Comparison of Sodium Hydroxide and Sodium Carbonate as Alkali Catalysts in Ethanol Organosolv Treatment of Cotton Stalks for the Release of Hydroxycinnamates
Abstract
1. Introduction
2. Results and Discussion
2.1. Treatment Severity and Catalyst Effects
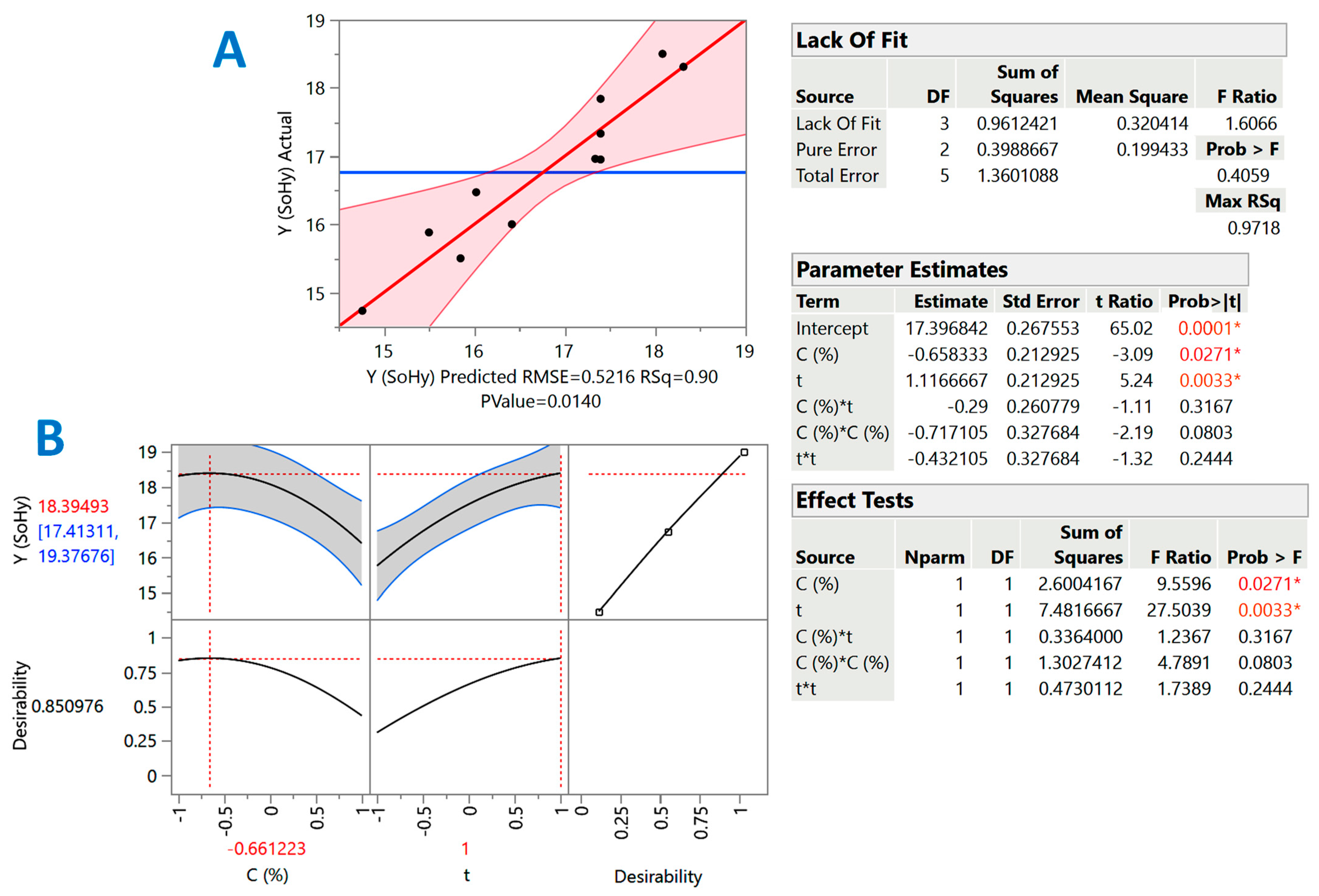
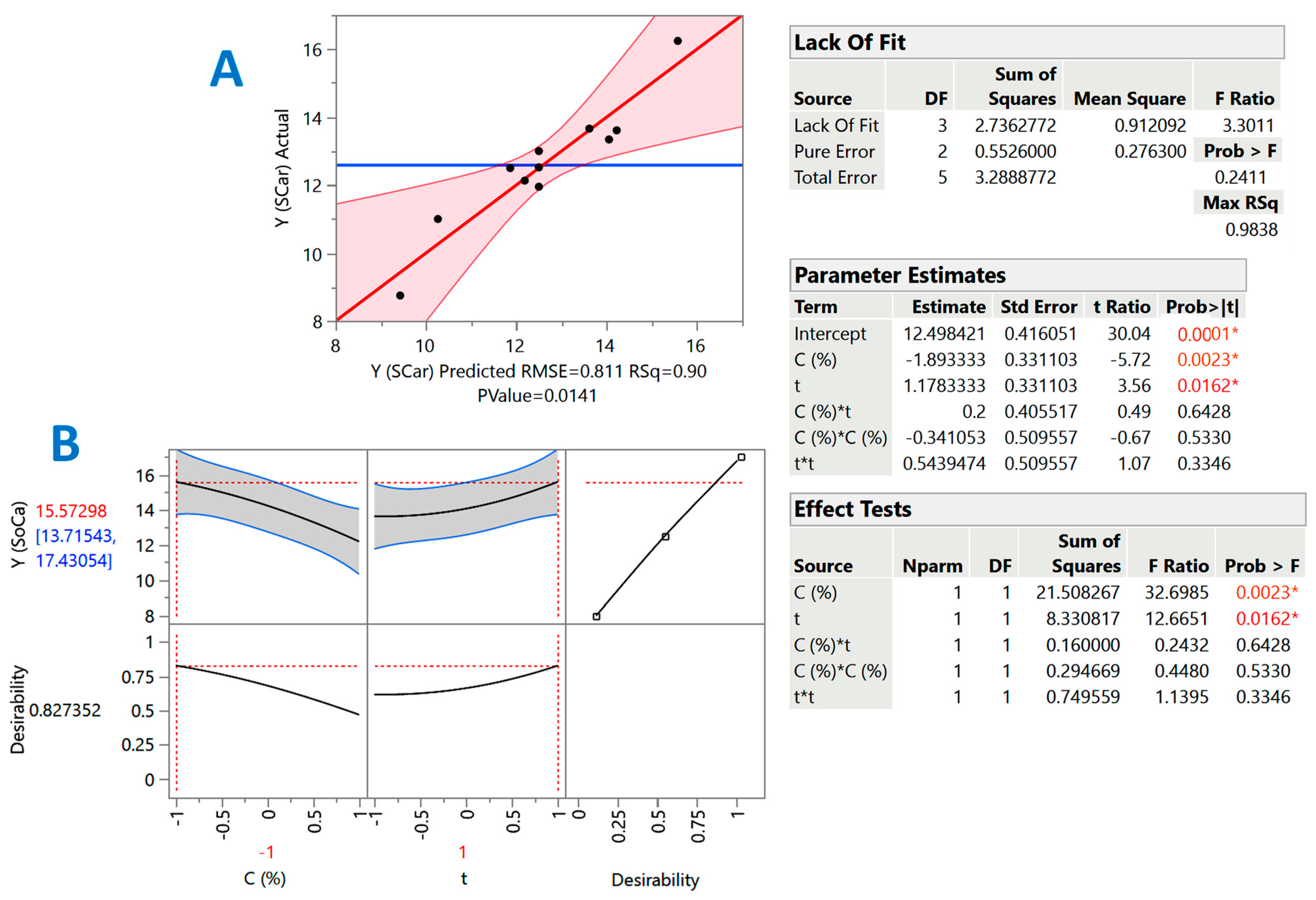
2.2. Treatment Optimization
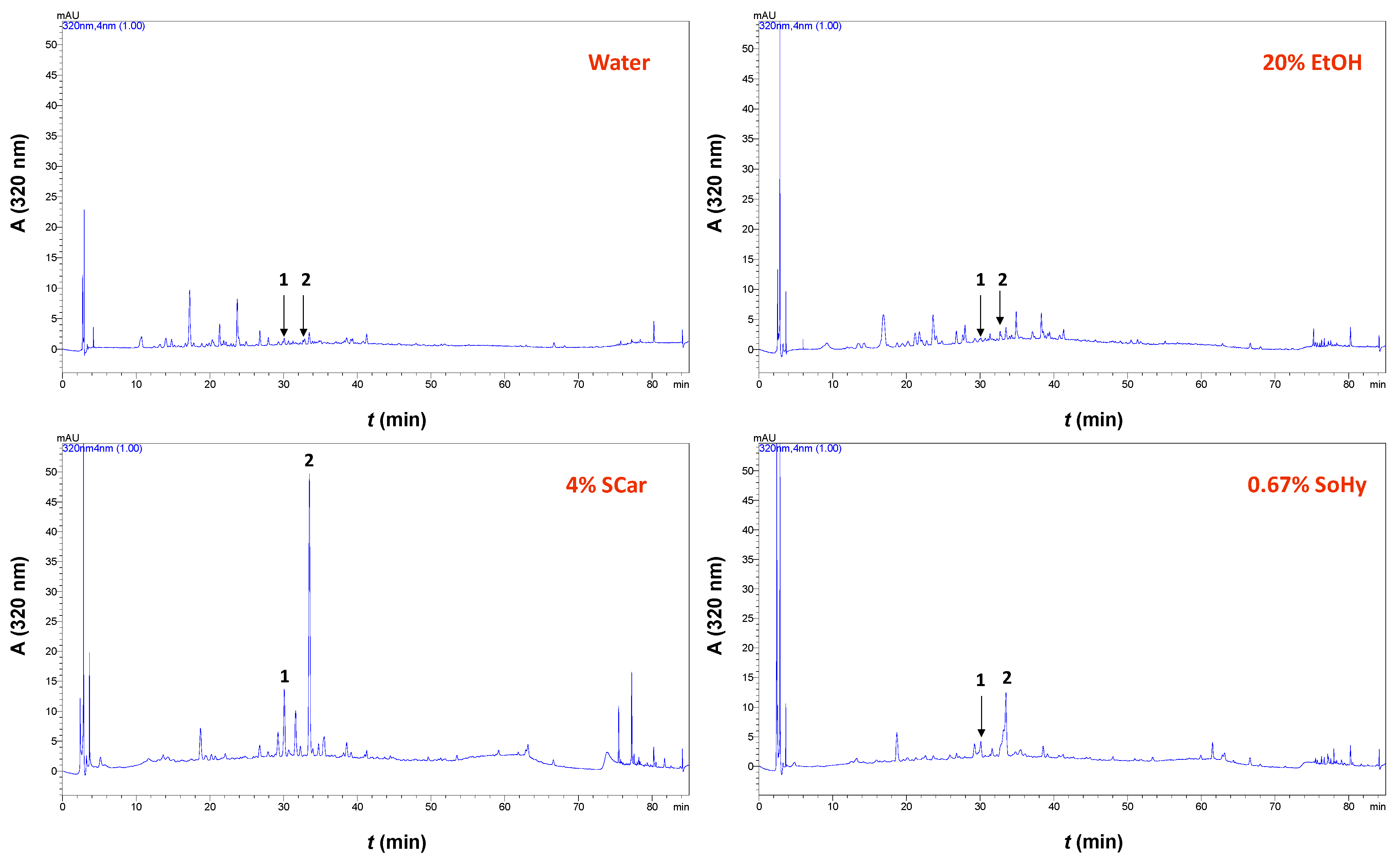
2.3. Polyphenolic Composition
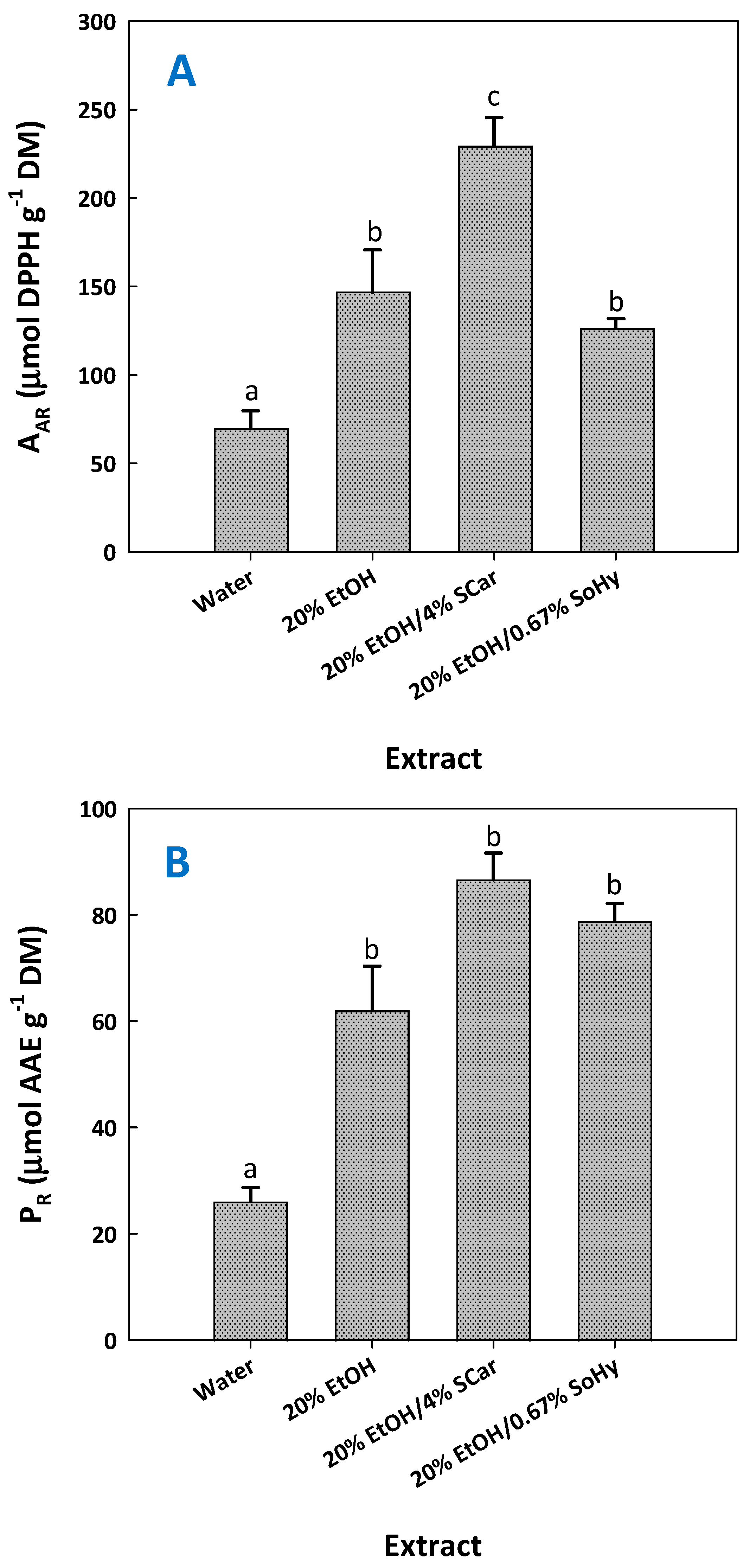
2.4. Antioxidant Activity
3. Materials and Methods
3.1. Chemicals
3.2. Cotton Stalks
3.3. Alkali-Catalyzed Ethanol Organosolv Treatment
3.4. Severity-Based Examination
3.5. Organosolv Treatment Optimization
3.6. Total Polyphenol Yield and Antioxidant Activity Measurements
3.7. Chromatographic Determinations
3.8. Statistical Handling and Processing
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Esparza, I.; Jiménez-Moreno, N.; Bimbela, F.; Ancín-Azpilicueta, C.; Gandía, L.M. Fruit and vegetable waste management: Conventional and emerging approaches. J. Environ. Manag. 2020, 265, 110510. [Google Scholar] [CrossRef]
- Zuin, V.G.; Ramin, L.Z. Green and sustainable separation of natural products from agro-industrial waste: Challenges, potentialities, and perspectives on emerging approaches. In Chemistry and Chemical Technologies in Waste Valorization; Springer: Cham, Switzerland, 2018; pp. 229–282. [Google Scholar]
- Torres, A.F.; Xu, X.; Nikiforidis, C.V.; Bitter, J.H.; Trindade, L.M. Exploring the treasure of plant molecules with integrated biorefineries. Front. Plant Sci. 2019, 10, 478. [Google Scholar] [CrossRef]
