Anaerobic Co-Digestion of Cattle Manure and Brewer’s Residual Yeast: Process Stability and Methane and Hydrogen Sulfide Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Inoculum and Feedstock
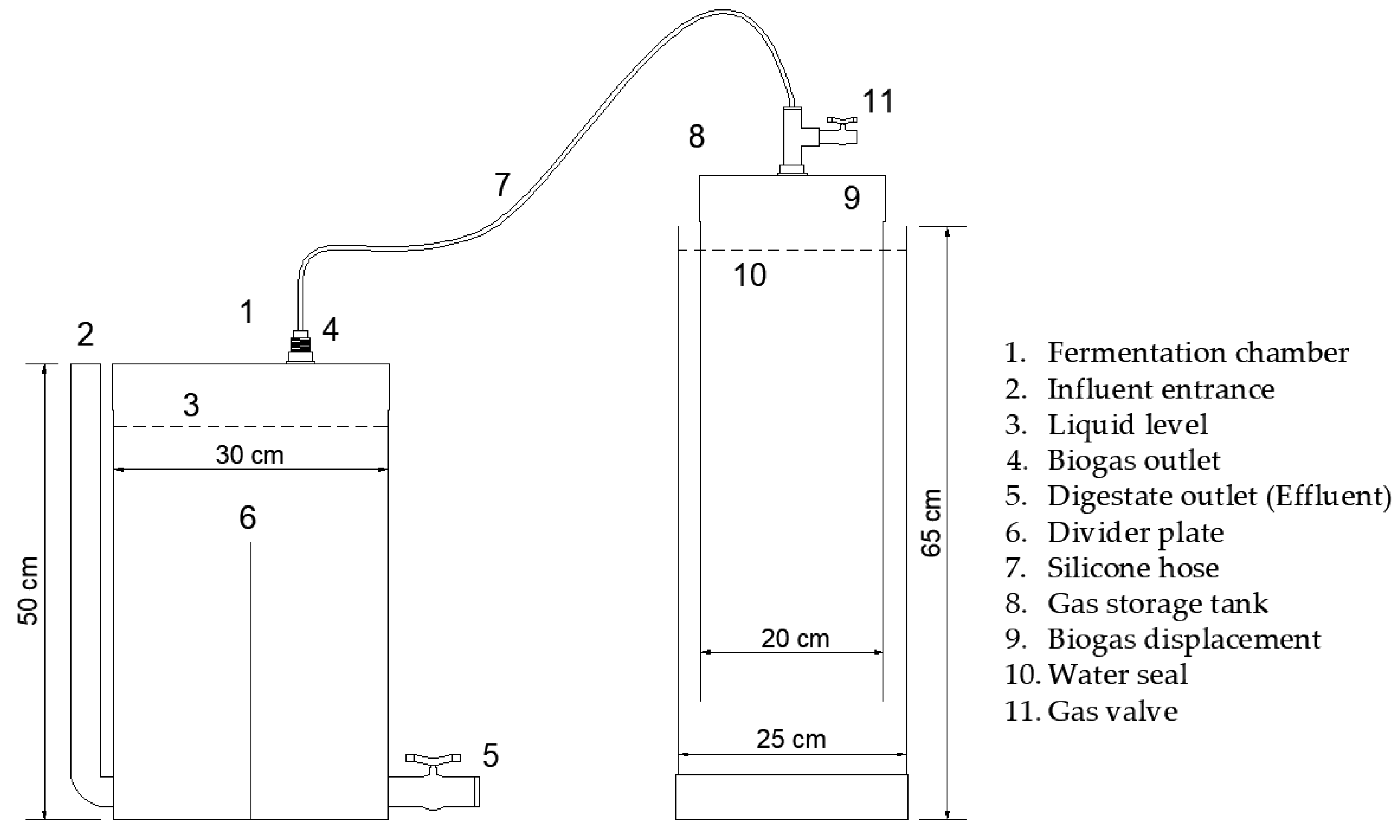
2.3. Semi-Continuous Biodigester Description
2.4. Treatment Descriptions
2.5. Analytical Methods
2.6. Biogas Monitoring
2.7. Statistical Analysis
3. Results and Discussion
Multivariate Analysis: Clusters and Principal Components
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CIBiogás. Centro Internacional de Energias Renováveis—Biogás. In Panorama do Biogás no Brasil 2022; CIBiogás (Brasil) Relatório Técnico no 001/2023; CIBiogás: Foz do Iguaçu, Brazil, 2023; p. 12. Available online: https://materiais.cibiogas.org/webinar-panorama-do-biogas-no-brasil-2022 (accessed on 19 October 2023).
