Abstract
A new insight into the synthesis of the herbal plant (White poplar, Poplus alba) leave extract using chitosan nanocapsule was studied. The in vitro antibacterial activity of chitosan white poplar nanocapsule (CWPNC) against Streptococcus agalactiae (S. agalactiae) was determined. About 120 fish were categorized for 7 days into four groups. The first and second (CWPNC) groups were treated with 0 mg/L and 3 mg/L CWPNC in the water, respectively, without being challenged; the first group was a control. The third (S. agalactiae) and fourth (CWPNC + S. agalactiae) groups were treated with 0 and 3 mg/L CWPNC, respectively, and challenged with S. agalactiae (0.5 × 107 CFU/mL). The obtained results revealed that CWPNC had an in vitro antibacterial activity against S. agalactiae. Moreover, S. agalactiae infection caused a significant elevation (p < 0.05) in the lipid peroxidation (malondialdehyde) and hepatorenal biomarkers, as well as the lowest significant (p < 0.05) survival rate (33.33%). Moreover, a significant depletion (p < 0.05) in the level of antioxidants (catalase and superoxide dismutase) and the immune indicators (immunoglobulin, lysozyme activity, and complement 3) were the consequences of S. agalactiae infection. Treatment of the infected fish with 3 mg/L CWPNC alleviated these bad circumstances.
Keywords:
CWPNC; antimicrobial activity; Streptococcus agalactia; in vitro assay; in vivo assay; oxidative stress Key Contribution:
Chitosan white poplar nanocapsule (CWPNC) at a dose of 3 mg/L can be used as water additives for mitigating the Streptococcus agalactiae challenge in Oreochromis niloticus.
1. Introduction
Aquaculture production on a global scale has increased significantly in recent decades. This is largely attributable to a variety of developments that have improved control and competitiveness over the production process. In this regard, the tissue investigation in fish specimens represents a valuable tool for assessing their health status. Further, hematological and biochemical research aid in the study of the relationship between blood properties and habitat, as well as the species’ ability to adapt to the environment [1,2]. The global production in aquaculture scale rose from 2.6 million metric tonnes (mt) in 1970 to 87.5 million mt in 2020 [3].
The Nile tilapia (Oreochromis niloticus) is the third most widely cultivated fish in the world, producing 4.5 million metric tonnes in 2018 [4]. Remarkably, our understanding of genetic variation within and among populations is still quite restricted, given the significance of O. niloticus to aquaculture and commercial fisheries, particularly in Africa [5].
Bacterial infections are one of the main causes and primary factors leading to the enormous losses every year in the fish farming business, which are estimated at billions of dollars [6]. The greatest barrier to the growth of fish aquaculture is bacterial illness [7].
Streptococcus spp. is a Gram-positive bacterium that is extremely pathogenic because it can infect various fish species worldwide, including fresh, marine, and brackish water fishes. It is important to note that Streptococcus has cost aquaculture around the world millions of dollars in economic damages. Additionally, tilapia is thought to be the ideal host for Streptococcus infection [8]. Streptococcus spp. affects the production of tilapia, and S. agalactiea is a widely distributed species [9].
Bacterial diseases control is a significant threat to the fish farming industry, and it is typically accomplished by using antibiotics, which the European Union forbade because they could lead to the development of bacterial strains which resistant to antibiotics and leave residues in fish muscles and aquatic water systems [10,11]. Recently, the use of antibiotics against outbreaks of aquatic diseases has been prohibited globally because of their apparent accumulation in tissues, which encourages the growth of bacteria that are resistant to them [12]. Thus, it has become necessary to find safe alternatives for antibiotics for controlling the most common fish pathogen.
Recently, medicinal plants have been thought to be a better alternative to antibiotics [13]. The white poplar tree (Populus alba L., P. alba) belongs to the family Salicaceae consisting of some three hundred species [14]. White poplar’s abundant and diverse secondary metabolite composition makes them potent antimicrobial agents [15].
Nanotechnology science is concerned with creating, designing, analyzing, and utilizing materials whose dimension is on the nanometer scale (one billionth of a meter) [16]. Nanotechnology has enormous potential to advance fields as varied as medical research, water purification, the technology of communication and information, as well as the creation of smaller and more powerful materials; nanoscience and nanotechnologies could confirm to be a godsend for human health [17,18,19,20]. The medical sciences will use the advancements of nanotechnology for patient care, drug delivery systems, and diagnostic procedures over the next two years. Nanotechnology is widely expected to continue evolving and expanding in many fields of life and science [21].
Chitosan, a biodegradable carbohydrate, is among the most widely used substances in the field of nanotechnology since it has a variety of uses [22]. There have been several papers on the use of herbal nanoparticles or nanocapsules in fish diets [23]. In addition, some nanomaterials had used for controlling bacterial infection, Aeromonas hydrophila in O. niloticus [24], and controlling fungal infection, Saprolegnia spp. in O. niloticus [23]. Chitosan is inexpensive, non-toxic, biocompatible, and biodegradable. These advantages have led to its application in a variety of sectors, such as biotechnology and medicine [25]. However, nothing is known regarding the use of chitosan white poplar nanocapsule (CWPNC) in controlling bacterial pathogens in the fish field. Therefore, this study was designated to investigate the in vitro antibacterial activity of CWPNC against S. agalactiae with the in vivo approach in Nile tilapia.
2. Material and Methods
2.1. Preparation of the White Poplar Leave Extract
About 5 g of the White poplar leaves were dissolved in 150 mL of deionized water and subjected to sonication power of 60 kHz and cycle 0.65 using a sonicator prop instrument (Hielscher UP400S, German) for 0.5 h. Finally, white poplar extract was filtered by Whatman No.1.
2.2. Preparation of the CWPNC
CWPNC was synthesized by ionic gelation method-assessing by the Sono-chemical method. However, 0.5 g of chitosan (CS) was added to 300 mL doubled deionized water and heated at 80 °C for 1 h with vigorous stirring, then an acetic acid solution (2% v/v) was added and stirred for 1 h until colorlessness was obtained. 50 mL of white poplar extract was added with continuous stirring until obtaining a homogenous solution, then a TPP solution (2% w/v) was added, drop by drop, until a whitish mixture was obtained. The mixture was washed by centrifugation method three times.
2.3. Characterization of the CWPNC
Characterization of CWPNC was carried out using atomic force microscopy (AFM instrument manufacturer, Agilent technology; model 5600LS), scanning electron microscopy (SEM), and transmission electron microscopy (TEM instrument manufacturer, JEOL; high-resolution model JEM-2100) for determining the morphology, composition, particle size distribution, pore size, surface area, and surface charge.
2.3.1. Characterization of CWPNC Using AFM
The sample was subjected to ultrasound waves for 0.5 h, a condition of 57 kHz, at an amplitude of 73% and 0.73 of a cycle (Up 400s manufactured by Hielscher, German). Afterward, a thin film was created by Spain coater instrument model Laurell-650Sz at the condition of 800 rpm under vacuum. AFM images were analyzed by Gwydion software. AFM images and data profiles were done for 100 nm × 100 nm through tap mode, gold tap, 0.2 In/S speed, I. gain 50, and P. gain 45.
2.3.2. Characterization of CWPNC Using TEM
Adding CWPNC was to double deionized water and sonication for 11 min through ultrasound prop with a 51 kHz, at an amplitude of 51% and 0.51 of a cycle (Up 400s, Hielscher, German). Afterward, 1 to 2 microns of desperation were placed onto a carbon-coated copper grid.
2.3.3. Characterization of CWPNC Using XRD
Thermo Scientific’s EQUINOX 1000 XRD instrument was utilized to analyze the production of CWPNC without any secondary products. Cu Kα radiation with a current of 34 mA and an applied voltage of 34 kV served as the light source. The scan speed was 0.1°/min, and the two angles ranged from 5 to 80°.
2.3.4. Characterization of CWPNC Using BET Surface Area and DA Pore Size
To determine the BET surface area and DA pore size of CWPNC, a Quanta Chrome Company model of the Nova Touch 4 L surface area and pore size analyzer was used. CWPNC was degassed for 1 h at 55 °C. Following the completion of the adsorption equilibrium, the materials were made to absorb nitrogen gas. The amount of gas that had been adsorbed was calculated using the applied pressure and the universal gas law.
