2.4 above) were fixed by adding 200 μL of 99% methanol for 15 min. The supernatant was then discarded. Microtiter plates were air-dried for 5 min and then 200 μL of 0.4% crystal violet solution (stock solution diluted in PBS; Sigma-Aldrich) were added to each well and the plates then incubated at room temperature for 20 min. After discarding the crystal violet solution, the wells were washed once with PBS to remove excess stain and the biomass in each well was then decolorized by adding 200 μL of 33% acetic acid for 5 min. One hundred microliters of the acetic acid solution were transferred to a new 96-well plate and the absorbance measured at 590 nm using a microplate reader (SpectraMax M3; Molecular Devices) [21].
2.5. Biofilm Architecture: Confocal Laser Scanning Microscopy (CLSM) Assay
Biofilms were formed on a polystyrene surface and treated as described above with different concentrations of micafungin (0.5–2.0 mg/L). Then, the biofilms were stained with Calcofluor white (Sigma-Aldrich) solution (5 µg/mL) for 1 h at room temperature and protected from the light [21,22,23]. Subsequently, the biofilms were washed twice with PBS and covered with n-propyl-gallate for observation using a confocal microscope (Leica TCS SP5 with OBS, Berlin, Germany). Fiji ImageJ2 software (UW-Madison LOCI, Madison, WI, USA), was used to obtain three-dimensional (3-D) reconstitutions of the biofilms [21,24]. In this way, image analysis was performed using z-series image stacks from five randomly chosen spots on each biofilm [21].
2.6. Literature Review
This exercise involved the compilation of available data regarding the susceptibility of the C. haemulonii species complex to echinocandins. The literature search was performed on 19 July 2020 using the following four databases: PubMed (https://pubmed.ncbi.nlm.nih.gov), Web of Science (https://webofknowledge.com), Google Scholar (https://scholar.google.com) and Scielo (https://scielo.org/). The term “Candida haemulonii” was added in the category “title/abstract” in the PubMed Advanced Search Builder and in the Web of Science databases, while in Google Scholar the search was conducted in the advanced search area, including the term “Candida haemulonii” and selecting the option “with the exact phrase in the title”; finally, for the Scielo database, we only used the search term “Candida haemulonii” in the general search. Papers available in English and published after the reclassification of the C. haemulonii complex by Cendejas-Bueno et al. [5] were selected. Subsequently, the list of results from each database was exported to the EndNote® software (version X1), using the “Output Records” tool in order to eliminate possibly duplicated references by means of the “Find Duplicates” tool. Finally, the papers were individually analyzed in order to select those that described either MIC or geometric-mean (GM)-MIC values of the C. haemulonii complex for echinocandins.
2.7. Statistics
All experiments were performed in triplicate, in three independent experimental sets. The results were analyzed statistically by the Analysis of Variance One-Way ANOVA (comparisons between three or more groups). All analyzes were performed using the GraphPad Prism5 program. For all analyses, p values of 0.05 or less were considered statistically significant.
3. Results and Discussion
3.1. Susceptibility of Planktonic Cells of the C. haemulonii Species Complex to Echinocandins
According to the breakpoints suggested in the M27S3 document published by CLSI, the planktonic cells of all clinical isolates of the C. haemulonii complex tested herein were considered susceptible to echinocandins, with MIC values ranging from 0.125 to 0.5 mg/L for caspofungin and 0.25–0.5 mg/L for micafungin (Table 1). For instance, a recent report described the successful use of caspofungin (MIC of ≤0.125 mg/L) in the treatment of a case of catheter-related candidemia caused by C. haemulonii in a pediatric patient in Mexico [25], whose fungal isolate exhibited in vitro high MICs for azoles (fluconazole MIC ≥256 mg/L, posaconazole ≥8 mg/L, itraconazole, ketoconazole and voriconazole ≥16 mg/L) and amphotericin B (MIC 1–2 mg/L). Some years before, a catheter-related candidemia in an adult patient hospitalized for a long period was only resolved when fluconazole treatment was replaced by caspofungin [4].

Table 1.
MIC values of echinocandins against the C. haemulonii species complex studied herein.
In general, echinocandins are highly active in vitro against species comprising the C. haemulonii complex [7,26,27,28,29], but the existence of isolates resistant to this class of antifungals has already been reported [4,5,30]. Herein, we conducted a careful review of the literature regarding the susceptibility of the C. haemulonii species complex to the three clinically available echinocandins, including only papers published after the species reclassification and the creation of the C. haemulonii complex [5]. Using the keyword “Candida haemulonii” in the search section, 148, 63, 46 and 5 publications were located from the Web of Science, PubMed, Google Scholar and Scielo databases, respectively (Table 2). However, only a small fraction of these published papers (varying from 12.2%–28.3%) cited the in vitro susceptibility profile of the C. haemulonii species complex against echinocandins. In this sense, we recovered a total of 21 distinct papers that fitted our established criteria and, for these reasons, they were selected for data extraction as follows: 5 (23.8%) papers studied the three members forming the C. haemulonii complex, 6 (28.6%) studied only two species (C. haemulonii and C. duobushaemulonii) and 10 (47.6%) studied only one species (C. haemulonii, n = 6, C. duobushaemulonii, n = 3, C. haemulonii var. vulnera, n = 1). Furthermore, 13 (61.9%) papers detailed the MIC value for each isolate investigated, while the remaining studies (n = 8; 38.1%) only presented the geometric mean (GM)-MIC and/or the range of MIC values for the fungal isolates against the test echinocandins. Finally, 12 (57.1%) papers tested the three echinocandins, 5 (23.8%) used two and 4 (19.1%) tested only one echinocandin, with caspofungin being the most frequently evaluated.

Table 2.
Number of publications retrieved from database searches using the term “Candida haemulonii”.
The results emanating from this literature review revealed that micafungin and anidulafungin appeared to be more effective than caspofungin against the three species forming the C. haemulonii complex (Table 3) [5,7,25,29,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47]. In this respect, 89.8% of the isolates of C. haemulonii exhibited susceptibility to caspofungin, while 96.3% and 98.4% were susceptible to micafungin and anidulafungin, respectively. Regarding C. duboushaemulonii, 95.5% of the isolates were susceptible to caspofungin, 99.1% to anidulafungin and 100.0% to micafungin. Finally, considering the clinical isolates of C. haemulonii var. vulnera, 85.0% were susceptible to caspofungin, 91.7% to micafungin and 97.1% to anidulafungin. Indeed, the MIC frequency distribution demonstrated that the modal MIC of echinocandins against the C. haemulonii complex was ≤0.12 mg/L in almost all cases (Table 4).

