Abstract
Bioisosteres of amide bonds such as 4,5-disubstituted-1,2,3-triazoles (4,5-DS-1,2,3-Ts) and 1,5-disubstituted tetrazoles (1,5-DST) are present in compounds with important biological activities like those that are antineoplastic, antibacterial, antifungal, and antiparasitic, and antifungal, antiparasitic, antiviral, and anti-inflammatory. In the present work, we describe the synthesis of tetrazole–triazole bis-heterocycles via the Ugi–Azide strategy. The target molecules were synthesized with moderate yields, under mild conditions, employing 2H-1,2,3-triazole aldehyde as an input.
1. Introduction
Multicomponent reactions (MCRs) have proven to be efficient synthetic tools compared to traditional multistep syntheses. They are defined as one-pot processes where three or more reagents interact together under the same reaction conditions [1]. MCRs are considered domino reactions [2].
MCR products are complex and exhibit great molecular diversity, allowing for the generation of libraries of compounds with importance for several fields such as optics, agrochemistry, and medicinal chemistry, among others [3]. Heterocyclic chemistry is an important topic in the MCR field, where heterocycles can be either synthesized via a multicomponent process or via MCR-post transformation or functionalized via MCRs [4].
Among MCRs, those based on isocyanide chemistry (IMCRs) are among the more important and widely used due to the versatility of isocyanide to react as a nucleophile and electrophile at the same carbon [5]. The Ugi reaction is one of the well-known IMCRs [6]. However, several variations have been reported, for example, Ugi–Azide, where carboxylic acid is replaced with hydrazoic acid. This reaction is the method of choice for 1,5-disubstituted tetrazole synthesis [7].
Tetrazoles are heterocyclic compounds formed by four nitrogen atoms with potential applications in medicine, agriculture, chemistry, and pharmacology, among others [8]. In medicinal chemistry, two types of tetrazoles are highlighted: 5-substituted tetrazole and 1,5-disubstituted tetrazole, the latter being considered as a bioisostere of amide bonds. This property is associated with bond angles and lengths, and it is beneficial as it improves metabolic resistance to peptidases [9].
