Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change
Abstract
1. Introduction
- Hg inputs into the Mediterranean from widespread geochemical, volcanic, geothermal, and anthropogenic sources and an evaluation of air–sea exchanges;
- The impact of the warming and acidification of seawater on the biogeochemical cycle of Hg;
- Possible changes in the composition of biotic communities and food webs and in bioecological processes involved in MMHg biomagnification.
2. The Unique Features of the Mediterranean Sea
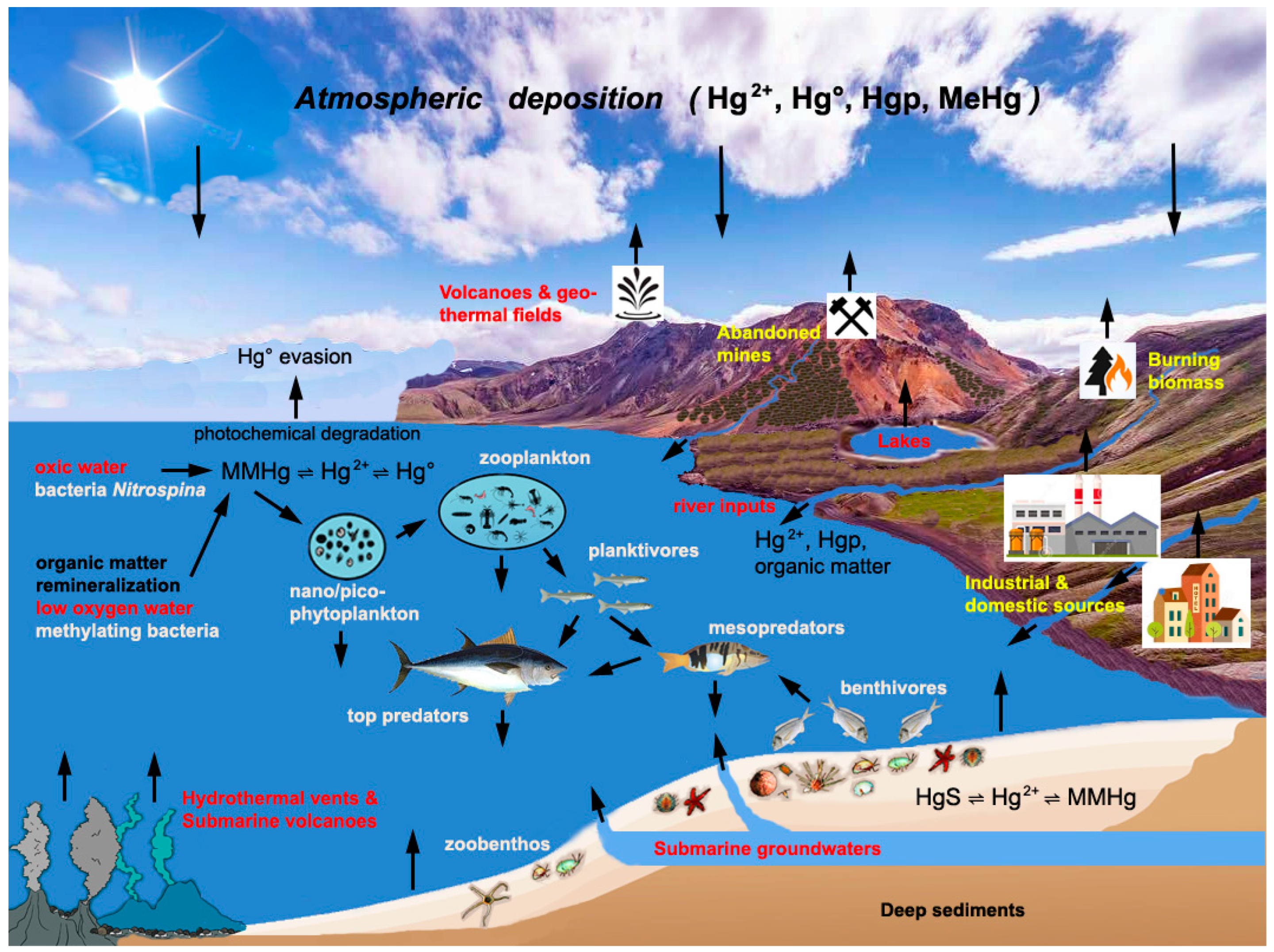
3. Sources and Cycling of Hg in the Mediterranean Sea
4. Methylation and Demethylation of Mercury
5. Methylmercury Biomagnification in Mediterranean Food Webs and Potential Risks for Human Health
6. Climate Change and the Biogeochemical Cycle of Hg in the Mediterranean
7. Future Directions and Research Needs
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jensen, S.; Jernelöv, A. Biological methylation of mercury in aquatic organisms. Nature 1969, 223, 753–754. [Google Scholar] [CrossRef]
- Ciusa, W.; Giaccio, M. Il contenuto in rame, zinco, mercurio e piombo di alcune specie ittiche dell’Adriatico. Quad. Merceol. 1971, 11, 1–12. [Google Scholar]
- Thibaud, Y. Tenure en mercure dans quelques poissons de consommation courante. Sci. Peche 1971, 209, 1–10. [Google Scholar]
- Bernhard, M.; Renzoni, A. Mercury concentrations in Mediterranean marine organisms and their environment: Natural or anthropogenic origin. Thalass. Jugosl. 1977, 13, 265–300. [Google Scholar]
- Stoeppler, M.; Bernhard, M.; Backhaus, F.; Schulte, E.K. Comparative studies on trace metal levels ion marine biota. I. Mercury in marine organisms from western Italian coast, the Strait of Gibraltar, and the North Sea. Sci. Total Environ. 1979, 13, 209–223. [Google Scholar] [CrossRef]
- Aston, S.R.; Fowler, S.W. Mercury in the open Mediterranean: Evidence of contamination? Sci. Total Environ. 1985, 43, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Renzoni, A.; Focardi, S.; Fossi, C.; Leonzio, C.; Mayol, J. Comparison between concentrations of mercury and other contaminants in eggs and tissues of Cory’s shearwater Calonectris diomedea collected on Atlantic and Mediterranean islands. Environ. Pollut. 1986, 40, 17–35. [Google Scholar] [CrossRef]
- Renzoni, A.; Zino, F.; Franchi, E. Mercury levels along the food chain and risk for exposed populations. Environ. Res. 1998, 77, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Leonzio, C.; Focardi, S.; Fossi, M.C. Heavy metals and selenium in stranded dolphins of the northern Tyrrhenian (NW Mediterranean). Sci. Total Environ. 1992, 119, 77–84. [Google Scholar] [CrossRef]
- UNEP. Mediterranean Action Plan (MAP); UN Environment Programme, Chemicals and Health Branch: Geneva, Switzerland, 2019; Available online: https://www.unep.org/unepmap/resources/publications (accessed on 2 October 2023).
- Minamata Convention on Mercury. Available online: https://www.mercuryconvention.org (accessed on 10 October 2023).
- Cinnirella, S.; Bruno, D.E.; Pirrone, N.; Horvat, M.; Zivkovic, I.; Evers, D.C.; Johnson, S.; Sunderland, E.M. Mercury concentrations in biota in the Mediterranean Sea, a compilation of 40 years of surveys. Sci. Data 2019, 6, 205. [Google Scholar] [CrossRef]
- Costantini, D.; Bustamante, P.; Bault-Favrou, M.; Dell’Olmo, G. Pattern of mercury exposure and relationships with isotopes and markers of oxidative status in chicks of a Mediterranean seabird. Environ. Pollut. 2020, 260, 114095. [Google Scholar] [CrossRef]
- Rosati, G.; Canu, D.; Lazzari, P.; Solidoro, C. Assessing the spatial and temporal variability of methylmecury biogeochemistry and bioaccumulation in the Mediterranean Sea with a coupled 3D model. Biogeosciences 2022, 19, 3663–3682. [Google Scholar] [CrossRef]