Table 3.
Literature compilation regarding the distribution (%) of the susceptible (S) and non-susceptible (NS) isolates belonging to the C. haemulonii complex against echinocandins described in published papers available until 19 July 2020.

Table 4.
MIC distribution of C. haemulonii complex isolates obtained from the literature review against the three echinocandins.
Comparing the GM-MIC values of our clinical isolates (Table 1) with those compiled from the literature reports (for these comparisons, we used the arithmetic mean of the GM-MIC values of the selected works, as summarized in Table 5), we observed that the GM-MIC values of caspofungin for our isolates of C. haemulonii, C. duobushaemulonii and C. haemulonii var. vulnera were higher than those reported in the literature (0.33 mg/L versus 0.18 mg/L for C. haemulonii, 0.18 mg/L versus 0.11 mg/L, for C. duobushaemulonii and 0.32 mg/L versus 0.21 mg/L for C. haemulonii var. vulnera). Similarly, GM-MIC values for micafungin calculated from the literature reports were lower than ours (0.18 mg/L versus 0.33 mg/L for C. haemulonii, 0.17 mg/L versus 0.30 mg/L for C. duobushaemulonii, and 0.13 mg/L versus 0.25 mg/L for C. haemulonii var. vulnera). Finally, based on the analysis of the literature data, anidulafungin also produced low GM-MIC values for the three fungal species of the C. haemulonii complex (GM-MICs of 0.16, 0.32 and 0.06 mg/L for C. haemulonii, C. duobuhaemulonii and C. haemulonii var. vulnera, respectively).

Table 5.
Literature review on the antifungal susceptibility of different isolates of the C. haemulonii complex to echinocandins.
In summary, the majority of literature reported GM-MIC concentration values of <0.5 mg/L for the three echinocandins against the C. haemulonii species complex. Nevertheless, two works warranted specific attention: Cendejas-Bueno et al. [5], in which the GM-MIC values for caspofungin for the three members of the C. haemulonii complex were disproportionately high in comparison to our present results and those given in the other literature publications; and Isla et al. [36], in which the GM-MIC value obtained for caspofungin against the C. duobushaemulonii isolates was considerably higher (Table 5). A possible explanation for the high MIC values found in the aforementioned papers is the possible occurrence of paradoxical growth effect (also known as the Eagle effect), that is characterized by reduced activity of the antifungal agents at high concentrations. In