Abstract
Simple Summary
Our study describes several species of Phaeosphaeriaceae found in terrestrial habitats in Sichuan Province, China. We used morphological and molecular data to accurately delimit these species and determine their taxonomic relationships within the family. Our findings contribute to understanding fungal diversity in China and provide a basis for future studies exploring the ecological roles and biotechnological potential of these fungi. Additionally, our multi-gene phylogenetic approach provides increased accuracy and resolution in the delimitation of species boundaries within Phaeosphaeriaceae.
Abstract
The family Phaeosphaeriaceae is a diverse group of ascomycetous fungi that are commonly found in terrestrial habitats worldwide. In recent years, there has been increasing interest in the biodiversity of Phaeosphaeriaceae in China, particularly in Sichuan Province, which has not been fully explored for its high fungal diversity. In our study, we conducted extensive surveys in Sichuan Province to identify and describe new species of Ascomycota with diverse habitats. Here, we present a taxonomic revision of Phaeosphaeriaceae with taxonomic novelties from terrestrial habitats in Sichuan Province based on a multi-gene phylogenetic approach. Our study focuses on the description of four new species of Phaeosphaeriaceae, representing novel additions to the diversity of this fungal family. Using a combination of morphological and molecular data, we provide detailed descriptions of the new taxa and their placement within the family. Additionally, we discuss the phylogenetic relationships of these new taxa with other members of Phaeosphaeriaceae, providing insights into the correct taxonomic classification of the family. Our taxonomic revision contributes to understanding fungal diversity in China and provides a foundation for future studies investigating the taxonomy and ecological roles of Phaeosphaeriaceae fungi. Furthermore, our multi-gene phylogenetic approach provides increased resolution and accuracy in the delimitation of species boundaries within the family. Our study highlights the importance of continued exploration and taxonomic revision in order to fully understand the diversity and distribution of fungal species in China and beyond. New species: Paraloratospora sichuanensis, Phaeosphaeria chengduensis, P. sichuanensis, and Septoriella shoemakeri. New combinations: Paraloratospora breonadiae, P. fructigena, Septoriella ammophilicola, S. asparagicola, S. festucae, S. luzulae, and S. verrucispora. New names: Septoriella paradactylidis, and S. neomuriformis.
1. Introduction
The family Phaeosphaeriaceae comprises a diverse group of ascomycetous fungi that are commonly found in terrestrial habitats worldwide [1]. Despite their abundance, the taxonomy of Phaeosphaeriaceae remains obscure, with many species remaining undescribed or poorly characterized. In recent years, there has been an increasing interest in the biodiversity of Phaeosphaeriaceae. Researchers have conducted extensive surveys of the mycobiota, resulting in the discovery of over 135 new taxa in Phaeosphaeriaceae within the last five years: 2019–2023 [2]. In particular, several new species of Phaeosphaeria have been identified, representing novel additions to the diversity of this fungal family. However, the taxonomy of Phaeosphaeriaceae is still challenging due to the high degree of morphological variability within the family. Many species have been described based on only a single specimen, and the use of molecular data to aid in the delimitation of species boundaries for the species published prior to the 1990s is still inadequate. Currently, the family consists of 84 genera [3], but their exact classification is debatable. The phylogenetic relationships within the family are also not well resolved. Twenty-one of the genera are represented by a single strain in the family (viz., Acericola, Alloneottiosporina, Arezzomyces, Banksiophoma, Bhagirathimyces, Bhatiellae, Camarosporioides, Equiseticola, Hydeopsis, Jeremyomyces, Kwanghwaensis, Longispora, Melnikia, Neosphaerellopsis, Ophiosimulans, Pseudophaeosphaeria, Scolicosporium, Vittaliana, Vrystaatia, Xenophaeosphaeria, and Xenophoma), whereas thirty of the genera are represented by a single species (monotypic). Additionally, some of the species are not monophyletic with their type (i.e., Muriphaeosphaeria, Loratospora, and Phaeosphaeria). Since the last revision [4], there has been no proper attempt to resolve the inter- and intra-generic classification of Phaeosphaeriaceae, indicating the need for a new taxonomic revision of the family.
Collecting species in Phaeosphaeriaceae is crucial because it enables more accurate and precise identification of its species, which is particularly important for linking their holomorphs and finding DNA-based sequence data for old herbarium specimens. Moreover, this helps fill the gap in our understanding of their diversity and evolution [5]. Since Phaeosphaeriaceae are cosmopolitan and ecologically important, studying their ecological relationships is essential to understand them in a broader sense [1,4]. In addition, some genera (i.e., Polydomus and Setophoma) have significant practical applications, including as sources of biological activities and biotechnological products [6,7,8]. Therefore, conducting research on Phaeosphaeriaceae is an important step towards unlocking their potential applications. Accurate identification of Phaeosphaeriaceae species is also necessary for assessing their conservation status and identifying areas of high biodiversity that require protection.
This paper presents a taxonomic revision of Phaeosphaeriaceae, focusing on the genera Paraloratospora, Phaeosphaeria, and Septoriella. Four new species, seven new combinations, and two new names are proposed, supported by detailed morphological and molecular analyses to establish their placement within this family. Furthermore, we investigate the phylogenetic relationships of these novel taxa with other members of Phaeosphaeriaceae and their current taxonomic classification. Additionally, we confirm the species list that includes DNA data in Phaeosphaeria sensu stricto. This study highlights the significance of ongoing exploration and taxonomic revision to comprehensively understand fungal species diversity and distribution in China and beyond. It contributes to our knowledge of the diversity and evolution of Phaeosphaeriaceae in terrestrial habitats and establishes a foundation for future taxonomic and ecological investigations within this important fungal family.
2. Materials and Methods
2.1. Isolates and Specimens
During our fieldwork in Sichuan Province, China, we collected typical black ascomata/conidiomata from dead twigs across various regions including Chenghua District, Qingyang District, and Dayi County during both wet (August 2021) and dry (January 2023) seasons. The collected samples were placed in Ziploc bags and transported to the mycology laboratory of the Kunming Institute of Botany, where they were stored in paper envelopes. Single spore isolation was conducted according to the methods described in Wanasinghe et al. [9], and germinated spores were handled appropriately. Dried specimens were preserved in the fungarium of the Cryptogams Kunming Institute of Botany, Academia Sinica (KUN-HKAS), and representative cultures were deposited in the Kunming Institute of Botany Culture Collection (KUMCC) and the University of Electronic Science and Technology Culture Collection (UESTCC), Chengdu, China. Nomenclatural data for fungal novelties were deposited in MycoBank [10].
2.2. Morphological Observations
To examine the ascomata, conidiophores, and conidia from natural substrates, we rehydrated them with tap water and viewed them using a Motic SMZ 168 series stereo-microscope (Motic Asia, Kowloon, Hong Kong). Morphological characteristics were evaluated by hand-sectioning sporocarps placed on water-mounted glass slides. We assessed various characteristics such as the diameter, height, color, and shape of the ascomata/conidiomata, as well as the width of the peridium and the height and diameter of the ostioles. Microscopic photography was carried out using a Nikon ECLIPSE Ni (Nikon Corporation, Konan, Minato-ku, Tokyo, Japan) compound microscope with differential interference contrast (DIC) and phase contrast (PC) illumination. Images of microscopic structures were captured using a Canon EOS 600D (Canon Inc., Ota, Tokyo, Japan) camera. Additionally, macroscopic images of colonies were documented using an iPhone XS Max (Apple Inc., Cupertino, CA, USA) in daylight. We used the Tarosoft (R) Image Frame Work program to measure dimensions, and images used for figures were processed using Adobe Photoshop CS6 (Adobe Systems, San Jose, CA, USA).
2.3. DNA Extraction, PCR Amplifications, and Sequencing
To extract genomic DNA, we followed the methods described by Wanasinghe et al. [11] using the Biospin Fungus Genomic DNA Extraction Kit-BSC14M1 (BIOER, Hangzhou Bioer Technology Co., Ltd., Hangzhou, China) according to the manufacturer’s instructions. Reference DNA for the polymerase chain reaction (PCR) was stored at 4 °C for regular use and duplicated at −20 °C for long-term storage. We used primers ITS5/ITS4, LR0R/LR5, NS1/NS4, EF1-983F/EF1-2218R, and fRPB2-5f/fRPB2-7cR to amplify the DNA sequences of the internal transcribed spacers (ITS), partial 28S large subunit rDNA (LSU), partial 18S small subunit rDNA (SSU), translation elongation factor 1-α (tef1), and RNA polymerase II second largest subunit (rpb2) [12,13,14,15,16,17]. The protocols used for PCR amplification (SSU, LSU, ITS, tef1, and rpb2) were the same as those described in Wanasinghe and Mortimer [18]. The amplified PCR fragments were sent to a private company (BGI, Ltd., Shenzhen, China) for sequencing.
2.4. Molecular Phylogenetic Analyses
2.4.1. Sequencing and Sequence Alignment
To analyze the sequences generated from different primers of the five genes, we conducted a BLAST search to identify sequences with high similarity indices and find the closest matches with taxa in Phaeosphaeriaceae, following recent publications [19,20,21,22]. Later, we analyzed them with other sequences retrieved from GenBank (Table 1). We used MAFFT v. 7 [23] to automatically generate multiple alignments of all consensus sequences as well as the reference sequences. We manually corrected the alignments using BioEdit v. 7.0.5.2 [24] where necessary.
2.4.2. Phylogenetic Analyses
We examined the single-locus datasets for any topological incongruence among the loci for members of the analyses. The resulting alignments were then concatenated into a multi-locus alignment and analyzed using maximum likelihood (ML) and Bayesian (BI) phylogenetic methods in the CIPRES Science Gateway [25]. We obtained an ML tree using RAxML-HPC2 on XSEDE v. 8.2.10 [26] with a GTR + G + I model and calculated support values with 1000 bp replicates [27]. Nodes with ML bootstrap values equal to or greater than 75% are indicated above each node.