On the other hand, 1,2,3-triazoles are five-membered heterocycles with three nitrogen atoms. In recent years, they have attracted interest from several fields due to their antineoplastic, antibacterial, antifungal, antiviral, and antiparasitic potential [10]. From the medicinal chemistry point of view, 1,4 and 1,5-disubstituted-1,2,3-triazoles are capable of mimicking trans and cis amide bonds, respectively, which provides a high metabolic resistance [11].
Bis-heterocycles are hybrid molecular systems where two heterocycles are present [12]. Their connectivity can be either linked, spaced, bound, fused, or merged [13]. Among the plethora of these kind of compounds, those which incorporate nitrogen heterocycles are highlighted because around 59% of the FDA-approved drugs until 2014 contained at least one nitrogen heterocycle [14].
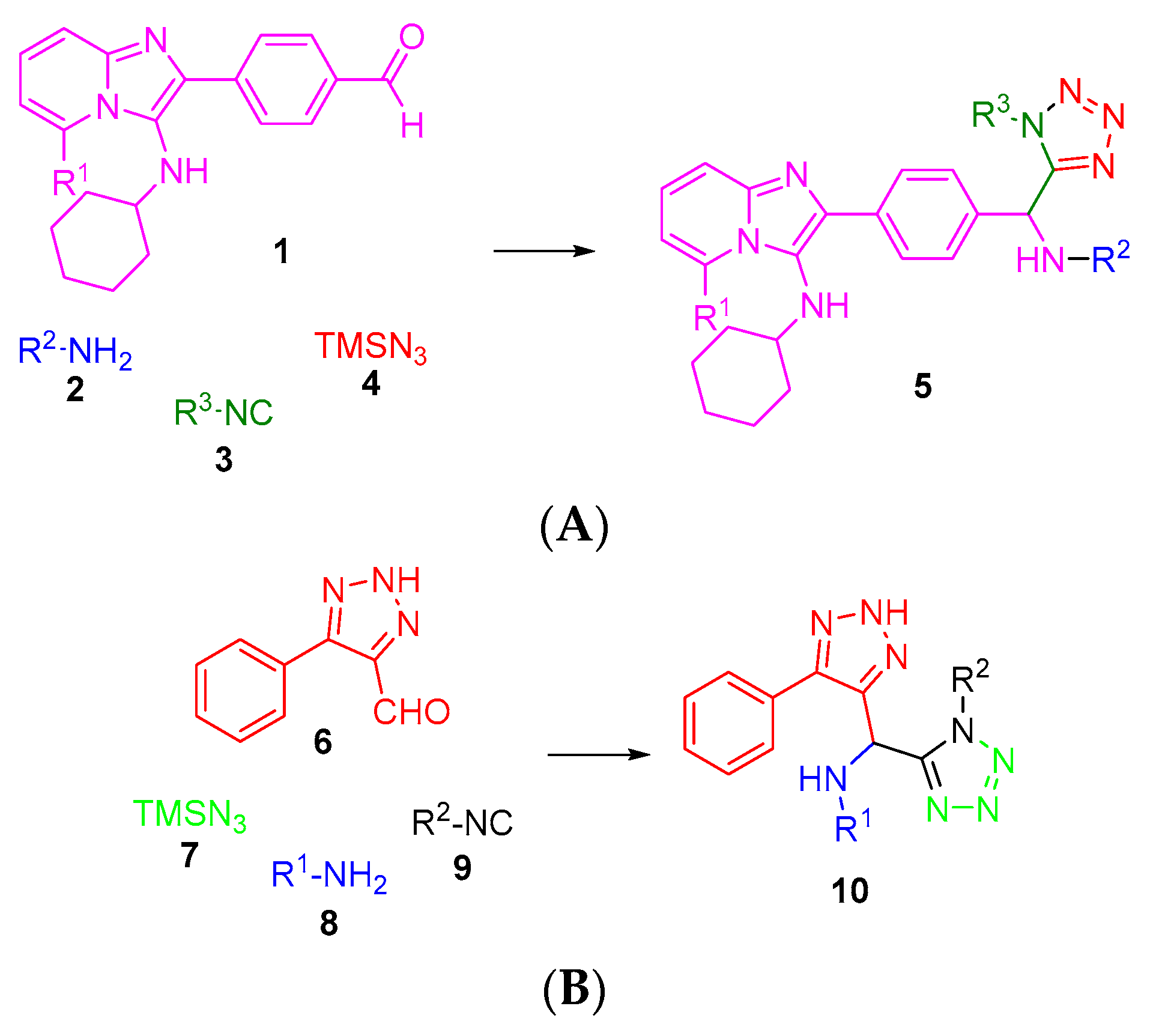
The incorporation of a heterocycle as part of the components in an IMCRs process such as the Ugi–Azide reaction is an important alternative to the construction of bis-heterocycles with linked connectivity, like 1,5-disubstituted-tetrazole-1,5-disubstituted-triazole (Figure 1).
Figure 1.
Synthesis of complex tetrazoles linked to other heterocycles. (A) Closer look at the work by Sharisa, A. et al. (2013) [15]; (B) This work.
2. Results and Discussion
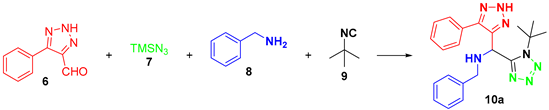
In the present work, the synthesis of a small library of bis-heterocycles containing the 1,5-disubstituted 1,2,3-triazole and 1,5-disubstituted tetrazole moieties via the Ugi–Azide reaction is presented. For the reaction optimization, 5-phenyl-2H-1,2,3-triazole-4-carbaldehyde (6), trimethylsilylazide (7), benzylamine (8a), and tert-butyl isocyanide (9a) were chosen as components, to synthesize bis-heterocycle 10a. The optimization experiments are described in Table 1.

Table 1.
Screening conditions for the synthesis of target molecule 16a.