fact, Cendejas-Bueno et al. [5] stressed this discussion in their study, but in a superficial way. A recent study conducted with 106 clinical isolates of C. auris demonstrated that the vast majority of isolates were susceptible to the echinocandins; however, they exhibited different intensities of paradoxical growth effect in the presence of caspofungin, whilst four isolates were resistant to echinocandins and had a mutation in hot spot region 1 of the FKS gene [48]. Interestingly, those isolates presenting paradoxical growth effect were susceptible to caspofungin at doses used in human treatment, while those with FKS1 mutation were still resistant in a murine model of invasive candidiasis, demonstrating that only the isolates with the mutations display in vivo echinocandin resistance [48].
3.2. Effects of Echinocandins on the Biofilm Formed by C. haemulonii Species Complex
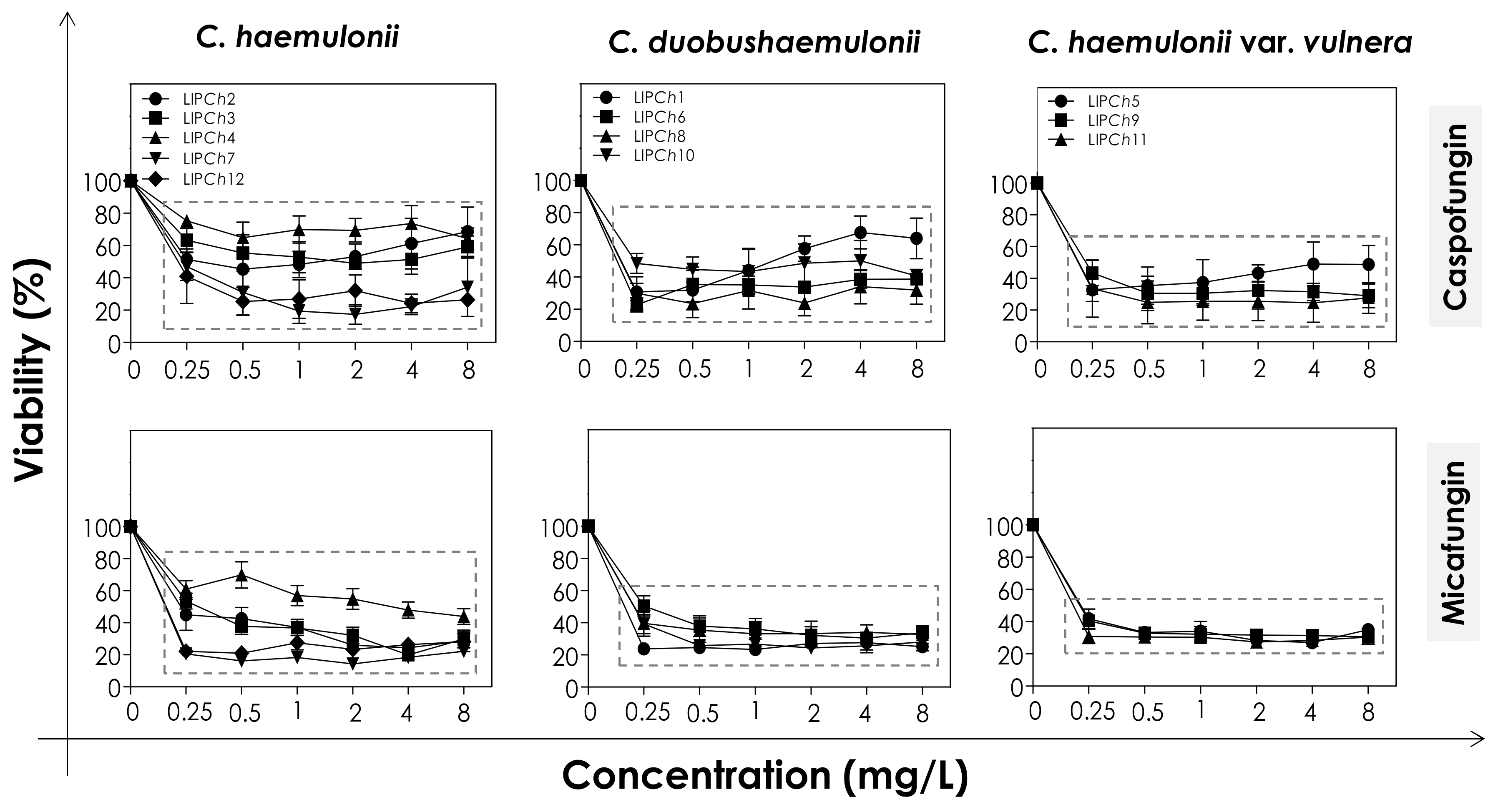
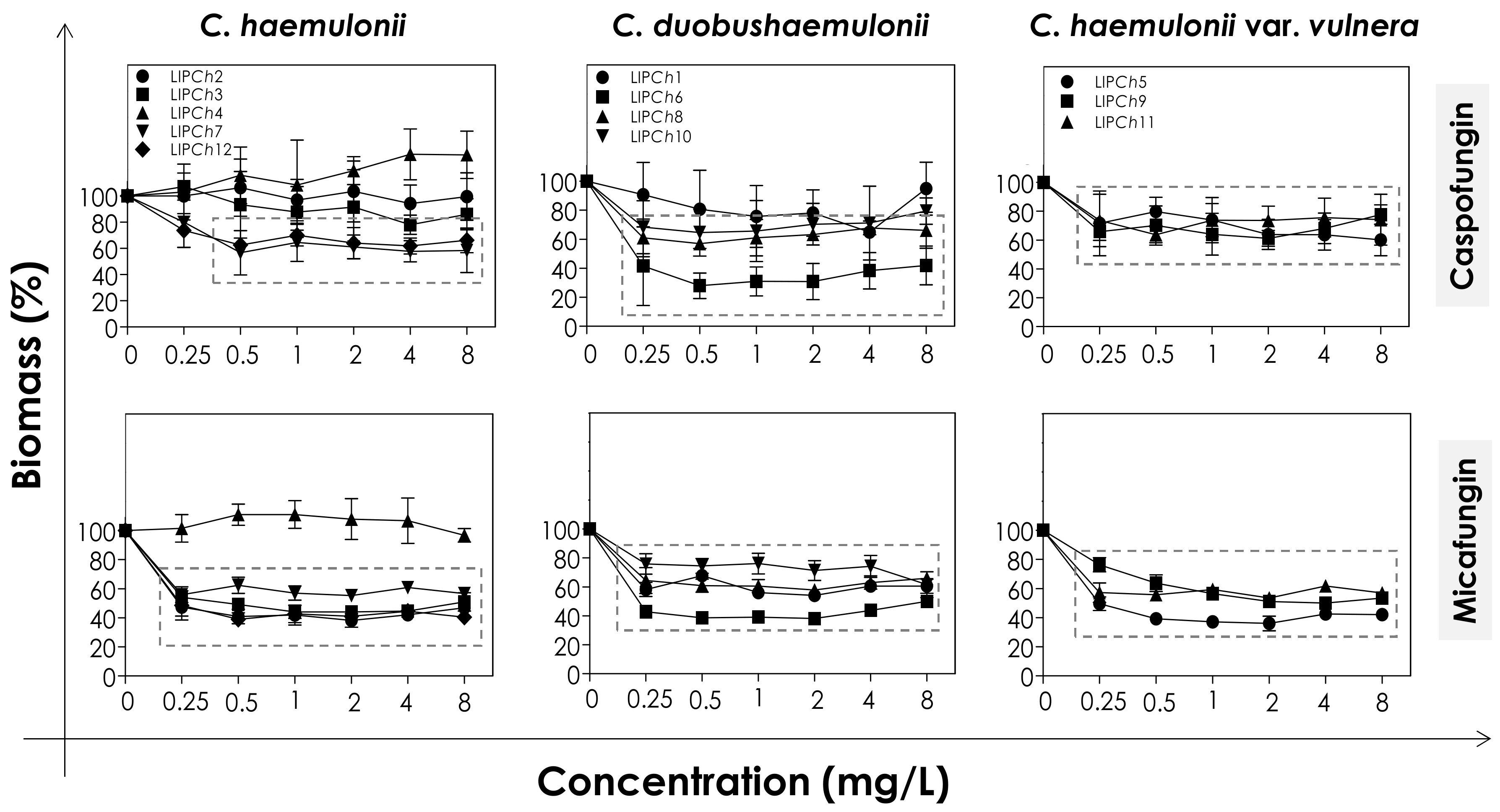
In order to evaluate the effects of echinocandins (caspofungin and micafungin) on the viability and biomass of the biofilms formed by the clinical isolates of the C. haemulonii complex, the mature biofilms were firstly incubated with different concentrations of the antifungals and then analyzed. The metabolic activity of viable fungal cells was assessed by their ability to reduce XTT to formazan, whilst the decrease in biofilm biomass was measured spectroscopically by looking at the incorporation of crystal violet into methanol-fixed, non-viable cells (Figure 1 and Figure 2). In general, the test echinocandins were found to be more efficient at reducing cell viability than decreasing the biomass of the C. haemulonii complex biofilms.
Figure 1.
Cell viability of biofilms formed by clinical isolates comprising the C. haemulonii complex exposed to different concentrations of echinocandins (caspofungin and micafungin). The results were assessed spectroscopically (492 nm) by XTT reduction and expressed as the mean of metabolic activity percentages compared to untreated biofilms (control), which correspond to 100%. The graphs exhibit the mean ± standard deviation of three independent experiments. The dashed boxes represent the concentrations of echinocandins that caused statistically significant reduction of cell viability in relation to the respective control (p < 0.05; One-way ANOVA analysis of variance, Dunnett’s multiple comparison test).
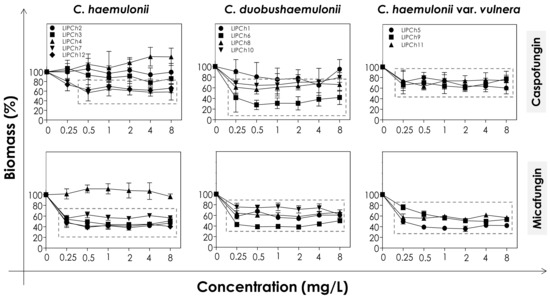
Figure 2.