- Cossa, D.; Knoery, J.; Bănaru, D.; Harmelin-Vivien, M.; Sonke, J.E.; Hedgecock, I.M.; Bravo, A.G.; Rosati, G.; Canu, D.; Horvat, M.; et al. Mediterranean mercury assessment 2022: An updated budget, health consequences, and research perspectives. Environ. Sci. Technol. 2022, 56, 3840–3862. [Google Scholar] [CrossRef] [PubMed]
- UNEP. Global Mercury Assessment 2018; UN Environment Programme, Chemicals and Health Branch: Geneva, Switzerland, 2019; Available online: https://www.unep.org/resources/publication/global-mercury-assessment-2018 (accessed on 2 October 2023).
- Albertos, S.; Brerenguer, N.I.; Sánchez-Virosta, P.; Gómez-Ramírez, P.; Jiménez, P.; Torres-Chaparro, M.Y.; Valverde, I.; Navas, I.; María-Mojica, P.; Garcíá-Fernández, A.; et al. Mercury exposure in birds linked to marine ecosystems in the Western Mediterranean. Arch. Environ. Contam. Toxicol. 2020, 79, 435–453. [Google Scholar] [CrossRef]
- Gustin, M.S.; Bowman, K.; Branfireum, B.; Chetelat, J.M.; Eckley, C.S.; Hammerschmidt, C.R.; Lamborg, C.; Lyman, S.; Martinez-Cortizas, A.; Sommar, J.; et al. Mercury biogeochemical cycling: A synthesis of recent scientific advances. Sci. Total Environ. 2020, 737, 139619. [Google Scholar] [CrossRef]
- Bowman, K.L.; Lamborg, C.H.; Agather, A.M. A global perspective on mercury cycling in the ocean. Sci. Total Environ. 2020, 710, 136166. [Google Scholar] [CrossRef] [PubMed]
- Sonke, J.E.; Angot, H.; Zhang, Y.; Poulain, A.; Björn, E.; Schartup, A. Global change effects on biogeochemical mercury cycling. Ambio 2023, 52, 853–876. [Google Scholar] [CrossRef]
- Lee, C.S.; Lutcavage, M.; Chandler, E.; Madigan, D.J.; Cerrato, R.M.; Fisher, N.S. Declining mercury concentrations in bluefin tuna reflect reduced emissions to the North Atlantic Ocean. Environ. Sci. Technol. 2016, 50, 12825–12830. [Google Scholar] [CrossRef]
- Wang, F.; Outridge, P.M.; Feng, X.; Meng, B.; Heimbürger-Boavid, L.-E.; Mason, R.P. How closely do mercury trends in fish and other aquatic wildlife track those in the atmosphere? Implications for evaluating the effectiveness of the Minamata Convention. Sci. Total Environ. 2019, 674, 58–70. [Google Scholar] [CrossRef]
- Morris, A.D.; Wilson, S.J.; Fryer, R.J.; Thomas, P.J.; Hudelson, K.; Andreasen, B.; Blévin, P.; Bustamante, P.; Chastel, O.; Christensen, G.; et al. Temporal trends of mercury in Arctic biota: 10 more years of progress in Arctic monitoring. Sci. Total Environ. 2022, 839, 155803. [Google Scholar] [CrossRef]
- AMAP/UNEP. Technical Background Report for the Global Mercury Assessment 2018; Arctic Monitoring and Assessment Programme: Oslo, Norway; UN Environment Programme, Chemicals and Health Branch: Geneva, Switzerland, 2019; 426p. [Google Scholar]
- Bagnato, E.; Oliveri, E.; Acquavita, A.; Covelli, S.; Petranich, E.; Barra, M.; Italiano, F.; Parello, F.; Sprovieri, M. Hydrochemical mercury and air-sea exchange over the submarine hydrothermal vents off-shore Panarea Island (Aeolian Arc, Tyrrhenian Sea). Mar. Chem. 2017, 194, 63–78. [Google Scholar] [CrossRef]
- Andaloro, F.; Romeo, T.; Renzi, M.; Guerranti, C.; Perra, G.; Consoli, P.; Perzia, P.; Focardi, S.E. Alteration of potential harmful elements levels in sediments and biota from the central Mediterranean Sea (Aeolian Archipelago) following an episode of intense volcanic activity. Environ. Monit. Assess. 2012, 184, 4035–4047. [Google Scholar] [CrossRef]
- Baldi, F.; Bargagli, R. Mercury pollution in marine sediments near a chlor-alkali plant: Distribution and availability of the metal. Sci. Total Environ. 1984, 39, 15–26. [Google Scholar] [CrossRef]
- Covelli, S.; Faganelli, J.; Horvat, M.; Brambati, A. Mercury contamination of coastal sediments as the result of long-term cinnabar mining activity (Gulf of Trieste, northern Adriatic Sea). Appl. Geochem. 2001, 16, 541–558. [Google Scholar] [CrossRef]
