Abstract
Conditional power based on classical Brownian motion (BM) has been widely used in sequential monitoring of clinical trials, including those with the covariate adaptive randomization design (CAR). Due to some uncontrollable factors, the sequential test statistics under CAR procedures may not satisfy the independent increment property of BM. We confirm the invalidation of BM when the error terms in the linear model with CAR design are not independent and identically distributed. To incorporate the possible correlation structure of the increment of the test statistic, we utilize the fractional Brownian motion (FBM). We conducted a comparative study of the conditional power under BM and FBM. It was found that the conditional power under FBM assumption was mostly higher than that under BM assumption when the Hurst exponent was greater than 0.5.
1. Introduction
Clinical trials, aiming to evaluate the safety and efficacy of drugs and medical devices in target populations, play an important role in the development of public health and medicine [1]. Adaptive randomized clinical design improves the trial, based on the accumulated data and changing environment, making clinical trials more efficient, flexible, and ethically reasonable [2].
The treatment effects estimated from unadjusted models may not be appropriate when the covariates are not balanced. Meanwhile, some covariates, such as elevated values of biomarkers that were found to affiliate with disease status in the translational research, may be critical in determining the treatment effects in clinical trials [3]. For example, biomarker HR23B is closely related to and used to indicate the effectiveness of histone deacetylase inhibitors-based therapy for tumors [4]. To address the problem of covariate imbalance, a useful tool is the Covariate Adaptive Randomization (CAR) procedure in which participants are assigned to different treatment groups based on previous participants’ assignment, previous participants’ covariates, and current participants’ covariates, such that the asymmetries across the subgroups are minimized [5]. Although the complete randomization is good at eliminating the selection bias, the CAR design is a more reasonable comprise between reducing the selection bias and balancing covariates assignments [6]. The rigorous theory of covariate adaptive randomized clinical trials has been developed more recently [7]. Thereafter, progress has been made in research on statistical inference with CAR designs [8,9].
Traditionally, classical Brownian motion (BM) is a fundamental theory for monitoring outcome effects in clinical trials, including those with CAR designs [10,11,12,13,14]. It has been proved that the sequential test statistics of covariate adaptive clinical trials follow Brownian motion asymptotically under some regularized conditions [15].
A condition of performing the hypothesis testing of covariate adaptive randomized clinical trials is that the underlying error terms are independent and identically distributed (iid). In addition, independent increments are a property of classical Brownian motion [16]. However, the independent increment property of the test statistics may not be completely met in some situations. For example, some patients may enter the trial during the same season; some patients may be treated by the same hospital or the same physician. Therefore, the error terms from the model may be correlated and follow special covariance patterns. Given the situation that most of the previous theoretical research into sequential monitoring of CAR designs was based on the Brownian motion assumption, it is necessary to explore the stochastic properties of the sequential monitoring process when error structures are not independent and identically distributed. We propose fractional Brownian motion (FBM) as a more valid tool to investigate the outcomes of clinical trials with correlated error structures.
FBM, annotated as “”, is a Gaussian process with E(, and ½, where the Hurst coefficient (H), in the range of , is a parameter of the FBM, describing the long-term dependence of the process [17,18,19]. FBM is a Markov process when [20,21]. The maximum likelihood estimation (MLE) method was proposed for estimating the Hurst coefficient underlying FBM in clinical trials [22]. The log likelihood function of the observed value of was nlog log where is the variance covariance matrix of [22].
In this paper, comprehensive simulations of the sequential monitoring of CAR procedures were conducted to investigate the breakdown of BM when the independent increment assumption was not met. We further calculated the conditional power (CP) under the null hypothesis, with the BM assumption and with the FBM assumption, respectively.
Section 2 of this paper includes the test statistics and theoretical properties under covariate adaptive randomized clinical trials with correlated error structures. In Section 3, results from numerical simulations are provided to estimate the Hurst exponents for sequential monitoring of CAR procedures when error structures are not iid. Conditional powers are calculated and compared between BM and FBM assumptions in Section 4. Conclusions and discussions are found in Section 5.
3. Simulations for Misspecification Scenarios
Since the sequential test statistics cannot converge to asymptotically Brownian motion when error terms are correlated, we propose a larger class of fractional Brownian motion for the stochastic structure of the test statistic. Maximum likelihood method was used to estimate the Hurst exponents of the FBM for the sequential monitoring processes under the misspecification assumption. If the mean estimation of H values deviates significantly from 0.5, the sequential monitoring processes would be confirmed as not converging to BM. Error terms in the model (1) were assumed to follow specific correlated patterns. Increments of fractional Brownian motion, defined as , were used in the error terms ε of our simulations [24]. fbm() function in the R software is a way to create one dimension FBM series (t) [25]. Covariance of the increments of fractional Brownian motion is:
Incorrect estimators (4) and (5) and incorrect classical hypothesis test statistics (2) were used to build the sequential monitoring processes without considering the covariance terms.
In the Equation (1), , are the probability of success respectively in the Bernoulli distribution when the covariates , are binary variables. , were set up as 0.5, 0.5, 1, 1, 0.5, 0.5, respectively. 1000 replications were used for all the simulations. Patients were assumed to be sequentially randomized into two treatment groups by the block randomization (BR) (by blockrand() function in R software), stratified permuted block randomization (SPB) [26], and Pocock and Simon minimization designs (PS) [27] in the simulation studies consecutively. No covariate, two discrete covariates, and two continuous covariates situations were illustrated under misspecification scenarios in the simulation studies.
Assume 4000 patients were recruited in a clinical trial study with uniformly distributed enter time. An interim analysis would be done after every 100 new patients had finished the study. In total, 40 interim results were obtained. The maximum likelihood method was used to estimate the Hurst exponent (H) for normalized value from the sequential test (3) in the entire paper. When H, this indicated an uncorrelated process, corresponding to classical Brownian motion. It was shown that has a long range dependence property when 0.5 < H <1 [28,29].
