Effects of Trophic Modes on the Lipid Accumulation of Parachlorella kessleri TY
Abstract
:1. Introduction
2. Materials and Methods
2.1. Algal Strains and Cultivation
2.2. Determination of Biomass Concentration and Biomass Productivity
2.3. Determination of Pigment Contents
2.4. Determination of Chlorophyll Fluorescence
2.5. Determination of Total Carbohydrate Content
2.6. Determination of Total Protein Content
2.7. Determination of Neutral Lipid Content
2.8. Determination of Total Lipid Content and Fatty Acid Composition
2.8.1. Determination of Total Lipid Content
2.8.2. Determination of Fatty Acid Composition
2.9. Statistical Analysis
3. Results
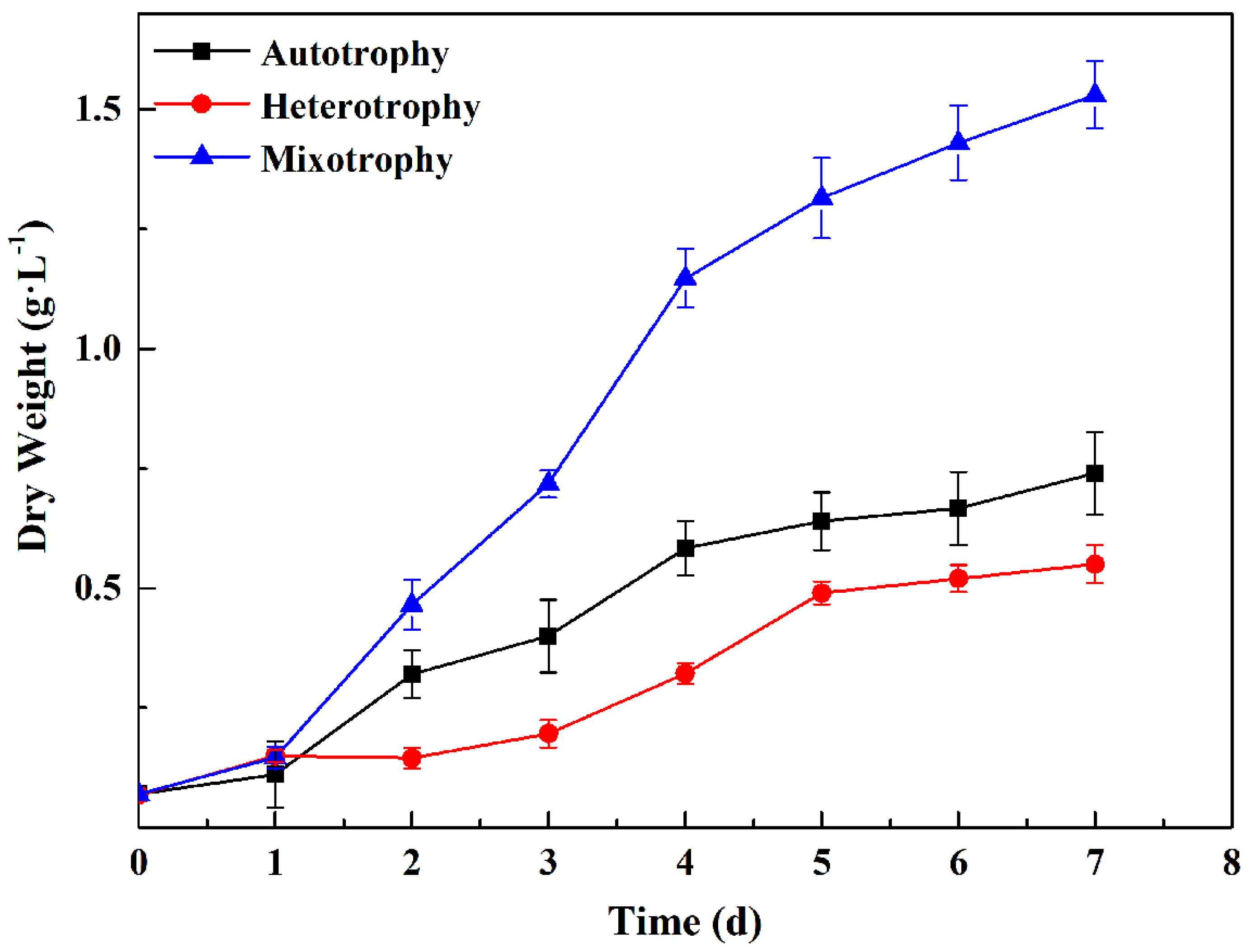
3.1. Analysis of Changes in Growth
3.1.1. The Effect of Glucose Concentration on the Growth of Parachlorella kessleri TY
3.1.2. The Effect of Heterotrophic Culture and Mixotrophic Culture on the Growth of P. kessleri TY
3.2. Analysis of Pigment Content
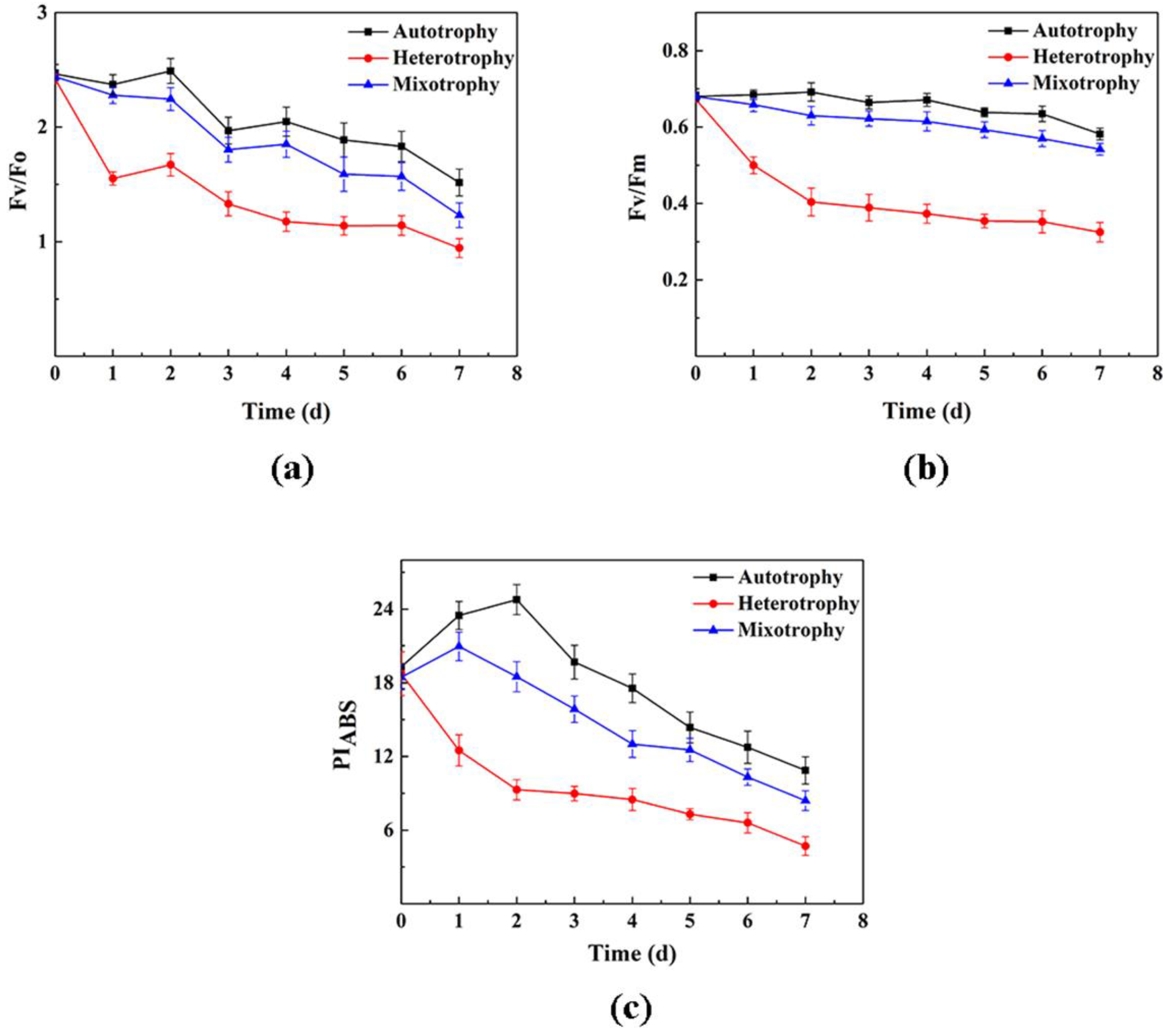
3.3. Analysis of Chlorophyll Fluorescence Characteristics
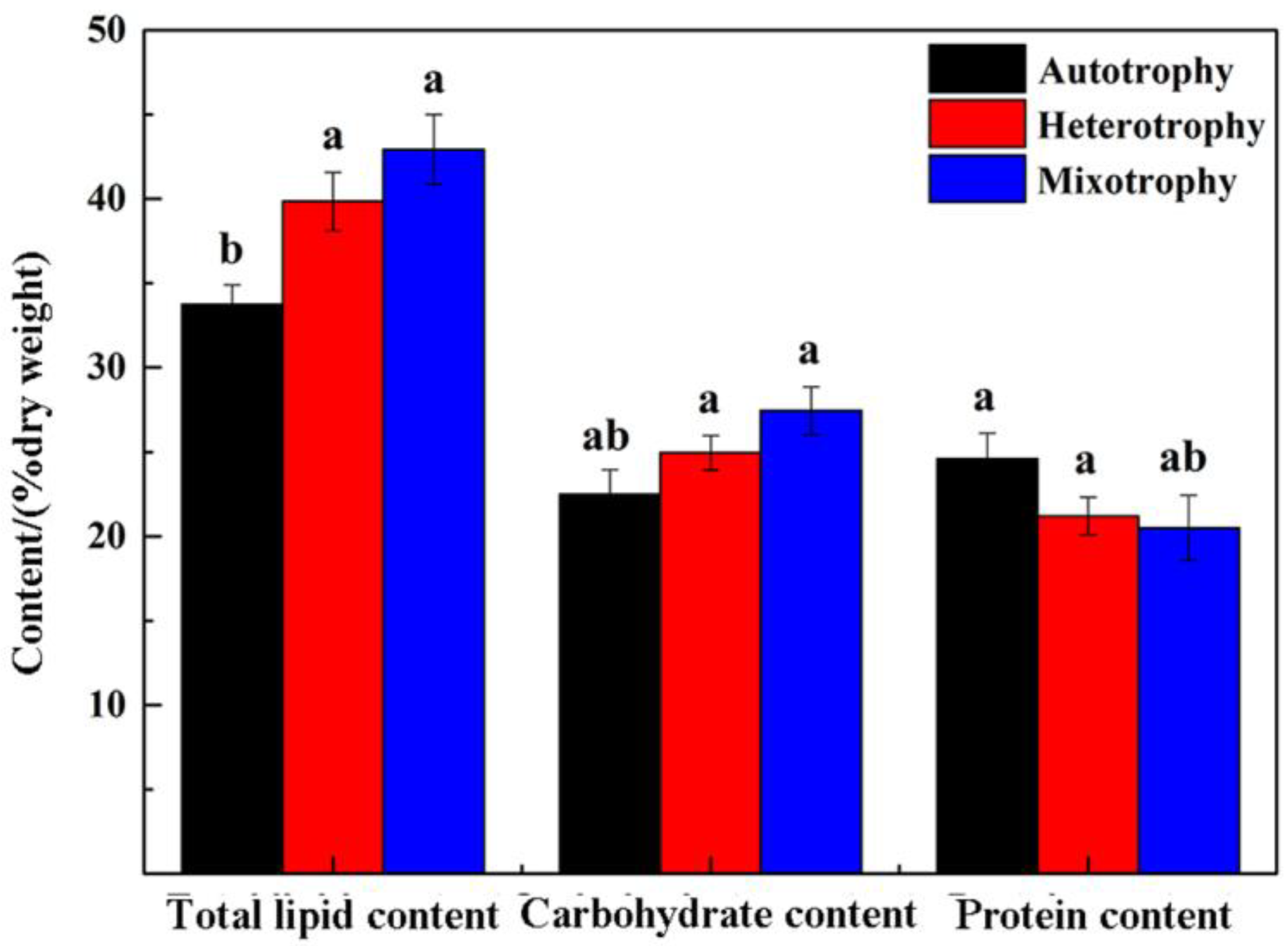
3.4. Analysis of Total Carbohydrate Content
3.5. Analysis of Total Protein Content
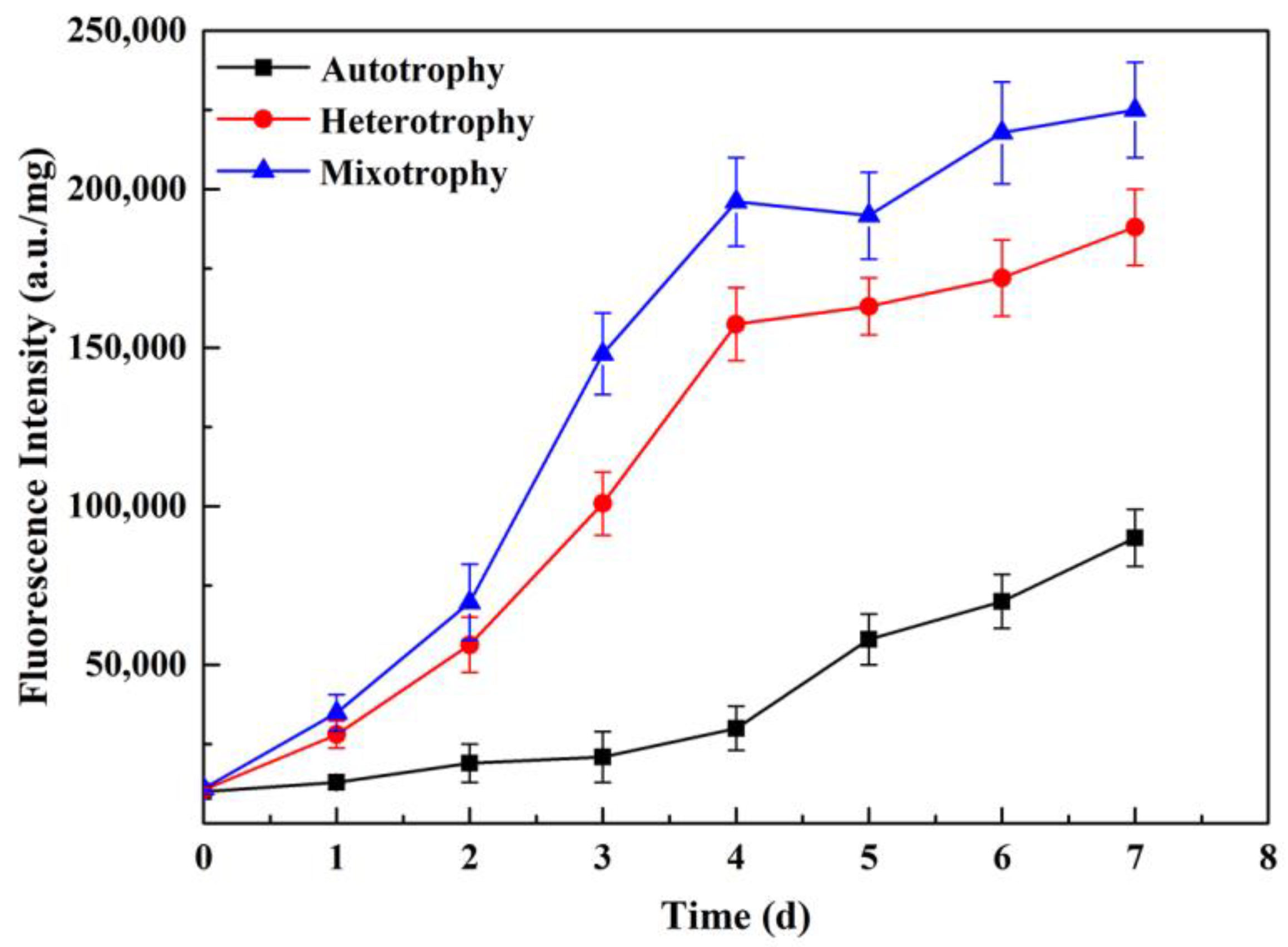
3.6. Analysis of Neutral Lipid Content
3.7. Analysis of Total Lipid Content and Fatty Acid Composition
3.7.1. Analysis of Total Lipid Content
3.7.2. Analysis of Fatty Acid Composition
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parakh, S.K.; Tian, Z.; Wong, J.Z.E.; Tong, Y.W.F. From microalgae to bioenergy: Recent advances in biochemical conversion processes. Fermentation 2023, 9, 529–576. [Google Scholar] [CrossRef]
- Khan, S.; Das, P.; Quadir, A.M.; Thaher, M.I.; Mahata, C.; Sayadi, S.; Al-Jabri, H. Microalgal feedstock for biofuel production: Recent advances, challenges, and future perspective. Fermentation 2023, 9, 281–315. [Google Scholar] [CrossRef]
- Cunha, A.E.P.; Satiro, J.R.; Escobar, B.P.; Simoes, R.M. Chlorella vulgaris growth, pigment and lipid accumulation: Effect of progressive light and hydrogen peroxide exposure. J. Chem. Technol. Biotechnol. 2023, 98, 442–450. [Google Scholar] [CrossRef]
- Touliabah, E.S.; Almutairi, A.W. Effect of phytohormones supplementation under nitrogen depletion on biomass and lipid production of Nannochloropsis oceanica for integrated application in nutrition and biodiesel. Sustainability 2021, 13, 592. [Google Scholar] [CrossRef]