- Drescher, A.; Kienberger, M. A systematic review on waste as sustainable feedstock for bioactive molecules—Extraction as isolation technology. Processes 2022, 10, 1668. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Ravi, H.K. Toward petroleum-free with plant-based chemistry. Curr. Opin. Green Sustain. Chem. 2021, 28, 100450. [Google Scholar] [CrossRef]
- Ouro-Salim, O.; Guarnieri, P. Circular economy of food waste: A literature review. Environ. Qual. Manag. 2022, 32, 225–242. [Google Scholar] [CrossRef]
- Hendriks, A.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Biores. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Thoresen, P.P.; Matsakas, L.; Rova, U.; Christakopoulos, P. Recent advances in organosolv fractionation: Towards biomass fractionation technology of the future. Biores. Technol. 2020, 306, 123189. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Song, G.; Pan, Z.; Tu, M.; Kharaziha, M.; Zhang, X.; Show, P.-L.; Sun, F. Advances in organosolv modified components occurring during the organosolv pretreatment of lignocellulosic biomass. Biores. Technol. 2022, 368, 128356. [Google Scholar] [CrossRef] [PubMed]
- Houasni, A.; Grigorakis, S.; Kellil, A.; Makris, D.P. Organosolv treatment/polyphenol extraction from olive leaves (Olea europaea L.) using glycerol and glycerol-based deep eutectic solvents: Effect on metabolite stability. Biomass 2022, 2, 46–61. [Google Scholar] [CrossRef]
- Abdoun, R.; Grigorakis, S.; Kellil, A.; Loupassaki, S.; Makris, D.P. Process optimization and stability of waste orange peel polyphenols in extracts obtained with organosolv thermal treatment using glycerol-based solvents. ChemEngineering 2022, 6, 35. [Google Scholar] [CrossRef]