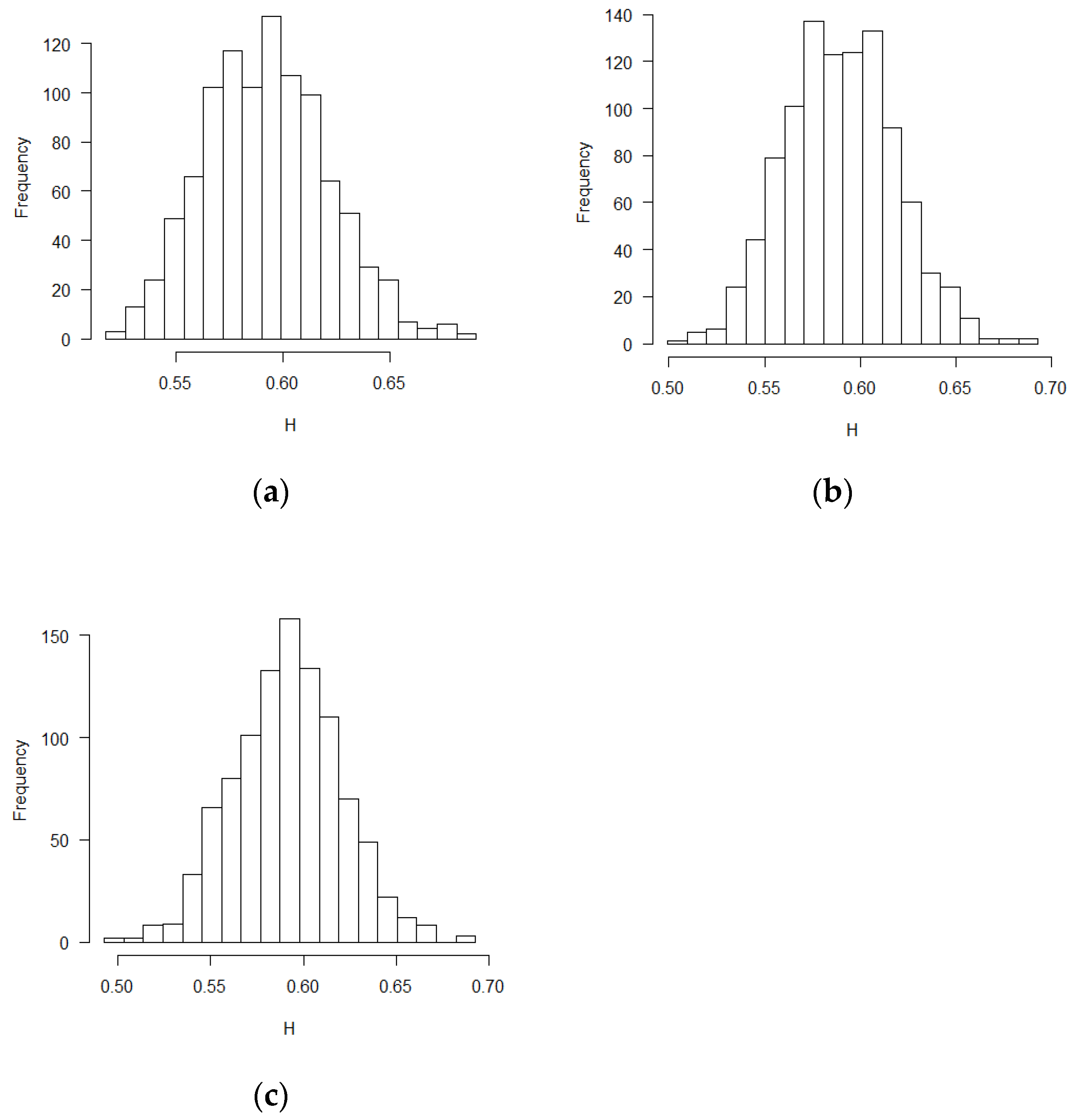
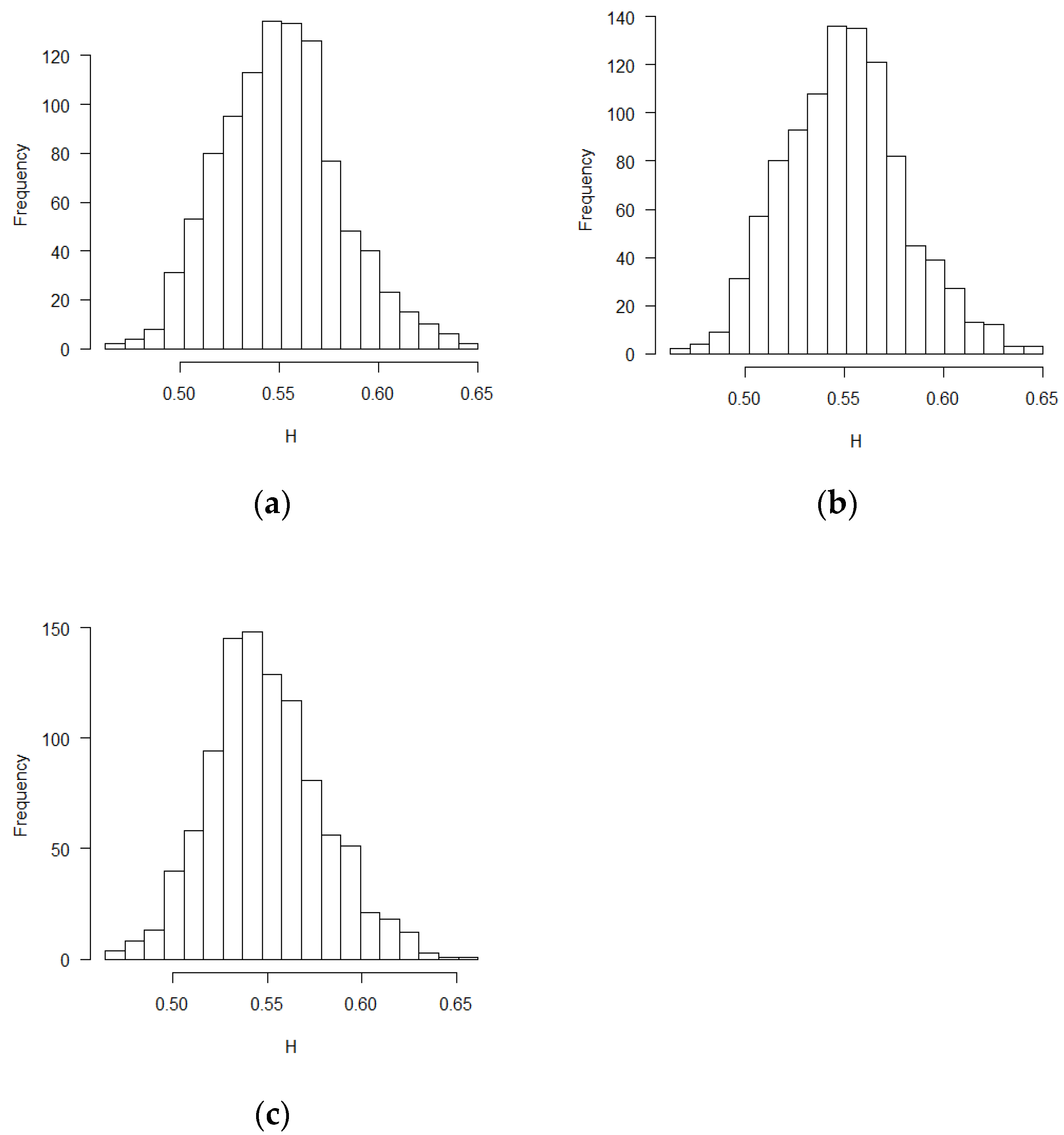
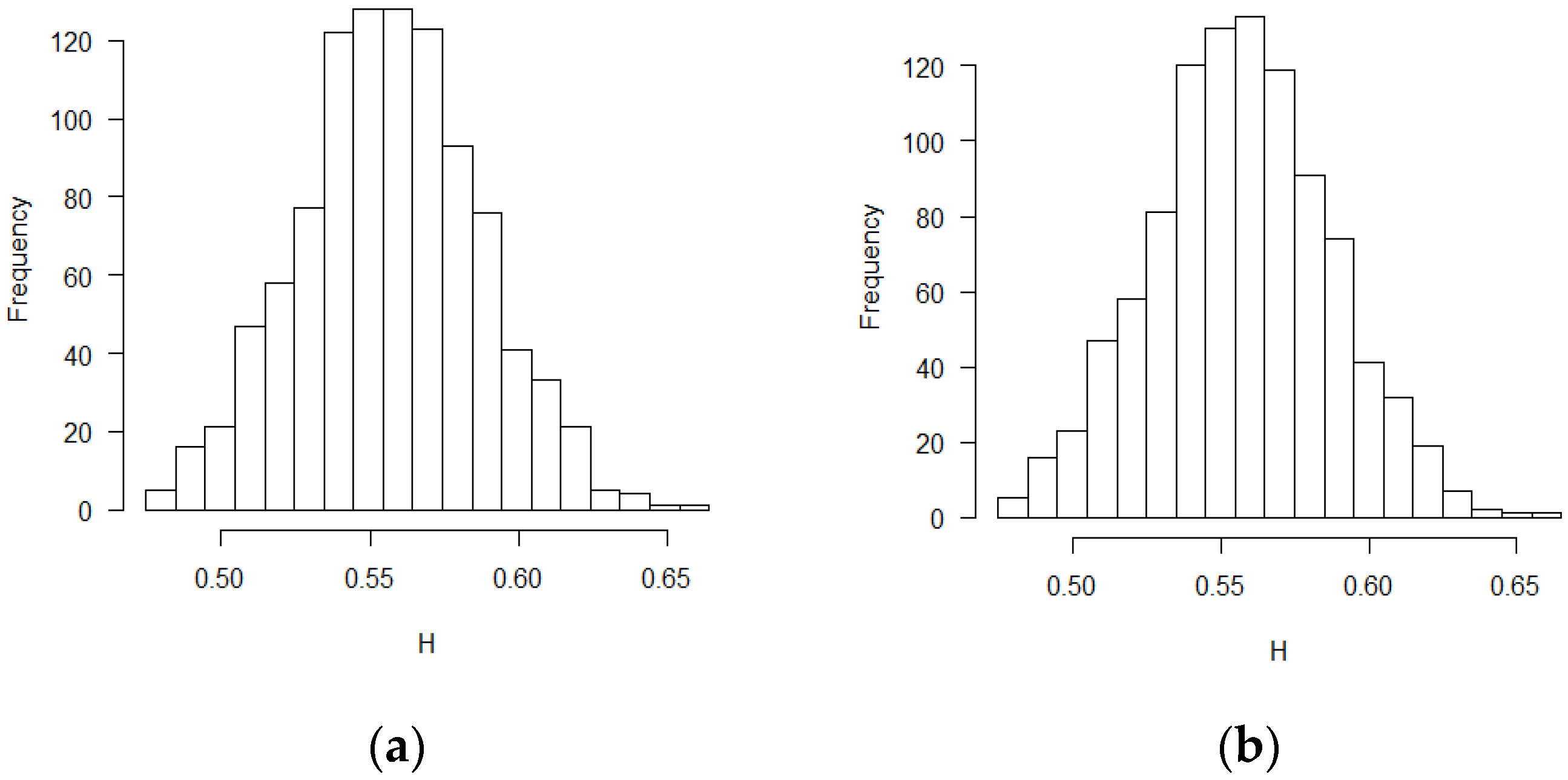
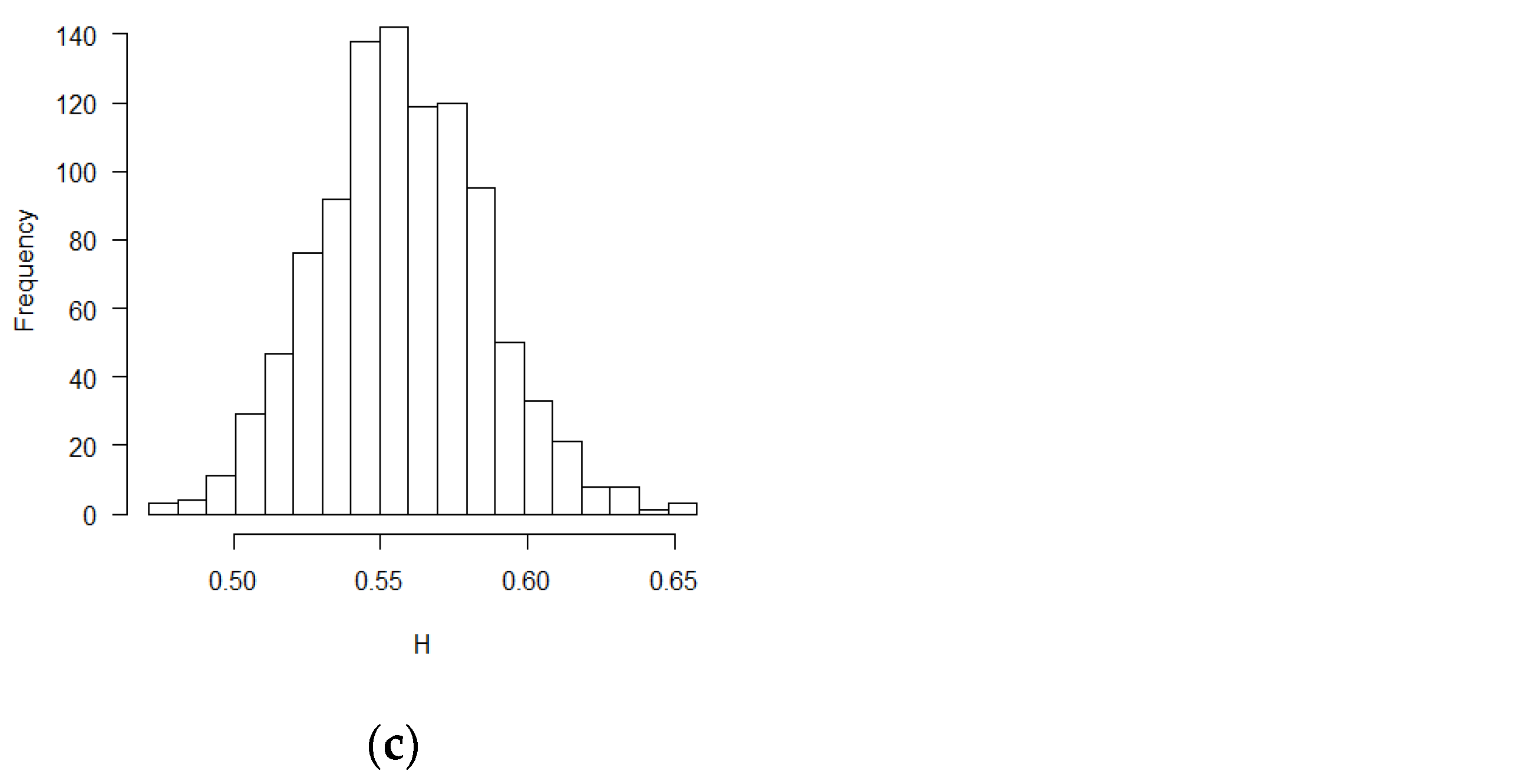
The mean and standard deviation of Hurst exponent estimations were tabulated in Table 1, in which another Hurst estimation method proposed by Peltler Lévy Véhel was used to validate the MLE results [30]. Two Hurst estimation methods reached similar results. The distribution of the estimates of H is close to normal distribution. The visual histograms are shown in Figure 1, Figure 2 and Figure 3. Mean estimated H values from the misspecification scenarios in Table 1 and Figure 1, Figure 2 and Figure 3 deviated from 0.5. All test of statistical significance test proved this conclusion