- Trivedi, J.; Atray, N.; Agrawal, D.; Ray, A. Enhanced lipid production in Scenedesmus obliquus via nitrogen starvation in a two-stage cultivation process and evaluation for biodiesel production. Fuel 2022, 316, 123418. [Google Scholar] [CrossRef]
- Pugliese, A.; Biondi, L.; Bartocci, P.; Fantozzi, F. Selenastrum capricornutum a new strain of algae for biodiesel production. Fermentation 2020, 6, 46–58. [Google Scholar] [CrossRef]
- Gao, Y.F.; Lv, J.P.; Feng, J.; Liu, Q.; Xie, S.L. Morphology, phylogeny and lipid components of an oil-rich microalgal strain. J. Appl. Bot. Food Qual. 2017, 90, 298–305. [Google Scholar] [CrossRef]
- Kalantaryan, N.K.; Harutyunyan, B.; Minasyan, E.V.; Goginyan, V. Comparative assessment of brewery wastewater treatment potential by microalgae Parachlorella kessleri and Chlorella vulgaris. Biol. J. Armen. 2021, 4, 72–79. [Google Scholar]
- Hasegawa, M.; Yoshida, T.; Yabuta, M.; Terazima, M.; Kumazaki, S. Anti-stokes fluorescence spectra of chloroplasts in Parachlorella kessleri and maize at room temperature as characterized by near-infrared continuous-wave laser fluorescence microscopy and absorption microscopy. J. Phys. Chem. B 2011, 115, 4184–4194. [Google Scholar] [CrossRef]
- Turmel, M.; Otis, C.; Lemieux, C. The chloroplast genomes of the green algae Pedinomonas minor, Parachlorella kessleri, and Oocystis solitaria reveal a shared ancestry between the Pedinomonadales and Chlorellales. Mol. Biol. Evol. 2009, 26, 2317–2331. [Google Scholar] [CrossRef]
- Beigbeder, J.B.; Sanglier, M.; Medeiros Dantas, J.M.; Lavoie, J.M. CO2 capture and inorganic carbon assimilation of gaseous fermentation effluents using Parachlorella kessleri microalgae. J. CO2 Util. 2021, 50, 101581. [Google Scholar] [CrossRef]
- Bauenova, M.O.; Sadvakasova, A.K.; Mustapayeva, Z.O.; Kokociński, M.; Zayadan, B.K.; Wojciechowicz, M.K.; Balouch, H.; Akmukhanova, N.R.; Alwasel, S.; Allakhverdiev, S.I. Potential of microalgae Parachlorella kessleri Bh-2 as bioremediation agent of heavy metals cadmium and chromium. Algal Res. 2021, 59, 102463. [Google Scholar] [CrossRef]
- Kadukova, J. Surface sorption and nanoparticle production as a silver detoxification mechanism of the freshwater alga Parachlorella kessleri. Bioresour. Technol. 2016, 216, 406–413. [Google Scholar] [CrossRef]
- Velgosova, O.; Anna, M.; Elena, C.; Jaroslav, M. Green synthesis of Ag nanoparticles: Effect of algae life cycle on Ag nanoparticle production and long-term stability. Trans. Nonferr. Metal. Soc. 2018, 28, 974–979. [Google Scholar] [CrossRef]