The best-fit model was selected based on Bayesian Information Criterion (BIC) scores using the IQ-TREE web application at http://iqtree.cibiv.univie.ac.at (accessed on 27 June 2023) [28]. We restricted the pool of available models to JC, F81, HKY, SYM, and GTR for model selection. We performed BI with two parallel runs of 50 M generations, using four chains in each, and retaining one tree every 100 generations. The dataset was partitioned by gene, and HKY + I + G (SSU), SYM + I + G (LSU), and GTR + I + G (ITS, tef1, and rpb2) models were applied to each partition, ending the run automatically when the standard deviation of split frequencies dropped below 0.01 with a burn-in fraction of 0.25. A 50% majority rule consensus tree was obtained after discarding the first 25% of trees, and posterior probabilities were used as a measure of nodal support. Nodes with posterior probabilities in BI (BYPP) greater than 0.95 are indicated above each node. Finally, we visualized the phylograms using the FigTree v1.4.0 program [29] and reorganized them in Microsoft PowerPoint (2019).
3. Results
3.1. Phylogenetic Analyses
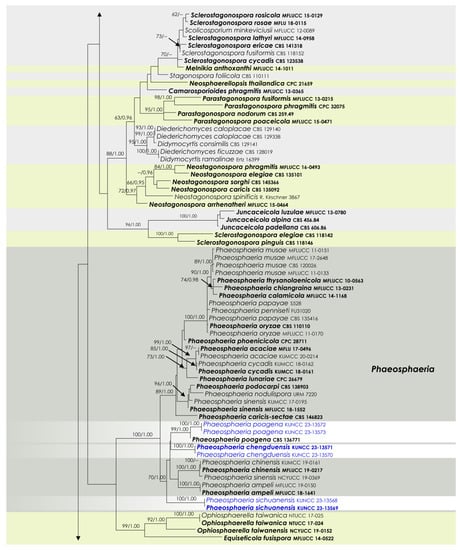
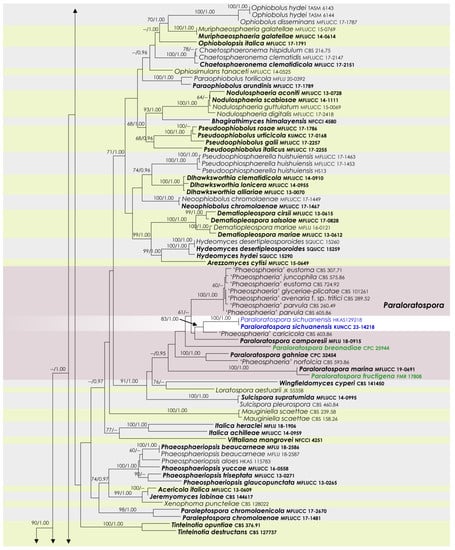
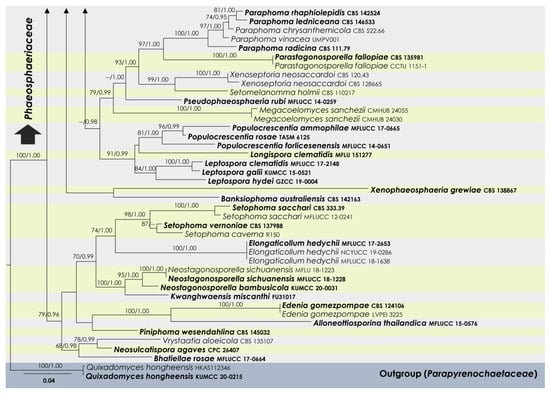
To examine the evolutionary relationships of our new strains within Phaeosphaeriaceae, we conducted phylogenetic analyses based on the combined SSU, LSU, ITS, tef1, and rpb2 DNA sequences of 258 representatives of the family. We used two strains from Quixadomyces hongheensis (Parapyrenochaetaceae) as the outgroup taxon. The full dataset consisted of 4413 characters, including gaps (SSU = 999 characters, LSU = 846, ITS1 + 5.8S + ITS2 = 663, tef1 = 871, and rpb2 = 1034). The RAxML analysis of the combined dataset yielded a best-scoring tree with a final ML optimization likelihood value of −60,293.418721. The matrix had 2211 distinct alignment patterns, with 39.5% undetermined characters or gaps. We used the GTR + I + G model of the combined amplicons for the analysis, with the following parameters: estimated base frequencies of A = 0.245652, C = 0.237255, G = 0.265783, and T = 0.251310; substitution rates of AC = 1.259144, AG = 3.770291, AT = 2.003190, CG = 0.806828, CT = 7.047351, and GT = 1.000; proportion of invariable sites I = 0.575954; and gamma distribution shape parameter α = 0.641179. The Bayesian analysis ran 40,280,000 generations before the average standard deviation for split frequencies reached below 0.01 (0.009998). The analyses generated 40,281 trees, from which we sampled 30,211 trees after discarding the first 25% as burn-in. The alignment contained a total of 2217 (SSU: 340, LSU: 325, ITS: 461, tef1: 415, and rpb2: 676) unique site patterns. Where applicable, the phylogenetic results obtained (Figure 1) are discussed in the descriptive notes below.
Figure 1.
RAxML tree based on a combined dataset of partial SSU, LSU, ITS, tef1, and rpb2 DNA sequence analyses in Phaeosphaeriaceae. The bootstrap support values for ML (MLB) greater than or equal to 70% and Bayesian posterior probabilities (BYPP) greater than or equal to 0.95 are indicated as MLB/BYPP above the corresponding nodes. The newly analyzed isolates are highlighted in blue. The scale bar in the figure represents the expected number of nucleotide substitutions per site.
3.2. Taxonomy
Pleosporales Luttr. ex M.E. Barr, Prodromus to class Loculoascomycetes: 67 (1987).
Phaeosphaeriaceae M.E. Barr, Mycologia 71: 948 (1979).
Paraloratospora Bundhun, Tennakoon, Phookamsak & K.D. Hyde, Fungal Diversity 100: 101 (2020).
Notes: This study presents an updated and comprehensive phylogenetic classification of the genus Paraloratospora, incorporating SSU, LSU, ITS, tef1, and rpb2 DNA sequence analyses. By combining morphological and phylogenetic considerations, we have identified a new species, Paraloratospora sichuanensis, as well as proposed two new combinations, Paraloratospora breonadiae (=Phaeosphaeria breonadiae) and P. fructigena (=Phaeosphaeria fructigena), within the genus. Detailed information regarding these taxonomic changes can be found in the note sections, where an additional discussion and supporting evidence are provided. The discovery of Paraloratospora sichuanensis adds to the known diversity of Paraloratospora species and expands our knowledge of the ecological and morphological characteristics within the genus. The two newly proposed combinations, Paraloratospora breonadiae and P. fructigena, reflect the revised taxonomic understanding based on the integration of molecular and morphological data. These taxonomic changes contribute to the overall refinement and accuracy of the classification system for Paraloratospora.
Paraloratosporabreonadiae (Crous & Jol. Roux) Maharachch. & Wanas. comb. nov.
MycoBank: MB 849359.
≡Phaeosphaeria breonadiae Crous & Jol. Roux, Persoonia 36: 399 (2016).
Holotype: South Africa, Limpopo Province, Wolkberg, on leaves of Breonadia microcephala, January 2015, J. Roux (CBS H-22631, holotype), ex-type, CPC 25944, CBS 141334.
Descriptions and illustrations: See Crous et al. [30].
Notes: The phylogenetic analysis conducted in this study yielded interesting results regarding the relationship between Phaeosphaeria breonadiae and P. fructigena with the species of Paraloratospora. The analysis revealed that type strains of these two species clustered together with Paraloratospora, forming a distinct group separate from the Phaeosphaeria sensu stricto (Figure 1). Based on these findings, it was determined that a reclassification of these two species within the genus Paraloratospora was warranted. Consequently, we propose the new combinations Paraloratospora breonadiae comb. nov. and P. fructigena comb. nov. for Phaeosphaeria breonadiae and P. fructigena, respectively. This reclassification aligns with the phylogenetic evidence and provides a more accurate taxonomic placement for these species within the Paraloratospora genus. The newly proposed combinations reflect the closer relationship and shared characteristics observed between these species and other members of Paraloratospora.
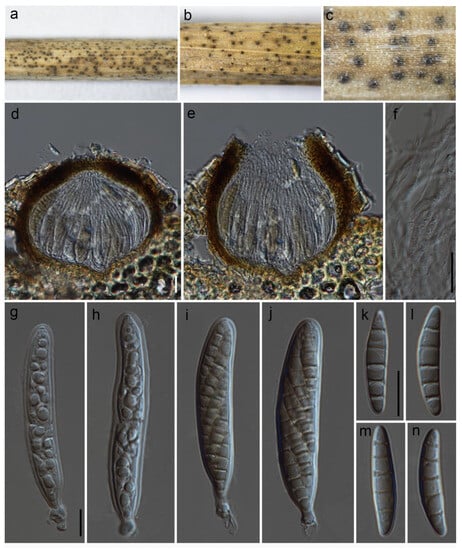
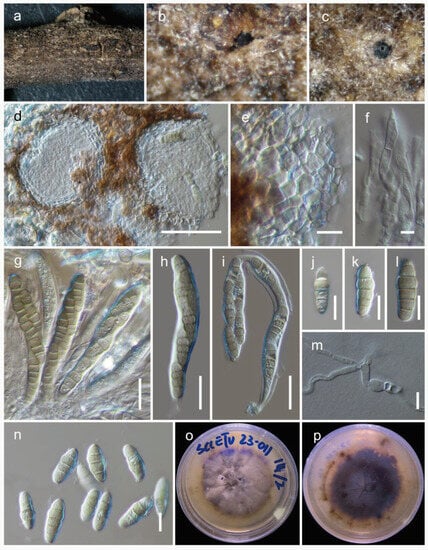
Paraloratospora sichuanensis Maharachch. & Wanas. sp. nov. (Figure 2).
Figure 2.
Paraloratospora sichuanensis (HKAS 129217, holotype): (a–c) ascomata on the host; (d,e) cross section of ascomata; (f) pseudoparaphyses; (g–j) asci; (j–n) ascospores. Scale bars: (d,f) 20 µm; (g,k) 10 µm (scale bar of (d) applies to (e), scale bar of (g) applies to (g–j), and scale bar of (k) applies to (k–n)).
MycoBank: MB 849432.
