Abstract
Evidence has shown the clear positive effects of nature-based products on biofilm control and improved gingival health. However, most studies have used in vitro models, have tested single natural components, or have not evaluated proteomic changes after treatment. This double-blind, parallel, randomized, and controlled clinical trial evaluated the benefits of a nature-based gel in controlling gingival inflammation and its effects on the proteomic gingival crevicular fluid (GCF) profile. Gingivitis patients were distributed into the following groups: (1) nature-based gel containing propolis, aloe vera, green tea, cranberry, and calendula (n = 10); (2) control—conventional toothpaste (n = 10). GCF was collected and evaluated by means of liquid chromatography coupled with tandem mass spectrometry (LC–MS/MS). At 3 months, the groups showed similar clinical benefits (p < 0.05). A total of 480 proteins were identified across all groups. In a pooled comparison of both groups at both time points, exclusive proteins were identified in the nature-based gel (78) and the control (21) groups. The exclusive proteins identified for the toothpaste mainly acted in wound healing, and those for the nature-based gel mainly acted on immune system processes. The nature-based gel achieved similar clinical outcomes to conventional toothpaste. However, the nature-based gel markedly changed the proteomic profile of GCF after treatment, showing a profile associated with a host response.
1. Introduction
Polymicrobial dysbiotic oral biofilms are the main etiologic factor triggering inflammatory chronic diseases on tooth-supporting tissues, known as periodontal diseases [1]. These prevalent conditions affect millions of people worldwide, showing increasing rates with age and negatively impacting people’s lives [2]. The biofilm’s biomass and its microbial composition act as chemical and physical “stress” factors on tooth-supporting tissues, triggering inflammatory responses and subsequent tissue damage [3]. This initial process is characterized by clinical gingival inflammation, known as gingivitis, which often progresses in severity and tissue damage [4]. Since it is a multifactorial condition, different risk, genetic, and systemic factors, as well as the intensity and duration of microbial challenges and host susceptibility, may promote the destruction of periodontal supporting tissue [5]. Oral hygiene procedures, such as toothbrushing and other mechanical cleansing methods, are effective in controlling biofilm accumulation and achieving gingival health [6]. Different antimicrobial agents and adjunctive therapies have been tested to control oral biofilm accumulation and reduce gingival inflammation [7], and the composition of toothpaste also plays an important role in achieving these outcomes [6].
Previous evidence suggests that specific chemical agents in toothpaste may present enhanced benefits in controlling gingival inflammation [6]. In fact, toothpaste composition may also affect local molecular parameters, such as microbial pathways at the metatranscriptomic level and the levels of pro-inflammatory components in gingival crevicular fluid (GCF) [8]. These new molecular and omics approaches provide new insights into the modulation of the abilities of new products and explain the effects of different agents on oral sites. However, although the proteomic profile is responsible for mediating molecular functions and biological outcomes, it has not been tested in new toothpastes, especially at the GCF level [9]. An understanding of the proteins involved in health and disease states provides significant potential in identifying new biomarkers, given the close relationship between molecular activity and biological/clinical outcomes [10].
In this sense, changes in toothpaste formulation should consider not only the clinical evaluation of gingival conditions and biofilm control but also the local molecular parameters that drive one toward a health-associated state. Incorporating new agents to enhance oral biofilm control and address its pathogenicity is a logical development in improving clinical outcomes [11]. Although traditional components have been used in most commercially available toothpastes, new formulations have been explored as agents, such as nature-based compounds. Thus, over the past decade, there has been increasing interest in nature-based products for oral health [12]. In the United States of America (USA), a study identified that 35% of the investigated population reported using some form of herbal medicine [13]. Furthermore, a high proportion of new drugs approved by the Food and Drug Administration (FDA) are natural or naturally derived products [14]. These products have shown high antimicrobial abilities, combined with additional benefits, such as anti-inflammatory and antioxidant properties [12]. Evidence shows the clear positive effects of nature-based products on biofilm control and improved gingival health, but most of this evidence comes from in vitro models [12]. Moreover, most studies have only tested products with single natural components [12], and combinations in a single product have not been widely explored, especially in terms of proteomic changes after treatments.
Therefore, this clinical trial aims to evaluate and compare the clinical benefits of a nature-based gel in controlling gingivitis and its effects on the proteomic GCF profile compared with a conventional toothpaste.
2. Results and Discussion
2.1. Clinical Outcomes
Natural products have shown promising antimicrobial effects against periopathogens, but most evidence has emerged from in vitro models [15]. These outcomes demonstrate the anti-inflammatory and antioxidant abilities of these agents, making them effective compounds in the manufacturing of nature-based products to control gingival inflammation. It is important to note that most of the clinical evidence has come from tests of nature-based products for oral biofilm control using mouthrinses as the vehicle [12]. This clinical study included 20 subjects (DESPLAC® group: 8 women and 2 men, 30–47 years of age; and Oral-B group: 7 women and 3 men, 31–48 years of age). There were no dropouts during the experimental period. All subjects who finished the study reported full adherence to the prescribed oral hygiene protocol. Throughout the study, no adverse effects on the soft or hard tissues of the oral cavity were detected by the examiner or reported by the subjects. Table 1 presents the mean (± SD) of full-mouth clinical parameters evaluated at baseline and 3 months after using the products. At the beginning, there were no clinical differences between the groups for any of the evaluated parameters (p > 0.05), demonstrating the homogeneity of the sample groups. At 3 months, no statistically significant differences were observed between the two groups (p > 0.05); however, a reduction in the gingival index was noted in both the nature-based gel and control toothpaste groups. Moreover, when evaluating the proportion of each score on the plaque index, there was a clear reduction trend after 3 months, with an approximately 20% increase in the number of sites/teeth categorized with a score of 0 on the plaque index (Table S1). Bleeding on probing, expressed as the percentage of sites bleeding, also did not show any differences between the groups or time points (Table S2). Therefore, the clinical findings showed similar outcomes for both tested products (nature-based gel and control toothpaste) after 3 months, with both being effective in reducing gingival inflammation.

Table 1.
Full-mouth clinical parameter evaluation of the study population at baseline and after 3 months. Average (± SD).
