Abstract
Traditional hard Xinotyri cheese was manufactured using raw or pasteurized goat milk, without starter cultures, and the changes in microbiological and biochemical characteristics were studied during ripening and storage. Mesophilic lactic acid bacteria (LAB) predominated (>8.5 log CFU/g) in freshly fermented Xinotyri cheeses (pH 4.5–4.6), regardless of milk pasteurization. Enterobacteria, pseudomonads and staphylococci were suppressed below 6 and 4–5 log CFU/g in fresh cheeses from raw and pasteurized milk, respectively. Salmonella and Listeria spp. were absent in 25 g cheese samples. Coagulase-positive staphylococci exceeded the 5-log safety threshold in fresh raw milk cheeses, which also had 10-fold higher levels of enterococci than pasteurized milk cheeses. Non-LAB groups declined <100 CFU/g, whereas yeasts increased to 5–6 log CFU/g in both cheeses during ripening. Milk pasteurization affected the protein, fat, ash, moisture, nitrogen fractions, total free fatty acids and total free amino acids content of cheeses. Primary proteolysis, detectable by urea-PAGE, was more intense in raw milk cheeses than in pasteurized milk cheeses. However, the hydrophilic and hydrophobic peptides and their ratio in the water-soluble fraction were similar in both cheeses. Cheeses discriminated clearly according to the milk kind (raw, pasteurized) and the stage of ripening, based on the examined biochemical characteristics.
1. Introduction
During ripening, a cheese undergoes microbiological and biochemical changes, resulting in the development of its characteristic texture and flavor. The microbiological changes include the death and lysis of starter cells, the growth of adventitious microbiota (non-starter lactic acid bacteria) and, in many varieties, the development of a secondary microbiota. The biochemical changes that occur during cheese ripening may be grouped into primary events, which are proteolysis, lipolysis and glycolysis, and secondary events, which include the metabolism of fatty acids and amino acids and affect the development of volatile flavor compounds. The biochemical transformations are catalyzed by enzymes from residual coagulant, milk (particularly plasmin) and proteinases and peptidases from bacteria, starters, non-starters and adjunct cultures [1].
Cheeses made from raw milk generally develop a stronger aroma and more intense taste than the same cheeses made from pasteurized milk. However, sometimes, raw milk cheeses can have a non-controlled microbiota, which may cause spoilage or food poisoning. Therefore, raw milk is often pasteurized to eliminate pathogenic and spoilage bacteria, although post-pasteurization contamination may occur. However, pasteurization can cause changes in milk, such as the inactivation of indigenous milk enzymes, elimination of milk microorganisms, slight denaturation of whey proteins and modifications in the activity of starter bacteria, which may affect the characteristics of the produced cheese [2].
Traditional cheeses have specific characteristics, which are linked to a particular geographical place and the historic and local cultural traditions of their production area. The maintenance and the development of their production are important since it can bring many economic, environmental and social benefits. General strategies such as marketing, or legislation could result in their larger-scale production and develop the local economies. Over the years, many efforts have been made to protect traditional food products [3,4,5].
In many countries, at present, goat cheeses have gained popularity due to the increased interest of consumers in both the traditional cheese-making practices and the sensorial and nutritional value attributed to goat milk. It is known that goat milk has unique characteristics such as antioxidant, anti-platelet aggregation and anti-microbial properties and easier digestion compared to cow milk [6,7]. Greece is the first among European countries in goat population and third in goat milk production [8]. In Greece, goat milk is usually added to sheep or cow milk to produce different types of cheeses (protected designation of origin or other traditional cheeses). The market for dairy products made from goat milk is increasing despite the seasonality of its production.
Hard Xinotyri cheese is a popular, traditional cheese, manufactured in Naxos island in the Aegean Sea (Cyclades complex), in late spring–early summer, from raw goat’s milk from indigenous breeds. It has a wrinkled rind with a yellow–grey colour, whereas the interior of the cheese is compact, with a white–yellow colour. It has an acidic and slightly salted taste, a yeast smell, a crumbly texture and a buttery mouthfeel. The identity (compositional, microbiological and biochemical changes) of this artisanal cheese has been studied previously [9,10]. This cheese is traditionally made from raw milk in small farms; however, to standardize the processing behaviour of the raw milk and obtain a constant-quality cheese that meets the current microbiological safety criteria, pasteurized milk could be used, resulting in its larger-scale production.
Therefore, the objective and the novelty of the present work were to study the influence of milk pasteurization prior to cheese-making on the microbiological, physico-chemical, proteolytic and lipolytic changes in hard Xinotyri cheese during ripening. This information could be of great interest for the eventual manufacture of this cheese in an industrial scale under controlled conditions.
2. Materials and Methods
2.1. Cheese-Making
Hard Xinotyri cheese was manufactured with the technology described by Bontinis et al. [9]. In brief, raw (R) or pasteurized (P) goat’s milk (63 °C for 30 min) was used for the cheese production in Naxos island, in the Cyclades complex, Aegean Sea. No starter cultures were added to the pasteurized milk, as is current practice in the manufacture of this cheese. Commercial calf rennet (1:10,000 strength) was used (3–4 mL for 100 L of milk). Curdling was achieved in about 24 h at room temperature (20–25 °C). Then, the cheese curd was transferred in cloth to pierced plastic and remained there for 2–3 h until most of the whey had been drained. Dry salt was added (1.5% w/w) with manual kneading for its uniform distribution in the curd. The salted curd was transferred to pierced plastic truncated conical moulds and remained there for 3–4 days. The cheese moulds were turned over every day, until the crust was hardened. Afterwards, the cheeses were transferred to ripening rooms (16 °C and relative humidity 70–80%) for 45 days, and then to storage rooms (4 °C) for up to 90 days. Three cheese-making trials were conducted on different production days.
2.2. Sampling
The cheeses from each batch (raw or pasteurized) were sampled on days 1, 20, 45, 60 and 90 after manufacture. Day 1 refers to the fresh cheese curd samples before salting, according to the flow diagram of traditional hard Xinotyri cheese production illustrated by Bontinis et al. [9]. Five whole cheeses were produced and, on each sampling day, one was randomly selected for use. The surface layer, which was approximately 0.5 to 1.0 cm, was discarded. Then, two slices of about 100 g, one from an edge and one from the middle of the cheese block, were cut and blended to a homogeneous paste, and small portions were taken for the analyses in duplicate.
2.3. Microbiological Analyses
Cheese samples (25 g) were aseptically transferred to stomacher bags, then 225 mL of 0.1% buffered peptone water (BPW; Merck, Darmstadt, Germany) was added and the mixture was homogenized in a stomacher (Lab Blender, Seward, London, UK) for 60 sec at room temperature. The homogenates were serially diluted in 0.1% BPW and spread (0.1 mL samples) or poured (1 mL samples) in duplicate on different agar media plates, following the microbial quantification methods applied for the analysis of fresh soft, acid-curd Xinotyri cheese that was previously made from raw or pasteurized goat’s milk [11]. In specific, total viable mesophilic bacteria were enumerated on casein–peptone soymeal–peptone (CASO) agar (Merck) with 0.6% yeast extract (Neogen Media, formerly Lab M, Bury, Lancashire, UK), aerobically incubated at 30 °C for 72 h; total dairy lactic acid bacteria (LAB), able to ferment lactose, on M17 agar (Lab M), aerobically incubated at 37 °C for 48 h; total mesophilic, aciduric LAB, able to grow well in the presence of acetate, on de Man, Rogosa, Sharpe (MRS) agar (Lab M), anaerobically incubated (Gas-Pack system, BBL, Becton, Dickinson, Sparks, MD, USA) at 30 °C for 72 h; enterococci on kanamycin aesculin azide (KAA) agar (Lab M), aerobically incubated at 37 °C for 48 h; total staphylococci on Baird–Parker (BP) agar base with egg yolk tellurite, aerobically incubated at 37 °C for 48 h; pseudomonad-like bacteria on Pseudomonas agar base with cephalothin-fucidin-cetrimide (CFC; supplement X108, Lab M), aerobically incubated at 25 °C for 48 h; total Enterobacteriaceae by pouring 1 mL samples into melted (45 °C) violet red bile glucose (VRBG) agar, overlayed with 5 mL of the same agar to create microaerophilic conditions, and incubated at 37 °C for 24 h; yeasts on rose bengal chloramphenicol (RBC) agar (Merck), incubated at 25 °C for 5 days. The lowest detection limit of the above analyses was 2.0 log CFU/g, except for enterobacteria, which had a lowest detection limit of 1 log CFU/g in the poured VRBG agar plates.