2.3.5. Characterization of CWPNC Using DLS and Zeta Potential
Malvern Company’s Nano Sight NS500 zeta seizer instrument was used to measure dynamic light scattering (DLS) and zeta potential.
2.4. Isolation and Preparation of the Bacterial Isolates
S. agalactiae strain was isolated from diseased O. niloticus and cultured on tryptic soy broth (TSB) (Difco, Detroit, MI, USA) with supplementation with 3% NaCl and incubation at 25 °C for 24–48 h. Identification of the bacterial isolate was carried out by conventional tests and VITEK® 2 compact (BioMérieux) biochemical Identification (VITEK2 Compact System, BIOMERIEUX, and France). The criteria used to detect the isolates are based on the morphology of colonies which are characterized by gram staining of the microorganisms.
2.5. In Vitro Assay
2.5.1. Antibacterial Inhibition Activity by Disc Diffusion Assay
The antibacterial activity of CWPNC against S. agalactiae was carried out by the disc diffusion method [26]. Overnight culture of the bacterium isolate was spread on Muller Hinton agar (MHA). Sterile discs were soaked in 50 μL of CWPNC (0.1 mmole), and further sterile discs were soaked in 50 μL of chitosan (as control). The discs were placed in an incubator to dry and then placed on the MHA plates. The plates were then incubated at 28 °C for 24 h. Finally, measurement of the inhibition zones was carried out.
2.5.2. Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
CWPNC were subjected to the MIC according to the protocol of Aliasghari et al. [27] assay in triplicate for the determination of MIC of CWPNC solution; the micro-dilution broth method was followed. Overnight, the bacterium isolate cultures in TSB were adjusted to 106 CFU/mL. CWPNC and chitosan (as positive control) solutions were prepared in tenfold serial dilution in broth to achieve final concentrations of 0 to 100 µg/mL. About 100 mL of each dilution of both solutions was added to the well containing 100 µL of the bacterial suspension in sterile 96-well plates. After 24 h of anaerobic incubation at 37 °C, turbidity measurements were taken for each well. The MBC is defined as the lowest concentration of antibacterial agents that completely kill the bacteria. The MBC was carried out by placing the suspension from each well of the microtiter plates onto an MHA for 24 h; the plates were incubated at 37 °C. The MBC value was determined as the lowest concentration on the MHA plate without any discernible growths.
2.6. In Vivo-Assay
2.6.1. Fish and Rearing Conditions
A total of 210 cultured O. niloticus fish that appeared to be in good health (average body weight: 20 ± 1.5 g) were used for the in vivo assay. Fish were purchased from the Fish Research Unit at the Zagazig University of Egypt’s, Faculty of Veterinary Medicine. Before the study, fish were monitored and acclimated for two weeks. To confirm that the fish were healthy, a medical examination was also performed. In glass aquariums (40 × 30 × 80 cm), the fish were kept at a ratio of 10 fish to 60 L of dechlorinated tap water. The laboratory’s water quality parameters, including dissolved oxygen (6.54 ± 0.20 mg/L), pH (6.7 ± 0.3), temperature (25 ± 1.4 °C), and ammonia (0.020 ± 0.02 mg/L), were maintained using a controlled day to night period (12 h dark: 12 h light). The fish received a basal diet (at a rate of 3% of their body mass). The experimental protocol, which adhered to NIH guidelines for the use and care of laboratory animals, was authorized (ZU-IACUC/2/F/310/2022) by the animal usage in the research committee at Zagazig University in Egypt.
2.6.2. Identifying CWPNC Therapeutic Dosage
About 90 fish were subjected to nine different levels of CWPNC (0, 1, 2, 3, 4, 5, 6, 7, and 8 mg/L) to determine the preliminary concentration (Table 1). The clinical manifestations and mortalities were recorded daily. The safe level of CWPNC was 3 mg/L.

Table 1.
Effect of exposure to different concentrations of CWPNC on mortality and clinical observations of O. niloticus for seven days.
2.6.3. Experimental Setup
The procedures used by Ibrahim et al. [28] were used to determine the lethal dose (LD50) of S. agalactiae, which was found to be 1 × 107 CFU/mL. The challenge used sub-lethal dosages of 0.5 × 107, and the outcomes were confirmed using a drop plate assay [29].
About 120 fish were randomly assigned into four groups for 7 days. The first group served as the control group, which was exposed with the second (CWPNC) group to 0 mg/L and 3 mg/L of CWPNC in water, respectively, without being challenged. The third (S. agalactiae) and fourth (S. agalactiae + CWPNC) groups were inoculated intraperitoneally with 0.2 mL of S. agalactiae (1 × 106 CFU) and subjected to 0 and 3 mg/L CWPNC, respectively. Throughout the trial, any clinical complaints, post-mortem lesions, and deaths were all recorded (7 days).
2.6.4. Blood Sampling
The fish (three fish/replicate; nine fish/group) were anesthetized (100 mg/L benzocaine solution) for blood sample collections. Blood sampling was carried out after the end of the experiment (7 days) from the caudal blood arteries of fish without anticoagulant. Centrifugation at 3000 rpm for 15 min to separate the serum was carried to the blood samples for evaluation of the immunological and biochemical indices. Additionally, the fish were euthanized using an overdose of a benzocaine solution (500 mg/L) for tissue sampling. Fresh samples of the liver for assessing the hepatic antioxidant activity were directly collected from euthanized fish following the instructions for fish use in the research [30].
2.6.5. Serum Oxidative Stress Assay
Superoxide dismutase (SOD) and catalase (CAT) were measured in liver homogenates using the techniques provided by Nishikimi et al. [31] and Aebi [32]. Malondialdehyde (MDA) was measured using the thiobarbituric acid reaction, as previously described by Ohkawa et al. [33].
2.6.6. Hepato-Renal Related Parameters
Serum activity of aspartate aminotransferase (AST) was measured according to the method used by Murray [34], and alanine aminotransferase (ALT) was measured according to the method used by Burtis and Ashwood [35]. The creatinine level was determined colorimetrically according to the Bartles and Bohmer [36] method.
2.6.7. Immune-Related Parameters Assay
According to the manufacturer’s instructions, Tina-quant IgM Gen.2 procedure utilizing Roche Cobas company diagnostic kits (Indianapolis, IN, USA) was used to estimate IgM levels (IgM, REF; 035071190) after an immunoturbidimetric test employing Anti-IgM antibodies. With an ELISA reader (Lambda EZ201; Perkin Elmer), the optical density (OD) value was calculated at 340 nm. Additionally, following a previously established technique [37], a commercial assay kit was used to assess the serum lysozyme activity, and the absorbance at 450 nm was calculated spectrophotometrically. Following the manufacturer’s instructions, serum complement 3 (C3) was evaluated using a fish-specific ELISA kit with CAT. NO. MBS005953 (sensitivity: 1 g/mL, control of the reaction: 31.2–1000 g/mL) (MyBioSource Company, Diego, CA, USA).
2.7. Data Analysis
Shapiro-test Wilk’s was applied to ensure the data’s normality. The Kaplan-Meier model was applied to calculate the survival probability of fish in each group. The log-rank test was used to look for any differences between groups in pairwise comparisons. Using SPSS version 18, a two-way analysis of variance (ANOVA) was used to examine the results of the biochemical, immunological, and oxidant/antioxidant indices (SPSS, Chicago, IL, USA). At a significance level of 0.05, Duncan’s multiple range tests were employed to find differences between means (N = 9/group).
3. Results
3.1. Characterization of the CWPNC
XRD curve illustrates the amorphous nature of CWPNC where there are not any peaks. Although the chitosan nanoparticles have characteristic XRD peaks, the Carley coating of white poplar extract on chitosan nanoparticles hides the XRD peaks of chitosan (Supplementary Material Figure S1A). TEM images confirmed AFM images shape where different fiber-shaped particles illustrate at different sizes (Supplementary Material Figure S1B). AFM images 3D (Supplementary Material Figure S2A) and 2D (Supplementary Material Figure S2B) illustrate the fiber shape of CWPNC with wide particle size distribution. The surface area determined by the multi-point BET method illustrates the high value of CWPNC. However, the BET surface area value is 61.1 m2/g (Supplementary Material Figure S3A), with pore volume determined by the DH method as 1.1 cc/nm (Supplementary Material Figure S3B). DLS (dynamic light scattering) illustrates the wide particle size distribution due to the presence of more than one peak in the DLS curve at 18 nm and 37 nm may be to fiber-shaped particles that have two different dimensions (Supplementary Material Figure S3C). The zeta potential value is −37 mV (Supplementary Material Figure S3D).