DESPLAC® gel (Premium Oral Gel) includes a combination of different natural agents in its formula: propolis, aloe vera, green tea, cranberry, and calendula. Although the individual antimicrobial, anti-inflammatory, and/or antioxidant benefits of these natural agents are well known, their combination in a single product has not been widely explored [16,17,18,19]. Previous in vitro studies have explored the antimicrobial ability of DESPLAC® in a multispecies biofilm model [16,17,18,19]. The results demonstrated its potential to reduce bacterial metabolic activity and the levels of two important periopathogens: Tannerella forsythia and Porphyromonas gingivalis [20,21]. In our clinical setting with gingivitis patients, the nature-based gel was clinically effective, controlling gingival inflammation over 3 months of use. However, the clinical outcomes of the two tested products were similar, with the nature-based gel showing no additional benefit compared with conventional toothpaste. In fact, a systematic review demonstrated that mechanically removing biofilm through toothbrushing is more important than the presence of toothpaste [22]. However, toothpaste may contain other important chemical agents crucial for oral health, such as fluoride, which is well known for its ability to control dental caries but is absent in the DESPLAC® product [23]. Importantly, clinical trials like the one conducted here, comparing different gel/toothpaste formulations for oral biofilm and gingival inflammation control, may be helpful in further developing clinical guidelines, particularly regarding the use of nature-based products for periodontal health and respecting patients’ preferences and characteristics [12]. Moreover, although a 3-month follow-up period is commonly used in clinical trials to identify significant changes in gingival health recovery post-treatment, aligning with the gingival healing timeline and the recommended interval for maintenance visits, longer periods should be considered in future studies to evaluate the maintenance of outcomes [24,25]. Furthermore, the reduction in gingival inflammation was not accompanied by a reduced plaque index after 3 months. However, the plaque index was expressed as an average of scores, and when evaluating the proportion of each score, there was a clear improvement in plaque control, with an increased proportion of sites scoring 0 after 3 months (Supplementary Materials). The lack of differences in the average values for this outcome may be attributable to the sample size, necessitating further investigation.
