Abstract
In order to explore the variation in volatile compounds and aroma profiles of different varieties of sugarcane wine, volatile compounds of 14 different varieties of sugarcane wine were analyzed by headspace solid-phase microextraction–gas chromatography–mass spectrometry (HS-SPME-GC-MS) and an electronic sensory system. The differences in flavor substances of different cultivars of sugarcane were assessed by orthogonal partial least squares discriminant analysis (OPLS-DA) discriminant model and relative odor activity value (ROAV) combined with multivariate statistical methods. The results showed that a total of sixty major volatile compounds, i.e., 27 esters, 15 alcohols, eight acids, three phenols, four aldehydes and ketones, and four others, were identified in 14 types of sugarcane wine. Seven key aroma compounds were screened out: ethyl caprylate, ethyl caprate, ethyl acetate, ethyl laurate, n-decanoic acid, 2,4-di-tert-butylphenol, and 2-phenylethanol and three differential aromas, i.e., ethyl palmitate, isobutyl alcohol, and caprylic acid. The electronic nose and electronic tongue analysis technology can effectively distinguish the aroma and taste of 14 sugarcane wines. It is confirmed that the aroma and taste of 14 sugarcane wines have differences in distribution patterns, and the results are consistent with the analysis and assessment of volatile compounds of sugarcane wine. The results of this study provide technical support for the production and quality improvement of sugarcane wine.
1. Introduction
Sugarcane (Saccharum spp. Hybrid) is an important agricultural cash crop around the world, mainly growing in arid and semiarid regions. It is mainly cultivated in different provinces in China, such as Guangxi, Guangdong, Yunnan, Hainan, etc. [1,2]. More than 80% of the world’s annual raw sugar production and 40% of the ethanol are produced by sugarcane plants [3,4]. Sugarcane juice is a major source of sucrose, fructose, and glucose, which are quickly absorbed by the body into the blood and provide a quick energy boost. It can effectively boost metabolic activities and reduce body temperature, promoting body fluids, moisturizing and relieving cough, strengthening the stomach and spleen, antioxidation, anti-aging, and preventing cancer cell differentiation [5,6,7,8]. It contains bioactive ingredients like polyphenols and flavonoids and has extremely high nutritional and flavor values [9]. Sugarcane is rich in nutrients and develops sugarcane wine [10,11], fruit vinegar [12,13,14], daily use beverages [11,15], and other products that can not only promote the diversified production of the sugarcane industry and promote added value but also extend the industrial chain of sugarcane processing.
Nowadays, research on sugarcane wine production mainly focuses on the optimization of the wine fermentation process, strain screening, and analysis of a specific type of chemical compound. Tang et al. [11] studied the mixed bacteria inoculation and fermentation process on lemon sugarcane wine. Lin et al. [16] and Chen et al. [12] conducted research on sugarcane wine and obtained low-alcohol sugarcane wine. The wine is clear and transparent, with a fresh sugarcane aroma and a soft taste. However, previous studies have mainly focused on the optimization of fermentation, and limited studies have assessed the effect of the sugarcane variety on the aroma characteristics. As an emerging fruit wine, sugarcane wine lacks reference values for its flavor characteristics, and there are limited research reports on how to improve and enhance the aroma of sugarcane wine. The aroma of fruit wine is an important indicator that affects consumers’ acceptance of fruit wine [13].
Raw materials greatly affect the flavor quality and sensory characteristics of fruit wine. The aroma composition ratio of fruit wines brewed from different varieties is different, which will affect the flavor and taste of the wine [17,18,19,20]. A study on the physical and chemical indicators and volatile compounds of five varieties of pear wine found that late autumn yellow pear is more suitable for brewing pear wine [21]. Wang et al. [22] analyzed the effect of different varieties of kiwifruit on the volatile and aroma compounds of their fermented wine based on the previous research carried out on the flavor substances of fermented sugarcane wine of different cultivars. The headspace solid-phase microextraction (HS-SPME) and gas chromatography–mass spectrometry (GC-MS) methods were applied to qualitatively and quantitatively analyze the volatile compounds of 14 different varieties of sugarcane wine. The orthogonal partial least squares discriminant analysis (OPLS-DA) discriminant model, relative odor activity value (ROAV), and principal component analysis (PCA) combined with multivariate statistical strategies were applied to analyze the differences of flavor substances and the characteristics of the key aroma of sugarcane wine. Combined with electronic nose and electronic tongue detection, it was further confirmed that there were significant differences in the aroma and taste of different varieties of sugarcane wine, providing technical knowledge and data support for its processing and production.
2. Materials and Methods
2.1. Plant Material
Fourteen sugarcane cultivars were used in this experiment, such as New Taiwan Sugar 22, Guitang 44, Guitang 46, Guitang 49, Guitang 58, Guitang 42, Guiliu 05136, Guitang 55, Guitang 59, Guitang 2022, Guitang 218, Guitang 60, Guiguozhe 1, and Guihuangpi 1 provided by the Sugarcane Research Institute of Guangxi Academy of Agricultural Sciences, Nanning, Guangxi, China (Table 1). Fresh sugarcane was squeezed to extract juice and stored in the freezer for further analysis.

Table 1.
List of sugarcane cultivars used in the experiment and serial number of fermented sugarcane wine.
2.2. Equipment and Reagents
White sugar, pectinase enzyme, and Angel SY fruit wine yeast were used. Ethanol, sodium nitrite, aluminum nitrate, sodium hydroxide, glacial acetic acid, sodium chloride, and phenolphthalein were provided by Tianjin Damao Chemical Reagent Factory, Tianjin, China. 2-octanol standard (purity ≥ 98%), folin, gallic acid, and rutin standard (purity ≥ 98%) were provided by Shanghai Yuanye Biotechnology Co., Ltd., Shanghai, China. HS-SPME-GC-MS (Agilent 8890-7000C, Santa Clara, CA, USA) with multi-function (PAL RSI 120) autosampler (Agilent Technologies, Inc., Santa Clara, CA, USA), SA-402B electronic tongue (Insent, Atsugi, Japan), PEN3 electronic nose (Airsense, Schwerin, Germany), solid phase microextraction fiber head 65 μm (PDMS/DVB) (Supelco Inc., Bellefonte, PA, USA), CAHT-50L ultrapure water device (Beijing Zhongke Zhiheng Technology Co., Ltd., Beijing, China), UV–visible spectrophotometer (UV-6100) (Shanghai Yuanxi Instrument Co., Ltd., Shanghai, China), D3500S-DTH ultrasonic cleaner (Branson Ultrasonic, Shanghai Co., Ltd., Shanghai, China), BIC-300 artificial climate chamber (Shanghai Boxun Industrial Co., Ltd., Shanghai, China), VORTEX GENIUS 3 Vortex oscillator (IKA, Staufen, Germany), PAL-1 saccharimeter (ATAGO, Tokyo, Japan), etc.
2.3. Sugarcane Fermentation Process
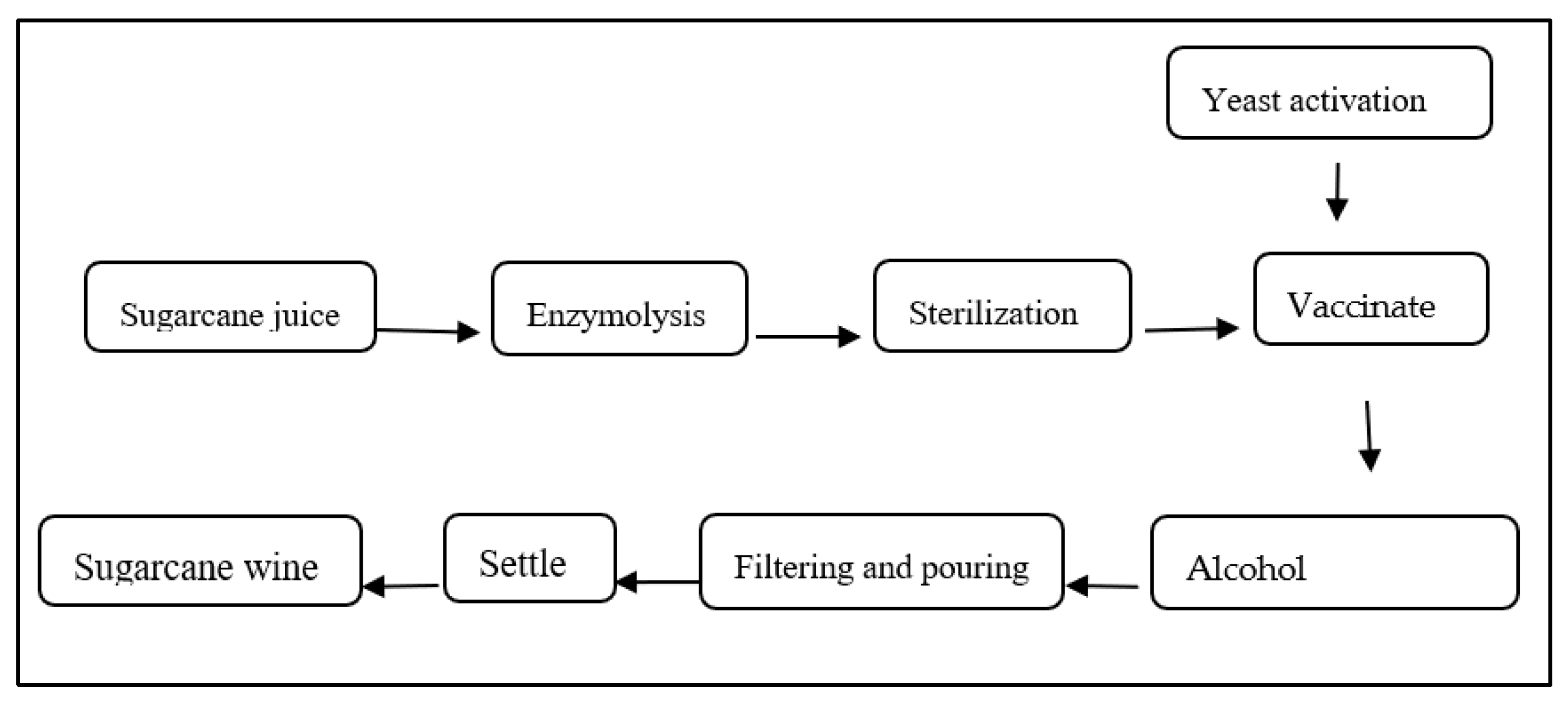
The sugarcane fermentation process was performed according to Chen et al. [10]. Firstly, take fresh sugarcane juice of different cultivars, add 0.10% (by mass volume ratio of sugarcane juice) of pectinase, perform enzymolysis at 40 °C for 1 h, then sterilize in a 70 °C water bath for 30 min, and cool at room temperature (RT), add activated yeast (by mass volume ratio of sugarcane juice, 0.10%) to the sugarcane juice, and ferment at fermentation temperature (25 °C). After 10 days of fermentation, the main fermentation was terminated, and filtration, backflow, aging, and other treatment processes were carried out. All wine samples were centrifuged at 534× g for 5 min; supernatants were collected and stored for further analysis (Figure 1).
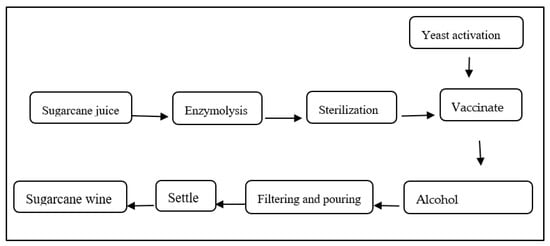
Figure 1.
Outline of the fermentation process of sugarcane wine.
2.4. Determination of Physical and Chemical Indicators
Sugar content and pH value were determined by a handheld sugar and pH meter. The alcohol content was observed in accordance with GB/T 15038-2006 [23] “General Analysis Method for Grape and Fruit Wine”, and total acid content was analyzed in accordance with the first method of GB/T 12456-2021 [24]. The acid–base indicator and total acid content were calculated using acetic acid. Total phenol and total flavonoid content were determined [25].
2.5. Determination of Aroma Profile in Sugarcane Wine
The solid-phase microextraction method was applied as 5 mL of wine sample, 10 μL of 2-octanol (concentration 0.1001 g/L), and 1 g of NaCl were placed in a 20 mL solid-phase microextraction bottle. The shaking speed was 400 r/min, equilibrated at 50 °C for 10 min, extracted for 40 min, desorbed for 4 min, and then analyzed by GC-MS. Three parallel samples (n = 3) were performed for each sample.
GC-MS analysis employed an HP-INNOWAX column (60 mm × 0.25 mm × 0.25 µm, Agilent, Santa Clara, CA, USA) with high-purity helium carrier gas (≥99.999%) at a constant flow rate of 1.0 mL/min. The injection mode was set to a split ratio of 5:1, with an inlet temperature of 250 °C. The temperature program started at 50 °C for 1 min, was enhanced to 220 °C at 3 °C/min, and then 220 °C was held for 5 min.
Mass spectrometry conditions: electron impact (EI) ion source; ionization energy 70 eV, ion source temperature 230 °C, transfer line temperature 250 °C, quadrupole temperature 150 °C; scanning mode SCAN and mass range 30–350 (m/z).
2.6. Analysis of Aroma Compounds
Qualitative analysis: The retention index and the peaks in the total ion flow were searched by the mass spectrometry computer data system. The spectrum was manually analyzed, components with a matching degree greater than 80% were screened out, and the qualitative analysis was combined with relevant literature [26]. The retention index was a mixed standard of normal alkanes (C7–C26).
Quantitative analysis: The internal standard method was applied for semi-quantitative analysis, and 2-octanol was used as the internal standard. The aroma compounds were calculated according to Li et al. [27].