Etymology: The specific epithet is derived from Sichuan, where this fungus is collected.
Holotype: HKAS 129217
The fungus is a saprobe, found on dead clumps of Lolium perenne L. (Poaceae). In its sexual morph, Ascomata are 130–190 μm high and have a 120–210 μm diam. (M = 165 × 170 µm, n = 10), are semi-immersed to erumpent, solitary, scattered, and uniloculate, and can be globose to subglobose in shape, with a brown to dark brown color. The ostioles, which can reach up to 3–40 µm in diam., are centrally located and have a minute papilla. They are dark brown and consist of hyaline periphyses. The peridium is of unequal thickness, with a thicker portion near the apex (15–24 µm) and a thinner portion at the base (10–16 µm). It is composed of two layers, with the outer part consisting of dark brown thick-walled cells arranged in a textura angularis pattern and the inner layer made up of pale brown to hyaline thin-walled cells also arranged in a textura angularis pattern. The hamathecium is 2.5–4.5 µm wide, unbranched, septate, and composed of pseudoparaphyses that are constricted at the septum. The asci 55–90 × 7–14 µm (M = 62.3 × 11 µm, n = 15) are eight-spored, bitunicate, fissitunicate, cylindrical, sessile to subsessile, with a rounded apex and a thick wall. They are short pedicellate and possess a developed ocular chamber. The ascospores 20–28 × 3–5.5 µm (M = 23 × 4.5 µm, n = 25) are arranged in overlapping 1–2 seriate fashion and are 3–5 septate (mostly 5). The ascospores are hyaline to pale yellowish in color and have a fusiform shape with rounded ends. They can be straight to slightly curved and have a smooth surface. They are surrounded by a sheath. Asexual morph: the asexual form of this fungus is undetermined.
Culture characteristics: The ascospores germinated on PDA within 24 h. Following a two-week incubation period at 25 °C, the colonies on PDA medium reached a diameter of 5 cm. These colonies exhibited an undulate margin, initially appearing creamy whitish and transitioning to a creamy grey shade at the center. Towards the periphery, the color lightened further. The colonies were slightly raised in texture, with a creamy orange hue at the center and a creamy grey coloration towards the periphery when observed from the reverse side.
Known distribution: China (Sichuan) on Lolium perenne in terrestrial habitats.
Material examined: China, Sichuan, Chengdu, Chenghua District, The Chengdu Research Base of Giant Panda Breeding, on dead culms of Lolium perenne, 13 August 2021, Q Wang, W90-4-3 (HKAS 129217, holotype), ex-type culture, KUNCC 23-14218. ibid. W90-4-1 (HKAS 129218).
Notes: Based on the phylogenetic analysis of multiple genes, Paraloratospora sichuanensis shares a close relationship with a putatively named strain called ‘Phaeosphaeria’ caricicola (CBS 603.86). However, it is important to note that de Gruyter et al. [31] provided sequence data for CBS 603.86 without establishing a connection to any type material. In terms of morphology, Paraloratospora sichuanensis does not significantly differ from Phaeosphaeria caricicola in several aspects. Both species exhibit similar dimensions in terms of ascomata size (130–190 μm high, 120–210 μm diameter for P. sichuanensis, compared to 120–140 μm high, 120–140 μm diameter for Phaeosphaeria caricicola), asci size (55–90 × 7–14 µm for P. sichuanensis, compared to 60–70 × 6–8 µm for Phaeosphaeria caricicola), and ascospore size (20–28 × 3–5.5 µm for P. sichuanensis, compared to 15–20 × 3.5–4 µm for Phaeosphaeria caricicola) [32,33]. However, a notable difference lies in the number of septa observed in the ascospores of these two species. The ascospores of P. sichuanensis predominantly possess five septa, whereas Phaeosphaeria caricicola typically has ascospores with four septa [33]. It is worth noting that further investigation is required to establish the exact phylogenetic placement of the type of Phaeosphaeria caricicola.
Paraloratospora fructigena (Magaña-Dueñas, Cano-Lira & Stchigel) Maharachch. & Wanas. comb. nov.
MycoBank: MB 849360.
≡Phaeosphaeria fructigena Magaña-Dueñas, Cano-Lira & Stchigel, Journal of Fungi 7 (12, no. 1102): 11 (2021).
Holotype: Spain, Tarragona Province, Capafonts (41.29598, 1.02753), from freshwater submerged plant debris, March 2019, V Magaña-Dueñas and II González (CBS H-24910), ex-type FMR 17808, CBS 148658.
Descriptions and illustrations: See Magaña-Dueñas et al. [34].
Notes: Phaeosphaeria fructigena, which was isolated from plant debris submerged in freshwater, is characterized by the production of clavate asci and fusiform ascospores. The sexual stage of P. fructigena has only been observed in both the original material and pure cultures by Magaña-Dueñas et al. [34]. In our phylogenetic analysis, P. fructigena was grouped under Paraloratospora. Please refer to the notes under Paraloratospora breonadiae for further information.
Phaeosphaeria I. Miyake, Bot. Mag. (Tokyo) 23: 93 (1909).
Notes: The genus Phaeosphaeria is renowned for its significant diversity, encompassing numerous described species and many more yet to be discovered. The presence of Phaeosphaeria species has been reported in various ecosystems, including terrestrial and freshwater environments [34,35]. These fungi exhibit adaptability to a wide range of climatic conditions and substrates, allowing their distribution in both temperate and tropical regions. Within this genus, some species play a crucial role as saprophytes, participating in vital processes such as decomposition and nutrient cycling. They are commonly associated with decaying plant material, dead wood, or organic debris of monocotyledons. On the other hand, certain Phaeosphaeria species are recognized as plant pathogens, causing diseases in various hosts, including agricultural crops and forest trees [4,35].
In our study, we observed that six strains of our newly isolated species exhibit close resemblance to Phaeosphaeria strains based on BLAST similarity indices in GenBank. These findings underscore the importance of conducting further investigations and taxonomic revisions to ensure accurate classification and a better understanding of the relationships within the Phaeosphaeria genus. By utilizing a combination of morphological and molecular data from these new collections, we provide detailed descriptions of two new species within Phaeosphaeria. Additionally, we revised the species description for Phaeosphaeria poagena to update the characteristics of its sexual form.
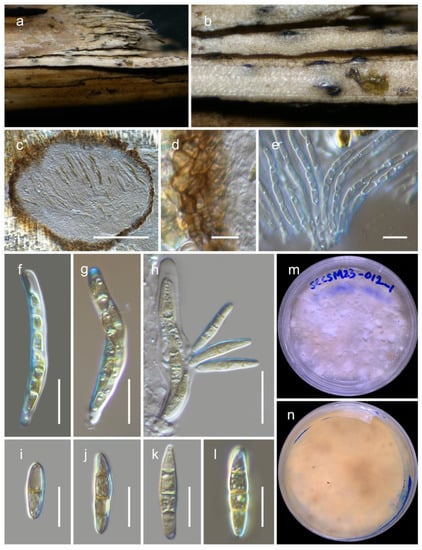
Phaeosphaeria chengduensis Wanas. & Maharachch. sp. nov. (Figure 3).
Figure 3.
Phaeosphaeria chengduensis (HKAS 129197, holotype): (a–c) herbarium specimen; (d) vertical section through ascomata; (e) peridium; (f) pseudoparaphyses; (g–i) asci; (j–n) ascospores (m germinated spore); (o,p) colonies on PDA after 6 weeks. Scale bars: (d) 50 µm; (e,j–n) 10 µm; (f) 5 µm; (g–i) 20 µm.
MycoBank: MB 849352.
Etymology: The specific epithet is derived from Chengdu, where this fungus was collected.
Holotype: HKAS 129197.
The fungus is saprobic, found on a dead twig of an unidentified deciduous host. Sexual state: The ascomata are 100–170 μm high, 90–150 μm in diam., and mostly scattered and immersed, with a uniloculate structure that is globose to subglobose and brown to dark brown in color. The peridium is 10–20 μm wide and consists of two layers: an outer layer composed of brown to dark brown cells arranged in a textura angularis pattern and an inner layer with loosely arranged, hyaline cells in a textura angularis to textura globulosa pattern. The hamathecium is made up of pseudoparaphyses, measuring 2–3 μm in width, which are cellular and indistinctly constricted at the septa. The asci measure 80–100 × 12–15 μm (M = 90.6 × 13.6 μm, n = 15) and are eight-spored, bitunicate, fissitunicate, broadly cylindrical, and have a pedicel. They are rounded at the apex and possess an ocular chamber. Ascospores measure 18–26 × 6.5–9 μm (M = 20.6 × 7.8 μm, n = 20) and are arranged as overlapping 1–2 seriate. They are phragmosporous, ellipsoidal with rounded ends, and initially yellowish brown but becoming pale brown as they mature. The ascospores are 4–5 septate and exhibit slight constriction at the septa, with the upper cell next to the middle septum being enlarged. Their surfaces are smooth walled. The asexual state was not observed.
Culture characteristics: Colonies on PDA reach a diameter of up to 3 cm after four weeks at 25 °C. These colonies have an irregular, flattened to slightly raised appearance, with a greenish-grey edge and a grey center. They also display various color sectors ranging from grey to dark grey. The reverse side of the colonies appears dark brown.
Known distribution: China (Sichuan) on dead twigs of deciduous hosts in terrestrial habitats.
Material examined: China, Sichuan, Chengdu, Chenghua, 30.748056 N, 103.928889 E, 533 m, on dead twigs of an unknown deciduous host, 03 January 2023, D.N. Wanasinghe, SCCETU23-011-4 (HKAS 129197, holotype), ex-holotype culture, KUNCC 23-13571. ibid. 30.747694 N, 103.928749 E, 03 January 2023, SCCETU23-011-1 (HKAS 129198), living culture, KUNCC 23-13570.