with p value less than 0.0001 by t-test (Student’s t-Test) function in R software. According to the theoretical derivation results and simulation results, sequential test statistics do not follow a Brownian motion in the covariate adaptive randomized clinical trials sequential monitoring processes when error terms are correlated. Models with different covariate types reached similar conclusions.

Table 1.
Hurst exponent estimations for covariate adaptive randomized clinical trials, ε’s ~increments of FBM, (0.5, 0.5, 1, 1, 0.5, 0.5).
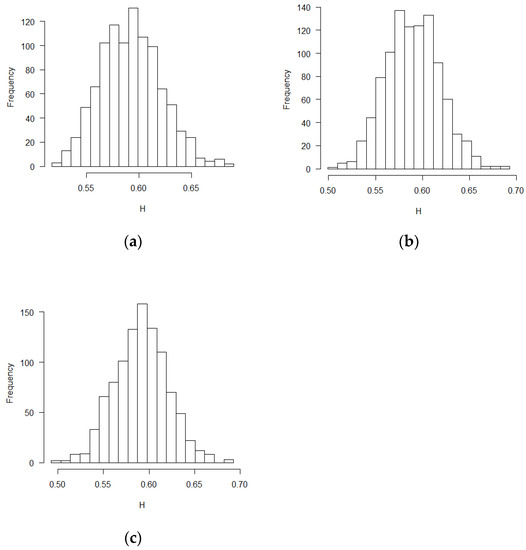
Figure 1.
Histograms for estimated H values (BR design, ε’s~ increments of FBM, MLE method): (a) no covariate, (b) two discrete covariates, (c) two continuous covariates.