- ABiogás. Associação Brasileira de Biogás e Biometano. ABiogás Divulga novo Potencial do Biogás para o Mercado Brasileiro. São Paulo. 2021. Available online: https://abiogas.org.br/potencial-do-biogas-no-brasil (accessed on 19 October 2023).
- Júnior, M.A.P.O.; Orrico, A.C.A.; de Lucas Júnior, J. Produção animal e o meio ambiente: Uma comparação entre potencial de emissão de metano dos dejetos e a quantidade de alimento produzido. Eng. Agríc. 2011, 31, 399–410. [Google Scholar] [CrossRef]
- Hagos, K.; Zong, J.; Li, D.; Liu, C.; Lu, X. Anaerobic co-digestion process for biogas production: Progress, challenges and perspectives. Renew. Sustain. Energy Rev. 2017, 76, 1485–1496. [Google Scholar] [CrossRef]
- Neshat, S.A.; Mohammadi, M.; Najafpour, G.D.; Lahijani, P. Anaerobic co-digestion of animal manures and lignocellulosic residues as a potent approach for sustainable biogas production. Renew. Sustain. Energy Rev. 2017, 79, 308–322. [Google Scholar] [CrossRef]
- Dareioti, M.A.; Tsigkou, K.; Vavouraki, A.I.; Kornaros, M. Hydrogen and Methane Production from Anaerobic Co-Digestion of Sorghum and Cow Manure: Effect of pH and Hydraulic Retention Time. Fermentation 2022, 8, 304. [Google Scholar] [CrossRef]
- Kunatsa, T.; Zhang, L.; Xia, X. Biogas potential determination and production optimisation through optimal substrate ratio feeding in co-digestion of water hyacinth, municipal solid waste and cow dung. Biofuels 2022, 13, 631–641. [Google Scholar] [CrossRef]
- Abid, M.; Wu, J.; Seyedsalehi, M.; Hu, Y.Y.; Tian, G. Novel insights of impacts of solid content on high solid anaerobic digestion of cow manure: Kinetics and microbial community dynamics. Bioresour. Technol. 2021, 333, 125205. [Google Scholar] [CrossRef]
- Song, Y.; Qiao, W.; Westerholm, M.; Huang, G.; Taherzadeh, M.J.; Dong, R. Microbiological and Technological Insights on Anaerobic Digestion of Animal Manure: A Review. Fermentation 2023, 9, 436. [Google Scholar] [CrossRef]
- Jiang, Y.; McAdam, E.; Zhang, Y.; Heaven, S.; Banks, C.; Longhurst, P. Ammonia inhibition and toxicity in anaerobic digestion: A critical review. J. Water Process Eng. 2019, 32, 100899. [Google Scholar] [CrossRef]
- Xu, S.; Bi, G.; Liu, X.; Yu, Q.; Li, D.; Yuan, H.; Chen, Y.; Xie, J. Anaerobic Co-Digestion of Sugarcane Leaves, Cow Dung and Food Waste: Focus on Methane Yield and Synergistic Effects. Fermentation 2022, 8, 399. [Google Scholar] [CrossRef]
- Montoro, S.B.; Lucas, J.; Santos, D.F.L.; Costa, M.S.S.M. Anaerobic co-digestion of sweet potato and dairy cattle manure: A technical and economic evaluation for energy and biofertilizer production. J. Clean. Prod. 2019, 226, 1082–1091. [Google Scholar] [CrossRef]
- Rabii, A.; Aldin, S.; Dahman, Y.; Elbeshbishy, E. A review on anaerobic co-digestion with a focus on the microbial populations and the effect of multi-stage digester configuration. Energies 2019, 12, 1106. [Google Scholar] [CrossRef]
- Piñas, J.A.V.; Venturini, O.J.; Silva Lora, E.E.; Calle Roalcaba, O.D. Technical assessment of mono-digestion and co-digestion systems for the production of biogas from anaerobic digestion in Brazil. Renew. Energy 2018, 117, 447–458. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Dosta, J.; Romero-Güiza, M.S.; Fonoll, X.; Peces, M.; Astals, S. A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew. Sustain. Energy Rev. 2014, 36, 412–427. [Google Scholar] [CrossRef]