Notes: Within Phaeosphaeria, our novel fungus closely resembles P. arenaria (≡Leptosphaena arenaria) and P. hiemalis (≡Leptosphaeria hiemalis) based on the presence of 4–5 septate, pigmented, ellipsoidal ascospores with rounded ends [33]. Phaeosphaeria hiemalis was collected from Equisetum hyemale (Equisetaceae) in Canada and USA. Phaeosphaeria arenaria was collected from Festuca arenaria (Poaceae) and Phleum arenarium (Poaceae), although the exact location is unknown [33]. Both of these species lack DNA-based sequence data for molecular comparisons. In our phylogenetic analysis, Phaeosphaeria chengduensis shows a close phylogenetic affinity to P. poagena. Morphologically, Phaeosphaeria poagena produces conical, fusiform spores with 3 septa, whereas P. chengduensis has ellipsoidal spores with rounded ends and 4–5 septa. However, their affiliation is not statistically supported, with a greater than or equal to 70% MLB or 0.95 BYPP. The comparison of nucleotide differences of ITS, tef1, and rpb2 between Phaeosphaeria chengduensis and P. poagena were 17/492 (3.4%), 25/869 (2.9%), and 64/621 (10.3%), respectively.
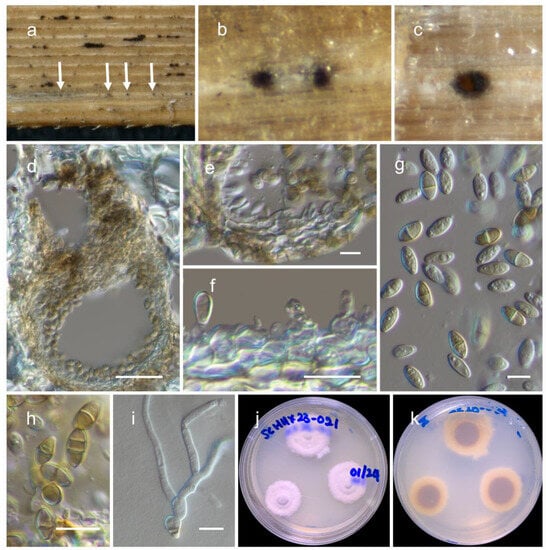
Figure 4.
Phaeosphaeria poagena (HKAS 129196): (a,b) ascomata on the bamboo culms; (c) vertical section through an ascoma; (d) peridium; (e) pseudoparaphyses; (f–h) asci ((h) showing a fissitunicate ascus); (i–l) ascospores; (m,n) colonies on PDA after 6 weeks. Scale bars: (c) 100 µm; (d,e,i–l) 10 µm; (f–h) 20 µm.
MycoBank: MB 808889.
The fungus is saprobic, found on deceased bamboo (Poaceae). Sexual state: Ascomata are 200–250 μm high, 240–280 μm in diam., and mostly scattered. They are typically scattered, partially embedded, or semi-erupted, appearing as small black dots on the surface of the host. These ascomata are unilocular, globose to subglobose, and range in color from brown to dark brown. The peridium, 15–25 μm wide, consists of thin-walled cells arranged in a textura angularis pattern, and is composed of brown to dark brown cells. The hamathecium is made up of numerous pseudoparaphyses, measuring 2–3 μm in width, that are cellular and contain guttulate material. These pseudoparaphyses are constricted at the septa. The asci measure 65–80 × 8.5–10.5 μm (M = 71.6 × 9.8 μm, n = 15), are eight-spored, bitunicate, fissitunicate, broadly cylindrical, and have a short pedicel. They are rounded at the apex and possess an ocular chamber. Ascospores measure 18–26 × 4–5 μm (M = 23.2 × 4.6 μm, n = 20) and are arranged as overlapping 1–2 seriate. They are phragmosporous, fusiform with conical ends, and initially pale yellowish-brown but becoming yellowish-brown as they mature. The ascospores are three septate and exhibit slight constriction at the septa. Their surfaces are rough walled. Asexual morph [36]: The pycnidial conidiomata are globose, black, erumpent, and possess a central ostiole. The pycnidial wall consists of 2–3 layers of brown textura angularis. Conidiophores are reduced to conidiogenous cells, which are hyaline, smooth, and doliiform. These cells exhibit prominent periclinal thickening or tightly aggregated percurrent proliferations. The conidia are solitary, brown, smooth, fusoid-ellipsoidal to subcylindrical, (1-)3 septate, and slightly constricted at the septa, with a subobtuse apex and a truncate base. They measure (8–)12–14(–16) × (2.5–)3(–3.5) µm.
Culture characteristics: After four weeks at 25 °C, colonies on potato dextrose agar (PDA) attained a diameter of up to 4 cm. These colonies exhibit an irregular, flattened to slightly raised morphology and display various color sectors ranging from white to creamy orange. The reverse side of the colonies appears creamy orange, with occasional dark patches that can be observed.
Known distribution: Crous et al. [36] identified this fungus in the Netherlands (specifically in Raalte), where it was found on Poa sp. (Poaceae) in a terrestrial habitat. In this study, we found it in China (specifically in Sichuan) on deceased bamboo culms (Poaceae). The habitat where it was discovered is terrestrial and typically covered with snow throughout the year.
Material examined: China, Sichuan, Dayi County, Xiling snow mountain, 30.684110 N, 103.164559 E, 3162 m, on dead bamboo, 06 January 2023, D.N. Wanasinghe, SCCSM23-012A-2 (HKAS 129196), living culture, KUNCC 23-13572. ibid. 30.684444 N, 103.164444 E, 2980 m, 6 January 2023, SCCSM23-012A-3 (HKAS 129195), living culture, KUNCC 23-13573.
Notes: The asexual form of Phaeosphaeria poagena was isolated from Poa sp. (Poaceae) and was introduced by Crous et al. [36]. Phaeosphaeria poagena is described as a new species because its small conidial dimensions do not match any of the asexual forms previously documented on Poa [32,33,37]. In our study, two strains from a sexually reproductive fungus formed a monophyletic clade with the type strain of Phaeosphaeria poagena (CBS 136771) with 99% MLB and 1.00 BYPP support values. A comparison of the ITS nucleotides between our new strains and the type strain of Phaeosphaeria poagena revealed only four base pair differences. Unfortunately, no protein-coding sequence data from the type strain are available. Instead of introducing a new species, we propose considering the host similarities (Poaceae) and ITS sequences to link these asexual and sexual forms as a single species. Therefore, we revised the species description to update the characteristics of its sexual form. This finding represents a new record of this fungus from bamboo and provides protein sequence data (tef1 and rpb2) for the species. Please refer to the notes under Phaeosphaeria chengduensis for further details.
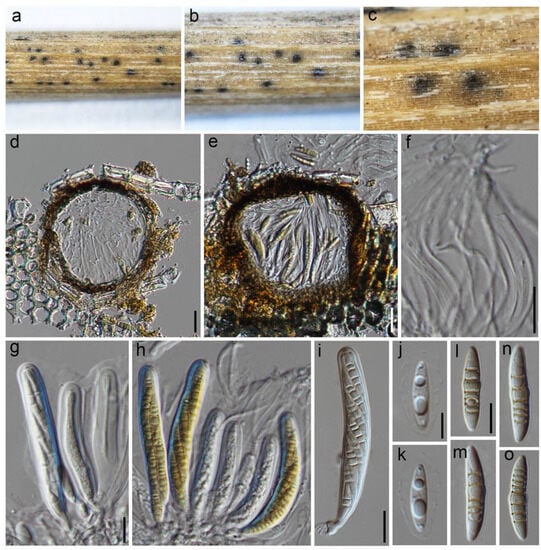
Phaeosphaeria sichuanensis Wanas. & Maharachch. sp. nov. (Figure 5).
Figure 5.
Phaeosphaeria sichuanensis (HKAS 129194, holotype): (a,b) conidiomata on the dead Pandanaceae leaves (arrowed); (c) horizontal section of conidiomata; (d) vertical section through a conidioma; (e,f) pycnidial wall showing immature and mature conidia attached to conidiogenous cells; (g,h) conidia; (i) germinated conidium; (j,k) colonies on PDA after 6 weeks. Scale bars: (d) 50 µm; (e–i) 10 µm.
MycoBank: MB 849353.
Etymology: The specific epithet is derived from Sichuan, the location where this fungus was collected.
Holotype: HKAS 129194.
It is saprobic on dead Pandanaceae leaves. The sexual state was not observed. The asexual state: conidiomata are 140–170 μm high, 150–200 μm in diam., pycnidial, scattered and immersed, and appearing as slightly raised small black dots on the surface of the host. They are uniloculate and can be globose to subglobose or irregular in shape, with a color ranging from brown to dark brown. The pycnidial wall is 10–15 μm wide and consists of two layers of brown cells. The outer margin is composed of cells arranged in a textura angularis pattern, while the inner layer is made up of hyaline to pale brown cells arranged in a textura angularis pattern. The conidiophores are reduced to conidiogenous cells, which are holoblastic, phialidic, and ampulliform in shape and measure 3–6 × 3.5–5.5 μm (M = 4.6 × 4.4 μm, n = 20). They can be either hyaline or pale brown. The conidia measure 7–10 × 3.5–5 μm (M = 8.4 × 4 μm, n = 30) and are ovoid to ellipsoid in shape, with a conically rounded apex and base. Initially, the conidia are hyaline and aseptate with guttules, but they become pale brown to brown and 1-2 septate as they mature. Occasionally, they may contain guttules. The conidia are not constricted at the septa and have a smooth surface.
Culture characteristics: Colonies on PDA reach a diameter of 2 cm after four weeks at 25 °C. Initially, the colonies are white but become creamy white when mature. They are dense in texture, and slight radiation can be observed. The reverse side of the colonies appears creamy orange at the edges, gradually becoming orange-brown to brown at the center.
Known distribution: China (Sichuan) on dead Pandanaceae leaves in terrestrial habitats.
Material examined: China, Sichuan, Chengdu, Qingyang, Chengdu Huanhuaxi Park, 30.662011 N, 104.026047 E, 511 m, on dead twigs of an unknown deciduous host, 4 January 2023, D.N. Wanasinghe, SCHHX23-021-4 (HKAS 129194, holotype), ex-type culture, KUNCC 23-13569. ibid. 30.661886 N, 104.025530 E, 506 m, 4 January 2023, SCHHX23-021-3 (HKAS 129193), living culture, KUNCC 23-13568.
