Evaluation of Organophosphate Pesticide Residues in Food Using the Partial Least Squares Method †
Abstract
:1. Introduction
2. Methods
2.1. Definition of Target Property and Structural Descriptors
2.2. Partial Least Squares (PLS) Method
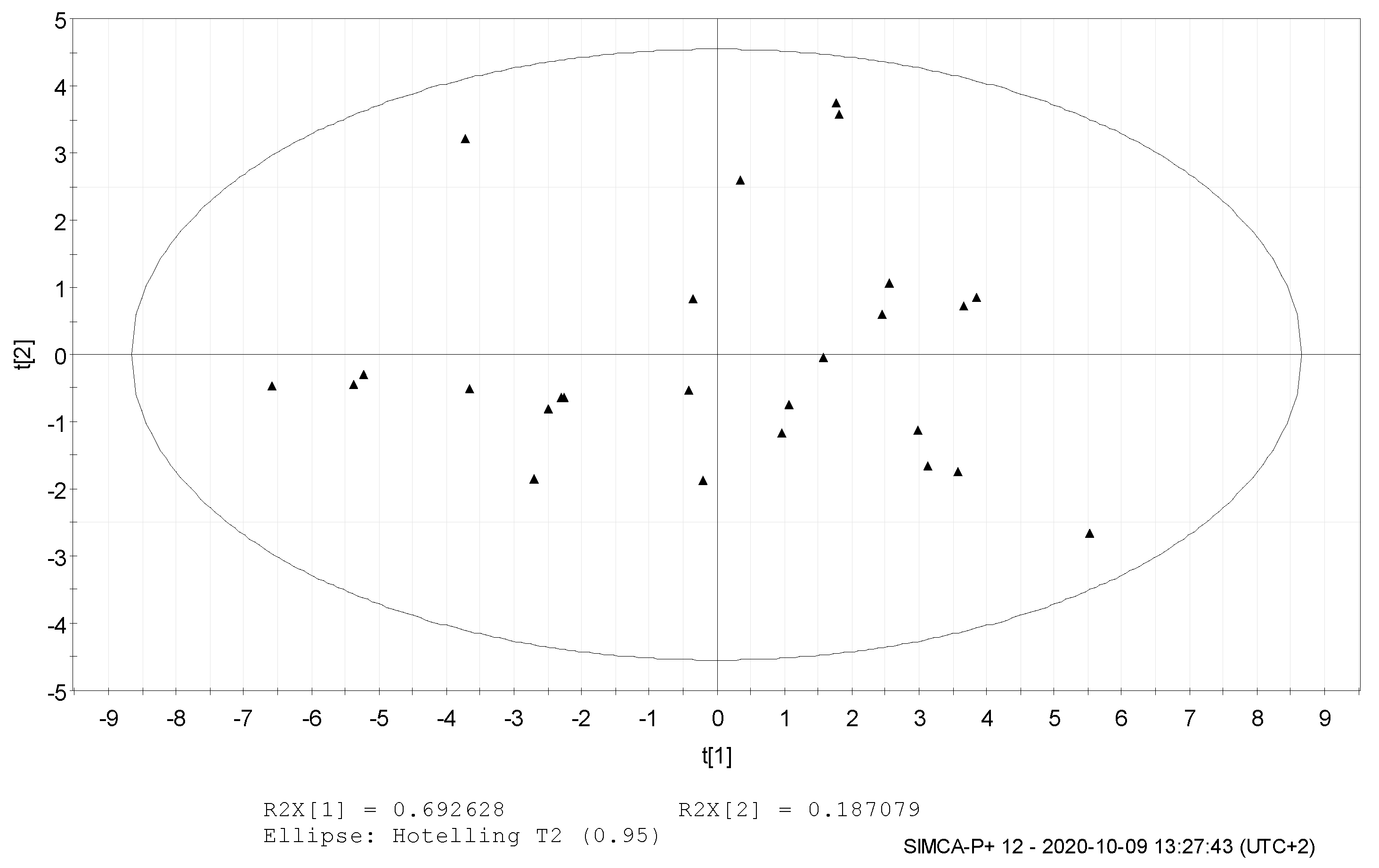
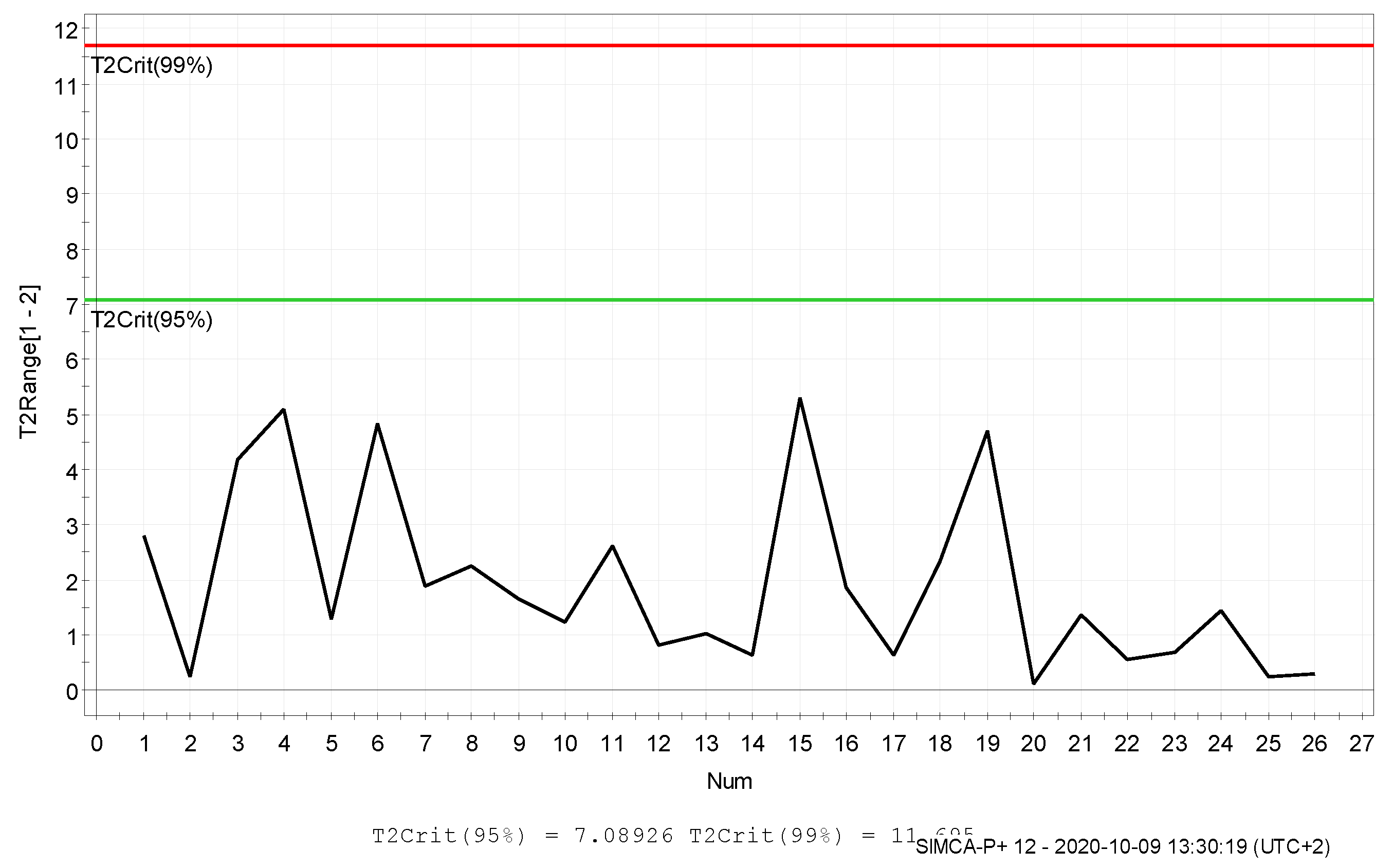
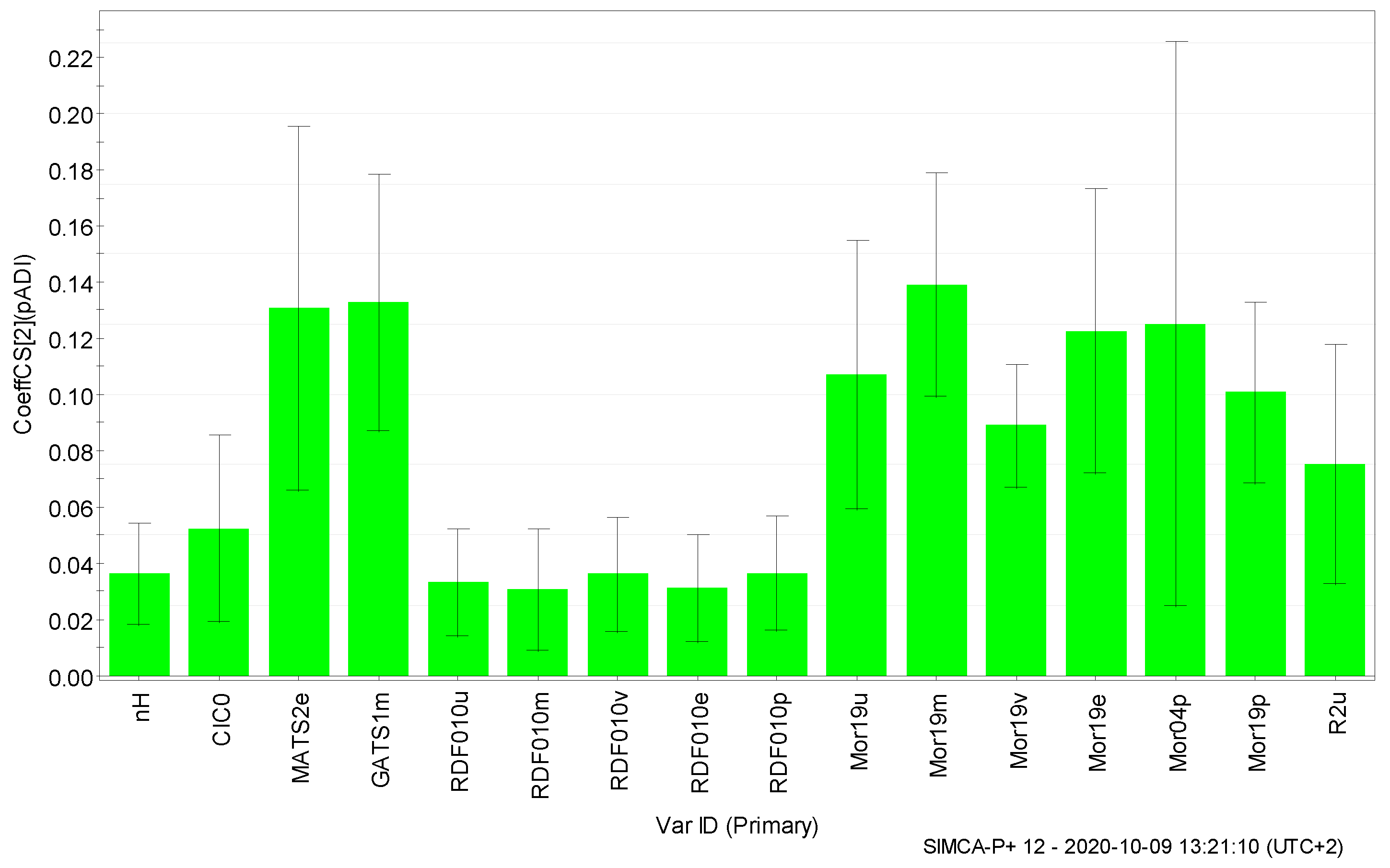
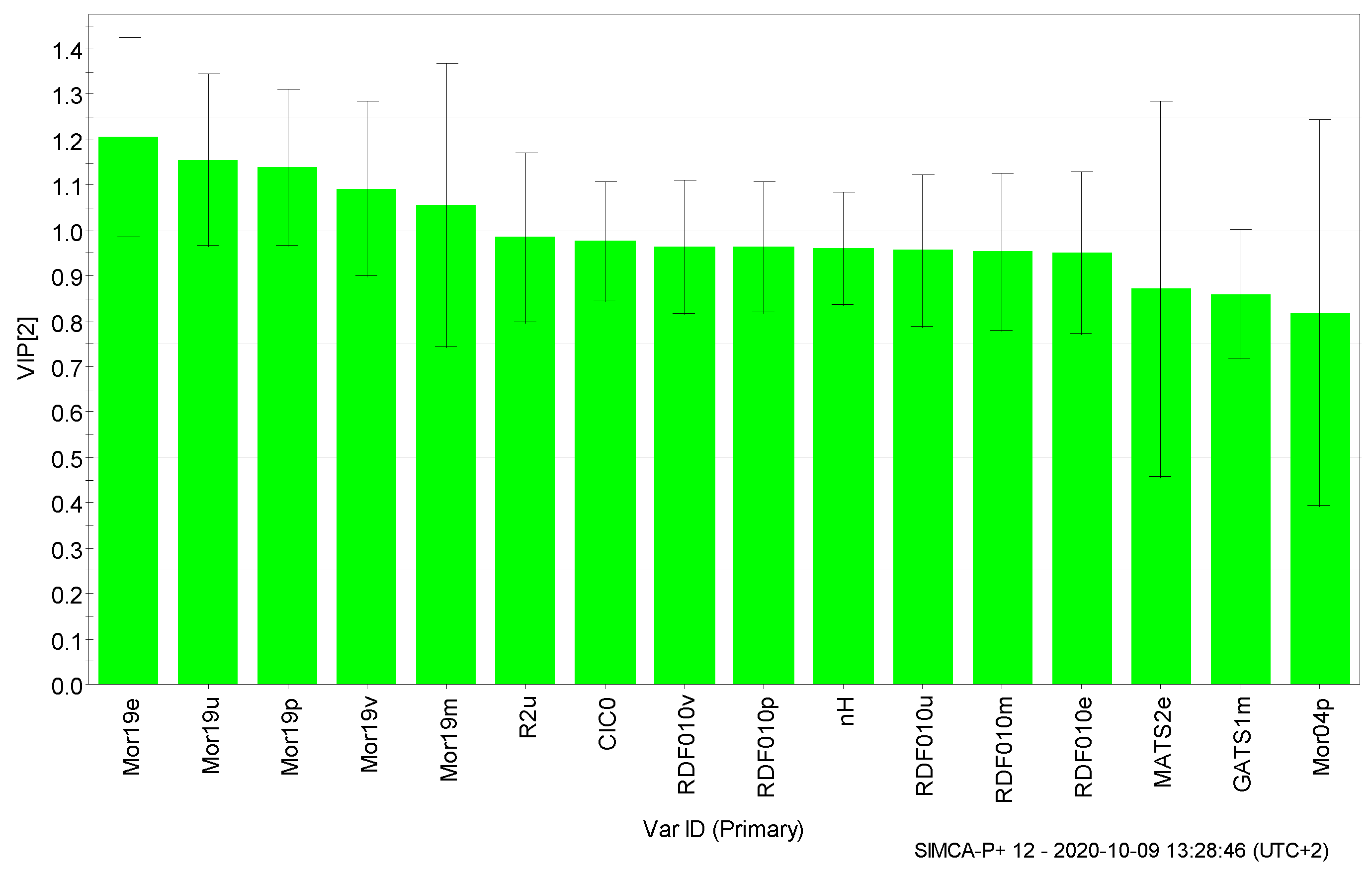
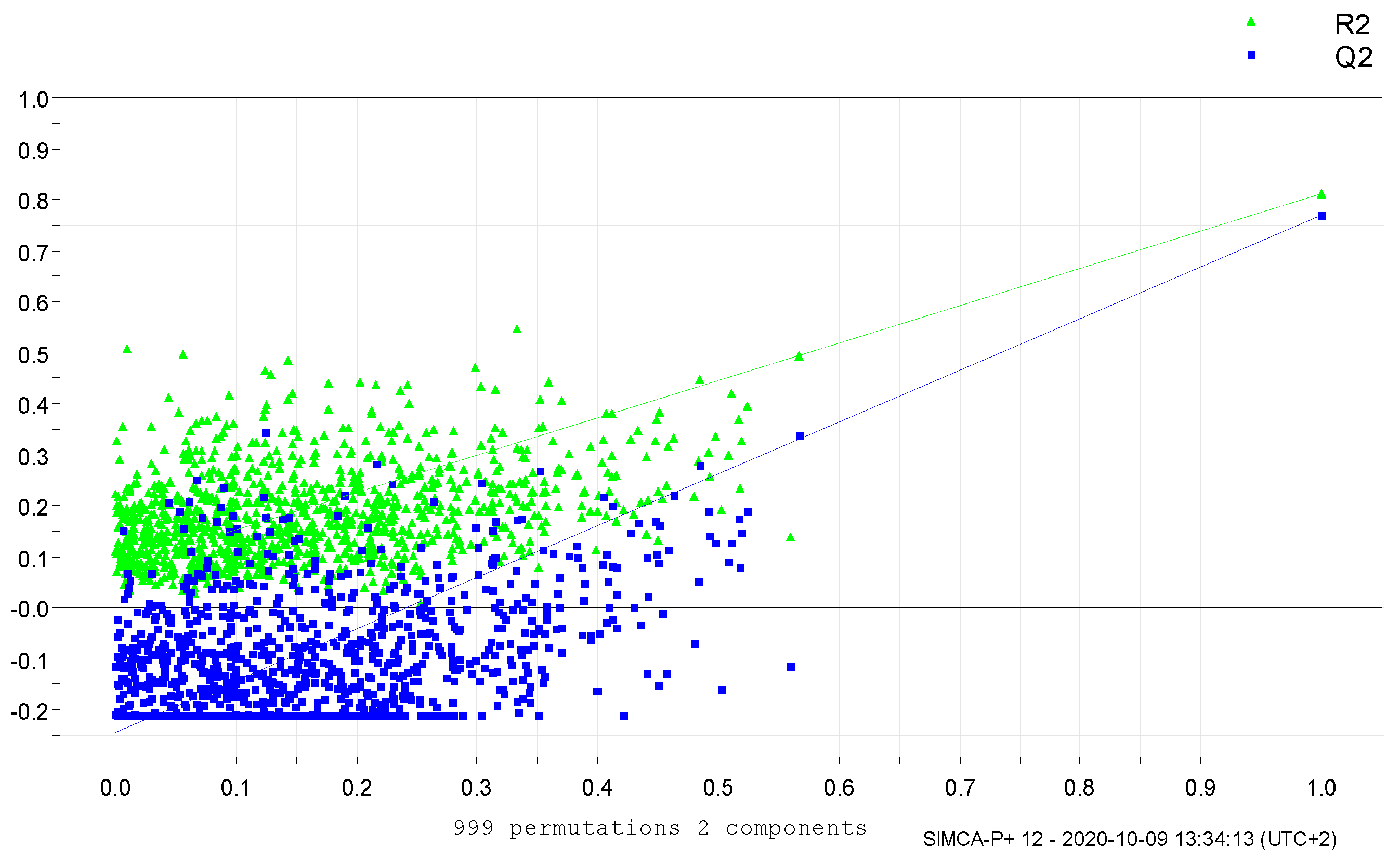
3. Results and Discussion
4. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Renwick, A.G. Pesticide residue analysis and its relationship to hazard characterisation (ADI/ARfD) and intake estimations (NEDI/NESTI). Pest Manag. Sci. 2002, 58, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Bhanti, M.; Taneja, A. Contamination of vegetables of