3.2. In Vitro Antibacterial Activity of CWPNC against S. agalactiae
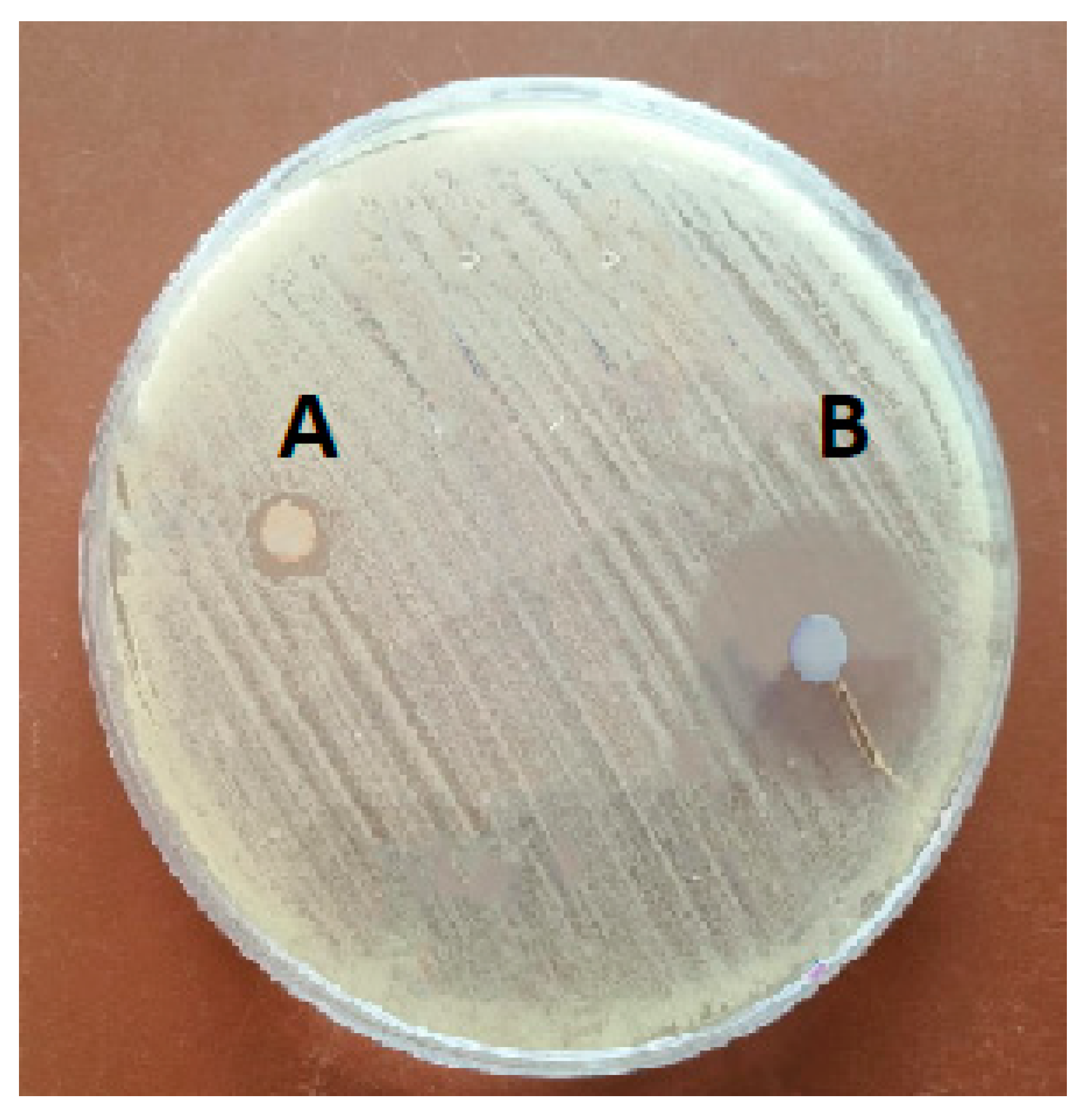
As shown in Figure 1 and Table 2, CWPNC showed antibacterial activity against S. agalactiae with an inhibition zone diameter of 22 ± 1.23 mm. Further, chitosan exhibited an antibacterial activity with an inhibition zone diameter of 2.5 ± 0.20 mm. The MIC of CWPNC and chitosan were 10 and 80 µg/mL, respectively. While the MBC of CWPNC and chitosan were 20 and 100 µg/mL, respectively.
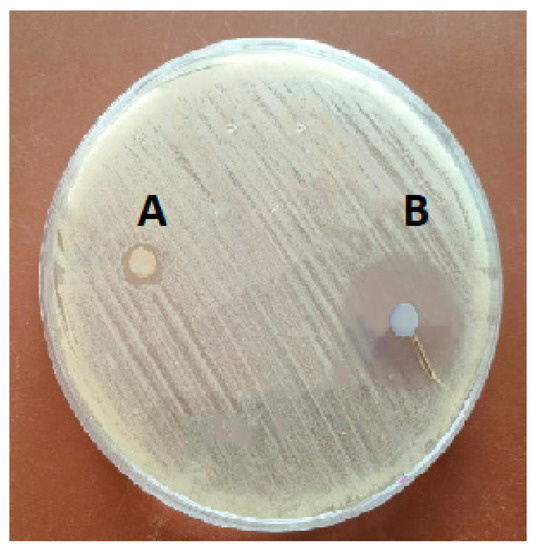
Figure 1.
Inhibition zone of chitosan against S. agalactia (A) and CWPNC (B).

Table 2.
The inhibition zone, MIC, and MBC of CWPNC and chitosan against S. agalactiae.