2.2. Composition of Toothpaste Modulated the Proteomic Profile in GFC
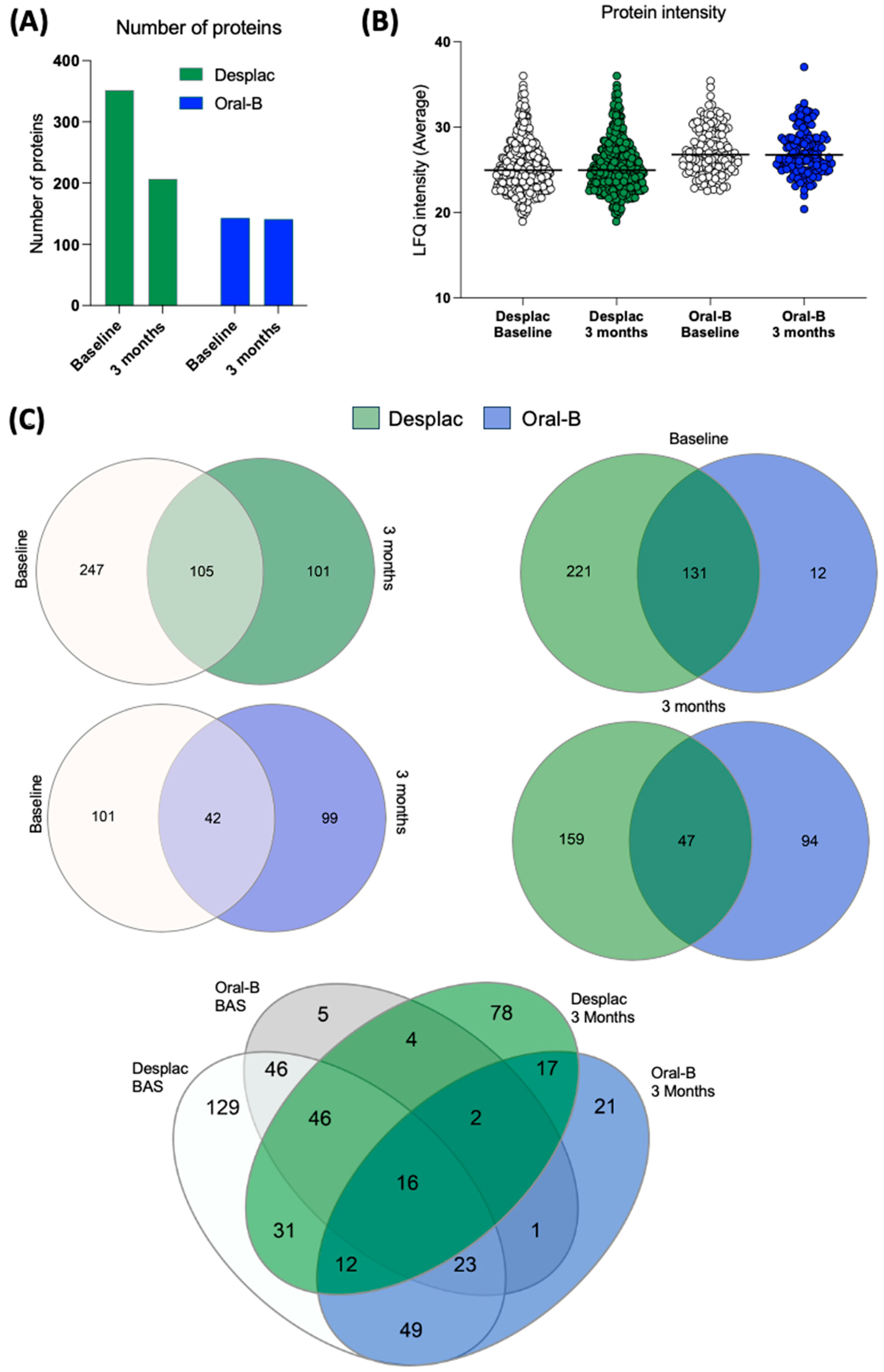
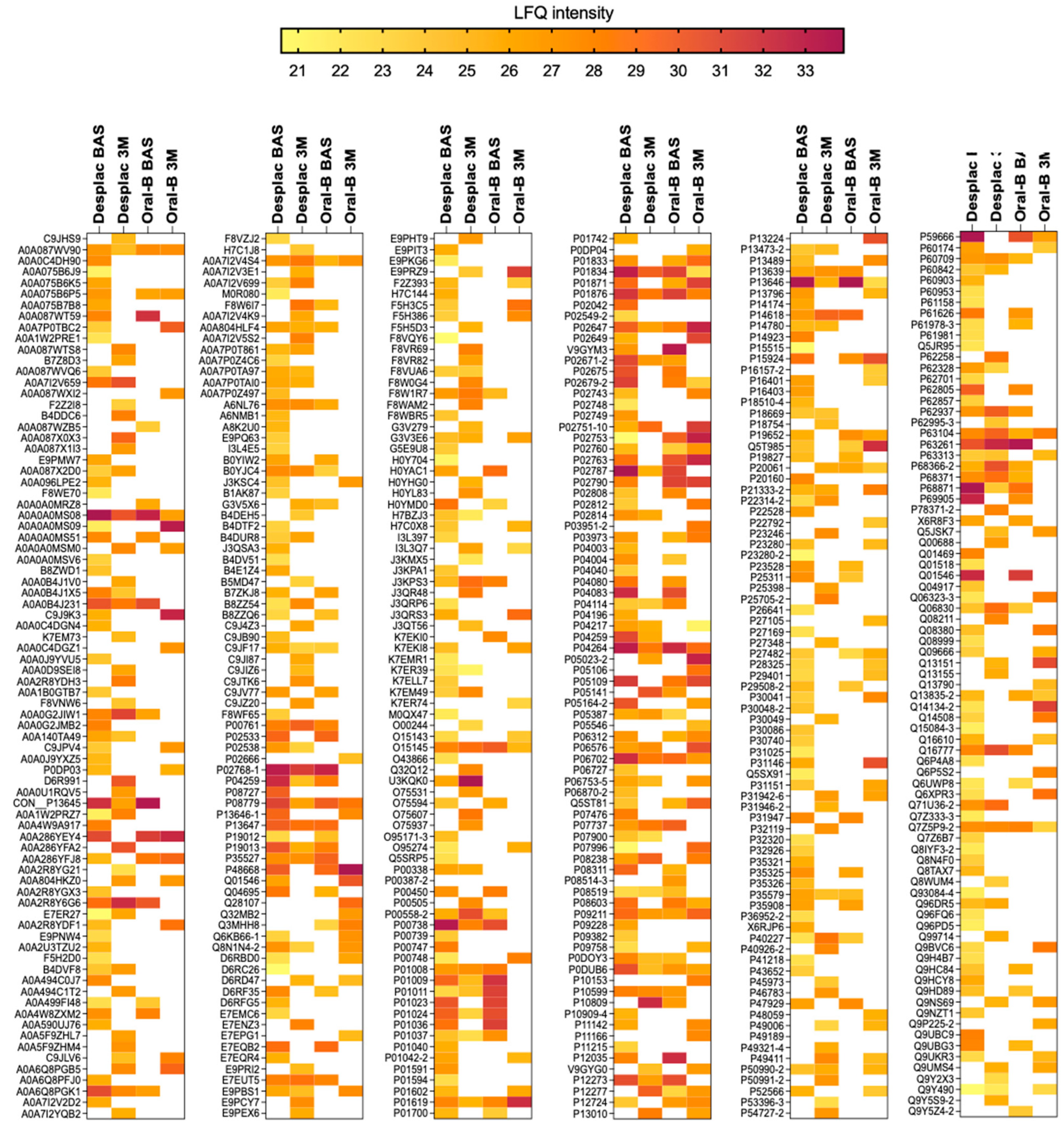
Our pioneering study explored the effect of a nature-based product on the GCF proteomic profile of gingivitis patients. A total of 480 proteins were identified across all groups at both time points. The conventional toothpaste group presented 143 proteins at baseline and 141 after 3 months (Figure 1A). For the nature-based gel group, 352 proteins were identified at baseline and 206 after 3 months (Figure 1A). Although the number of proteins somewhat decreased after 3 months, their intensities were similar for both groups, with no significant differences over time (Figure 1B). Importantly, both groups showed notable differences in the proteomic profile of GCF after 3 months compared with baseline (Figure 1C). Exclusive proteins were identified in the conventional toothpaste group (99 proteins) and the nature-based gel group (101 proteins) after 3 months within the same-group comparison. When comparing the two groups at each time point, our findings showed a lower level of similarity, with approximately 36% of proteins being shared at baseline and only 15.6% after 3 months (Figure 1C). In the pooled comparison of the groups at each time point, exclusive proteins were identified in both, indicating differences in proteomic profile according to time and treatment. These results demonstrate important differences in the proteomic profile of GCF during gingivitis treatment/control, which were also modulated by the toothpaste/gel applied. Despite differences in proteomic composition and the presence of exclusive proteins in each group, the heatmap shows that even among shared proteins, some differences in intensities were identified between both groups (Figure 2).
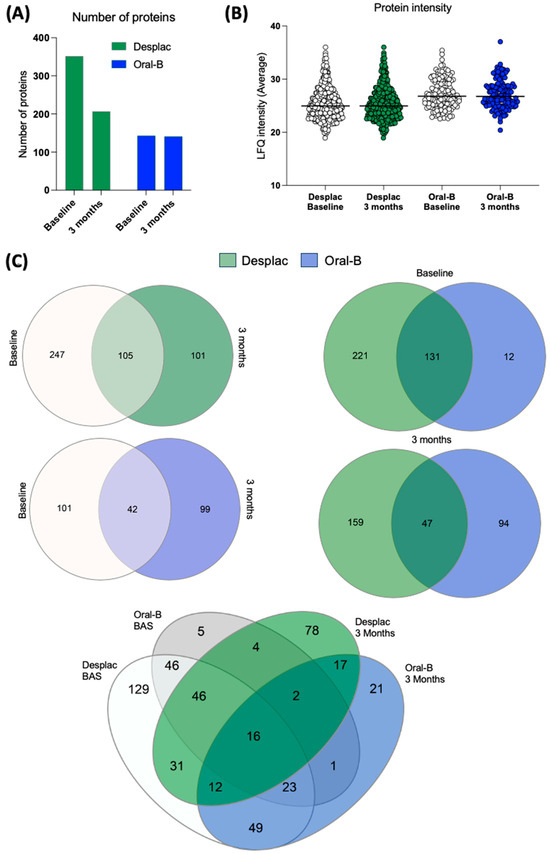
Figure 1.
Proteomic profile of gingival crevicular fluid of patients treated with nature-based gel (DESPLAC®) or conventional toothpaste (Oral-B). (A) Total proteins identified for each group and time point (baseline and after 3 months). Proteomic profile was evaluated using liquid chromatography coupled with tandem mass spectrometry. (B) Average LFQ intensity of proteins identified for each group and time point. (C) Venn diagrams comparing the groups and time points.
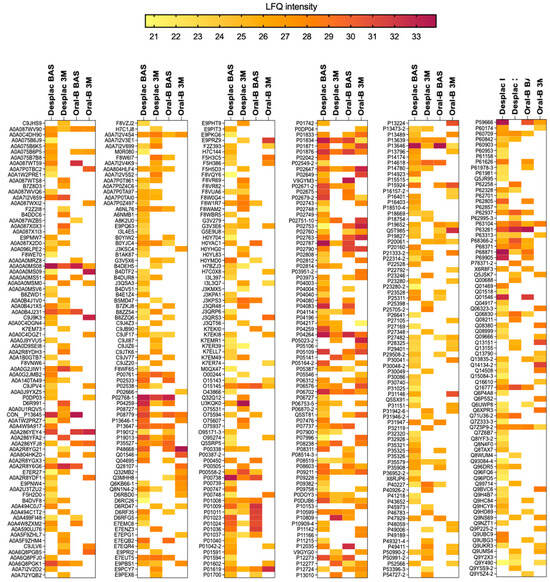
Figure 2.