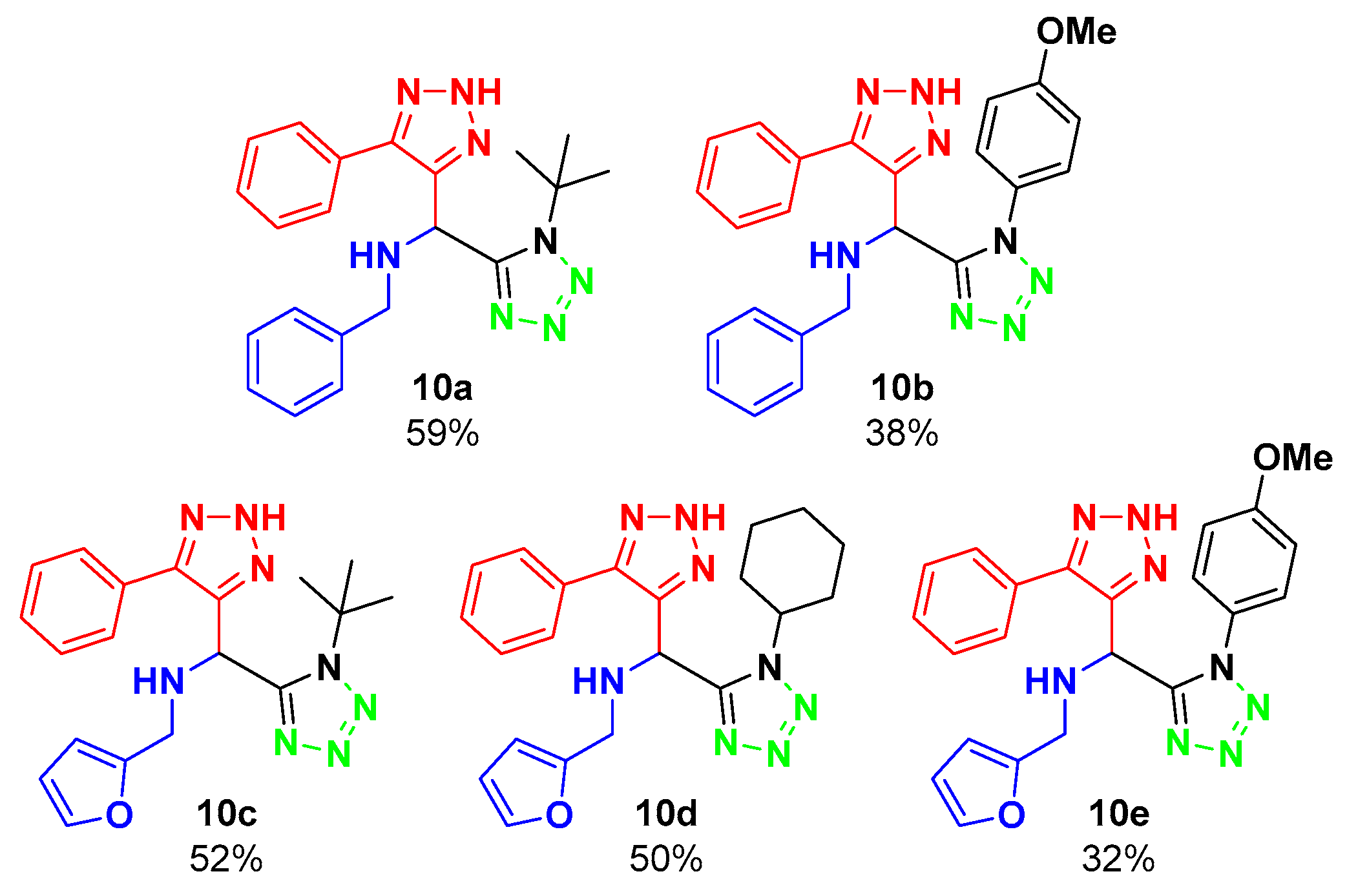
In the first experiment, ethanol was used as a solvent for the Ugi–Azide reaction for 24 h, at room temperature, obtaining a moderate yield of 59%. Further optimization of the reaction was attempted by using water as a solvent, and with a solvent-free experiment; however, neither of the two reactions proceeded, as the starting materials were recovered. Taking the experiment with ethanol as the optimized condition, the reaction scope was evaluated, varying the amine and isocyanide components (Figure 2).