- Karki, R.; Chuenchart, W.; Surendra, K.C.; Shrestha, S.; Raskin, L.; Sung, S.; Hashimoto, A.; Khanal, S.K. Anaerobic co-digestion: Current status and perspectives. Bioresour. Technol. 2021, 330, 125001. [Google Scholar] [CrossRef]
- Manyi-Loh, C.E.; Lues, R. Anaerobic Digestion of Lignocellulosic Biomass: Substrate Characteristics (Challenge) and Innovation. Fermentation 2023, 9, 755. [Google Scholar] [CrossRef]
- Ma, G.; Ndegwa, P.; Harrison, J.H.; Chen, Y. Methane yields during anaerobic co-digestion of animal manure with other feedstocks: A meta-analysis. Sci. Total Environ. 2020, 728, 138224. [Google Scholar] [CrossRef]
- Neira, K.; Jeison, D. Anaerobic co-digestion of surplus yeast and wastewater to increase energy recovery in breweries. Water Sci. Technol. 2010, 61, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Ekpeni, L.E.N.; Nkem-Ekpeni, F.F.; Benyounis, K.Y.; Aboderheeba, A.K.M.; Stokes, J.; Olabi, A.G. Yeast: A potential biomass substrate for the production of cleaner energy (Biogas). Energy Procedia 2014, 61, 1718–1731. [Google Scholar] [CrossRef][Green Version]
- Sosa-Hernández, O.; Parameswaran, P.; Alemán-Nava, G.S.; Torres, C.I.; Parra-Saldívar, R. Evaluating biochemical methane production from brewer’s spent yeast. J. Ind. Microbiol. Biotechnol. 2016, 43, 1195–1204. Available online: https://academic.oup.com/jimb/article/43/9/1195/5995653 (accessed on 19 October 2023). [CrossRef]
- Pettigrew, L.; Gutbrod, A.; Domes, H.; Groß, F.; Méndez-Contreras, J.M.; Delgado, A. Modified ADM1 for high-rate anaerobic co-digestion of thermally pre-treated brewery surplus yeast wastewater. Water Sci. Technol. 2017, 76, 542–554. [Google Scholar] [CrossRef]
- Vitanza, R.; Cortesi, A.; Gallo, V.; Colussi, I.; De Arana-Sarabia, M.E. Biovalorization of brewery waste by applying anaerobic digestion. Chem. Biochem. Eng. Q. 2016, 30, 351–357. [Google Scholar] [CrossRef]
- Mainardis, M.; Flaibani, S.; Mazzolini, F.; Peressotti, A.; Goi, D. Techno-economic analysis of anaerobic digestion implementation in small Italian breweries and evaluation of biochar and granular activated carbon addition effect on methane yield. J. Environ. Chem. Eng. 2019, 7, 103184. [Google Scholar] [CrossRef]
- Caballero-Córdoba, G.M.; Sgarbieri, V.C. Nutritional and toxicological evaluation of yeast (Saccharomyces cerevisiae) biomass and a yeast protein concentrate. J. Sci. Food Agric. 2000, 80, 341–351. [Google Scholar] [CrossRef]
- Costa, A.G.; Magnani, M.; Castro-Gomez, R.J.H. Obtenção e caracterização de manoproteínas da parede celular de leveduras de descarte em cervejaria. Acta Sci. Biol. Sci. 2012, 34, 77–84. [Google Scholar] [CrossRef]
- Jacob, F.F.; Striegel, L.; Rychlik, M.; Hutzler, M.; Methner, F.J. Yeast extract production using spent yeast from beer manufacture: Influence of industrially applicable disruption methods on selected substance groups with biotechnological relevance. Eur. Food Res. Technol. 2019, 245, 1169–1182. [Google Scholar] [CrossRef]
- Ferreira, I.M.P.L.V.O.; Pinho, O.; Vieira, E.; Tavarela, J.G. Brewer’s Saccharomyces yeast biomass: Characteristics and potential applications. Trends Food Sci. Technol. 2010, 21, 77–84. [Google Scholar] [CrossRef]