3.3. In Vivo Assay
3.3.1. Clinical Observation, Behavior Response, and Survival Rate
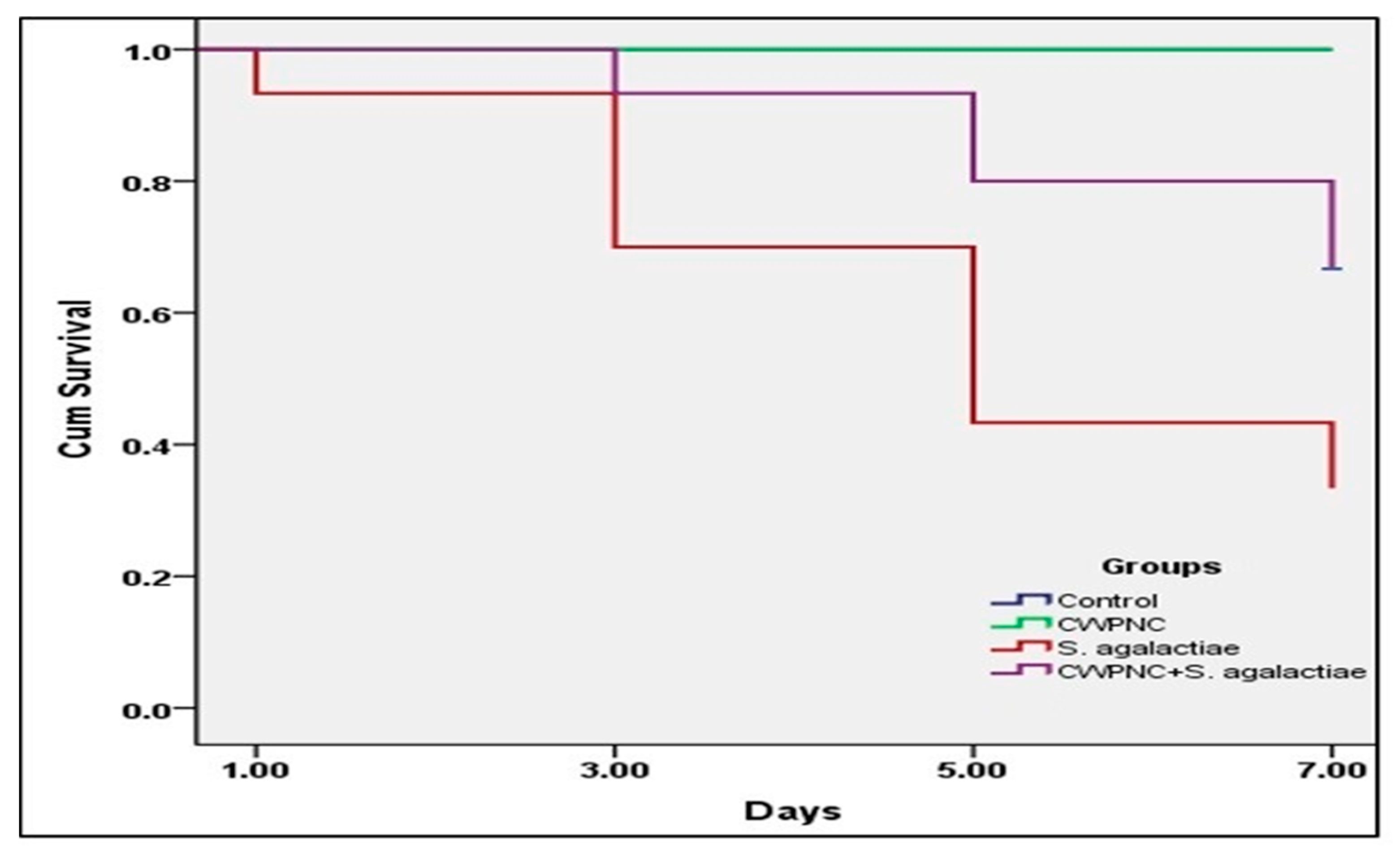
The non-infected fish (control and CWPNC groups) that were exposed to 0 and 3 mg/L CWPNC, respectively, had no mortality and revealed no abnormal signs (Figure 2A) or behaviors (Table 3). S. agalactiae-infected fish suffered a decrease in swimming activity, dark body coloration, fin rot, hemorrhages on numerous body parts, and skin ulcerations (Figure 2B,C) with congested internal organs. Likewise, exposure of infected fish to 3 mg/L CWPNC (CWPNC + S. agalactiae) group restored the prior clinical signs, except for fin rot (Figure 2D). Fish survival rates in the control, CWPNC, S. agalactiae, and CWPNC + S. agalactiae groups were 100%, 100%, 33.33%, and 66.67%, respectively, according to the Kaplan-Meier curves. The S. agalactiae group recorded the lowest significant (p < 0.05) survival rate (Figure 3). Furthermore, statistically significant differences (p < 0.05) were across the groups.

Figure 2.
Effect of CWPNC as water exposure on clinical observation of experimentally infected O. niloticus with S. agalactiae for 7 days. (A) Fish in the control group or CWPNC group demonstrated normal appearance. (B,C) Fish in the S. agalactiae group showed skin ulcers (blue arrows), hemorrhagic patches on the body, and fin rot (yellow arrows). (D) Fish in the CWPNC + S. agalactiae group showed some fin rot (yellow arrow).

Table 3.
Effect of exposure to 3 mg/L CWPNC on behavioral responses of O. niloticus infected with S. agalactiae for 7 days.
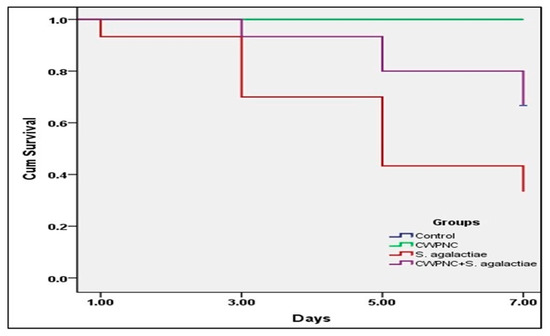
Figure 3.
Survival curves (Kaplan–Meier) of experimentally infected O. niloticus with S. agalactiae and treated with CWPNC as water exposure for 7 days.
3.3.2. The Results of the Shapiro-Wilk Test
The normal distributions of MDA, CAT, SOD, AST, ALT, creatinine, IgM, LYZ, and C3 were tested using the Shapiro-Wilk method (Table 4). According to the Shapiro-Wilk test, all variables followed a normal distribution (p values greater than 0.05).

Table 4.
Shaprio-Wilk test results for the measured parameters.
3.3.3. Oxidative Stress-Related Parameters
The infected groups showed a significant increase (p < 0.05) in the serum level of MDA and a significant decrease in the levels of CAT and SOD compared to the non-infected ones. Concerning the effects of CWPNC exposure, 3 mg/L exposure level reduced the level of MDA and increased the level of SOD (p < 0.05) compared to the 0 mg/L level and did not affect (p < 0.05) the level of CAT (Table 5). Regarding the interaction between the S. agalactiae infection and CWPNC exposure, the infected group exposed to 0 mg/L CWPNC showed the highest serum MDA level and the lowest CAT and SOD values, followed by the infected one and exposed to 3 mg/L CWPNC when compared to the non-infected groups treated with 3 and 0 mg/L CWPNC, respectively.

Table 5.
Effect of S. agalactiae infection and/or CWPNC as water exposure on oxidant/antioxidant biomarkers of O. niloticus for seven days.
3.3.4. Hepato-Renal Related Parameters
According to Table 6, infected groups showed a significant increase (p < 0.05) in the levels of hepatic function indicators (ALT and AST) compared to the non-infected ones. Regarding the effects of CWPNC exposure, the 3 mg/L exposure level decreased significantly (p < 0.05) the level of hepatic function indicators compared to the 0 mg/L exposure level. Neither the S. agalactiae infection nor the CWPNC exposure affected (p > 0.05) the level of kidney function indicator (creatinine). Regarding the interaction between the S. agalactiae infection and the CWPNC exposure, the infected groups exposed to 0 mg/L CWPNC showed a significant increase (p < 0.05) in the levels of hepatorenal function indicators (ALT, AST, and creatinine) followed by the infected one exposed to 3 mg/L CWPNC level, then the non-infected groups exposed to 3 and 0 mg/L, CWPNC.

Table 6.
Effect of S. agalactiae infection and/or CWPNC as water exposure on hepatorenal biomarkers of O. niloticus for seven days.
3.3.5. Immune-Related Parameters
As indicated in Table 7, the immunological parameters (IgM, LYZ, and C3) in the infected group noticeably declined (p < 0.05) in comparison to those of non-infected fish. Regarding the CWPNC exposure, the 3 mg/L exposure level significantly increased (p < 0.05) the immunological parameters compared to 0 mg/L levels. Regarding the interaction between the S. agalactiae infection and the CWPNC exposure, the infected group exposed to the 0 mg/L CWPNC showed a significant decrease (p < 0.05) in the immunological parameters (IgM, LYZ, and C3). These parameters were improved in the infected group by treatment with 3 mg/L CWPNC. The non-infected group exposed to 3 mg/L CWPNC showed the highest values of the immunological parameters, followed by the non-infected one exposed to 0 mg/L CWPNC.

Table 7.
Effect of S. agalactiae infection and/or CWPNC as water exposure on immunological biomarkers of O. niloticus for seven days.
4. Discussion
Despite the significant growth that the aquaculture industry has seen over the past three decades, infectious illnesses are now a barrier to sustainable development [38]. Antibiotic resistance has grown and spread as a result of the extensive and regular usage of antibiotics in aquaculture. Due to the health risks associated with the use of antibiotics in animal husbandry, there is a growing awareness that they should be administered with greater caution. This is demonstrated by the most recent adoption of stricter rules on the use of antibiotics for preventative purposes and the presence of antibiotic residues in aquaculture products [39].
Innovative bacterial infection control methods are required for the aquaculture industry to grow sustainably in the future. Several recently developed alternative control strategies are evaluated in this review. Few of these techniques have been tested in actual aquaculture environments. To increase the likelihood of successfully preserving the animals and minimize resistance development, it is critical to continue developing various techniques that might be combined or employed in rotation. Nanoparticles, which use completely different antibacterial activity pathways than conventional antibiotics, offer a strong substitute [40]. Therefore, in this investigation, an innovative substitute for antibiotic therapy was tested against S. agalactiae both in vitro and in vivo.