The electivity of the BP, CFC and RBC agar media was checked by subjecting at least 10 representatives of the different colony types, in case of growth, to rapid tests for their microscopic appearance and gram-stain, catalase and oxidase reactions, as was most recently described by Samelis et al. [12]. Moreover, staphylococcal colonies surrounded by characteristic zones of lecithinase activity were confirmed for agglutination using the rapid Staph Microscreen test (Microgen Bioproducts, Camberley, UK). Regarding the native Xinotyri cheese LAB biota predominantly grown on CASO, MRS, M17 and KAA agar plates, 5 to 10 random colonies from each medium were periodically transferred for growth in tubes, with 5 mL MRS broth incubated at 45 °C overnight to check for the presence of thermophilic LAB types in the samples during ripening and storage.
Additionally, the presence of Salmonella spp. and Listeria spp./L. monocytogenes in 25 g cheese samples at the beginning (day 1) and the end (day 45) of Xinotyri ripening was determined by culture enrichment, as reported previously [9].
2.4. Biochemical Analyses
Cheeses were analyzed for moisture, salt, fat, total nitrogen (TN) and ash content with established methods, as described by Bontinis, et al. [9]. The fat-in-dry-matter (FDM) content was calculated by Equation (1):
fat-in-dry-matter% = fat × 100/100-moisture
The protein content was calculated by Equation (2):
protein% = TN% × 6.38.
The pH of the samples was measured using a pH meter (model micro-pH 2001, Crison, Barcelona, Spain).
Water-soluble nitrogen (WSN), nitrogen soluble in 5% phosphotungstic acid (PTA-N) and nitrogen soluble in 12% trichloroacetic acid (TCA-N), as the percentage of TN of the cheese, were determined by the methods described by Mallatou et al. [13].
To monitor the degradation of as- and β-caseins during ripening and storage, cheese samples were analyzed by urea-polyacrylamide gel electrophoresis (PAGE) as described by Mallatou et al. [13]. The levels of residual αs- and β-casein in the ripened cheeses were calculated in comparison with the level present in a reference sample of 1-day-old cheese.
Peptide profiles of the water-soluble fraction of the cheeses were determined by RP-HPLC using a WATERS HPLC system (WATERS Associates, Milford, MA, USA), as described by Mallatou et al. [13]. After each run, the integration area of peptides was determined and divided into two regions, with the criterion being the elution time of peaks. The first group consists of the hydrophilic peptides (HL) with retention times from 0 to 67.5 min (0–55% eluent B). The second group consists of hydrophobic peptides (HB) with retention times ranging from 67.6 to 110 min (55.1–100% eluent B).
The free amino acids (FAA) were extracted from the cheese, as described by Pappa and Sotirakoglou [14], derivatized as phenylthiocarbamyl (PTC)-derivatives using the PICO-Tag amino acid analysis system (Waters, Milford, MA, USA), separated and quantified on an HPLC (Waters, Milford, MA, USA). The results were expressed on a dry matter basis.
The free fatty acids (FFA) of the cheese samples were extracted, separated and quantified as described previously [15], using a Shimadzu model GC-17A gas chromatograph (Shimadzu Scientific Instruments Inc. Columbia, MD, USA), equipped with an on-column injector and a flame ionization detector (FID).
2.5. Statistical Analysis
In order to analyze the response variables relating to chemical and microbiological data, a two-way, repeated measures analysis of variance was applied considering the ripening time (1, 20, 45, 60 and 90 days) as a repeated measure and the type of milk (raw and pasteurized) as an independent factor. Multiple comparisons between the interventions were tested using the Least Significant Difference (LSD) test. Principal component analysis (PCA) was used to reduce the dimensionality of the chemical data, to detect the relationships between the variables and investigate if the cheeses can be distinguished according to the type of milk and the stage of ripening. For all tests, the level of significance was set at 0.05. A statistical analysis was performed using the software STATGRAPHICS Plus for Windows version 5.2, Manugistics, Inc. (Rockville, MD, USA).
3. Results and Discussion
3.1. Microbiological Characteristics of Hard Xinotyri Cheeses during Ripening and Storage
Table 1 summarizes the changes in the population of the main microbial types during the ripening and storage of three batches of hard Xinotyri cheese made from raw (R) or pasteurized (P) milk. The fresh (day-1), unsalted R and P cheese curd samples did not significantly differ (p > 0.05) in total mesophilic viable counts (TVC), which were higher than 8.5 log CFU/g. Along with the acid pH values of both cheese types, shown in Table 2, the very high TVCs shown above reflected the predominance of an adventitious LAB biota that had already evolved well above the 8-log level in all freshly fermented cheese samples, regardless of previous milk pasteurization or the aerobic or anaerobic incubation of the M17 and MRS agar plates at 37 °C or 30 °C, respectively (Table 1). Conversely, the levels of enterococci, which constitute an important part of the native microbiota in traditional raw or thermized milk cheeses made in Greece or other Mediterranean countries [16,17,18], were, on average, 6.4 log CFU/g in fresh raw milk cheeses, and 0.7-log units lower in the pasteurized milk cheeses (Table 1). Notably, except for enterococci, all prevalent LAB colony types tested from CASO, M17 and MRS agar plates failed to grow at 45 °C in MRS broth. Thus, the microbiota of all Xinotyri cheese curds before salting was dominated by mesophilic LAB types, which had grown abundantly after 24 h of milk fermentation at 20–25 °C, followed by 2–3 h of draining at 16 °C. Based on the literature, this prolific LAB growth was normal in the raw milk cheese curds [19], but this speed was surprising in their pasteurized counterparts in the absence of commercial or natural starters [20,21]. Probably, the prolific LAB growth in the pasteurized cheese samples was either because the traditional open-batch pasteurization (63 °C; 30 min) process was poorly monitored, or because the pasteurized milk was cross-contaminated with adventitious LAB from the plant environment or subjected to ‘back-slope’ inoculation [22], although the local cheese processor denied the application of such technique. This is emphasized because, in traditional semi-hard or hard thermized milk cheeses [23,24,25], as well as in fresh or ripened acid-curd cheeses [12,26], the technological microbiota during fermentation and early ripening stages is dominated by thermophilic LAB species, primarily Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus, acquired from the natural yoghurt or whey starters applied to enhance the acidification of the curd [21,27]; however, thermophilic LAB other than enterococci did not occur in hard or soft acid-curd Xinotyri cheese varieties [9,11]. Instead, regardless of the sources of the mesophilic technological LAB biota during curdling of the pasteurized milks, their prolific growth and good milk-acidifying capacity in the curd were beneficial for the microbial quality and safety of the naturally fermented and ripened hard Xinotyri cheeses of this study. Indeed, apart from enterococci, which belong to the beneficial cheese LAB biota but may also be considered undesirable biota and hygienic indicators [18], all non-LAB groups, i.e., enterobacteria, pseudomonads and total staphylococci, were suppressed below 6 log CFU/g and 4–5 log CFU/g in the fresh cheese samples from raw and pasteurized milk, respectively (Table 1). In contrast, the fresh curds of most artisan Greek cheese varieties harbor high levels of enterobacteria, staphylococci and other non-LAB biota during fermentation and early ripening stages [28,29,30]. In the present study, the growth suppression or mitigation of the acid-sensitive Gram-negative or Gram-positive non-LAB biota was apparently due to the rapid and competitive LAB growth and the low pH (4.5–4.6) of the fresh Xinotyri cheese curds (Table 2), and was more pronounced in the pasteurized milk cheese samples (Table 1). In particular, mean levels of total staphylococci were more than 2.5 log units higher in the fresh (day 1) unsalted cheeses from raw than pasteurized milk (Table 1). Most importantly, based on the latex agglutination testing of ca. 10% of the lecithinase-positive colonies enumerated on countable BP agar plates, coagulase-positive staphylococci were estimated to occur at levels slightly above the 5-log safety threshold in the curd of two raw milk cheese batches, as was found in fresh, soft Xinotyri cheese from raw milk [11] and in raw milk Kashkaval cheese before the ‘pasta filata’ heat process [31]. In contrast, all fresh pasteurized milk cheeses had less than 100 CFU/g of coagulase-positive staphylococci in the curd (data not shown), confirming our previous findings [11,31].