- Fillaudeau, L.; Blanpain-Avet, P.; Daufin, G. Water, wastewater and waste management in brewing industries. J. Clean. Prod. 2006, 14, 463–471. [Google Scholar] [CrossRef]
- Pinto, M.; Coelho, E.; Nunes, A.; Brandão, T.; Coimbra, M.A. Valuation of brewers spent yeast polysaccharides: A structural characterization approach. Carbohydr. Polym. 2015, 116, 215–222. [Google Scholar] [CrossRef]
- Jaeger, A.; Arendt, E.K.; Zannini, E.; Sahin, A.W. Brewer’s Spent Yeast (BSY), an Underutilized Brewing By-Product. Fermentation 2020, 6, 123. [Google Scholar] [CrossRef]
- Huige, N.J. Brewery By-Products and Effluents. In Handbook of Brewing, 2nd ed.; Priest, F.G., Steward, G.G., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 656–713. Available online: https://www.taylorfrancis.com/chapters/edit/10.1201/9781420015171-22/brewery-products-effluents-nick-huige (accessed on 19 October 2023).
- Bocher, B.T.; Agler, M.T.; Garcia, M.L.; Beers, A.R.; Angenent, L.T. Anaerobic digestion of secondary residuals from an anaerobic bioreactor at a brewery to enhance bioenergy generation. J. Ind. Microbiol. Biotechnol. 2008, 35, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Zupančič, G.D.; Škrjanec, I.; Marinšek Logar, R. Anaerobic co-digestion of excess brewery yeast in a granular biomass reactor to enhance the production of biomethane. Bioresour. Technol. 2012, 124, 328–337. [Google Scholar] [CrossRef]
- Oliveira, J.V.; Alves, M.M.; Costa, J.C. Biochemical methane potential of brewery by-products. Clean Technol. Environ. Policy 2018, 20, 435–440. [Google Scholar] [CrossRef]
- Suwannarat, J.; Ritchie, R.J. Anaerobic digestion of food waste using yeast. Waste Manag. 2015, 42, 61–66. [Google Scholar] [CrossRef]
- Syaichurrozi, I.; Rusdi, R.; Hidayat, T.; Bustomi, A. Kinetics studies impact of initial pH and addition of yeast Saccharomyces cerevisiae on biogas production from tofu wastewater in Indonesia. Int. J. Eng. Trans. B Appl. 2016, 29, 1037–1046. [Google Scholar]
- Li, D.; Song, L.; Fang, H.; Shi, Y.; Li, Y.-Y.; Liu, R.; Niu, Q. Effect of temperature on the anaerobic digestion of cardboard with waste yeast added: Dose-response kinetic assays, temperature coefficient and microbial co-metabolism. J. Clean. Prod. 2020, 275, 122949. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0959652620329942 (accessed on 19 October 2023). [CrossRef]
- Rasmeni, Z.Z.; Madyira, D.M.; Matheri, A.N. Optimum loading ratio for co-digested wastewater sludge and brewery spent yeast. Energy Rep. 2022, 8, 1141–1149. [Google Scholar] [CrossRef]
- Islas-Espinoza, M.; de las Heras, A.; Vázquez-Chagoyán, J.C.; Salem, A.Z.M. Anaerobic cometabolism of fruit and vegetable wastes using mammalian fecal inoculums: Fast assessment of biomethane production. J. Clean. Prod. 2017, 141, 1411–1418. [Google Scholar] [CrossRef]
- Amaral, A.C.d.; Steinmetz, R.L.R.; Kunz, A. (Eds.) Os biodigestores. In Fundamentos da Digestão Anaeróbia, Purificação do Biogás, Uso e Tratamento do Digestato, 2nd ed.; Sbera, Embrapa Suínos e Aves: Concórdia, Brazil, 2022; pp. 43–70. Available online: http://www.alice.cnptia.embrapa.br/alice/handle/doc/1141618 (accessed on 19 October 2023).