Proteomic profile of gingival crevicular fluid of patients treated with nature-based gel (DESPLAC®) or conventional toothpaste (Oral-B) at different time points (BAS—baseline; or 3M—3 months). Heatmap of LFQ intensity of proteins identified for each group and time point.
Periodontal disease progression markedly changes the proteomic profile of GCF [26]. Moreover, changes in GCF protein levels caused by inflammation and disease resolution have been reported [27]. However, an important level of overlap in proteomic profile between healthy individuals and those with periodontal disease has been found [28], as reported in our study comparing scores at baseline and after 3 months. Our findings show that the nature-based gel increased the number of proteins identified in GCF, and compared with conventional toothpaste, it showed only 15.6% similarity after 3 months. Importantly, the pooled comparison (including both groups and time points) revealed that specific proteins could be uniquely identified for each group: 78 proteins were exclusive to the nature-based gel group, while 21 proteins were unique to the conventional toothpaste group at 3 months. This shows that the composition of the products highly affected the proteomic composition of GCF after 3 months of use. Notably, GCF is a complex mixture with various components, and its composition is affected by factors such as aging and biofilm composition [29]. Although all patients were standardized in terms of clinical parameters, molecular factors may have contributed to increased protein content in the nature-based gel group at baseline. However, after 3 months, the groups showed close protein quantities, with differences primarily driven by composition, which should be further investigated at the screening stage. Moreover, there was a clear change in the proteomic profile within the nature-based gel group when comparing the baseline and the 3-month follow-up, suggesting that the identified proteomes are a result of the treatment rather than the initial composition.
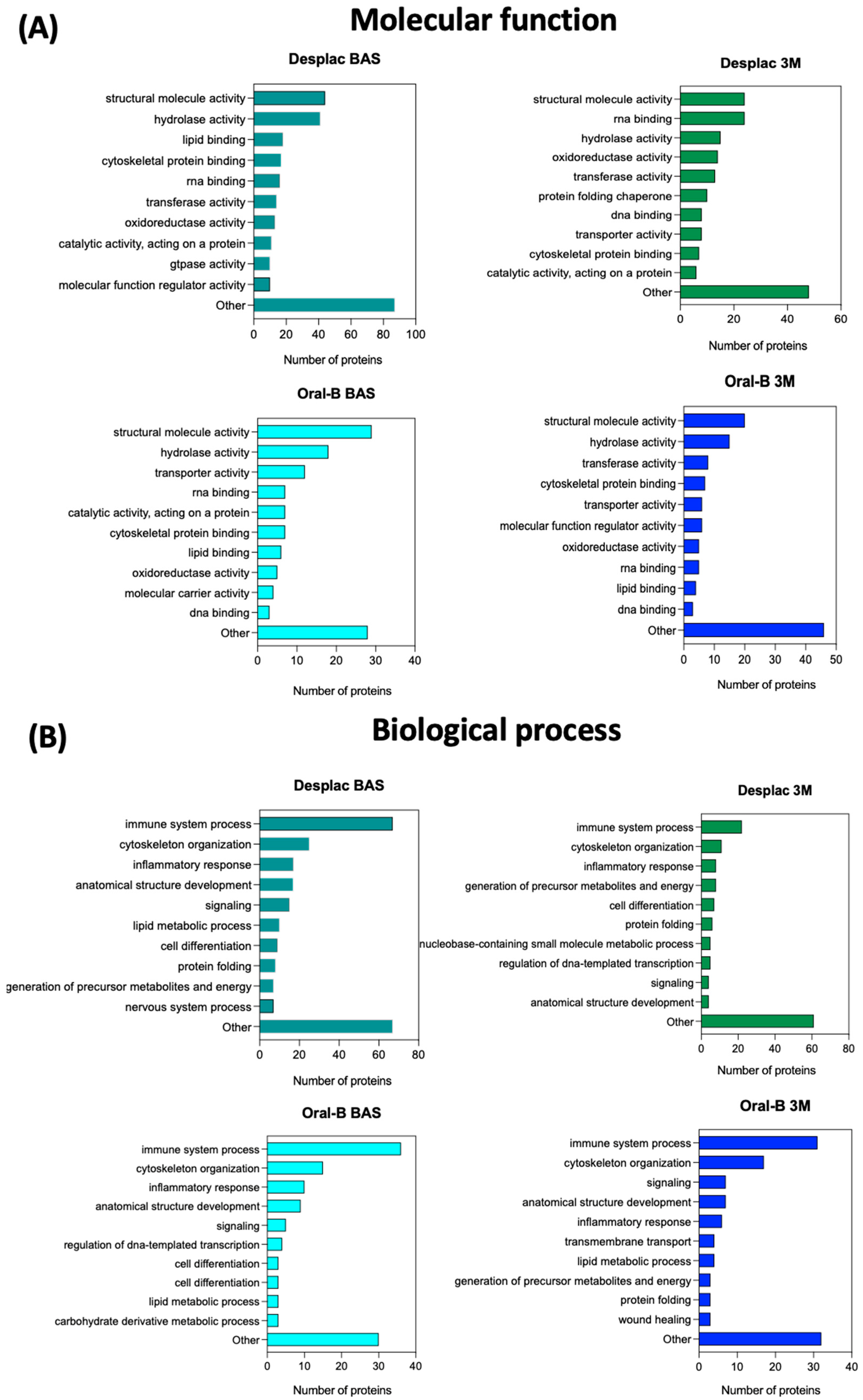
The exclusive proteins identified for each group in the pooled comparison (all groups and time points—Figure 1C, bottom diagram) are described in Table 2. After 3 months, the exclusive proteins identified for the conventional toothpaste mainly acted in wound healing, the most common biological process identified. In contrast, for the nature-based gel group, the exclusive proteins identified after 3 months primarily mediated immune system processes (Table 2). Therefore, considering that each protein has a specific molecular function and mediates specific biological processes, the profile for all proteins (not only exclusive ones) identified in each group and at each time point was also explored (Figure 3A,B). Although there is a high level of similarity between the groups at both time points in terms of the top 10 most common molecular functions and biological processes related to the identified proteins, some slight differences were observed. The inflammatory response process was among the top three biological processes for the nature-based gel group after 3 months but not for the conventional toothpaste group. Furthermore, cell differentiation was a common biological process identified for the nature-based gel but not for the conventional toothpaste. However, wound healing was only found among the common biological processes for the conventional toothpaste (Figure 3B). Therefore, the differences in proteomic profile led to slight differences in the main molecular functions and biological processes mediated by the identified proteins. The immune response process plays a key role in restoring homeostasis in injured tissues [30]. Therefore, the nature-based product and its composition may enhance the host response, and some of its components—such as propolis, with its well-recognized anti-inflammatory properties—may also contribute to faster inflammation resolution [30,31]. Although immune-response-related proteins were highly associated with the nature-based gel, clinical parameters, as measured by the gingival index, suggest the resolution of the inflammatory process, similar to what was observed with the conventional toothpaste, indicating no delay in the healing process for this group. Notably, the same protein can exhibit a wide range of molecular functions and be involved in different biological processes. While proteins may have primary functions, they can play roles in various processes. Moreover, the presence of a specific protein does not necessarily indicate a high concentration; some components of GCF may show similar levels immediately after therapy compared with the baseline but then exhibit reduced levels during the maintenance phase [32].