2.7. Analysis of Key Flavor Compounds
The relative odor activity value (ROAV) method was used to assess the contribution of individual compounds to the overall aroma of fruit wine. The key flavor components were determined by the size of the ROAV [28]. ROAV = 100 was defined as the maximum odor activity value among the aroma components of the sample. The ROAV value was calculated according to Chen et al. [13].
An electronic nose was used to determine different varieties of sugarcane wine [29,30]; 15 mL of sugarcane wine was placed in a headspace bottle, sealed for 60 min, and then observed by the electronic nose (Table 2). The electronic nose determination conditions are as follows: room temperature 25 °C, air intake 400 mL/min, sampling time 60 s, and data from 54 to 57 were taken for aroma analysis.

Table 2.
Electronic nose sensor array.
2.8. Statistical Analysis and Data Processing
Microsoft Office Excel 2016 was used for data collection. Multivariate analysis of variance was used to assess the differences in aroma and flavor quality between different varieties of sugarcane wine. The aroma and flavor of different sugarcane wines were analyzed using principal component analysis (PCA). Orthogonal partial least squares-discriminant analysis (OPLS-DA) was performed using SIMCA 14.1 software to calculate the variable importance in projection (VIP). SPSS19 software was used for univariate analysis to screen the differential aroma components under the conditions of p< 0.05 and VIP > 1. Origin2021 software was used for graphical design.
3. Results and Discussion
3.1. Quality Analysis of Sugarcane Wine Fermented from Sugarcane Juice of Different Cultivars
Alcohol content is a key indicator for the assessment of sugarcane wine quality. As shown in Table 3, the alcohol content of 14 sugarcane wines prepared from the different sugarcane cultivars ranged from 8.70 to 13.07% vol, all of which met the requirements of NY/T 1508-2017 [31] Green Food Wine. There were significant differences in different varieties (p < 0.05), among which GJ05 found the highest (13.07% vol) and GJ07 lowest alcohol content (8.70% vol). The initial sugar content ranged from 18.0 to 22.7 °Brix, except for GJ06, GJ08, and GJ10. There were no significant differences among the other varieties. The residual sugar content of the fruit wine after fermentation was 6.0 to 8.9 °Brix. The raw material for alcohol metabolism is sugar, and the sugar content affects the degree and frequency of the fermentation process. Therefore, within the same fermentation time, the difference in alcohol content can be caused by different initial sugar contents among varieties. The effects of apple varieties on the fermentation process were that the production of alcohol content was mainly associated with the content of fermentable sugar [32].

Table 3.
Key results of quality components of sugarcane wine prepared from the different cultivars of sugarcane.
Total acid content plays a major role in shaping the flavor of wine, and it is largely determined by the quality characteristics of the fruit [33]. The pH value and total acidity of 14 sugarcane wines prepared from sugarcane cultivars ranged from 3.87 to 4.99 and 2.71 to 4.51 g/L, respectively (Table 3). Among them, GJ12 found the highest pH value (4.99), which was significantly higher than other varieties (3.87 to 4.86). The highest total acid content was observed in GJ02 and the lowest in GJ01. The total acid content and pH value of the 14 sugarcane wines were inconsistent due to some acids and alcohols reacting in the form of esters during the fermentation process. This result is similar to that of Robles et al. [34].
Flavonoids and polyphenols are important bioactive substances in sugarcane wine [35] and are also important indicators for measuring the differences in functional properties between different sugarcane cultivars [21]. The total phenol and flavonoid contents of different sugarcane varieties were significantly different (p < 0.05), with GJ12 having the optimum total phenol content (756.24 mg/L) and GJ08 and GJ06 having the lowest (468.64 and 480.91 mg/L). The maximum coefficient of variation of total flavonoid content among different varieties of sugarcane wine was 14.81%, with GJ10 and GJ02 being the highest (109.79 and 109.05 mg/L), and GJ05 being the lowest total flavonoid content (62.76 mg/L), as shown in Table 3. Aroma is one of the important indicators of fruit wine quality. It is composed of a large number of volatile aromatic compounds and the flavor characteristics of fruit wine [36,37].
3.2. Analysis of Aroma of Different Varieties of Sugarcane Wine by GC-MS
Headspace solid-phase microextraction–gas chromatography–mass spectrometry (HS-SPME-GC-MS) was used to analyze the volatile compounds (VCs) of 14 sugarcane wines. The analytical results are shown in Table 4.

Table 4.
Content of volatile compounds in different sugarcane wines.
It can be seen from Table 4 that sixty-one VCs were found in sugarcane wine: 27 esters, 15 alcohols, three phenols, eight acids, four aldehydes and ketones, and four others. Different types of VCs were observed in the different varieties of sugarcane wine ranging from 40 to 55, among which GJ01 contained the most VCs (55), and GJ12 had the least VCs (40), which was similar to the results of Chen et al. [13]. The total aroma content of the 14 sugarcane wines was different, ranging from 1.579 to 4.174 mg/L. GJ11 observed the highest aroma content (4.174 mg/L), and GJ08 had the lowest aroma content (1.579 mg/L). The VCs of each sugarcane wine also have differences. There are 30 aroma compounds in total, i.e., 13 esters, eight alcohols, five acids, two phenols, and one other. The four aroma compounds with the highest content are ethyl octanoate, ethyl decanoate, isopentanol, and 2-phenylethanol. It is not completely consistent with the results of Oliveira et al. [38]. The three compounds with the highest content in sugarcane wine are isopentanol, ethyl octanoate, and 2-methyl-1-butanol. This can be related to the different yeast species.
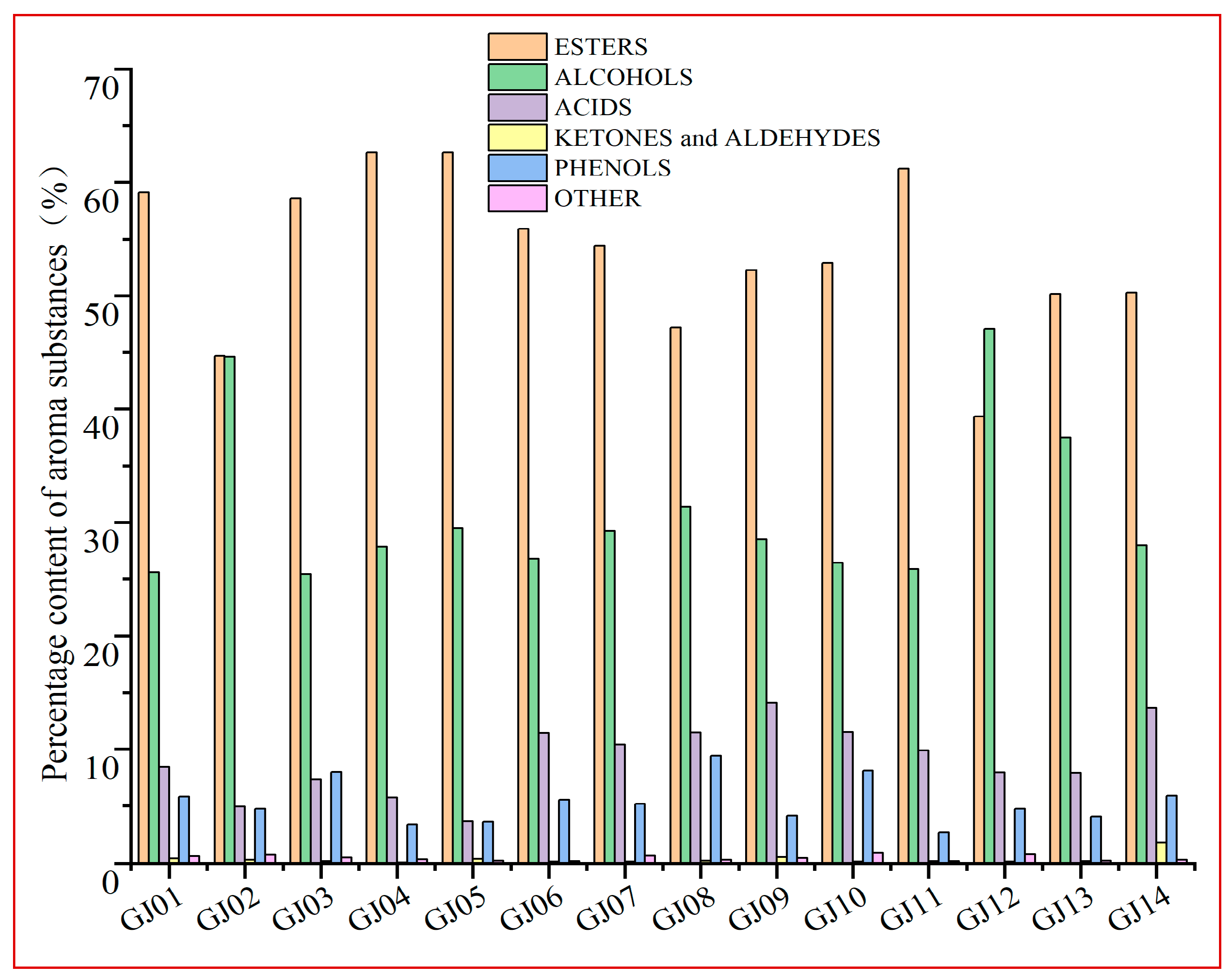
The percentages of the main VCs in different sugarcane wines are shown in Figure 2. The main VCs in sugarcane wine are esters and alcohols, followed by acids and phenols. There are specific differences in the percentage of VCs in different varieties of sugarcane wine, and the difference between their contents affects the flavor of the wine (Figure 2). The percentage of esters (39.34–64.32%) was observed in the 14 types of sugarcane wine, with the percentage of alcohols (25.42–47.07%), acids (4.95–13.77%), phenols (3.43–8.85%), and aldehydes and ketones (0.12–0.46%). Esters and alcohols are the main aroma compounds of sugarcane wine, which are similar research findings to Lin et al. [16] and other main wine aromas. However, the changes in different classifications between different varieties are relatively more numerous, among which GJ04 has a higher percentage of esters, GJ12 has the highest percentage of alcohols, and GJ02 has little difference in the esters and alcohols. This shows that the variety of raw materials has a significant impact on the classification and composition of sugarcane wine flavors.
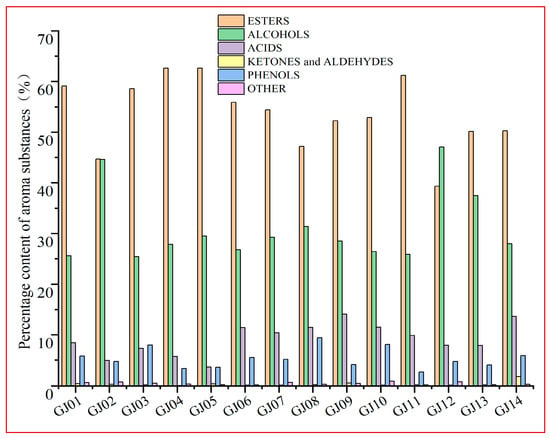
Figure 2.
Percentage content of volatile compounds in a variety of sugarcane wines.
Esters are the main components of the aroma of fruit wine, which are mainly produced during the yeast fermentation process with richer and more flavor [39]. A total of 13 ester components were observed in 14 sugarcane wines. Ethyl caprylate, ethyl caprate, ethyl caproate, ethyl acetate, isoamyl acetate, and ethyl laurate were assessed in all 14 wine samples, and the content was relatively high. The sugarcane wine produced a fat aroma, brandy aroma, strawberry aroma, and banana aroma [40]. However, phenylethyl acetate, ethyl myristic acid, and ethyl palmitate were also found in sugarcane wine. The apple aroma, vanilla aroma, rose aroma, honey and cream aroma are consistent with the aroma compounds of sugarcane wine [10] and others. However, methyl salicylate was assessed in GJ01 and GJ14, which produced the fruit wine with a light mint aroma. Ethyl heptanoate and ethyl lactate were found in GJ05, GJ06, and GJ13, which can produce fruit wine with pineapple and rum aromas [15].
Alcohols are important aroma compounds in the flavor characteristics of fruit wine. They are mainly derived from amino acid metabolism or sugar metabolism during the fermentation process [41]. The content of alcohol substances and the ratio between various alcohols also affect the flavor of wine [26,35]. A total of 15 alcohol substances were found in 14 types of sugarcane wine, of which there were eight compounds, namely isopentanol, isobutanol, n-hexanol, 1-octanol, and 2-phenylethanol, which produce sugarcane wine apple aroma, grass aroma, jasmine aroma, lemon aroma, and rose aroma [15]. 2-phenylethanol found in sugarcane wine has a rose aroma, and citronellol has rich floral aromas such as rose and clove. Trans-nerolidol found in GJ06 and GJ14 produces sugarcane wine with an apple aroma and wood aroma.
Acids are important associated compounds of the aroma of fruit wine. Appropriate amounts of acid improve the taste and color of fruit wine [42] and are essential for the balance of aroma compounds in wine. A total of eight acids were observed in different sugarcane wine samples, of which three were common compounds, i.e., caprylic acid, caproic acid, and isobutyric acid. Caprylic acid is the volatile acid found in optimum content in fruit wine. After dilution, it has a fruity aroma and produces the wine of strawberry, peach, and other fruit flavors [15,43]. Caproic acid produces the wine of fat and cheese flavor, and isobutyric acid is the wine of cheese flavor [44].
As shown in Table 4, esters, alcohols and acids, phenols, aldehydes, ketones, and other aroma compounds were examined in sugarcane wine. Although their content is low, they still contribute to the flavor of wine. 2-Methoxy-4-vinylphenol and 2,4-tert-butylphenol were found in all different types of sugarcane wine, which can produce the sugarcane wine of clove and sweet aromas. 2-octanone was detected in GJ01 and GJ02, which produce the sugarcane wine of fruity and floral aromas. Eugenol was detected in the GJ01 sample, which can produce the sugarcane wine of clove flavor [45].