The results of this study investigated the preparation of CWPNC and their characterization by different techniques [41]. Similar preparation methods had conducted in various studies using chitosan nanocapsules; Abdel Rahman et al. [23] had been studied the preparation of Ocimium basilicum using chitosan nanocapsules. Another study synthesized the neem leave extract using a chitosan nanocapsule [42]. The results showed that CWPNC had an amorphous nature with a BET surface area value is 61.1 m2/g with a pore volume determined by the DH method is 1.1 cc/nm. The microporous and high specific surface area of chitosan–white poplar nanocapsules affect its bioactivity due to increasing chemical activity. CWPNC had wide particle size distribution due to the presence of more than one peak in the DLS curve at 18 nm and 37 nm may be due to fiber-shaped particles that have two different dimensions. The results of zeta potential indicated that CWPNC had a high zeta potential of about −37 mV, which explains the colloidal of CWPNC and negative charge due to the coating of white poplar extract where chitosan has a positive charge.
The results of the in vitro study revealed that CWPNC showed antibacterial properties against S. agalactiae. Further, chitosan exhibited a lower antibacterial activity against S. agalactiae compared to CWPNC. The antibacterial properties of CWPNC in this study could be explained by the synergistic effects of both white poplar leaf extract and chitosan; both have antimicrobial activity against various bacterial strains. A previous study had reported the antibacterial activity of white poplar extract against Mycobacterium spp. [43]. Several studies have reported the antibacterial activity of chitosan [44,45,46]. In addition, the presence of phenolic substances in the white poplar leave extract may prevent the nucleic acid synthesis of the bacterial cell [47,48] or via cytoplasmic membrane modification [43,49,50]. Additionally, phenolic chemicals are in charge of inhibiting the bacterial enzymes that produce and modify energy metabolic mechanisms by interfering with the ATP production cycle and by interfering with the transfer of nutrients or metabolites [49]. Moreover, chitosan interacts with surface molecules in addition to inhibiting mRNA by binding to bacterial DNA [51]. As stated by Ganan et al. [52] and Raafat et al. [53], chitosan has amine groups (NH3) of glucosamine, which may be a key factor in its ability to interact with negatively charged surface elements of many microorganisms and cause extensive surface alterations that lead to the leakage of intracellular substances and cell death.
The results of the in vivo exposure demonstrated that S. agalactiae produced worth effects on O. niloticus through disruption of the behavior response (decrease the swimming behavior and loss of the escape reflex), lowered survivability (33.33%), as well as producing serious disease signs, including hemorrhagic skin and fin rot. Similar results had been recorded in Nile tilapia infected with S. agalactiae [54,55,56]. These pathogenic consequences could be attributed to the virulence components of S. agalactiae (adhesins, invasins, and immune evasins), which are responsible for their pathogenicity in fish [57]. Adhesins enable the attachment of S. agalactiae to the fish cell wall [58,59]. Invasins are responsible for the crossing of the bacteria to the mucosal and blood-brain barrier [60]. Immune evasins are responsible for the escape from host immunity [61]. CWPNC therapy notably retrieved the previous clinical signs and enhanced the survivability (66.67%) of the infected fish; this could be attributed to the CWPNC antibacterial activity that was proven in this study by the in vitro assay.
Oxidative stress is caused by the imbalance between the antioxidant enzymes and the excessive reactive oxygen species (ROS) production [62,63,64]. Fish has an enzymatic antioxidant mechanism that protects them from oxidative stress [65,66,67]. MDA is the end product of lipid peroxidation caused by oxidative damage [68]. Since CAT is a sensitive enzyme, different factors, such as an abundance of superoxide radicals, might affect its activity. H2O2 and this enzyme combine to create water and oxygen molecules [69]. Antioxidant enzymes known as SOD serve as the first lines of defense against ROS [70]. On this subject, the S. agalactiae-infected group in this study exhibited lower levels of CAT and SOD but higher levels of MDA. The virulence components and metabolites of S. agalactiae could implicate in the cell wall increased lipid peroxidation (MDA) and ROS production, which could impair the antioxidant system and lead to oxidative stress. S. agalactiae infection elicited comparable features in Nile tilapia [71]. On the other hand, the application of CWPNC increased the activity of antioxidant enzymes in S. agalactiae-infected fish, proving the antioxidant activity of CWPNC. The previous study proved that white polar leaf has abundant phenolics which destroy free radicals, prevent lipid peroxidation, and display a variety of physiological antioxidant functions [72].
Increased hepato-renal function indicators (ALT, AST, and creatinine) in the serum indicate liver and kidney dysfunction [73,74,75]. S. agalactiae infection increased the hepatorenal function indicators in this study. In line with this study, a previous study proved that S. agalactiae produced severe pathological effects in the hepatorenal tissue [76]. Moreover, CWPNC therapy modulates the hepatorenal function indicators in the infected fish; these results could be related to the antioxidant properties of CWPNC proved here in the current study.
Immune function indicators (IgM, LYZ, and C3) were assessed in this study. LYZ is a bacteriolytic enzyme that is considered an indicator of a non-specific immune response against microbial invasion [75,77,78]. The primary systemic immunoglobulin usually seen in fish serum is IgM. Through several processes, including neutralization, complement pathway activation, and phagocytosis, IgM protects the fish against infection [79]. In fish and other vertebrates, the complement system is a collection of proteins that interact with both innate and adaptive immune systems. The complement system is a prominent and essential actor in the humeral component of innate immunity [80,81]. Immune function indicators (IgM, LYZ, and C3) were lowered in the S. agalactiae-infected fish; these results could be attributed to the virulence component of the bacteria. Several previously documented that S. agalactiae suppresses the immune functions of fish [82,83,84]. Treatment of S. agalactiae-infected fish with 3 mg/L CWPNC retrieved immune functions produced by the infection. Similar through enhancing the levels of IgM, LYZ, and C3, as well as enhancing survivability. These results could be attributed to the antibacterial properties of CWPNC that prevent disease progression and enhance the antioxidant and immune status of the infected fish.
5. Conclusions
It can be concluded that CWPNC had greater antibacterial activity against S. agalactiae based on the results of disc diffusion and MIC and MBC assays. CWPNC at a level of 3 mg/L ameliorated the negative effects of S. agalactiae challenge in O. niloticus health. Future studies are recommended for looking into different fish pathogens’ responses to CWPNC’s antimicrobial capabilities for promoting the sustainable development of the aquaculture industry.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/fishes8040199/s1, Figure S1. The XRD curve of CWPNC (A), TEM image of CWPNC (B); Figure S2. The 3D AFM image of CWPNC (A), 2D AFM image of CWPNC (B); Figure S3. Isotherm of CWPNC (A), pore size and pore volume according to DH method for CWPNC (B), DLS curve of CWPNC (C), and Zeta potential of CWPNC (D).
Author Contributions
Conceptualization: A.N.A.R., S.H.I., M.M.S.F., A.A.A., E.M.Y., S.S.K., M.M.E.-S., A.E.A., S.J.D. and R.E.I. Methodology: A.N.A.R., S.H.I., M.M.S.F., A.A.A., E.M.Y., M.M.E.-S., A.E.A., S.J.D. and R.E.I. Software and data curation: R.E.I. Writing—Original draft preparation: R.E.I. Writing—Reviewing and Editing: A.N.A.R., S.S.K. and R.E.I. All authors have read and agreed to the published version of the manuscript.
Funding
The APC was funded by the Researches Supporting Project (RSPD2023R700), King Saud University, Riyadh, Saudi Arabia.
Institutional Review Board Statement
The experimentation was performed in the Aquatic Animal Medicine Department, Faculty of Veterinary Medicine, Zagazig University, and supervised by the Animal Use in Research Committee with ethical approval code ZU-IACUC/2/F/310/2022. All experimental procedures were conducted in compliance with the ethical guidelines approved by the National Institutes of Health for Use and Treatment of Laboratory Animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated or analyzed during the current study are not publicly available but are available from the corresponding author upon reasonable request.