- Velmourougane, K.; Blaise, D.; Savitha, S.; Waghmare, V. Valorization of cotton wastes for agricultural and industrial applications: Present status and future prospects. In Valorization of Agri-Food Wastes and by-Products; Elsevier: Amsterdam, The Netherlands, 2021; pp. 665–692. [Google Scholar]
- Egbuta, M.A.; McIntosh, S.; Waters, D.L.; Vancov, T.; Liu, L. Biological importance of cotton by-products relative to chemical constituents of the cotton plant. Molecules 2017, 22, 93. [Google Scholar] [CrossRef]
- Yosef, E.; Ben-Ghedalia, D.; Miron, J.; Huttermann, A.; Majcherczyk, A.; Milstein, O.; Ludemann, H.D.; Frund, R. Characterization of some cell wall components of untreated and ozone-treated cotton stalks. J. Agric. Food Chem. 1994, 42, 86–90. [Google Scholar] [CrossRef]
- Kirkan, B.; Sarikurkcu, C.; Copuroglu, M.; Cengiz, M.; Tepe, B. Is it possible to use the stalks of Gossypium hirsitum L., an important by-product of cotton cultivation, as an alternative source of bioactive components? Eur. Food Res. Technol. 2018, 244, 1065–1071. [Google Scholar] [CrossRef]
- Pazo-Cepeda, V.; Benito-Román, Ó.; Navarrete, A.; Alonso, E. Valorization of wheat bran: Ferulic acid recovery using pressurized aqueous ethanol solutions. Waste Biomass Valor. 2020, 11, 4701–4710. [Google Scholar] [CrossRef]
- Papadaki, E.S.; Palaiogiannis, D.; Lalas, S.I.; Mitlianga, P.; Makris, D.P. Polyphenol release from wheat bran using ethanol-based organosolv treatment and acid/alkaline catalysis: Process modeling based on severity and response surface optimization. Antioxidants 2022, 11, 2457. [Google Scholar] [CrossRef]
- Mathew, S.; Abraham, T.E. Ferulic acid: An antioxidant found naturally in plant cell walls and feruloyl esterases involved in its release and their applications. Crit. Rev. Biotech 2004, 24, 59–83. [Google Scholar] [CrossRef]