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 21st ed.; Andrew, D., Eaton, M.A.H.F., Eds.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2005; 8p. [Google Scholar]
- Ripley, L.E.; Boyle, W.C.; Converse, J.C. Improved Alkalimetric Monitoring for Anaerobic Digestion of High-Strength Wastes. J. Water Pollut. Control Fed. 1986, 58, 406–411. Available online: http://www.jstor.org/stable/25042933 (accessed on 19 October 2023).
- Carmo, D.L.d.; Silva, C.A. Métodos de quantificação de carbono e matéria orgânica em resíduos orgânicos. Rev. Bras. Cienc. do Solo. 2012, 36, 1211–1220. [Google Scholar] [CrossRef]
- Vieira, W.; Silva, F.C.d. Análises de fertilizantes minerais, organominerais e corretivos. In Manual de Análises Químicas de Solos, Plantas e Fertilizantes, 2nd ed.; da Silva, F.C., Ed.; Embrapa Informação Tecnológica: Brasília, Brazil, 2009; p. 627. [Google Scholar]
- Hongyu, K.; Sandanielo, V.L.M.; de Oliveira Junior, G.J. Análise de Componentes Principais: Resumo Teórico, Aplicação e Interpretação. E&S Eng. Sci. 2016, 5, 83–90. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: http://www.r-project.org/index.html (accessed on 19 October 2023).
- Tewelde, S.; Eyalarasan, K.; Radhamani, R.; Karthikeyan, K. Biogas Production from Co-digestion of Brewery Wastes [BW] and Cattle Dung [CD]. Int. J. Latest Trends Agric. Food Sci. 2012, 2, 90–93. [Google Scholar]
- Akyol, Ç.; Ince, O.; Bozan, M.; Ozbayram, E.G.; Ince, B. Fungal bioaugmentation of anaerobic digesters fed with lignocellulosic biomass: What to expect from anaerobic fungus Orpinomyces sp. Bioresour. Technol. 2019, 277, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.K. Bioenergy Generation from Residues of Biofuel Industries. In Anaerobic Biotechnology for Bioenergy Production: Principles and Applications; Khanal, S.K., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 161–188. [Google Scholar] [CrossRef]
- Caballero-Córdoba, G.M.; Pacheco, M.T.B.; Sgarbieri, V.C. Composição química da biomassa de levedura integral (Saccharomyces sp.) e determinação do valor nutritivo da proteína em células íntegras ou rompidas mecanicamente. Food Sci. Technol. 1997, 17, 102–106. [Google Scholar] [CrossRef]
- Callado, N.H.; Damianovic, M.H.Z.; Foresti, E. Influência da razão DQO/[SO42-] e da concentração de Na+ na remoção de matéria orgânica e sulfato em reator UASB. Eng. Sanit. Ambient. 2017, 22, 381–390. [Google Scholar] [CrossRef]
- Westerholm, M.; Hansson, M.; Schnürer, A. Improved biogas production from whole stillage by co-digestion with cattle manure. Bioresour. Technol. 2012, 114, 314–319. [Google Scholar] [CrossRef]
- Paula, D.R.; Foresti, E. Sulfide toxicity kinetics of a uasb reactor. Brazilian J. Chem. Eng. 2009, 26, 669–675. [Google Scholar] [CrossRef]
- Deublein, D.; Steinhauser, A. Gas Preparation. In Biogas from Waste and Renewable Resources; Deublein, D., Steinhauser, A., Eds.; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 333–355. Available online: https://onlinelibrary.wiley.com/doi/book/10.1002/9783527621705 (accessed on 19 October 2023).
- Silva, M.L.B.d.; Mezzari, M.P. Tratamento e purificação de biogás. In Fundamentos da Digestão Anaeróbia, Purificação do Biogás, Uso e Tratamento do Digestato, 2nd ed.; Kunz, A., Steinmetz, R.L.R., do Amaral, A.C., Eds.; Sbera, Embrapa Suínos e Aves: Concórdia, Brazil, 2022; pp. 71–95. Available online: http://www.alice.cnptia.embrapa.br/alice/handle/doc/1141618 (accessed on 19 October 2023).
- ANP. Agência Nacional do Petróleo, Gás Natural e Biocombustíveis. Resolução ANP n. 886, de 29 de Setembro de 2022. Brasília; 2022. Available online: https://www.in.gov.br/en/web/dou/-/resolucao-anp-n-886-de-29-de-setembro-de-2022-432620215 (accessed on 19 October 2023).