Acknowledgments
This work was supported by the Researches Supporting Project (RSPD2023R700), King Saud University, Riyadh, Saudi Arabia. The authors thank the Aquatic Animal Medicine Department, Faculty of Veterinary Medicine, Zagazig University, for their kind help during the experimental procedures.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Faggio, C.; Piccione, G.; Marafioti, S.; Arfuso, F.; Fortino, G.; Fazio, F. Metabolic response to monthly variations of Sparus aurata reared in Mediterranean on-shore tanks. Turk. J. Fish. Aquat. Sci. 2014, 14, 567–574. [Google Scholar]
- Faggio, C.; Arfuso, F.; Piccione, G.; Zumbo, A.; Fazio, F. Effect of three different anticoagulants and storage time on haematological parameters of Mugil cephalus (Linneaus, 1758). Turk. J. Fish. Aquat. Sci. 2014, 14, 615–621. [Google Scholar]
- Afewerki, S.; Asche, F.; Misund, B.; Thorvaldsen, T.; Tveteras, R. Innovation in the Norwegian aquaculture industry. Rev. Aquac. 2023, 15, 759–771. [Google Scholar] [CrossRef]
- Department, A.O. The State of World Fisheries and Aquaculture, 2000; Food & Agriculture Organization: Rome, Italy, 2000; Volume 3. [Google Scholar]
- Falk, T.M.; Abban, E.K. Genetic diversity of the Nile tilapia Oreochromis niloticus (Teleostei, Cichlidae) from the Volta System in Ghana. Biodivers. Manag. Util. West Afr Fishes. 2004, 13–15. [Google Scholar]
- Pridgeon, J.W.; Klesius, P.H. Major bacterial diseases in aquaculture and their vaccine development. CABI Rev. 2012, 11, 1–16. [Google Scholar] [CrossRef]
- Rajme-Manzur, D.; Gollas-Galvan, T.; Vargas-Albores, F.; Martínez-Porchas, M.; Hernández-Oñate, M.Á.; Hernández-López, J. Granulomatous bacterial diseases in fish: An overview of the host’s immune response. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2021, 261, 111058. [Google Scholar] [CrossRef]
- Amal, M.; Zamri-Saad, M. Streptococcosis in tilapia (Oreochromis niloticus): A review. Pertanika J. Trop. Agric. Sci. 2011, 34, 195–206. [Google Scholar]
- Ghetas, H.; Neiana, A.; Khalil, R.; AM, H.; Khallaf, M. Streptococcus agalactiae Isolation and Characterization in Nile Tilapia (Oreochromis niloticus) with Histopathological Studies. J. Curr. Vet. Res. 2021, 3, 70–79. [Google Scholar] [CrossRef]
- Cabello, F.C. Heavy use of prophylactic antibiotics in aquaculture: A growing problem for human and animal health and for the environment. Environ. Microbiol. 2006, 8, 1137–1144. [Google Scholar] [CrossRef]
- Maron, D.; Smith, T.; Nachman, K. Restrictions on antimicrobial use in food animal production: An international regulatory and economic survey. Glob. Health 2013, 9, 48–58. [Google Scholar] [CrossRef]
- Binh, V.N.; Dang, N.; Anh, N.T.K.; Thai, P.K. Antibiotics in the aquatic environment of Vietnam: Sources, concentrations, risk and control strategy. Chemosphere 2018, 197, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Forouzi, A.; Ghasemnezhad, A.; Nasrabad, R.G. Phytochemical response of Stevia plant to growth promoting microorganisms under salinity stress. S. Afr. J. Bot. 2020, 134, 109–118. [Google Scholar] [CrossRef]
- Karrenberg, S.; Edwards, P.J.; Kollmann, J. The life history of Salicaceae living in the active zone of floodplains. Freshw. Biol. 2002, 47, 733–748. [Google Scholar] [CrossRef]
- Nassima, B.; Nassima, B.; Riadh, K. Antimicrobial and antibiofilm activities of phenolic compounds extracted from Populus nigra and Populus alba buds (Algeria). Brazilian J. Pharm. Sci. 2019, 55, e18114. [Google Scholar] [CrossRef]
- Emerich, D.F.; Thanos, C.G. Targeted nanoparticle-based drug delivery and diagnosis. J. Drug Target. 2007, 15, 163–183. [Google Scholar] [CrossRef]
- Shaffer, C. Nanomedicine transforms drug delivery. Drug Discov. Today 2005, 10, 1581–1582. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Tsutsumi, Y.; Nakagawa, S. Development of nanomedicine using intracellular DDS. Nihon. Rinsho. 2006, 64, 247–252. [Google Scholar]
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Nanomedicine: Current status and future prospects. FASEB J. 2005, 19, 311–330. [Google Scholar] [CrossRef]
- Emerich, D.F. Nanomedicine–prospective therapeutic and diagnostic applications. Expert Opin. Biol. Ther. 2005, 5, 1–5. [Google Scholar] [CrossRef]
- Sahoo, S.; Parveen, S.; Panda, J. The present and future of nanotechnology in human health care. Nanomedicine 2007, 3, 20–31. [Google Scholar] [CrossRef]
- Pillai, C.K.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Ibrahim, R.E.; Elshopakey, G.E.; Abd El-Rahman, G.I.; Ahmed, A.I.; Altohamy, D.E.; Zaglool, A.W.; Younis, E.M.; Abdelwarith, A.A.; Davies, S.J.; Al-Harthi, H.F. Palliative role of colloidal silver nanoparticles synthetized by moringa against Saprolegnia spp. infection in Nile Tilapia: Biochemical, immuno-antioxidant response, gene expression, and histopathological investigation. Aquac. Rep. 2022, 26, 101318. [Google Scholar] [CrossRef]
- El-Houseiny, W.; Mansour, M.F.; Mohamed, W.A.; Al-Gabri, N.A.; El-Sayed, A.A.; Altohamy, D.E.; Ibrahim, R.E. Silver nanoparticles mitigate Aeromonas hydrophila-induced immune suppression, oxidative stress, and apoptotic and genotoxic effects in Oreochromis niloticus. Aquaculture 2021, 535, 736430. [Google Scholar] [CrossRef]
- Ma, Z.; Garrido-Maestu, A.; Jeong, K.C. Application, mode of action, and in vivo activity of chitosan and its micro-and nanoparticles as antimicrobial agents: A review. Carbohydr. Polym. 2017, 176, 257–265. [Google Scholar] [CrossRef]
- Liu, C.; Wang, L.; Xu, H.; Wang, S.; Gao, S.; Ji, X.; Xu, Q.; Lan, W. “One pot” green synthesis and the antibacterial activity of g-C3N4/Ag nanocomposites. Mater. Lett. 2016, 164, 567–570. [Google Scholar] [CrossRef]
- Aliasghari, A.; Rabbani Khorasgani, M.; Vaezifar, S.; Rahimi, F.; Younesi, H.; Khoroushi, M. Evaluation of antibacterial efficiency of chitosan and chitosan nanoparticles on cariogenic streptococci: An in vitro study. Iran. J. Microbiol. 2016, 8, 93–100. [Google Scholar]
- Ibrahim, R.E.; Amer, S.A.; Farroh, K.Y.; Al-Gabri, N.A.; Ahmed, A.I.; El-Araby, D.A.; Ahmed, S.A. The effects of chitosan-vitamin C nanocomposite supplementation on the growth performance, antioxidant status, immune response, and disease resistance of Nile tilapia (Oreochromis niloticus) fingerlings. Aquaculture 2021, 534, 736269. [Google Scholar] [CrossRef]
- Zetterberg, C.; Öfverholm, T. Carpal tunnel syndrome and other wrist/hand symptoms and signs in male and female car assembly workers. Int. J. Ind. Ergon. 1999, 23, 193–204. [Google Scholar] [CrossRef]
- Jenkins, J.A.; Bart, H., Jr.; Bowker, J.D.; Bowser, P.; MacMillan, J.; Nickum, J.; Rose, J.; Sorensen, P.; Whitledge, G.; Rachlin, J.W.J.B.; et al. Guidelines for the Use of Fishes in Research; American Fisheries Society: Bethesda, MD, USA, 2014; p. 104. [Google Scholar]