- Costa, C.A.E.; Coleman, W.; Dube, M.; Rodrigues, A.E.; Pinto, P.C.R. Assessment of key features of lignin from lignocellulosic crops: Stalks and roots of corn, cotton, sugarcane, and tobacco. Ind. Crop. Prod. 2016, 92, 136–148. [Google Scholar] [CrossRef]
- Huang, C.; Zhan, Y.; Wang, J.; Cheng, J.; Meng, X.; Liang, L.; Liang, F.; Deng, Y.; Fang, G.; Ragauskas, A.J. Valorization of bamboo biomass using combinatorial pretreatments. Green Chem. 2022, 24, 3736–3749. [Google Scholar] [CrossRef]
- De Oliveira, D.M.; Finger-Teixeira, A.; Rodrigues Mota, T.; Salvador, V.H.; Moreira-Vilar, F.C.; Correa Molinari, H.B.; Craig Mitchell, R.A.; Marchiosi, R.; Ferrarese-Filho, O.; Dantas dos Santos, W. Ferulic acid: A key component in grass lignocellulose recalcitrance to hydrolysis. Plant Biotech. J. 2015, 13, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Buranov, A.U.; Mazza, G. Lignin in straw of herbaceous crops. Ind. Crop. Prod. 2008, 28, 237–259. [Google Scholar] [CrossRef]
- Linh, T.N.; Fujita, H.; Sakoda, A. Release kinetics of esterified p-coumaric acid and ferulic acid from rice straw in mild alkaline solution. Biores. Technol. 2017, 232, 192–203. [Google Scholar] [CrossRef]
- Zhou, Z.; Lei, F.; Li, P.; Jiang, J. Lignocellulosic biomass to biofuels and biochemicals: A comprehensive review with a focus on ethanol organosolv pretreatment technology. Biotech. Bioeng. 2018, 115, 2683–2702. [Google Scholar] [CrossRef] [PubMed]