Biomass of biofilms formed by clinical isolates comprising the C. haemulonii species complex exposed to different concentrations of echinocandins (caspofungin and micafungin). The amount of crystal violet incorporated by the cells was assessed spectroscopically (absorbance at 590 nm) and the results expressed as the mean of biomass percentages compared to untreated biofilms (control), which correspond to 100%. The graphs show the mean ± standard deviation of three independent experiments. The dashed boxes represent the concentrations of echinocandins that caused a statistically significant reduction in biomass in relation to the respective control (p < 0.05; One-way ANOVA analysis of variance, Dunnett’s multiple comparison test).
The decrease of both viability and biomass parameters by caspofungin was isolate-dependent. At the lowest concentration used (0.25 mg/L) this echinocandin caused a statistically significant reduction in the viability of all of the fungal cells tested (p < 0.05; One-way ANOVA analysis of variance, Dunnett’s multiple comparison test), varying from 30–80% among the different isolates (Figure 1). However, caspofungin was unable to reduce the biomass of some of the C. haemulonii isolates (LIPCh2, LIPCh3 and LIPCh4) even at the highest concentration used. Nevertheless, for the remaining fungal isolates the drug caused a biomass reduction of up to 60% (mainly against the C. duobushaemulonii isolates) (Figure 2). The isolates LIPCh2 (C. haemulonii), LIPCh1 (C. duobushaemulonii) and LIPCh5 (C. haemulonii var. vulnera) were less susceptible to caspofungin at the higher concentrations (Figure 1).
Micafungin proved to be more effective than caspofungin at disturbing both biofilm viability and biomass. A decrease in biofilm viability of up to 60% was seen among most of the clinical isolates, especially against C. duobushaemulonii and C. haemulonii var. vulnera (Figure 1). Unlike caspofungin, micafungin showed a decrease of up to 60% on the biofilm biomass of C. haemulonii isolates, with the exception of isolate LIPCh4, which forms a very dense and robust biofilm (Figure 2). For the C. duobushaemulonii and C. haemulonii var. vulnera isolates, micafungin reduced biomass in the range 20–60% (Figure 2). In summary, the lowest concentration of micafungin used was able to significantly reduce the cell viability and the biomasses of biofilms formed by all of the test isolates, expect for the biomass of one isolate.
The determination of MBEC, which was defined as the lowest antifungal concentration able to reduce the biofilm viability in 50% [19], revealed that the biofilms of all isolates remained susceptible to echinocandins, with the exception of the isolate LIPCh4 of C. haemulonii (Table 6). This fact could be explained by the ability of the isolate LIPCh4 to form very robust biofilm on polystyrene in comparison with the other isolates [18,21], hampering the action of echinocandins due to the high amount of fungal cells-forming the biofilm architecture as well as due to the high production of extracellular matrix that can block the antifungal penetration into the biofilm structure.

Table 6.
Minimal biofilm eradication concentration (MBEC) to echinocandins against C. haemulonii complex.
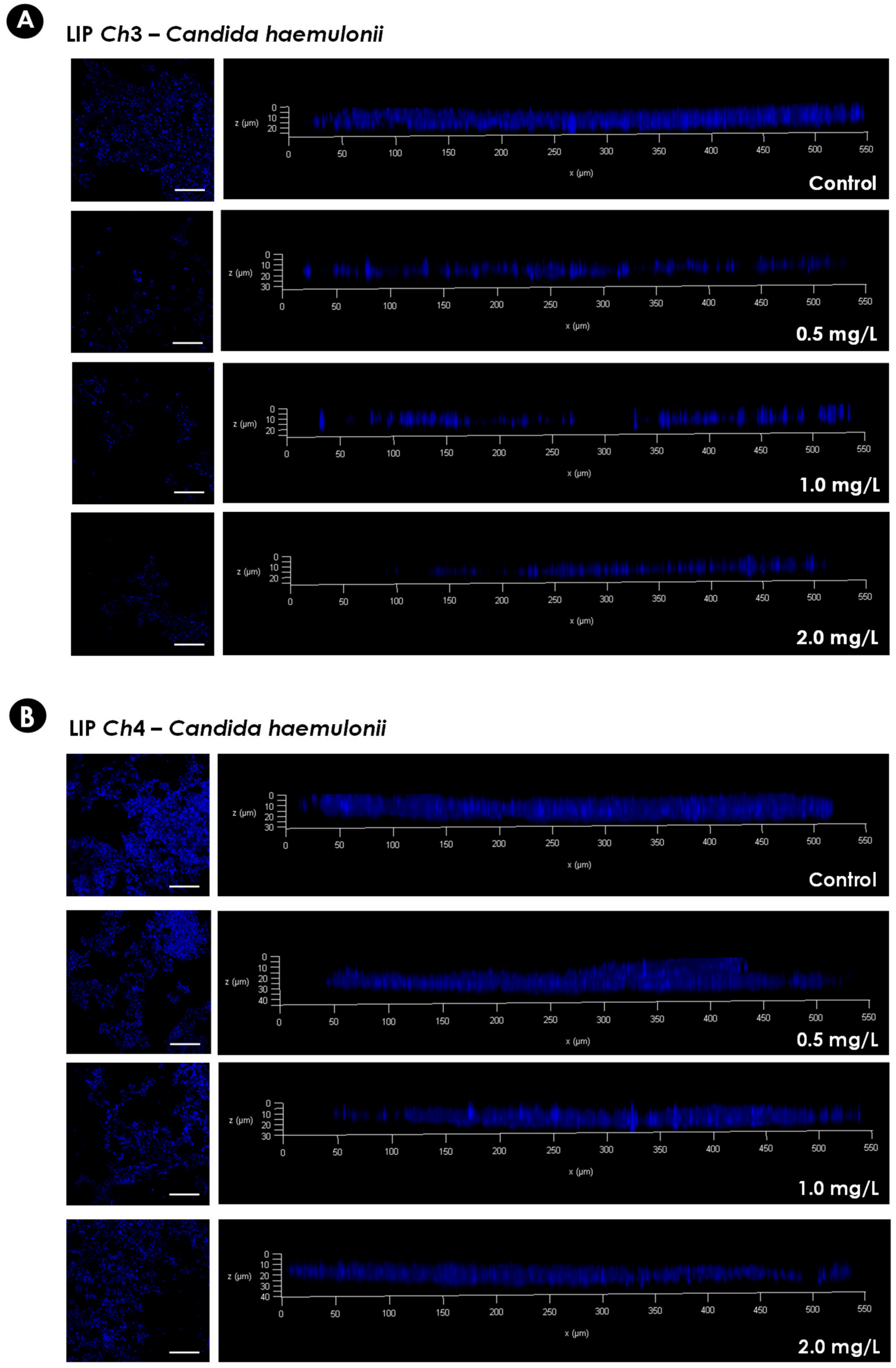
As micafungin was more active than caspofungin against the mature biofilms formed by the C. haemulonii species complex it was chosen for further studies. In order to verify the 3-D organization of the biofilms following exposure to micafungin two isolates of C. haemulonii were selected: LIPCh3, to represent the isolates having susceptible biofilms, and LIPCh4, to represent isolates forming resistant biofilms. CLSM analysis was conducted using Calcofluor white, which binds to the chitin in the fungal cell wall, to evidence the biofilm biomass. The CLSM analysis corroborated the results observed by crystal violet approach, with the lowest antifungal concentration used causing a drastic reduction in the biofilm biomass of LIPCh3, whilst even the highest concentration of micafungin failed to affect the biofilm formed by LIPCh4 (Figure 3).