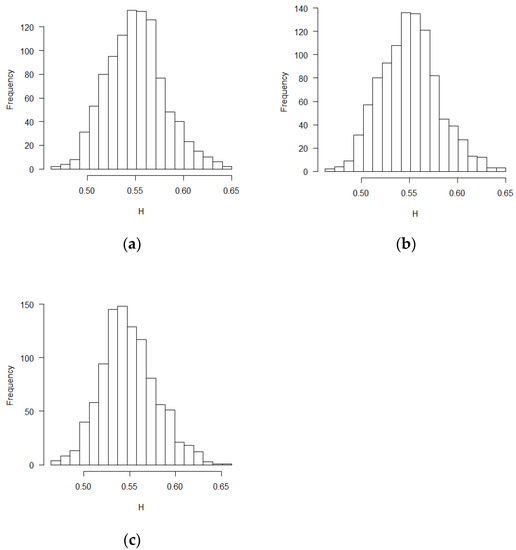
Figure 2.
Histograms for estimated H values (PS design, ε’s~ increments of FBM, MLE method): (a) no covariate, (b) two discrete covariates, (c) two continuous covariates.
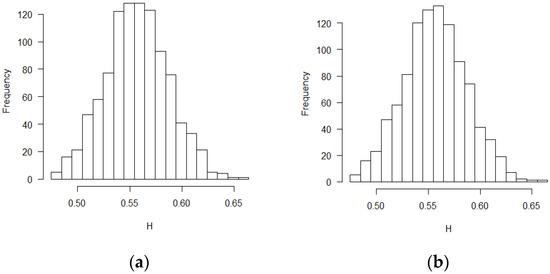
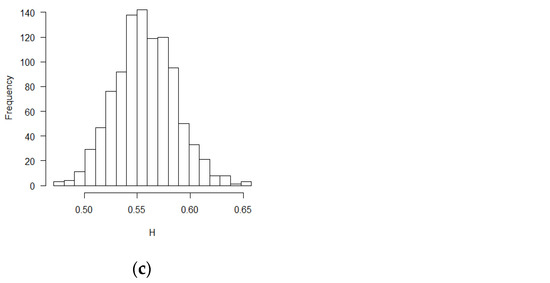
Figure 3.
Histograms for estimated H values (SPB design, ε’s~ increments of FBM, MLE method): (a) no covariate, (b) two discrete covariates, (c) two continuous covariates.
5. Conclusions and Discussions
In this study, we investigated the sequential monitoring properties in covariate adaptive randomized clinical trials under the misspecification scenarios. We also performed numerical simulations under various