- Rimondi, V.; Costagliola, P.; Lattanzi, P.; Morelli, G.; Cara, G.; Cencetti, C.; Fagotti, C.; Fredduzzi, A.; Marchetti, G.; Sconocchia, A.; et al. A 200 km-long mercury contamination of the Paglia and Tiber floodplain: Monitoring results and implications for environmental management. Environ. Pollut. 2019, 255, 113191. [Google Scholar] [CrossRef]
- Ozkan, E.Y.; Buyukisik, H.B.; Kontas, A.; Turkdogan, M. A survey of metal concentrations in marine sediment cores in the vicinity of an old mercury-mining area in Karaburun, Aegean Sea. Environ. Sci. Pollut. Res. 2017, 24, 13823–13836. [Google Scholar] [CrossRef]
- Degetto, S.; Schintu, M.; Contu, A.; Sbrignadello, G. Santa Gilla Lagoon (Italy): A mercury sediment pollution case study. Contamination assessment and restoration of the site. Sci. Total Environ. 1997, 204, 49–56. [Google Scholar] [CrossRef]
- Tessier, E.; Garnier, C.; Mullot, J.-U.; Lenoble, V.; Arnaud, M.; Raynaud, M.; Mounier, S. Study of the spatial and historical distribution of sediment inorganic contamination in the Toulon Bay (France). Mar. Pollut. Bull. 2011, 62, 2075–2086. [Google Scholar] [CrossRef] [PubMed]
- Rosati, G.; Solidoro, C.; Canu, D. Mercury dynamics in a changing coastal area over industrial and post-industrial phases: Lessons from the Venice Lagoon. Sci. Total Environ. 2020, 743, 140586. [Google Scholar] [CrossRef] [PubMed]
- Richon, C.; Dutay, J.-C.; Bopp, L.; Le Vu, B.; Orr, J.C.; Somot, S.; Dulac, F. Biogeochemical response of the Mediterranean Sea to the transient SRES-A2 climate change scenario. Biogeosciences 2019, 16, 135–165. [Google Scholar] [CrossRef]
- Calafat, F.M.; Frederiske, T.; Horsburgh, K. The sources of sea-level changes in the Mediterranean Sea since 1960. J. Geophys. Res. 2022, 127, e2022JC019061. [Google Scholar] [CrossRef]
- Zhang, Y.; Dutkiewicz, S.; Sunderland, E.M. Impact of climate change on methylmercury formations and bioaccumulation in the 21th century ocean. One Earth 2021, 4, 279–288. [Google Scholar] [CrossRef]
- MedECC. Climate and Environmental Change in the Mediterranean Basin—Current Situation and Risks for the Future: First Mediterranean Assessment Report; Cramer, W., Guiot, J., Marini, K., Eds.; Union for the Mediterranean, Plan Bleu, UNEP/MAP: Marseille, France, 2020; 632p. [Google Scholar] [CrossRef]
- Zenetos, A.; Galanidi, M. Mediterranean non indigenous species at the start of the 2020s: Recent changes. Mar. Biodivers. Rec. 2020, 13, 10. [Google Scholar] [CrossRef]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Ben Rais Lasram, F.; Aguzzi, J.; Ballesteros, E.; Nike Bianchi, C.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediterranean Sea: Estimates, patterns and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhang, Q.; Maavara, T.; Liu, S.; Wang, X.; Raymond, P.A. Rivers as the largest source of mercury to coastal oceans worldwide. Nat. Geosci. 2021, 14, 672–677. [Google Scholar] [CrossRef]
- Costa, V.; Sciuretti, V.; Consoli, P.; Manea, E.; Menini, E.; Andaloro, F.; Romeo, T.; Danovaro, R. Volcanic-associated ecosystems of the Mediterranean Sea: A systematic map and an interactive tool to support their conservation. PeerJ 2023, 11, e15162. [Google Scholar] [CrossRef]
- Bakalowicz, M. Coastal karst groundwater in the Mediterranean: A resource to be preferably exploited onshore, not from karst submarine springs. Geosciences 2018, 8, 258. [Google Scholar] [CrossRef]
- Lazzari, P.; Solidoro, C.; Salon, S.; Bolzon, G. Spatial variability of phosphate and nitrate in the Mediterranean Sea: A modeling approach. Deep Sea Res. 2016, 108, 39–52. [Google Scholar] [CrossRef]
- Sammartino, M.; Di Cicco, A.; Marullo, S.; Santoleri, R. Spatio-temporal variability of micro-, nano- and pico-phytoplankton in the Mediterranean Sea from satellite ocean colour data of SeaWiFS. Ocean Sci. 2015, 11, 759–778. [Google Scholar] [CrossRef]