Table 1.
Microbial characteristics (log CFU/g) of hard Xinotyri cheese made from raw (R) or pasteurized (P) milk, during ripening and storage (i).

Table 2.
Physicochemical characteristics of hard Xinotyri cheese made from raw (R) or pasteurized (P) milk, during ripening and storage (i).
During the ripening of Xinotyri, there was no further growth in LAB or other bacteria (Table 1) due to the acid cheese pH 4.2–4.5 (Table 2). Instead, the predominant LAB progressively declined below the 8-log level, but remained viable above the 7-log level in all cheeses (Table 1). Declines were significant (p < 0.05) by the end of ripening (day 45) and reflected the inability of a major portion of the LAB biota to survive the harsh conditions of high acidity and dehydration prevailing in hard Xinotyri cheese during ripening (Table 2). Even greater declines in the mesophilic LAB populations occurred in artisan-made Xinotyri cheeses from raw goats’ milk that were studied previously, probably because those batch products were much more dehydrated during ripening [9] than the present hard Xinotyri samples (Table 2). These results generally contrast with traditional Greek cheese varieties with a higher pH and moisture content than Xinotyri, where mesophilic non-starter LAB, particularly members of the Lactobacillus casei/paracasei and Lactobacillus plantarum species, promote significant growth during ripening [20,25,29,32,33,34,35,36].
Enterococci populations also declined below 6 log CFU/g during ripening, with their declines being greater (p < 0.05) in pasteurized milk cheeses than in raw milk cheeses. This finding was in agreement with the overall lowered levels of acid-sensitive enterococci in fresh and ripened Greek acid-curd cheeses [12,26]. Conversely, enterococci were found to occur at levels higher than 7-log units, or even predominate at levels above 8 log units, in Greek Graviera and other traditionally ripened hard cheeses with a final pH > 5.0 [16,28,34,35], depending on the starter culture type [25].
All non-LAB groups, including pathogenic staphylococci that exceeded the 5-log safety threshold in two fresh (day 1) raw milk cheeses, declined below 100 CFU/g in all ripened hard Xinotyri cheeses (Table 1), in agreement with relevant data from previous studies [9,20]. Only the population of acid-tolerant yeasts increased by ca. 2 log units by mid-ripening (day 20). Yeasts were stabilized at populations within 5–6 log CFU/g by the end of ripening and during the refrigerated storage of all ripened cheeses, regardless of previous milk pasteurization (Table 1). The ability of yeasts to proliferate in traditional food fermentations, particularly at the low pH of acid-curd fresh or ripened cheese varieties, is well established [37].
Finally, all fresh (day 1) and ripened (day 45) cheese samples were free of Salmonella and Listeria at 25 g (data not tabulated). Altogether the results of the present and previous studies indicated that the hygienic quality and safety of traditional hard Xinotyri was very good, and that the cheese’s acid pH, in combination with its low moisture (<40%), were the primary inhibitory factors against gram-negative milk spoilage and pathogenic bacteria during fermentation, ripening and storage (Table 1 and Table 2; [9]). The pasteurized milk Xinotyri cheeses of this study, which retained fewer than 1000 CFU/g of pathogenic staphylococci throughout processing, were in compliance with the current EC Regulation microbiological safety criteria for RTE cheese products [38].
3.2. Biochemical Analyses
3.2.1. Composition
The gross chemical composition of the pasteurized and raw hard Xinotyri cheeses during ripening and storage are presented in Table 2. The initial pH of the cheese was approximately 4.5. No significant differences were found in pH values between P and R cheeses at all sampling dates. The same trend was observed in Kashkaval cheese made from raw or pasteurized milk [17]. Although, in the present work, no lactic acid bacteria (starter culture) were added in the pasteurized milk, the pH of pasteurized cheese reached a pH of 4.53 the first day. For the cheese samples made from pasteurized milk, the post-thermal contamination of the milk with adventitious LAB under the artisanal cheesemaking conditions was likely high enough to reduce the fresh curd pH compared to that of the raw cheese samples after fermentation at an ambient temperature. The same was also observed in soft Xinotyri cheese made from raw or pasteurized milk [11]. In the present work, the pH values decreased in pasteurized cheese but did not significantly decrease in the first 20 days, and then remained stable throughout ripening and storage, whereas in raw cheese samples, pH decreased significantly (p < 0.05) in the beginning of ripening and increased (p < 0.05) on day 90. A reduction in pH values is expected at the beginning of ripening due to the metabolism of residual lactose to lactic acid, possibly followed by an increase in the pH due to the formation of ammonia or other compounds during the proteolysis [39]. The moisture content, which was high, at around 72% on day 1, decreased (p < 0.05) during ripening and storage. Moisture decrease resulted from acid development due to the microbial multiplication during ripening and storage, as well as due to the syneresis of cheeses. The reduced hydration of casein as the pH reaches its isoelectric point may also be a contributing factor to the decrease in moisture [27]. At 90 days, the moisture content in the raw and pasteurized hard Xinotyri cheese was 35.49% and 28.97% and differed significantly (p < 0.05). The salt content of the cheeses increased (p < 0.05) during ripening and storage; a rapid increase was observed on day 20. No significant differences (p > 0.05) were detected in the salt content between raw and pasteurized milk cheeses. The contents of protein increased until day 45 and then remained stable, following the opposite trend to moisture (Table 2). Pasteurization, generally, affected (p < 0.05) the moisture, fat, protein and ash content of mature cheeses. The same trend was found for Motal cheese made from raw or pasteurized milk [40]. In this study, the contents of the different physicochemical parameters of raw milk cheeses were, generally, lower than the respective values found for artisanal cheese [9], possibly because the use of raw milk did not result in a cheese with constant composition.
At 90 days of ripening and storage, raw and pasteurized hard Xinotyri cheese had 35.49% and 28.97% moisture, and 53.40% and 59.43% FDM content, respectively (Table 2). Therefore, the cheeses in the present study fulfilled the requirements of the Greek legislation and could be categorized as excellent quality (maximum moisture 35%, minimum FDM 47%) hard Xinotyri cheeses, according to the regulation for hard cheeses [41]. Therefore, in the present work, high-quality hard Xinotyri traditional cheese from goat milk was manufactured.
