An Investigation of the Synthesis of a Hybrid Homogeneous Structure of ZnO/Si Nanowires with Stable Energy Properties †
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of the Studied Samples
2.2. Characterization Methods
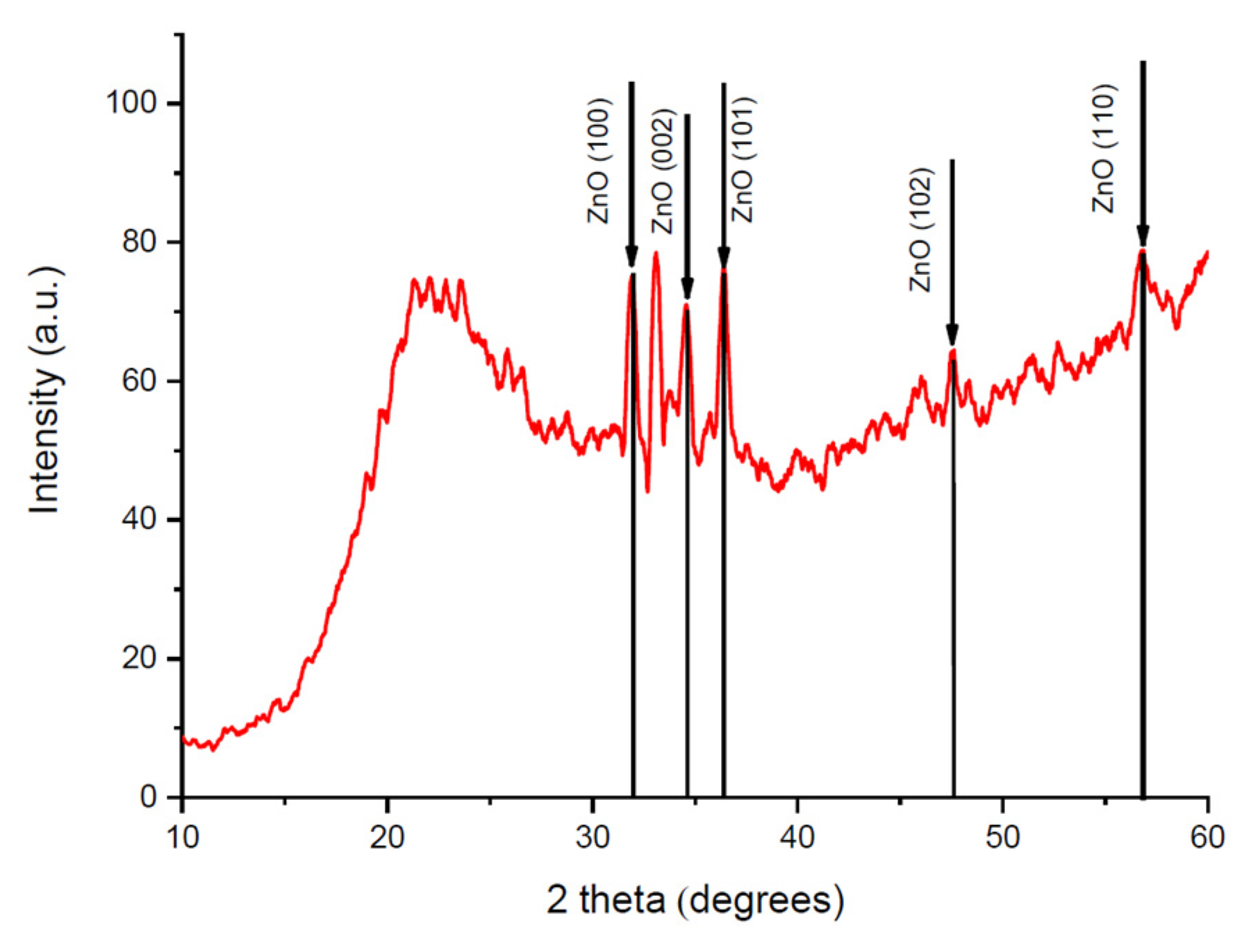
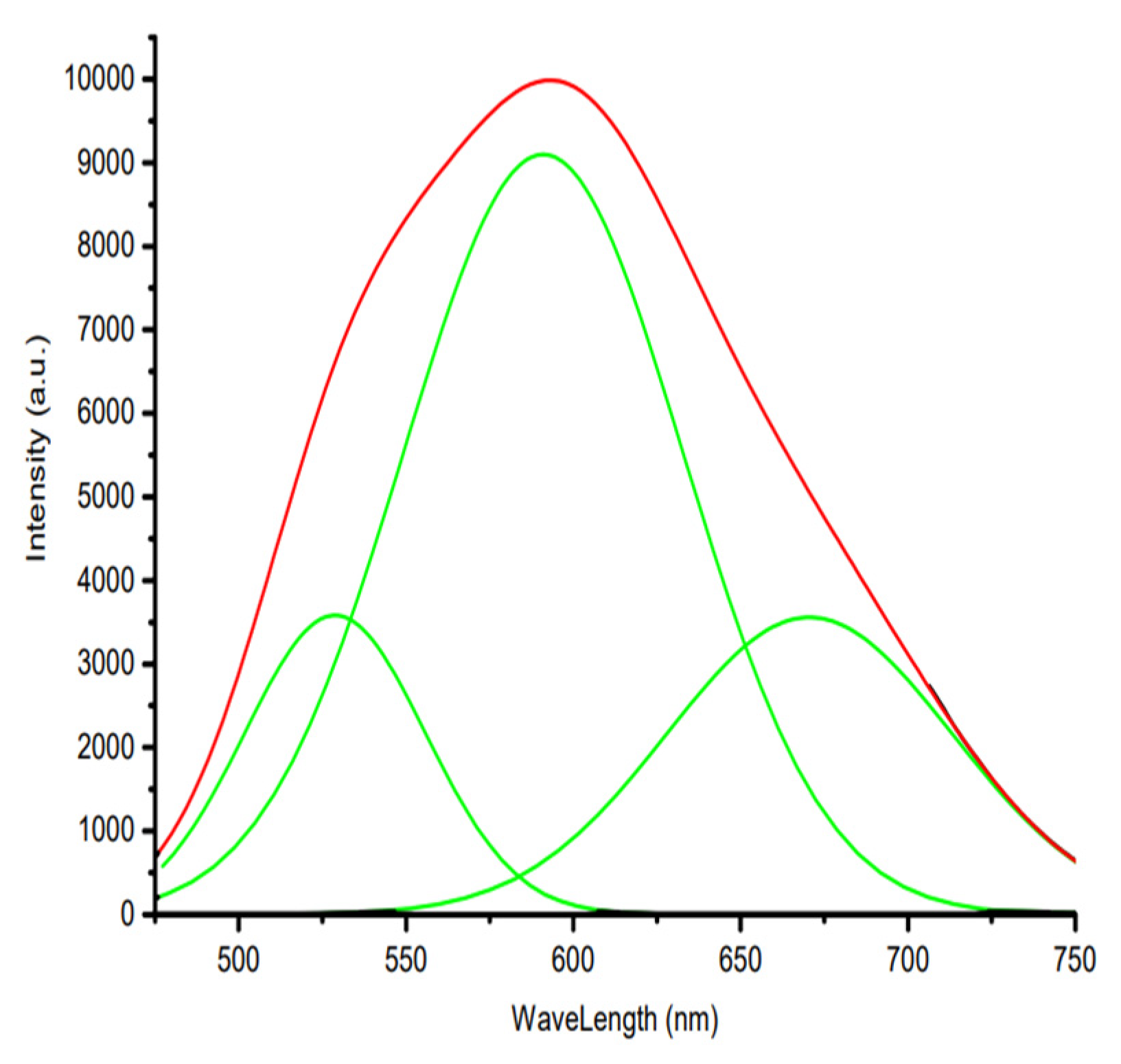
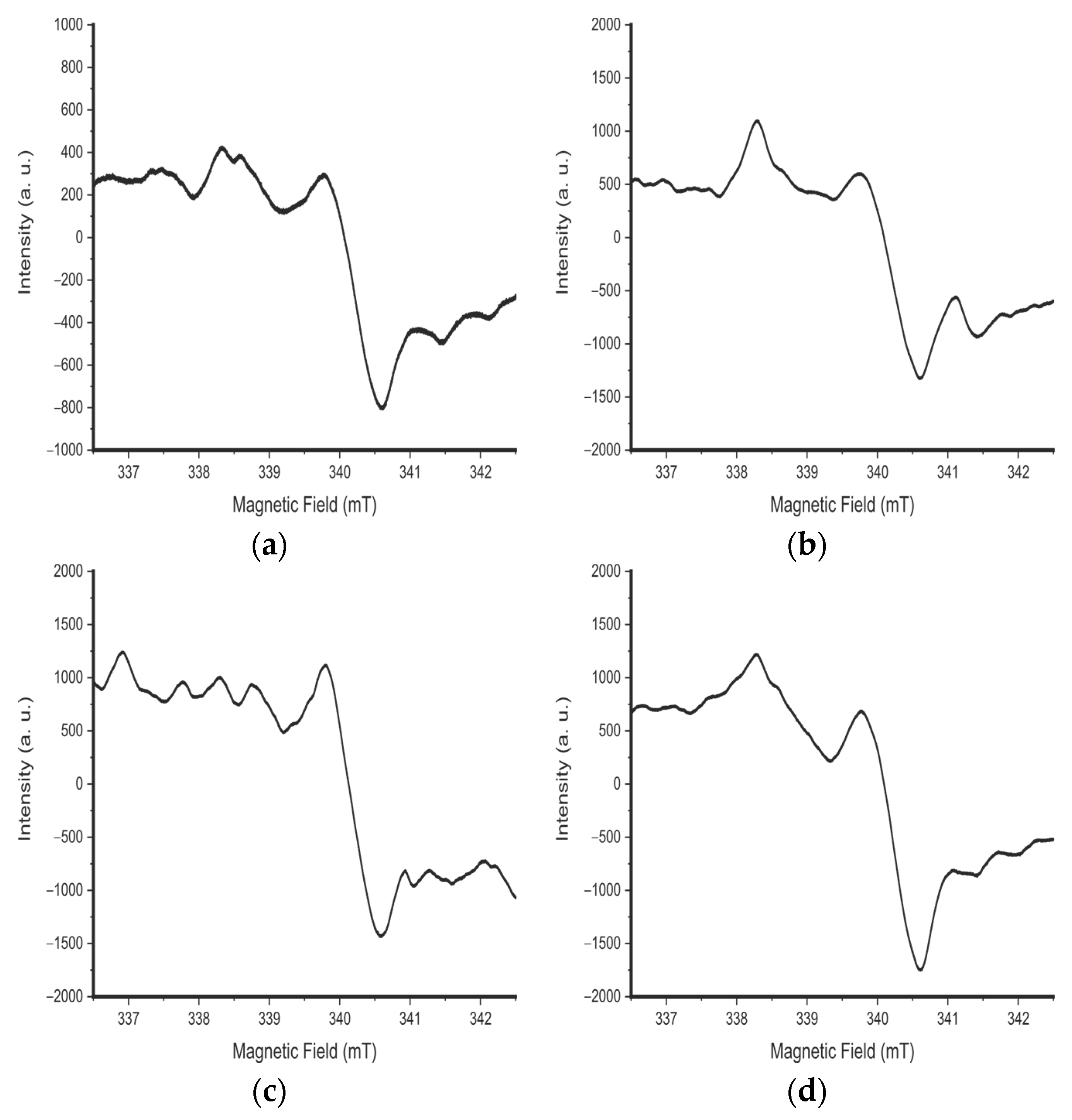
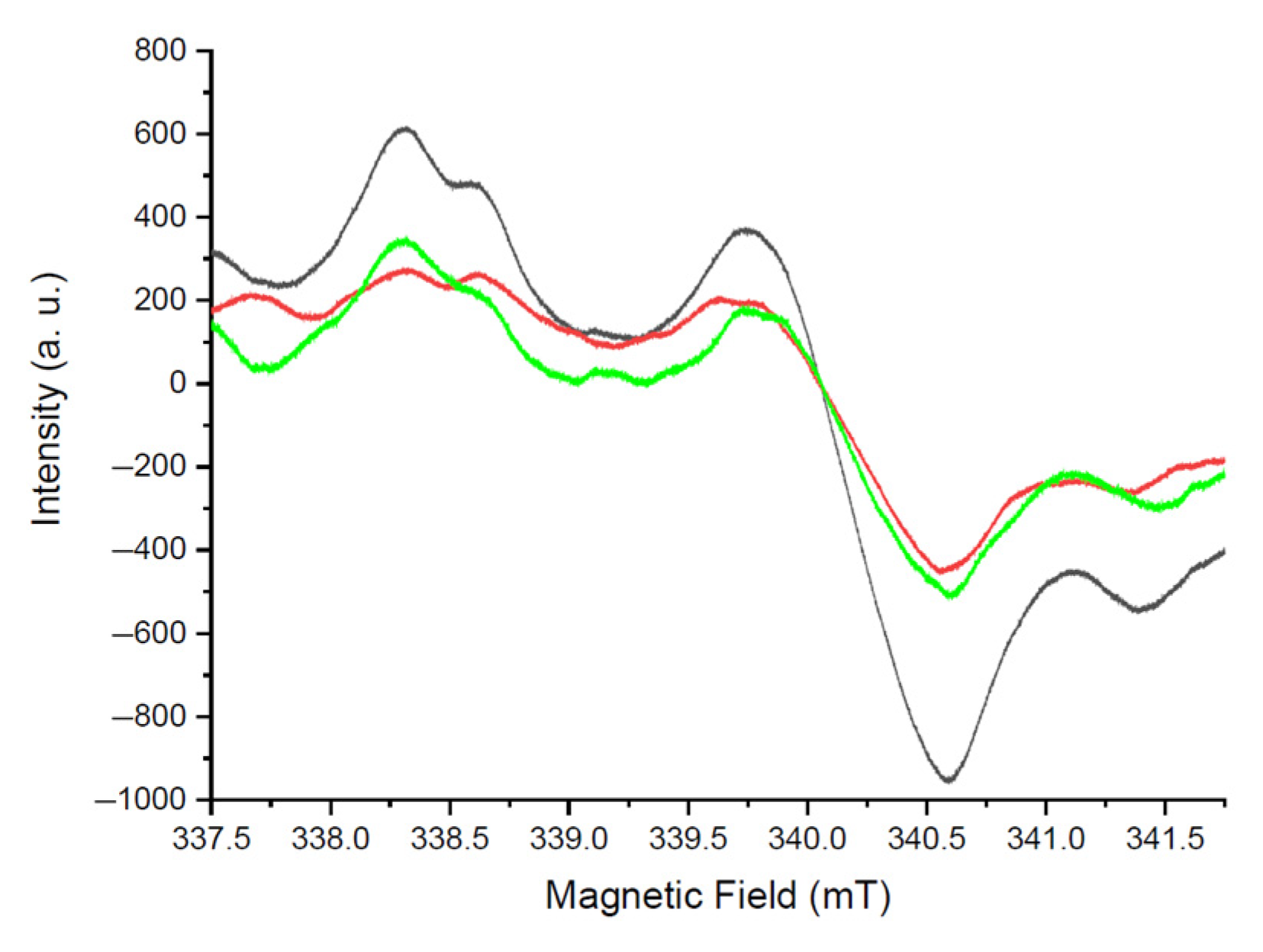
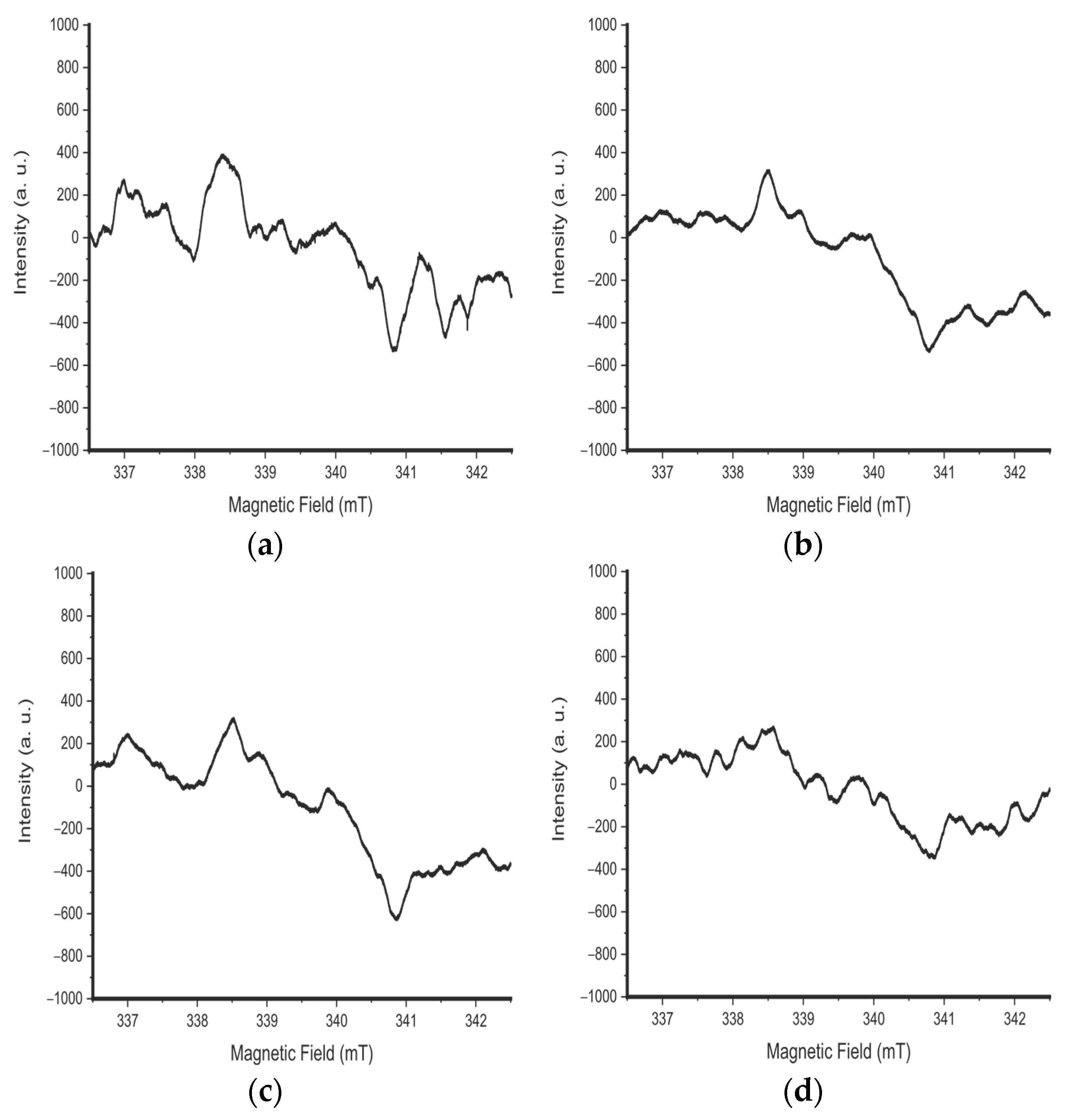
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, X.; Song, J.; Wang, Z.L. Nanowire and nanobelt arrays of zinc oxide from synthesis to properties and to novel devices. J. Mater. Chem. 2007, 17, 711–720. [Google Scholar] [CrossRef]
- Abdulgafour, H.I.; Hassan, Z.; Yam, F.K.; Jawad, M.J. Growth of ZnO nanowires without catalyst on porous silicon. AIP Conf. Proc. 2011, 1341, 18–20. [Google Scholar]