- Adloff, F.; Somot, S.; Sevault, F.; Jordà, G.; Aznar, R.; Dèqué, M.; Herrmann, M.; Marcos, M.; Dubois, C.; Padorno, E.; et al. Mediterranean Sea response to climate change in ensemble of twenty first century scenarios. Clim. Dyn. 2015, 45, 2775–2802. [Google Scholar] [CrossRef]
- Baumann, Z.; Jonsson, S.; Mason, R.P. Geochemistry of mercury in the marine environment. Encycl. Ocean. Sci. 2019, 6, 301–308. [Google Scholar] [CrossRef]
- Bargagli, R.; Barghigiani, C. Lichen biomonitoring of mercury emission and deposition in mining, geothermal and volcanic areas of Italy. Environ. Monit. Assess. 1991, 16, 265–275. [Google Scholar] [CrossRef]
- Edner, H.; Ragnarson, P.; Svanberg, S.; Wallinder, E.; Deliso, A.; Ferrara, R.; Maserti, B.E. Differential absorption lidar mapping of atmospheric atomic mercury in Italian geothermal fields. J. Geophys. Res. 1992, 97, 3779–3786. [Google Scholar] [CrossRef][Green Version]
- Bargagli, R.; Cateni, D.; Nelli, L.; Olmastroni, S.; Zagarese, B. Environmental impact of trace element emissions from geothermal power plants. Arch. Environ. Contam. Toxicol. 1997, 33, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, R.; Mazzolai, B.; Lanzillotta, E.; Nucaro, E.; Pirrone, N. Volcanoes as emission sources of atmospheric mercury in the Mediterranean basin. Sci. Total Environ. 2000, 259, 115–121. [Google Scholar] [CrossRef]
- Gencarelli, C.N.; De Simone, F.; Hedgecock, I.M.; Sprovieri, F.; Yang, X.; Pirrone, N. European and Mediterranean mercury modelling: Local and long-range contribution to the deposition flux. Atmos. Environ. 2015, 117, 162–168. [Google Scholar] [CrossRef]
- De Simone, F.; Gencarelli, C.N.; Hedgecock, I.M.; Pirrone, N. A modeling comparison of mercury deposition from current anthropogenic mercury emission inventories. Environ. Sci. Technol. 2016, 50, 5154–5162. [Google Scholar] [CrossRef]
- Baldi, F.; Bargagli, R. Chemical leaching and specific surface measurements of marine sediments in the evaluation of mercury contamination near cinnabar deposits. Mar. Environ. Res. 1982, 6, 69–82. [Google Scholar] [CrossRef]
- Kotnik, J.; Horvat, M.; Ogrinc, N.; Fajon, V.; Žagar, D.; Cossa, D.; Sprovieri, F.; Pirrone, N. Mercury speciation in the Adriatic Sea. Mar. Pollut. Bull. 2015, 96, 136–148. [Google Scholar] [CrossRef]
- Cossa, D.; Durrieu de Madron, X.; Schäfer, J.; Guédron, S.; Mrusczak, S.; Castelle, S.; Naudin, J.-J. Sources and exchange of mercury in the waters of the Northwestern Mediterranean margin. Progr. Oceanogr. 2018, 163, 172–183. [Google Scholar] [CrossRef]
- Širca, A.; Rajar, R.; Harris, R.C.; Horvat, M. Mercury transport and fate in the Gulf of Trieste (Northern Adriatic)—A two dimensional modelling approach. Environ. Model. Softw. 1999, 14, 645–655. [Google Scholar] [CrossRef]
- Žagar, D.; Knap, A.; Warwick, J.; Rajar, R.; Horvat, M.; Četina, M. Modelling of mercury transport and transformation processes in the Idrijca and Soča river system. Sci. Total Environ. 2006, 368, 149–163. [Google Scholar] [CrossRef]
- Heimbürger, L.-H.; Cossa, D.; Marty, J.-C.; Migon, C.; Averty, B.; Dufour, A.; Ras, J. Methyl mercury distributions in relation to the presence of nano- and picoplankton in an oceanic water column (Ligurian Sea, North-western Mediterranean). Geochim. Cosmochim. Acta 2010, 74, 5549–5559. [Google Scholar] [CrossRef]
- Jiskra, M.; Heimbürger-Boavida, L.-E.; Desgranges, M.-M.; Petrova, M.V.; Dufour, A.; Ferreira-Araujo, B.; Masbou, J.; Chmeleff, J.; Melilotus, T.; Point, D.; et al. Mercury stable isotopes constrain atmospheric sources to the ocean. Nature 2021, 597, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Munson, K.M.; Lamborg, C.H.; Swarr, G.J.; Saito, M.A. Mercury species concentrations and fluxes in the Central Tropical Pacific Ocean. Glob. Biogeochem. Cycles 2015, 29, 656–676. [Google Scholar] [CrossRef]