- Sun, Q.; Li, H.; Yan, J.; Liu, L.; Yu, Z.; Yu, X. Selection of appropriate biogas upgrading technology-a review of biogas cleaning, upgrading and utilisation. Renew. Sustain. Energy Rev. 2015, 51, 521–532. [Google Scholar] [CrossRef]
- Akhiar, A.; Battimelli, A.; Torrijos, M.; Carrere, H. Comprehensive characterization of the liquid fraction of digestates from full-scale anaerobic co-digestion. Waste Manag. 2017, 59, 118–128. [Google Scholar] [CrossRef]
- Amaral, A.C.d.; Steinmetz, R.L.R.; Kunz, A. O processo de biodigestão. In Fundamentos da Digestão Anaeróbia, Purificação do Biogás, Uso e Tratamento do Digestato, 2nd ed.; Kunz, A., Steinmetz, R.L.R., do Amaral, A.C., Eds.; Sbera, Embrapa Suínos e Aves: Concórdia, Brazil, 2022; pp. 15–28. Available online: http://www.alice.cnptia.embrapa.br/alice/handle/doc/1141618 (accessed on 19 October 2023).
- Huang, X.; Yun, S.; Zhu, J.; Du, T.; Zhang, C.; Li, X. Mesophilic anaerobic co-digestion of aloe peel waste with dairy manure in the batch digester: Focusing on mixing ratios and digestate stability. Bioresour. Technol. 2016, 218, 62–68. [Google Scholar] [CrossRef] [PubMed]





| Parameters | Fresh Cattle Manure | CM * | RY | |
|---|---|---|---|---|
| pH | pH unit | 6.10 ± 0.20 | 6.88 ± 0.27 | 4.30 ± 0.21 |
| Total solids | % (NM) | 15.10 ± 0.79 | 1.51 ± 0.13 | 17.63 ± 0.47 |
| Volatile solids | % of the TS | 84.37 ± 2.64 | 72.95 ± 2.96 | 97.80 ± 0.80 |
| Ash | % of the TS | 15.70 ± 3.33 | 26.82 ± 2.79 | 2.20 ± 0.47 |
| COD | g O2 L−1 | 58.42 ± 8.79 | 12.09 ± 0.8 | 268.96 ± 35.10 |
| TOC | % of the TS | 46.87 ± 1.46 | 40.53 ± 1.64 | 54.33 ± 0.45 |
| TKN | % of the TS | 3.00 ± 0.51 | 1.61 ± 0.14 | 7.11 ± 0,12 |
| Phosphorus | % of the TS | - | 2.45 ± 0.09 | 0.95 ± 0.01 |
| Potassium | % of the TS | - | 1.51 ± 0.02 | - |
| C/N ratio | dimensionless | 15.62 ± 0.40 | 25.21 ± 1,02 | 7.64 ± 0.18 |
| NDF | % of the TS | - | - | 10.7 ± 0.20 |
| ADF | % of the TS | - | - | 6.8 ± 0.16 |
| Protein | % of the TS | - | 10.06 ± 0.10 | 44.44 ± 0.09 |
| Parameters | RY | ||||
|---|---|---|---|---|---|
| 0% | 12% | 22% | 32% | ||
| pH | Influ. | 6.88 ± 0.27 | 6.97 ± 0.25 | 7.07 ± 0.21 | 7.19 ± 0.21 |
| Efflu. | 7.16 ± 0.09 | 7.19 ± 0.09 | 7.33 ± 0.30 | 7.39 ± 0.29 | |
| TS (g L−1) | Influ. | 15.06 ± 1.34 | 17.43 ± 0.77 | 18.09 ± 1.71 | 19.02 ± 1.09 |
| Efflu. | 7.72 ± 0.63 | 6.43 ± 0.01 | 6.55 ± 0.31 | 6.47 ± 0.58 | |
| VS (g L−1) | Influ. | 10.96 ± 0.81 | 12.41 ± 0.57 | 13.22 ± 0.86 | 14.24 ± 0.65 |
| Efflu. | 4.32 ± 0.40 | 3.55 ± 0.07 | 3.48 ± 0.21 | 3.29 ± 0.36 | |