Table 2.
Protein ID, name, molecular function, and biological process of the exclusive proteins in each group and at each time when comparing the four groups’ proteomic profiles (data extracted from the UniProt database).
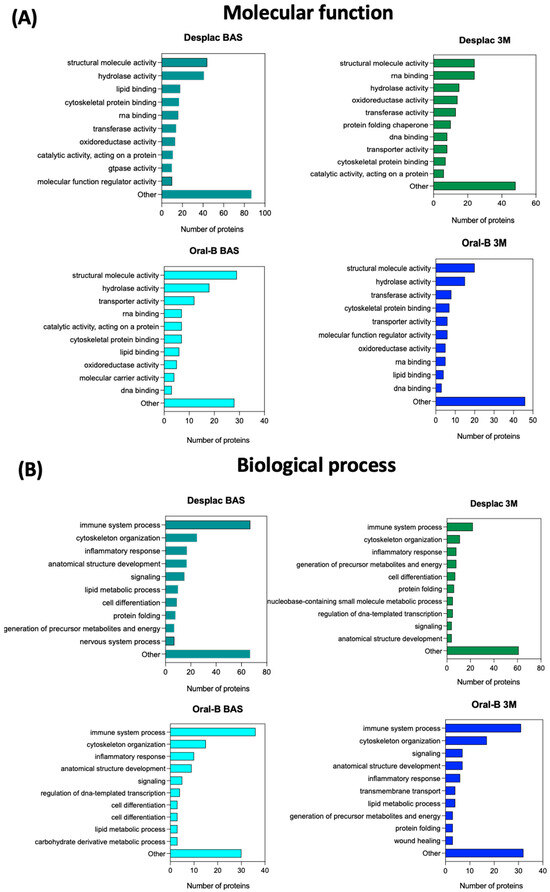
Figure 3.
Molecular functions and biological processes mediated by the proteins identified in each group and at each time point. Top 10 molecular functions (A) and biological processes (B) with the highest number of proteins adsorbed onto each substrate. Nature-based gel (DESPLAC®) or conventional toothpaste (Oral-B) at different time points (BA—baseline; or 3M—3 months).
The exclusive proteins identified in each group also need to be explored. For example, the conventional toothpaste group showed the exclusive protein integrin beta-3 at 3 months, which is part of a protein group associated with stimulating growth factors, forming granulation tissue, and modulating inflammatory processes [33]. The nature-based gel group also presented an important exclusive proteomic profile at 3 months, including the presence of Peroxiredoxin-2, which plays a role in immune system processes and has well-recognized antioxidant abilities [34]. These findings may provide important information on clinical changes at the molecular level and reveal new biomarkers for testing the effectiveness of therapeutic products or disease resolution.
Although these findings are promising in demonstrating the effectiveness of a nature-based product in controlling biofilm accumulation and gingival inflammation—and although 3 months of evaluation is commonly used for this type of trial [12]—further clinical trials should evaluate the maintenance of outcomes over longer periods. Moreover, the use of high-throughput techniques focusing on the oral microbiome should also be further explored and aligned with evaluating immune response pathways. Furthermore, the sample size used here may have reduced the chance of identifying differences between the groups, and further trials should consider larger samples. Clinical trials testing these new nature-based products are essential to elucidating the clinical efficacy of these formulations. However, patient profiles, the occurrence of other oral diseases, adverse effects, and the therapeutic protocols applied need to be considered before recommending these products. Moreover, patients need to be well informed about oral hygiene procedures and the importance of mechanical biofilm control through toothbrushing, regardless of toothpaste formulation.
3. Conclusions
We demonstrated that a nature-based gel achieved similar clinical outcomes to a conventional toothpaste in controlling gingival inflammation among gingivitis patients. However, the nature-based gel markedly changed the proteomic profile of GCF after treatment compared with the conventional toothpaste, increasing the number of proteins and showing a profile associated with a host response.
4. Materials and Methods
4.1. Study Design
This study protocol was developed following SPIRIT (Standard Protocol Items: Recommendations for Interventional Studies), and the manuscript was prepared according to the CONSORT checklist [35]. This was a double-blind, parallel, randomized, and controlled study. All subjects received and signed a form of informed consent. The study protocol was approved by the local Ethics Committee (IRB approval #51781621.9.0000.5506), and the clinical trial was registered (RBR-7dz5y9x—REBEC platform).
4.2. Treatment Groups
Using a table of equiprobable numbers, all selected individuals were randomly distributed to the following treatment groups: (1) test group (n = 10): nature-based gel (DESPLAC®, São Paulo, São Paulo, Brazil)—applied 2x/day during toothbrushing in the morning and evening; (2) control group (n = 10): Oral-B ProGengiva (Cincinnati, OH, EUA)—applied 2x/day during toothbrushing in the morning and evening. The DESPLAC® product (Premium Oral Gel) is the only toothpaste available on the Brazilian national market that includes a combination of natural agents in its formula: propolis, aloe vera, green tea, cranberry, and calendula. Considering the outcomes found for the average and standard deviation values of the nature-based gel group’s gingival index at baseline and after 90 days, the sample power was estimated using an α of 5% and an effect size of 1.23, resulting in a power of 90%.
4.3. Subject Population and Inclusion/Exclusion Criteria
Participants were selected from the population referred to the Dental Clinic of Guarulhos University (Guarulhos, SP, Brazil). The inclusion criteria were as follows: availability for the duration of the study; at least 15 natural teeth with minimal tooth restorations; good general health or health well controlled under a physician’s care; aged 18–65 years; and an average initial gingivitis index of at least 1.5 [36]. The exclusion criteria were as follows: a medical condition requiring premedication before dental visits/procedures; use of any medication that may affect salivary flow; xerostomia; carious lesions; sites with probing depths of >4 mm; use of orthodontic appliances; use of antibiotics within 6 months before or during the study; use of any over-the-counter medications that would affect the results, other than analgesics (i.e., aspirin, ibuprofen, acetaminophen, or naproxen) at the time of informed consent; pregnant or breastfeeding individuals; immune-compromised individuals; history of allergies to oral-care/personal-care consumer products or their ingredients; and a history of alcohol abuse, smoking, or drug abuse.