- Palanques, A.; Guillen, G.; Puig, P.; Durrieu de Madron, X. Storm-driven shelf-to-canyon suspended sediment transport at the southwestern Gulf of Lions. Cont. Shelf Res. 2008, 28, 1947–1956. [Google Scholar] [CrossRef]
- Amos, H.M.; Jacob, D.J.; Kocman, D.; Horowitz, H.M.; Zhang, Y.; Dutkiewicz, S.; Horvat, M.; Corbitt, E.S.; Krabbenhoft, D.P.; Sunderland, E.M. Global biogeochemical implications of mercury discharges from rivers and sediment burial. Environ. Sci. Technol. 2014, 48, 9514–9522. [Google Scholar] [CrossRef]
- Hsu-Kim, H.; Kucharzyk, K.H.; Zhang, T.; Deshusses, A. Mechanisms regulating mercury bioavailability for methylating microorganisms in the aquatic environment: A critical review. Environ. Sci. Technol. 2013, 47, 2441–2456. [Google Scholar] [CrossRef]
- Cossa, D.; Gobeil, C. Mercury speciation in the Lower St. Lawrence Estuary. Can. J. Fish. Aquat. Sci. 2000, 57, 138–147. [Google Scholar] [CrossRef]
- Oliveri, E.; Salvagio Manta, D.; Bonsignore, M.; Cappello, S.; Tranchida, G.; Bagnato, E.; Sabatino, N.; Santisi, S.; Sprovieri, M. Mobility of mercury in contaminated marine sediments: Biogeochemical pathways. Mar. Chem. 2016, 186, 1–10. [Google Scholar] [CrossRef]
- Thian, L.; Guan, W.; Ji, Y.; He, X.; Chen, W.; Alvarez, P.J.J.; Zhang, T. Microbial methylation potential of mercury sulphide particles dictated by surface structure. Nat. Geosci. 2021, 14, 409–416. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Z.; Fang, Q.; Qiao, R.; Zhang, T. Extracellular polymeric substances enhance dissolution and microbial methylation of mercury sulphide minerals. Environ. Sci. Process. Impacts 2023, 25, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Cheng, Q.; He, D.; Sun, J.; Li, J.; Pan, X. Recent advances in microbial mercury methylation: A review on methylation habitat, methylator, mechanism, and influencing factors. Proc. Saf. Environ. Prot. 2023, 170, 286–296. [Google Scholar] [CrossRef]
- Regnell, O.; Watras, C.J. Microbial mercury methylation in aquatic environments: A critical review of published field and laboratory studies. Environ. Sci. Technol. 2019, 53, 4–19. [Google Scholar] [CrossRef] [PubMed]
- Drott, A.; Lambertsson, L.; Björn, E.; Skyllberg, U. Importance of dissolved neutral mercury sulphides for methyl mercury production in contaminated sediments. Environ. Sci. Technol. 2007, 41, 2270–2276. [Google Scholar] [CrossRef]
- Hammerschmidt, C.R.; Fitzgerald, W.F. Iron-mediated photochemical decomposition of methylmercury in the Arctic Alaskan lake. Environ. Sci. Technol. 2010, 44, 6138–6143. [Google Scholar] [CrossRef]
- Mason, R.P.; Choi, A.L.; Fitzgerald, W.F.; Hammerschmidt, C.R.; Lamborg, C.H.; Soerense, A.L.; Sunderland, E.M. Mercury biogeochemical cycling in the ocean and policy implications. Environ. Res. 2012, 119, 101–117. [Google Scholar] [CrossRef]
- Villar, E.; Cabrol, L.; Heimbürger-Boavida, L.-E. Widespread microbial mercury methylation genes in the global ocean. Environ. Microbiol. Rep. 2020, 12, 277–287. [Google Scholar] [CrossRef]
- Blum, J.D. Marine mercury breakdown. Nat. Geosci. 2011, 4, 139–140. [Google Scholar] [CrossRef]
- Motta, L.C.; Blum, J.D.; Johnson, M.W.; Umhau, B.P.; Popp, B.N.; Washburn, S.J.; Drazen, J.C.; Benitez-Nelson, C.R.; Hannides, C.C.S.; Close, H.G.; et al. Mercury cycling in the North Pacific subtropical gyre as revealed by mercury stable isotope ratios. Glob. Biogeochem. Cycles 2019, 33, 777–794. [Google Scholar] [CrossRef]
- Gionfriddo, C.M.; Tate, M.T.; Wick, R.R.; Schultz, M.B.; Zemia, A.; Thelen, M.P.; Schofield, R.; Krabbenhoft, D.P.; Holt, K.E.; Moreau, J.W. Microbial mercury methylation in Antarctic sea ice. Nat. Microbiol. 2016, 1, 16127. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Fisher, N.S. Methylmercury uptake by diverse marine phytoplankton. Limnol. Oceanog. 2016, 61, 1626–1639. [Google Scholar] [CrossRef]