- Najafabadi, H.A.; Malekzadeh, M.; Jalilian, F.; Vossoughi, M.; Pazuki, G. Effect of various carbon sources on biomass and lipid production of Chlorella vulgaris during nutrient sufficient and nitrogen starvation conditions. Bioresour. Technol. 2015, 180, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Ananthi, V.; Raja, R.; Carvalho, I.S.; Brindhadevi, K.; Pugazhendhi, A.; Arun, A. A realistic scenario on microalgae based biodiesel production: Third generation biofuel. Fuel 2021, 284, 118965. [Google Scholar] [CrossRef]
- He, Y.; Hong, Y.; Liu, X.; Zhang, Q.; Liu, P.; Wang, S. Influences of carbon and nitrogen sources and metal ions on the heterotrophic culture of Scenedesmus sp. LX1. Environ. Sci. Pollut. R 2019, 26, 13381–13389. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.F.; Hu, H.; Ma, L.L. FAMEs production from Scenedesmus obliquus in autotrophic, heterotrophic and mixotrophic cultures under different nitrogen conditions. Environ. Sci. Wat. Res. 2018, 4, 461–468. [Google Scholar] [CrossRef]
- Oliveira, C.Y.B.; D’Alessandro, E.B.; Filho, N.R.A.; Lopes, R.G.; Derner, R.B. Synergistic effect of growth conditions and organic carbon sources for improving biomass production and biodiesel quality by the microalga Choricystis minor var. minor. Sci. Total Environ. 2021, 759, 143476. [Google Scholar] [CrossRef]
- Leon-Vaz, J.R.S. Using agro-industrial wastes for mixotrophic growth and lipids production by the green microalga Chlorella sorokiniana. N. Biotechnol. 2019, 51, 31–38. [Google Scholar] [CrossRef]
- Ende, S.S.W.; Noke, A. Heterotrophic microalgae production on food waste and by-products. J. Appl. Phycol. 2019, 31, 1565–1571. [Google Scholar] [CrossRef]
- Katiyar, R.; Gurjar, B.R.; Bharti, R.K.; Kumar, A.; Biswas, S.; Pruthi, V. Heterotrophic cultivation of microalgae in photobioreactor using low cost crude glycerol for enhanced biodiesel production. Renew. Energy 2018, 113, 1359–1365. [Google Scholar] [CrossRef]
- Lin, T.S.; Wu, J.Y. Effect of carbon sources on growth and lipid accumulation of newly isolated microalgae cultured under mixotrophic condition. Bioresour. Technol. 2015, 184, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Shu, C.H.; Tsai, C.C. Enhancing oil accumulation of a mixed culture of Chlorella sp. and Saccharomyces cerevisiae using fish waste hydrolysate. J. Taiwan Inst. Chem. E 2016, 67, 377–384. [Google Scholar] [CrossRef]