The contribution of wine aroma compounds to the overall flavor depends not only on their content but also on the relative aroma threshold (ROAV) [28]. Therefore, the ROAV size is used to evaluate the contribution of VCs to the aroma of sugarcane wine. The aroma thresholds of 44 VCs in sugarcane wine were obtained from the literature [42,44], and the ROAV was calculated accordingly. Only the compounds with the threshold were analyzed, and the results are shown in Table 5.

Table 5.
Relative odor activity values of volatile compounds in different wines of sugarcane.
The compounds with larger ROAV values contribute more to the overall flavor of sugarcane wine. Compounds with 1 ≤ ROAV ≤ 100 are the key aroma compounds of sugarcane wine and compounds with 0.1 ≤ ROAV < 1 have an important modifying effect on the overall flavor of sugarcane wine [46]. As shown in Table 4, there are 23 substances with ROAV > 1 in 14 different varieties of sugarcane aroma compounds, including 13 esters, four alcohols, four acids, and two phenols, respectively. There are seven important compounds with ROAV > 10 that affect the flavor of sugarcane wine. There are 13 substances with 0.1 ≤ ROAV < 1, including four esters, seven alcohols, one acid, and one ketone.
Esters are the main compounds of the aroma of sugarcane wine. The main key aromas with ROAV > 10 are ethyl octanoate, ethyl hexanoate, ethyl acetate, ethyl decanoate, and isopentyl pentadecanoate. Research findings are similar to those of Munoz-Gonzalez et al. [47], Matijasevic et al. [48], and Ivic et al. [49]. The key aroma compounds in sugarcane wine are ethyl decanoate and ethyl octanoate, and there are three more esters, such as ethyl hexanoate, ethyl acetate, and isopentyl pentadecanoate, which produce sugarcane wine rich in fruity and floral aromas [45].
Alcohol modifies the effect of aromas. When the isoamyl alcohol content is appropriate, it has obvious grassy and plant aromas, increases the association of wine, and produces the mellow feeling of wine [50]. Its content in sugarcane wine is relatively high, but its aroma threshold is also higher(0.1 ≤ ROAV < 1), which modifies the flavor of sugarcane wine. 2-phenylethanol ROAV > 10 produces wine sweetness and rose and honey flavors, which helps to improve the quality of sugarcane wine [50].
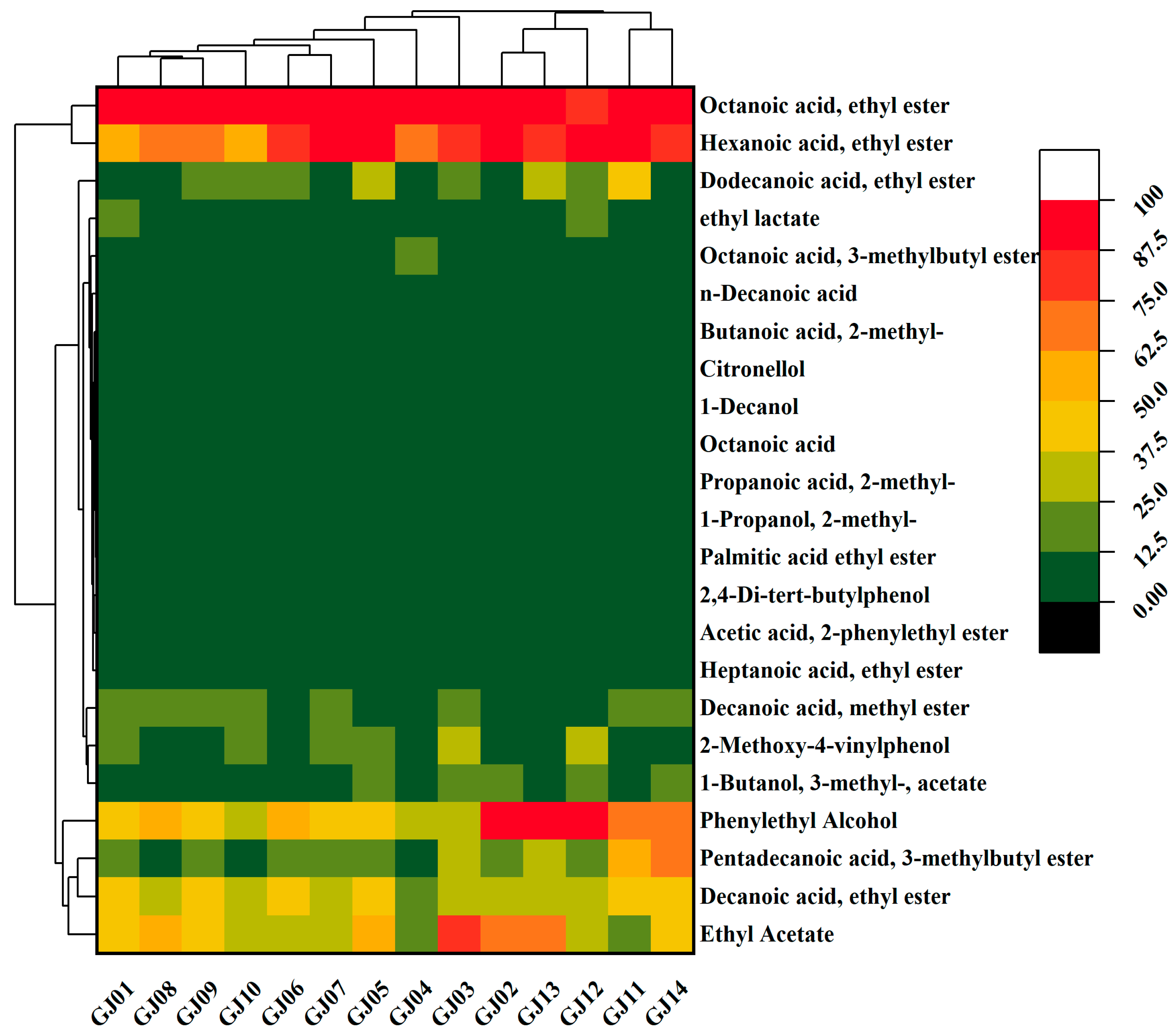
The heat map of 23 VCs with ROAV > 1 in different varieties of sugarcane wine is shown in Figure 3. The data are visualized, and the colors from green to red represent the increasing content of compounds. Among the main aroma compounds of sugarcane wine, esters have the optimum content, mainly ethyl octanoate, ethyl hexanoate, ethyl acetate, ethyl decanoate, etc., followed by 2-phenylethanol and 2,4-di-tert-butylphenol.
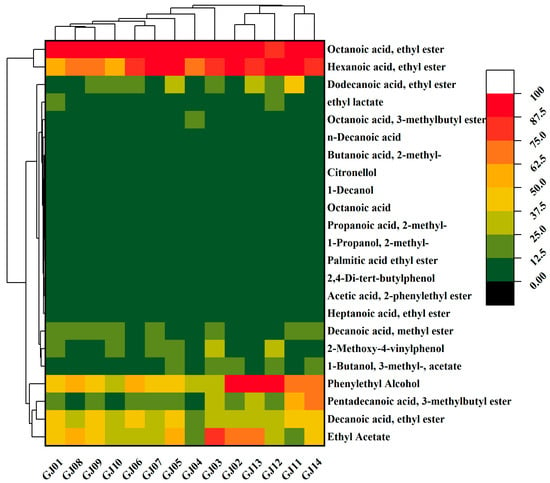
Figure 3.
Heat map of key volatile flavor compounds in sugarcane wines.
3.3. Screening of Markers for Differences in Volatile Compounds of Different Varieties of Sugarcane Wine
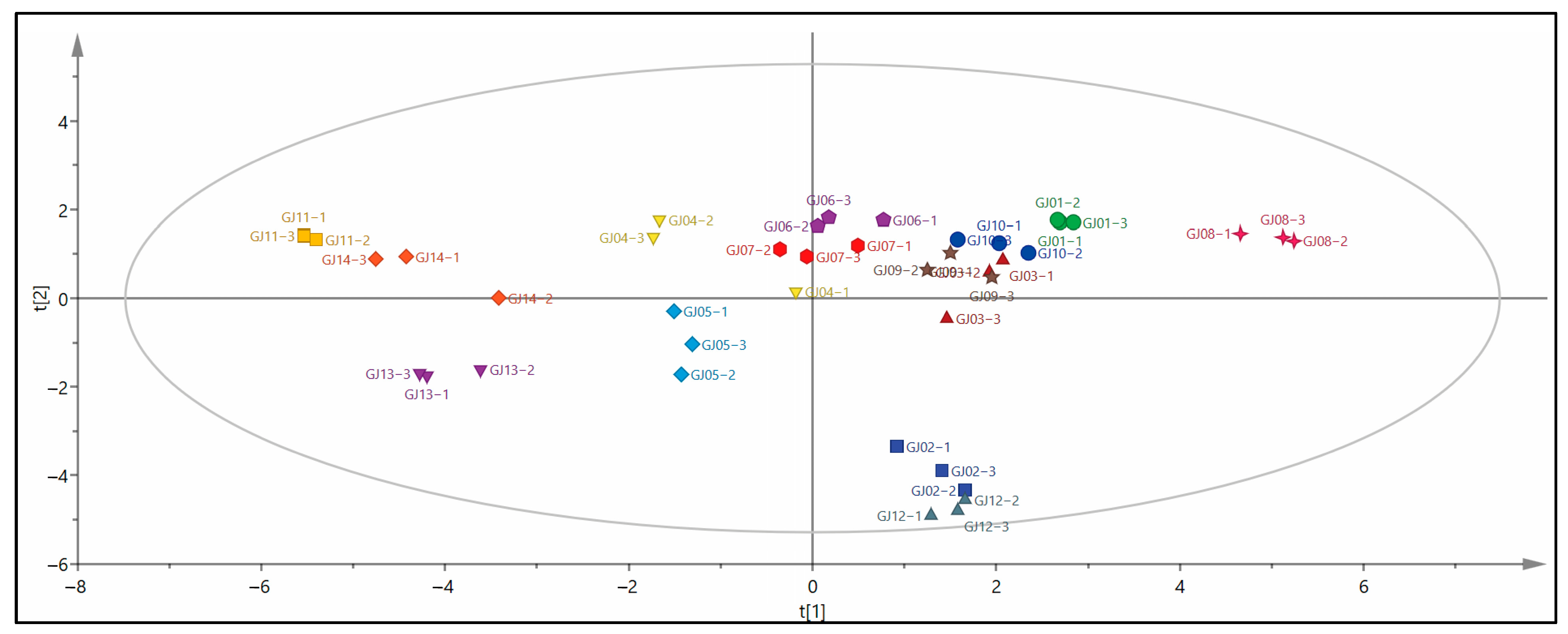
In order to explore the characteristic VCs of different varieties of sugarcane wine, the aroma compounds were used as dependent variables and different varieties of sugarcane wine were applied as independent variables. The OPLS-DA model was established to distinguish 14 types of sugarcane wine (Figure 4). All VCs of sugarcane wine are within the 95% Hotelling T2 confidence interval, indicating that the model is reliable and can be used as OPLS-DA model parameters for distinguishing sugarcane wines, and the model-related parameters R2X = 0.985, R2Y = 0.910, Q2 = 0.754, which meet the requirements of R2X − R2Y < 0.3 and Q2 > 0.5, indicating that the model is stable and has good predictive ability [7]. GJ08 is in the upper right corner of the first quadrant, GJ13 and GJ05 are in the third quadrant, GJ12 and GJ02 are in the fourth quadrant, GJ11 and GJ14 are in the second quadrant, there is a specific distance between them, they can be clearly distinguished, and the rest are distributed in the first quadrant and the center. It can be seen that there is a specific degree of distinction between different varieties of sugarcane wine and differences between samples. The distance between sugarcane wines in the same quadrant is relatively close, indicating that their aroma compounds have specific similarities.
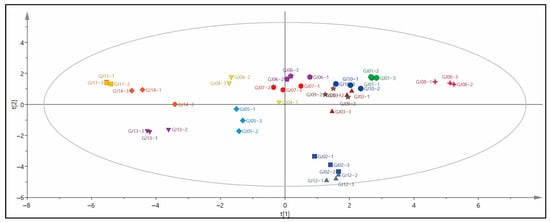
Figure 4.
OPLS-DA model scores of different sugarcane wines.