different seasons with organophosphorous pesticides and related health risk assessment in northern India. Chemosphere 2007, 69, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Galloway, T.; Handy, R. Immunotoxicity of organophosphorous pesticides. Ecotoxicology 2003, 12, 345–363. [Google Scholar] [CrossRef] [PubMed]
- Guodong, D.; Pei, W.; Ying, T.; Jun, Z.; Yu, G.; Xiaojin, W.; Rong, S.; Guoquan, W.; Xiaoming, S. Organophosphate pesticide exposure and neurodevelopment in young Shanghai children. Environ. Sci. Technol. 2012, 46, 2911–2917. [Google Scholar] [CrossRef] [PubMed]
- Casarett and Doull’s Toxicology. The Basic Science of Poisons, 9th ed.; Klaassen, C.D., Amdur, M.O., Doull, J., Eds.; McGraw-Hill Education: New York, NY, USA; Chicago, IL, USA; San Francisco, CA, USA; Athens, Greece; London, UK; Madrid, Spanish; Mexico City, Mexico; Milan, Italy; New Delhi, Indian; Singapore; Sydney, Australia; Toronto, ON, Canada, 2019. [Google Scholar]
- Casida, J.E.; Quistad, G.B. Serine hydrolase targets of organophosphorus toxicants. Chem. Biol. Interact. 2005, 157, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Chun, O.K.; Kang, H.G. Estimation of risks of pesticide exposure, by food intake, to Koreans. Food Chem. Toxicol. 2003, 41, 1063–1076. [Google Scholar] [CrossRef]
- Joint FAO/WHO Expert Consultation on the Application of Risk Analysis to Food Standards Issues (1995: Geneva, Switzerland), World Health Organization. Food Safety Team & Food and Agriculture Organization of the United Nations. Application of Risk Analysis to Food Standards Issues: Report of the