3.2.2. Proteolysis
Cheese proteolysis involves the initial hydrolysis of caseins, which are primarily catalyzed by residual coagulant and, to a lesser extent, by plasmin, resulting in the formation of large and intermediate-sized peptides that are subsequently degraded by enzymes from the starter and non-starter flora of the cheese. The production of small peptides and free amino acids results from the action of bacterial proteinases and peptidases [1,39].
The WSN, TCA-N and PTA-N fractions were used to determine the extent of proteolysis of the experimental cheeses in this study. The changes in the above casein fractions during the ripening of hard Xinotyri cheese are shown in Table 3. Generally, when differences were observed, the amounts of WSN%TN, TCA-N%TN and PTA-N% TN were significantly higher (p < 0.05) in raw than in pasteurized cheese, in accordance with the results of others [40,42]. Considering the soluble N fractions levels, proteolysis in hard Xinotyri cheese increased during ripening, as similar changes were also observed in other cheese types [43,44]. In the present work, the evolution of the nitrogen fractions during the ripening of hard Xinotyri cheese could be attributed to the activity of the native flora as no starters were used during the cheese manufacturing. Raw, hard Xinotyri cheese of the present study generally showed higher levels of soluble fractions than were previously reported by Bontinis et al. [10], possibly due to the variations in the composition of raw milk.

Table 3.
Changes in nitrogenous fractions of hard Xinotyri cheese made from raw (R) or pasteurized (P) milk, during ripening and storage (i).
Intact caseins and high-molecular-weight peptides (derived from the proteolysis of caseins) can be separated into various protein bands on an electrophoresis gel. The intensity of staining in each band reflects the amount of each protein or peptide that is present in the cheese. Urea-PAGE electropherograms showing the separation of cheese proteins during the ripening of raw and pasteurized hard Xinotyri cheese are presented in Figure 1. Table 4 presents the hydrolysis of individual casein fractions expressed as a percentage of the concentration of the corresponding casein present at first day. The results showed that the degradation of β-casein decreased during aging, but not as rapidly as that observed for αs-casein. The lower degradation of β-casein compared to that of αs-casein may be attributed to the structure of β-casein, which renders the molecule less accessible to enzymes [45]. As can be seen from Figure 1 and Table 4, the pasteurization of cheese-milk significantly (p < 0.05) affected both the residual αs- and β-casein, and this is in agreement with the results found by others [44,46]. However, the opposite results were observed by Trujillo et al. [47], possibly due to the many factors affecting cheese manufacture, such as the type of milk and cultures, the cheesemaking technology and the ripening conditions used.

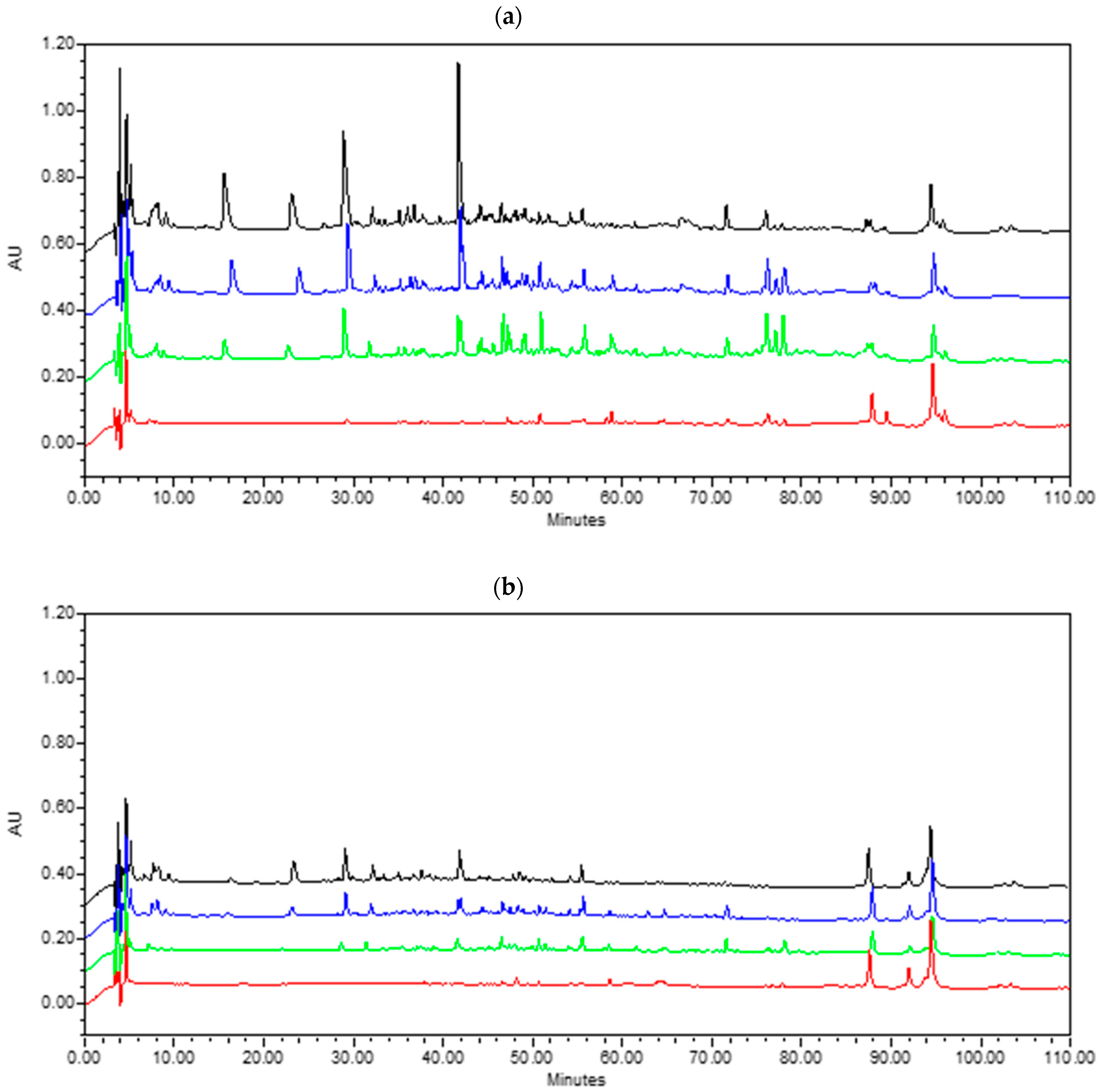
Figure 1.
Urea-polyacrylamide gel electrophoretograms of hard Xinotyri cheese made with raw (A) or pasteurized (B) milk during ripening and storage. Lanes 1: total goat casein; 23: 1-d cheese; 4–5: 20-d cheese; 6–7: 45-d cheese; 8–9: 60-d cheese; 10–11: 90-d cheese.

Table 4.
Residual αs- and β-casein of hard Xinotyri cheese made from raw (R) or pasteurized milk (P), during ripening and storage (i).
Table 5 presents the changes in, hydrophilic (HI), hydrophobic (HO) peptides (expressed as a percentage of the total area of the chromatograms) and their ratio (HO/HI) in the water-soluble fraction of hard Xinotyri cheese made from raw or pasteurized milk during ripening and storage. Figure 2 shows the peptide profiles of Xinotyri cheese made from raw or pasteurized milk. It can be seen that as the age of cheeses increased, new peaks appeared, while peaks that existed at the initial stage of ripening increased or decreased in size. The days of ripening significantly (p < 0.05) affected the area of HI and HO peptides; the level of HI peptides significantly increased with time, while HO peptides and the HO/HI ratio decreased with cheese age. The great decrease in the HO/HI ratio observed between 1 d and 20 d of aging could mainly be attributed to the degradation of water-soluble HO peptides and the formation of HI peptides, as well as the highly HO peptides that are no longer water-soluble [48]. In Table 5, it can be seen that the pasteurization of milk had no significant (p > 0.05) effects on the levels of hydrophobic and hydrophilic peptides or on their ratio in the water-soluble fraction of hard Xinotyri cheese. Similar results were reported by others [11,47]. There were some characteristic differences in certain peaks of the peptide profiles of hard Xinotyri cheese, due to pasteurization. From Figure 2, it can be observed that the area of peaks with retention times of 15 min, 30 min and 42 min was higher in raw cheese than that of pasteurized cheese, while the area of peaks with a retention time of 89 min was higher in pasteurized cheese than in raw milk.