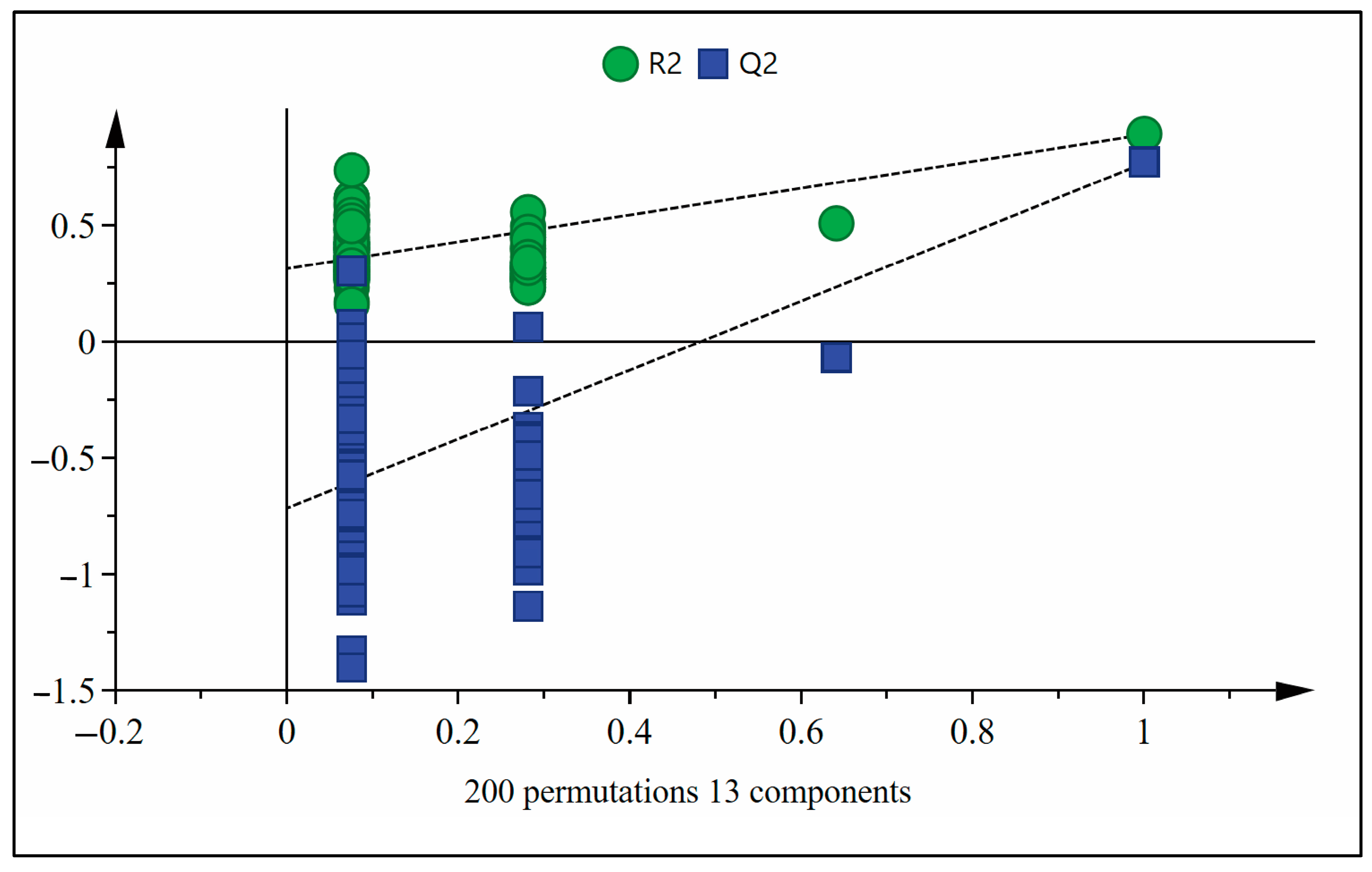
The OPLS-DA model was subjected to 200 permutation tests to verify whether the OPLS-DA model was overfitting. The verification results are shown in Figure 5. R2 = 0.313, Q2 = −0.716, the intercept of Q2 with the Y-axis was less than zero, and all Q2 values on the left were smaller than those on the right, indicating that the model fits the data well and is stable [51].
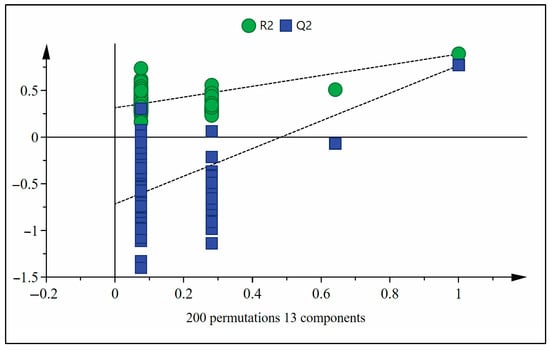
Figure 5.
OPLS-DA model permutation analysis on sugarcane wines.
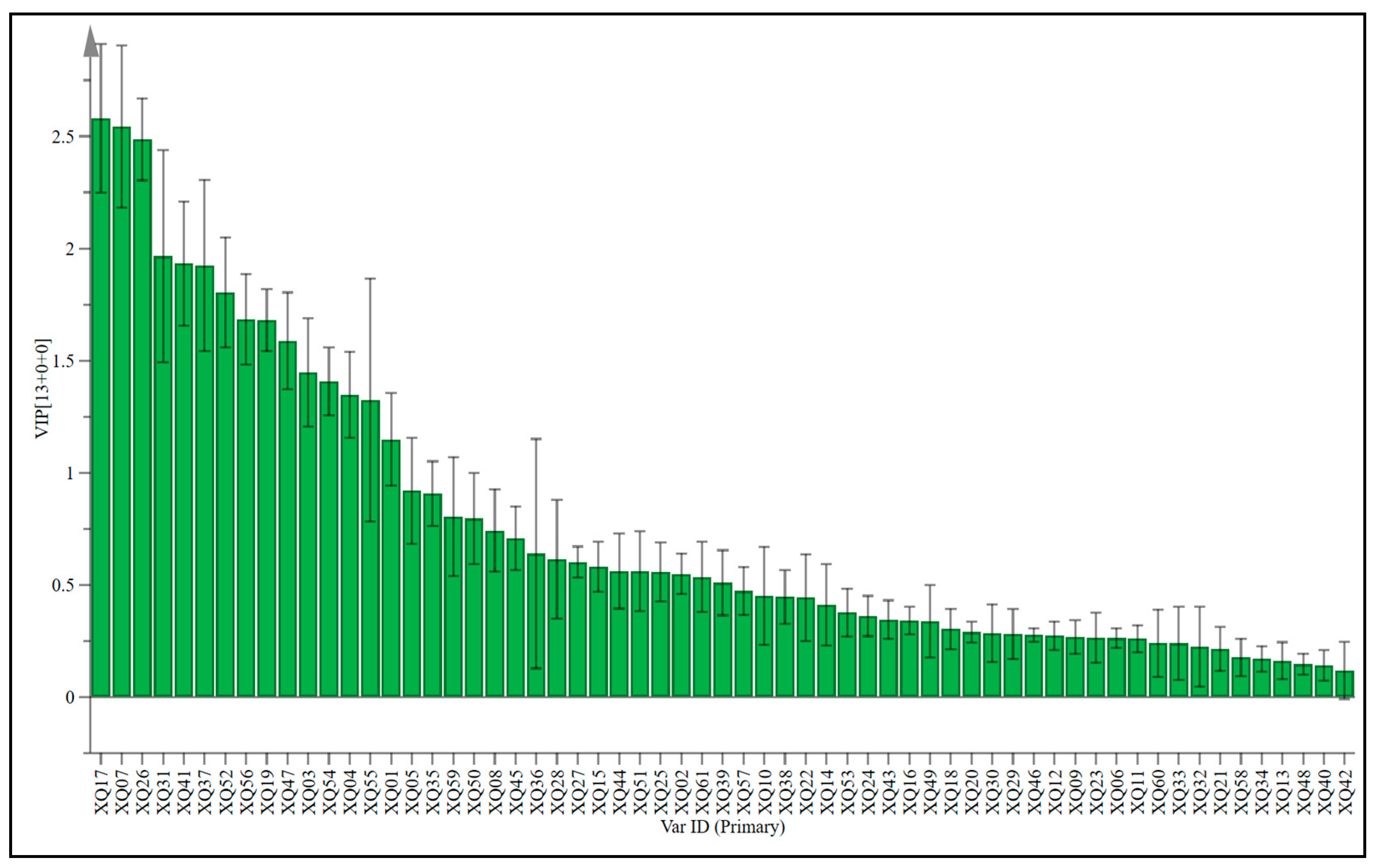
The weighted important variables (VIP) can provide an important reference for identifying variables that have a significant impact on classification. They are usually used to explain the contribution of variables to the model. Variables with a VIP value greater than 1 are important variables [39,52]. Using VIP > 1 and p < 0.05 as the screening criteria, 14 different varieties of sugarcane wine were screened for different aroma compounds (Figure 6 and Table 6). From the changes in their relative contents, it can be seen that the contents of 15 aroma compounds in different varieties of sugarcane wines are different to varying degrees. The main aroma compounds are Octanoic acid, ethyl ester, 1-Butanol, 3-methyl, Decanoic acid, ethyl ester, Ethyl 9-decenoate, Dodecanoic acid, ethyl ester, Palmitic acid ethyl ester, 2,4-Di-tert-butylphenol, Acetic acid, Octanoic acid, 2-Butanol, n-Decanoic acid, 1-Propanol, 2-methyl-, Ethyl 9-hexadecenoate, and Ethyl acetate. However, OPLS-DA and one-way ANOVA can be used to screen the above compounds to identify the characteristic differences in compounds of wines prepared from different cultivars of sugarcane.
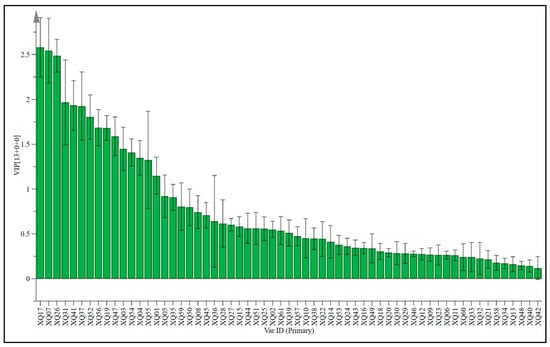
Figure 6.
OPLS-DA modeling of VIP on various sugarcane wines.

Table 6.
Influence of volatile compounds with significant differences (n = 3).
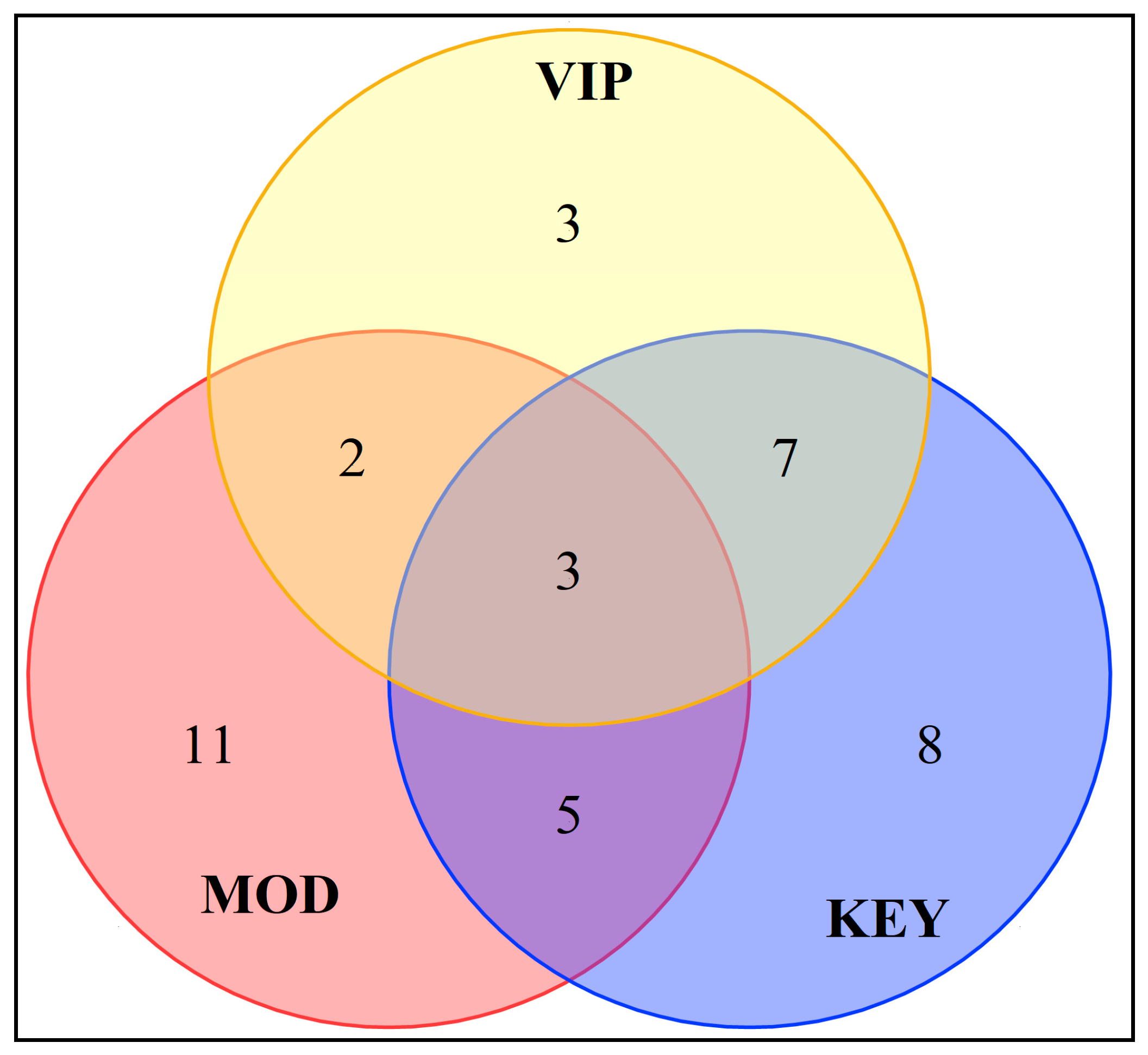
In order to further analyze the differential aroma compounds of different varieties of sugarcane wine, the 15 compounds with VIP values > 1 obtained by screening were intersected with the key and modifying aroma compounds, and a total of three aroma compounds were obtained, i.e., ethyl palmitate, isobutanol, and caprylic acid (Figure 7). These key characteristic aroma compounds can better identify and justify different varieties of sugarcane wine. Among the 14 types of sugarcane wine, the content of ethyl palmitate in GJ02 was the highest (305.794 mg/L) and the lowest (106.677 mg/L) in GJ08. The content of isobutanol in GJ01 was the highest (99.163 mg/L) and the lowest (9.769 mg/L) in GJ08. The caprylic acid in GJ14 was found to be the highest (209.563 mg/L) and the lowest (42.495 mg/L) in GJ02. Their contents were significantly different in different types of sugarcane wine. There are seven compounds with VIP values > 1 that intersect with the key aroma compounds, such as ethyl caprylate, ethyl caprate, ethyl acetate, ethyl laurate, n-decanoic acid, 2,4-di-tert-butylphenol, and 2-phenylethanol. These compounds are the key characteristic aroma compounds of different varieties of sugarcane wine.