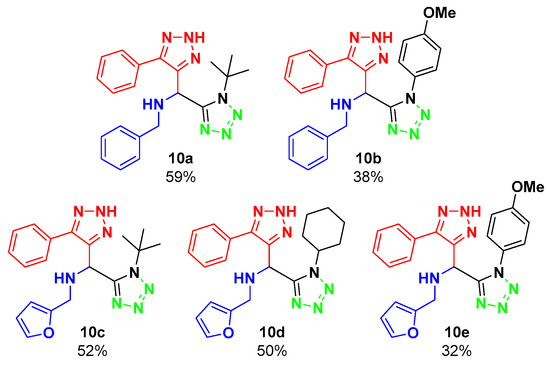
Figure 2.
Substrate scope.
3. Conclusions
The incorporation of a nitrogen heterocycle in one of the components in an IMCR is an efficient strategy for the functionalization of heterocycles. It is highlighted that the target molecules incorporate bis-heterocycles in their structure. The developed strategy is the first report on the use of 5-phenyl-2H-1,2,3-triazole-4-carbaldehyde as a component in the Ugi–Azide reaction. The developed procedure has advantages such as being carried out under mild and environmentally friendly reaction conditions, using a green solvent.
4. Experimental Section
4.1. General information, Chemicals, and Instrumentation
Bruker Avance III spectrometers (500 and 125 MHz, respectively) were used for acquisition of 1H and 13C NMR spectra. Deuterated chloroform (CDCl3) was used as the solvent for NMR experiments. Chemical shifts (δ) are given in ppm relative to tetramethylsilane (TMS). Coupling constants are reported in Hertz (Hz). Multiplicities of the signals are described using standard abbreviations: singlet (s), doublet (d), triplet (t), quartet (q), and multiplet (m). NMR spectra were analyzed using MestReNova software version 12.0.0-20080. Reaction progress was monitored by thin-layer chromatography (TLC) on pre-coated silica gel F254 aluminum sheets. The spots were visualized under UV light at 254 nm. Column chromatography was performed using silica gel (230–400 mesh) as a stationary phase. Mixtures of hexanes and ethyl acetate were used as mobile phases for column chromatography and in TLC for reaction progress monitoring. All reagents were purchased from Sigma Aldrich and were used without further purification. Chemical names and drawings were obtained using the ChemDraw 22.2.0.3300 software package.
4.2. General Procedure
In a sealed vial, 5-phenyl-2H-1,2,3-triazole-4-carbaldehyde (6, 1.0 equiv.), trimethylsilylazide (7, 1.0 equiv.), amine (8a,b, 1.0 equiv.), and isocyanide (9a–c, 1.0 equiv.) were dissolved in EtOH (0.5 M) and stirred for 24 h at room temperature. The solvent was evaporated under reduced pressure and the product was purified by flash chromatography using mixtures of ethylacetate in hexanes as mobile phase and silica gel as stationary phase to obtain the corresponding bis-heterocycles 10a–e.
4.3. Spectral Data
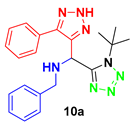
N-benzyl-1-(1-(tert-butyl)-1H-tetrazol-5-yl)-1-(5-phenyl-2H-1,2,3-triazol-4-yl)-methanamine (10a): Yellow solid; 1H (500 MHz, CDCl3) δ 7.58 (m, 2H), 7.35 (m, 3H), 7.27 (m, 3H), 7.21 (m, 3H), 5.53 (s, 1H), 3.86 (d, J = 12.7 Hz, 1H), 3.75 (d, J = 12.7 Hz, 1H), 1.44 (s, 9H).; 13C NMR (125 MHz, CDCl3) δ 154.55, 144.12, 141.20, 138.30, 129.62, 129.91, 128.85, 128.80, 128.43, 128.01, 127.55, 61.98, 51.63, 49.33, 29.60.