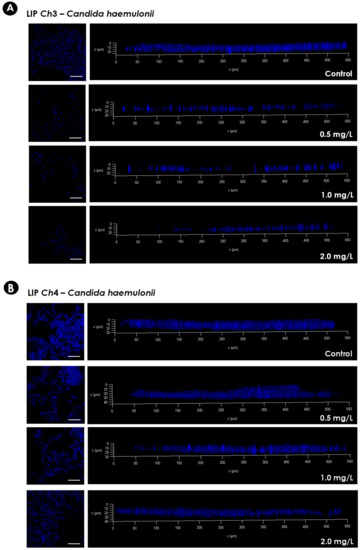
Figure 3.
Representative confocal laser scanning microscopy (CLSM) images of the biofilms formed by C. haemulonii on a polystyrene surface. Yeasts (200 μL containing 106 cells) were placed to interact with the polystyrene for 48 h at 37 °C. Subsequently, the supernatant fluids were removed and washed with PBS, and 200 µL of RPMI 1640 medium containing different concentrations of micafungin were added. The biofilms were incubated at 37 °C for an additional 48 h. Afterwards, the supernatant fluids were carefully removed again, and the wells were washed twice with PBS to remove non-adherent cells. Finally, the biofilms were stained with Calcofluor white in order to evidence the fungal biomass. The panels on the left represent the top view images of the fungal biofilms visualized by Confocal Laser Scanning Microscopy (CLSM) (bars represent 5 µm). The graphs on the right represent the three-dimensional reconstruction of the biofilms formed. The isolate LIPCh3 of C. haemulonii (A) was chosen to represent susceptible biofilms, while the isolate LIPCh4 of C. haemulonii (B) represents resistant biofilms.
Until now, no information has been available in the literature regarding the activity of conventional antifungal agents against the biofilm formed by the C. haemulonii species complex. A recent study conducted with C. auris, which belongs to the C. haemulonii clade, showed that, despite the susceptibility of planktonic cells to echinocandins and amphotericin B, the biofilms were not vulnerable, exhibiting MBECs which were 512-fold higher than their planktonic MIC counterparts [19]. Actually, the biofilm formed by C. auris is not as robust as those arising from C. albicans and C. glabrata, but its tolerance to the major classes of antifungal agents is notable, especially for amphotericin B and micafungin, which are the recommended antifungal therapeutics for infections caused by C. albicans biofilms [49]. The antifungal tolerance of the C. auris biofilm has been shown to be phase-dependent, with the mature biofilms resistant to the three available antifungal drug classes [50]. On the other hand, micafungin has been shown to be effective against both planktonic and biofilm-forming C. albicans cells, while its effectiveness against C. parapsilosis was considered to be moderate [51]. Additionally, micafungin concentrations >2 mg/L prevented the regrowth of Candida biofilm cells [51]. Regarding the C. parapsilosis complex, caspofungin was more active against biofilms of C. orthopsilosis than C. parapsilosis sensu strictu, with 20% and 86% of isolates resistant to this antifungal, respectively, suggesting that a treatment of catheter-related candidemia caused by C. orthopsilosis with caspofungin would be more effective than against C. parapsilosis sensu strictu [52]. A study, conducted with five different Candida species recovered from cases of bloodstream infections demonstrated both species-specific and drug-specific differences in Candida biofilms regarding their susceptibility to echinocandins [53]. In this sense, while C. albicans and C. krusei biofilms were susceptible to the three clinically available echinocandins, C. lusitaniae, C. guilliermondii and C. parapsilosis were quite resistant to them [53]. In addition, micafungin seemed to be the most effective echinocandin against C. parapsilosis biofilms, presenting lower MBECs against this Candida species in comparison to caspofungin and anidulafungin [53]. These observations reinforce the need to determine the correct identification of the actual fungal species causing the candidiasis infection and, further, to assess its antifungal susceptibility profile against both planktonic and biofilm-forming cells in order to choose the best therapeutic option for each case.
Furthermore, we observed that one isolate of each species forming the C. haemulonii complex showed a smaller reduction in cell viability when incubated in the presence of higher concentrations of the echinocandins. This phenomenon is called paradoxical growth, and it corresponds to the decreased sensitivity to echinocandins in the presence of concentrations higher than the MIC values. To date, the evidence strongly suggests that this paradoxical effect is more commonly associated with caspofungin than either micafungin or anidulafungin [54]. This effect has already been documented for biofilms formed by other Candida species, such as C. albicans [53,55], C. parapsilosis [53], C. tropicalis [55] and C. dubliniensis [56].
To finalize, we recognize some of the limitations associated with the present study, such as the limited number of isolates used and the exclusion of anidulafungin. The experiments were conducted with only 12 clinical isolates of the C. haemulonii complex due to the difficulties in obtaining more isolates, since it is quite a rare fungal complex. Additionally, we tested only two of the three echinocandins currently in clinical use, and this was because at the time the experiments were conducted anidulafungin was not available for scientific research purposes.
4. Conclusions
In addition to their own clinical conditions, hospitalized patients are at constant risk of acquiring contagions associated with the hospital environment. Biofilm-related Candida infections represent an important and worrisome threat to these patients, and there is a limited number of available antifungal agents of sufficient potency to break down these highly resistant structures. In this sense, echinocandins are considered highly active against various Candida species and the results presented herein reinforce the potential of echinocandins to treat biofilm-related infections caused by the emergent and multidrug-resistant species comprising the C. haemulonii complex.