- Silva Benavides, A.M.; Campos Rudin, M.; Villalobos, N.; Touloupakis, E.; Torzillo, G. Growth and hydrogen production by three Chlamydomonas strains cultivated in a commercial fertilizer. Int. J. Hydrogen. Energ. 2019, 44, 9849–9855. [Google Scholar] [CrossRef]
- Lv, J.P.; Guo, J.Y.; Feng, J.; Liu, Q.; Xie, S.L. Effect of sulfate ions on growth and pollutants removal of self-flocculating microalga Chlorococcum sp. GD in synthetic municipal wastewater. Bioresour. Technol. 2017, 234, 289–296. [Google Scholar] [CrossRef]
- Mera, R.; Torres, E.; Abalde, J. Effects of sodium sulfate on the freshwater microalga Chlamydomonas moewusii: Implications for the optimization of algal culture media. J. Phycol. 2016, 52, 75–88. [Google Scholar] [CrossRef]
- Markou, G.; Muylaert, K. Effect of light intensity on the degree of ammonia toxicity on PSII activity of Arthrospira platensis and Chlorella vulgaris. Bioresour. Technol. 2016, 216, 453–461. [Google Scholar] [CrossRef]
- Li, H.S. Principles and Techniques of Plant Physiological and Biochemical Experiments; Higher Education Press: Beijing, China, 2000; pp. 184–199. [Google Scholar]
- Prajapati, S.K.; Kaushik, P.; Malik, A.; Vijay, V.K. Phycoremediation and biogas potential of native algal isolates from soil and wastewater. Bioresour. Technol. 2013, 135, 232–238. [Google Scholar] [CrossRef]
- He, Q.N.; Yang, H.J.; Hu, C.X. Effects of temperature and its combination with high light intensity on lipid production of Monoraphidium dybowskii Y2 from semi-arid desert areas. Bioresour. Technol. 2018, 265, 407–414. [Google Scholar] [CrossRef]
- Guo, Y.N. Optimizing Cultivation and Mechanism of Lipid Accumulation Research in Chlorococcum sphacosum GD. Master’s Thesis, Shanxi University, Taiyuan, China, 2017. [Google Scholar]
- Chen, W.; Zhang, C.; Song, L.; Sommerfeld, M.; Hua, Q. A high throughput Nile red method for quantitative measurement of neutral lipids in microalgae. J. Microbiol. Meth. 2015, 77, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Karima, A.; Silalahi, M.D.; Rinanti, A. Increasing content of lipid in tropical microalgae Chlorella sorokiniana and Closterium sp. with variation of nitrogen content and extraction temperature. Appl. Sci. Eng. Conf. 2018, 197, 13019. [Google Scholar] [CrossRef]