Table 5.
Hydrophobic (HO) and hydrophilic (HI) peptides (%TA) in the water-soluble fraction of hard Xinotyri cheese made from raw (R) or pasteurized milk (P), during ripening and storage (i).
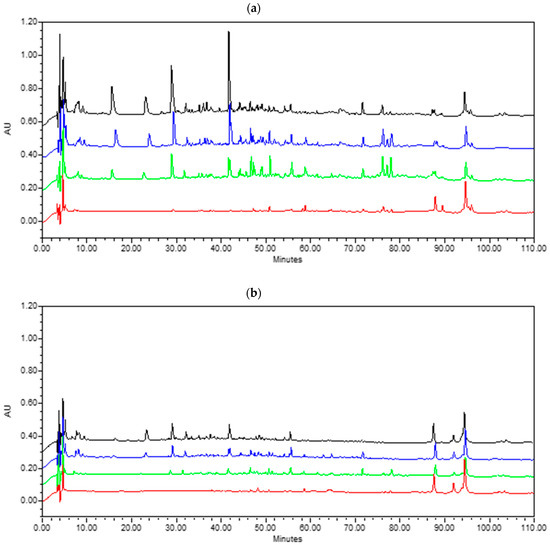
Figure 2.
RP-HPLC chromatograms of water-soluble fractions of hard Xinotyri cheese made from raw (a) or pasteurized (b) milk, during ripening (colour black: 90 days, blue: 45 days, green: 20 days, red: 1 day).
The FAA composition of hard Xinotyri cheese made with raw or pasteurized milk during ripening is shown in Table 6. It is known that each type of cheese has its own characteristic FAA pattern, which results from the enzymatic degradation of peptides by microorganisms, as well as from the interconversion, excretion or degradation of amino acids [49]. The essential amino acid lysine, leucine and threonine, as well as the non-essential alanine and citrulline, were the principal FAAs in mature Xinotyri cheeses (Table 6); high quantities of these free amino acids were found in other type of cheeses [32,50,51,52]. It is known that citrulline, as well as a- and γ-aminobutyric acid and ornithine, do not originate from casein, but accumulate as metabolic products of microorganisms [53]. From Table 6, it can be seen that, generally, raw cheeses showed higher (p < 0.05) levels of total FAA (TFAA) than cheeses made from pasteurized milk, in agreement with the results of others [46,54]. This is usually linked to the significant amino peptidase activities of indigenous bacteria, which increase the level and modify the composition of FAA in R cheeses [2]. In this work, hard Xinotyri cheese made with raw milk showed significantly lower (p < 0.05) values for Asp, and Gln and higher levels for Ala, Arg, Gly, Ile, Tyr and Val in relation to pasteurized milk cheeses (Table 6); these differences could point to quantitative and qualitative differences between the peptidase activity of the indigenous microbiota, since no starter cultures were used. The TFAA content of R and P hard Xinotyri cheese increased (p < 0.05) at all stages of ripening (Table 6). This finding is in accordance with the results found for the nitrogen that was soluble in 5% PTA (Table 2).

Table 6.
Free amino acid content (mg/100 g of dry matter) of hard Xinotyri cheese made from raw (R) or pasteurized milk (P) during ripening and storage (i).
3.2.3. Lipolysis
Free fatty acids (FFA) are usually released by the actions of lipases (from different sources) during lipolysis. Lipolysis is very important for the sensorial properties of cheese as the free fatty acids can be precursors of the volatile compounds that contribute to cheese flavour [1].
The free fatty acid content of raw and pasteurized hard Xinotyri cheese at different sampling days is shown in Table 7. Total FFA in raw milk cheeses was found in higher levels than in pasteurized cheeses, in agreement with the results of others [43,55]. Jandal [56] reported that milk pasteurization destroys most activities of the indigenous lipoprotein lipase. The predominant free fatty acids in all cheese samples were palmitic (C16), stearic (C18), oleic (C18:1) and linoleic (C18:2) acids (Table 7); generally, high quantities of these FFA were found in other cheeses [15,20,55,57]. The previously studied raw Xinotyri cheese [10] showed a greater increase in total FFA during ripening and storage; palmitic, stearic, oleic, myristic and capric acids were the most abundant FFAs. In the present study, although the amounts of all FFA (TFFA) in the raw and pasteurized cheeses increased (p < 0.05) during ripening and storage, their relative increases were different. As a result, the percent composition of short- (C6 to C10), medium- (C12 and C14) and long- (C16 to C18:3) chain FFA changed during ripening. Short-chain FFA (SCFFA), medium-chain FFA (MCFFA) and long-chain FFA (LCFFA) increased (p < 0.05) during ripening and storage in all cheeses. The values of SCFFA did not significantly differ between P and R cheeses (Table 7). The values of MCFFA were higher in R than in P cheeses on the 1st and 90th only, whereas the values of LCFFA were higher (p < 0.05) in raw cheeses at all sampling dates. These results most likely reflect the changes in the microbial lipolytic activity. Despite the quantitative importance of long-chain FFA, they do not contribute to cheese flavour as much as short-chain fatty acids [57].

Table 7.
Free fatty acid content (mg/kg) of hard Xinotyri cheese made from raw (R) or pasteurized milk (P) during ripening and storage (i).
The sensory analysis of cheeses (results not shown) indicated that Xinotyri cheese made from raw or pasteurized goat milk was very much appreciated, although raw milk cheeses developed a more intense aroma and had higher flavour scores than pasteurized cheeses; the latter were characterized as “rather mild”.