situations in which the mean estimates of Hurst coefficient by MLE from the sequential test statistics under misspecification scenarios deviated from 0.5. Brownian motion is satisfied only when . Therefore, the independent increment assumption was violated and Brownian motion was not appropriate for the sequential process. However, clinical researchers may not know the existence of the covariance in the error terms, and hence use the original classical statistic test under the misspecification scenarios, leading to non-Brownian motion trajectory of the test statistics under sequential analysis. Therefore, it is necessary to estimate the Hurst coefficient.
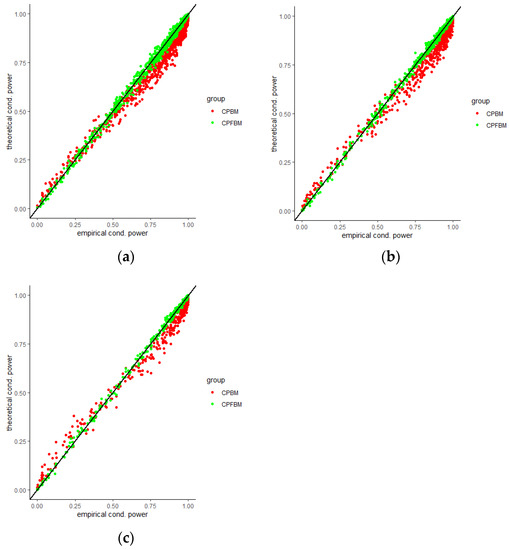