| VS/TS | Efflu. | 0.56 ± 0.01 | 0.55 ± 0.01 | 0.53 ± 0.02 | 0.51 ± 0.01 |
| TOC (g L−1) | Influ. | 6.09 ± 0.45 | 6.60 ± 0.43 | 7.11 ± 0.41 | 7.62 ± 0.38 |
| Efflu. | 1.34 ± 0.14 | 1.09 ± 0.04 | 1.03 ± 0.08 | 0.93 ± 0.12 | |
| NKT(g L−1) | Influ. | 0.24 ± 0.02 | 0.34 ± 0.02 | 0.43 ± 0.02 | 0.53 ± 0.02 |
| Efflu. | 0.10 ± 0.01 | 0.71 ± 0.01 | 0.71 ± 0.03 | 0.70 ± 0.06 | |
| C/N ratio | Influ. | 25.17 ± 1.02 | 19.59 ± 0.55 | 16.47 ± 0.42 | 14.48 ± 0.35 |
| Efflu. | 13.27 ± 0.34 | 1.53 ± 0.06 | 1.45 ± 0.08 | 1.32 ± 0.07 | |
| Parameters | RY | p | CV | ||||
|---|---|---|---|---|---|---|---|
| 0% | 12% | 22% | 32% | ||||
| Biogas | L d−1 | 4.2 d | 5.0 c | 5.7 b | 6.5 a | <0.001 | 4.78 |
| L kgTSadded−1 | 260.0 c | 269.1 bc | 292.5 ab | 318.7 a | 0.0011 | 4.99 | |
| L kgVSadded−1 | 357.2 c | 378.1 bc | 400.3 ab | 425.7 a | 0.0041 | 4.96 | |
| Methane | L d−1 | 2.71 d | 3.22 c | 3.59 b | 4.12 a | <0.001 | 4.10 |
| L kgTSadded−1 | 180.2 b | 184.5 b | 198.4 ab | 216.6 a | <0.001 | 4.28 | |
| L kgVSadded−1 | 247.5 b | 259.2 b | 271.4 ab | 289.3 a | 0.0033 | 4.26 | |
| TSred. | % | 48.7 b | 63.1 a | 63.8 a | 66.0 a | <0.001 | 5.69 |
| VSred. | % | 60.6 b | 71.4 a | 73.6 a | 76.9 a | <0.001 | 4.36 |
| Methane | % | 64.3 a | 64.3 a | 63.4 a | 63.2 a | 0.0667 | 1.03 |
| Hydrogen sulfide | ppm | 24.8 b | 44.1 ab | 55.3 a | 59.0 a | 0.0026 | 20.58 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akamine, L.A.; Passini, R.; Sousa, J.A.S.; Fernandes, A.; Moraes, M.J.d. Anaerobic Co-Digestion of Cattle Manure and Brewer’s Residual Yeast: Process Stability and Methane and Hydrogen Sulfide Production. Fermentation 2023, 9, 993. https://doi.org/10.3390/fermentation9120993
Akamine LA, Passini R, Sousa JAS, Fernandes A, Moraes MJd. Anaerobic Co-Digestion of Cattle Manure and Brewer’s Residual Yeast: Process Stability and Methane and Hydrogen Sulfide Production. Fermentation. 2023; 9(12):993. https://doi.org/10.3390/fermentation9120993
Chicago/Turabian StyleAkamine, Luana Alves, Roberta Passini, João Antônio Silva Sousa, Aline Fernandes, and Maria Joselma de Moraes. 2023. "Anaerobic Co-Digestion of Cattle Manure and Brewer’s Residual Yeast: Process Stability and Methane and Hydrogen Sulfide Production" Fermentation 9, no. 12: 993. https://doi.org/10.3390/fermentation9120993
APA StyleAkamine, L. A., Passini, R., Sousa, J. A. S., Fernandes, A., & Moraes, M. J. d. (2023). Anaerobic Co-Digestion of Cattle Manure and Brewer’s Residual Yeast: Process Stability and Methane and Hydrogen Sulfide Production. Fermentation, 9(12), 993. https://doi.org/10.3390/fermentation9120993