3.2.4. Principal Component Analysis
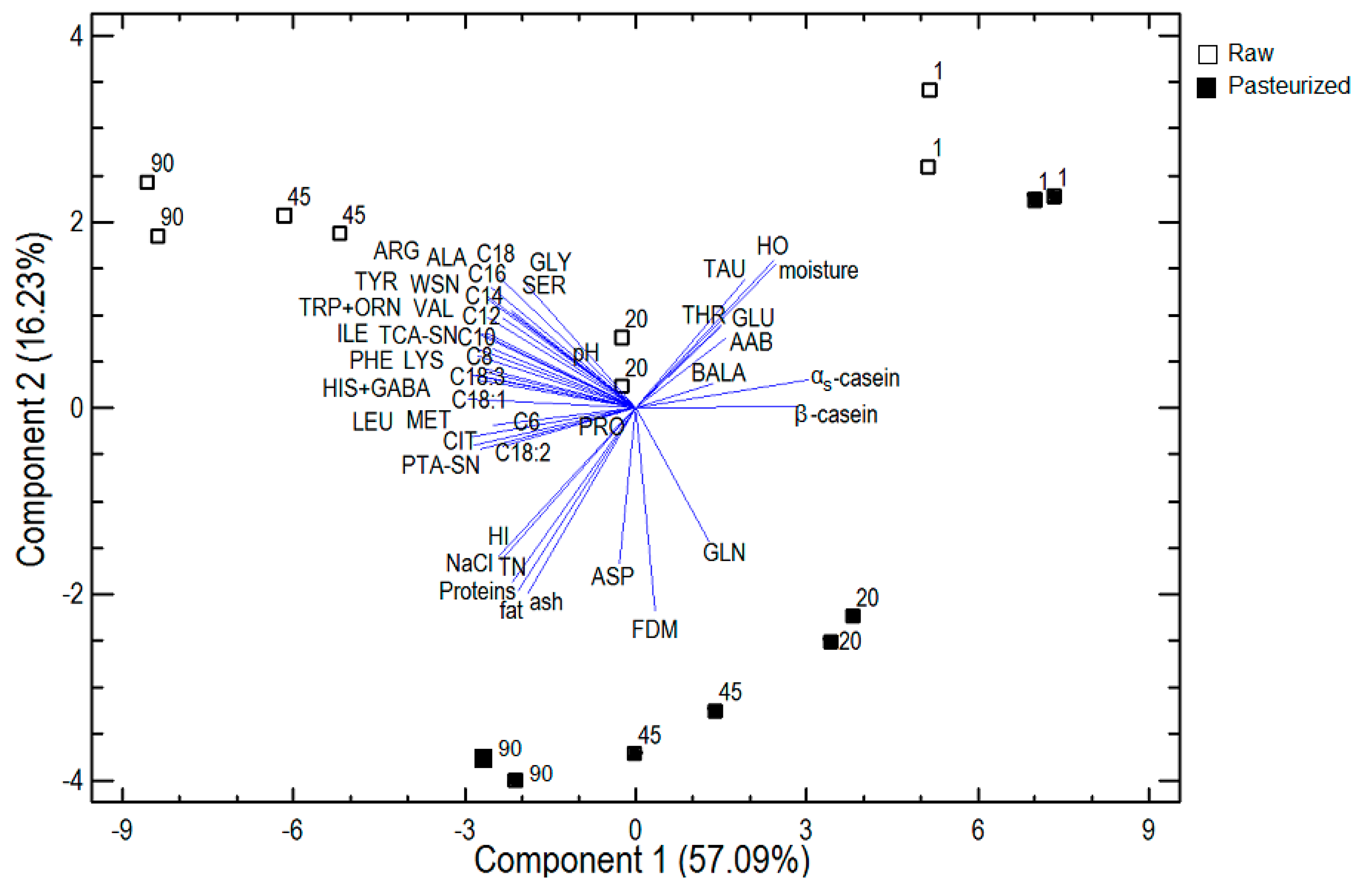
Principal component analysis (PCA) was applied to the pooled data of cheese composition, nitrogenous fractions, αs- and β-casein, hydrophobic and hydrophilic peptides, free amino acid and free fatty acid content to reduce the dimensionality of the data and detect the most important causes of variability, since a large correlation was noticed between the variables, and to establish the variables capable of discriminating cheeses according to the type of milk (raw, pasteurized) and the stage of ripening. The PCA of the 22 variables resulted in five principal components with eigenvalues greater than 1.0, a common statistical cut-off point. The five selected components accounted for 91.95% of the total variability. In Figure 3, a plot of both the first and second principal components is shown. The first principal component (PC1) explained 57.09% of the total variability and was mainly defined by the majority of free fatty acids and αs- and β-casein. Fatty acids were located away from the axis origin, suggesting that they were well represented by the first PC, and were placed close together on the negative side of PC1, indicating that they were positively correlated with each other. However, αs- and β-casein were placed on the positive side of PC1, opposite to free fatty acids and were, therefore, negatively correlated with them. The first PC could be considered as a representative of free fatty acids and caseins, and these factors are mainly related to the stage of ripening, because all 1-day-old cheeses were clustered on the positive side of PC1, while cheeses that were 20 or 45 days old were placed in the middle of PC1 and 90-day-old cheeses were located on the negative side of PC1, near most of the free amino acids and free fatty acids, with the highest contents of these acids. αs- and β-casein were located near 1-day-old cheeses, indicating that they have the highest values in these cheeses and that these values decrease during ripening. The second principal component (PC2) explained another 16.23% of the total variability and was mainly defined by physicochemical characteristics and hydrophobic and hydrophilic peptides. Proteins, fat, ash, NaCl and hydrophilic peptides (HI) were located close together on the negative side of PC2, indicating a strong positive correlation and near-mature cheeses that were produced from pasteurized milk; therefore, these cheeses have the highest contents of those physicochemical characteristics and HI. Hydrophobic peptides (HO) were placed near 1-day-old cheeses made from raw milk, indicating that they have the highest values in these cheeses. The second PC could be considered as a representative of the physicochemical characteristics and hydrophobic and hydrophilic peptides. The third principal component explained another 10.60% of the total variability and was mainly defined by the majority of free amino acids. Most were clustered on the left side of Figure 3, indicating a strong positive correlation with each other and near-mature cheeses that were made from raw milk; therefore, these cheeses have the highest contents of those free amino acids. The fourth and fifth principal components explained another 4.47% and 3.57%, respectively, of the total variability and were mainly defined by nitrogenous fractions. WSN, TCA-SN and PTA-SN were placed on the left side of Figure 3, near the mature cheeses that were produced from raw milk. In conclusion, as can be seen in Figure 3, cheeses could clearly be discriminated according to the kind of milk (raw, pasteurized) and the stage of ripening, based on the examined characteristics.
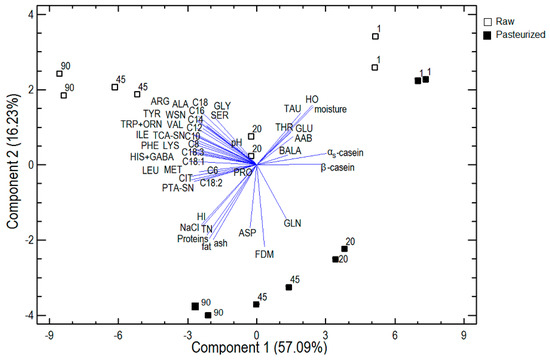
Figure 3.
Principal component analysis. Plot of the two first principal components.
4. Conclusions
Besides the cheeses that are protected under European regulations, other cheeses play an important role in the socioeconomic development of the rural regions in which they are produced. The results obtained during this study showed that high-quality hard Xinotyri traditional cheese products from goat milk were manufactured using either raw or pasteurized milk. Throughout the ripening period, the pasteurization of milk had a decrease and/or delay effect on both the lipolysis and proteolysis of Xinotyri. However, pasteurization can ensure cheese safety. In addition, the acidification that the curd underwent during natural fermentation determines the low pH-value range of Xinotyri cheese. The combination of an acid pH and low moisture resulted in lowered levels of technological mesophilic LAB biota and the elimination of spoilage and pathogenic bacteria during ripening, particularly from the pasteurized milk cheeses. Therefore, the further investigation of the use of pasteurized milk in the production of hard Xinotyri cheese with the addition of starter cultures is suggested to study all the changes that take place during ripening and storage.
Author Contributions
Experimental design, E.C.P.; methodology, E.C.P., J.S. and K.S.; formal analysis, E.C.P., T.G.B., J.S.; writing—original draft preparation, E.C.P., J.S. and K.S.; writing—review and editing, E.C.P., J.S. and K.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Greek Dairy Organization; grant number 4250/06052005.
Acknowledgments
The authors would like to thank the Greek Dairy Organization for the financial support of this study. The contribution, the useful discussions of Helen Mallatou and the technical assistance of Athanasia Kakouri with the microbiological analyses are gratefully acknowledged.
Conflicts of Interest
The authors declare no conflict of interest.