Joint FAO/WHO Expert Consultation, Geneva, Switzerland, 13–17 March 1995. World Health Organization. Available online: https://apps.who.int/iris/handle/10665/58913 (accessed on 4 March 2020).
- Joint FAO/WHO Expert Committee on Food Additives. World Health Organization & Food and Agriculture Organization of the United Nations: Procedures for the Testing of Intentional Food Additives to Establish Their Safety for Use: Second Report of the Joint FAO/WHO Expert Committee on Food Additives [Meeting Held in Geneva from 17 to 24 June 1957]. WHO Techn. Rep. Ser., No. 144, FAO Nutrition Meetings. Report Series, No. 17. 1958. Available online: https://apps.who.int/iris/handle/10665/40403 (accessed on 4 March 2020).
- WHO Expert Committee on Pesticide Residues. World Health Organization & Food and Agriculture Organization of the United Nations: Principles Governing Consumer Safety in Relation to Pesticide Residues: Report of a Meeting of a WHO Expert Committee on Pesticide Residues Held Jointly with the FAO Panel of Experts on the Use of Pesticides in Agriculture [Meeting held in Rome from 9 to 16 October 1961]. WHO Tech. Rep. Ser., No. 240. 1962. Available online: https://apps.who.int/iris/handle/10665/40536 (accessed on 4 March 2020).
- Joint FAO/WHO Expert Committee on Food Additives; World Health Organization & Food and Agriculture Organization of the United Nations. Evaluation of the Toxicity of a Number of Antimicrobials and Antioxidants: Sixth Report of the Joint FAO/WHO Expert Committee on Food Additives [Meeting Held in Geneva from 5 to 12 June 1961]. WHO Tech. Rep. Ser. No. 228. FAO Nutrition Meetings. Report Series No. 31. 1962. Available online: https://apps.who.int/iris/handle/10665/40518 (accessed on 4 March 2020).
- WHO International Programme on Chemical Safety; Joint FAO/WHO Expert Committee on Food Additives; WHO Task Group on Updating the Principles for the Safety Assessment of Food Additives and Contaminants in Food; International Labour Organization. Principles for the Safety Assessment of Food Additives and Contaminants in Food, Published under the Joint Sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization in Collaboration with the Food and Agriculture Organization of the United Nations. WHO, Environmental Health Criteria No. 70. 1987. Available online: https://apps.who.int/iris/handle/10665/37578 (accessed on 4 March 2020).