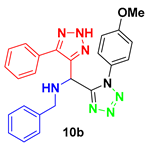
N-benzyl-1-(1-(4-methoxyphenyl)-1H-tetrazol-5-yl)-1-(5-phenyl-2H-1,2,3-triazol-4-yl)-methanamine (10b). Yellow solid: 1H NMR (400 MHz, CDCl3, 25 °C, TMS): δ 7.22 (m, 7H), 7.09 (m, 3H), 6.97 (d, J = 6.9 Hz, 2H), 6.69 (d, J = 6.9 Hz, 2H), 5.34 (s, 1H), 3.79 (d, J = 4.8 Hz, 2H), 3.77 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 160.83, 155.23, 138.10, 129.12, 128.75, 128.66, 128.57, 128.44, 127.78, 127.41, 126.28, 125.56, 114.60, 55.55, 51.23, 47.02.
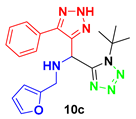
1-(1-(tert-butyl)-1H-tetrazol-5-yl)-N-(furan-2-yl-methyl)-1-(5-phenyl-2H-1,2,3-triazol-4-yl)-methanamine (10c): brown solid; 1H NMR (400 MHz, CD3OD) 7.65 (m, 2H), 7.50 (m, 1H), 7.44 (m, 3H), 6.37 (m, 1H), 6.19 (d, J = 3.1 Hz, 1H), 5.61 (s, 1H), 3.98 (d, J = 14.3 Hz, 1H), 3.83 (d, J = 14.4 Hz, 1H), 1.38 (s, 9H); 13C NMR (100 MHz, CD3OD) δ 155.76, 153.74, 143.87, 130.13, 130.01, 129.03, 111.46, 109.78, 63.36, 50.20, 44.27, 29.69.
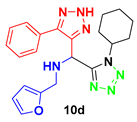
1-(1-cyclohexyl-1H-tetrazol-5-yl)-N-(furan-2-yl-methyl)-1-(5-phenyl-2H-1,2,3-triazol-4-yl)-methanamine (10d): colorless oil: 1H NMR (400 MHz, CD3OD) 7.65 (m, 2H), 7.50 (m, 1H), 7.44 (m, 3H), 6.37 (m, 1H), 6.19 (d, J = 3.1 Hz, 1H), 5.61 (s, 1H), 3.98 (d, J = 14.3 Hz, 1H), 3.83 (d, J = 14.4 Hz, 1H), 1.38 (s, 9H); 13C NMR (100 MHz, CD3OD) δ 155.76, 153.74, 143.87, 130.13, 130.01, 129.03, 111.46, 109.78, 63.36, 50.20, 44.27, 29.69.
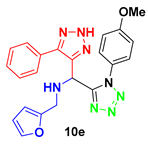
N-(furan-2-yl-methyl)-1-(1-(4-methoxyphenyl)-1H-tetrazol-5-il)-1-(5-phenyl-2H-1,2,3-triazol-4-yl)-methanamine (10e): Colorless oil: 1H NMR (500 MHz, CD3OD) 7.33 (m, 1H), 7.26 (t, J = 7.2 Hz, 1H), 7.21 (t, J = 7.4 Hz, 2H), 7.12 (d, J = 7.3 Hz, 2H), 6.85 (d, J = 8.9 Hz, 1H), 6.67 (d, J = 8.9 Hz, 1H), 6.22 (m, 1H), 6.01 (d, J = 2.8 Hz, 1H), 5.29 (s, 1H), 3.86 (d, J = 14.6 Hz, 1H), 3.82 (d, J = 14.5 Hz, 1H), 3.68 (s, 3H); 13C NMR (125 MHz, CD3OD) δ 161.18, 155.39, 152.12, 142.43, 128.47, 127.49, 126.26, 125.35, 114.34, 109.98, 108.27, 54.72, 46.78, 42.76.
Author Contributions
Conceptualization, R.G.-M.; methodology, C.Z.-H. and M.A.R.-G.; software, F.R.-L.; validation, R.G.-M.; formal analysis, R.G.-M.; investigation C.Z.-H., M.A.R.-G., and R.G.-M.; resources, R.G.-M.; data curation, C.Z.-H. and M.A.R.-G.; writing—original draft preparation, F.R.-L.; writing—review and editing, R.G.-M.; visualization, R.G.-M.; supervision, R.G.-M.; project administration, R.G.-M.; funding acquisition, R.G.-M.. All authors have read and agreed to the published version of the manuscript.