References
- McSweeney, P.L.H. Biochemistry of cheese ripening. Int. J. Dairy Technol. 2004, 57, 127–144. [Google Scholar] [CrossRef]
- Grappin, R.; Beuvier, E. Possible implications of milk pasteurization on the manufacture and sensory quality of ripened cheese. Int. Dairy J. 1997, 7, 751–761. [Google Scholar] [CrossRef]
- Committee of the Regions. Opinion of the Committee of the Regions on Promoting and Protecting Local Products—A Trump-Card for the Regions. 1996. Available online: https://op.europa.eu/en/publication-detail/-/publication/dcd34e04-fce5-4af8-9966-b3d35be2f52d (accessed on 7 October 2021).
- Manzo, N.; Santini, A.; Pizzolongo, F.; Aiello, A.; Marrazzo, A.; Meca, G.; Durazzo, A.; Lucarini, M.; Romano, R. Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco. Sustainability 2019, 11, 2520. [Google Scholar] [CrossRef] [Green Version]
- Donelly, C. The Oxford Companion to Cheese; Oxford University Press: New York, NY, USA, 2016. [Google Scholar]
- Tagliazucchi, D.; Martini, S.; Shamsia, S.; Helel, A.; Conte, A. Biological activities and peptidomic profile of in vitro-digested cow, camel, goat and sheep milk. Int. Dairy J. 2018, 81, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Haenlein, G.F.W. Goat milk in human nutrition. Small Rum. Res. 2004, 51, 155–163. [Google Scholar] [CrossRef]
- FAO STAT. 2018. Available online: www.fao.org (accessed on 7 October 2021).
- Bontinis, T.G.; Mallatou, H.; Alichanidis, E.; Kakouri, A.; Samelis, J. Physicochemical, microbiological and sensory changes during ripening and storage of Xinotyri, a traditional Greek cheese from raw goat’s milk. Int. J. Dairy Technol. 2008, 61, 229–236. [Google Scholar] [CrossRef]
- Bontinis, T.G.; Mallatou, H.; Pappa, E.C.; Massouras, T.; Alichanidis, E. Study of proteolysis, lipolysis and volatile profile of a traditional Greek goat cheese (Xinotyri) during ripening. Small Rum. Res. 2012, 105, 193–201. [Google Scholar] [CrossRef]
- Pappa, E.C.; Bontinis, T.G.; Tasioula-Margari, M.; Samelis, J. Microbial Quality of and Biochemical Changes in Fresh Soft, Acid-Curd Xinotyri Cheese Made from Raw or Pasteurized Goat’s Milk. Food Technol. Biotechnol. 2017, 55, 496–510. [Google Scholar] [CrossRef]
- Samelis, J.; Doulgeraki, A.I.; Bikouli, V.; Pappas, D.; Kakouri, A. Microbiological and metagenomic characterization of a retail delicatessen Galotyri-like fresh acid-curd cheese product. Fermentation 2021, 7, 67. [Google Scholar] [CrossRef]
- Mallatou, H.; Pappa, E.C.; Boumba, V.A. Proteolysis in Teleme cheese made from sheep’s, goats’ or a mixture of sheep’s and goats’ milk. Int. Dairy J. 2004, 14, 977–987. [Google Scholar] [CrossRef]
- Pappa, E.C.; Sotirakoglou, K. Changes of free amino acid content of Teleme cheese made with different types of milk and culture. Food Chem. 2008, 111, 606–615. [Google Scholar] [CrossRef]
- Mallatou, H.; Pappa, E.C.; Massouras, T. Changes in free fatty acids during ripening of Teleme cheese made with ewes’, goats’, cows’ or mixture of ewes’ and goats’ milk. Int. Dairy J. 2003, 13, 211–219. [Google Scholar] [CrossRef]
- Moreno, M.R.F.; Sarantinopoulos, P.; Tsakalidou, E.; De Vuyst, L. The role and application of enterococci in food and health. Int. J. Food Microbiol. 2006, 106, 1–24. [Google Scholar] [CrossRef]
- Pappa, E.C.; Kondyli, E.; Samelis, J. Microbiological and biochemical characteristics of Kashkaval cheese produced using pasteurised or raw milk. Int. Dairy J. 2019, 89, 60–67. [Google Scholar] [CrossRef]
- Dapkevicious, M.L.E.; Sgardioli, B.; Câmara, S.P.A.; Poeta, P.; Malcata, F.X. Current trends of enterococci in dairy products: A comprehensive review of their multiple roles. Foods 2021, 10, 821. [Google Scholar] [CrossRef]
- Montel, M.C.; Buchin, S.; Mallet, A.; Delbés-Paus, C.; Vuitton, D.A.; Desmasures, N.; Berthier, F. Traditional cheeses: Rich and diverse microbiota with associated benefits. Int. J. Food Microbiol. 2014, 177, 136–154. [Google Scholar] [CrossRef]
- Albenzio, M.; Corbo, M.R.; Rehman, S.U.; Fox, P.F.; De Angelis, M.; Corsetti, A.; Sevi, A.; Gobbetti, M. Microbiological and biochemical characteristics of Canestrato Pugliese cheese made from raw milk, pasteurized milk or by heating the curd in hot whey. Int. J. Food Microbiol. 2001, 67, 35–48. [Google Scholar] [CrossRef]
- Beresford, T.P.; Fitzsimons, N.A.; Brennan, N.L.; Cogan, T.M. Recent advances in cheese microbiology. Int. Dairy. J. 2001, 11, 259–274. [Google Scholar] [CrossRef]
- Joishy, T.K.; Dehingia, M.; Khan, M.R. Bacterial diversity and metabolite profiles of curd prepared by natural fermentation of raw milk and back sloping of boiled milk. World J. Microbiol. Biotechnol. 2019, 35, 102. [Google Scholar] [CrossRef]
- Di Cagno, R.; Upadhyay, V.K.; Mc Sweeney, P.L.H.; Corbo, M.R.; Faccia, M.; Gobbetti, M. Microbiological, compositional and biochemical characterization of PDO Canestrato Pugliese cheese. Ital. J. Food Sci. 2004, 16, 45–58. [Google Scholar]
- Piraino, P.; Zotta, T.; Ricciardi, A.; Parente, E. Discrimination of commercial Caciocavallo cheeses on the basis of the diversity of lactic microflora and primary proteolysis. Int. Dairy J. 2005, 15, 1138–1149. [Google Scholar] [CrossRef]
- Vandera, E.; Kakouri, A.; Koukkou, A.I.; Samelis, J. Major ecological shifts within the dominant nonstarter lactic acid bacteria in mature Greek Graviera cheese as affected by the starter culture type. Int. J. Food Microbiol. 2019, 290, 15–26. [Google Scholar] [CrossRef]
- Samelis, J.; Kakouri, A. Major technological differences between an industrial-type and five artisan-type Greek PDO Galotyri market cheeses as revealed by great variations in their lactic acid microbiota. AIMS Agric. Food 2019, 4, 685–710. [Google Scholar] [CrossRef]
- Turner, K.W.; Morris, H.A.; Martley, F.G. Swiss-type cheese II. The role of thermophilic lactobacilli in sugar fermentation. N. Z. J. Dairy Sci. Technol. 1983, 18, 117–124. [Google Scholar]
- Litopoulou-Tzanetaki, E.; Tzanetakis, N. Microbiological characteristics of Greek traditional cheeses. Small Rum. Res. 2011, 101, 17–32. [Google Scholar] [CrossRef]
- Litopoulou-Tzanetaki, E.; Tzanetakis, N. The microfloras of traditional Greek cheeses. Microbiol. Spectr. 2014, 2, 1–34. [Google Scholar] [CrossRef] [Green Version]