Notes: In our phylogenetic analysis, two strains (KUNCC 23-13568 and KUNCC 23-13569) from an asexual morphic fungus were found to be grouped with Phaeosphaeria species. Specifically, they formed the basal lineage of a monophyletic sister clade that included Phaeosphaeria ampeli (MFLUCC 19-0150 and MFLUCC 18-1641), P. chengduensis (KUNCC 23-13570 and KUNCC 23-13571), P. chinensis (KUMCC 19-0161 and MFLUCC 19-0217), P. poagena (KUNCC 23-13572 and KUNCC 23-13573), and P. sinensis (NCYUCC 19-0369). Morphologically, the conidia of the new fungus differ from other Phaeosphaeria species in terms of shape and the number of septa. Typically, Phaeosphaeria species produce fusiform conidia with multiple vertical septa [32,33]. However, this new fungus has ovoid to ellipsoid conidia with only 1–2 vertical septa. Nevertheless, the ampulliform, holoblastic, and conidiogenous cells of the new species bear resemblance to the asexual morphs in Phaeosphaeria.
Septoriella Oudem., Ned. Kruidk. Arch. ser. 2, 5 (3): 504 (1889).
=Allophaeosphaeria Ariyaw., Camporesi & K.D. Hyde, Fungal Diversity 72: 137 (2015).
=Dactylidina Wanas., Camporesi & K.D. Hyde, Fungal Diversity 89: 107 (2018).
=Hydeopsis J.F. Zhang, J.K. Liu & Z.Y. Liu, Mycosphere 8: 211 (2019) syn. nov.
=Naemostroma Höhn., Berichte der Deutschen Botanischen Gesellschaft 37: 114 (1919).
=Phaeopoacea Thambug., Dissan. & K.D. Hyde, Mycosphere 8: 752 (2017) syn. nov.
=Poaceicola W.J. Li, Camporesi, Bhat & K.D. Hyde, Mycosphere 6 (6): 696 (2015).
=Vagicola Chethana & K.D. Hyde, Fungal Diversity 75: 113 (2015).
Notes: Septoriella was initially described by Oudemans [38], but it received limited attention, and only a few species were recognized. However, a recent study by Crous et al. [39] redefined the genus and established an epitype (CBS H-22281) and an ex-epitype strain (CBS 140065) for the type species, Septoriella phragmitis. The genus exhibits a wide distribution and encompasses a diverse array of species. Septoriella species are commonly associated with various host plants, including grasses, cereals, and other herbaceous plants [40]. Over the years, the taxonomy and classification of Septoriella have undergone significant revisions, with the discovery of new species and re-evaluation of existing ones using molecular and morphological data [40,41,42,43]. In the study of Marin-Felix [40], the ex-type strains of the sexual genera Allophaeosphaeria, Poaceicola, and Vagicola were found within the clade representing the genus Septoriella. Consequently, these genera were synonymized with Septoriella in their research. Subsequent to the research conducted by Crous et al. [39], later studies have contributed to the addition of 29 recognized species to the genus Septoriella [2]. However, it is crucial to note that this number is subject to change, as new species are continually being discovered and identified. Taxonomic studies have utilized various approaches, including molecular phylogenetics, morphology, and host specificity, to delineate and classify different species within Septoriella. In this study, we propose the synonymization of Amarenographium ammophilicola, Amarenomyces dactylidis, Dactylidina shoemakeri, Hydeopsis verrucispora, Loratospora luzulae, Phaeopoacea asparagicola, P. festucae, and P. muriformis under Septoriella.
Septoriella ammophilicola (Dayar., E.B.G. Jones & K.D. Hyde) Wanas. & Maharachch., comb. nov.
MycoBank: MB 849361.
≡Amarenographium ammophilicola Dayar., E.B.G. Jones & K.D. Hyde, in Dayarathne et al., Mycosphere 11(1): 59 (2020).
Holotype: UK, Wales, Carmarthenshire, Cefn Sidan, on a leaf of Marram grass (Ammophila arenaria) in a sand dune, 15 October 2017, E.B. Gareth Jones, GJ448 (MFLU 17-2571, holotype).
Descriptions and illustrations. See Dayarathne et al. [44].
Notes: Dayarathne et al. [44] introduced Amarenographium ammophilicola based on its morphological characteristics and phylogenetic analysis. In our phylogenetic analysis, the type strain of A. ammophilicola clustered with other Septoriella species within the Phaeosphaeriaceae (Figure 1). Therefore, we propose the transfer of Amarenographium ammophilicola to the genus Septoriella, and subsequently it should be recognized as Septoriella ammophilicola, comb. nov.
Septoriella asparagicola (Phukhams., Akulov & K.D. Hyde) Wanas. & Maharachch., comb. nov.
MycoBank: MB 849362.
≡Phaeopoacea asparagicola Phukhams., Akulov & K.D. Hyde, in Hyde et al., Fungal Diversity 96: 57 (2019).
Holotype: Ukraine, Odessa Region, Lyman District, Tiligulskyt Regional Landscape Park, on the overwintered stems of Asparagus sp., 1 May 2014, A. Akulov, EX CWU (MYC) AS 5825 (MFLU 18-1380, holotype), ex-type MFLUCC 16-0379.
Descriptions and illustrations: See Hyde et al. [19].
Notes: Thambugala et al. [42] established the genus Phaeopoacea to accommodate P. festucae and P. phragmiticola (≡Phaeosphaeria phragmiticola). The type species, P. festucae, was previously known only from its asexual morph, which forms pycnidial conidiomata that are globose to subglobose or linear in rows on the host, producing brown to dark brown, oblong conidia [42]. Meanwhile, Phaeosphaeria phragmiticola was previously considered a synonym of Septoriella leuchtmannii by Crous et al. [39]. However, based on multi-gene phylogenetic analyses conducted by Li et al. [41] and Thambugala et al. [42], Phaeosphaeria phragmiticola was placed within Phaeopoacea. Hyde et al. [19,45] later included two additional species, P. asparagicola and P. muriformis, in this genus. The phylogenetic analysis by various authors revealed that Phaeopoacea does not form a clearly defined clade and consistently clusters with the genera Allophaeosphaeria, Amarenographium, Amarenomyces, Dactylidina, Poaceicola, Septoriella, and Vagicola [35]. In our phylogenetic analysis, the three type strains of Phaeopoacea, P. asparagicola (MFLUCC 16-0379), P. festucae (MFLUCC 17-0056), and P. muriformis (MFLUCC 17-0372) were found to be distantly related to each other within Septoriella. To provide better clarity and avoid unresolved taxonomic circumscriptions, we propose the synonymization of these three species under Septoriella. As a result, we established a new combination by classifying Phaeopoacea asparagicola within Septoriella.
Septoriella festucae (Dissan. & K.D. Hyde) Wanas. & Maharachch., comb. nov.
MycoBank: MB 849363.
≡Phaeopoacea festucae Dissan. & K.D. Hyde, in Thambugala et al., Mycosphere 8(4): 752 (2017).
Holotype: Italy, Province of Forlì-Cesena, near Santa Sofia, on a dead aerial stem of Festuca pratensis, 16 July 2013, Erio Camporesi IT 1384 (MFLU 17– 0121), ex-type MFLUCC 17–0056.
Descriptions and illustrations: See Thambugala et al. [42].
Notes: Our study findings have led us to propose the reclassification of Phaeopoacea into the genus Septoriella, resulting in the establishment of Septoriella festucae, comb. nov. For more information, please refer to the note provided under Septoriella asparagicola.
Septoriella luzulae (Jayasiri, Camporesi & K.D. Hyde) Wanas. & Maharachch., comb. nov.
MycoBank: MB 849364.
≡Loratospora luzulae Jayasiri, Camporesi & K.D. Hyde, in Ariyawansa et al., 75: 108 (2015).
Holotype: Italy, Province of Forlì-Cesena, Campigna-Santa Sofia, on dead stems of Luzula nivea, 8 June 2014, E. Camporesi, IT 1918 (MFLU 15-1394, holotype), ex-type MFLUCC 14-0826.
Descriptions and illustrations: See Ariyawansa et al. [46].
Notes: The monotypic genus Loratospora was established by Kohlmeyer and Volkmann-Kohlmeyer [47] to accommodate L. aestuarii. Later, Ariyawansa et al. [46] introduced Loratospora luzulae as the second species in this genus. Although their phylogenetic analysis showed that these two species were closely related, they were not monophyletic (page 98, Figure 40). In this study, the type strain of Loratospora luzulae (MFLUCC 14-0826) grouped with Septoriella species, particularly showing a sister relationship to Septoriella callistemonis (CBS 146822) and S. camporesii (KUMCC 16-0113). Therefore, we have assigned Loratospora luzulae to the Septoriella genus as a new combination (S. luzulae).
Septoriellaneomuriformis Wanas. & Maharachch., nom. nov.
MycoBank: MB 849365.
≡Phaeopoacea muriformis Karun. & K.D. Hyde, in Hyde et al., Fungal Diversity 87: 83 (2017).
Holotype: China, Yunnan Province, Kunming Institute of Botany, Botanical Garden, on stems of unidentified grass, 28 November 2016, K.V.A. Karunarathna, AKKIB 49 (MFLU 17-0372, holotype; HKAS 97365, isotype), ex-type living cultures MFLUCC 17-1382, KUMCC 16-0234.
Descriptions and illustrations: See Hyde et al. [45].
Notes: Our study findings have led us to propose the reclassification of Phaeopoacea into the genus Septoriella, resulting in the establishment of Septoriella muriformiae, nom. nov. Since the name ‘Septoriella muriformis’ is already in use within the Septoriella genus, we have employed a nomenclature novelty (Septoriella neomuriforme) for this synonymization. For more information, please refer to the note provided under Septoriella asparagicola.
Septoriella paradactylidis Wanas. & Maharachch., nom. nov. (Figure 6).
Figure 6.
Septoriella paradactylidis (HKAS 129216): (a–c) ascomata on the host; (d,e) cross section of ascomata; (f) pseudoparaphyses; (g–i) asci; (j–o) ascospores. Scale bars: (d,f,g,i) 20 µm; (j,l) 10 µm, scale bar of (d) applies to (e), scale bar of (g) applies to (g–i), scale bar of (j) applies to (k), and scale bar of (l) applies to (l–o).