- Nishikimi, M.; Rao, N.A.; Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Aebi, H. [13] Catalase in vitro. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1984; Volume 105, pp. 121–126. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analyt. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Murray, R. Aspartate aminotransferase. In Clinical Chemistry; Kaplan, L.A., Pesce, A.J., Eds.; The CV Mosby Co.: Toronto, ON, Canada, 1984; pp. 418, 437, 1257–1260. [Google Scholar]
- Burtis, C.A.; Ashwood, E.R. Tietz Textbook of Clinical Chemistry; Amer Assn for Clinical Chemistry: Washington, DC, USA, 1994. [Google Scholar]
- Bartles, H.; Bohmer, M.; Heirli, C. Colorimetric kinetic method for creatinine determination in serum and urine. Clin. Chem. Acta 1972, 37, 193–195. [Google Scholar]
- Caruso, D.; Schlumberger, O.; Dahm, C.; Proteau, J.-P. Plasma lysozyme levels in sheatfish Silurus glanis (L.) subjected to stress and experimental infection with Edwardsiella tarda. Aquac. Res. 2002, 33, 999–1008. [Google Scholar] [CrossRef]
- Pérez-Sánchez, T.; Mora-Sánchez, B.; Balcázar, J.L. Biological Approaches for Disease Control in Aquaculture: Advantages, Limitations and Challenges. Trends Microbiol. 2018, 26, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Defoirdt, T.; Sorgeloos, P.; Bossier, P. Alternatives to antibiotics for the control of bacterial disease in aquaculture. Curr. Opin. Microbiol. 2011, 14, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Seil, J.T.; Webster, T.J. Antimicrobial applications of nanotechnology: Methods and literature. Int. J. Nanomed. 2012, 7, 2767–2781. [Google Scholar]
- Banna, A.H.E.; Youssef, F.S.; Elzorba, H.Y.; Soliman, A.M.; Mohamed, G.G.; Ismail, S.H.; Mousa, M.R.; Elbanna, H.A.; Osman, A.S. Evaluation of the wound healing effect of neomycin-silver nano-composite gel in rats. Int. J. Immunopathol. Pharmacol. 2022, 36, 03946320221113486. [Google Scholar] [PubMed]
- Rajendran, R.; Radhai, R.; Balakumar, C.; Ahamed, H.A.M.; Vigneswaran, C.; Vaideki, K. Synthesis and characterization of neem chitosan nanocomposites for development of antimicrobial cotton textiles. J. Eng. Fiber. Fabr. 2012, 7, 155892501200700116. [Google Scholar] [CrossRef]
- Amina, C.H.; Souraya, E.G.; Abdellatif, H.; Suzanna, D.; Hakima, S.; Saad, I.; Mohammed, I. Antimycobacterial activity of Populus alba leaf extracts. J. Med. Plant. Res. 2013, 7, 1015–1021. [Google Scholar]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Yildirim-Aksoy, M.; Beck, B. Antimicrobial activity of chitosan and a chitosan oligomer against bacterial pathogens of warmwater fish. J. Appl. Microbiol. 2017, 122, 1570–1578. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.-C.; Su, Y.P.; Chen, C.-C.; Jia, G.; Wang, H.L.; Wu, J.G.; Lin, J.G. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharmacol. Sin. 2004, 25, 932–936. [Google Scholar]
- Ulanowska, K.; Tkaczyk, A.; Konopa, G.; Węgrzyn, G. Differential antibacterial activity of genistein arising from global inhibition of DNA, RNA and protein synthesis in some bacterial strains. Arch. Microbiol. 2006, 184, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Bouarab-Chibane, L.; Forquet, V.; Lantéri, P.; Clément, Y.; Léonard-Akkari, L.; Oulahal, N.; Degraeve, P.; Bordes, C. Antibacterial Properties of Polyphenols: Characterization and QSAR (Quantitative Structure-Activity Relationship) Models. Front. Microbiol. 2019, 10, 829. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, S.; Ahmed, S.; Wang, F.; Gu, Y.; Zhang, C.; Chai, X.; Wu, Y.; Cai, J.; Cheng, G. Antimicrobial activity and resistance: Influencing factors. Front. Pharm. 2017, 8, 364. [Google Scholar] [CrossRef] [PubMed]
- Chusnie, T.; Lamb, A.J. Antimicrobial activity of flavonoid. Int. J. Antimicrob. Agent 2005, 26, 343–356. [Google Scholar]
- Goy, R.C.; Britto, D.d.; Assis, O.B. A review of the antimicrobial activity of chitosan. Polímeros 2009, 19, 241–247. [Google Scholar] [CrossRef]
- Ganan, M.; Carrascosa, A.; Martinez-Rodriguez, A. Antimicrobial activity of chitosan against Campylobacter spp. and other microorganisms and its mechanism of action. J. Food Prot. 2009, 72, 1735–1738. [Google Scholar] [CrossRef]
- Raafat, D.; Von Bargen, K.; Haas, A.; Sahl, H.-G. Insights into the mode of action of chitosan as an antibacterial compound. Appl. Environ. Microbiol. 2008, 74, 3764–3773. [Google Scholar] [CrossRef]
- Figueiredo, H.; Carneiro, D.; Faria, F.; Costa, G. Streptococcus agalactiae associado à meningoencefalite e infecção sistêmica em tilápia-do-Nilo (Oreochromis niloticus) no Brasil. Arq. Bras. Med. Vet. Zootec. 2006, 58, 678–680. [Google Scholar] [CrossRef]
- Pretto-Giordano, L.G.; Müller, E.E.; Freitas, J.C.d.; Silva, V.G.d. Evaluation on the Pathogenesis of Streptococcus agalactiae in Nile Tilapia (Oreochromis niloticus). Brazilian Arch. Biol. Technol. 2010, 53, 87–92. [Google Scholar] [CrossRef]
- Salvador, R.; Muller, E.E.; Freitas, J.C.d.; Leonhadt, J.H.; Pretto-Giordano, L.G.; Dias, J.A. Isolation and characterization of Streptococcus spp. group B in Nile tilapias (Oreochromis niloticus) reared in hapas nets and earth nurseries in the northern region of Parana State, Brazil. Ciência Rural 2005, 35, 1374–1378. [Google Scholar] [CrossRef]
- Lin, F.P.-Y.; Lan, R.; Sintchenko, V.; Gilbert, G.L.; Kong, F.; Coiera, E. Computational bacterial genome-wide analysis of phylogenetic profiles reveals potential virulence genes of Streptococcus agalactiae. PLoS ONE 2011, 6, e17964. [Google Scholar] [CrossRef] [PubMed]
- Pietrocola, G.; Visai, L.; Valtulina, V.; Vignati, E.; Rindi, S.; Arciola, C.R.; Piazza, R.; Speziale, P. Multiple interactions of FbsA, a surface protein from Streptococcus agalactiae, with fibrinogen: Affinity, stoichiometry, and structural characterization. Biochemistry 2006, 45, 12840–12852. [Google Scholar] [CrossRef] [PubMed]
- Buscetta, M.; Papasergi, S.; Firon, A.; Pietrocola, G.; Biondo, C.; Mancuso, G.; Midiri, A.; Romeo, L.; Teti, G.; Speziale, P. FbsC, a novel fibrinogen-binding protein, promotes Streptococcus agalactiae-host cell interactions. J. Biol. Chem. 2014, 289, 21003–21015. [Google Scholar] [CrossRef]
- Landwehr-Kenzel, S.; Henneke, P. Interaction of Streptococcus agalactiae and cellular innate immunity in colonization and disease. Front. Immunol. 2014, 5, 519. [Google Scholar] [CrossRef]
- Kannika, K.; Pisuttharachai, D.; Srisapoome, P.; Wongtavatchai, J.; Kondo, H.; Hirono, I.; Unajak, S.; Areechon, N. Molecular serotyping, virulence gene profiling and pathogenicity of Streptococcus agalactiae isolated from tilapia farms in Thailand by multiplex PCR. J. Appl. Microbiol. 2017, 122, 1497–1507. [Google Scholar] [CrossRef]