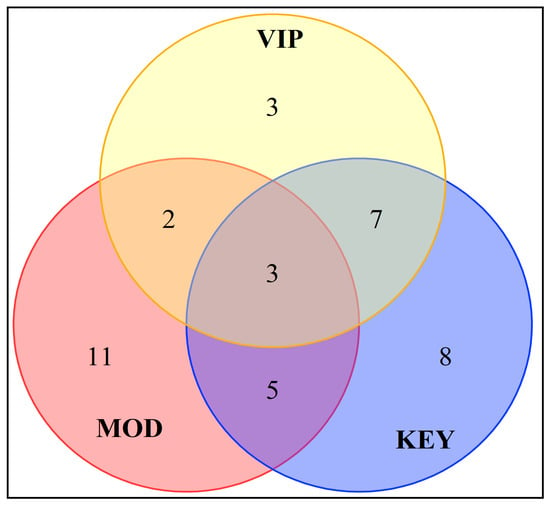
Figure 7.
Venn diagram of key and modifying aroma and volatile components with VIP > 1.
3.4. Electronic Nose Analysis of the Aroma of Sugarcane Wine Develop from Different Sugarcane Cultivars
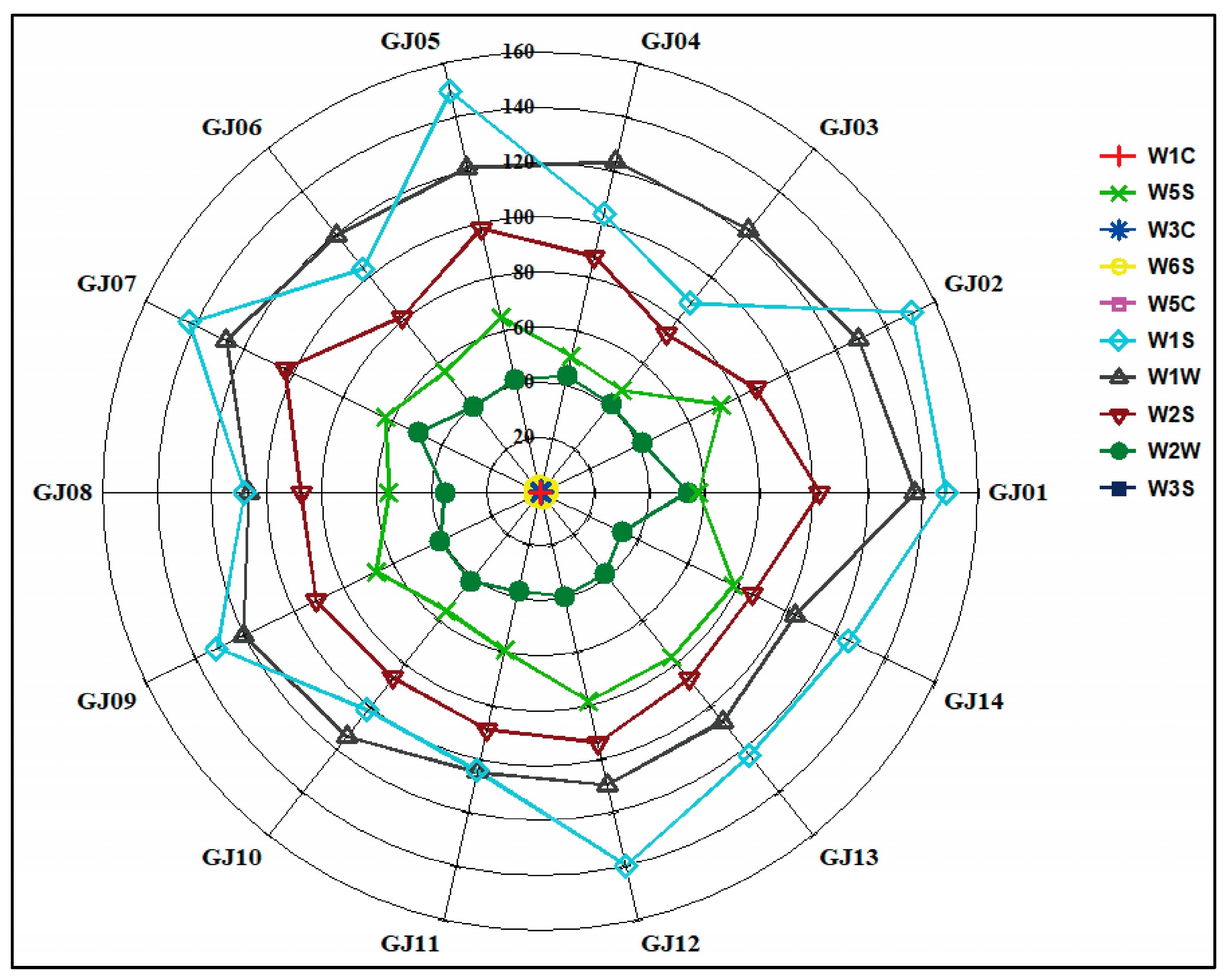
In order to further explore the differences and distribution characteristics of aroma compounds in sugarcane wine produced from different sugarcane cultivars, a highly sensitive electronic nose was applied to conduct the research analysis. Based on the response values of different sugarcane wines on 10 sensors of the electronic nose, the radar fingerprint of sugarcane wines is shown in Figure 8.
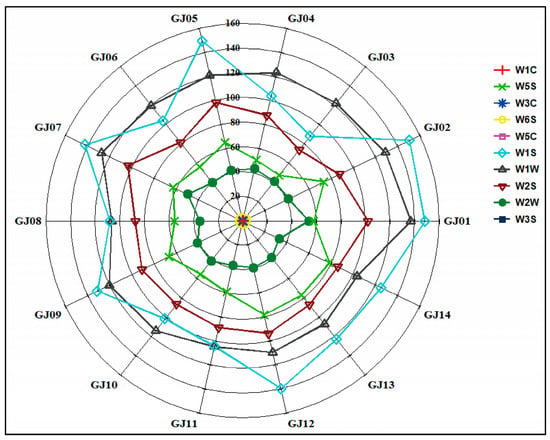
Figure 8.
Influence of radar plot of relative aroma intensity on different cultivars of sugarcane wines.
The response intensity of the electronic nose sensors W1S, W1W, W2S, W5S, and W1W to the 14 sugarcane wines are significantly higher than that of other sensors, and the difference is reliable. The response intensity of the sensors W5C, W1C, W3C, W3S, and W6S to the 14 sugarcane wines is low, and the difference is not reliable. Analysis showed that the aroma differences of the 14 sugarcane wines are mainly focused on the aromatics, methyls, alcohols, aldehydes and ketones, nitrogen oxides, and organic sulfides, while the difference in aroma between hydrides, alkanes, ammonia, and aromatic compounds are not reliable. These findings are consistent with the GC-MS analysis, which shows that the main aroma compounds are esters, alcohols, aldehydes, and ketones (Figure 8). The response value of the aroma compounds on the corresponding sensors can be used as an indicator for identifying the volatile odor compounds of different sugarcane wines.
Principal Component Analysis of Different Varieties of Sugarcane Wine
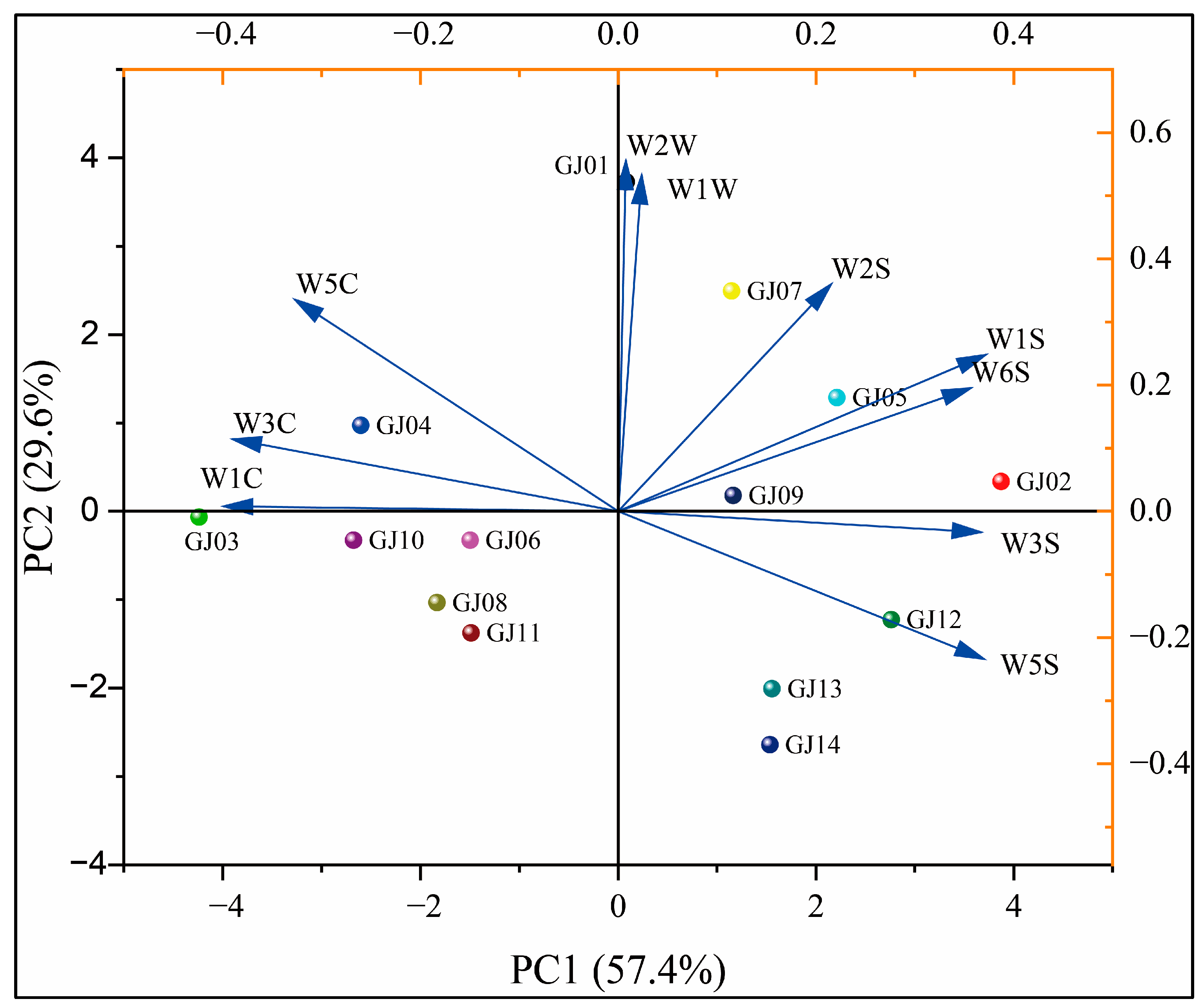
In order to further analyze the differences in the aroma of different varieties of sugarcane wines by each sensor of the electronic nose, principal component analysis (PCA) was applied for the response values of 14 types of sugarcane wines on 10 sensors of the electronic nose (Figure 9). The results showed that the information of principal component analysis is mainly focused on the first two principal components, with the variance contribution rate of the first principal component being 57.4%. The variance contribution rate of the second principal component is 29.6%, and the cumulative contribution rate is 87.0%. This main information can reflect the differences in the overall aroma between the different types of sugarcane wine [35].
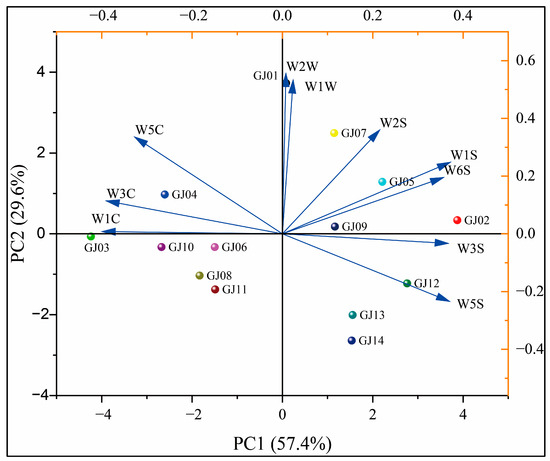
Figure 9.
Principal component analysis of wine aroma on different sugarcane cultivars.
As shown in Figure 9, the distances between GJ01, GJ02, and GJ03 and other fruit wines are relatively large, indicating that they have reliable differences in aroma from other fruit wines. The distances between GJ08 and GJ11 are relatively close, indicating that there is a specific similarity in the aroma between these two sugarcane wines. From PC1, GJ13 and GJ14 overlap and cannot be well distinguished, but they can be well distinguished through PC2. Overall, the distances between the different types of sugarcane wines are relatively scattered, and there is no overlap. Principal component analysis can determine that there are differences in aroma between different varieties of sugarcane wine. It can be seen that W1C has a positive effect on GJ03 and a negative effect on GJ02, indicating that the ester content of GJ03 is high and the ester content of GJ02 is low. The results are consistent with the VCs results [53].
3.5. Analysis of Sugarcane Wine Taste by Electronic Tongue
Taste is one of the main indicators for assessing the quality of fruit wine. An electronic tongue is one of the commonly used methods for taste assessment due to its accuracy, objectivity, and high sensitivity [54,55].