- Sherstnev, A.I.; Chubenko, E.B.; Redko, S.V.; Petrovich, V.A.; Pilipenko, V.A.; Bondarenko, V.P. Formation of nanocomposite materials based on porous silicon and zinc oxide by electrochemical method. Rep. Belarusian State Univ. Inform. Radioelectron. 2016, 95, 82–88. [Google Scholar]
- Hsu, H.C.; Cheng, C.S.; Chang, C.C.; Yang, S.; Chang, C.S.; Hsieh, W.F. Orientation-enhanced growth and optical properties of ZnO nanowires grown on porous silicon substrates. Nanotechnology 2005, 16, 297. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.J.; Pickett, M.D.; Li, X.; Ohlberg, D.A.; Stewart, D.R.; Williams, R.S. Memristive switching mechanism for metal/oxide/metal nanodevices. Nat. Nanotechnol. 2008, 3, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Sawa, A. Resistive switching in transition metal oxides. Mater. Today 2008, 11, 28–36. [Google Scholar] [CrossRef]
- Zoolfakar, A.S.; Kadir, R.A.; Rani, R.A.; Balendhran, S.; Liu, X.; Kats, E.; Bhargava, S.K.; Bhaskaran, M.; Sriram, S.; Zhuiykov, S.; et al. Engineering electrodeposited ZnO films and their memristive switching performance. Phys. Chem. Chem. Phys. 2013, 15, 10376–10384. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Chen, B.; Gao, B.; Zhang, F.; Chen, Y.; Liu, X.; Kang, J. Engineering oxide resistive switching materials for memristive device application. Appl. Phys. A 2011, 102, 991–996. [Google Scholar] [CrossRef]
- Martínez, L.; Ocampo, O.; Kumar, Y.; Agarwal, V. ZnO-porous silicon nanocomposite for possible memristive device fabrication. Nanoscale Res. Lett. 2014, 9, 437. [Google Scholar] [CrossRef] [PubMed]
- Gagarina, A.Y.; Bogoslovskaya, L.S.; Spivak, Y.M.; Novikova, K.N.; Kuznetsov, A.; Moshnikov, V.A. Synthesis of arrays nanostructured porous silicon wires in electron conductivity type silicon with crystallographic orientation (111). Tech. Phys. 2023, 68, 254. [Google Scholar] [CrossRef]