- Pappa, E.C.; Samelis, J.; Kondyli, E.; Pappas, A.C. Characterization of Urda whey cheese: Evolution of main biochemical and microbiological parameters during ripening and vacuum packaged cold storage. Int. Dairy J. 2016, 58, 54–57. [Google Scholar] [CrossRef]
- Samelis, J.; Kakouri, A.; Kondyli, E.; Pappa, E.C. Effects of curd heating with or without previous milk pasteurization on the microbiological quality and safety of craft-made ‘Pasta Filata’ Kashkaval cheese curds. Int. J. Dairy Technol. 2019, 72, 447–455. [Google Scholar]
- Gobbetti, M.; Folkertsma, B.; Fox, P.F.; Corsetti, A.; Smacchi, E.; De Angelis, M.; Rossi, J.; Kilcawley, K.; Cortini, M. Microbiology and biochemistry of Fossa pit cheese. Int. Dairy J. 1999, 9, 763–773. [Google Scholar] [CrossRef]
- De Angelis, M.; Corsetti, A.; Tosti, N.; Rossi, J.; Corbo, M.R.; Gobbetti, M. Characterization of non-starter lactic acid bacteria from Italian ewe cheeses based on phenotypic, genotypic and cell wall protein analyses. Appl. Environ. Microbiol. 2001, 67, 2011–2020. [Google Scholar] [CrossRef] [Green Version]
- Settanni, L.; Moschetti, G. Non-starter lactic acid bacteria used to improve cheese quality and provide health benefits. Food Microbiol. 2010, 27, 691–697. [Google Scholar] [CrossRef]
- Samelis, J.; Kakouri, A.; Pappa, E.C.; Matijašic, B.B.; Georgalaki, M.D.; Tsakalidou, E.; Rogelj, I. Microbial stability and safety of traditional Greek Graviera cheese: Characterization of the lactic acid bacterial flora and culture-independent detection of bacteriocin genes in the ripened cheeses and their microbial consortia. J. Food Prot. 2010, 73, 1294–1303. [Google Scholar] [CrossRef]
- Bottari, B.; Levante, A.; Neviani, E.; Gatti, M. How the fewest become the greatest. L. casei’s impact on long ripened cheeses. Front. Microbiol. 2018, 9, 2866. [Google Scholar] [CrossRef] [Green Version]
- Tofalo, R.; Fusco, V.; Böhnlein, C.; Kabisch, J.; Logrieco, A.F.; Habermann, D.; Cho, G.S.; Benomar, N.; Abriouel, H.; Schmidt-Heydt, M.; et al. The life and times of yeasts in traditional food fermentations. Crit. Rev. Food Sci. Nutr. 2020, 60, 3103–3132. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 1441/2007 of 5 December 2007 amending Regulation (EC) No. 2073/2005 on microbiological criteria for foodstuffs. Off. J. Europ. Union 2007, L322, 12–29. [Google Scholar]
- Fox, P.F. Proteolysis during cheese manufacture and ripening. J. Dairy Sci. 1989, 72, 1379–1400. [Google Scholar] [CrossRef]
- Rezaei, A.; Alirezalu, K.; Damirchi, S.A.; Hesari, J.; Papademas, P.; Domínguez, R.; Lorenzo, J.M.; Yaghoubi, M. Effect of Pasteurization and Ripening Temperature on Chemical and Sensory Characteristics of Traditional Motal Cheese. Fermentation 2020, 6, 95. [Google Scholar] [CrossRef]
- Greek Codex Alimentarius. Official Journal of the Hellenic Republic; Volume B, Article 83, No. 899, Paragraphs 1.6–1.8; National Printing Office: Athens, Greek, 2009. [Google Scholar]
- Ballesteros, C.; Poveda, J.M.; Gonzalez-Vinas, M.A.; Cabezas, L. Microbiological, biochemical and sensory characteristics of artisanal and industrial Manchego cheeses. Food Contr. 2006, 17, 249–255. [Google Scholar] [CrossRef]
- Rashtchi, P.; Bazmi, A.; Noshirvani, N.; Moosavy, M.H. Comparison of the microbial, physicochemical, and sensorial properties of raw and pasteurized Lighvan cheeses during ripening time. Food Sci. Nutr. 2021, 9, 5527–5535. [Google Scholar] [CrossRef]
- Moatsou, G.; Κandarakis, I.; Moschopoulou, Ε.; Anifantakis, Ε.; Alichanidis, Ε. Effect of technological parameters onthe characteristics of Κasseri cheese made from raw and pasteurized ewes milk. Int. J. Dairy Technol. 2001, 54, 69–77. [Google Scholar] [CrossRef]
- Phelan, J.A.; Guiney, J.; Fox, P.F. Proteolysis of β-casein in Cheddar cheese. J. Dairy Res. 1973, 40, 105–112. [Google Scholar] [CrossRef]
- Gaya, P.; Sanchez, C.; Nunez, M.; Fernandez-Garcia, E. Proteolysis during ripening of Manchego cheese made from raw or pasteurized ewes’ milk. Seasonal variation. J. Dairy Res. 2005, 72, 287–295. [Google Scholar] [CrossRef]
- Trujillo, A.J.; Buffa, Μ.; Casals, Ι.; Fernández, P.; Guamis, Β. Proteolysis in goat cheese made from raw, pasteurized or pressure-treated milk. Innov. Food Sci. Emerg. Technol. 2002, 3, 309–319. [Google Scholar] [CrossRef]
- Lau, K.Y.; Barbano, D.M.; Rasmussen, R.R. Influence of pasteurization of milk on protein breakdown in Chaddar cheese during aging. J. Dairy Sci. 1991, 74, 727–740. [Google Scholar] [CrossRef]
- Polo, C.; Ramos, M.; Sanchez, R. Free amino acids by high performance liquid chromatography and peptides by gel electrophoresis in Mahon cheese during ripening. Food Chem. 1985, 16, 85–96. [Google Scholar] [CrossRef]
- Paskas, S.; Miocinovic, J.; Savic, M.; Jesic, G.; Raseta, M.; Becskei, Z. Comparison of the chemical composition of whey cheeses: Urda and Ricotta. Maced. Vet. Rev. 2019, 42, 151–161. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez de Llno, D.; Polo, M.C.; Ramos, M.; Martin-Alvarez, P. Free and total amino acids in the non-protein fraction of an artisanal blue cheese during ripening. Z. Lebensm.-Unters. Forsch. 1991, 193, 529–532. [Google Scholar] [CrossRef]
- Skeiea, S.; Lindberg, C.; Narvhus, J. Development of amino acids and organic acids in Norvegia, influence of milk treatment and adjunct Lactobacillus. Int. Dairy J. 2001, 11, 399–411. [Google Scholar] [CrossRef]
- Butikofer, U. Cheese characterization with free amino acids pattern in several hard and semi-hard cheeses. Bull. Int. Dairy Fed. 1996, 317, 22. [Google Scholar]
- Buffa, M.; Guamis, B.; Trujillo, A.J. Specific effect of high-pressure treatment of milk on cheese proteolysis. J. Dairy Res. 2005, 72, 385–392. [Google Scholar] [CrossRef]
- Buffa, M.; Guamis, B.; Pavia, M.; Trujillo, A.J. Lipolysis in cheese made from raw, pasteurized or high-pressure treated goats’ cheese. Int. Dairy J. 2001, 11, 175–179. [Google Scholar] [CrossRef]
- Jandal, J.M. Some factors affecting lipase activity in goat milk. Small Rum. Res. 1995, 16, 87–91. [Google Scholar] [CrossRef]
- Freitas, A.C.; Malcata, F.X. Lipolysis in Picante cheese: Influence of milk type and ripening time on free fatty acid profile. Lait 1998, 78, 251–258. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).