3.5.1. Analysis of Fruit Wine Taste Prepared from Different Cultivars of Sugarcane
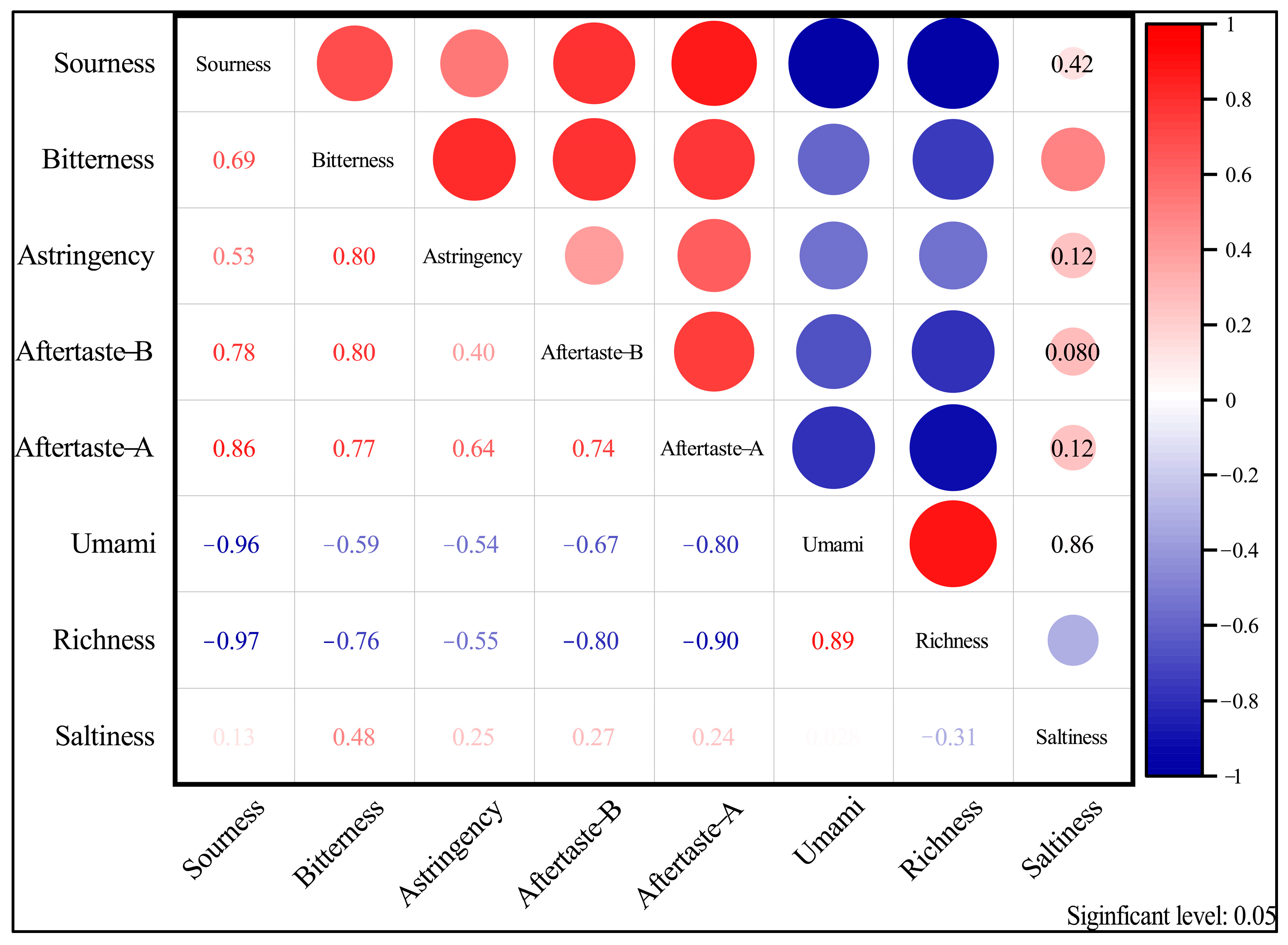
The electronic tongue was used to analyze the differences and correlations of the five basic tastes sourness, bitterness, astringency, umami, and saltiness, and three aftertastes of fruit wine produced from different cultivars of sugarcane as aftertaste A (aftertaste of astringency), aftertaste B (aftertaste of bitterness), and abundance (aftertaste of umami). The results are shown in Figure 10.
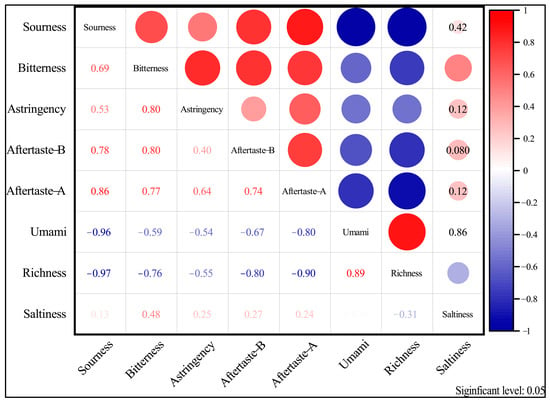
Figure 10.
Coherence analysis of the response value of each electronic tongue taste indicator on a variety of wines (p < 0.05).
As shown in Figure 10, except for saltiness, there was no significant difference in the taste, aftertaste B, and astringency of different varieties of sugarcane wine (p > 0.05). Sourness, bitterness, astringency, umami, aftertaste A, aftertaste B, and abundance data were significantly different in the taste of different varieties of sugarcane wine (p < 0.05). Based on the response values of different sugarcane wines on eight sensors of the electronic tongue, the radar fingerprint of sugarcane wines is shown in Figure 10.
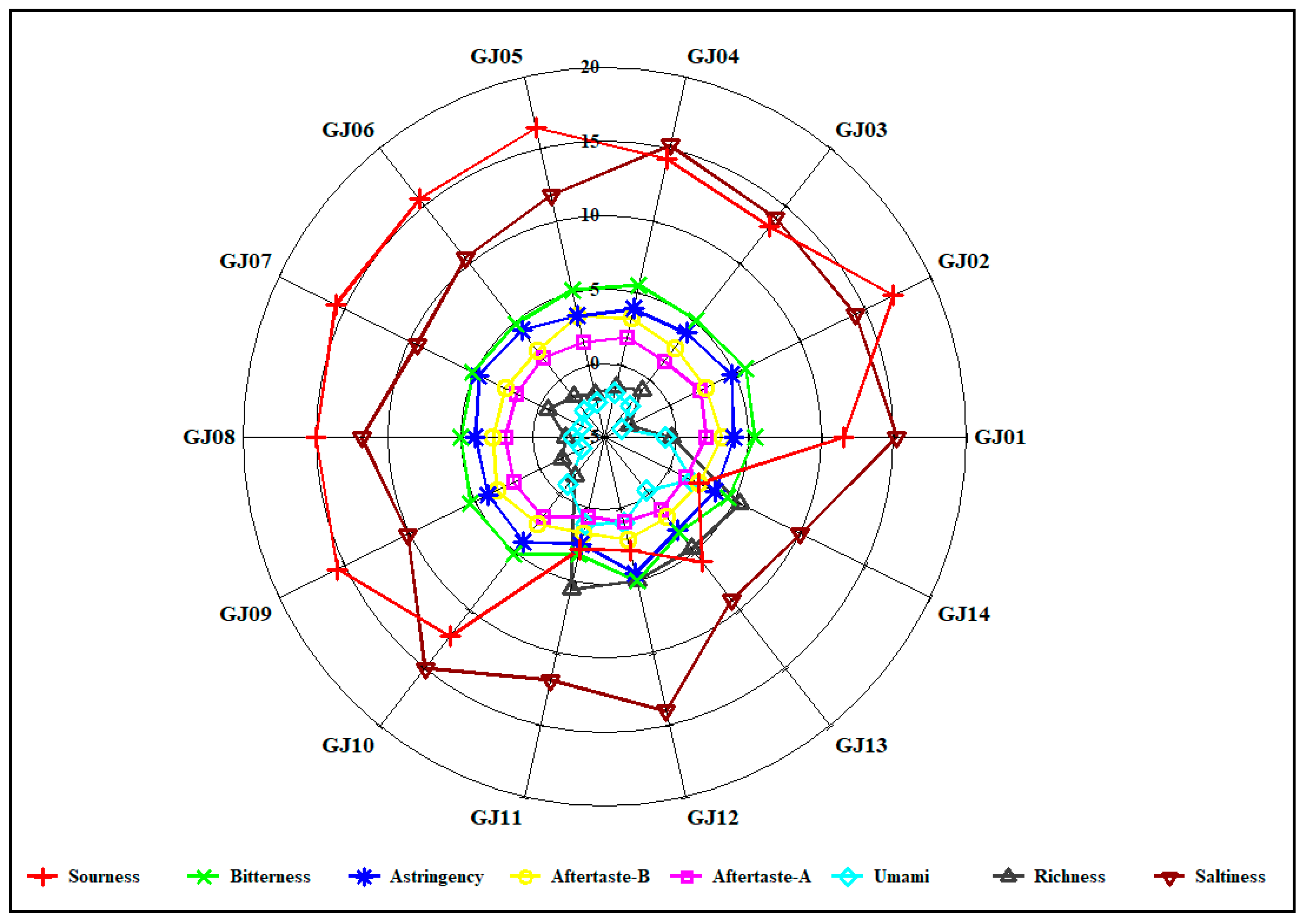
As shown in Figure 11, the differences in the three electronic tongue taste indices of sourness, saltiness, and astringency among the 14 sugarcane wines are obvious, while the corresponding intensities of the five electronic tongue taste indices of aftertaste A, aftertaste B, richness, bitterness, and umami are limited and the differences are not reliable. Therefore, only using the response intensity of the electronic tongue taste index to analyze the difference in sugarcane wines cannot completely distinguish the taste differences between different varieties of sugarcane wine.
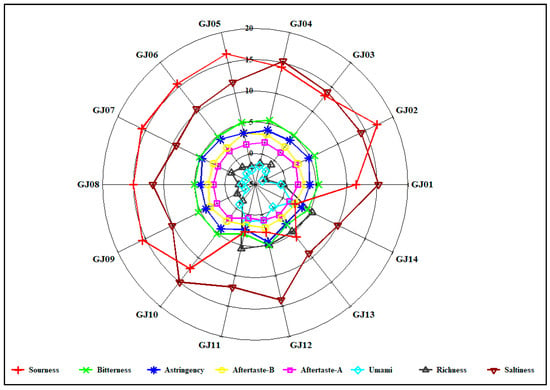
Figure 11.
Radar plot of relative taste intensity on different sugarcane wines.
3.5.2. Principal Component Analysis of the Electronic Tongue
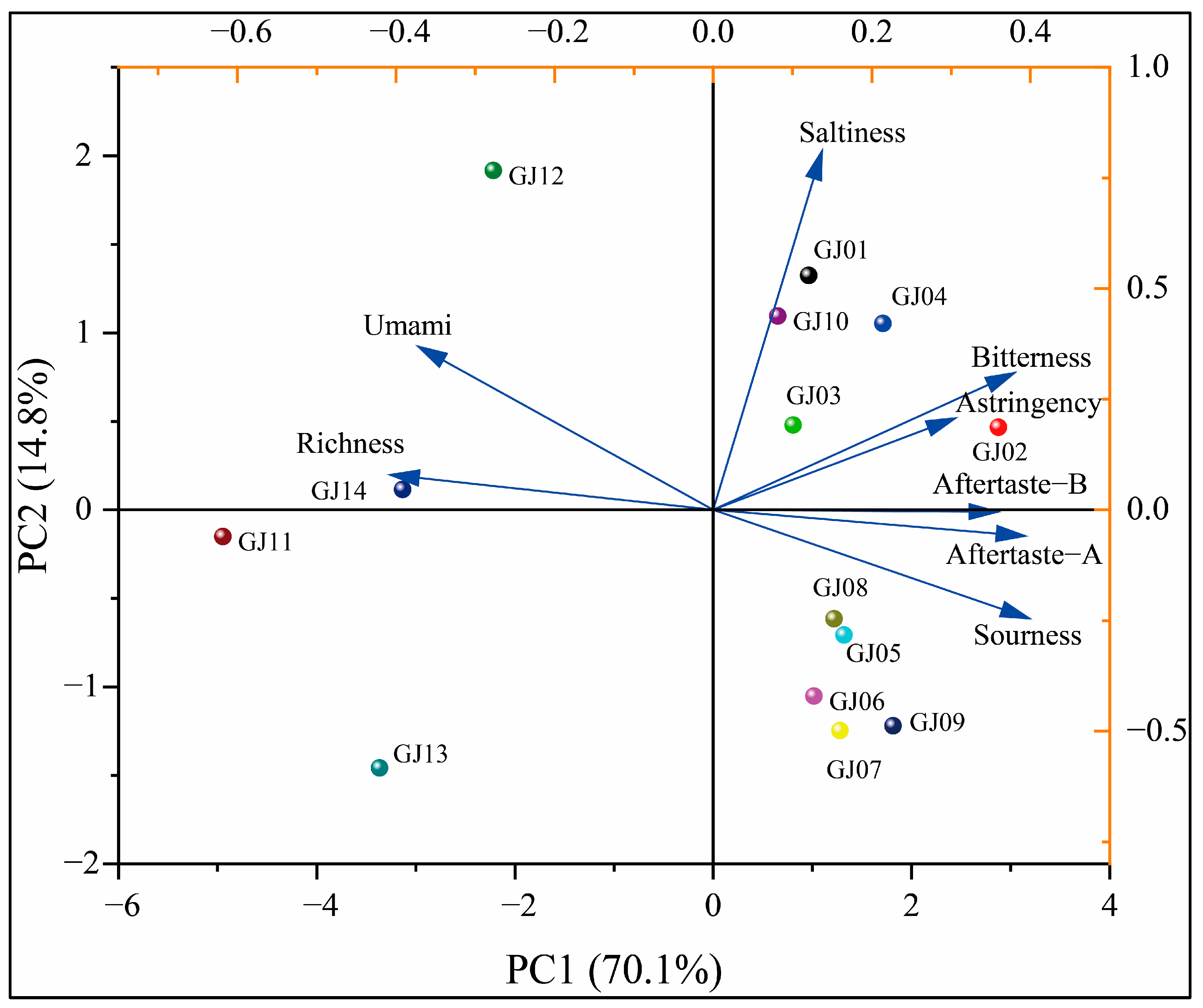
In order to further analyze the differences in the taste of different varieties of sugarcane wine, PCA was used to assess the differences in the taste of different varieties of wine. The principal component analysis and loading diagram are shown in Figure 12.