Author Contributions
All authors conceived and designed the experiments. L.S.R. performed the experiments. All authors analyzed the data. M.H.B. and A.L.S.S. contributed reagents/materials/analysis tools. All authors wrote and revised the paper. All authors contributed to the research and approved the final version of the manuscript. All authors agree to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grants from Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Financial code 001).
Acknowledgments
The authors would like to thank Denise Rocha de Souza (UFRJ) for technical assistance in the experiments and Grasiella Matioszek (UFRJ) for confocal analyses. The authors would like to thank Malachy McCann (Department of Chemistry at the National University of Ireland Maynooth—NUIM, Ireland) for his valuable contribution to the critical review and editing of English.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Khan, Z.U.; Al-Sweih, N.A.; Ahmad, S.; Al-Kazemi, N.; Khan, S.; Joseph, L.; Chandy, R. Outbreak of fungemia among neonates caused by Candida haemulonii resistant to amphotericin B, itraconazole, and fluconazole. J. Clin. Microbiol. 2007, 45, 2025–2027. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J.; Gibbs, D.L.; Newell, V.A.; Ellis, D.; Tullio, V.; Rodloff, A.; Fu, W.; Ling, T.A. Results from the ARTEMIS DISK Global Antifungal Surveillance Study, 1997 to 2007: A 10.5-year analysis of susceptibilities of Candida Species to fluconazole and voriconazole as determined by CLSI standardized disk diffusion. J. Clin. Microbiol. 2010, 48, 1366–1377. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lopez, A.; Buitrago, M.J.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. in vitro antifungal susceptibility pattern and ergosterol content in clinical yeast strains. Rev. Iberoam. Micol. 2011, 28, 100–103. [Google Scholar] [CrossRef]
- Kim, S.; Ko, K.S.; Moon, S.Y.; Lee, M.S.; Lee, M.Y.; Son, J.S. Catheter-related candidemia caused by Candida haemulonii in a patient in long-term hospital care. J. Korean Med. Sci. 2011, 26, 297–300. [Google Scholar] [CrossRef]
- Cendejas-Bueno, E.; Kolecka, A.; Alastruey-Izquierdo, A.; Theelen, B.; Groenewald, M.; Kostrzewa, M.; Cuenca-Estrella, M.; Gomez-Lopez, A.; Boekhout, T. Reclassification of the Candida haemulonii complex as Candida haemulonii (C. haemulonii group I), C. duobushaemulonii sp. nov. (C. haemulonii group II), and C. haemulonii var. vulnera var. nov.: Three multiresistant human pathogenic yeasts. J. Clin. Microbiol. 2012, 50, 3641–3651. [Google Scholar] [CrossRef] [PubMed]
- Ramos, L.S.; Figueiredo-Carvalho, M.H.; Barbedo, L.S.; Ziccardi, M.; Chaves, A.L.; Zancope-Oliveira, R.M.; Pinto, M.R.; Sgarbi, D.B.; Dornelas-Ribeiro, M.; Branquinha, M.H.; et al. Candida haemulonii complex: Species identification and antifungal susceptibility profiles of clinical isolates from Brazil. J. Antimicrob. Chemother. 2015, 70, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Xiao, M.; Chen, S.C.; Wang, H.; Cheng, J.W.; Chen, X.X.; Xu, Z.P.; Fan, X.; Kong, F.; Xu, Y.C. Identification and antifungal susceptibility profiles of Candida haemulonii species complex clinical isolates from a Multicenter Study in China. J. Clin. Microbiol. 2016, 54, 2676–2680. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, R.S.; Robbins, N.; Cowen, L.E. Regulatory circuitry governing fungal development, drug resistance, and disease. Microbiol. Mol. Biol. Rev. 2011, 75, 213–267. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical Practice Guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2015, 62, e1–e50. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Slavin, M.A.; Sorrell, T.C. Echinocandin antifungal drugs in fungal infections: A comparison. Drugs 2011, 71, 11–41. [Google Scholar] [CrossRef]
- Kuhn, D.M.; George, T.; Chandra, J.; Mukherjee, P.K.; Ghannoum, M.A. Antifungal susceptibility of Candida biofilms: Unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrob. Agents Chemother. 2002, 46, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.W.; Shin, J.H.; Jung, S.I.; Park, K.H.; Cho, D.; Kee, S.J.; Shin, M.G.; Suh, S.P.; Ryang, D.W. Species-specific differences in the susceptibilities of biofilms formed by Candida bloodstream isolates to echinocandin antifungals. Antimicrob. Agents Chemother. 2007, 51, 1520–1523. [Google Scholar] [CrossRef] [PubMed]
- Lazzell, A.L.; Chaturvedi, A.K.; Pierce, C.G.; Prasad, D.; Uppuluri, P.; Lopez-Ribot, J.L. Treatment and prevention of Candida albicans biofilms with caspofungin in a novel central venous catheter murine model of candidiasis. J. Antimicrob. Chemother. 2009, 64, 567–570. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, CLSI Standard M27, 4th ed.; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. Third Informational Supplement, CLSI Document M27-S3; CLSI: Wayne, PA, USA, 2018; Volume 28. [Google Scholar]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. Fourth Informational Supplement, CLSI Document M27S4; CLSI: Wayne, PA, USA, 2012. [Google Scholar]
- CLSI. CLSI Document M60. In Performance Standards for Antifungal Susceptibility Testing of Yeasts, 2nd ed.; CLSI: Wayne, PA, USA, 2020; Volume 40. [Google Scholar]