- Woiciechowski, A.L.; Neto, C.J.D.; de Souza Vandenberghe, L.P.; de Carvalho Neto, D.P.; Sydney, A.C.N.; Letti, L.A.J.; Karp, S.G.; Torres, L.A.Z.; Soccol, C.R. Lignocellulosic biomass: Acid and alkaline pretreatments and their effects on biomass recalcitrance—Conventional processing and recent advances. Biores. Technol. 2020, 304, 122848. [Google Scholar] [CrossRef] [PubMed]
- Borand, M.N.; Karaosmanoğlu, F. Effects of organosolv pretreatment conditions for lignocellulosic biomass in biorefinery applications: A review. J. Renew. Sustain. Energy 2018, 10, 033104. [Google Scholar] [CrossRef]
- Verma, B.; Hucl, P.; Chibbar, R. Phenolic acid composition and antioxidant capacity of acid and alkali hydrolysed wheat bran fractions. Food Chem. 2009, 116, 947–954. [Google Scholar] [CrossRef]
- Valério, R.; Cadima, M.; Crespo, J.G.; Brazinha, C. Extracting ferulic acid from corn fibre using mild alkaline extraction: A pilot scale study. Waste Biomass Valor. 2021, 13, 287–297. [Google Scholar] [CrossRef]
- Pazo-Cepeda, M.V.; Aspromonte, S.G.; Alonso, E. Extraction of ferulic acid and feruloylated arabinoxylo-oligosaccharides from wheat bran using pressurized hot water. Food Biosci. 2021, 44, 101374. [Google Scholar] [CrossRef]
- Mateo Anson, N.; van den Berg, R.; Havenaar, R.; Bast, A.; RMM Haenen, G. Ferulic acid from aleurone determines the antioxidant potency of wheat grain (Triticum aestivum L.). J. Agric. Food Chem. 2008, 56, 5589–5594. [Google Scholar] [CrossRef]
- Kim, K.-H.; Tsao, R.; Yang, R.; Cui, S.W. Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effect of hydrolysis conditions. Food Chem. 2006, 95, 466–473. [Google Scholar] [CrossRef]
- Overend, R.P.; Chornet, E. Fractionation of lignocellulosics by steam-aqueous pretreatments. Phil. Trans. R. Soc. Lond. Ser. A Math. Phys. Sci. 1987, 321, 523–536. [Google Scholar]