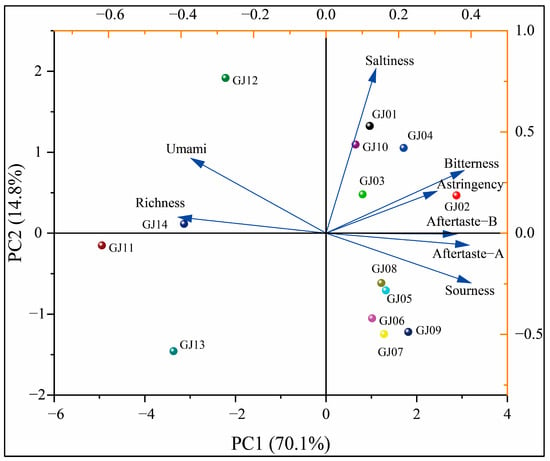
Figure 12.
Principal component analysis of taste on different cultivars of sugarcane wines.
As shown in Figure 12, PCA showed that the sum of the cumulative contribution rate of PC1 and PC2 is 84.9%, the recognition index exceeds 80%, and the 14 different varieties of sugarcane wine are relatively uniform in the PCA without cross-overlapping. However, the electronic tongue can distinguish the taste differences of different varieties of sugarcane wine. GJ02, GJ12, GJ11, and GJ13 are distributed in different quadrants and far away from other wines, indicating that they have reliable taste differences from other wines. GJ08 and GJ05 are close and in the same quadrant, indicating that there is a certain similarity in the taste between these two sugarcane wines. The loading chart showed that the GJ12 and GJ14 have outstanding umami and richness. GJ01 and GJ10 have outstanding saltiness, and GJ11 has the lowest bitterness, which is consistent with the results of the radar chart analysis. Combined with the above analysis and assessment of the response intensity values of different varieties of sugarcane wine by electronic tongue and PCA, it can be seen that there are differences in the flavor substances of different varieties of sugarcane wine, indicating that the electronic tongue can be used to distinguish different types of sugarcane wine.
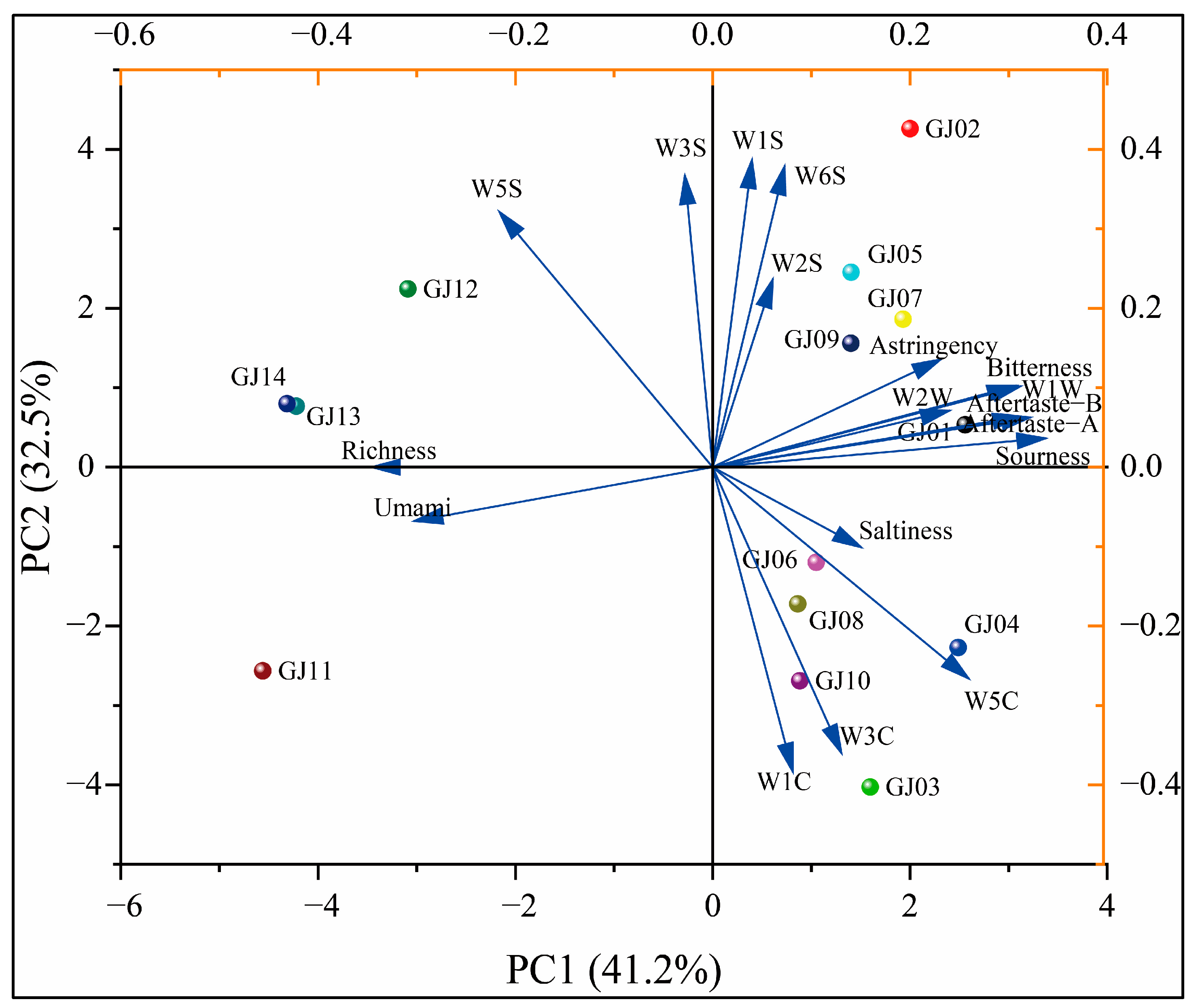
3.6. Principal Component Analysis of Aroma and Flavor of Sugarcane Wine
In order to further analyze the differences and correlations of aroma and flavor of different varieties of sugarcane wine, PCA was applied to assess the differences in aroma and flavor of different varieties of wine.
As shown in Figure 13, the PCA showed that the characteristics are mainly focused on the first two principal components. The variance contribution rate of the first principal component is 41.2%, the variance contribution rate of the second principal component is 32.5%, and the cumulative contribution rate is 73.7%. This main information can reflect the differences in the overall aroma between the different sugarcane wines. The 14 different varieties of sugarcane wines are relatively scattered in the PCA, except that GJ13 and GJ14 are relatively close and in the same quadrant, indicating that the aroma and taste of these two sugarcane wines are similar. GJ11 is alone in the third quadrant, and its loading diagram is positive with umami and richness, indicating that its aroma and taste are different from others, and the umami is prominent. GJ01 has prominent sourness and bitterness, and the aroma of organic sulfur and aldehyde ketone compounds are prominent. Other fruit wines are more evenly distributed in the second and fourth quadrants and can be better distinguished.
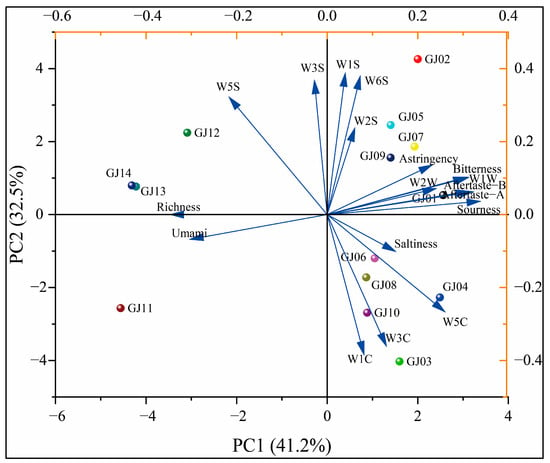
Figure 13.
Principal component analysis of aroma and taste profile on different cultivars of sugarcane wine and flavor.
4. Conclusions
This study used headspace solid-phase microextraction–gas chromatography–mass spectrometry (HS-SPME-GC-MS) combined with the electronic nose and electronic tongue detection technology to analyze the physical and chemical parameters, taste, and aroma compounds of fruit wine developed from sugarcane juice of 14 main sugarcane varieties in Guangxi province, China. By establishing the orthogonal partial least squares discriminant analysis (OPLS-DA) discriminant model, relative odor activity value (ROAV), and principal component analysis combined with multivariate statistical methods, the difference analysis of flavor substances of different cultivars of sugarcane was carried out, and the characteristics of key aroma compounds in sugarcane wine were characterized. The results showed that a total of 61 main aroma compounds were found in different types of sugarcane wines. The types and contents of VCs of different sugarcane wines were significantly different (p < 0.05). Based on the OPLS-DA analysis, the fifteen potential VCs with significant differences were screened by VIP values > 1.0, and the flavor of the different sugarcane wines was differentially analyzed with the combination of ROAV. The results showed that the 14 types of sugarcane wines contained seven key aroma substances, i.e., ethyl caprylate, ethyl caprate, ethyl acetate, ethyl laurate, 2-phenylethanol, 2,4-di-tert-butylphenol, and n-decanoic acid, which are the main aroma sources of sugarcane wine. Ethyl palmitate, isobutanol, and caprylic acid were determined with significant differences. The research results revealed the different aroma and key aroma compounds for different varieties of sugarcane wine. The differences in aroma and taste substances between different varieties of sugarcane wine were consistent with the results of GC-MS analysis of VCs of wine, which can be used to distinguish different varieties of sugarcane wine. The present research findings can provide technical references for the selection of sugarcane cultivars for sugarcane wine processing and the improvement of wine quality, as well as theoretical knowledge and data support for the development of sugarcane industries and high-value-added products in years to come.
Author Contributions
Conceptualization, Y.Y., J.C., F.Z. and G.C.; methodology, Y.Y., J.C., B.L. and F.W.; writing—original draft preparation, Y.Y., J.C. and B.L.; writing—review and editing, F.Z., K.K.V. and G.C.; electrochemical part, Y.Y., J.C., B.L. and F.W.; analytical part, J.C., F.W. and K.K.V.; Project management, Funding and Supervision, F.Z. and G.C. All authors have read and agreed to the published version of the manuscript.
Funding
This study was financially supported by the Guangxi Major Science and Technology Program (GK-AA22117015-3), the earmarked fund for China Agriculture Research System Guangxi Innovation Team—Specialty Fruilt (nycytxgxcxtd-2024-17), the Guangxi Academy of Agricultural Sciences Basic Research Business Project (GNK2021YT117).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available within the article.