- Bacci, E. Mercury in the Mediterranean. Mar. Pollut. Bull. 1989, 20, 59–63. [Google Scholar] [CrossRef]
- Tesán-Onrubia, J.A.; Heimbürger-Boavida, L.-E.; Dufour, A.; Harmelin-Vivien, M.; García-Arévalo, I.; Knoery, J.; Thomas, B.; Carlotti, F.; Tedetti, M.; Bănaru, D. Bioconcentration, bioaccumulation and biomagnification of mercury in plankton of the Mediterranean Sea. Mar. Pollut. Bull. 2023, 194, 115439. [Google Scholar] [CrossRef]
- Buckman, K.L.; Lane, O.; Kotnik, J.; Bratkic, A.; Sprovieri, F.; Horvat, M.; Pirrone, N.; Evers, D.C.; Chen, C.Y. Spatial and taxonomic variation of mercury concentration in low trophic level fauna from Mediterranean Sea. Ecotoxicology 2018, 27, 1341–1352. [Google Scholar] [CrossRef]
- Bargagli, R.; Monaci, F.; Sanchez-Hernandez, J.C.; Cateni, D. Biomagnification of mercury in an Antarctic marine coastal food web. Mar. Ecol. Prog. Ser. 1998, 169, 65–76. [Google Scholar] [CrossRef]
- Girolametti, F.; Frapiccini, E.; Annibaldi, A.; Illuminati, S.; Panfili, M.; Marini, M.; Santojanni, A.; Truzzi, C. Total mercury (THg) content in red mullet (Mullus barbatus) from Adriatic Sea (central Mediterranean Sea): Relation to biological parameters, sampling area and human health risk assessment. Appl. Sci. 2022, 12, 10083. [Google Scholar] [CrossRef]
- Choy, C.A.; Popp, B.N.; Kaneko, J.J.; Drazen, J.C. The influence of depth on mercury levels in pelagic fishes and their prey. Proc. Natl. Acad. Sci. USA 2009, 106, 13865–13869. [Google Scholar] [CrossRef]
- Tseng, C.-M.; Ang, S.-J.; Chen, Y.-S.; Shiao, J.-S.; Lamborg, C.H.; He, X.; Reinferder, J.R. Bluefin tuna reveal global patterns of mercury pollution and bioavailability in the world’s oceans. Proc. Natl. Acad. Sci. USA 2021, 118, e2111205118. [Google Scholar] [CrossRef]
- Madigan, D.J.; Li, M.; Yin, R.; Baumann, H.; Snodgrass, O.E.; Dewar, H.; Krabbenhoft, D.P.; Baumann, Z.; Fisher, N.S.; Balcom, P.; et al. Mercury stable isotopes reveal influence of foraging depth on mercury concentrations and growth in Pacific bluefin tuna. Environ. Sci. Technol. 2018, 52, 6256–6264. [Google Scholar] [CrossRef]
- Capodiferro, M.; Marco, E.; Grimalt, J.O. Wild fish and seafood species in the western Mediterranean Sea with low safe mercury concentrations. Environ. Pollut. 2022, 314, 120274. [Google Scholar] [CrossRef] [PubMed]
- Aissioui, S.; Poirier, L.; Amara, R.; Ramdane, Z. Concentrations of lead, cadmium and mercury in sardines, Sardina pilchardus (Walbaum, 1972) from the Algerian coast and health risks. J. Food Compos. Anal. 2022, 109, 104490. [Google Scholar] [CrossRef]
- Aissioui, S.; Poirier, L.; Amara, R.; Ramdane, Z. Concentrations of lead, cadmium, and mercury in Mullus barbatus from the Algerian coast and health risk associated to its consumption. Reg. Stud. Mar. Sci. 2021, 47, 101959. [Google Scholar] [CrossRef]
- El-Sherbiny, M.M.; Sallam, K.I. Residual content and health risk assessment of mercury, lead and cadmium in sardine and mackerel from Mediterranean Sea coast, Egypt. J. Food Compos. Anal. 2021, 2021, 103749. [Google Scholar] [CrossRef]
- Renieri, E.A.; Safenkova, I.V.; Alegakis, A.K.; Slutskaya, E.S.; Kokaraki, V.; Kentouri, M.; Dzantiev, B.B.; Tsatsakis, A.M. Cadmium, lead and mercury in muscle tissue of gilthead seabream and seabass: Risk evaluation for consumers. Food Chem. Toxicol. 2019, 124, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Abbass, K.; Huusko, A.; Knutsen, H.K.; Nieminen, P.; Myllynen, P.; Meltzer, H.M.; Vahakangas, K.; Rautio, A. Quantitative estimation of mercury intake by toxicokinetic modelling based on total mercury levels in humans. Environ. Int. 2018, 114, 1–11. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013. Off. J. Eur. Union 2013, L. 226, 1–17. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32013L0039 (accessed on 1 October 2023).