- Lu, F.C. Acceptable daily intake: Inception, evolution, and application. Regul. Toxicol. Pharmacol. 1988, 8, 45–60. [Google Scholar] [CrossRef]
- Rubery, E.D.; Barlow, S.M.; Steadman, J.H. Criteria for setting quantitative estimates of acceptable intakes of chemicals in food in the U.K. Food Addit. Contam. 1990, 7, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, P.C.D.; Skillman, A.G.; Warren, G.L.; Ellingson, B.A.; Stahl, M.T. Conformer generation with OMEGA: Algorithm and validation using high quality structures from the Protein Databank and Cambridge Structural Database. J. Chem. Inf. Model. 2010, 50, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, P.C.D.; Nicholls, A. Conformer generation with OMEGA: Learning from the data set and the analysis of failures. J. Chem. Inf. Model. 2012, 52, 2919–2936. [Google Scholar] [CrossRef] [PubMed]
- Wold, H.; Kotz, S.; Johnson, N.L. (Eds.) Encyclopedia of Statistical Sciences; Wiley: New York, NY, USA, 1985; Volume 6, p. 581. [Google Scholar]
- Roy, P.P.; Paul, S.; Mitra, I.; Roy, K. On two novel parameters for validation of predictive QSAR models. Molecules 2009, 14, 1660–1701. [Google Scholar]
- Roy, K.; Kar, S.; Ambure, P. On a simple approach for determining applicability domain of QSAR models. Chemometr. Intell. Lab. Syst. 2015, 145, 22–29. [Google Scholar] [CrossRef]


| No | Structure | pADIexp | PmodXPS + [2] | No | Structure | pADIexp | PmodXPS + [2] |
|---|---|---|---|---|---|---|---|
| 1 * | 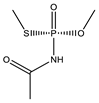 | 6.79 | 0.424 | 24 | 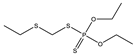 | 7.11 | 0.593 |
| 2 * |  | 7.8 | 0.292 | 25 | 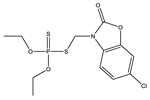 | 8.54 | 0.657 |
| 3 | 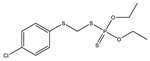 | 6.96 | 0.729 | 26* | 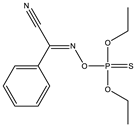 | 7.22 | 0.183 |
| 4 * | 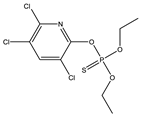 | 6.99 | 0.138 | 27 | 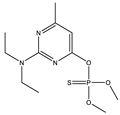 | 8.22 | 0.903 |
| 5 * |  | 8.84 | 0.084 | 28 | 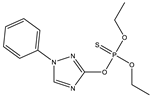 | 7.6 | 0.607 |
| 6 |  | 8.25 | 0.181 | 29 * | 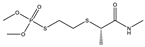 | 7.55 | 0.055 |
| 7 * | 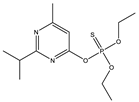 | 7.54 | 0.676 | 30 * |  | 8.48 | 0.739 |
| 8 | 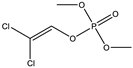 | 7.51 | 0.962 | 31 | 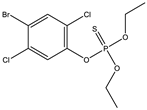 | 8.17 | 0.093 |
| 9 *,** | 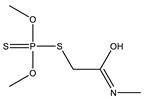 | 6.46 | 0.039 | 32 | 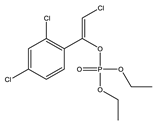 | 8.57 | 0.894 |
| 10 | 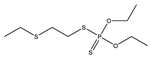 | 8.89 | 0.129 | 33 * | 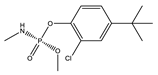 | 8.85 | 0.368 |
| 11 |  | 8.18 | 0.245 | 34 | 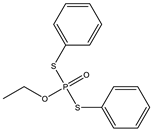 | 7.77 | 0.658 |