- Thang, D.V.; Faruq, A.; Thomas-Hall, S.R.; Simon, Q.; Ekaterina, N.; Schenk, P.M. High protein and high lipid-producing microalgae from northern Australia as potential feedstock for animal feed and biodiesel. Front. Bioeng. Biotech. 2015, 3, 53–60. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.; Sun, Z.; Zhu, Y.J. Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: Assessment of algal oils for biodiesel production. Bioresour. Technol. 2011, 102, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.X.; Deng, D.Y.; Liu, J.P.; Cheng, K.; Xu, Y.J. Field trial multiple comparison results alphabetic difference counting method and its letter abbreviation. Seeds 2018, 5, 131–132. [Google Scholar] [CrossRef]
- Yan, W.J. Characteristics of Coccomyxa subellipsoidea C-169 in Trophic Growth and Evaluation of Efficiency on Yeast Wastewater Treatment. Master’s Thesis, South China University of Technology, Guangzhou, China, 2016. [Google Scholar]
- Li, C.L.; Yang, H.L.; Li, Y.J.; Wang, W. Effect of culture models on metabolism and protein components of microalgae Chlorella vulgaris. J. Food Sci. Biotechnol. 2014, 33, 56–62. [Google Scholar] [CrossRef]
- Shokravi, Z.; Shokravi, H.; Atabani, A.E.; Lau, W.J.; Chyuan, O.H.; Ismail, A.F. Impacts of the harvesting process on microalgae fatty acid profiles and lipid yields: Implications for biodiesel production. Renew. Sust. Energy Rev. 2022, 161, 112410. [Google Scholar] [CrossRef]
- Li, T.T.; Zheng, Y.; Yu, L.; Chen, S.L. Mixotrophic cultivation of a Chlorella sorokiniana strain for enhanced biomass and lipid production. Biomass Bioenergy 2014, 66, 204–213. [Google Scholar] [CrossRef]
- Abreu, A.P.; Morais, R.C.; Kazmerski, L. A comparison between microalgal autotrophic growth and metabolite accumulation with heterotrophic, mixotrophic and photoheterotrophic cultivation modes. Renew. Sust. Energy Rev. 2022, 159, 112247. [Google Scholar] [CrossRef]
- Wang, J.; Yang, H.; Wang, F. Mixotrophic cultivation of microalgae for biodiesel production: Status and prospects. Appl. Biochem. Biotechnol. 2014, 172, 3307–3329. [Google Scholar] [CrossRef]
- Yan, S.K. The Effect of Nitrogen and Phosphorus Conditions and Culture Modes on the Growth and Lipid Content of Microalgae. Master’s Thesis, Anhui Normal University, Hefei, China, 2019. [Google Scholar]
- Ma, D.D.; Li, Y.F. Heterotrophic culture of Chlorella sp. and Dunaliella salina using alginate oligosaccharide and its growth promoting mechanism. Period. Ocean. Univ. China 2020, 50, 40–45. [Google Scholar] [CrossRef]