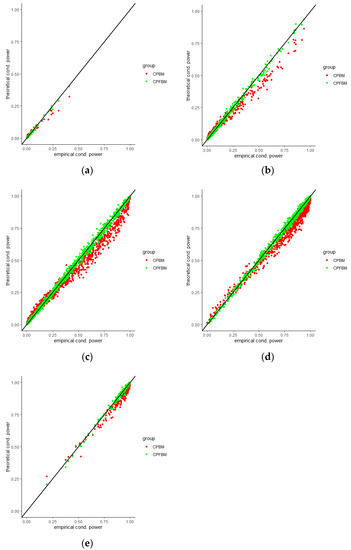
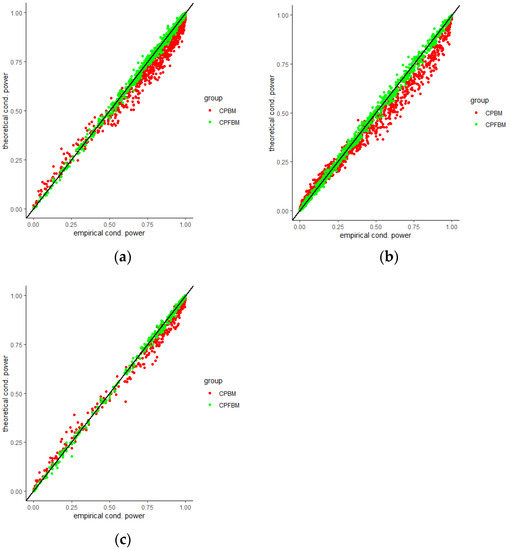
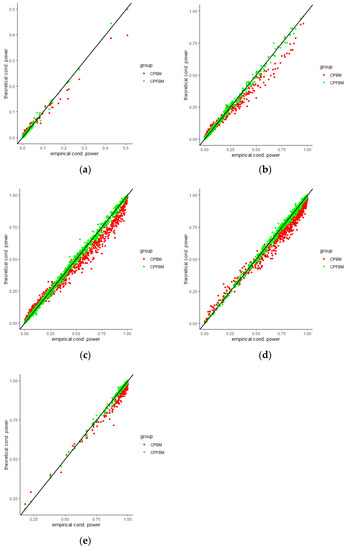
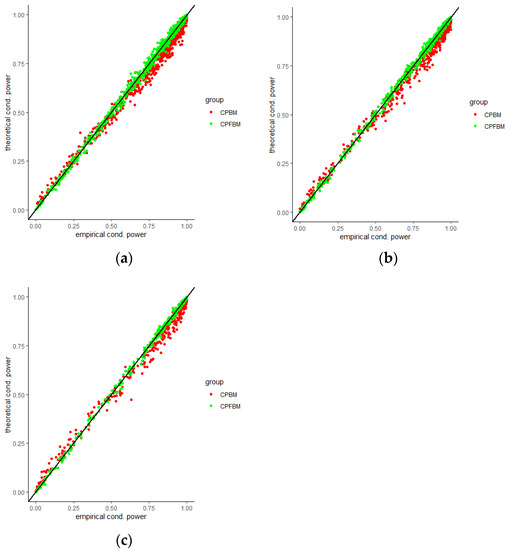
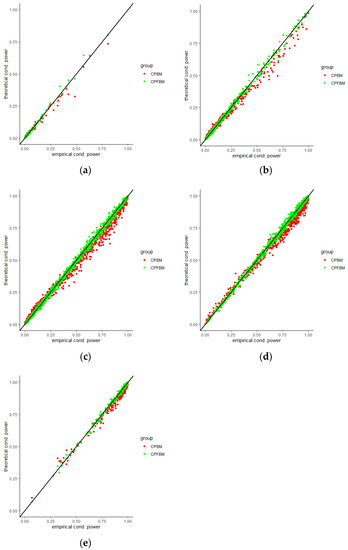
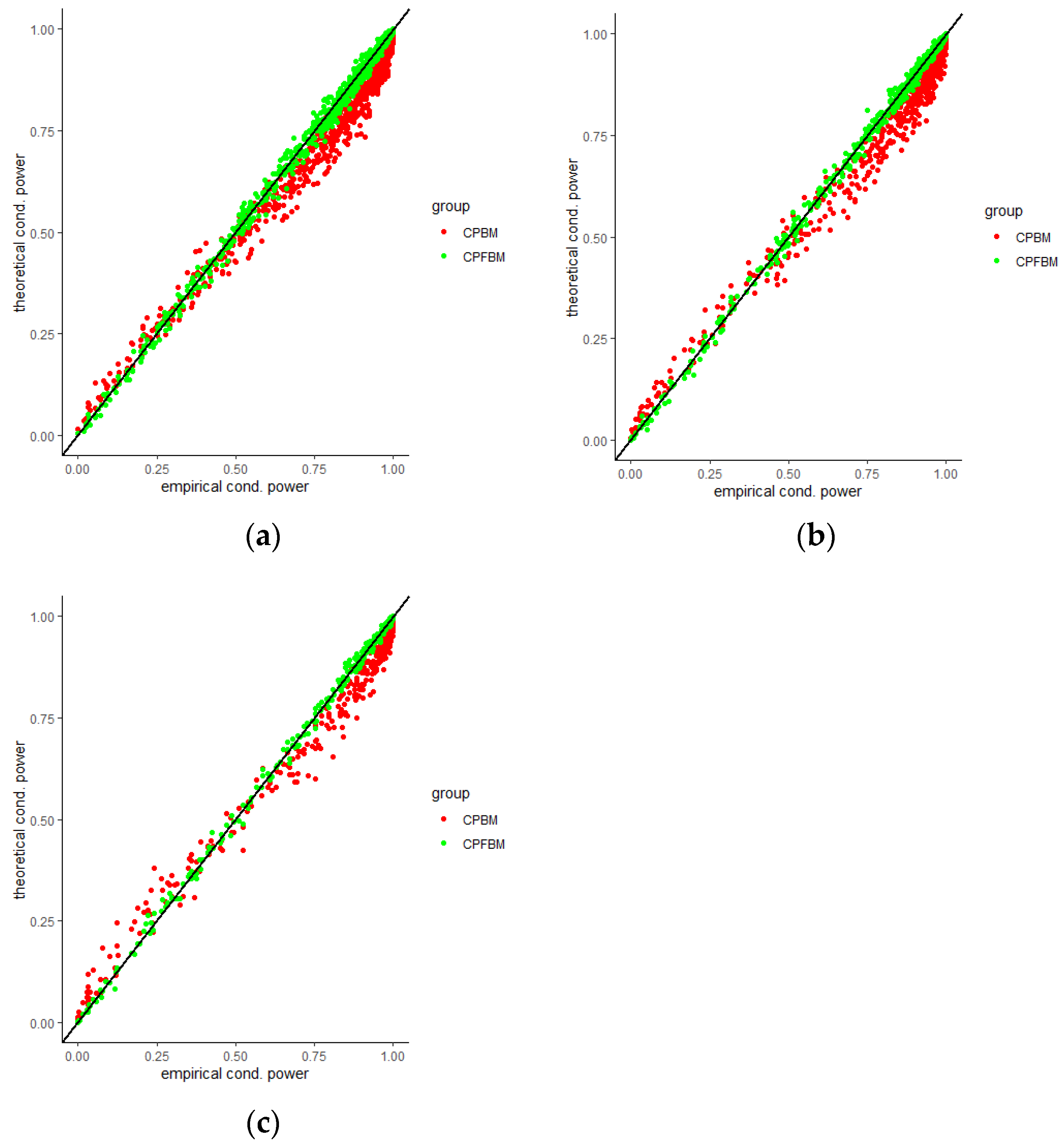
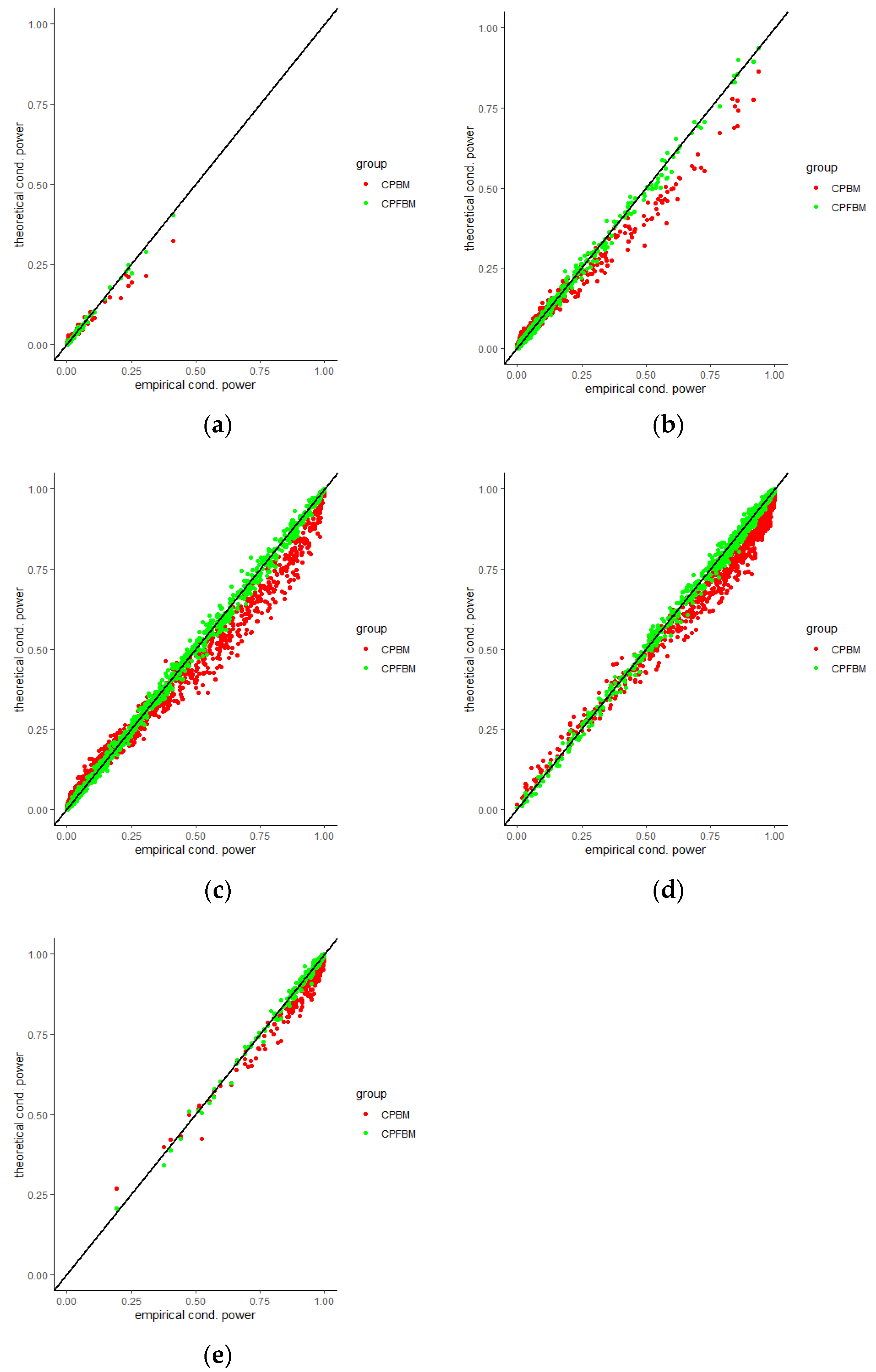
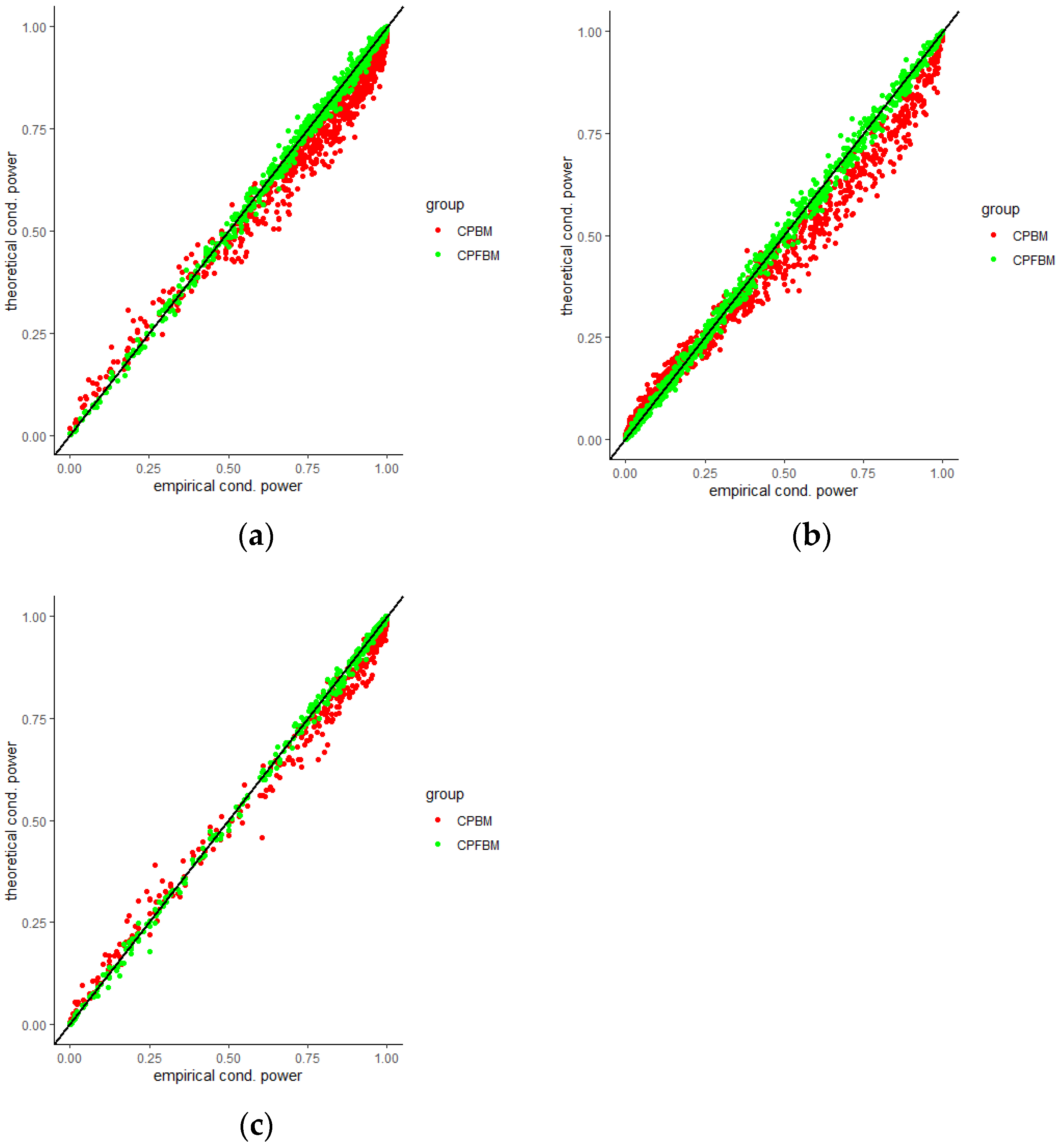
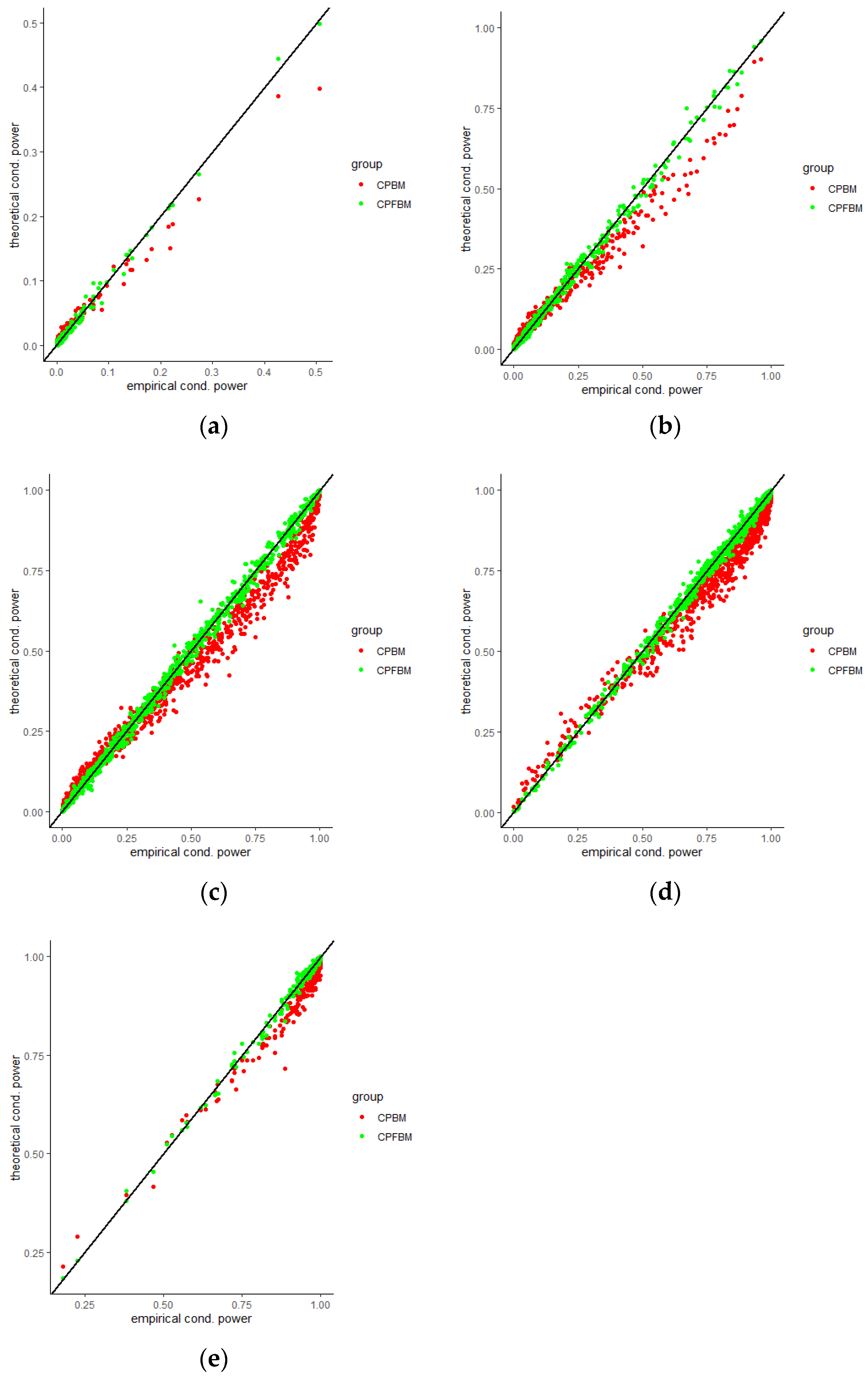
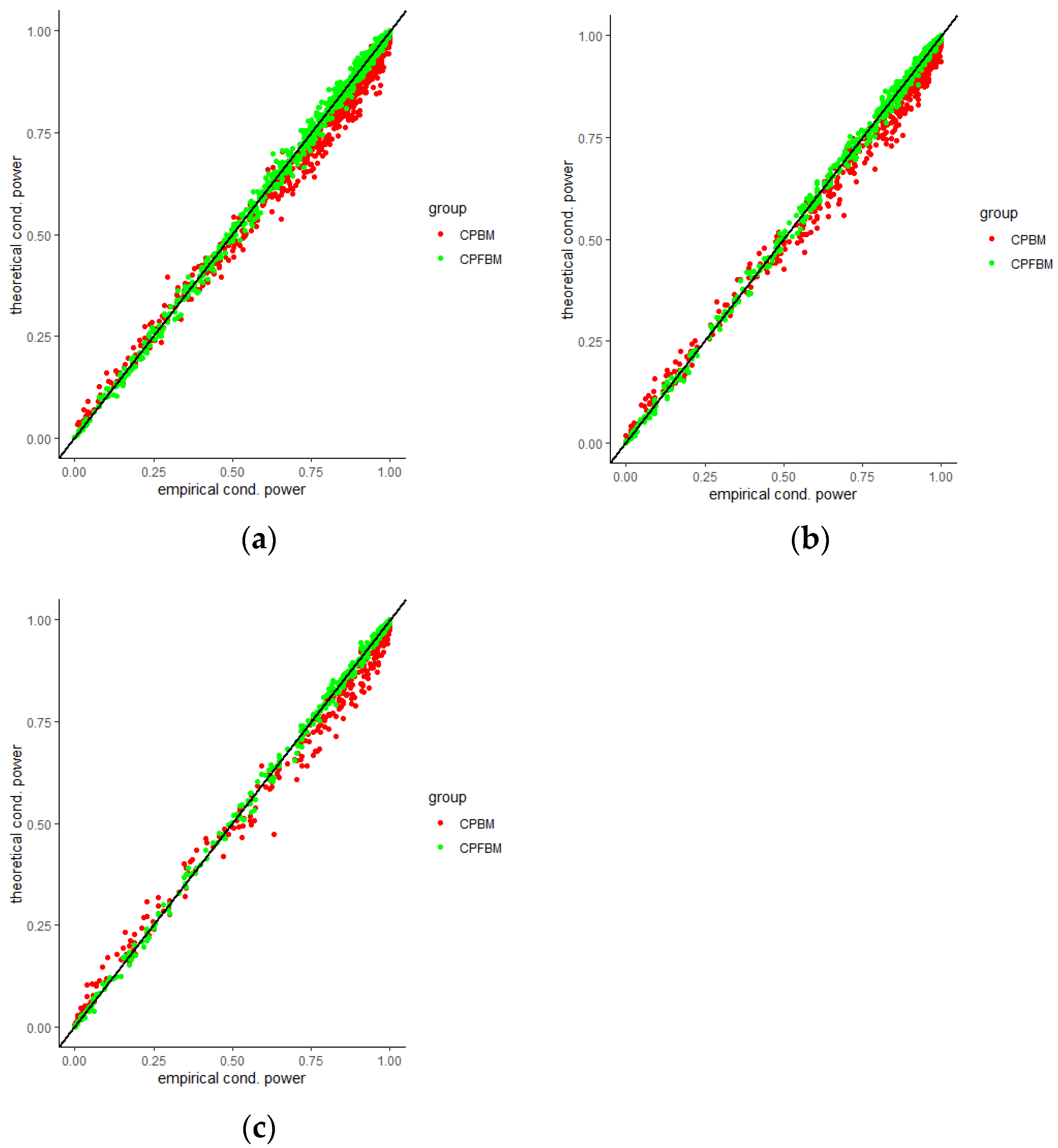
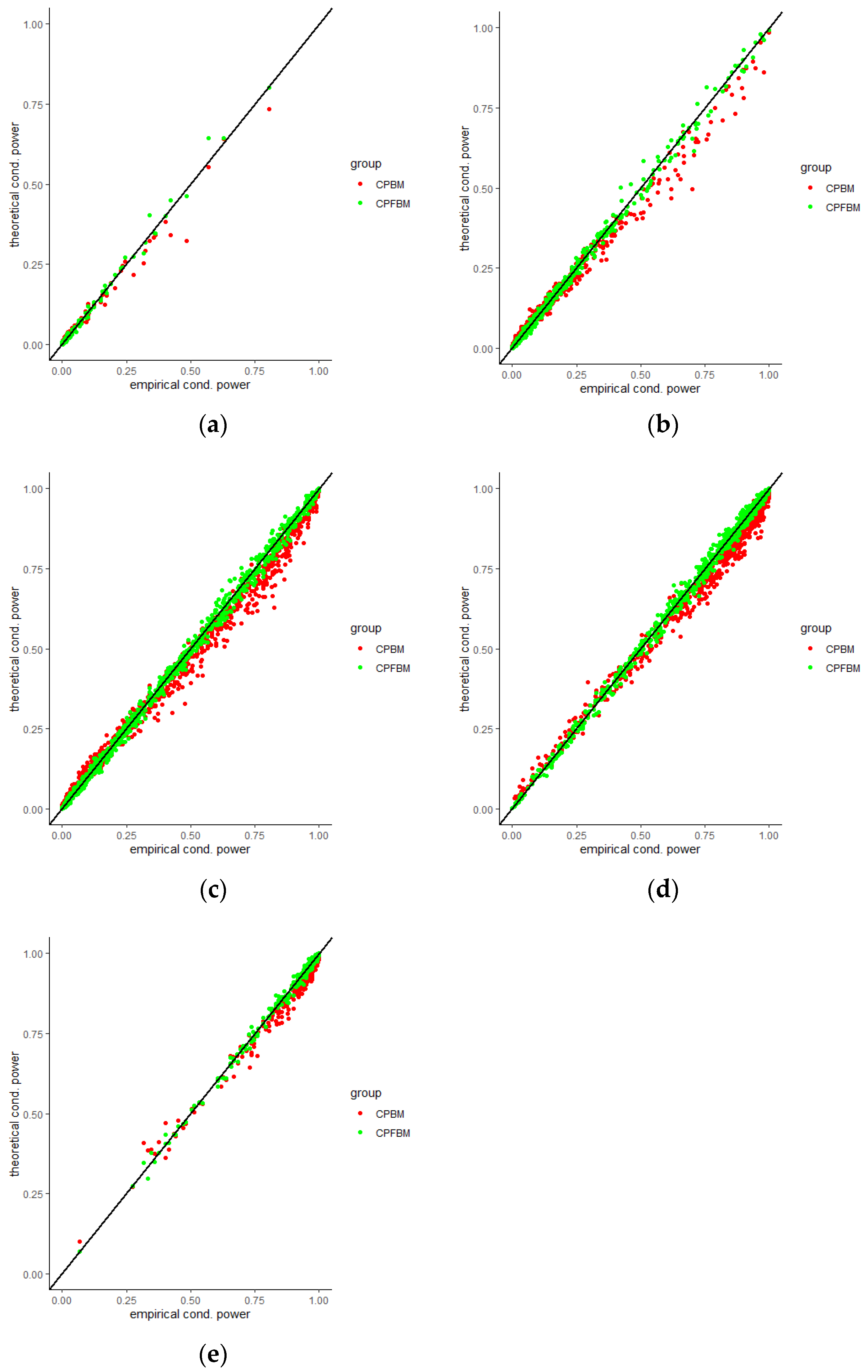
We calculated conditional powers for covariate adaptive randomized clinical trials with mis-specified error structures of the model under different covariate types, adaptive designs, drift parameters, and interim time points. Conditional powers based on the fractional Brownian motion (CP (FBM)) assumption resulted in better consistency with the standard empirical value (CP (empirical)) than conditional powers under the classical Brownian motion (CP (BM)) assumption. When the , most conditional powers under the FBM assumption were greater than the conditional powers under the classical Brownian motion assumption. The fractional Brownian motion, incorporating a dependent increment assumption, would be a reasonable approach for the clinical trial sequential analyses. Even if the sequential procedure actually follows the Brownian motion, the application of the fractional Brownian motion technique would still be useful, since BM is a special case of FBM with .