Funding
F.R.-L. is grateful to CONACYT-Mexico for a scholarship (764724). R.G.-M. is grateful for financial support from UG CIIC 005/2022, 132/2023, and CONACYT (CB-2016-285622).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ruijter, E.; Orru, R.V.A. Discovery of MCRs. In Multicomponent Reactions in Organic Synthesis, 1st ed.; Zhu, J., Wang, Q., Wang, M., Eds.; Wiley-VCH: Weinheim, Germany, 2015; pp. 13–15. [Google Scholar]
- Tietze, L.F.; Brasche, G.; Gericke, K.M. Domino Reactions in Organic Synthesis, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; p. 542. [Google Scholar]
- Ugi, I.; Dömling, A.; Ebert, B. Combinatorial Chemistry of Multicomponent Reactions. In Combinatorial Chemistry: Synthesis, Analysis, Screening, 1st ed.; Günther, J., Ed.; WYLE-VCH: Weinheim, Germany, 1999; pp. 124–125. [Google Scholar]
- Vicente-García, E.; Kielland, N.; Lavilla, R. Functionalization of Heterocycles by MCRs. In Multicomponent Reactions in Organic Synthesis, 1st ed.; Zhu, J., Wang, Q., Wang, M., Eds.; Wiley-VCH: Weinheim, Germany, 2015; pp. 159–206. [Google Scholar]
- Banfi, L.; Basso, A.; Riva, R. Synthesis of Heterocycles Through Classical Ugi and Passerini Reactions Followed by Secondary Transformations Involving One or Two Additional Functional Groups. In Synthesis of Heterocycles via Multicomponent Reactions I, 1st ed.; Orru, R.V.A., Ruijter, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 23, pp. 1–8. [Google Scholar]
- Ugi, I.; Meyr, R.; Fetzer, U.; Steinsbrückner, C. Versuche mit Isonitrikn. Angew. Chem. 1959, 71, 386. [Google Scholar]
- Ugi, I.; Steinbruckner, C. Reaktion von isonitrilen mit carbonylverbindungen, aminen und stickstoffwasserstoffsäure. Chem. Ber. 1961, 94, 734–742. [Google Scholar] [CrossRef]
- Wei, C.X.; Bian, M.; Gong, G.H. Tetrazolium compounds: Synthesis and applications in medicine. Molecules 2015, 20, 5528–5553. [Google Scholar] [CrossRef] [PubMed]
- Neochoritis, C.G.; Zhao, T.; Dömling, A. Tetrazoles via multicomponent reactions. Chem. Rev. 2019, 119, 1970–2042. [Google Scholar] [CrossRef] [PubMed]
- Dheer, D.; Singh, V.; Shankar, R. Medicinal attributes of 1, 2, 3-triazoles: Current developments. Bioorg. Chem. 2017, 71, 30–54. [Google Scholar] [CrossRef] [PubMed]
- Schröder, D.C.; Kracker, O.; Fröhr, T.; Góra, G.; Jewginski, M.; Nieß, A.; Antes, I.; Latajka, R.; Marion, A.; Sewald, N. 1, 4-Disubstituted 1 H-1, 2, 3-Triazole Containing Peptidotriazolamers: A New Class of Peptidomimetics With Interesting Foldamer Properties. Front. Chem. 2019, 7, 155. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.I. One-Pot Construction of Bis-Heterocycles through Isocyanide Based Multicomponent Reactions. ChemistrySelect 2020, 5, 8040–8061. [Google Scholar] [CrossRef]
- Soural, M.; Bouillon, I.; Krchňak, V. Combinatorial libraries of bis-heterocyclic compounds with skeletal diversity. J. Comb. Chem. 2008, 10, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among US FDA approved pharmaceuticals: Miniperspective. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef] [PubMed]
- Shahrisa, A.; Esmati, S. Three novel sequential reactions for the facile synthesis of a library of bisheterocycles possessing the 3-aminoimidazo [1,2-a] pyridine core catalyzed by bismuth (III) chloride. Synlett 2013, 24, 595–602. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).