MycoBank: MB 849366.
≡Amarenomyces dactylidis Mapook, Camporesi & K.D. Hyde, in Hyde et al., Fungal Diversity 87: 78 (2017).
It is saprobic on decaying clumps of Lolium perenne. The sexual morph: Ascomata are 120–190 μm high and 130–200 μm diam., are semi-immersed to erumpent, solitary, scattered, uniloculate, and have a globose to subglobose shape. They are dark brown in color, with a central ostiole and a minute papilla. The peridium is 13–25 μm wide and consists of 3–6 layers of brown to dark brown cells with a textura angularis structure. The hamathecium is made up of filamentous, cylindrical to filiform, septate pseudoparaphyses that are 2–4 μm wide and are embedded in a gelatinous matrix. The asci measure (70–)75–90(–110) × (9–)10–12(–15) μm (x = 83 × 9.5 μm, n = 10), with a developed ocular chamber. They are eight-spored, bitunicate, fissitunicate, cylindrical, and short pedicellate. The ascospores measure 20–27 × 5–6 μm (x = 24.5 × 5.5 μm, n = 40) and are broadly fusiform to inequilateral in shape. They are hyaline to pale yellowish, 5–8 septate, with the widest part at the middle cell. The ascospores are asymmetrical and have a smooth wall, surrounded by a hyaline gelatinous sheath. The asexual state was not observed.
Culture characteristics: The ascospores showed germination on PDA within 24 h. After a two-week incubation period at 25 °C, the colonies on PDA medium grew to a diameter of 4 cm. These colonies had an undulate margin and initially appeared whitish grey, transitioning to a grey shade with an irregularly raised texture at the center. Towards the periphery, the color became greenish grey. When observed from the reverse side, the colonies displayed a blackish brown color at the center and a creamy grey coloration towards the periphery.
Known distribution: China, Italy, on Dactylis glomerata, Lolium perenne, and unknown fern.
Material examined: China, Sichuan, Chengdu, Chenghua District, The Chengdu Research Base of Giant Panda Breeding, on dead culms of Lolium perenne L., 13 August 2021, Q Wang, W90-3 (HKAS 129216), culture, KUNCC 23-14219.
Notes: Our phylogenetic analyses have revealed that the new strain KUNCC 23-14219 is closely related to the other isolates of Septoriella paradactylidis (=Amarenomyces dactylidis), including the type strain (MFLU 17-0498). Further investigations comparing our isolate to the type species have revealed a similar size range of the ascomata, asci, and ascospores, as well as the ascospore septation [45]. Therefore, we are now reporting the presence of S. neodactylidis in Lolium perenne, which represents the first record of this fungus in that host and provides protein sequence data (tef1 and rpb2) for this species. Since the name ‘Septoriella dactylidis’ is already in use within the Septoriella genus, we have utilized a nomenclature novelty (Septoriella paradactylidis) for this synonymization.
Septoriella shoemakeri Wanas. & Maharachch., sp. nov.
MycoBank: MB 849358.
≡Dactylidina shoemakeri Wanas., Camporesi, E.B.G. Jones & K.D. Hyde, in Wanasinghe et al., Fungal Diversity 89: 109 (2018).
Holotype: Italy, Trento, Marilleva 1400, on dead aerial stems of Poa sp., 2 August 2013, Erio Camporesi IT 1932 (MFLU 16-0202, holotype); ex-type MFLUCC 14-0963.
Descriptions and illustrations: See Wanasinghe et al. [48].
Notes: In Wanasinghe et al. [48], they described Dactylidina shoemakeri based on its morphological traits and a conducted phylogenetic analysis. However, it should be noted that this species has not been validly published (Nom. inval., Art. 35.1 (Shenzhen)). In our phylogenetic analysis, we found that the strain MFLUCC 14-0963, which was previously invalidly published as Dactylidina shoemakeri, grouped with Septoriella species and showed a close affiliation to S. neodactylidis (MFLUCC 14-0966). Therefore, we propose reassigning Dactylidina shoemakeri to the genus Septoriella, and going forward, it should be referred to as Septoriella shoemakeri.
Septoriella verrucispora (J.F. Zhang, J.K. Liu & Z.Y. Liu) Wanas. & Maharachch., comb. nov.
MycoBank: MB 849368.
≡Hydeopsis verrucispora J.F. Zhang, J.K. Liu & Z.Y. Liu, in Zhang et al., Mycosphere 8(1): 211 (2019).
Holotype: China, Guizhou Province, Guiyang City, Huaxi District, dead culms of herbaceous plant, 3 April 2016, J.F. Zhang, SD-2016-5 (MFLU 18-2269; holotype); ex-type MFLUCC 19-0163, GZCC 19-0001.
Descriptions and illustrations: See Zhang et al. [49].
Notes: The monotypic genus Hydeopsis, typified by H. verrucispora, was introduced by Zhang et al. [49]. It exhibited a close phylogenetic relationship with Dactylidina and Phaeopoacea within the family Phaeosphaeriaceae. The authors differentiated Hydeopsis from these genera based on differences in pigmentation and the number of septa in ascospores. Our combined sequence phylogenetic analysis reveals that Hydeopsis verrucispora is phylogenetically positioned within the genus Septoriella. Additionally, pigmentation and the number of septa appear to be highly informative at the species level, although these characteristics alone cannot reliably distinguish between genera. Species of Septoriella also share more similar characteristics. Therefore, Hydeopsis is determined to be congeneric with Septoriella, and as a result, we propose assigning it to the genus Septoriella as a new combination.
4. Discussion
The delimitation of species in Phaeosphaeriaceae is typically based on a combination of morphological and molecular characteristics. Some of the major morphological features used to delimitate species in this family include fruiting body type, peridium, spores, asci, conidiogenous cells, and colony morphology [4]. The shape, size, and color of ascospores and conidia can vary between different species of Phaeosphaeriaceae. However, the usefulness of these morphological features for species delimitation can vary depending on the species and the taxonomic group being studied. There are several challenges in using morphological features for species delimitation in Ascomycota, including in Phaeosphaeriaceae. Morphological variation within species of Phaeosphaeriaceae can pose challenges in distinguishing closely related species. For instance, in the case of Septoriella species, there are variations in ascospore features [40]. These include the arrangement of 1–3 seriate in an ascus, colors ranging from hyaline to yellowish-brown, pale brown, brown, golden brown, or reddish-brown, and shapes that can be narrowly or broadly fusiform, oblong, or narrowly oblong. The surface can be smooth-walled or echinulate, with transverse or longitudinal septa and sometimes enlarged medium cells. The ends of the ascospores can be conical, obtuse, or rounded, and they may or may not have sheaths. Furthermore, even the conidia morphology of the asexual morphs within these species can vary [39], with shapes ranging from cylindrical to subcylindrical, fusiform, or subfusiform. The apex can be obtuse or subobtuse, the base truncate, and the conidia can be straight or curved. They are euseptate, pale brown to brown, and can have thin-walled, smooth surfaces or minutely verruculose surfaces. Additionally, they may bear mucoid appendages at both ends.
Cryptic or polyphyletic species within Phaeosphaeriaceae are morphologically similar but genetically distinct, making it challenging to differentiate them based solely on morphology. This can be observed in sexual morphs resembling Phaeosphaeria or Ophiobolus, as well as asexual morphs with a camarosporium-like resemblance (i.e., Melnikia, Camarosporioides, and Dlhawksworthia). Convergence, where different species develop similar traits due to similar ecological pressures, can contribute to this morphological similarity. Moreover, variations in methods used to observe and measure morphological features can lead to inconsistencies in species delineation. Careful examination of old literature is crucial to avoid mistakenly introducing existing species as new ones. Additionally, limited information on the morphological features of certain Phaeosphaeriaceae species in old-type materials makes accurate delimitation based on morphology alone difficult. To overcome these challenges, a combination of morphological and molecular approaches is essential for species delineation in Phaeosphaeriaceae. Molecular data can provide valuable insights into genetic variation and evolutionary relationships, aiding in the resolution of taxonomic uncertainties and differentiation between closely related species with similar morphology.
The taxonomy of the Phaeosphaeria genus is continuously evolving, with ongoing research and the utilization of molecular techniques aiding in the identification and classification of new species. Molecular phylogenetic analyses, in conjunction with morphological studies, have contributed to the clarification of species boundaries and relationships within the genus. The diversity of Phaeosphaeria is evident through the wide range of hosts, substrates, and ecological roles displayed by its various species. Recent phylogenetic analyses have revealed that Phaeosphaeria is polyphyletic, leading to the reclassification of many Phaeosphaeria sensu lato species into different genera within Phaeosphaeriaceae [37,40,43,45,46,50,51].
Based on our multi-gene phylogenetic analysis, strains of Phaeosphaeria were found to be distributed across several subclades within Phaeosphaeriaceae. Among all the Phaeosphaeria strains examined, only P. orae-maris (CBS 255.64: MH858433 and MH870063) displayed a distant relationship from the family Phaeosphaeriaceae in our primary analyses. Instead, it exhibited a close phylogenetic relationship to Lentitheciaceae. For our phylogenetic analysis, we included all the Phaeosphaeria strains that formed a monophyletic group with the type strain (Phaeosphaeria oryzae). Additionally, we incorporated other Phaeosphaeria strains that were closely associated with Paraloratospora and Septoriella. However, the remaining strains that showed close affiliations with other genera, namely Phaeosphaeria anchiala (5552A and 5547D), P. avenaria (AFTOL-ID 280), P. breonadiae (CPC 25944), P. eustoma (AFTOL-ID 1570), P. fructigena (FMR 17808), P. fusispora (LC6215 and LC5367), P. halima (RKDO787 and RKDO844), P. juncicola (CBS 110108), P. nardi (CBS 304.71), P. nigrans (CBS 307.79 and CBS 576.86), P. occulta (CBS 582.86), P. olivacea (AFTOL-ID 2206 and JK 5540Q), P. orae-maris (AFTOL-ID 1441), P. spartinae (CBS 254.64, RKDO785, RKDO806, and RKDO808), P. spartinicola (CBS 176.91, CBS 118215, and JK 5177A), and P. typharum (CBS 296.54), were excluded from the final analysis. These decisions were made to ensure the accuracy and validity of our phylogenetic analysis, focusing specifically on the relationships within the genera Paraloratospora, Phaeosphaeria, and Septoriella.