- Sidiras, D.; Politi, D.; Giakoumakis, G.; Salapa, I. Simulation and optimization of organosolv based lignocellulosic biomass refinery: A review. Biores. Technol. 2022, 343, 126158. [Google Scholar] [CrossRef]
- Pedersen, M.; Meyer, A.S. Lignocellulose pretreatment severity–relating pH to biomatrix opening. New Biotech. 2010, 27, 739–750. [Google Scholar] [CrossRef]
- Athanasiadis, V.; Palaiogiannis, D.; Bozinou, E.; Lalas, S.I.; Makris, D.P. β-Cyclodextrin-aided aqueous extraction of antioxidant polyphenols from peppermint (Mentha × piperita L.). Oxygen 2022, 2, 424–436. [Google Scholar] [CrossRef]
- Guenaoui, A.; Casasni, S.; Grigorakis, S.; Makris, D.P. Alkali-catalyzed organosolv treatment of oat bran for enhanced release of hydroxycinnamate antioxidants: Comparison of 1-and 2-propanol. Environments 2023, 10, 118. [Google Scholar] [CrossRef]
- Cicco, N.; Lanorte, M.T.; Paraggio, M.; Viggiano, M.; Lattanzio, V. A reproducible, rapid and inexpensive Folin–Ciocalteu micro-method in determining phenolics of plant methanol extracts. Microchem. J. 2009, 91, 107–110. [Google Scholar] [CrossRef]
- Lakka, A.; Grigorakis, S.; Karageorgou, I.; Batra, G.; Kaltsa, O.; Bozinou, E.; Lalas, S.; Makris, D.P. Saffron processing wastes as a bioresource of high-value added compounds: Development of a green extraction process for polyphenol recovery using a natural deep eutectic solvent. Antioxidants 2019, 8, 586. [Google Scholar] [CrossRef]



| CSCar (% w/v) | t (min) | CSF | CSF′ | YTP (mg CAE g−1 DM) |
|---|---|---|---|---|
| 4 | 60 | −10.45 b | 6.41 a | 13.7 ± 0.8 c |
| 180 | −9.97 c | 6.89 c | 13.3 ± 0.9 c | |
| 300 | −9.75 c | 7.11 b | 16.2 ± 1 b | |
| 7 | 60 | −10.47 a | 6.43 a | 12.5 ± 0.7 c |