- Kabir, F.; Gulfraz, M.; Raja, G.K.; Inam-Ul-Haq, M.; Shadloo, M.S. Screening of native hyper-lipid producing microalgae strains for biomass and lipid production. Renew. Energy 2020, 160, 1295–1307. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, D.; Wu, T.; Lo, Y.; Lee, Y.; Liu, J. The synergistic energy and carbon metabolism under mixotrophic cultivation reveals the coordination between photosynthesis and aerobic respiration in Chlorella zofingiensis. Algal Res. 2017, 25, 109–116. [Google Scholar] [CrossRef]
- Vidotti, A.D.S.; Riano-Pachon, D.M.; Mattiello, L.; Giraldi, L.A.; Franco, T.T. Analysis of autotrophic, mixotrophic and heterotrophic phenotypes in the microalgae Chlorella vulgaris using time-resolved proteomics and transcriptomics approaches. Algal Res. 2020, 51, 102060. [Google Scholar] [CrossRef]
- El-Sheekh, M.M.; Bedaiwy, M.Y.; Osman, M.E.; Ismail, M.M. Mixotrophic and heterotrophic growth of some microalgae using extract of fungal-treated wheat bran. Int. J. Recycl. Org. Waste Agric. 2012, 1, 12. [Google Scholar] [CrossRef]
- Lewitus, A.J.; Kana, T.M. Light respiration in six estuarine phytoplankton species: Contrasts under photoautotrophic and mixotrophic growth conditions. J. Phycol. 2010, 31, 754–761. [Google Scholar] [CrossRef]
- Huang, Y.; Li, P.; Huang, Y.; Xia, A.; Zhu, X.; Liao, Q. A synchronous photoautotrophic-heterotrophic biofilm cultivation mode for chlorella vulgaris biomass and lipid simultaneous accumulation. J. Clean. Prod. 2022, 336, 130453. [Google Scholar] [CrossRef]
- Karimian, A.; Mahdavi, M.A.; Gheshlaghi, R. Algal cultivation strategies for enhancing production of Chlorella sorokiniana IG-W-96 biomass and bioproducts. Algal Res. 2022, 62, 102630. [Google Scholar] [CrossRef]
- Liu, X.J. Charaeteristics of Photoautotrophy, Mixotrophy and Heterotrophy of Phaeodactylum tricornutum. Ph.D. Thesis, Jinan University, Guangzhou, China, 2008; pp. 58–88. [Google Scholar]
- Pleissner, D.; Eriksen, N.T. Effects of phosphorous, nitrogen, and carbon limitation on biomass composition in batch and continuous flow cultures of the heterotrophic dinoflagellate Crypthecodinium cohnii. Biotechnol. Bioeng. 2012, 109, 2005–2016. [Google Scholar] [CrossRef]
- Ogbonna, J.C.; Tanaka, H. Production of pure photosynthetic cell biomass for environmental biosensors. Mat. Sci. Eng. 2000, 12, 9–15. [Google Scholar] [CrossRef]