4.4. Procedures
All participants received the same oral hygiene instructions in terms of products to be used, a toothbrush with soft bristles, and the respective tube of toothpaste according to their assigned group. The study coordinator, who was uninvolved in the clinical evaluations, distributed the oral hygiene products to the participants. Allocation concealment was achieved using numbered, opaque, and sealed envelopes handled by the study coordinator. All products were stored in a sealed bag to eliminate any differences in product aesthetics and packaging between the study groups. Label information included a study group code, instructions for at-home use, and safety information (including emergency contact details). Participants were instructed to apply 1 cm of toothpaste to the brush and brush their teeth for 2 min twice a day (morning and evening) and to return their assigned products to the study site before receiving new products. They were instructed to follow these oral hygiene regimens for 90 days and to exclusively use the provided products. There were no specific instructions related to toothbrushing technique. They were not instructed to use dental floss to avoid introducing additional factors that could affect outcomes related to the toothpaste/gel composition and use. At the end of the 90 days, all participants returned to the dental clinic for a clinical evaluation and for the collection of gingival crevicular fluid samples. They were also instructed to refrain from performing any oral hygiene procedures for 4 h before the clinical evaluation at 90 days. The clinical evaluations and sample collections were conducted by calibrated examiners. Calibration was conducted according to a previously established protocol for periodontal studies [37]. The calibration phase was conducted before the study began, with examiners assessing specific clinical conditions in patients after familiarizing themselves with the protocol measures. At the first clinical appointment, an examiner assessed up to 42 sites per patient, followed by a second examiner who repeated the measurements in the same quadrant during the same appointment. Seven days later, at the second appointment, one of the original examiners and a third examiner repeated the measurements. The standard error of measurement was calculated, with intra-examiner variability at 0.26 mm and 0.28 mm for probing depth and clinical attachment level, respectively; inter-examiner values were 0.25 mm and 0.29 mm for probing depth and clinical attachment level, respectively, showing adequate reproducibility for periodontal evaluations. For categorical variables, such as plaque index and bleeding on probing, the level of agreement was estimated using Kappa, showing a 91% agreement rate.
4.5. Clinical Evaluation
At baseline and at 90 days, the presence or absence of visible plaque, marginal bleeding, and bleeding on probing, as well as probing depth and clinical attachment level, were assessed at six sites per tooth (excluding third molars) using a manual periodontal probe (North Carolina—Hu-Friedy, Chicago, IL, USA). Plaque accumulation and gingival inflammation were assessed for each tooth using standardized methods: the gingival index [36] and plaque index [38]. Each tooth was categorized by score, and an average score was calculated. The percentage of teeth assigned to each score was also calculated. Bleeding on probing was expressed as the average number of bleeding sites [39]. Probing depth and clinical attachment level were estimated in millimeters and expressed as an average.
4.6. Monitoring Compliance and Adverse Events
At the end of the study, participants were asked to return to the dental clinic and bring their tube(s) of toothpaste (empty or not), which were checked for any remaining toothpaste. The oral soft tissues of all subjects were evaluated during the clinical assessment at 90 days, and subjects were queried about any self-perceived side effects.
4.7. Gingival Crevicular Fluid (GCF) Sampling
The same GCF sampling procedure was performed for all patients. GCF was collected approximately one week after the clinical evaluation to avoid changes in the GCF due to blood contamination. Two non-adjacent shallow sites with a PD and CAL of ≤3 mm were randomly selected per patient. Supragingival biofilm was removed, and the sites were isolated and dried to avoid saliva contamination. GCF was sampled by inserting a standard absorbent paper cone approximately 2 mm into the sulcus/pocket for 60 s. After 20 s, a second absorbent paper cone was inserted in the same site for another 60 s. The two paper cones from the same site were pooled in a single dry microcentrifuge tube and stored at −80 °C. Strips contaminated with blood were discarded.
4.8. Protein Extraction
The absorbent papers containing the samples were placed in tubes with 150 μL of protein extraction solution (58% acetonitrile, 40% purified water, and 2% acetic acid). Each tube was agitated for 1 min, and the extraction procedure was performed in duplicate for each sample. After extraction, the solutions were pooled by group to increase protein concentration to a detectable level for the technique. The samples were then dried for 2 h, resuspended in ammonium bicarbonate (50 mM; pH 7.8), and dried again for an additional 1 h.
4.9. Liquid Chromatography Coupled with Tandem Mass Spectrometry
The proteomic profile was evaluated using liquid chromatography coupled with tandem mass spectrometry (LC–MS/MS), following a previous protocol [40,41,42]. Pooled samples were resuspended in urea (8 M). The Bradford method was used to quantify the total protein content. Then, the proteins were reduced, alkylated, digested with trypsin (1:50 w w-1), and subjected to LC–MS/MS. Samples were dried in a vacuum concentrator and immersed in 22.5 µL of 0.1% formic acid. An aliquot of 2 µL (0.88 µg) was analyzed on an LTQ Orbitrap Velos mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) connected to an EASY-nLC system (Proxeon Biosystem, West Palm Beach, FL, USA) through a Proxeon nanoelectrospray ion source. Peptides were separated by applying a 2–90% acetonitrile gradient in 0.1% formic acid in an analytical PicoFrit Column (20 cm × ID75 µm, 5 µm particle size) (New Objective Inc., Woburn, MA, USA) over 80 min [43]. The instrument methods were set up in the data-dependent acquisition mode. After accumulation to a target value of 1 × 106, full-scan MS spectra (m/z 300–1600) were made from an Orbitrap analyzer with a resolution of r = 60,000. A thousand (1000) counts was the signal threshold for triggering an MS/MS event. A size list of 500, a duration of 60 s, and a repeat count of 1 were enabled for dynamic exclusion.
Peptide sequences acquired were identified using MaxQuant (v.1.3.0.3—Martinsried, Munich, Germany), and MS/MS spectra were searched against the Human UniProt database. A maximum of 1%FDR was set for both protein and peptide identification. Protein quantification was performed using the LFQ algorithm implemented in MaxQuant, with a minimum ratio count of 2 and a window of 2 min for matching between runs. The list of peptides identified was filtered by a minimum 0.75 localization probability of containing at least one peptide with Perseus v.1.5. Reverse and contaminant entries were excluded from further analysis.
Exclusive proteins identified in each group were depicted using Venn diagrams constructed using a free web-based tool [44]. The name, molecular function, and biological process of each protein were checked using the UniProt database. Protein ID was used to search manually on UniProt for all information. Heatmaps were constructed using GraphPad Prism (version 8.0.0 for Windows, GraphPad Software, San Diego, CA, USA) to show LFQ intensity (protein intensity/expression) for each group and time.
4.10. Statistics
Prism (GraphPad) 8.0 and SPSS (20.0) were used to generate graphs and perform statistical analysis. Group comparisons, when necessary, were conducted using the t-test or repeated measures ANOVA (Tukey’s test). A significance level of 5% was adopted.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/gels10120772/s1, Table S1: Full-mouth gingival and plaque index evaluation of the study population at baseline and after 3 months according to scores. Proportion (%); Table S2: Percentage (%) of sites with bleeding on probing.