- World Health Organization; Food and Agriculture Organization. Evaluation of certain contaminants in food. Seventy-second Report of the Joint FAO/WHO Expert Committee on Food Additives; WHO-Technical report series 959. In Proceedings of the Seventy-Second Meeting of the Joint FAO/WHO Expert Committee on Food Additives, Rome, Italy, 16–25 February 2010; Available online: https://www.who.int/publications/i/item/9789241209595 (accessed on 1 October 2023).
- Rodríguez, J.; Andersson, A.; Björn, E.; Timonen, S.; Brugel, S.; Skrobonja, A.; Rowe, O. Inputs of dissolved organic matter enhance bacterial production and methylmercury formation in oxic coastal waters. Front. Microbiol. 2022, 13, 809166. [Google Scholar] [CrossRef]
- Bargagli, R.; Baldi, F. Mercury and methylmercury in higher fungi and their relation with the substrata in a cinnabar mining area. Chemosphere 1984, 13, 1059–1071. [Google Scholar] [CrossRef]
- Rodrigues, E.T.; Coelho, J.P.; Pereira, E.; Pardal, M.A. Are mercury in fishery products appropriate to ensure low risk to high fish-consumption populations? Mar. Pollut. Bull. 2023, 186, 114464. [Google Scholar] [CrossRef]
- Bradley, M.A.; Barst, B.D.; Basu, N. A review of mercury bioavailability in humans and fish. Int. J. Environ. Res. Public Health 2017, 14, 169. [Google Scholar] [CrossRef] [PubMed]
- Tinggi, U.; Perkins, A.V. Selenium status: Its interactions with dietary mercury exposure and implications in human health. Nutrients 2022, 14, 5308. [Google Scholar] [CrossRef]
- Zampetti, C.J.; Barndt, J.E. Co-considering selenium concentrations alters mercury-based fish and seafood consumption advice: A data compilation and critical assessment. Environ. Sci. Technol. Lett. 2023, 10, 179–185. [Google Scholar] [CrossRef]
- Wang, Y.; Li, L.; Yao, C.; Tian, X.; Wu, Y.; Xie, Q.; Wang, D. Mercury in human hair and its implications for health investigation. Curr. Opin. Environ. Sci. Health 2021, 22, 100271. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, P.; Zhang, Y. Climate-driven changes of global marine mercury cycles in 2100. Proc. Natl. Acad. Sci. USA 2023, 120, e2202488120. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, P.; Song, Z.; Huang, S.; Yuan, T.; Wu, P.; Shah, V.; Liu, M.; Chen, L.; Wang, X.; et al. An updated global mercury budget from a coupled atmosphere-land-ocean model: 40% more re-emissions buffer the effect of primary emission reductions. One Earth 2023, 6, 316–325. [Google Scholar] [CrossRef]
- Kvale, K.; Oschlies, A. Recovery from microplastic-induced marine deoxygenation make take centuries. Nat. Geosci. 2023, 16, 10–12. [Google Scholar] [CrossRef]
- Reale, M.; Cossarini, G.; Lazzari, P.; Lovato, T.; Bolzon, G.; Masina, S.; Solidoro, C.; Salon, S. Acidification, deoxygenation, and nutrient and biomass declines in a warming Mediterranean Sea. Biogeosciences 2022, 19, 4035–4065. [Google Scholar] [CrossRef]
- Schartup, A.T.; Thackray, C.P.; Qureshi, A.; Dassuncao, C.; Gillespie, K.; Hanke, A.; Sunderland, E.M. Climate change and overfishing increase neurotoxicant in marine predators. Nature 2019, 572, 648–650. [Google Scholar] [CrossRef]
- Pagés, R.; Baklouti, M.; Barrier, N.; Richon, C.; Dutay, J.-C.; Motin, T. Changes in rivers inputs during the last decades significantly impacted the biogeochemistry of the eastern Mediterranean basin: A modelling study. Progr. Oceanog. 2020, 181, 102242. [Google Scholar] [CrossRef]
- Zittis, G.; Almazroui, M.; Alpert, P.; Ciais, P.; Cramer, W.; Dahdal, Y.; Fnais, M.; Francis, D.; Hadjinicolaou, P.; Howari, F.; et al. Climate change and weather extremes in the Eastern Mediterranean and Middle East. Rev. Geophys. 2022, 60, e2021RG000762. [Google Scholar] [CrossRef]