- Nalimova, S.S.; Gagarina, A.I.; Spivak, I.M.; Bobkov, A.A.; Kondratev, V.M.; Bolshakov, A.D.; Moshnikov, V.A. Chemoresistive Gas Sensor and Method for Its Manufacture. Russian Patent RU 2806670 C1, 2 November 2023. [Google Scholar]
- Mukhamedshina, D.; Mit’, K.; Chuchvaga, N.; Tokmoldin, N. Fabrication and study of sol-gel ZnO films for use in Si-based 716 heterojunction photovoltaic devices. Mod. Electron. Mater. 2017, 3, 158–161. [Google Scholar] [CrossRef]
- Murzalinov, D.; Seredavina, T.; Kemelbekova, A.; Spivak, Y.; Moshnikov, V.; Mukhamedshina, D.; Fedosimova, A. Investigation of Surface Nanoclusters and Paramagnetic Centers of ZnO/Por-Si Structures as the Basis of Sensory Properties. Processes 2023, 11, 3332. [Google Scholar] [CrossRef]



| Fitting Coefficient: 0.3890 | |||
|---|---|---|---|
| Element | (kEV) | Mass% | At% |
| O | 0.525 | 2.48 | 7.77 |
| Si | 1.739 | 17.10 | 30.53 |
| Zn | 8.630 | 80.42 | 61.70 |
| Total | 100.00 | 100.00 | |
| Average Size of Crystallites in the Plane, nm | |||
|---|---|---|---|
| ZnO (100) | ZnO (002) | ZnO (101) | ZnO (102) |
| 38.87 | 41.78 | 26.71 | 22.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Begunov, M.; Gagarina, A.; Zhapakov, R.; Murzalinov, D.; Seredavina, T.; Novikova, K.; Kemelbekova, A.; Spivak, Y.; Moshnikov, V.; Dmitriyeva, E. An Investigation of the Synthesis of a Hybrid Homogeneous Structure of ZnO/Si Nanowires with Stable Energy Properties. Eng. Proc. 2024, 67, 28. https://doi.org/10.3390/engproc2024067028
Begunov M, Gagarina A, Zhapakov R, Murzalinov D, Seredavina T, Novikova K, Kemelbekova A, Spivak Y, Moshnikov V, Dmitriyeva E. An Investigation of the Synthesis of a Hybrid Homogeneous Structure of ZnO/Si Nanowires with Stable Energy Properties. Engineering Proceedings. 2024; 67(1):28. https://doi.org/10.3390/engproc2024067028
Chicago/Turabian StyleBegunov, Mikhail, Alena Gagarina, Rashid Zhapakov, Danatbek Murzalinov, Tatyana Seredavina, Kristina Novikova, Ainagul Kemelbekova, Yulia Spivak, Vyacheslav Moshnikov, and Elena Dmitriyeva. 2024. "An Investigation of the Synthesis of a Hybrid Homogeneous Structure of ZnO/Si Nanowires with Stable Energy Properties" Engineering Proceedings 67, no. 1: 28. https://doi.org/10.3390/engproc2024067028
APA StyleBegunov, M., Gagarina, A., Zhapakov, R., Murzalinov, D., Seredavina, T., Novikova, K., Kemelbekova, A., Spivak, Y., Moshnikov, V., & Dmitriyeva, E. (2024). An Investigation of the Synthesis of a Hybrid Homogeneous Structure of ZnO/Si Nanowires with Stable Energy Properties. Engineering Proceedings, 67(1), 28. https://doi.org/10.3390/engproc2024067028