Author Contributions
Conceptualization, D.L. and H.Z.; methodology, D.L. and H.Z.; software, Y.Y.; validation, D.L., H.Z. and Y.Y.; formal analysis, Y.Y.; investigation, D.L., H.Z. and Y.Y.; resources, D.L.; data curation, Y.Y.; writing—original draft preparation, Y.Y.; writing—review and editing, D.L. and H.Z.; visualization, Y.Y.; supervision, D.L.; project administration, D.L.; funding acquisition, D.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially supported by Cancer Prevention Research Institute of Texas (RP170668).
Acknowledgments
This work was partially supported by Cancer Prevention Research Institute of Texas (RP170668).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chow, S.-C.; Liu, J.-P. Design and Analysis of Clinical Trials: Concepts and Methodologies; John Wiley & Sons: Hoboken, NJ, USA, 2008; Volume 507. [Google Scholar]
- Pallmann, P.; Bedding, A.W.; Choodari-Oskooei, B.; Dimairo, M.; Flight, L.; Hampson, L.V.; Holmes, J.; Mander, A.P.; Odondi, L.; Sydes, M.R.; et al. Adaptive designs in clinical trials: Why use them, and how to run and report them. BMC Med. 2018, 16, 1–15. [Google Scholar] [CrossRef]
- Hu, Y.Q.; Hu, F.F. Asymptotic Properties of Covariate-Adaptive Randomization. Ann. Stat. 2012, 40, 1794–1815. [Google Scholar] [CrossRef]
- Khan, O.; Fotheringham, S.; Wood, V.; Stimson, L.; Zhang, C.L.; Pezzella, F.; Duvic, M.; Kerr, D.J.; La Thangue, N.B. HR23B is a biomarker for tumor sensitivity to HDAC inhibitor-based therapy. Pro. Natl. Acad. Sci. USA 2010, 107, 6532–6537. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Lai, D.J. Sequential monitoring of covariate adaptive randomized clinical trials with sample size re-estimation. Contemp Clin. Trials 2019, 87, 105874. [Google Scholar] [CrossRef]
- Efron, B. Forcing a sequential experiment to be balanced. Biometrika 1971, 58, 403–417. [Google Scholar] [CrossRef]
- Ma, W.; Hu, F.F.; Zhang, L.X. Testing Hypotheses of Covariate-Adaptive Randomized Clinical Trials. J. Am. Stat. Assoc. 2015, 110, 669–680. [Google Scholar] [CrossRef]
- Bugni, F.A.; Canay, I.A.; Shaikh, A.M. Inference Under Covariate-Adaptive Randomization. J. Am. Stat. Assoc. 2018, 113, 1784–1796. [Google Scholar] [CrossRef]
- Ma, W.; Qin, Y.C.; Li, Y.; Hu, F.F. Statistical Inference for Covariate-Adaptive Randomization Procedures. J. Am. Stat. Assoc. 2020, 115, 1488–1497. [Google Scholar] [CrossRef]
- Lan, K.K.; Wittes, J. The B-value: A tool for monitoring data. Biometrics 1988, 44, 579–585. [Google Scholar] [CrossRef]
- Proschan, M.A.; Lan, K.G.; Wittes, J.T. Statistical Monitoring of Clinical Trials: A Unified Approach; Springer: New York, NY, USA, 2006. [Google Scholar]
- Zhu, H.J.; Hu, F.F. Sequential Monitoring of Covariate-Adaptive Randomized Clinical Trials. Stat. Sin. 2019, 29, 265–282. [Google Scholar]
- Abundo, M.; Pirozzi, E. On the Integral of the Fractional Brownian Motion and Some Pseudo-Fractional Gaussian Processes. Mathematics 2019, 7, 991. [Google Scholar] [CrossRef]
- Wang, P.G.; Xu, Y. Averaging Method for Neutral Stochastic Delay Differential Equations Driven by Fractional Brownian Motion. J. Funct. Space 2020, 2020, 5212690. [Google Scholar] [CrossRef]
- Yang, Y. Clinical trial designs and monitoring with fractional Brownian motion. Proquest Diss. Publ. 2020. [Google Scholar]
- Ferguson, T.S.; Klass, M.J. A representation of independent increment processes without Gaussian components. Ann. Math. Stat. 1972, 43, 1634–1643. [Google Scholar] [CrossRef]
- Hurst, H.E. Long-term storage capacity of reservoirs. Trans. Am. Soc. Civ. Eng. 1951, 116, 770–799. [Google Scholar] [CrossRef]
- Taqqu, M.S. Fractional Brownian motion and long-range dependence. In Theory and Applications of Long-Range Dependence; Doukhan, P., Oppenheim, G., Taqqu, M.S., Eds.; Birkhäuser: Boston, MA, USA, 2003. [Google Scholar]
- Yerlikaya-Ozkurt, F.; Vardar-Acar, C.; Yolcu-Okur, Y.; Weber, G.W. Estimation of the Hurst parameter for fractional Brownian motion using the CMARS method. J. Comput. Appl. Math. 2014, 259, 843–850. [Google Scholar] [CrossRef]
- Mandelbrot, B.B.; Van Ness, J.W. Fractional Brownian motions, fractional noises and applications. Siam Rev. 1968, 10, 422–437. [Google Scholar] [CrossRef]
- McCauley, J.L.; Gunaratne, G.H.; Bassler, K.E. Hurst exponents, Markov processes, and fractional Brownian motion. Phys. A 2007, 379, 1–9. [Google Scholar] [CrossRef]
- Lai, D.J. Estimating the Hurst effect and its application in monitoring clinical trials. Comput. Stat. Data 2004, 45, 549–562. [Google Scholar] [CrossRef]
- Jennison, C.; Turnbull, B.W. Group Sequential Methods with Applications to Clinical Trials; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Qian, H. Fractional Brownian motion and fractional Gaussian noise. In Processes with Long-Range Correlations; Springer: Berlin/Heidelberg, Germany, 2003; pp. 22–33. [Google Scholar]
- Huang, J. Somebm: Some Brownian Motions Simulation Functions. R Package Version 0.1. Available online: https://CRAN.R-project.org/package=somebm (accessed on 5 September 2021).
- Zelen, M. The randomization and stratification of patients to clinical trials. J. Chronic. Dis. 1974, 27, 365–375. [Google Scholar] [CrossRef]
- Pocock, S.J.; Simon, R. Sequential treatment assignment with balancing for prognostic factors in the controlled clinical trial. Biometrics 1975, 31, 103–115. [Google Scholar] [CrossRef]
- Ortigueira, M.D.; Batista, A.G. A fractional linear system view of the fractional Brownian motion. Nonlinear Dynam. 2004, 38, 295–303. [Google Scholar] [CrossRef][Green Version]
- Beran, J. Statistical methods for data with long range dependence. Stat. Sci. 1992, 7, 4404–4427. [Google Scholar]
- Peltier, R.F.; Lévy Véhel, J. A new method for estimating the parameter. Research Report. 1994. RR-2396, inria (inria-00074279). Available online: https://hal.inria.fr/inria-00074279 (accessed on 5 September 2021).
- Lachin, J.M. A review of methods for futility stopping based on conditional power. Stat. Med. 2005, 24, 2747–2764. [Google Scholar] [CrossRef] [PubMed]
- Lai, D.J.; Moye, L.A.; Davis, B.R.; Brown, L.E.; Sacks, F.M. Brownian motion and long-term clinical trial recruitment. J. Stat. Plan Infer. 2001, 93, 239–246. [Google Scholar] [CrossRef]
- Moyé, L.A. Statistical Monitoring of Clinical Trials: Fundamentals for Investigators; Springer: New York, NY, USA, 2006. [Google Scholar]
- Lai, D.J.; Davis, B.R.; Hardy, R.J. Fractional Brownian motion and clinical trials. J. Appl. Stat. 2000, 27, 103–108. [Google Scholar] [CrossRef]
- Huang, V.J.; Lai, D.J. Interim Analysis of Clinical Trials: Simulation Studies of Conditional Power under Fractional Brownian Motion. Fractals 2019, 27, 1950133. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).