| 180 | −9.99 c | 6.91 c | 12.5 ± 0.9 c | |
| 300 | −9.77 a | 7.13 b | 13.6 ± 1 c | |
| 10 | 60 | −10.50 a | 6.46 a | 8.8 ± 0.6 a |
| 180 | −10.02 c | 6.94 c | 11.2 ± 1.3 c | |
| 300 | −9.80 a | 7.16 b | 12.1 ± 1 c |
| CSoHy (% w/v) | t (min) | CSF | CSF′ | YTP (mg CAE g−1 DM) |
|---|---|---|---|---|
| 0.5 | 60 | −11.33 b | 7.29 a | 15.9 ± 1.1 c |
| 180 | −10.85 b | 7.77 c | 17 ± 1.3 c | |
| 300 | −10.63 b | 7.99 c | 18.3 ± 1.5 b | |
| 1 | 60 | −11.56 a | 7.52 a | 15.5 ± 1 a |
| 180 | −11.08 b | 8.00 c | 17.3 ± 1.6 c | |
| 300 | −10.86 b | 8.22 b | 18.5 ± 1.5 b | |
| 1.5 | 60 | −11.61 a | 7.57 a | 14.7 ± 1 a |
| 180 | −11.13 b | 8.05 c | 16.5 ± 0.9 c | |
| 300 | −10.91 b | 8.27 b | 16 ± 1.5 c |
| Design Point | Independent Variables | Response (YTP, mg CAE g−1 DM) | ||||
|---|---|---|---|---|---|---|
| X1 (C) | X2 (t) | SoHy | SCar | |||
| Measured | Predicted | Measured | Predicted | |||
| 1 | −1 | −1 | 15.9 | 15.5 | 13.7 | 13.6 |
| 2 | −1 | 1 | 18.3 | 18.3 | 16.2 | 15.6 |
| 3 | 1 | −1 | 14.7 | 14.8 | 8.8 | 9.4 |
| 4 | 1 | 1 | 16 | 16.4 | 12.1 | 12.2 |
| 5 | −1 | 0 | 17 | 17.3 | 13.3 | 14 |
| 6 | 1 | 0 | 16.5 | 16 | 11.1 | 10.3 |
| 7 | 0 | −1 | 15.5 | 15.9 | 12.5 | 11.9 |
| 8 | 0 | 1 | 18.5 | 18.1 | 13.6 | 14.2 |
| 9 | 0 | 0 | 17.3 | 17.4 | 12.5 | 12.5 |
| 10 | 0 | 0 | 16.9 | 17.4 | 13 | 12.5 |
| 11 | 0 | 0 | 17.8 | 17.4 | 12 | 12.5 |
| Solvent | FA (μg g−1 DM) | p-CouA (μg g−1 DM) | Total (μg g−1 DM) |
|---|---|---|---|
| Water | 24.8 ± 0.5 a | 14.6 ± 0.3 a | 39.4 a |
| 20% Ethanol | 25.5 ± 0.7 a | 13.3 ± 0.4 a | 38.7 a |
| 20% Ethanol/4% Sodium Carbonate | 233.1 ± 9 b | 55.6 ± 1.9 b | 288.6 b |
| 20% Ethanol/0.67% Sodium Hydroxide | 81.1 ± 5.3 c | 25.6 ± 1 a | 106.8 c |
