Schiff bases are the most extensively used versatile ligands, capable of coordinating several chemical elements and stabilizing them in numerous oxidation states. Coordination complexes of Cu(II) with Schiff base ligands have been extensively studied due to their properties, structural aspects, and potential biological activity [1]. Three new copper Schiff base complexes have been synthesized: a 1-D copper coordination polymer, [Cu2(L)2(μ-ClO4)]n, 1, a mononuclear copper complex, [Cu(L)]ClO4, 2, and a 1-D copper-tin coordination polymer, [Cu2(L)(μ4-tma)Ph3SnCl]n, 3, where L = 4-Chloro-6(N-2-picolyl-1′-iminomethyl)phenol and tma = benzene-1,3,5-tricarboxylate. The newly obtained compounds were synthesized and then characterized by elemental analysis, IR and UV-Vis spectral studies. Their crystal structures were determined by single-crystal X-ray diffraction.
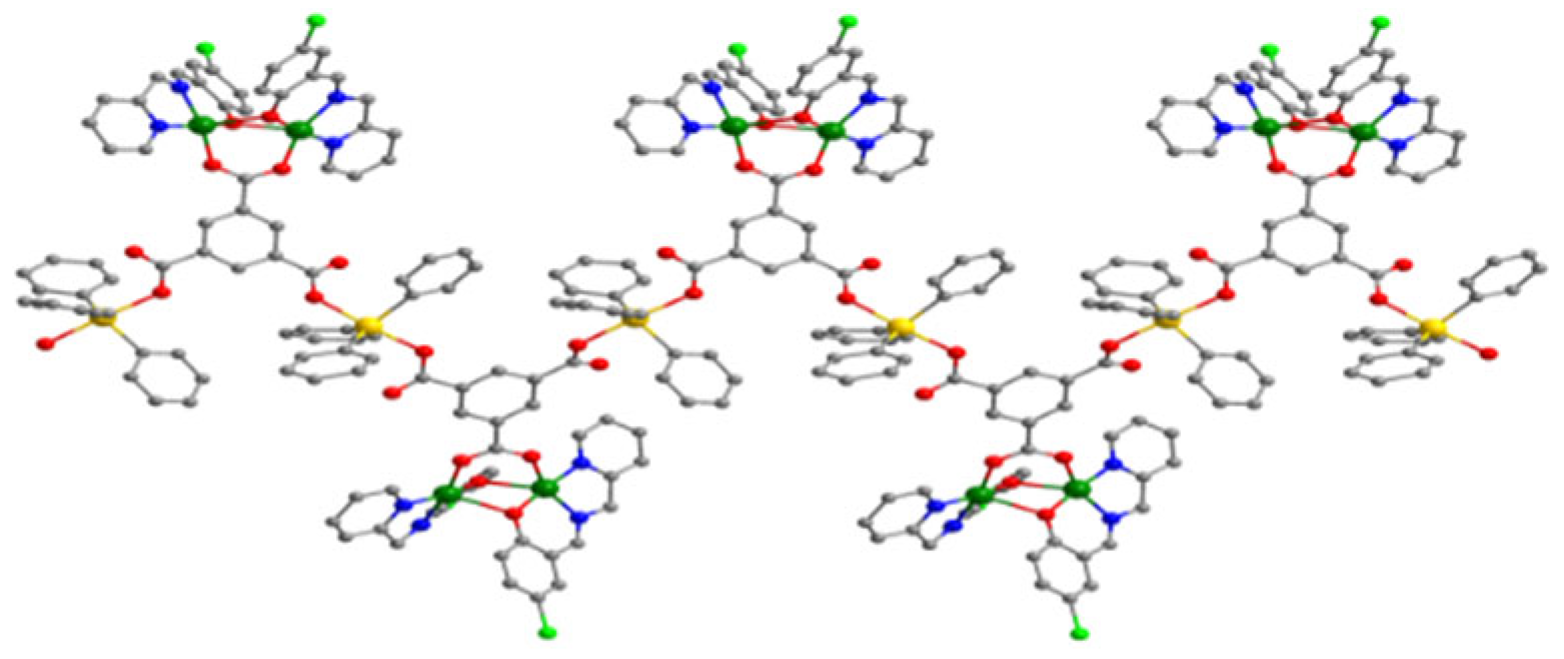
Compound 1 is a coordination polymer bridged with a bidentate perchlorate ligand and it was obtained from Cu(ClO4)2·6H2O and the condensation product of 5-chlorosalicylaldehyde and 2-picolylamine. The copper atom is hexacoordinated: Four of the coordination sites are occupied by the three donor atoms of the Schiff base ligand and one oxygen atom from a water molecule that have a square-planar arrangemet and the apical sites are occupied by two perchlorato ligands. Complex 2 was obtained from the first one, overtime. The experimental research has shown that after a few days, in the absence of any interfering factors, the polymer crystals change their shape turning into a mononuclear copper complex where the geometry around the copper(II) ion is square pyramidal. Compound 3 was also prepared by the reaction of 5-chlorosalicylaldehyde and 2-picolylamine in the presence of a copper salt—Cu(ClO4)2·6H2O, Ph3SnCl and deprotonated trimesic acid. From the stereochemical point of view (Figure 1), two copper atoms are triply bridged by one of the trimesic acid carboxylate group and by the phenolic oxygen atoms of two tridentate Schiff base ligands. Each copper(II) atom is five-coordinated with a square pyramidal geometry. Tin(IV) atoms are pentacoordinated having a trigonal bipyramid geometry.
Figure 1.
Structure of compound 3.
In conclusion, three new coordination compounds containing Schiff base ligands have been synthesized and characterized.
References
- Zoubi, W.A. Biological Activities of Schiff Bases and Their Complexes: A Review of Recent Works. Int. J. Org. Chem. 2013, 73, 95. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).