Author Contributions
Conceptualization: L.C.F., J.A.S. and B.B.-S.; data collection and analysis: L.C.F., J.G.S.S., G.D., N.F.F., D.F.d.C., M.H.R.B., D.H. and T.A.; manuscript writing: L.C.F., J.G.S.S. and V.A.R.B.; critical review: L.C.F., J.G.S.S., J.A.S., V.A.R.B., D.H. and T.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the local Ethics Committee (protocol code #51781621.9.0000.5506 and date of approval) for studies involving humans.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
Acknowledgments
We thank the Mass Spectrometry Facility at the Brazilian Biosciences National Laboratory (LNBio) at the Brazilian Center of Research in Energy and Materials (CNPEM) for proteomics analysis.
Conflicts of Interest
Authors Doron Haim and Thabet Asbi were employed by the company Maccabi-Dent Research Department. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S173–S182. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.B.; Yap, A.U.; Allen, P.F. Periodontal disease and quality of life: Umbrella review of systematic reviews. J. Periodontal Res. 2021, 56, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Marsh, P.D.; Moter, A.; Devine, D.A. Dental plaque biofilms: Communities, conflict and control. Periodontol. 2000 2011, 55, 16–35. [Google Scholar] [CrossRef] [PubMed]
- Chapple, I.L.C.; Mealey, B.L.; Van Dyke, T.E.; Bartold, P.M.; Dommisch, H.; Eickholz, P.; Geisinger, M.L.; Genco, R.J.; Glogauer, M.; Goldstein, M.; et al. Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: Consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S74–S84. [Google Scholar] [CrossRef]
- Socransky, S.S.; Haffajee, A.D. Implications of periodontal microbiology for the treatment of periodontal infections. J. Clin. Periodontol. 1994, 18, 688–693. [Google Scholar]
- Valkenburg, C.; Van der Weijden, F.A.; Slot, D.E. Plaque control and reduction of gingivitis: The evidence for dentifrices. Periodontol. 2000 2019, 79, 221. [Google Scholar] [CrossRef]
- James, P.; Worthington, H.V.; Parnell, C.; Harding, M.; Lamont, T.; Cheung, A.; Whelton, H.; Riley, P. Chlorhexidine mouthrinse as an adjunctive treatment for gingival health. Cochrane Database Syst. Rev. 2017, 3, CD008676. [Google Scholar]
- Gumber, H.K.; Louyakis, A.S.; Sarma, T.; Fabijanic, K.I.; Paul, R.; Mellenbruch, K.; Kilpatrick-Liverman, L. Effect of a Stannous Fluoride Dentifrice on Biofilm Composition, Gene Expression and Biomechanical Properties. Microorganisms 2022, 10, 1691. [Google Scholar] [CrossRef]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding protein adsorption phenomena at solid surfaces. Adv. Colloid. Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, Z.; Li, J.; Wang, Y.; Li, H.; Lv, R. Identification of ACTA2 as a key contributor to venous malformation. Front. Cell Dev. Biol. 2021, 9, 755409. [Google Scholar] [CrossRef]
- Davies, R.M. Toothpaste in the control of plaque/gingivitis and periodontitis. Periodontol. 2000 2008, 48, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Laleman, I.; Teughelsm, W. Novel natural product-based oral topical rinses and toothpastes to prevent periodontal diseases. Periodontol. 2000 2020, 84, 102–123. [Google Scholar] [CrossRef] [PubMed]
- Rashrash, M.; Schommer, J.C.; Brown, L.M. Prevalence and predictors of herbal medicine use among adults in the United States. J. Patient Exp. 2017, 4, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770. [Google Scholar] [CrossRef]
- López-Valverde, N.; López-Valverde, A.; Montero, J.; Rodríguez, C.; Macedo de Sousa, B.; Aragoneses, J.M. Antioxidant, anti-inflammatory and antimicrobial activity of natural products in periodontal disease: A comprehensive review. Front. Bioeng. Biotechnol. 2023, 11, 1226907. [Google Scholar] [CrossRef]
- Lima, M.D.R.; Lopes, A.P.; Martins, C.; Brito, G.A.C.; Carneiro, V.C.; Goes, P. The Effect of Calendula officinalis on Oxidative Stress and Bone Loss in Experimental Periodontitis. Front. Physiol. 2017, 8, 440. [Google Scholar] [CrossRef]
- Kranz, S.; Guellmar, A.; Olschowsky, P.; Tonndorf-Martini, S.; Heyder, M.; Pfister, W.; Reise, M.; Sigusch, B. Antimicrobial Effect of Natural Berry Juices on Common Oral Pathogenic Bacteria. Antibiotics 2020, 9, 533. [Google Scholar] [CrossRef]
- Rezaei-Tazangi, F.; Forutan Mirhosseini, A.; Fathi, A.; Roghani-Shahraki, H.; Arefnezhad, R.; Vasei, F. Herbal and nano-based herbal medicine: New insights into their therapeutic aspects against periodontitis. Avicenna J. Phytomed. 2024, 14, 430–454. [Google Scholar]
- Macedo, T.T.; Malavazi, L.M.; Vargas, G.Q.; Gonçalves, F.J.D.S.; Gomes, A.P.A.P.; Bueno, M.R.; Aguiar da Silva, L.D.; Figueiredo, L.C.; Bueno-Silva, B. Combination of Neovestitol and Vestitol Modifies the Profile of Periodontitis-Related Subgingival Multispecies Biofilm. Biomedicines 2024, 12, 1189. [Google Scholar] [CrossRef]
- Figueiredo, L.C.; Freitas Figueiredo, N.; da Cruz, D.F.; Baccelli, G.T.; Sarachini, G.E.; Bueno, M.R.; Feres, M.; Bueno-Silva, B. Propolis, Aloe Vera, Green Tea, Cranberry, Calendula, Myrrha and Salvia Properties against Periodontal Microorganisms. Microorganisms 2022, 10, 2172. [Google Scholar] [CrossRef]
- Bueno-Silva, B.; Kiausinus, K.R.; Gonçalves, F.J.D.S.; Moreira, M.V.C.; de Oliveira, E.G.; Brugnera Junior, A.; Feres, M.; Figueiredo, L.C. Antimicrobial activity of Desplac® oral gel in the subgingival multispecies biofilm formation. Front. Microbiol. 2023, 14, 1122051. [Google Scholar] [CrossRef] [PubMed]