Strains belong to eighteen existing Phaeosphaeria species, namely P. acaciae (KUMCC 20-0214 and MFLU 17-0496), P. ampeli (MFLUCC 18-1641 and MFLUCC 19-0150), P. calamicola (MFLUCC 14-1168), P. caricis-sectae (CBS 146823), P. chiangraina (MFLUCC 13-0231), P. chinensis (KUMCC 19-0161 and MFLUCC 19-0217), P. cycadis (KUMCC 18-0161 and KUMCC 18-0162), P. lunariae (CPC 26679), P. musae (CBS 120026, MFLUCC 11-0133, MFLUCC 11-0151, and MFLUCC 17-2648), P. nodulispora (URM 7220), P. oryzae (CBS 110110 and MFLUCC 11-0170), P. papayae (CBS 135416 S528), P. penniseti (FU31020), P. phoenicicola (CPC 28711), P. poagena (CBS 136771), P. podocarpi (CBS 138903), P. sinensis (KUMCC 17-0195, MFLUCC 18-1552, and NCYUCC 19-0369), and P. thysanolaenicola (MFLUCC 10-0563), which are grouped in Phaeosphaeria sensu stricto (Figure 1). Additionally, two of our new strains (KUNCC 23-13572 and KUNCC 23-13573) were grouped with the type strain of Phaeosphaeria poagena (CBS 136771), and four new strains formed two distinct monophyletic lineages that we have introduced as two new species, namely Phaeosphaeria chengduensis and P. sichuanensis.
Based on our multi-gene phylogenetic analysis, we observed that Paraloratospora is closely related to Loratospora aestuarii (JK 5535B), Sulcispora pleurospora (CBS 460.84), S. supratumida (MFLUCC 14-0995), and Wingfieldomyces cyperi (CBS 141450), forming a sister relationship. However, it is important to note that these species were represented by single strains. Therefore, further evaluation of their inter-generic relationships should be conducted with additional species/collections and through DNA-based sequence data analyses. Currently, only three species are accepted in Paraloratospora, viz., P. camporesii, P. gahniae, and P. marina [52]. In our analysis, two type strains of Phaeosphaeria breonadiae (CPC 25944) and P. fructigena (FMR 17808) were grouped among these three Paraloratospora species. Additionally, two of our new strains (KUNCC 23-14218 and HKAS 129218) also clustered with Paraloratospora species. Consequently, we synonymized Phaeosphaeria breonadiae and P. fructigena under Paraloratospora, and introduced our new strains as belonging to a new species. Furthermore, strains provisionally named as Phaeosphaeria avenaria f. sp. tritici (CBS 289.52), P. caricicola (CBS 603.86), P. eustoma (CBS 307.71 and CBS 724.92), P. glyceriae-plicatae (CBS 101261), P. juncophila (CBS 575.86), P. norfolcia (CBS 593.86), and P. parvula (CBS 260.49 and CBS 605.86) were also grouped within the genus Paraloratospora. However, since these strains are not related to their type materials, we did not transfer these species to Paraloratospora in our analysis. Further studies are needed to resolve the phylogenetic classification of the aforementioned species.
In our analysis, we primarily utilized single-gene phylogenetic analyses but obtained a more robust topology through the combined gene analysis (SSU + LSU + ITS + tef1 + rpb2). For molecular comparisons, ITS sequences were available for 97.7% of the species in the family, while LSU sequences were available for 98.8%. However, the availability of sequence data for protein-coding genes, such as tef1 and rpb2, was limited in previous studies, with tef1 available for 54.6% of the species and rpb2 available for 32.7%. By incorporating more informative gene data and conducting extensive sampling worldwide, it is anticipated that most of the monotypic species will be transferred to a few genera within the family. One example of this is the synonymization of Hydeopsis under Septoriella in this study. Based on morphological, phylogenetic, and ecological evidence, we have introduced four new species, seven new combinations, and two new names in the genera Paraloratospora, Phaeosphaeria, and Septoriella. Despite the advancements in our understanding of these genera, several challenges still persist. Differentiating closely related species remains a significant obstacle, necessitating further investigation and the integration of multiple data sources. Moreover, the limited knowledge regarding the life cycles, host range, and geographic distribution of many of these species hinders our comprehension of their ecological roles. To overcome these challenges, future research should prioritize the utilization of advanced molecular techniques such as genomics, transcriptomics, and metagenomics. These approaches can provide insights into the genetic diversity, evolutionary history, and ecological functions of Paraloratospora, Phaeosphaeria, and Septoriella. By employing these cutting-edge methods, we can unravel the intricate complexities within these genera and gain a more comprehensive understanding of their significance in the ecosystem.
Author Contributions
Conceptualization, D.N.W. and S.S.N.M.; resources, S.S.N.M.; writing—original draft preparation, D.N.W.; writing—review and editing, S.S.N.M.; funding acquisition, S.S.N.M. All authors have read and agreed to the published version of the manuscript.
Funding
Support for this study was provided by the CAS President’s International Fellowship Initiative (number 2021FYB0005), the National Science Foundation of China (NSFC) under the project code 32150410362, the Postdoctoral Fund from Human Resources and Social Security Bureau of Yunnan Province, and the University of Electronic Science and Technology of China Talent Introduction and Cultivation Project (A1098531023601245).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated for this study can be found in the NCBI GenBank and MycoBank.
Acknowledgments
We acknowledge the Kunming Institute of Botany, Chinese Academy of Sciences, and the University of Electronic Science and Technology of China for providing the laboratories and instruments for molecular work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Phookamsak, R.; Wanasinghe, D.N.; Hongsanan, S.; Phukhamsakda, C.; Huang, S.K.; Tennakoon, D.S.; Norphanphoun, C.; Camporesi, E.; Bulgakov, T.S.; Promputtha, I.; et al. Towards a natural classification of Ophiobolus and ophiobolus-like taxa; introducing three novel genera Ophiobolopsis, Paraophiobolus and Pseudoophiobolus in Phaeosphaeriaceae (Pleosporales). Fungal Divers. 2017, 87, 299–339. [Google Scholar] [CrossRef]
- Index Fungorum. Available online: http://www.indexfungorum.org/Names/Names.asp (accessed on 28 June 2023).
- Wijayawardene, N.N.; Hyde, K.D.; Dai, D.Q.; Sánchez-García, M.; Goto, B.T.; Saxena, R.K.; Erdoğdu, M.; Selçuk, F.; Rajeshkumar, K.C.; Aptroot, A.; et al. Outline of Fungi and fungus-like taxa—2021. Mycosphere 2022, 13, 53–453. [Google Scholar] [CrossRef]
- Phookamsak, R.; Liu, J.-K.; McKenzie, E.H.C.; Manamgoda, D.S.; Ariyawansa, H.; Thambugala, K.M.; Dai, D.-Q.; Camporesi, E.; Chukeatirote, E.; Wijayawardene, N.N.; et al. Revision of Phaeosphaeriaceae. Fungal Divers. 2014, 68, 159–238. [Google Scholar] [CrossRef]
- Liu, J.-K.; Hyde, K.D.; Jeewon, R.; Phillips, A.; Maharachchikumbura, S.S.N.; Ryberg, M.; Liu, Z.-Y.; Zhao, Q. Ranking higher taxa using divergence times: A case study in Dothideomycetes. Fungal Divers. 2017, 84, 75–99. [Google Scholar] [CrossRef]
- El-Demerdash, A.; El-Demerdash, A. Chemical diversity and biological activities of Phaeosphaeria fungi genus: A systematic review. J. Fungi 2018, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Norouzi, H.; Sohrabi, M.; Yousefi, M.; Boustie, J. Tridepsides as potential bioactives: A review on their chemistry and the global distribution of their lichenic and non-lichenic natural sources. Front. Fungal Biol. 2023, 4, 1088966. [Google Scholar] [CrossRef]
- Ashrafi, S.; Wennrich, J.P.; Becker, Y.; Maciá-Vicente, J.G.; Brißke-Rode, A.; Daub, M.; Thünen, T.; Dababat, A.A.; Finckh, M.R.; Stadler, M. Polydomus karssenii gen nov. sp. nov. is a dark septate endophyte with a bifunctional lifestyle parasitising eggs of plant parasitic cyst nematodes (Heterodera spp.). IMA Fungus 2023, 14, 6. [Google Scholar]
- Wanasinghe, D.N.; Mortimer, P.E.; Xu, J. Insight into the systematics of microfungi colonizing dead woody twigs of Dodonaea viscosa in Honghe (China). J. Fungi 2021, 7, 180. [Google Scholar] [CrossRef]
- MycoBank. Available online: http://www.mycobank.org (accessed on 28 June 2023).