- Ramos, L.S.; Oliveira, S.S.C.; Souto, X.M.; Branquinha, M.H.; Santos, A.L.S. Planktonic growth and biofilm formation profiles in Candida haemulonii species complex. Med. Mycol. 2017, 55, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Romera, D.; Aguilera-Correa, J.J.; Gadea, I.; Vinuela-Sandoval, L.; Garcia-Rodriguez, J.; Esteban, J. Candida auris: A comparison between planktonic and biofilm susceptibility to antifungal drugs. J. Med. Microbiol. 2019, 68, 1353–1358. [Google Scholar] [CrossRef] [PubMed]
- Peeters, E.; Nelis, H.J.; Coenye, T. Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. J. Microbiol. Methods 2008, 72, 157–165. [Google Scholar] [CrossRef]
- Ramos, L.S.; Mello, T.P.; Branquinha, M.H.; Santos, A.L.S. Biofilm formed by Candida haemulonii species complex: Structural analysis and extracellular matrix composition. J. Fungi 2020, 6, 46. [Google Scholar] [CrossRef]
- Chandra, J.; Mukherjee, P.K.; Ghannoum, M.A. In vitro growth and analysis of Candida biofilms. Nat. Protoc. 2008, 3, 1909–1924. [Google Scholar] [CrossRef]
- Ramage, G.; Rajendran, R.; Sherry, L.; Williams, C. Fungal biofilm resistance. Int. J. Microbiol. 2012, 2012, 528521. [Google Scholar] [CrossRef]
- Beauvais, A.; Schmidt, C.; Guadagnini, S.; Roux, P.; Perret, E.; Henry, C.; Paris, S.; Mallet, A.; Prevost, M.C.; Latge, J.P. An extracellular matrix glues together the aerial-grown hyphae of Aspergillus fumigatus. Cell. Microbiol. 2007, 9, 1588–1600. [Google Scholar] [CrossRef]
- Reséndiz-Sánchez, J.; Ortiz-Alvarez, J.; Casimiro-Ramos, A.; Hernandez-Rodriguez, C.; Villa-Tanaca, L. First report of a catheter-related bloodstream infection by Candida haemulonii in a children’s hospital in Mexico City. Int. J. Infect. Dis. 2020, 92, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.N.; Shin, J.H.; Sung, H.; Lee, K.; Kim, E.C.; Ryoo, N.; Lee, J.S.; Jung, S.I.; Park, K.H.; Kee, S.J.; et al. Candida haemulonii and closely related species at 5 university hospitals in Korea: Identification, antifungal susceptibility, and clinical features. Clin. Infect. Dis. 2009, 48, e57–e61. [Google Scholar] [CrossRef] [PubMed]
- Ruan, S.Y.; Kuo, Y.W.; Huang, C.T.; Hsiue, H.C.; Hsueh, P.R. Infections due to Candida haemulonii: Species identification, antifungal susceptibility and outcomes. Int. J. Antimicrob. Agents 2010, 35, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Crouzet, J.; Sotto, A.; Picard, E.; Lachaud, L.; Bourgeois, N. A case of Candida haemulonii osteitis: Clinical features, biochemical characteristics, and antifungal resistance profile. Clin. Microbiol. Infect. 2011, 17, 1068–1070. [Google Scholar] [CrossRef]
- De Almeida, J.N., Jr.; Assy, J.G.; Levin, A.S.; Del Negro, G.M.; Giudice, M.C.; Tringoni, M.P.; Thomaz, D.Y.; Motta, A.L.; Abdala, E.; Pierroti, L.C.; et al. Candida haemulonii complex species, Brazil, January 2010–March 2015. Emerg. Infect. Dis. 2016, 22, 561–563. [Google Scholar] [CrossRef] [PubMed]
- Muro, M.D.; Motta Fde, A.; Burger, M.; Melo, A.S.; Dalla-Costa, L.M. Echinocandin resistance in two Candida haemulonii isolates from pediatric patients. J. Clin. Microbiol. 2012, 50, 3783–3785. [Google Scholar] [CrossRef]
- Almeida, J.N., Jr.; Motta, A.L.; Rossi, F.; Abdala, E.; Pierrotti, L.C.; Kono, A.S.; Diz Mdel, P.; Benard, G.; Del Negro, G.M. First report of a clinical isolate of Candida haemulonii in Brazil. Clinics 2012, 67, 1229–1231. [Google Scholar] [CrossRef]
- Kathuria, S.; Singh, P.K.; Sharma, C.; Prakash, A.; Masih, A.; Kumar, A.; Meis, J.F.; Chowdhary, A. Multidrug-resistant Candida auris misidentified as Candida haemulonii: Characterization by matrix-assisted laser desorption ionization-time of flight mass spectrometry and DNA sequencing and its antifungal susceptibility profile variability by Vitek 2, CLSI broth microdilution, and Etest method. J. Clin. Microbiol. 2015, 53, 1823–1830. [Google Scholar] [CrossRef]
- Kumar, A.; Prakash, A.; Singh, A.; Kumar, H.; Hagen, F.; Meis, J.F.; Chowdhary, A. Candida haemulonii species complex: An emerging species in India and its genetic diversity assessed with multilocus sequence and amplified fragment-length polymorphism analyses. Emerg Microbes Infect. 2016, 5, e49. [Google Scholar] [CrossRef]
- Fang, S.Y.; Wei, K.C.; Chen, W.C.; Lee, S.J.; Yang, K.C.; Wu, C.S.; Sun, P.L. Primary deep cutaneous candidiasis caused by Candida duobushaemulonii in a 68-year-old man: The first case report and literature review. Mycoses 2016, 59, 818–821. [Google Scholar] [CrossRef]
- Ben-Ami, R.; Berman, J.; Novikov, A.; Bash, E.; Shachor-Meyouhas, Y.; Zakin, S.; Maor, Y.; Tarabia, J.; Schechner, V.; Adler, A.; et al. Multidrug-resistant Candida haemulonii and C. auris, Tel Aviv, Israel. Emerg. Infect. Dis. 2017, 23. [Google Scholar] [CrossRef] [PubMed]
- Isla, G.; Taverna, C.G.; Szusz, W.; Vivot, W.; García-Effron, G.; Davel, G. Candida haemulonii sensu lato: Update of the determination of susceptibility profile in Argentina and literature review. Curr. Fungal Infect. Rep. 2017, 11, 203–208. [Google Scholar] [CrossRef]
- Ramos, R.; Caceres, D.H.; Perez, M.; Garcia, N.; Castillo, W.; Santiago, E.; Borace, J.; Lockhart, S.R.; Berkow, E.L.; Hayer, L.; et al. Emerging multidrug-resistant Candida duobushaemulonii infections in Panama hospitals: Importance of laboratory surveillance and accurate identification. J. Clin. Microbiol. 2018, 56, e00371-18. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.F.; Gade, L.; Chow, N.A.; Loparev, V.N.; Juieng, P.; Berkow, E.L.; Farrer, R.A.; Litvintseva, A.P.; Cuomo, C.A. Genomic insights into multidrug-resistance, mating and virulence in Candida auris and related emerging species. Nat. Commun. 2018, 9, 5346. [Google Scholar] [CrossRef]