- Fornasaro, S.; Morelli, G.; Rimondi, V.; Fagotti, C.; Friani, R.; Lattanzi, P.; Costagliola, P. The extensive mercury contamination in soil and legacy sediments of the Paglia River basin (Tuscany, Italy): Interplay between Hg-mining waste discharges along rivers, 1960s economic boom, and ongoing climate change. J. Soils Sediments 2022, 22, 656–671. [Google Scholar] [CrossRef]
- Antonioli, F.; De Falco, G.; Lo Presti, V.; Moretti, L.; Scardino, G.; Anzidei, M.; Mastronuzzi, G. Relative sea-level rise and potential submersion risk for 2100 on 16 coastal plains of the Mediterranean Sea. Waters 2020, 12, 2173. [Google Scholar] [CrossRef]
- Krabbenhoft, D.P.; Sunderland, E.M. Global change and mercury. Science 2013, 341, 1457–1458. [Google Scholar] [CrossRef] [PubMed]

| Location | Depth | Total Hg | MeHg | References |
|---|---|---|---|---|
| Mediterranean Sea | ||||
| Ligurian Sea | From surface to 2200 m | NA | 0.30 ± 0.17 | [58] |
| Adriatic Sea | From surface to 1000 m | 0.78–6.97 | 0.12–2.48 | [54] |
| Marseille (Endoume Pier) | surface | 4.9–6.7 | NA | [59] |
| 20 km off-shore | From surface to 1400 m | 0.8–1.1 | 0.04–0.40 | |
| Gulf of Lion | Inner shelf | 1.52 ± 1.00 | NA | [15] |
| Continental slope | 1.09 ± 0.15 | NA | ||
| W Mediterranean | From 500 to 3000 m | 1.02 ± 0.12 | <0.02–0.71 | |
| E Mediterranean | Intermediate waters | 1.02 ± 0.12 | <0.02–0.23 | |
| Atlantic Ocean | ||||
| North Atlantic | From surface to 4500 m | 0.46–0.83 | 0.04–0.4 | [59] |
| North Atlantic | <150 m | 0.69 ± 0.39 | 0.13 ± 0.12 | [19] |
| 150–1000 m | 0.96 ± 0.34 | 0.22 ± 0. 05 | ||
| Gulf of Cadiz | From surface to 4000 m | 0.86 ± 0.27 | NA | [15] |
| Pacific Ocean | ||||
| Central North | <150 m | 0.47 ± 0.36 | 0.07 ± 0.03 | [60] |
| >150–1000 m | 1.1 ± 0.29 | 0.21 ± 0.02 | ||
| Equatorial | <150 m | 0.45 ± 0.31 | 0.08 ± 0.02 | |
| >150–1000 m | 0.93 ± 0.23 | 0.14 ± 0.01 |
| Fishing Area | Species | Length (cm) Mean or Range | Total Hg Mean ± SD | References |
|---|---|---|---|---|
| NW Mediterranean Spain, France, Italy | Engraulis encrasicolus | 14 | 0.077 ± 0.05 | [86] |
| Sardina pilchardus | 16 | 0.084 ± 0.04 | ||
| Mullus surmuletus | 20 | 0.18 ± 0.12 | ||
| Sparus aurata | 36 | 0.17 ± 0.13 | ||
| Solea solea | - | 0.10 (median) | ||
| Scomber scombrus | - | 0.41 (median) | ||
| Trachurus trachurus | - | 0.35 (median) | ||
| Conger conger | - | 0.38 (median) | ||
| Dentex dentex | - | 1.00 (median) | ||
| Adriatic Sea | Mullus barbatus | 10–17 | 0.20 ± 0.15 | [82] |
| Algerian coast | Sardina pilchardus | 12–15 | 0.10 ± 0.03 | [87] |
| Mullus barbatus | 13–16 | 0.18 ± 0.07 | [88] | |
| Egypt coast | Sardina pilchardus | 17.6 ± 8.9 | 0.27 ± 0.02 | [89] |
| Aegean Sea | Sparus aurata | 29.5 ± 0.8 | 0.037 ± 0.022 | [90] |
| Dicentrarchus labrax | 34.4 ± 1.4 | 0.034 ± 0.016 | ||
| Global | Order | No. locations | Total Hg | [24] |
| Clupeiformes | 142 | 0.06 ± 0.18 | ||
| Perciformes | 1430 | 0.37 ± 0.48 | ||
| Pleuronectiformes | 112 | 0.26 ± 0.28 | ||
| Gadiformes | 85 | 0.12 ± 0.14 | ||
| Mugiliformes | 91 | 0.08 ± 0.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bargagli, R.; Rota, E. Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change. Coasts 2024, 4, 89-107. https://doi.org/10.3390/coasts4010006
Bargagli R, Rota E. Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change. Coasts. 2024; 4(1):89-107. https://doi.org/10.3390/coasts4010006
Chicago/Turabian StyleBargagli, Roberto, and Emilia Rota. 2024. "Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change" Coasts 4, no. 1: 89-107. https://doi.org/10.3390/coasts4010006
APA StyleBargagli, R., & Rota, E. (2024). Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change. Coasts, 4(1), 89-107. https://doi.org/10.3390/coasts4010006






