Abstract
Methane (CH4) emissions are a leading cause of global warming, and precise monitoring of and reduction in these emissions are important. To achieve these goals, miniaturized low-power sensor systems with improved precision are necessary. To this end, we present a novel room-temperature chemiresistive CH4 gas sensor that employs Zn-hexahydroxytriphenylene-based metal–organic frameworks (Zn-HHTP MOFs) as detection materials. The high surface area and porosity of Zn-HHTP MOFs enable effective detection of low atmospheric levels (1.2 ppm) of CH4.
1. Introduction
CH4 is a potent greenhouse gas, and detecting low concentrations of odorless CH4 is challenging due to its non-polarity and high enthalpy of C–H bonds [1]. Therefore, developing miniaturized low-cost and sensitive gas sensors remains an active research goal. Although electrochemical, infrared, and chemiresistive are the most common gas detection technologies, chemiresistive sensors are advantageous thanks to their low cost, simple operation, high sensitivity, and lifetime [2]. Despite intensive research on advanced materials, metal oxide-based chemiresistive sensors, which are used most, still face major challenges related to poor selectivity and high working temperature [2]. Replacing traditional metal oxides with advanced conductive MOFs as detection materials could address these challenges. MOFs, made up of metal nodes and organic linkers, have a rigid cage-like structure with a high surface area, porosity, and crystallinity [3]. These properties make them excellent for gas detection, particularly for chemiresistive sensors that rely on surface reactions. Therefore, the primary goal of this study is to detect low levels of CH4 by utilizing advanced MOFs as the detection material. In this respect, Zn-HHTP MOFs were synthesized by coordinating a prominent group of triphenylene with Zn2+ ions using a simple solvothermal method, and they were employed for the first time as active sensing material to detect low levels of CH4 at room temperature.
2. Materials and Methods
To synthesize Zn-HHTP MOFs, a mixture of HHTP-ligand and Zn-acetate in distilled water (DW) was sonicated for 10 mins, and then dimethylformamide was added and sonicated for another 15 min. After incubating at 80 °C for 6 h, the powder was collected by centrifugation, washed with DW and ethanol, and dried. The chemical structure of the Zn-HHTP MOFs is shown in Figure 1a. For sensor fabrication, 1 mg of Zn-HHTP MOF powder was dispersed into 1 mL of distilled water and sonicated for 30 min. Later, the suspension was deposited on the interdigited chips with Au electrodes and dried in air prior to being used as a detection element.
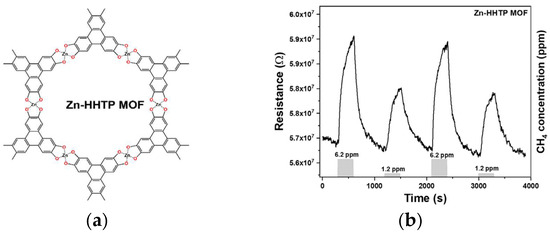
Figure 1.
(a) Chemical structure of Zn-HHTP MOF; (b) chemiresistive study of Zn-HHTP MOF sensor to 6.2 and 1.2 ppm of CH4 at room temperature, proving its reversibility.
3. Discussion
Room-temperature chemiresistive gas sensing measurements on Zn-HHTP MOFs were performed regarding various CH4 concentrations via a dynamic gas sensing setup under laboratory conditions. A sensor response was calculated as the ratio between the resistance of Zn-HHTP MOF in air (Ra) to the resistance of Zn-HHTP MOF in CH4 gas (Rg), i.e., response (%) = [|Ra − Rg|/Ra] × 100. Before CH4 exposure, the Zn-HHTP MOF sensor was stabilized by aging in dry synthetic air for 3 hours. Figure 1b shows the reversibility plot of the Zn-HHTP sensor to successive 6.2 and 1.2 ppm of CH4 at room temperature, where the sensor electrical resistance values were found to be increased upon the interaction of CH4 gas with complete recovery features. Notably, the Zn-HHTP sensor is capable of effectively detecting a very low CH4 concentration down to 1.2 ppm (response = 2.5%), which is lower than the atmospheric CH4 concentration (1.9 ppm) specified by the Global Monitoring Laboratory for the year 2022 [4]. The observed response is mainly due to the MOF’s high surface area, porosity, and crystallinity, enabling effective surface reactions with CH4 molecules. Furthermore, the sensor’s affinity for CH4 may have been enhanced by the coordination of triphenylene with Zn2+ ions. Overall, based on our findings, Zn-HHTP MOFs show promise as advanced sensing materials for room-temperature chemiresistive CH4 sensors. Our current focus is to study the influence of interfering gases, such as water vapor, on the sensor response, considering its practical applications.
Author Contributions
Conceptualization, S.N. and I.F.-G.; methodology, S.N. and I.F.-G.; validation, S.N., I.F.-G. and A.R.-R.; investigation, S.N., I.F.-G. and Y.M.; data curation, S.N. and I.F.-G.; writing—original draft preparation, S.N.; writing—review and editing, D.S., A.V.-F. and A.R.-R.; visualization, I.F.-G., P.P. and M.M.; supervision, A.R.-R.; project administration, S.N. and A.R.-R.; funding acquisition, S.N. and A.R.-R. All authors have read and agreed to the published version of the manuscript.
Funding
This research has received funding from the European Union’s Horizon 2020 research and innovation programme under Marie Sklodowska-Curie grant agreement No. 101030015 and from the Spanish Ministerio de Economía e Innovación through project PID2019-107697RB C41/AEI/10.13039/501100011033.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bezdeka, M.; Luo, S.; Ku, K.; Swager, T. A chemiresistive methane sensor. Proc. Natl. Acad. Sci. USA 2021, 118, e2022515118. [Google Scholar] [CrossRef] [PubMed]
- Dey, A. Semiconductor metal oxide gas sensors: A review. Mater. Sci. Eng. B 2018, 229, 206–217. [Google Scholar] [CrossRef]
- Koo, W.; Jang, J.; Kim, L. Metal-organic frameworks for chemiresistive sensors. Chem 2019, 5, 1938–1963. [Google Scholar] [CrossRef]
- Lan, X.; Thoning, K.W.; Dlugokencky, E.J. Trends in globally-averaged CH4, N2O, and SF6 determined from NOAA Global Monitoring Laboratory measurements. Version 2024-03. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).