- Frías-De-León, M.G.; Martinez-Herrera, E.; Acosta-Altamirano, G.; Arenas, R.; Rodriguez-Cerdeira, C. Superficial candidosis by Candida duobushaemulonii: An emerging microorganism. Infect. Genet. Evol. 2019, 75, 103960. [Google Scholar] [CrossRef]
- Zhang, H.; Niu, Y.; Tan, J.; Liu, W.; Sun, M.A.; Yang, E.; Wang, Q.; Li, R.; Wang, Y.; Liu, W. Global screening of genomic and transcriptomic factors associated with phenotype differences between multidrug-resistant and -susceptible Candida haemulonii strains. mSystems 2019, 4. [Google Scholar] [CrossRef]
- Di Luca, M.; Koliszak, A.; Karbysheva, S.; Chowdhary, A.; Meis, J.F.; Trampuz, A. Thermogenic characterization and antifungal susceptibility of Candida auris by microcalorimetry. J. Fungi 2019, 5, 103. [Google Scholar] [CrossRef]
- Bastos, R.W.; Rossato, L.; Valero, C.; Lagrou, K.; Colombo, A.L.; Goldman, G.H. Potential of Gallium as an antifungal agent. Front. Cell Infect. Microbiol. 2019, 9, 414. [Google Scholar] [CrossRef]
- Xiao, M.; Chen, S.C.; Kong, F.; Xu, X.L.; Yan, L.; Kong, H.S.; Fan, X.; Hou, X.; Cheng, J.W.; Zhou, M.L.; et al. Distribution and antifungal susceptibility of Candida species causing candidemia in China: An update from the CHIF-NET Study. J. Infect. Dis. 2020, 221, S139–S147. [Google Scholar] [CrossRef]
- Gade, L.; Muñoz, J.F.; Sheth, M.; Wagner, D.; Berkow, E.L.; Forsberg, K.; Jackson, B.R.; Ramos-Castro, R.; Escandón, P.; Dolande, M.; et al. Understanding the emergence of multidrug-resistant Candida: Using Whole-Genome Sequencing to describe the population structure of Candida haemulonii species complex. Front. Genet. 2020, 11, 554. [Google Scholar] [CrossRef]
- Lima, S.L.; Francisco, E.C.; De Almeida Júnior, J.N.; Santos, D.W.C.L.; Carlesse, F.; Queiroz-Telles, F.; Melo, A.S.A.; Colombo, A.L. Increasing prevalence of multidrug-resistant Candida haemulonii species complex among all yeast cultures collected by a reference laboratory over the past 11 years. J. Fungi 2020, 6, 110. [Google Scholar] [CrossRef] [PubMed]
- Coles, M.; Cox, K.; Chao, A. Candida haemulonii: An emerging opportunistic pathogen in the United States? IDCases 2020, 21, e00900. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.S.; Gazara, R.K.; Passarelli-Araujo, H.; Valengo, A.E.; Pontes, P.V.M.; Nunes-da-Fonseca, R.; De Souza, R.F.; Venancio, T.M.; Dalla-Costa, L.M. First genome sequences of two multidrug-resistant Candida haemulonii var. vulnera isolates from pediatric patients with candidemia. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Kordalewska, M.; Lee, A.; Park, S.; Berrio, I.; Chowdhary, A.; Zhao, Y.; Perlin, D.S. Understanding echinocandin resistance in the emerging pathogen Candida auris. Antimicrob. Agents Chemother. 2018, 62, e00238-18. [Google Scholar] [CrossRef] [PubMed]
- Sherry, L.; Ramage, G.; Kean, R.; Borman, A.; Johnson, E.M.; Richardson, M.D.; Rautemaa-Richardson, R. Biofilm-forming capability of highly virulent, multidrug-resistant Candida auris. Emerg. Infect. Dis. 2017, 23, 328–331. [Google Scholar] [CrossRef]
- Kean, R.; Delaney, C.; Sherry, L.; Borman, A.; Johnson, E.M.; Richardson, M.D.; Rautemaa-Richardson, R.; Williams, C.; Ramage, G. Transcriptome assembly and profiling of Candida auris reveals novel insights into biofilm-mediated resistance. mSphere 2018, 3, e00334-18. [Google Scholar] [CrossRef]
- Guembe, M.; Guinea, J.; Marcos-Zambrano, L.J.; Fernandez-Cruz, A.; Pelaez, T.; Munoz, P.; Bouza, E. Micafungin at physiological serum concentrations shows antifungal activity against Candida albicans and Candida parapsilosis biofilms. Antimicrob. Agents Chemother. 2014, 58, 5581–5584. [Google Scholar] [CrossRef]
- Ziccardi, M.; Souza, L.O.; Gandra, R.M.; Galdino, A.C.; Baptista, A.R.; Nunes, A.P.; Ribeiro, M.A.; Branquinha, M.H.; Santos, A.L.S. Candida parapsilosis (sensu lato) isolated from hospitals located in the Southeast of Brazil: Species distribution, antifungal susceptibility and virulence attributes. Int. J. Med. Microbiol. 2015, 305, 848–859. [Google Scholar] [CrossRef]
- Simitsopoulou, M.; Peshkova, P.; Tasina, E.; Katragkou, A.; Kyrpitzi, D.; Velegraki, A.; Walsh, T.J.; Roilides, E. Species-specific and drug-specific differences in susceptibility of Candida biofilms to echinocandins: Characterization of less common bloodstream isolates. Antimicrob. Agents Chemother. 2013, 57, 2562–2570. [Google Scholar] [CrossRef]
- Melo, A.S.; Colombo, A.L.; Arthington-Skaggs, B.A. Paradoxical growth effect of caspofungin observed on biofilms and planktonic cells of five different Candida species. Antimicrob. Agents Chemother. 2007, 51, 3081–3088. [Google Scholar] [CrossRef]
- Ferreira, J.A.; Carr, J.H.; Starling, C.E.; De Resende, M.A.; Donlan, R.M. Biofilm formation and effect of caspofungin on biofilm structure of Candida species bloodstream isolates. Antimicrob. Agents Chemother. 2009, 53, 4377–4384. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, M.D.; Whyte, J.A.; Odds, F.C. Candida albicans and Candida dubliniensis respond differently to echinocandin antifungal agents in vitro. Antimicrob. Agents Chemother. 2007, 51, 1882–1884. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).