| 12 | 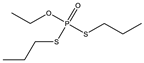 | 7.74 | 0.519 | 35 | 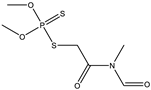 | 7.12 | 0.999 |
| 13 | 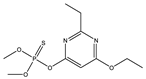 | 7.36 | 0.139 | 36 |  | 8.03 | 0.853 |
| 14 *,** | 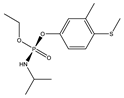 | 8.96 | 0.016 | 37 | 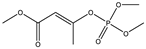 | 9.11 | 0.764 |
| 15 * | 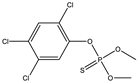 | 8.01 | 0.561 | 38 * | 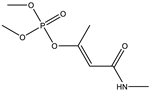 | 8.57 | 0.981 |
| 16 | 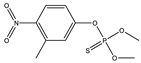 | 8.28 | 0.625 | 39 * | 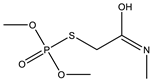 | 7.2 | 0.400 |
| 17 |  | 8.91 | 0.808 | 40 | 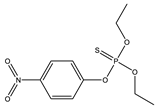 | 8.78 | 0.741 |
| 18 | 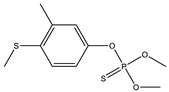 | 7.99 | 0.162 | 41 | 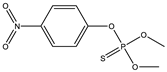 | 8.47 | 0.112 |
| 19 *,** |  | 8.78 | 0.001 | 42 *,** | 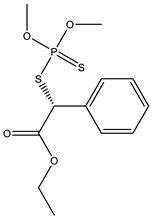 | 7.01 | 0.006 |
| 20 |  | 7.51 | 0.882 | 43 * | 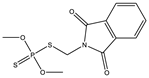 | 7.91 | 0.409 |
| 21 |  | 7.74 | 0.996 | 44 | 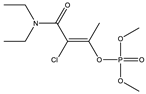 | 8.5 | 0.122 |
| 22 | 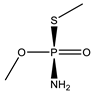 | 9.01 | 0.166 | 45 *,** | 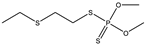 | 7.41 | 0.000 |
| 23 * |  | 8.44 | 0.408 | 46 * | 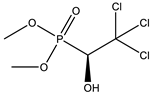 | 7.56 | 0.081 |
| No | Variable ID * | CoefCS [2] | VIP [2] |
|---|---|---|---|
| 1 | CIC0 | 0.052 | 0.976 |
| 2 | GATS1m | 0.133 | 0.860 |
| 3 | MATS2e | 0.131 | 0.872 |
| 4 | Mor04p | 0.125 | 0.818 |
| 5 | Mor19e | 0.123 | 1.205 |
| 6 | Mor19m | 0.139 | 1.057 |
| 7 | Mor19p | 0.101 | 1.139 |
| 8 | Mor19u | 0.107 | 1.156 |
| 9 | Mor19v | 0.089 | 1.093 |
| 10 | nH | 0.036 | 0.961 |
| 11 | R2u | 0.075 | 0.985 |
| 12 | RDF010e | 0.031 | 0.951 |
| 13 | RDF010m | 0.031 | 0.953 |
| 14 | RDF010p | 0.036 | 0.963 |
| 15 | RDF010u | 0.033 | 0.957 |
| 16 | RDF010v | 0.036 | 0.963 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilia, G.; Funar-Timofei, S. Evaluation of Organophosphate Pesticide Residues in Food Using the Partial Least Squares Method. Chem. Proc. 2021, 3, 19. https://doi.org/10.3390/ecsoc-24-08307
Ilia G, Funar-Timofei S. Evaluation of Organophosphate Pesticide Residues in Food Using the Partial Least Squares Method. Chemistry Proceedings. 2021; 3(1):19. https://doi.org/10.3390/ecsoc-24-08307
Chicago/Turabian StyleIlia, Gheorghe, and Simona Funar-Timofei. 2021. "Evaluation of Organophosphate Pesticide Residues in Food Using the Partial Least Squares Method" Chemistry Proceedings 3, no. 1: 19. https://doi.org/10.3390/ecsoc-24-08307
APA StyleIlia, G., & Funar-Timofei, S. (2021). Evaluation of Organophosphate Pesticide Residues in Food Using the Partial Least Squares Method. Chemistry Proceedings, 3(1), 19. https://doi.org/10.3390/ecsoc-24-08307