Acknowledgments
The authors would like to thank the Guangxi Subtropical Crops Research Institute and the Guangxi Academy of Agricultural Sciences, Nanning, Guangxi, China, for providing the necessary facilities for this study.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Luo, T.; Lakshmanan, P.; Zhou, Z.; Deng, Y.; Deng, Y.; Yang, L.; Huang, D.; Song, X.; Liu, X.; Cong, W.F.; et al. Sustainable sugarcane cropping in China. Front. Agric. Sci. Eng. 2022, 9, 272–283. [Google Scholar] [CrossRef]
- Chen, G.L.; Zheng, F.J.; Lin, B.; Yang, Y.X.; Fang, X.C.; Verma, K.K.; Yang, L.F. Vinegar: A potential source of healthy and functional food with special reference to sugarcane vinegar. Front. Nutr. 2023, 10, 1145862. [Google Scholar] [CrossRef] [PubMed]
- Canton, H. Food and agriculture organization of the United Nations—FAO. In The Europa Directory of International Organizations; Routledge: London, UK, 2021; pp. 297–305. [Google Scholar] [CrossRef]
- Talukdar, D.; Verma, D.K.; Malik, K.; Mohapatra, B.; Yulianto, R. Sugarcane as a Potential Biofuel Crop. In Sugarcane Biotechnology: Challenges and Prospects; Mohan, C., Ed.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Wang, B.S.; Chang, L.W.; Kang, Z.C.; Chu, H.L.; Tai, H.M.; Huang, M.H. Inhibitory effects of molasses on mutation and nitric oxide production. Food Chem. 2011, 126, 1102–1107. [Google Scholar] [CrossRef]
- Ali, S.E.; Yuan, Q.; Wang, S.; Farag, M.A. More than sweet: A phytochemical and pharmacological review of sugarcane (Saccharum officinarum L.). Food Biosci. 2021, 44, 101431. [Google Scholar] [CrossRef]
- Wang, Y.; He, T.; Wang, J.; Wang, L.; Ren, X.; He, S.; Liu, X.; Dong, Y.; Ma, J.; Song, R.; et al. High performance liquid chromatography fingerprint and headspace gas chromatography-mass spectrometry combined with chemometrics for the species authentication of Curcumae Rhizoma. J. Pharm. Biomed. Anal. 2021, 202, 114144. [Google Scholar] [CrossRef]
- Zheng, F.-J.; Lin, B.; Yang, Y.-X.; Fang, X.-C.; Verma, K.K.; Chen, G.-L. Efficacy and functionality of sugarcane original vinegar on mice. Front. Microbiol. 2023, 14, 1224666. [Google Scholar] [CrossRef]
- Duarte-Almeida, J.M.; Salatino, A.; Genovese, M.I.; Lajolo, F.M. Phenolic composition and antioxidant activity of culms and sugarcane (Saccharum officinarum L.) products. Food Chem. 2011, 125, 660–664. [Google Scholar] [CrossRef]
- Chen, G.L.; Zheng, F.J.; Lin, B.; Wang, T.S.; Li, Y.R. Preparation and characteristics of sugarcane low alcoholic drink by submerged alcoholic fermentation. Sugar Tech 2013, 15, 412–416. [Google Scholar] [CrossRef]
- Tang, L.; Jin, C.; Tang, J.; Li, J.; Liu, D.; Zou, Q. Optimization of fermentation condition for mixed bacteria of lemon-cane wine. Food Ferment. Sci. Technol. 2022, 58, 108–112. [Google Scholar]
- Chen, G.L.; Zheng, F.J.; Sun, J.; Li, Z.C.; Lin, B.; Li, Y.R. Production and characteristics of high quality vinegar from sugarcane juice. Sugar Tech 2015, 17, 89–93. [Google Scholar] [CrossRef]
- Chen, G.L.; Zheng, F.J.; Lin, B.; Lao, S.B.; He, J.; Huang, Z.; Zeng, Y.; Sun, J.; Verma, K.K. Phenolic and Volatile Compounds in the Production of Sugarcane Vinegar. ACS Omega 2020, 5, 30587–30595. [Google Scholar] [CrossRef] [PubMed]
- Lohan, V.; Pawar, K.; Kumari, A.; Gehlot, R. Optimization of fermentation factors for vinegar preparation from sugarcane jaggery using response surface methodology. Asian J. Dairy Food Res. 2022, 43, 441–447. [Google Scholar] [CrossRef]
- Ma, Y.; Meng, A.; Liu, P.; Chen, Y.; Yuan, A.; Dai, Y.; Ye, K.; Yang, Y.; Wang, Y.; Li, Z. Reflux extraction optimization and antioxidant activity of phenolic compounds from Pleioblastus amarus (Keng) shell. Molecules 2021, 27, 362. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Li, B.; Liao, M.; Liu, J.; Zhou, Y.; Liang, Y.; Yuan, H.; Li, K.; Li, H. The physicochemical attributes, volatile compounds, and antioxidant activities of five plum cultivars in Sichuan. Foods 2022, 12, 3801. [Google Scholar] [CrossRef]
- Huang, Y.; Xu, M.; Li, J.; Chen, K.; Xia, L.; Wang, W.; Ren, P.; Huang, X. Ex vivo to in vivo extrapolation of syringic acid and ferulic acid as grape juice proxies for endothelium-dependent vasodilation: Redefining vasoprotective resveratrol of the French paradox. Food Chem. 2021, 363, 130323. [Google Scholar] [CrossRef]
- Sui, X.; Zhu, Z.; Cheng, F.; Sun, Y.; Jiang, X. Evaluation of physicochemical properties, antioxidant capacity, and volatile flavor compounds during the wine fermentation of Mei (Prunus mume) fruit. LWT 2024, 201, 116177. [Google Scholar] [CrossRef]
- Chen, G.L.; Lin, B.; Zheng, F.J.; Fang, X.C.; Yang, Y.X.; Verma, K.K.; Yang, L.F. Assessment of acid production efficiency and aroma volatile compounds by immobilized fermentation of different carriers on sugarcane original vinegar. LWT-Food Sci. Technol. 2024, 203, 116285. [Google Scholar] [CrossRef]
- Chen, G.L.; Chen, J.; Lin, B.; Zhao, L.Z.; Zheng, F.J.; Verma, K.K.; Li, Y.R. Research overview on sugarcane deep processing and comprehensive utilization in China. Sugar Tech 2024, 26, 982–991. [Google Scholar] [CrossRef]
- Xiang, N.; Chang, X.; Qin, L.; Li, K.; Wang, S.; Guo, X. Insights into tissue-specific anthocyanin accumulation in Japanese plum (Prunus salicina L) fruits: A comparative study of three cultivars. Food Chem. Mol. Sci. 2023, 7, 100178. [Google Scholar] [CrossRef]
- Wang, L.; Wang, P.; Deng, W.; Cai, J.; Chen, J. Evaluation of aroma characteristics of sugarcane (Saccharum officinarum L.) juice using gas chromatography-mass spectrometry and electronic nose. Lebensm. Wiss. Technol. (LWT) 2019, 108, 400–406. [Google Scholar] [CrossRef]
- CNSM Committee. GB/T 15038-2006; General Analysis Methods for Wine and Fruit Wine. China Standard Press: Beijing, China, 2006.
- GB/T 12456-2021; Determination of Total Acid in Foods. Standards Press of China: Beijing, China, 2021.
- Xiao, Z.; Fang, L.; Niu, Y.; Yu, H. Effect of cultivar and variety on phenolic compounds and antioxidant activity of cherry wine. Food Chem. 2015, 186, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Joshna, K.; Gopal, V.; Kavitha, B. Analysis of Bitter honey using gas chromatography and Tandem Mass Spectrometry. Bioinformation 2022, 18, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yuan, L.; Liu, H.; Liu, H.; Zhou, Y.; Li, M.; Gao, R. Analysis of the changes of volatile flavor compounds in a traditional Chinese shrimp paste during fermentation based on electronic nose, SPME-GC-MS and HS-GC-IMS. Food Sci. Hum. Wellness 2022, 12, 173–182. [Google Scholar] [CrossRef]
- Zhu, Y.; Chen, J.; Chen, X.; Chen, D.; Deng, S. Use of relative odor activity value (ROAV) to link aroma profiles to volatile compounds: Application to fresh and dried Eel (Muraenesox Cinereus). Int. J. Food Prop. 2020, 23, 2257–2270. [Google Scholar] [CrossRef]
- Chen, J.; Lin, B.; Zheng, F.J.; Fang, X.C.; Ren, E.F.; Wu, F.F.; Verma, K.K.; Chen, G.L. Characterization of the pure black tea wine fermentation process by electronic nose and tongue-based techniques with nutritional characteristics. ACS Omega 2023, 8, 12538–12547. [Google Scholar] [CrossRef]
- Wu, F.; Lin, B.; Chen, J.; Zheng, F.; Fang, X.; Luo, L.; Chen, H.; Verma, K.K.; Chen, G. Characteristic aroma screening among green tea varieties and electronic sensory evaluation of green tea wine. Fermentation 2024, 10, 449. [Google Scholar] [CrossRef]
- NY/T 1508-2017; Determination of Green Food-Fruit Wine. Standard Press of China: Beijing, China, 2017.
- Tarko, T.; Kostrz, M.; Duda-Chodak, A.; Semik-Szczurak, D.; Sroka, P.; Senczyszyn, T. The effect of apple cultivars and yeast strains on selected quality parameters and antioxidant activity of fermented apple beverages. CyTA–J. Food 2018, 16, 892–900. [Google Scholar] [CrossRef]
- Zhang, Q.; Ma, Y.; Wan, F.; Cai, Z.; Zeng, R.; Tang, J.; Nie, X.; Jiang, X.; Zhu, C.; Laghi, L. Comprehensive comparison of flavor and metabolomic profiles in kiwi wine fermented by kiwifruit flesh with different colors. LWT 2024, 208, 116719. [Google Scholar] [CrossRef]
- Robles, A.; Fabjanowicz, M.; Chmiel, T.; Płotka-Wasylka, J. Determination and identification of organic acids in wine samples. Problems and challenges. TrAC Trends Anal. Chem. 2019, 120, 115630. [Google Scholar] [CrossRef]
- Li, P.; Jia, Y.; Cai, D.; Wang, X.; Liu, J.; Zhu, R.; Wang, Z.; He, Y.; Wen, L. Study on the relationship between flavor components and quality of ice wine during freezing and brewing of ‘beibinghong’ grapes. Food Chem. X 2023, 20, 101016. [Google Scholar] [CrossRef]
- Chen, G.; Yuan, Y.; Tang, S.; Yang, Z.; Wu, Q.; Liang, Z.; Chen, S.; Li, W.; Lv, X.; Ni, L. Comparative analysis of microbial communities and volatile flavor components in the brewing of Hongqu rice wines fermented with different starters. Curr. Res. Food Sci. 2022, 7, 100628. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Ji, M.; Gong, J.; Chitrakar, B. The formation of volatiles in fruit wine process and its impact on wine quality. Appl. Microbiol. Biotechnol. 2024, 108, 420. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, E.R.; Caliari, M.; Soares Júnior, M.S.; Oliveira, A.R.; Marques Duarte, R.C.; Boas, B.V. Assessment of chemical and sensory quality of sugarcane alcoholic fermented beverage. J. Food Sci. Technol. 2017, 55, 72–81. [Google Scholar] [CrossRef]
- Hou, Z.; Wei, Y.; Sun, L.; Xia, R.; Xu, H.; Li, Y.; Feng, Y.; Fan, W.; Xin, G. Effects of drying temperature on umami taste and aroma profiles of mushrooms (Suillus granulatus). J. Food Sci. 2022, 87, 1983–1998. [Google Scholar] [CrossRef] [PubMed]
- Lee, S. Quality characteristics and antioxidant activities of six types of Korean white wine. Foods 2022, 12, 3246. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, M.G.; Pretorius, I.S. Yeast and its importance to wine aroma—A review. S. Afr. J. Enol. Vitic. 2000, 21, 97–129. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, Z.; Lu, X.; Yi, Y.; Tian, Q.; Deng, J.; Jiang, D.; Tang, J.; Laghi, L. Effects of Saccharomyces cerevisiae strains on the metabolomic profiles of Guangan honey pear cider. LWT 2023, 182, 114816. [Google Scholar] [CrossRef]
- Lv, Z.; Liu, H.; Yang, W.; Zhang, Q.; Chen, D.; Jiao, Z.; Liu, J. Comprehensive analysis of physicochemical properties and volatile compounds in different strawberry wines under various pre-treatments. Molecules 2024, 29, 2045. [Google Scholar] [CrossRef]
- Zhao, D.; Shi, D.; Sun, J.; Li, A.; Sun, B.; Zhao, M.; Chen, F.; Sun, X.; Li, H.; Huang, M.; et al. Characterization of key aroma compounds in Gujinggong Chinese Baijiu by gas chromatography–olfactometry, quantitative measurements, and sensory evaluation. Food Res. Int. 2018, 105, 616–627. [Google Scholar] [CrossRef]
- Wang, L.; Wu, L.; Xiang, D.; Huang, H.; Han, Y.; Zhen, P.; Shi, B.; Chen, S.; Xu, Y. Characterization of key aroma compounds in aged Qingxiangxing baijiu by comparative aroma extract dilution analysis, quantitative measurements, aroma recombination, and omission studies. Food Chem. 2023, 419, 136027. [Google Scholar] [CrossRef]
- Lan, T.; Wang, J.; Yuan, Q.; Lei, Y.; Peng, W.; Zhang, M.; Li, X.; Sun, X.; Ma, T. Evaluation of the color and aroma characteristics of commercially available Chinese kiwi wines via intelligent sensory technologies and gas chromatography-mass spectrometry. Food Chem. X 2022, 15, 100427. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Gonzalez, C.; Perez-Jiménez, M.; Criado, C.; Pozo-Bayon, M.A. Effects of ethanol concentration on oral aroma release after wine consumption. Molecules 2019, 24, 3253. [Google Scholar] [CrossRef] [PubMed]
- Matijasevic, S.; Ristić, R.; Ćirković, D.; Ćirković, B.; Popović, T. Volatile aroma compounds of brandy ‘Lozovača′ produced from muscat table grapevine cultivars (Vitis vinifera L.). Molecules 2019, 24, 2485. [Google Scholar] [CrossRef]
- Ivic, I.; Kopjar, M.; Jukic, V.; Bošnjak, M.; Maglica, M.; Mesić, J.; Pichler, A. Aroma profile and chemical composition of reverse osmosis and nanofiltration concentrates of red wine cabernet sauvignon. Molecules 2021, 26, 874. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Zhang, X.; Guo, S.; Yan, H.; Wang, J.; Zhou, J.; Yang, J.; Duan, J. Headspace GC/MS and fast GC e-nose combined with chemometric analysis to identify the varieties and geographical origins of ginger (Zingiber officinale Roscoe). Food Chem. 2022, 396, 133672. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Z.; Han, B.; Wu, W.; Zhao, Q.; Wei, C.; Liu, W. Comprehensive analysis of volatile compounds in cold-pressed safflower seed oil from Xinjiang, China. Food Sci. Nutr. 2020, 8, 903–914. [Google Scholar] [CrossRef]
- Hou, C.Y.; Huang, P.H.; Lai, Y.T.; Lin, S.P.; Liou, B.K.; Lin, H.W.; Hsieh, C.W.; Cheng, K.C. Screening and identification of yeasts from fruits and their coculture for cider production. Fermentation 2021, 8, 1. [Google Scholar] [CrossRef]
- Zheng, P.; Qin, X.; Chen, H.; Li, X.; Liu, X.; Liao, B.; Li, N.; Qin, X.; Chen, H.; Li, X.; et al. Research on co-production technology of rum and sugarcane vinegar. China Food Addit. 2022, 33, 178–184. [Google Scholar]
- Lu, L.; Hu, Z.; Hu, X.; Li, D.; Tian, S. Electronic tongue and electronic nose for food quality and safety. Food Res. Int. 2022, 162, 112214. [Google Scholar] [CrossRef]
- Liu, J.; Qian, J.; Adil, M.; Bi, Y.; Wu, H.; Hu, X.; Wang, Z.; Zhang, W. Bioinspired integrated triboelectric electronic tongue. Microsyst. Nanoeng. 2024, 10, 57. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).