- Wanasinghe, D.N.; Wijayawardene, N.N.; Xu, J.C.; Cheewangkoon, R.; Mortimer, P.E. Taxonomic novelties in Magnolia-associated pleosporalean fungi in the Kunming Botanical Gardens (Yunnan, China). PLoS ONE 2020, 15, e0235855. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.J.W.T.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. Appl. 1990, 18, 315–322. [Google Scholar]
- Rehner, S.A.; Samuels, G.J. Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol. Res. 1994, 98, 625–634. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [PubMed]
- Rehner, S.A.; Buckley, E.A. Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 2005, 97, 84–98. [Google Scholar] [CrossRef]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among ascomycetes evidence from an RNA polymerase II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Sung, G.H.; Sung, J.M.; Hywel-Jones, N.L.; Spatafora, J.W. A multi-gene phylogeny of Clavicipitaceae (Ascomycota, Fungi): Identification of localized incongruence using a combinational bootstrap approach. Mol. Phylogenet. Evol. 2007, 44, 1204–1223. [Google Scholar] [CrossRef]
- Wanasinghe, D.N.; Mortimer, P.E. Taxonomic and phylogenetic insights into novel Ascomycota from forest woody Litter. Biology 2022, 11, 889. [Google Scholar] [CrossRef]
- Hyde, K.D.; Tennakoon, D.S.; Jeewon, R.; Bhat, D.J.; Maharachchikumbura, S.S.N.; Rossi, W.; Leonardi, M.; Lee, H.B.; Mun, H.Y.; Houbraken, J.; et al. Fungal diversity notes 1036–1150: Taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers. 2019, 96, 1–242. [Google Scholar] [CrossRef]
- Tennakoon, D.S.; Thambugala, K.M.; Wanasinghe, D.N.; Gentekaki, E.; Promputtha, I.; Kuo, C.H.; Hyde, K.D. Additions to Phaeosphaeriaceae (Pleosporales): Elongaticollum gen. nov., Ophiosphaerella taiwanensis sp. nov., Phaeosphaeriopsis beaucarneae sp. nov. and a new host record of Neosetophoma poaceicola from Musaceae. MycoKeys 2020, 70, 59–88. [Google Scholar] [CrossRef]
- Devadatha, B.; Mehta, N.; Wanasinghe, D.N.; Baghela, A.; Sarma, V.V. Vittaliana mangrovei, gen. nov., sp. nov. (Phaeosphaeriaceae) from mangroves near Pondicherry (India), based on morphology and multigene phylogeny. Cryptogam. Mycol. 2019, 40, 117–132. [Google Scholar] [CrossRef]
- Maharachchikumbura, S.S.N.; Ariyawansa, H.A.; Wanasinghe, D.N.; Dayarathne, M.C.; Al-Saady, N.A.; Al-Sadi, A.M. Phylogenetic classification and generic delineation of Hydeomyces desertipleosporoides gen. et sp. nov., (Phaeosphaeriaceae) from Jebel Akhdar Mountain in Oman. Phytotaxa 2019, 391, 28–38. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Proceedings of the 2010 Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; pp. 1–8. [Google Scholar]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Phylogenies and the comparative method. Am. Nat. 1985, 125, 1–15. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef]
- Rambaut, A. FigTree Version 1.4.0. Available online: http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 15 June 2023).
- Crous, P.W.; Wingfield, M.J.; Richardson, D.M. Fungal Planet description sheets: 400–468. Persoonia 2016, 36, 316–458. [Google Scholar] [CrossRef]
- De Gruyter, J.; Aveskamp, M.M.; Woudenberg, J.H.; Verkley, G.J.; Groenewald, J.Z.; Crous, P.W. Molecular phylogeny of Phoma and allied anamorph genera: Towards a reclassification of the Phoma complex. Mycol. Res. 2009, 113, 508–519. [Google Scholar] [CrossRef]
- Leuchtmann, A. Über Phaeosphaeria Miyake und andere bitunicate Ascomyceten mit mehrfach querseptierten Ascosporen. Ph.D. Thesis, ETH Zurich, Zurich, Switzerland, 1984. [Google Scholar]
- Shoemaker, R.A.; Babcock, C.E. Phaeosphaeria. Can. J. Bot. 1989, 67, 1500–1599. [Google Scholar] [CrossRef]
- Magaña-Dueñas, V.; Cano-Lira, J.F.; Stchigel, A.M. New Dothideomycetes from freshwater habitats in Spain. J. Fungi 2021, 7, 1102. [Google Scholar] [CrossRef]
- Hongsanan, S.; Hyde, K.D.; Phookamsak, R.; Wanasinghe, D.N.; McKenzie, E.H.C.; Sarma, V.V.; Boonmee, S.; Lücking, R.; Bhat, D.J.; Liu, N.G. Refined families of Dothideomycetes: Dothideomycetidae and Pleosporomycetidae. Mycosphere 2020, 11, 1533–2107. [Google Scholar] [CrossRef]
- Crous, P.W.; Shivas, R.G.; Quaedvlieg, W.; van der Bank, M.; Zhang, Y.; Summerell, B.A.; Guarro, J.; Wingfield, M.J.; Wood, A.R.; Alfenas, A.C.; et al. Fungal Planet description sheets: 214–280. Persoonia 2014, 32, 184–306. [Google Scholar] [CrossRef] [PubMed]
- Quaedvlieg, W.; Verkley, G.; Shin, H.-D.; Barreto, R.; Alfenas, A.; Swart, W.; Groenewald, J.; Crous, P. Sizing up Septoria. Stud. Mycol. 2013, 75, 307–390. [Google Scholar] [CrossRef] [PubMed]
- Oudemans, C.A.J.A. Contributions à la flore mycologique des Pays-Bas. XIII. Ned. Kruidkd. Arch. Ser. 2 1889, 5, 454–519. [Google Scholar]
- Crous, P.W.; Carris, L.M.; Giraldo, A.; Groenewald, J.Z.; Hawksworth, D.L.; Hemández-Restrepo, M.; Jaklitsch, W.M.; Lebrun, M.H.; Schumacher, R.K.; Stielow, J.B.; et al. The Genera of Fungi-fixing the application of the type species of generic names-G2: Allantophomopsis, Latorua, Macrodiplodiopsis, Macrohilum, Milospium, Protostegia, Pyricularia, Robillarda, Rotula, Septoriella, Torula, and Wojnowicia. IMA Fungus 2015, 6, 163–198. [Google Scholar] [CrossRef] [PubMed]
- Marin-Felix, Y.; Hernández-Restrepo, M.; Iturrieta-González, I.; García, D.; Gené, J.; Groenewald, J.Z.; Cai, L.; Chen, Q.; Quaedvlieg, W.; Schumacher, R.K.; et al. Genera of phytopathogenic fungi: GOPHY 3. Stud. Mycol. 2019, 94, 1–124. [Google Scholar] [CrossRef]
- Li, W.J.; Bhat, D.J.; Camporesi, E.; Tian, Q.; Wijayawardene, N.N.; Dai, D.Q.; Phookamsak, R.; Chomnunti, P.; Bahkali, A.H.; Hyde, K.D. New asexual morph taxa in Phaeosphaeriaceae. Mycosphere 2015, 6, 681–708. [Google Scholar] [CrossRef]
- Thambugala, K.M.; Wanasinghe, D.N.; Phillips, A.J.L.; Camporesi, E.; Bulgakov, T.S.; Phukhamsakda, C.; Ariyawansa, H.A.; Goonasekara, I.D.; Phookamsak, R.; Dissanayake, A.; et al. Mycosphere notes 1–50: Grass (Poaceae) inhabiting Dothideomycetes. Mycosphere 2017, 8, 697–796. [Google Scholar] [CrossRef]
- Hyde, K.D.; Dong, Y.; Phookamsak, R.; Jeewon, R.; Bhat, D.J.; Jones, E.B.G.; Liu, N.G.; Abeywickrama, P.D.; Mapook, A.; Wei, D.P.; et al. Fungal diversity notes 1151–1276: Taxonomic and phylogenetic contributions on genera and species of fungal taxa. Fungal Divers. 2020, 100, 5–277. [Google Scholar] [CrossRef]
- Dayarathne, M.C.; Jones, E.B.G.; Maharachchikumbura, S.S.N.; Devadatha, B.; Sarma, V.V.; Khongphinitbunjong, K.; Chomnunti, P.; Hyde, K.D. Morpho-molecular characterization of microfungi associated with marine based habitats. Mycosphere 2020, 11, 1–188. [Google Scholar] [CrossRef]
- Hyde, K.D.; Norphanphoun, C.; Abreu, V.P.; Bazzicalupo, A.; Chethana, K.W.T.; Clericuzio, M.; Dayarathne, M.C.; Dissanayake, A.J.; Ekanayaka, A.H.; He, M.-Q.; et al. Fungal diversity notes 603–708: Taxonomic and phylogenetic notes on genera and species. Fungal Divers. 2017, 87, 1–235. [Google Scholar] [CrossRef]
- Ariyawansa, H.A.; Hyde, K.D.; Jayasiri, S.C.; Buyck, B.; Chethana, K.W.T.; Dai, D.Q.; Dai, Y.C.; Daranagama, D.A.; Jayawardena, R.S.; Lücking, R.; et al. Fungal diversity notes 111–252—Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2015, 75, 27–274. [Google Scholar] [CrossRef]
- Kohlmeyer, J.; Volkmann-Kohlmeyer, B. Atrotorquata and Loratospora: New ascomycete genera on Juncus roemerianus. Syst. Ascomycetum 1993, 12, 7–22. [Google Scholar]
- Wanasinghe, D.N.; Phukhamsakda, C.; Hyde, K.D.; Jeewon, R.; Lee, H.B.; Jones, G.E.B.; Tibpromma, S.; Tennakoon, D.S.; Dissanayake, A.J.; Jayasiri, S.C.; et al. Fungal diversity notes 709–839: Taxonomic and phylogenetic contributions to fungal taxa with an emphasis on fungi on Rosaceae. Fungal Divers. 2018, 89, 1–236. [Google Scholar] [CrossRef]
- Zhang, J.F.; Liu, J.K.; Jeewon, R.; Wanasinghe, D.N.; Liu, Z.Y. Fungi from Asian Karst formations III. Molecular and morphological characterization reveal new taxa in Phaeosphaeriaceae. Mycosphere 2019, 10, 202–220. [Google Scholar] [CrossRef]
- Tanaka, K.; Hirayama, K.; Yonezawa, H.; Sato, G.; Toriyabe, A.; Kudo, H.; Hashimoto, A.; Matsumura, M.; Harada, Y.; Kurihara, Y.; et al. Revision of the Massarineae (Pleosporales, Dothideomycetes). Stud. Mycol. 2015, 82, 75–136. [Google Scholar] [CrossRef] [PubMed]
- Tennakoon, D.S.; Hyde, K.D.; Phookamsak, R.; Wanasinghe, D.N.; Camporesi, E.; Promputtha, I. Taxonomy and phylogeny of Juncaceicola gen. nov. (Phaeosphaeriaceae, Pleosporinae, Pleosporales). Cryptogam. Mycol. 2016, 37, 135–156. [Google Scholar] [CrossRef]
- Devadatha, B.; Jones, E.B.G.; Wanasinghe, D.N.; Bahkali, A.H.; Hyde, K.D. Characterization of novel estuarine Ascomycota based on taxonomic and phylogenetic evaluation. Bot. Mar. 2023, 66, 281–300. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).