- Ibrahim, R.E.; Amer, S.A.; Shahin, S.A.; Darwish, M.I.; Albogami, S.; Abdelwarith, A.A.; Younis, E.M.; Abduljabbar, M.H.; Davies, S.J.; Attia, G.A. Effect of fish meal substitution with dried bovine hemoglobin on the growth, blood hematology, antioxidant activity and related genes expression, and tissue histoarchitecture of Nile tilapia (Oreochromis niloticus). Aquac. Rep. 2022, 26, 101276. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Ibrahim, R.E.; Elshopakey, G.E.; Khamis, T.; Abdel-Ghany, H.M.; Abdelwarith, A.A.; Younis, E.M.; Davies, S.J.; Elabd, H.; Elhady, M. Immune-antioxidant trait, growth, splenic cytokines expression, apoptosis, and histopathological alterations of Oreochromis niloticus exposed to sub-lethal copper toxicity and fed thyme and/or basil essential oils enriched diets. Fish Shellfish Immunol. 2022, 131, 1006–1018. [Google Scholar] [CrossRef]
- Amer, S.A.; Farahat, M.; Khamis, T.; Abdo, S.A.; Younis, E.M.; Abdel-Warith, A.-W.A.; Reda, R.; Ali, S.A.; Davies, S.J.; Ibrahim, R.E. Evaluation of Spray-Dried Bovine Hemoglobin Powder as a Dietary Animal Protein Source in Nile Tilapia, Oreochromis niloticus. Animals 2022, 12, 3206. [Google Scholar] [CrossRef]
- Fontagné-Dicharry, S.; Lataillade, E.; Surget, A.; Larroquet, L.; Cluzeaud, M.; Kaushik, S. Antioxidant defense system is altered by dietary oxidized lipid in first-feeding rainbow trout (Oncorhynchus mykiss). Aquaculture 2014, 424, 220–227. [Google Scholar] [CrossRef]
- Abdel Rahman, A.N.; Van Doan, H.; Elsheshtawy, H.M.; Dawood, A.; Salem, S.M.; Sheraiba, N.I.; Masoud, S.R.; Abdelnaeim, N.S.; Khamis, T.; Alkafafy, M. Dietary Salvia officinalis leaves enhances antioxidant-immune-capacity, resistance to Aeromonas sobria challenge, and growth of Cyprinus carpio. Fish Shellfish Immunol. 2022, 127, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Abdel Rahman, A.N.; Shakweer, M.S.; Algharib, S.A.; Abdelaty, A.I.; Kamel, S.; Ismail, T.A.; Daoush, W.M.; Ismail, S.H.; Mahboub, H.H. Silica nanoparticles acute toxicity alters ethology, neuro-stress indices, and physiological status of African catfish (Clarias gariepinus). Aquac. Rep. 2022, 23, 101034. [Google Scholar] [CrossRef]
- Garcia, D.; Lima, D.; da Silva, D.G.H.; de Almeida, E.A. Decreased malondialdehyde levels in fish (Astyanax altiparanae) exposed to diesel: Evidence of metabolism by aldehyde dehydrogenase in the liver and excretion in water. Ecotoxicol. Environ. Saf. 2020, 190, 110107. [Google Scholar] [CrossRef]
- Ozmen, I.; Bayir, A.; Cengiz, M.; Sirkecioglu, A.; Atamanalp, M. Effects of water reuse system on antioxidant enzymes of rainbow trout (Oncorhynchus mykiss W., 1792). Vet. Med. 2004, 49, 373. [Google Scholar] [CrossRef]
- Zelko, I.N.; Mariani, T.J.; Folz, R.J. Superoxide dismutase multigene family: A comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic. Biol. Med. 2002, 33, 337–349. [Google Scholar] [CrossRef]
- Baldissera, M.D.; Souza, C.F.; da Silva, A.S.; Velho, M.C.; Ourique, A.F.; Baldisserotto, B. Benefits of nanotechnology: Dietary supplementation with nerolidol-loaded nanospheres increases survival rates, reduces bacterial loads and prevents oxidative damage in brains of Nile tilapia experimentally infected by Streptococcus agalactiae. Microb. Pathog. 2020, 141, 103989. [Google Scholar] [CrossRef] [PubMed]
- Danise, T.; Innangi, M.; Curcio, E.; Piccolella, S.; Fioretto, A.; Pacifico, S. White poplar (Populus alba L.) leaf waste recovery and intercropping outcome on its polyphenols. Ind. Crops Prod. 2021, 171, 113866. [Google Scholar] [CrossRef]
- Mohamed, W.A.; El-Houseiny, W.; Ibrahim, R.E.; Abd-Elhakim, Y.M. Palliative effects of zinc sulfate against the immunosuppressive, hepato-and nephrotoxic impacts of nonylphenol in Nile tilapia (Oreochromis niloticus). Aquaculture 2019, 504, 227–238. [Google Scholar] [CrossRef]
- Abdel Rahman, A.N.; Mohamed, A.A.-R.; Dahran, N.; Farag, M.F.; Alqahtani, L.S.; Nassan, M.A.; AlThobaiti, S.A.; El-Naseery, N.I. Appraisal of sub-chronic exposure to lambada-cyhalothrin and/or methomyl on the behavior and hepato-renal functioning in Oreochromis niloticus: Supportive role of taurine-supplemented feed. Aquat. Toxicol. 2022, 250, 106257. [Google Scholar] [CrossRef]
- Abdel Rahman, A.N.; El-Bouhy, Z.; Wahbah, M.; Ahmed, S. Fisheries. Effects of dietary turmeric and clove powder on growth and immune response of the Nile tilapia. Egypt. J. Aquat. Biol. Fish. 2020, 24, 589–608. [Google Scholar] [CrossRef]
- Zamri-Saad, M.; Amal, M.N.A.; Siti-Zahrah, A. Pathological Changes in Red Tilapias (Oreochromis spp.) Naturally Infected by Streptococcus agalactiae. J. Comp. Pathol. 2010, 143, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Uribe, C.; Folch, H.; Enríquez, R.; Moran, G. Innate and adaptive immunity in teleost fish: A review. Vet. Med. 2011, 56, 486. [Google Scholar] [CrossRef]
- Reda, R.M.; Maricchiolo, G.; Quero, G.M.; Basili, M.; Aarestrup, F.M.; Pansera, L.; Mirto, S.; Abd El-Fattah, A.H.; Alagawany, M.; Abdel Rahman, A.N. Rice protein concentrate as a fish meal substitute in Oreochromis niloticus: Effects on immune response, intestinal cytokines, Aeromonas veronii resistance, and gut microbiota composition. Fish Shellfish Immunol. 2022, 126, 237–250. [Google Scholar] [CrossRef]
- Jones, E.M.; Oliver, L.P.; Ma, J.; Leeuwis, R.H.J.; Myrsell, V.; Arkoosh, M.R.; Dietrich, J.P.; Schuster, C.M.; Hawkyard, M.; Gamperl, A.K.; et al. Production of a monoclonal antibody specific to sablefish (Anoplopoma fimbria) IgM and its application in ELISA, western blotting, and immunofluorescent staining. Fish Shellfish Immunol. 2022, 130, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Kania, P.W.; Buchmann, K. Complement Activation in Fish with Emphasis on MBL/MASP. In Principles of Fish Immunology; Springer: Berlin/Heidelberg, Germany, 2022; pp. 279–300. [Google Scholar]
- Mahboub, H.H.; Shahin, K.; Mahmoud, S.M.; Altohamy, D.E.; Husseiny, W.A.; Mansour, D.A.; Shalaby, S.I.; Gaballa, M.M.; Shaalan, M.; Alkafafy, M.; et al. Silica nanoparticles are novel aqueous additive mitigating heavy metals toxicity and improving the health of African catfish, Clarias gariepinus. Aquat. Toxicol. 2022, 249, 106238. [Google Scholar] [PubMed]
- Wang, J.; Lu, D.-Q.; Jiang, B.; Luo, H.-L.; Lu, G.-L.; Li, A.-X. The effect of intermittent hypoxia under different temperature on the immunomodulation in Streptococcus agalactiae vaccinated Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2018, 79, 181–192. [Google Scholar] [CrossRef]
- Wu, Y.-R.; Gong, Q.-F.; Fang, H.; Liang, W.-W.; Chen, M.; He, R.-J. Effect of Sophora flavescens on non-specific immune response of tilapia (GIFT Oreochromis niloticus) and disease resistance against Streptococcus agalactiae. Fish Shellfish Immunol. 2013, 34, 220–227. [Google Scholar] [CrossRef]
- Lin, Y.-S.; Saputra, F.; Chen, Y.-C.; Hu, S.-Y. Dietary administration of Bacillus amyloliquefaciens R8 reduces hepatic oxidative stress and enhances nutrient metabolism and immunity against Aeromonas hydrophila and Streptococcus agalactiae in zebrafish (Danio rerio). Fish Shellfish Immunol. 2019, 86, 410–419. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).