- Pereira, M.I.B.; Chagas, B.M.E.; Sassi, R.; Medeiros, G.F.; Rangel, A.H.N. Mixotrophic cultivation of Spirulina platensis in dairy wastewater: Effects on the production of biomass, biochemical composition and antioxidant capacity. PLoS ONE 2019, 14, e0224294. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.K.; Sahoo, P.K.; Singhal, S.; Patel, A. Impact of various media and organic carbon sources on biofuel production potential from Chlorella spp. 3 Biotech 2016, 6, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Pancha, I.; Chokshi, K.; George, B.; Ghosh, T.; Paliwal, C.; Maurya, R.; Mishra, S. Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour. Technol. 2014, 156, 146–154. [Google Scholar] [CrossRef]
- Rios, L.F.; Klein, B.C.; Luz, L.F.J.; Filho, R.M.; Maciel, M.R.F. Nitrogen starvation for lipid accumulation in the microalga species Desmodesmus sp. Appl. Biochem. Biotechnol. 2015, 175, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Siaut, M.; Cuine, S.; Cagnon, C.; Fessler, B.; Nguyen, M.; Carrier, P.; Beyly, A.; Beisson, F. Oil accumulation in the model green alga Chlamydomonas reinhardtii: Characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol. 2011, 11, 7. [Google Scholar] [CrossRef]
- Zhu, S.N.; Huang, W.; Xu, J.; Wang, Z.M.; Xu, J.L.; Yuan, Z.H. Metabolic changes of starch and lipid triggered by nitrogen starvation in the microalga Chlorella zofingiensis. Bioresour. Technol. 2014, 152, 292–298. [Google Scholar] [CrossRef]
- Yun, H.S.; Kim, Y.S.; Yoon, H.S. Effect of different cultivation modes (photoautotrophic, mixotrophic, and heterotrophic) on the growth of Chlorella sp. and biocompositions. Front. Bioeng. Biotechnol. 2021, 9, 774143. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.; Fan, K.W.; Jiang, Y.; Zhong, Y.; Sun, Z.; Chen, F. Production potential of Chlorella zofingienesis as a feedstock for biodiesel. Bioresour. Technol. 2010, 101, 8658–8663. [Google Scholar] [CrossRef]
- Cheng, P.F.; Huang, J.; Song, X.; Yao, T.; Jiang, J.; Zhou, C.; Yan, X.J.; Ruan, R. Heterotrophic and mixotrophic cultivation of microalgae to simultaneously achieve furfural wastewater treatment and lipid production. Bioresour. Technol. 2022, 349, 126888. [Google Scholar] [CrossRef]
- Leong, W.H.; Saman, N.A.M.; Kiatkittipong, W.; Assabumrungrat, S.; Najdanovic-Visak, V.; Wang, J. Photoperiod-induced mixotrophic metabolism in Chlorella vulgaris for high biomass and lipid to biodiesel productions using municipal wastewater medium. Fuel J. Fuel Sci. 2022, 313, 123052. [Google Scholar] [CrossRef]