| Process Variables | Codes | Coded Variable Level | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| t (min) | X1 | 60 | 180 | 300 |
| CSCar (% w/v) * | X2 | 4 | 7 | 10 |
| CSoHy (% w/v) * | 0.5 | 1 | 1.5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadimitriou, G.; Zarnavalou, V.; Chatzimitakos, T.; Palaiogiannis, D.; Athanasiadis, V.; Lalas, S.I.; Makris, D.P. Comparison of Sodium Hydroxide and Sodium Carbonate as Alkali Catalysts in Ethanol Organosolv Treatment of Cotton Stalks for the Release of Hydroxycinnamates. Recycling 2024, 9, 21. https://doi.org/10.3390/recycling9020021
Papadimitriou G, Zarnavalou V, Chatzimitakos T, Palaiogiannis D, Athanasiadis V, Lalas SI, Makris DP. Comparison of Sodium Hydroxide and Sodium Carbonate as Alkali Catalysts in Ethanol Organosolv Treatment of Cotton Stalks for the Release of Hydroxycinnamates. Recycling. 2024; 9(2):21. https://doi.org/10.3390/recycling9020021
Chicago/Turabian StylePapadimitriou, Georgia, Vassiliki Zarnavalou, Theodoros Chatzimitakos, Dimitrios Palaiogiannis, Vassilis Athanasiadis, Stavros I. Lalas, and Dimitris P. Makris. 2024. "Comparison of Sodium Hydroxide and Sodium Carbonate as Alkali Catalysts in Ethanol Organosolv Treatment of Cotton Stalks for the Release of Hydroxycinnamates" Recycling 9, no. 2: 21. https://doi.org/10.3390/recycling9020021
APA StylePapadimitriou, G., Zarnavalou, V., Chatzimitakos, T., Palaiogiannis, D., Athanasiadis, V., Lalas, S. I., & Makris, D. P. (2024). Comparison of Sodium Hydroxide and Sodium Carbonate as Alkali Catalysts in Ethanol Organosolv Treatment of Cotton Stalks for the Release of Hydroxycinnamates. Recycling, 9(2), 21. https://doi.org/10.3390/recycling9020021