- Valkenburg, C.; Slot, D.E.; Bakkerm, E.W.P.; Weijdenm, F.A.V.D. Does dentifrice use help to remove plaque? A systematic review. J. Clin. Periodontol. 2016, 43, 1050. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.; Worthington, H.V.; Glenny, A.M.; Marinho, V.C.; Jeroncic, A. Fluoride toothpastes of different concentrations for preventing dental caries. Cochrane Database Syst. Rev. 2019, 3, CD007868. [Google Scholar] [CrossRef] [PubMed]
- Shick, R.A. Maintenance phase of periodontal therapy. J. Periodontol. 1981, 52, 576–583. [Google Scholar] [CrossRef]
- Brecx, M.C.; Lehmann, B.; Siegwart, C.M.; Gehr, P.; Lang, N.P. Observations on the initial stages of healing following human experimental gingivitis. A clinical and morphometric study. J. Clin. Periodontol. 1988, 15, 123–129. [Google Scholar] [CrossRef]
- Torres, A.; Michea, M.A.; Végvári, Á.; Arce, M.; Morales, A.; Lanyon, E.; Alcota, M.; Fuentes, C.; Vernal, R.; Budini, M.; et al. Proteomic profile of human gingival crevicular fluid reveals specific biological and molecular processes during clinical progression of periodontitis. J. Periodontal Res. 2023, 58, 1061–1081. [Google Scholar] [CrossRef]
- Grant, M.M.; Creese, A.J.; Barr, G.; Ling, M.R.; Scott, A.E.; Matthews, J.B.; Griffiths, H.R.; Cooper, H.J.; Chapple, I.L. Proteomic analysis of a noninvasive human model of acute inflammation and its resolution: The twenty-one day gingivitis model. J. Proteome Res. 2010, 9, 4732–4744. [Google Scholar] [CrossRef]
- Carneiro, G.C.; Nouhm, H.; Salih, E. Quantitative gingival crevicular fluid proteome in health and periodontal disease using stable isotope chemistries and mass spectrometry. J. Clin. Periodontol. 2014, 41, 733–747. [Google Scholar] [CrossRef]
- Barros, S.P.; Williams, R.; Offenbacher, S.; Morelli, T. Gingival crevicular fluid as a source of biomarkers for periodontitis. Periodontol. 2000 2016, 70, 53–64. [Google Scholar] [CrossRef]
- Cekici, A.; Kantarci, A.; Hasturk, H.; Van Dyke, T.E. Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontol. 2000 2014, 64, 57–80. [Google Scholar] [CrossRef]
- Zulhendri, F.; Lesmana, R.; Tandean, S.; Christoper, A.; Chandrasekaran, K.; Irsyam, I.; Suwantika, A.A.; Abdulah, R.; Wathoni, N. Recent Update on the Anti-Inflammatory Activities of Propolis. Molecules 2022, 27, 8473. [Google Scholar] [CrossRef] [PubMed]
- Kinane, D.F.; Darby, I.B.; Said, S.; Luoto, H.; Sorsa, T.; Tikanoja, S.; Mäntylä, P. Changes in gingival crevicular fluid matrix metalloproteinase-8 levels during periodontal treatment and maintenance. J. Periodontal. Res. 2003, 38, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, L.; Heino, J.; Hakkinen, L.; Larjava, H. Integrins in Wound Healing. Adv. Wound Care (New Rochelle) 2014, 3, 762–783. [Google Scholar] [CrossRef] [PubMed]
- Sadowska-Bartosz, I.; Bartosz, G. Peroxiredoxin 2: An Important Element of the Antioxidant Defense of the Erythrocyte. Antioxidants 2023, 12, 1012. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.W.; Tetzlaff, J.M.; Altman, D.G.; Laupacis, A.; Gøtzsche, P.C.; Krleža-Jerić, K.; Hróbjartsson, A.; Mann, H.; Dickersin, K.; Berlin, J.A.; et al. SPIRIT 2013 statement: Defining standard protocol items for clinical trials. Ann. Intern Med. 2013, 158, 200–207. [Google Scholar] [CrossRef]
- Loe, H.; Silness, J. Periodontal disease in pregnancy. I. Prevalence and severity. Acta Odontol. Scand. 1963, 21, 533–551. [Google Scholar] [CrossRef]
- Araujo, M.W.; Hovey, K.M.; Benedek, J.R.; Grossi, S.G.; Dorn, J.; Wactawski-Wende, J.; Genco, R.J.; Trevisan, M. Reproducibility of probing depth measurement using a constant-force electronic probe: Analysis of inter- and intraexaminer variability. J. Periodontol. 2003, 74, 1736–1740. [Google Scholar] [CrossRef]
- Loe, H.; Silness, J. Periodontal disease in pregnancy. II. correlation between oral hygiene and periodontal condtion. Acta Odontol. Scand. 1964, 22, 121–135. [Google Scholar]
- Tamashiro, N.S.; Duarte, P.M.; Miranda, T.S.; Maciel, S.S.; Figueiredo, L.C.; Faveri, M.; Feres, M. Amoxicillin Plus Metronidazole Therapy for Patients with Periodontitis and Type 2 Diabetes: A 2-year Randomized Controlled Trial. J. Dent. Res. 2016, 95, 829–836. [Google Scholar] [CrossRef]
- Siqueira, W.L.; Oppenheim, F.G. Small molecular weight proteins/peptides present in the in vivo formed human acquired enamel pellicle. Arch. Oral. Biol. 2009, 54, 437–444. [Google Scholar] [CrossRef]
- Souza, J.G.S.; Bertolini, M.; Costa, R.C.; Lima, C.V.; Barão, V.A.R. Proteomic profile of the saliva and plasma protein layer adsorbed on Ti-Zr alloy: The effect of sandblasted and acid-etched surface treatment. Biofouling 2020, 36, 428–441. [Google Scholar] [CrossRef] [PubMed]
- Miranda, L.F.B.; Lima, C.V.; Pagin, R.; Costa, R.C.; Pereira, M.M.A.; de Avila, E.D.; Bertolini, M.; Retamal-Valdes, B.; Shibli, J.A.; Feres, M.; et al. Effect of Processing Methods of Human Saliva on the Proteomic Profile and Protein-Mediated Biological Processes. J. Proteome Res. 2023, 22, 857–870. [Google Scholar] [CrossRef] [PubMed]
- Carnielli, C.M.; Macedo, C.C.S.; De Rossi, T.; Granato, D.C.; Rivera, C.; Domingues, R.R.; Pauletti, B.A.; Yokoo, S.; Heberle, H.; Busso-Lopes, A.F.; et al. Combining discovery and targeted proteomics reveals a prognostic signature in oral cancer. Nat. Commun. 2018, 9, 3598. [Google Scholar] [CrossRef] [PubMed]
- Heberle, H.; Meirelles, G.V.; da Silva, F.R.; Telles, G.P.; Minghim, R. InteractiVenn: A web-based tool for the analysis of sets through Venn diagrams. BMC Bioinform. 2015, 16, 169. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).