- Ahmad, F.T.; Seyed, K.M.; Meisam, T.; Masoud, T.; Abdolreza, B.; Mehrshad, Z.; Hossein, H.M.; Mehrdad, M.; Saeid, M.; Shiva, B. Fatty acids profiling: A selective criterion for screening microalgae strains for biodiesel production. Algal Res. 2013, 2, 258–267. [Google Scholar] [CrossRef]


| Trophic Modes | Biomass Productivity (mg·L−1d−1) |
|---|---|
| autotrophic | 105.76 b ± 1.22 |
| heterotrophic | 77.95 c ± 5.82 |
| mixotrophic | 218.43 a ± 10.00 |
| Trophic Modes | Lipid Productivity (mg·L−1 d−1) |
|---|---|
| autotrophic | 35.69 b ± 0.78 |
| heterotrophic | 31.00 c ± 0.96 |
| mixotrophic | 93.61 a ± 0.37 |
| Fatty Acids Composition | Autotrophic (% DW) | Heterotrophic (% DW) | Mixotrophic (% DW) |
|---|---|---|---|
| C16:0 | 22.00 c ± 0.26 | 23.60 b ± 0.17 | 24.00 a ± 0.07 |
| C16:1 | 2.30 c ± 0.05 | 3.31 b ± 0.07 | 3.91 a ± 0.05 |
| C16:2 | 6.81 ± 0.06 | - | - |
| C18:0 | 7.71 c ± 0.05 | 8.00 b ± 0.08 | 8.32 a ± 0.05 |
| C18:1 | 5.31 c ± 0.14 | 15.51 b ± 0.05 | 15.84 a ± 0.03 |
| C18:2 | 14.64 c ± 0.11 | 20.50 b ± 0.06 | 21.10 a ± 0.03 |
| C18:3 | 28.79 a ± 0.25 | 19.70 b ± 0.07 | 17.95 c ± 0.11 |
| C16 + C18 | 87.56 b ± 0.50 | 90.60 a ± 0.08 | 91.12 a ± 0.06 |
| others | 12.44 a ± 0.50 | 9.40 b ± 0.80 | 8.88 b ± 0.06 |
| SFA | 33.93 c ± 0.06 | 34.88 b ± 0.24 | 35.47 a ± 0.15 |
| MUFA | 8.69 c ± 0.06 | 20.77 b ± 0.14 | 21.67 a ± 0.08 |
| PUFA | 57.38 a ± 0.11 | 44.37 b ± 0.06 | 42.85 c ± 0.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Y.; Li, Y.; Yang, Y.; Feng, J.; Ji, L.; Xie, S. Effects of Trophic Modes on the Lipid Accumulation of Parachlorella kessleri TY. Fermentation 2023, 9, 891. https://doi.org/10.3390/fermentation9100891
Gao Y, Li Y, Yang Y, Feng J, Ji L, Xie S. Effects of Trophic Modes on the Lipid Accumulation of Parachlorella kessleri TY. Fermentation. 2023; 9(10):891. https://doi.org/10.3390/fermentation9100891
Chicago/Turabian StyleGao, Yifan, Yuan Li, Yan Yang, Jia Feng, Li Ji, and Shulian Xie. 2023. "Effects of Trophic Modes on the Lipid Accumulation of Parachlorella kessleri TY" Fermentation 9, no. 10: 891. https://doi.org/10.3390/fermentation9100891
APA StyleGao, Y., Li, Y., Yang, Y., Feng, J., Ji, L., & Xie, S. (2023). Effects of Trophic Modes on the Lipid Accumulation of Parachlorella kessleri TY. Fermentation, 9(10), 891. https://doi.org/10.3390/fermentation9100891






